Proteases, Mucus, and Mucosal Immunity in Chronic Lung Disease
Abstract
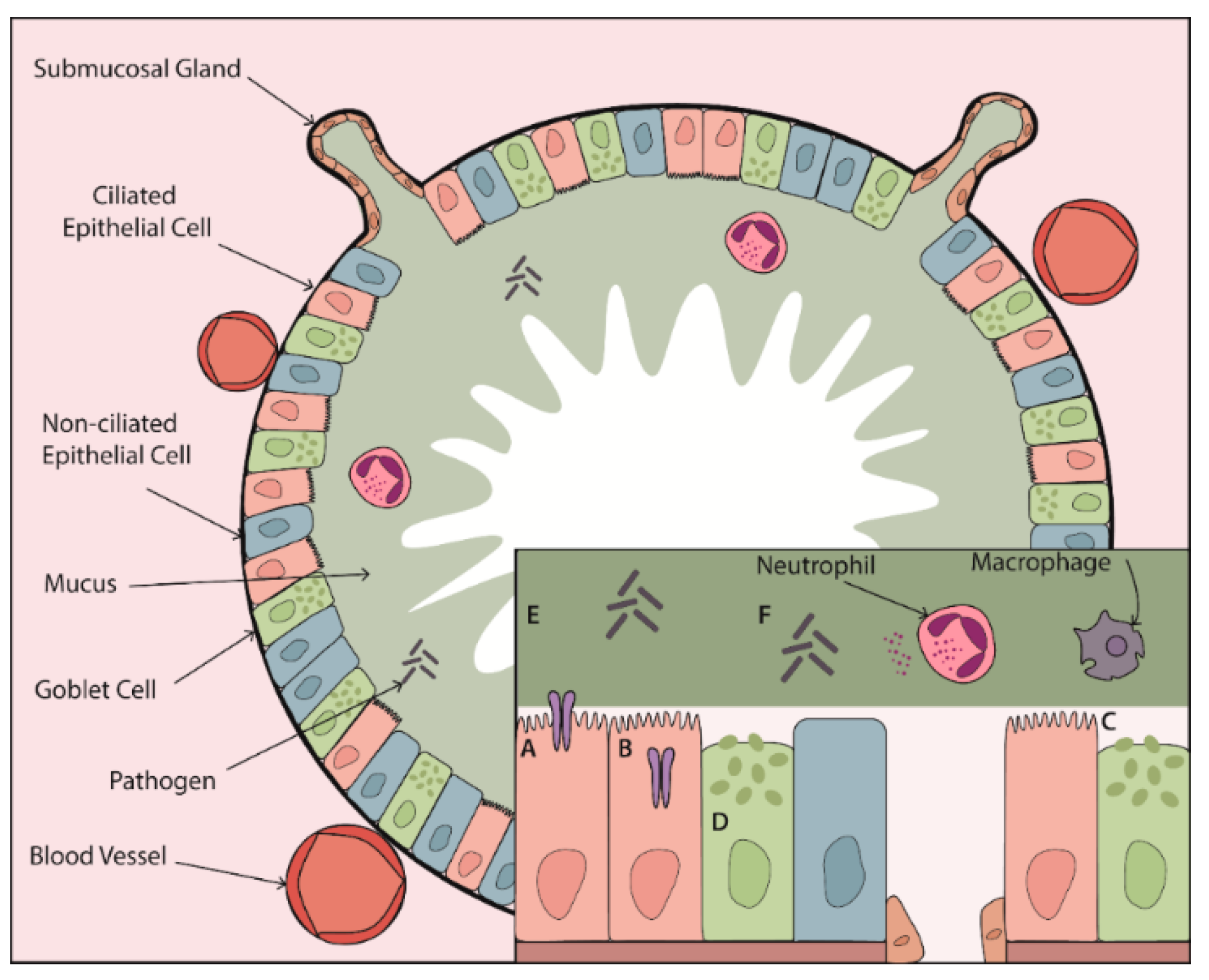
:1. Introduction
2. Proteases and Mucus
2.1. Proteases and Ion Transport
2.2. Proteases and Ciliary Function
2.3. Proteases and Mucus Properties
2.3.1. Mucin Expression
2.3.2. Mucin Secretion
2.3.3. Mucus Viscoelastic Properties
3. Proteases and Mucosal Immunity
3.1. Mucus and Mucosal Immunity
3.2. Proteases as Regulators of Host Defence
3.3. Self-Perpetuating Protease Activity
3.4. Protease Signalling and Epithelial Integrity
4. Targeting Proteases in Muco-Obstructive Lung Disease
5. Conclusions
Funding
Conflicts of Interest
Abbreviations
| A1-AT | α1-antitrypsin |
| ADAM | A disintegrin and a metalloprotease |
| AEC | Airway epithelial cell |
| AMP | Antimicrobial peptide |
| ASL | Airway surface liquid |
| βENaC-Tg | Transgenic mouse overexpressing the β-subunit of epithelial sodium channel |
| cAMP | Cyclic adenosine monophosphate |
| CAP | Channel-activating protease |
| CBF | Ciliary beat frequency |
| CF | Cystic fibrosis |
| CFTR | Cystic fibrosis transmembrane conductance regulator |
| CLD | Chronic lung disease |
| COPD | Chronic obstructive pulmonary disease |
| CTSB | Cathepsin B |
| CTSS | Cathepsin S |
| DNA | Deoxyribonucleic acid |
| ECM | Extracellular matrix |
| EGFR | Epidermal growth factor receptor |
| Egr | Early growth response |
| ENaC | Epithelial sodium channel |
| ERK | Extracellular receptor kinase |
| FRET | Fluorescence resonance energy transfer |
| HAT | Human airway trypsin-like protease |
| ICAM | Intercellular adhesion molecule |
| IMP | Investigational medical product |
| JNK | c-Jun N-terminal kinase |
| MARCKS | Myristoylated alanine-rich C kinase substrate |
| MCC | Mucociliary clearance |
| MMP | Matrix metalloprotease |
| mRNA | Messenger ribonucleic acid |
| NE | Neutrophil elastase |
| NET | Neutrophil extracellular trap |
| NF-κB | Nuclear factor κB |
| NSP | Neutrophil serine protease |
| PAR | Protease-activated receptor |
| PCD | Primary ciliary dyskinesia |
| PK | Pharmacokinetic |
| PKC | Protein kinase C |
| PGF | Placental growth factor |
| SLPI | Secretory leukocyte protease inhibitor |
| TIMP | Tissue inhibitor of metalloproteinases |
| TLR | Toll-like receptor |
| TMPRSS | Transmembrane protease, serine |
| TNF | Tumour necrosis factor |
References
- Kappelhoff, R.; Puente, X.S.; Wilson, C.H.; Seth, A.; López-Otín, C.; Overall, C.M. Overview of transcriptomic analysis of all human proteases, non-proteolytic homologs and inhibitors: Organ, tissue and ovarian cancer cell line expression profiling of the human protease degradome by the CLIP-CHIPTM DNA microarray. BBA-Mol. Cell Res. 2017, 1864, 2210–2219. [Google Scholar] [CrossRef]
- Taggart, C.; Mall, M.A.; Lalmanach, G.; Cataldo, D.; Ludwig, A.; Janciauskiene, S.; Heath, N.; Meiners, S.; Overall, C.M.; Schultz, C.; et al. Protean proteases: At the cutting edge of lung diseases. Eur. Respir. J. 2017, 49, 1501200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taggart, C.C.; Greene, C.M.; Carroll, T.P.; O’Neill, S.J.; McElvaney, N.G. Elastolytic proteases: Inflammation resolution and dysregu-lation in chronic infective lung disease. Am. J. Respir. Crit. Care Med. 2005, 171, 1070–1076. [Google Scholar] [CrossRef] [PubMed]
- Greene, C.M.; McElvaney, N.G. Proteases and antiproteases in chronic neutrophilic lung disease-relevance to drug discovery. Br. J. Pharmacol. 2009, 158, 1048–1058. [Google Scholar] [CrossRef] [Green Version]
- McKelvey, M.C.; Weldon, S.; McAuley, D.F.; Mall, M.A.; Taggart, C.C. Targeting Proteases in Cystic Fibrosis Lung Disease. Paradigms, Progress, and Potential. Am. J. Respir. Crit. Care Med. 2020, 201, 141–147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Knowles, M.R.; Boucher, R.C. Mucus clearance as a primary innate defense mechanism for mammalian airways. J. Clin. Investig. 2002, 109, 571–577. [Google Scholar] [CrossRef] [PubMed]
- Mall, M.A. Role of Cilia, Mucus, and Airway Surface Liquid in Mucociliary Dysfunction: Lessons from Mouse Models. J. Aerosol Med. Pulm. Drug Deliv. 2008, 21, 13–24. [Google Scholar] [CrossRef]
- Bustamante-Marin, X.M.; Ostrowski, L.E. Cilia and Mucociliary Clearance. Cold Spring Harb. Perspect. Biol. 2017, 9, a028241. [Google Scholar] [CrossRef] [PubMed]
- Fahy, J.V.; Dickey, B.F. Airway Mucus Function and Dysfunction. N. Engl. J. Med. 2010, 363, 2233–2247. [Google Scholar] [CrossRef] [Green Version]
- Boucher, R.C. Muco-Obstructive Lung Diseases. N. Engl. J. Med. 2019, 380, 1941–1953. [Google Scholar] [CrossRef] [PubMed]
- Anderson, W.H.; Coakley, R.D.; Button, B.; Henderson, A.G.; Zeman, K.L.; Alexis, N.E.; Peden, D.B.; Lazarowski, E.R.; Davis, C.W.; Bailey, S.; et al. The Relationship of Mucus Concentration (Hydration) to Mucus Osmotic Pressure and Transport in Chronic Bronchitis. Am. J. Respir. Crit. Care Med. 2015, 192, 182–190. [Google Scholar] [CrossRef] [Green Version]
- Button, B.; Anderson, W.H.; Boucher, R.C. Mucus Hyperconcentration as a Unifying Aspect of the Chronic Bronchitic Phenotype. Ann. Am. Thorac. Soc. 2016, 13, S156–S162. [Google Scholar]
- Henderson, A.G.; Ehre, C.; Button, B.; Abdullah, L.H.; Cai, L.-H.; Leigh, M.W.; DeMaria, G.C.; Matsui, H.; Donaldson, S.H.; Davis, C.W.; et al. Cystic fibrosis airway secretions exhibit mucin hyperconcentration and increased osmotic pressure. J. Clin. Investig. 2014, 124, 3047–3060. [Google Scholar] [CrossRef] [Green Version]
- Tarran, R. Regulation of Airway Surface Liquid Volume and Mucus Transport by Active Ion Transport. Proc. Am. Thorac. Soc. 2004, 1, 42–46. [Google Scholar] [CrossRef]
- Tarran, R.; Button, B.; Picher, M.; Paradiso, A.M.; Ribeiro, C.M.; Lazarowski, E.R.; Zhang, L.; Collins, P.L.; Pickles, R.J.; Fredberg, J.J.; et al. Normal and cystic fibrosis airway surface liquid homeostasis: The effects of phasic shear stress and viral infections. J. Biol. Chem. 2005, 280, 35751–35759. [Google Scholar] [CrossRef] [Green Version]
- Kunzelmann, K.; Greger, R. Na+ and Cl− conductances in airway epithelial cells: Increased Na+ conductance in cystic fibrosis. Pflügers Archiv-Eur. J. Physiol. 1995, 431, 4371. [Google Scholar] [CrossRef]
- Riordan, J.R.; Rommens, J.M.; Kerem, B.; Alon, N.; Rozmahel, R.; Grzelczak, Z.; Zielenski, J.; Lok, S.; Plavsic, N.; Chou, J.L.; et al. Identification of the cystic fibrosis gene: Cloning and characterization of complementary DNA. Science 1989, 245, 1066–1073. [Google Scholar] [CrossRef] [PubMed]
- Hughey, R.P.; Bruns, J.B.; Kinlough, C.L.; Harkleroad, K.L.; Tong, Q.; Carattino, M.D.; Johnson, J.P.; Stockand, J.D.; Kleyman, T.R. Epithelial Sodium Channels Are Activated by Furin-dependent Proteolysis. J. Biol. Chem. 2004, 279, 18111–18114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bruns, J.B.; Carattino, M.D.; Sheng, S.; Maarouf, A.B.; Weisz, O.A.; Pilewski, J.M.; Hughey, R.P.; Kleyman, T.R. Epithelial Na+ Channels Are Fully Activated by Furin- and Prostasin-dependent Release of an Inhibitory Peptide from the γ-Subunit. J. Biol. Chem. 2007, 282, 6153–6160. [Google Scholar] [CrossRef] [Green Version]
- Haerteis, S.; Krappitz, M.; Bertog, M.; Krappitz, A.; Baraznenok, V.; Henderson, I.; Lindström, E.; Murphy, J.E.; Bunnett, N.W.; Korbmacher, C. Proteolytic activation of the epithelial sodium channel (ENaC) by the cysteine protease cathepsin-S. Pflügers Archiv-Eur. J. Physiol. 2012, 464, 353–365. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- García-Caballero, A.; Dang, Y.; He, H.; Stutts, M.J. ENaC Proteolytic Regulation by Channel-activating Protease 2. J. Gen. Physiol. 2008, 132, 521–535. [Google Scholar] [CrossRef] [Green Version]
- Caldwell, R.A.; Boucher, R.C.; Stutts, M.J. Serine protease activation of near-silent epithelial Na+ channels. Am. J. Physiol. Physiol. 2004, 286, C190–C194. [Google Scholar] [CrossRef] [Green Version]
- Larionov, A.; Dahlke, E.; Kunke, M.; Rodriguez, L.Z.; Schiessl, I.M.; Magnin, J.; Kern, U.; Alli, A.A.; Mollet, G.; Schilling, O.; et al. Cathepsin B increases ENaC activity leading to hypertension early in nephrotic syndrome. J. Cell. Mol. Med. 2019, 23, 6543–6553. [Google Scholar] [CrossRef] [Green Version]
- Caldwell, R.A.; Boucher, R.C.; Stutts, M.J. Neutrophil elastase activates near-silent epithelial Na+ channels and increases airway epithelial Na+ transport. Am. J. Physiol. Cell. Mol. Physiol. 2005, 288, L813–L819. [Google Scholar] [CrossRef] [Green Version]
- Shuto, T.; Kamei, S.; Nohara, H.; Fujikawa, H.; Tasaki, Y.; Sugahara, T.; Ono, T.; Matsumoto, C.; Sakaguchi, Y.; Maruta, K.; et al. Pharmacological and genetic reappraisals of protease and oxidative stress pathways in a mouse model of obstructive lung diseases. Sci. Rep. 2016, 6, 39305. [Google Scholar] [CrossRef] [Green Version]
- Butterworth, M.B.; Zhang, L.; Liu, X.; Shanks, R.M.; Thibodeau, P.H. Modulation of the Epithelial Sodium Channel (ENaC) by Bacterial Metalloproteases and Protease Inhibitors. PLoS ONE 2014, 9, e100313. [Google Scholar] [CrossRef] [Green Version]
- Mall, M.; Grubb, B.R.; Harkema, J.R.; O’Neal, W.K.; Boucher, R.C.; Neal, W.K.O. Increased airway epithelial Na+ absorption produces cystic fibrosis-like lung disease in mice. Nat. Med. 2004, 10, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Liang, X.; Zhao, M.; Liu, S.-L.; Huang, Y.; Idell, S.; Li, X.; Ji, H.-L. Correlation of Apical Fluid-Regulating Channel Proteins with Lung Function in Human COPD Lungs. PLoS ONE 2014, 9, e109725. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mall, M.A.; Harkema, J.R.; Trojanek, J.B.; Treis, D.; Livraghi, A.; Schubert, S.; Zhou, Z.; Kreda, S.M.; Tilley, S.L.; Hudson, E.J.; et al. Development of chronic bronchitis and emphysema in β-epithelial Na+ channel-overexpressing mice. Am. J. Respir. Crit. Care Med. 2008, 177, 730–742. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kerem, E.; Bistritzer, T.; Hanukoglu, A.; Hofmann, T.; Zhou, Z.; Bennett, W.; MacLaughlin, E.; Barker, P.; Nash, M.; Quittell, L.; et al. Pulmonary Epithelial Sodium-Channel Dysfunction and Excess Airway Liquid in Pseudohypoaldosteronism. N. Engl. J. Med. 1999, 341, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Averna, M.; Stifanese, R.; Grosso, R.; Pedrazzi, M.; De Tullio, R.; Salamino, F.; Pontremoli, S.; Melloni, E. Role of calpain in the regulation of CFTR (cystic fibrosis transmembrane conductance regulator) turnover. Biochem. J. 2010, 430, 255–263. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matos, A.M.; Pinto, F.R.; Barros, P.; Amaral, M.D.; Pepperkok, R.; Matos, P. Inhibition of calpain 1 restores plasma membrane stability to pharmacologically rescued Phe508del-CFTR variant. J. Biol. Chem. 2019, 294, 13396–13410. [Google Scholar] [CrossRef] [PubMed]
- Averna, M.; Stifanese, R.; De Tullio, R.; Minicucci, L.; Cresta, F.; Palena, S.; Salamino, F.; Pontremoli, S.; Melloni, E. Evidence for alteration of calpain/calpastatin system in PBMC of cystic fibrosis patients. Biochim. Biophys. Acta Mol. Basis Dis. 2011, 1812, 1649–1657. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Averna, M.; Pedrazzi, M.; Minicucci, L.; De Tullio, R.; Cresta, F.; Salamino, F.; Pontremoli, S.; Melloni, E. Calpain Inhibition Promotes the Rescue of F508del-CFTR in PBMC from Cystic Fibrosis Patients. PLoS ONE 2013, 8, e66089. [Google Scholar] [CrossRef] [Green Version]
- Le Gars, M.; Descamps, D.; Roussel, D.; Saussereau, E.; Guillot, L.; Ruffin, M.; Tabary, O.; Hong, S.S.; Boulanger, P.; Paulais, M.; et al. Neutrophil elastase degrades cystic fibrosis transmembrane conductance regulator via calpains and disables channel function in vitro and in vivo. Am. J. Respir. Crit. Care Med. 2013, 187, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Mall, M.A.; Hartl, D. CFTR: Cystic fibrosis and beyond. Eur. Respir. J. 2014, 44, 1042–1054. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bodas, M.; Min, T.; Mazur, S.; Vij, N. Critical Modifier Role of Membrane-Cystic Fibrosis Transmembrane Conductance Regula-tor-Dependent Ceramide Signaling in Lung Injury and Emphysema. J. Immunol. 2011, 186, 602–613. [Google Scholar] [CrossRef] [PubMed]
- Cantin, A.M.; Hanrahan, J.W.; Bilodeau, G.; Ellis, L.; Dupuis, A.; Liao, J.; Zielenski, J.; Durie, P. Cystic Fibrosis Transmembrane Conductance Regulator Function Is Suppressed in Cigarette Smokers. Am. J. Respir. Crit. Care Med. 2006, 173, 1139–1144. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clunes, L.A.; Davies, C.M.; Coakley, R.D.; Aleksandrov, A.A.; Henderson, A.G.; Zeman, K.L.; Worthington, E.N.; Gentzsch, M.; Kreda, S.M.; Cholon, D.; et al. Cigarette smoke exposure induces CFTR internalization and insolubility, leading to airway surface liquid dehydration. FASEB J. 2011, 26, 533–545. [Google Scholar] [CrossRef] [Green Version]
- Pezzulo, A.A.; Tang, X.X.; Hoegger, M.J.; Abou Alaiwa, M.H.; Ramachandran, S.; Moninger, T.O.; Karp, P.H.; Wohlford-Lenane, C.L.; Haagsman, H.P.v.E.M.; Bánfi, B.; et al. Reduced airway surface pH impairs bacterial killing in the porcine cystic fibrosis lung. Nature 2012, 487, 109–113. [Google Scholar] [CrossRef]
- Haitchi, H.M.; Yoshisue, H.; Ribbene, A.; Wilson, S.J.; Holloway, J.W.; Bucchieri, F.; Hanley, N.A.; Wilson, D.I.; Zummo, G.; Holgate, S.T.; et al. Chronological expression of Ciliated Bronchial Epithelium 1 during pulmonary development. Eur. Respir. J. 2009, 33, 1095–1104. [Google Scholar] [CrossRef]
- Davis, C.W.; Dickey, B.F. Regulated Airway Goblet Cell Mucin Secretion. Annu. Rev. Physiol. 2008, 70, 487–512. [Google Scholar] [CrossRef] [PubMed]
- Lucas, J.S.; Burgess, A.; Mitchison, H.M.; Moya, E.; Williamson, M.; Hogg, C.; on behalf of the National PCD Service, UK. Diagnosis and management of primary ciliary dyskinesia. Arch. Dis. Child. 2014, 99, 850–856. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wyatt, T.A.; Spurzem, J.R.; May, K.; Sisson, J.H. Regulation of ciliary beat frequency by both PKA and PKG in bovine airway epithelial cells. Am. J. Physiol. Cell. Mol. Physiol. 1998, 275, L827–L835. [Google Scholar] [CrossRef]
- Di Benedetto, G.; Magnus, C.J.; Gray, P.T.; Mehta, A. Calcium regulation of ciliary beat frequency in human respiratory epithelium in vitro. J. Physiol. 1991, 439, 103–113. [Google Scholar] [CrossRef]
- Sutto, Z.; Conner, G.E.; Salathe, M. Regulation of human airway ciliary beat frequency by intracellular pH. J. Physiol. 2004, 560, 519–532. [Google Scholar] [CrossRef]
- Satir, P.; Heuser, T.; Sale, W.S. A Structural Basis for How Motile Cilia Beat. Bioscience 2014, 64, 1073–1083. [Google Scholar] [CrossRef] [Green Version]
- Hingley, S.T.; Hastie, A.T.; Kueppers, F.; Higgins, M.L. Disruption of respiratory cilia by proteases including those of Pseudomonas aeruginosa. Infect. Immun. 1986, 54, 379–383. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amitani, R.; Wilson, R.; Rutman, A.; Read, R.; Ward, C.; Burnett, D.; Stockley, R.A.; Cole, P.J. Effects of Human Neutrophil Elastase andPseudomonas aeruginosaProteinases on Human Respiratory Epithelium. Am. J. Respir. Cell Mol. Biol. 1991, 4, 26–32. [Google Scholar] [CrossRef] [PubMed]
- McMahon, D.B.; Workman, A.D.; Kohanski, M.A.; Carey, R.M.; Freund, J.R.; Hariri, B.M.; Chen, B.; Doghramji, L.J.; Adappa, N.D.; Palmer, J.N.; et al. Protease-activated receptor 2 activates airway apical membrane chloride permeability and increases ciliary beating. FASEB J. 2018, 32, 155–167. [Google Scholar] [CrossRef] [Green Version]
- Gomperts, B.N.; Gong-Cooper, X.; Hackett, B.P. Foxj1 regulates basal body anchoring to the cytoskeleton of ciliated pulmonary epithelial cells. J. Cell Sci. 2004, 117, 1329–1337. [Google Scholar] [CrossRef] [Green Version]
- Roberts, R.E.; Martin, M.; Marion, S.; Elumalai, G.L.; Lewis, K.; Hallett, M.B. Ca2+-activated cleavage of ezrin visualised dynamically in living myeloid cells during cell surface area expansion. J. Cell Sci. 2020, 133. [Google Scholar] [CrossRef]
- Kirkham, S.; Sheehan, J.K.; Knight, D.; Richardson, P.S.; Thornton, D.J. Heterogeneity of airways mucus: Variations in the amounts and glycoforms of the major oligomeric mucins MUC5AC and MUC5B. Biochem. J. 2002, 361, 537–546. [Google Scholar] [CrossRef]
- Hogg, J.C.; Chu, F.S.; Tan, W.C.; Sin, D.D.; Patel, S.A.; Pare, P.D.; Martinez, F.J.; Rogers, R.M.; Make, B.J.; Criner, G.J.; et al. Survival after lung volume reduction in chronic obstructive pulmonary disease: Insights from small airway pathology. Am. J. Respir. Crit. Care Med. 2007, 176, 454–459. [Google Scholar] [CrossRef] [Green Version]
- Hill, D.B.; Long, R.F.; Kissner, W.J.; Atieh, E.; Garbarine, I.C.; Markovetz, M.R.; Fontana, N.C.; Christy, M.; Habibpour, M.; Tarran, R.; et al. Pathological mucus and impaired mucus clearance in cystic fibrosis patients result from increased concentration, not altered pH. Eur. Respir. J. 2018, 52, 1801297. [Google Scholar] [CrossRef]
- Ramsey, K.A.; Chen, A.C.H.; Radicioni, G.; Lourie, R.; Martin, M.; Broomfield, A.; Sheng, Y.H.; Hasnain, S.Z.; Radford-Smith, G.; Simms, L.A.; et al. Airway Mucus Hyperconcentration in Non–Cystic Fibrosis Bronchiectasis. Am. J. Respir. Crit. Care Med. 2020, 201, 661–670. [Google Scholar] [CrossRef] [PubMed]
- Yuan, S.; Hollinger, M.; Lachowicz-Scroggins, M.E.; Kerr, S.C.; Dunican, E.M.; Daniel, B.M.; Ghosh, S.; Erzurum, S.C.; Willard, B.; Hazen, S.L.; et al. Oxidation increases mucin polymer cross-links to stiffen airway mucus gels. Sci. Transl. Med. 2015, 7, 276ra27. [Google Scholar] [CrossRef] [Green Version]
- Roy, M.G.; Livraghi-Butrico, A.; Fletcher, A.A.; McElwee, M.M.; Evans, S.E.; Boerner, R.M.; Alexander, S.N.; Bellinghausen, L.K.; Song, A.S.; Petrova, Y.M.; et al. Muc5b is required for airway defence. Nat. Cell Biol. 2014, 505, 412–416. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ja, S.K.; Kim, Y.D.; Jetten, A.M.; Belloni, P.; Nettesheim, P. Overexpression of mucin genes induced by interleukin-1β, tumor necrosis factor-α, lipopolysaccharide, and neutrophil elastase is inhibited by a retinoic acid receptor α antagonist. Exp. Lung Res. 2002, 28, 315–332. [Google Scholar]
- Voynow, J.A.; Rosenthal Young, L.; Wang, Y.; Horger, T.; Rose, M.C.; Fischer, B.M. Neutrophil elastase increases MUC5AC mRNA and protein expression in respiratory epithelial cells. Am. J. Physiol. Lung Cell. Mol. Physiol. 1999, 276, L835–L843. [Google Scholar] [CrossRef] [Green Version]
- Fischer, B.; Voynow, J. Neutrophil elastase induces MUC5AC messenger RNA expression by an oxidant-dependent mecha-nism. Chest 2000, 117, 317S–320S. [Google Scholar] [CrossRef]
- Shao, M.X.G.; Nadel, J.A. Neutrophil Elastase Induces MUC5AC Mucin Production in Human Airway Epithelial Cells via a Cascade Involving Protein Kinase C, Reactive Oxygen Species, and TNF-α-Converting Enzyme. J. Immunol. 2005, 175, 4009–4016. [Google Scholar] [CrossRef] [Green Version]
- Deshmukh, H.S.; Case, L.M.; Wesselkamper, S.C.; Borchers, M.T.; Martin, L.D.; Shertzer, H.G.; Nadel, J.A.; Leikauf, G.D. Metalloproteinases Mediate Mucin 5AC Expression by Epidermal Growth Factor Receptor Activation. Am. J. Respir. Crit. Care Med. 2005, 171, 305–314. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chokki, M.; Yamamura, S.; Eguchi, H.; Masegi, T.; Horiuchi, H.; Tanabe, H.; Kamimura, T.; Yasuoka, S. Human Airway Trypsin-Like Protease Increases Mucin Gene Expression in Airway Epithelial Cells. Am. J. Respir. Cell Mol. Biol. 2004, 30, 470–478. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kamei, S.; Fujikawa, H.; Nohara, H.; Ueno-Shuto, K.; Maruta, K.; Nakashima, R.; Kawakami, T.; Matsumoto, C.; Sakaguchi, Y.; Ono, T.; et al. Zinc Deficiency via a Splice Switch in Zinc Importer ZIP2/SLC39A2 Causes Cystic Fibrosis-Associated MUC5AC Hypersecretion in Airway Epithelial Cells. EBioMedicine 2018, 27, 304–316. [Google Scholar] [CrossRef] [Green Version]
- Oguma, T.; Asano, K.; Tomomatsu, K.; Kodama, M.; Fukunaga, K.; Shiomi, T.; Ohmori, N.; Ueda, S.; Takihara, T.; Shiraishi, Y.; et al. Induction of Mucin and MUC5AC Expression by the Protease Activity of Aspergillus fumigatus in Airway Epithelial Cells. J. Immunol. 2011, 187, 999–1005. [Google Scholar] [CrossRef] [Green Version]
- Saunders, R.V.; Modha, D.E.; Claydon, A.; Gaillard, E.A. Chronic Aspergillus fumigatus colonization of the pediatric cystic fibrosis airway is common and may be associated with a more rapid decline in lung function. Med. Mycol. 2016, 54, 537–543. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, X.; Lee, B.; Zhu, L.; Ding, Z.; Chen, Y. Exposure to mold proteases stimulates mucin production in airway epithelial cells through Ras/Raf1/ERK signal pathway. PLoS ONE 2020, 15, e0231990. [Google Scholar] [CrossRef] [Green Version]
- Woodruff, P.G.; Modrek, B.; Choy, D.F.; Jia, G.; Abbas, A.R.; Ellwanger, A.; Arron, J.R.; Koth, L.L.; Fahy, J.V. T-helper Type 2–driven Inflammation Defines Major Subphenotypes of Asthma. Am. J. Respir. Crit. Care Med. 2009, 180, 388–395. [Google Scholar] [CrossRef]
- Bonser, L.R.; Zlock, L.; Finkbeiner, W.; Erle, D.J. Epithelial tethering of MUC5AC-rich mucus impairs mucociliary transport in asthma. J. Clin. Investig. 2016, 126, 2367–2371. [Google Scholar] [CrossRef]
- Ostedgaard, L.S.; Moninger, T.O.; McMenimen, J.D.; Sawin, N.M.; Parker, C.P.; Thornell, I.M.; Powers, L.S.; Gansemer, N.D.; Bouzek, D.C.; Cook, D.P.; et al. Gel-forming mucins form distinct morphologic structures in airways. Proc. Natl. Acad. Sci. USA 2017, 114, 6842–6847. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seibold, M.A.; Wise, A.L.; Speer, M.C.; Steele, M.P.; Brown, K.K.; Loyd, J.E.; Fingerlin, T.E.; Zhang, W.; Gudmundsson, G.; Groshong, S.D.; et al. A Common MUC5B Promoter Polymorphism and Pulmonary Fibrosis. N. Engl. J. Med. 2011, 364, 1503–1512. [Google Scholar] [CrossRef] [Green Version]
- Duerr, J.; Leitz, D.H.W.; Szczygiel, M.; Dvornikov, D.; Fraumann, S.G.; Kreutz, C.; Zadora, P.K.; Agircan, A.S.; Konietzke, P.; Engelmann, T.A.; et al. Conditional deletion of Nedd4–2 in lung epi-thelial cells causes progressive pulmonary fibrosis in adult mice. Nat. Commun. 2020, 11, 2012. [Google Scholar] [CrossRef] [PubMed]
- Jaramillo, A.M.; Azzegagh, Z.; Tuvim, M.J.; Dickey, B.F. Airway Mucin Secretion. Ann. Am. Thorac. Soc. 2018, 15, S164–S170. [Google Scholar] [CrossRef]
- Shumilov, D.; Popov, A.; Fudala, R.; Akopova, I.; Gryczynski, I.; Borejdo, J.; Gryczynski, Z.; Grygorczyk, R. Real-time imaging of exocytotic mucin release and swelling in Calu-3 cells using acridine orange. Methods 2014, 66, 312–324. [Google Scholar] [CrossRef] [Green Version]
- Davis, C.W.; Dowell, M.L.; Lethem, M.; Van Scott, M. Goblet cell degranulation in isolated canine tracheal epithelium: Response to exogenous ATP, ADP, and adenosine. Am. J. Physiol. Cell Physiol. 1992, 262, C1313–C1323. [Google Scholar] [CrossRef] [PubMed]
- Lethem, M.I.; Dowell, M.L.; Van Scott, M.; Yankaskas, J.R.; Egan, T.; Boucher, R.C.; Davis, C.W. Nucleotide Regulation of Goblet Cells in Human Airway Epithelial Explants: Normal Exocytosis in Cystic Fibrosis. Am. J. Respir. Cell Mol. Biol. 1993, 9, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Vestbo, J.; Prescott, E.; Lange, P. Association of chronic mucus hypersecretion with FEV1 decline and chronic obstructive pulmonary disease morbidity. Am. J. Respir. Crit. Care Med. 1996, 153, 1530–1535. [Google Scholar] [CrossRef]
- Martínez-Rivera, C.; Crespo, A.; Pinedo-Sierra, C.; García-Rivero, J.L.; Pallarés-Sanmartín, A.; Marina-Malanda, N.; Pascual-Erquicia, S.; Padilla, A.; Mayoralas-Alises, S.; Plaza, V.; et al. Mucus hypersecretion in asthma is associated with rhinosinusitis, polyps and exacerbations. Respir. Med. 2018, 135, 22–28. [Google Scholar] [CrossRef] [Green Version]
- Lemjabbar, H.; Basbaum, C. Platelet-activating factor receptor and ADAM10 mediate responses to Staphylococcus aureus in epithelial cells. Nat. Med. 2002, 8, 41–46. [Google Scholar] [CrossRef]
- Nadel, J.A. Role of Mast Cell and Neutrophil Proteases in Airway Secretion. Am. Rev. Respir. Dis. 1991, 144, S48–S51. [Google Scholar] [CrossRef]
- Lundgren, J.D.; Rieves, R.D.; Mullol, J.; Logun, C.; Shelhamer, J.H. The effect of neutrophil proteinase enzymes on the release of mucus from feline and human airway cultures. Respir. Med. 1994, 88, 511–518. [Google Scholar] [CrossRef]
- Witko-Sarsat, V.; Halbwachs-Mecarelli, L.; Schuster, A.; Nusbaum, P.; Ueki, I.; Canteloup, S.; Lenoir, G.; Descamps-Latscha, B.; Nadel, J.A. Proteinase 3, a Potent Secretagogue in Airways, Is Present in Cystic Fibrosis Sputum. Am. J. Respir. Cell Mol. Biol. 1999, 20, 729–736. [Google Scholar] [CrossRef] [PubMed]
- Takeyama, K.; Agustí, C.; Ueki, I.; Lausier, J.; Cardell, L.O.; Nadel, J.A. Neutrophil-dependent goblet cell degranulation: Role of membrane-bound elastase and adhesion molecules. Am. J. Physiol. Content 1998, 275, L294–L302. [Google Scholar] [CrossRef]
- Park, J.-A.; He, F.; Martin, L.D.; Li, Y.; Chorley, B.N.; Adler, K.B. Human Neutrophil Elastase Induces Hypersecretion of Mucin from Well-Differentiated Human Bronchial Epithelial Cells in Vitro via a Protein Kinase Cδ-Mediated Mechanism. Am. J. Pathol. 2005, 167, 651–661. [Google Scholar] [CrossRef]
- Zhong, T.; Perelman, J.M.; Kolosov, V.P.; Zhou, X.-D. MiR-146a negatively regulates neutrophil elastase-induced MUC5AC secretion from 16HBE human bronchial epithelial cells. Mol. Cell. Biochem. 2011, 358, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Klinger, J.D.; Tandler, B.; Liedtke, C.M.; Boat, T.F. Proteinases of Pseudomonas aeruginosa evoke mucin release by tracheal epithe-lium. J. Clin. Investig. 1984, 74, 1669–1678. [Google Scholar] [CrossRef]
- Puchelle, E.; Zahm, J.-M.; De Bentzmann, S.; Grosskopf, C.; Shak, S.; Mougel, D.; Polu, J.-M. Effects of rhDNase on purulent airway secretions in chronic bronchitis. Eur. Respir. J. 1996, 9, 765–769. [Google Scholar] [CrossRef] [Green Version]
- Henke, M.O.; John, G.; Rheineck, C.; Chillappagari, S.; Naehrlich, L.; Rubin, B.K. Serine Proteases Degrade Airway Mucins in Cystic Fibrosis. In Proceedings of the American Thoracic Society 2011 International Conference, Denver, CO, USA, 13–18 May 2011; Volume 79, pp. 3438–3444. [Google Scholar] [CrossRef]
- Majewski, P.; Majchrzak-Gorecka, M.; Grygier, B.; Skrzeczynska-Moncznik, J.; Osiecka, O.; Cichy, J. Inhibitors of Serine Proteases in Regulating the Production and Function of Neutrophil Extracellular Traps. Front. Immunol. 2016, 7, 261. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dubois, A.V.; Gauthier, A.; Bréa, D.; Varaigne, F.; Diot, P.; Gauthier, F.; Attucci, S. Influence of DNA on the Activities and Inhibition of Neutrophil Serine Proteases in Cystic Fibrosis Sputum. Am. J. Respir. Cell Mol. Biol. 2012, 47, 80–86. [Google Scholar] [CrossRef]
- Guerra, M.; Halls, V.S.; Schatterny, J.; Hagner, M.; Mall, M.A.; Schultz, C. Protease FRET Reporters Targeting Neutrophil Extracellular Traps. J. Am. Chem. Soc. 2020, 142, 20299–20305. [Google Scholar] [CrossRef] [PubMed]
- Robinson, C.V.; Elkins, M.R.; Bialkowski, K.M.; Thornton, D.J.; Kertesz, M.A. Desulfurization of mucin by Pseudomonas aeruginosa: Influence of sulfate in the lungs of cystic fibrosis patients. J. Med. Microbiol. 2012, 61, 1644–1653. [Google Scholar] [CrossRef]
- Cowley, A.C.; Thornton, D.J.; Denning, D.W.; Horsley, A. Aspergillosis and the role of mucins in cystic fibrosis. Pediatr. Pulmonol. 2016, 52, 548–555. [Google Scholar] [CrossRef] [Green Version]
- Seibold, M.A.; Wise, A.L.; Speer, M.C.; Steele, M.P.; Brown, K.K.; Loyd, J.E.; Fingerlin, T.E.; Zhang, W.; Gudmundsson, G.; Groshong, S.D.; et al. Hypoxic epithelial necrosis triggers neutrophilic inflammation via IL-1 receptor signaling in cystic fibrosis lung disease. Am. J. Respir. Crit. Care Med. 2015, 191, 902–913. [Google Scholar]
- Rosen, B.H.; Evans, T.I.A.; Moll, S.R.; Gray, J.S.; Liang, B.; Sun, X.; Zhang, Y.; Jensen-Cody, C.W.; Swatek, A.M.; Zhou, W.; et al. Infection is not required for mucoinflammatory lung disease in CFTR-Knockout ferrets. Am. J. Respir. Crit. Care Med. 2018, 197, 1308–1318. [Google Scholar] [CrossRef] [PubMed]
- Sly, P.D.; Brennan, S.; Gangell, C.; De Klerk, N.; Murray, C.; Mott, L.; Stick, S.M.; Robinson, P.J.; Robertson, C.F.; Ranganathan, S.C. Lung Disease at Diagnosis in Infants with Cystic Fibrosis Detected by Newborn Screening. Am. J. Respir. Crit. Care Med. 2009, 180, 146–152. [Google Scholar] [CrossRef] [PubMed]
- Sly, P.D.; Gangell, C.L.; Chen, L.; Ware, R.S.; Ranganathan, S.; Mott, L.S.; Murray, C.P.; Stick, S.M. Risk factors for bronchiectasis in children with cystic fibrosis. N. Engl. J. Med. 2013, 368, 1963–1970. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou-Suckow, Z.; Duerr, J.; Hagner, M.; Mall, M.A. Airway mucus, inflammation and remodeling: Emerging links in the patho-genesis of chronic lung diseases. Cell Tissue Res. 2017, 367, 537–550. [Google Scholar] [CrossRef] [PubMed]
- Trojanek, J.B.; Cobos-Correa, A.; Diemer, S.; Kormann, M.; Schubert, S.C.; Zhou-Suckow, Z.; Agrawal, R.; Duerr, J.; Wagner, C.J.; Schatterny, J.; et al. Airway Mucus Obstruction Triggers Macrophage Activation and Matrix Metalloproteinase 12–Dependent Emphysema. Am. J. Respir. Cell Mol. Biol. 2014, 51, 709–720. [Google Scholar] [CrossRef]
- Hiemstra, P.S.; McCray, P.B., Jr.; Bals, R. The innate immune function of airway epithelial cells in inflammatory lung disease. Eur. Respir. J. 2015, 45, 1150–1162. [Google Scholar] [CrossRef] [Green Version]
- Jasper, A.E.; McIver, W.J.; Sapey, E.; Walton, G.M. Understanding the role of neutrophils in chronic inflammatory airway disease. F1000Research 2019, 8, 557. [Google Scholar] [CrossRef]
- Hoenderdos, K.; Condliffe, A. The Neutrophil in Chronic Obstructive Pulmonary Disease. Too Little, Too Late or Too Much, Too Soon? Am. J. Respir. Cell Mol. Biol. 2013, 48, 531–539. [Google Scholar] [CrossRef] [PubMed]
- Bedi, P.; Davidson, D.J.; McHugh, B.J.; Rossi, A.G.; Hill, A.T. Blood Neutrophils Are Reprogrammed in Bronchiectasis. Am. J. Respir. Crit. Care Med. 2018, 198, 880–890. [Google Scholar] [CrossRef] [PubMed]
- Laval, J.; Ralhan, A.; Hartl, D. Neutrophils in cystic fibrosis. Biol. Chem. 2016, 397, 485–496. [Google Scholar] [CrossRef] [PubMed]
- Kapellos, T.S.; Bassler, K.; Aschenbrenner, A.C.; Fujii, W.; Schultze, J.L. Dysregulated Functions of Lung Macrophage Populations in COPD. J. Immunol. Res. 2018, 2018, 2349045. [Google Scholar] [CrossRef] [Green Version]
- Hartl, D.; Tirouvanziam, R.; Laval, J.; Greene, C.M.; Habiel, D.; Sharma, L.; Yildirim, A.Ö.; Cruz, C.S.D.; Hogaboam, C.M. Innate Immunity of the Lung: From Basic Mechanisms to Translational Medicine. J. Innate Immun. 2018, 10, 487–501. [Google Scholar] [CrossRef] [PubMed]
- Berenson, C.S.; Kruzel, R.L.; Eberhardt, E.; Sethi, S. Phagocytic Dysfunction of Human Alveolar Macrophages and Severity of Chronic Obstructive Pulmonary Disease. J. Infect. Dis. 2013, 208, 2036–2045. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lévêque, M.; Le Trionnaire, S.; Del Porto, P.; Martin-Chouly, C. The impact of impaired macrophage functions in cystic fibrosis disease progression. J. Cyst. Fibros. 2017, 16, 443–453. [Google Scholar] [CrossRef] [Green Version]
- Freeman, C.M.; Curtis, J.L. Lung dendritic cells: Shaping immune responses throughout chronic obstructive pulmonary disease progression. Am. J. Respir. Cell Mol. Biol. 2017, 56, 152–159. [Google Scholar]
- Ni, L.; Dong, C. Roles of Myeloid and Lymphoid Cells in the Pathogenesis of Chronic Obstructive Pulmonary Disease. Front. Immunol. 2018, 9. [Google Scholar] [CrossRef]
- David, B.; Bafadhel, M.; Koenderman, L.; De Soyza, A. Eosinophilic inflammation in COPD: From an inflammatory marker to a treatable trait. Thorax 2021, 76, 188–195. [Google Scholar] [CrossRef]
- Borger, J.G.; Lau, M.; Hibbs, M.L. The Influence of Innate Lymphoid Cells and Unconventional T Cells in Chronic Inflammatory Lung Disease. Front. Immunol. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Vethanayagam, R.R.; Almyroudis, N.G.; Grimm, M.J.; Lewandowski, D.C.; Pham, C.T.N.; Blackwell, T.S.; Petraitiene, R.; Petraitis, V.; Walsh, T.J.; Urban, C.F.; et al. Role of NADPH Oxidase versus Neutrophil Proteases in Antimicrobial Host Defense. PLoS ONE 2011, 6, e28149. [Google Scholar] [CrossRef]
- Clancy, D.M.; Sullivan, G.P.; Moran, H.B.; Henry, C.M.; Reeves, E.P.; McElvaney, N.G.; Lavelle, E.C.; Martin, S.J. Extracellular Neutrophil Proteases Are Efficient Regulators of IL-1, IL-33, and IL-36 Cytokine Activity but Poor Effectors of Microbial Killing. Cell Rep. 2018, 22, 2937–2950. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stapels, D.A.; Geisbrecht, B.V.; Rooijakkers, S.H. Neutrophil serine proteases in antibacterial defense. Curr. Opin. Microbiol. 2015, 23, 42–48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Standish, A.J.; Weiser, J.N. Human Neutrophils Kill Streptococcus pneumoniae via Serine Proteases. J. Immunol. 2009, 183, 2602–2609. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Belaaouaj, A.; McCarthy, R.T.; Baumann, M.L.; Gao, Z.; Ley, T.J.; Abraham, S.N.; Shapiro, S.D. Mice lacking neutrophil elastase reveal impaired host defense against gram negative bacterial sepsis. Nat. Med. 1998, 4, 615–618. [Google Scholar] [CrossRef] [PubMed]
- Hirche, T.O.; Benabid, R.; Deslee, G.; Gangloff, S.; Achilefu, S.; Guenounou, M.; Lebargy, F.; Hancock, R.E.; Belaaouaj, A. Neutrophil Elastase Mediates Innate Host Protection against Pseudomonas aeruginosa. J. Immunol. 2008, 181, 4945–4954. [Google Scholar] [CrossRef] [Green Version]
- Müller, S.; Faulhaber, A.; Sieber, C.; Pfeifer, D.; Hochberg, T.; Gansz, M.; Deshmukh, S.D.; Dauth, S.; Brix, K.; Saftig, P.; et al. The endolysosomal cysteine cathepsins L and K are involved in macrophage-mediated clearance of Staphylococcus aureus and the concomitant cytokine induction. FASEB J. 2013, 28, 162–175. [Google Scholar] [CrossRef]
- Szulc-Dąbrowska, L.; Bossowska-Nowicka, M.; Struzik, J.; Toka, F.N. Cathepsins in Bacteria-Macrophage Interaction: Defenders or Victims of Circumstance? Front. Cell. Infect. Microbiol. 2020, 10. [Google Scholar] [CrossRef]
- Ewald, S.E.; Engel, A.; Lee, J.; Wang, M.; Bogyo, M.; Barton, G.M. Nucleic acid recognition by Toll-like receptors is coupled to stepwise processing by cathepsins and asparagine endopeptidase. J. Exp. Med. 2011, 208, 643–651. [Google Scholar] [CrossRef] [PubMed]
- Tsukuba, T.; Yamamoto, S.; Yanagawa, M.; Okamoto, K.; Okamoto, Y.; Nakayama, K.I.; Kadowaki, T.; Yamamoto, K. Cathepsin E-deficient mice show in-creased susceptibility to bacterial infection associated with the decreased expression of multiple cell surface Toll-like receptors. J. Biochem. 2006, 140, 57–66. [Google Scholar] [CrossRef]
- Hsieh, C.-S.; DeRoos, P.; Honey, K.; Beers, C.; Rudensky, A.Y. A Role for Cathepsin L and Cathepsin S in Peptide Generation for MHC Class II Presentation. J. Immunol. 2002, 168, 2618–2625. [Google Scholar] [CrossRef]
- Padrines, M.; Wolf, M.; Walz, A.; Baggiolini, M. Interleukin-8 processing by neutrophil elastase, cathepsin G and proteinase-3. FEBS Lett. 1994, 352, 231–235. [Google Scholar] [CrossRef] [Green Version]
- Nufer, O.; Corbett, M.; Walz, A. Amino-Terminal Processing of Chemokine ENA-78 Regulates Biological Activity†. Biochemistry 1999, 38, 636–642. [Google Scholar] [CrossRef]
- Richter, R.; Bistrian, R.; Escher, S.; Forssmann, W.-G.; Vakili, J.; Henschler, R.; Spodsberg, N.; Frimpong-Boateng, A.; Forssmann, U. Quantum Proteolytic Activation of Chemokine CCL15 by Neutrophil Granulocytes Modulates Mononuclear Cell Adhesiveness. J. Immunol. 2005, 175, 1599–1608. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wittamer, V.; Bondue, B.; Guillabert, A.; Vassart, G.; Parmentier, M.; Communi, D. Neutrophil-Mediated Maturation of Chemerin: A Link between Innate and Adaptive Immunity. J. Immunol. 2005, 175, 487–493. [Google Scholar] [CrossRef]
- Schonbeck, U.; Mach, F.; Libby, P. Generation of biologically active IL-1 beta by matrix metalloproteinases: A novel caspa-se-1-independent pathway of IL-1 beta processing. J. Immunol. 1998, 161, 3340–3346. [Google Scholar] [PubMed]
- Lambrecht, B.N.; Vanderkerken, M.; Hammad, H. The emerging role of ADAM metalloproteinases in immunity. Nat. Rev. Immunol. 2018, 18, 745–758. [Google Scholar] [CrossRef]
- Gaggar, A.; Weathington, N. Bioactive extracellular matrix fragments in lung health and disease. J. Clin. Investig. 2016, 126, 3176–3184. [Google Scholar] [CrossRef] [Green Version]
- Turnbull, A.R.; Pyle, C.J.; Patel, D.F.; Jackson, P.L.; Hilliard, T.N.; Regamey, N.; Tan, H.-L.; Brown, S.; Thursfield, R.; Short, C.; et al. Abnormal pro-gly-pro pathway and airway neu-trophilia in pediatric cystic fibrosis. J. Cyst. Fibros. 2019, 2020, 40–48. [Google Scholar]
- Bonnans, C.; Chou, J.; Werb, Z. Remodelling the extracellular matrix in development and disease. Nat. Rev. Mol. Cell Biol. 2014, 15, 786–801. [Google Scholar] [CrossRef] [PubMed]
- Karakioulaki, M.; Papakonstantinou, E.; Stolz, D. Extracellular matrix remodelling in COPD. Eur. Respir. Rev. 2020, 29, 190124. [Google Scholar] [CrossRef] [PubMed]
- Taggart, C.C.; Greene, C.M.; Smith, S.G.; Levine, R.L.; McCray, P.B.; O’Neill, S.; McElvaney, N. Inactivation of human beta-defensins 2 and 3 by elastolytic cathepsins. J. Immunol. 2003, 171, 931–937. [Google Scholar] [CrossRef] [Green Version]
- Andrault, P.-M.; Samsonov, S.A.; Weber, G.; Coquet, L.; Nazmi, K.; Bolscher, J.G.M.; Lalmanach, A.-C.; Jouenne, T.; Brömme, D.; Pisabarro, M.T.; et al. Antimicrobial Peptide LL-37 Is Both a Substrate of Cathepsins S and K and a Selective Inhibitor of Cathepsin L. Biochemistry 2015, 54, 2785–2798. [Google Scholar] [CrossRef] [PubMed]
- Fischer, B.M.; Domowicz, D.A.L.; Zheng, S.; Carter, J.L.; McElvaney, N.G.; Taggart, C.; Lehmann, J.R.; Voynow, J.A.; Ghio, A.J. Neutrophil elastase increases airway epi-thelial nonheme iron levels. Clin. Transl. Sci. 2009, 2, 333–339. [Google Scholar] [CrossRef]
- Rubio, F.; Cooley, J.; Accurso, F.J.; Remold-O’Donnell, E. Linkage of neutrophil serine proteases and decreased surfactant protein-A (SP-A) levels in inflammatory lung disease. Thorax 2004, 59, 318–323. [Google Scholar] [CrossRef] [Green Version]
- Cooley, J.; McDonald, B.; Accurso, F.J.; Crouch, E.C.; Remold-O’Donnell, E. Patterns of neutrophil serine protease-dependent cleavage of surfactant protein D in inflammatory lung disease. J. Leukoc. Biol. 2008, 83, 946–955. [Google Scholar] [CrossRef]
- Bratcher, P.E.; Weathington, N.M.; Nick, H.J.; Jackson, P.L.; Snelgrove, R.J.; Gaggar, A. MMP-9 Cleaves SP-D and Abrogates Its Innate Immune Functions In Vitro. PLoS ONE 2012, 7, e41881. [Google Scholar] [CrossRef] [Green Version]
- Han, S.; Mallampalli, R.K. The Role of Surfactant in Lung Disease and Host Defense against Pulmonary Infections. Ann. Am. Thorac. Soc. 2015, 12, 765–774. [Google Scholar] [CrossRef] [Green Version]
- Linden, D.; Guo-Parke, H.; Coyle, P.V.; Fairley, D.; McAuley, D.F.; Taggart, C.C.; Kidney, J. Respiratory viral infection: A potential “missing link” in the pathogenesis of COPD. Eur. Respir. Rev. 2019, 28, 180063. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Böttcher-Friebertshäuser, E.; Freuer, C.; Sielaff, F.; Schmidt, S.; Eickmann, M.; Uhlendorff, J.; Steinmetzer, T.; Klenk, H.; Garten, W. Cleavage of Influenza Virus Hemagglutinin by Airway Proteases TMPRSS2 and HAT Differs in Subcellular Localization and Susceptibility to Protease In-hibitors. J. Virol. 2010, 84, 5605–5614. [Google Scholar] [CrossRef] [Green Version]
- Seth, S.; Batra, J.; Srinivasan, S. COVID-19: Targeting Proteases in Viral Invasion and Host Immune Response. Front. Mol. Biosci. 2020, 7, 215. [Google Scholar] [CrossRef]
- Simpson, A.; Maxwell, A.; Govan, J.; Haslett, C.; Sallenave, J.-M. Elafin (elastase-specific inhibitor) has anti-microbial activity against Gram-positive and Gram-negative respiratory pathogens. FEBS Lett. 1999, 452, 309–313. [Google Scholar] [CrossRef] [Green Version]
- Sallenave, J.-M. Secretory Leukocyte Protease Inhibitor and Elafin/Trappin-2. Am. J. Respir. Cell Mol. Biol. 2010, 42, 635–643. [Google Scholar] [CrossRef] [PubMed]
- Ries, C. Cytokine functions of TIMP-1. Cell. Mol. Life Sci. 2014, 71, 659–672. [Google Scholar] [CrossRef]
- Holloway, A.J.; Yu, J.; Arulanandam, B.P.; Hoskinson, S.M.; Eaves-Pyles, T. Cystatins 9 and C as a Novel Immunotherapy Treatment That Protects against Multidrug-Resistant New Delhi Metallo-Beta-Lactamase-1-ProducingKlebsiella pneumoniae. Antimicrob. Agents Chemother. 2017, 62. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guyot, N.; Butler, M.W.; McNally, P.; Weldon, S.; Greene, C.M.; Levine, R.L.; O’Neill, S.J.; Taggart, C.C.; McElvaney, N.G. Elafin, an Elastase-specific Inhibitor, Is Cleaved by Its Cognate Enzyme Neutrophil Elastase in Sputum from Individuals with Cystic Fibrosis. J. Biol. Chem. 2008, 283, 32377–32385. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weldon, S.; McNally, P.; McElvaney, N.G.; Elborn, J.S.; McAuley, D.F.; Wartelle, J.; Belaaouaj, A.; Levine, R.L.; Taggart, C.C. Decreased Levels of Secretory Leucoprotease Inhibitor in thePseudomonas-Infected Cystic Fibrosis Lung Are Due to Neutrophil Elastase Degradation. J. Immunol. 2009, 183, 8148–8156. [Google Scholar] [CrossRef] [Green Version]
- Kerrin, A.; Weldon, S.; Chung, A.H.-K.; Craig, T.; Simpson, A.J.; O’Kane, C.M.; McAuley, D.F.; Taggart, C.C. Proteolytic cleavage of elafin by 20S proteasome may contribute to inflammation in acute lung injury. Thorax 2013, 68, 315–321. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taggart, C.C.; Lowe, G.J.; Greene, C.M.; Mulgrew, A.T.; O’Neill, S.J.; Levine, R.L.; McElvaney, N.G. Cathepsin B, L, and S Cleave and Inactivate Secretory Leucoprotease Inhibitor. J. Biol. Chem. 2001, 276, 33345–33352. [Google Scholar] [CrossRef] [Green Version]
- Johnson, D.A.; Barrettli, A.J.; Masonli, R.W. Cathepsin L Inactivates Alpha 1-Proteinase Inhibitor by Cleavage in the Reactive Site Region. J. Biol. Chem. 1986, 261, 14748–14751. [Google Scholar] [CrossRef]
- Liu, Z.; Zhou, X.; Shapiro, S.D.; Shipley, J.; Twining, S.S.; Diaz, L.A.; Senior, R.M.; Werb, Z. The Serpin α1-Proteinase Inhibitor Is a Critical Substrate for Gelatinase B/MMP-9 In Vivo. Cell 2000, 102, 647–655. [Google Scholar] [CrossRef] [Green Version]
- Jackson, P.L.; Xu, X.; Wilson, L.; Weathington, N.M.; Clancy, J.P.; Blalock, J.E.; Gaggar, A. Human Neutrophil Elastase-Mediated Cleavage Sites of MMP-9 and TIMP-1: Implications to Cystic Fibrosis Proteolytic Dysfunction. Mol. Med. 2010, 16, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Buttle, D.J.; Abrahamson, M.; Burnett, D.; Mort, J.S.; Barrett, A.J.; Dando, P.M.; Hill, S.L. Human sputum cathepsin B degrades proteoglycan, is inhibited by α2-macroglobulin and is modulated by neutrophil elastase cleavage of cathepsin B precursor and cystatin C. Biochem. J. 1991, 276, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Dittrich, A.S.; Kühbandner, I.; Gehrig, S.; Rickert-Zacharias, V.; Twigg, M.; Wege, S.; Taggart, C.C.; Herth, F.; Schultz, C.; Mall, M.A. Elastase activity on sputum neutrophils correlates with severity of lung disease in cystic fibrosis. Eur. Respir. J. 2018, 51, 1701910. [Google Scholar] [CrossRef] [Green Version]
- Guerra, M.; Frey, D.; Hagner, M.; Dittrich, S.; Paulsen, M.; Mall, M.A.; Schultz, C. Cathepsin G Activity as a New Marker for Detecting Airway Inflammation by Microscopy and Flow Cytometry. ACS Central Sci. 2019, 5, 539–548. [Google Scholar] [CrossRef] [Green Version]
- Genschmer, K.R.; Russell, D.W.; Lal, C.; Szul, T.; Bratcher, P.E.; Noerager, B.D.; Roda, M.A.; Xu, X.; Rezonzew, G.; Viera, L.; et al. Activated PMN Exosomes: Pathogenic Entities Causing Matrix Destruction and Disease in the Lung. Cell 2019, 176, 113–126.e15. [Google Scholar] [CrossRef] [Green Version]
- Hu, H.-Y.; Gehrig, S.; Reither, G.; Subramanian, D.; Mall, M.A.; Plettenburg, O.; Schultz, C. FRET-based and other fluorescent proteinase probes. Biotechnol. J. 2014, 9, 266–281. [Google Scholar] [CrossRef]
- Hagner, M.; Frey, D.L.; Guerra, M.; Dittrich, A.S.; Halls, V.S.; Wege, S.; Herth, F.J.; Schultz, C.; Mall, M.A. New method for rapid and dynamic quantification of elastase activity on sputum neutrophils from patients with cystic fibrosis using flow cytometry. Eur. Respir. J. 2020, 55, 1902355. [Google Scholar] [CrossRef]
- Hamon, Y.; Legowska, M.; Hervé, V.; Dallet-Choisy, S.; Marchand-Adam, S.; Vanderlynden, L.; Demonte, M.; Williams, R.; Scott, C.J.; Si-Tahar, M.; et al. Neutrophilic Cathepsin C Is Maturated by a Multistep Proteolytic Process and Secreted by Activated Cells during Inflammatory Lung Diseases. J. Biol. Chem. 2016, 291, 8486–8499. [Google Scholar] [CrossRef] [Green Version]
- Méthot, N.; Rubin, J.; Guay, D.; Beaulieu, C.; Ethier, D.; Reddy, T.J.; Riendeau, D.; Percival, M.D. Inhibition of the Activation of Multiple Serine Proteases with a Cathepsin C Inhibitor Requires Sustained Exposure to Prevent Pro-enzyme Processing. J. Biol. Chem. 2007, 282, 20836–20846. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Garratt, L.W.; Sutanto, E.N.; Ling, K.-M.; Looi, K.; Iosifidis, T.; Martinovich, K.M.; Shaw, N.C.; Kicic-Starcevich, E.; Knight, D.A.; Ranganathan, S.; et al. Matrix metalloproteinase activation by free neutrophil elastase contributes to bronchiectasis progression in early cystic fibrosis. Eur. Respir. J. 2015, 46, 384–394. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heuberger, D.M.; Schuepbach, R.A. Protease-activated receptors (PARs): Mechanisms of action and potential therapeutic mod-ulators in PAR-driven inflammatory diseases. Thromb. J. 2019, 17, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mercer, P.F.; Williams, A.E.; Scotton, C.J.; José, R.J.; Sulikowski, M.; Moffatt, J.D.; Murray, L.A.; Chambers, R.C. Proteinase-Activated Receptor-1, CCL2 and CCL7 Regulate Acute Neutrophilic Lung Inflammation. Am. J. Respir. Cell Mol. Biol. 2013, 50, 144–157. [Google Scholar] [CrossRef] [Green Version]
- José, R.J.; Williams, A.E.; Mercer, P.F.; Sulikowski, M.G.; Brown, J.S.; Chambers, R.C. Regulation of Neutrophilic Inflammation by Pro-teinase-Activated Receptor 1 during Bacterial Pulmonary Infection. J. Immunol. 2015, 194, 6024–6034. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, C.; von der Thüsen, J.; Daalhuisen, J.; ten Brink, M.; Crestani, B.; van der Poll, T.; Borensztajn, K.; Spek, C.A. Pharmacological Targeting of Prote-ase-Activated Receptor 2 Affords Protection from Bleomycin-Induced Pulmonary Fibrosis. Mol. Med. 2015, 21, 576–583. [Google Scholar] [CrossRef] [PubMed]
- Asokananthan, N.; Graham, P.T.; Fink, J.; Knight, D.A.; Bakker, A.J.; McWilliam, A.S.; Thompson, P.J.; Stewart, G.A. Activation of Protease-Activated Receptor (PAR)-1, PAR-2, and PAR-4 Stimulates IL-6, IL-8, and Prostaglandin E2Release from Human Respiratory Epithelial Cells. J. Immunol. 2002, 168, 3577–3585. [Google Scholar] [CrossRef] [Green Version]
- Chambers, R.C.; Scotton, C.J. Coagulation Cascade Proteinases in Lung Injury and Fibrosis. Proc. Am. Thorac. Soc. 2012, 19, 96–101. [Google Scholar] [CrossRef]
- Small, D.M.; Brown, R.R.; Doherty, D.F.; Abladey, A.; Zhou-Suckow, Z.; Delaney, R.J.; Kerrigan, L.; Dougan, C.M.; Borensztajn, K.S.; Holsinger, L.; et al. Targeting of cathepsin S reduces cystic fibrosis-like lung disease. Eur. Respir. J. 2019, 53, 1801523. [Google Scholar] [CrossRef]
- Hou, H.-H.; Wang, H.-C.; Cheng, S.-L.; Chen, Y.-F.; Lu, K.-Z.; Yu, C.-J. MMP-12 activates protease-activated receptor-1, upregulates placenta growth factor, and leads to pulmonary emphysema. Am. J. Physiol. Cell. Mol. Physiol. 2018, 315, L432–L442. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Yamashita, C.; Zemans, R.L.; Briones, N.; Van Linden, A.; Downey, G.P. Leukocyte elastase induces lung epithelial apoptosis via a PAR-1-, NF-kappaB-, and p53-dependent pathway. Am. J. Respir. Cell Mol. Biol. 2009, 41, 742–755. [Google Scholar] [CrossRef] [Green Version]
- Kuliopulos, A.; Covic, L.; Seeley, S.K.; Sheridan, P.J.; Helin, J.; Costello, C.E. Plasmin Desensitization of the PAR1 Thrombin Receptor: Kinetics, Sites of Truncation, and Implications for Thrombolytic Therapy†. Biochemistry 1999, 38, 4572–4585. [Google Scholar] [CrossRef]
- Wittekindt, O.H. Tight junctions in pulmonary epithelia during lung inflammation. Pflügers Archiv-Eur. J. Physiol. 2017, 469, 135–147. [Google Scholar] [CrossRef] [Green Version]
- Huguenin, M.; Müller, E.J.; Trachsel-Rösmann, S.; Oneda, B.; Ambort, D.; Sterchi, E.E.; Lottaz, D. The Metalloprotease Meprinβ Processes E-Cadherin and Weakens Intercellular Adhesion. PLoS ONE 2008, 3, e2153. [Google Scholar] [CrossRef] [Green Version]
- Bao, J.; Yura, R.E.; Matters, G.L.; Bradley, S.G.; Shi, P.; Tian, F.; Bond, J.S. Meprin A impairs epithelial barrier function, enhances monocyte migration, and cleaves the tight junction protein occludin. Am. J. Physiol. Physiol. 2013, 305, F714–F726. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brune, K.; Frank, J.A.; Schwingshackl, A.; Finigan, J.H.; Sidhaye, V.K. Pulmonary epithelial barrier function: Some new players and mechanisms. Am. J. Physiol. Cell. Mol. Physiol. 2015, 308, L731–L745. [Google Scholar] [CrossRef] [Green Version]
- Churg, A.; Wang, R.; Wang, X.; Onnervik, P.-O.; Thim, K.; Wright, J.L. Effect of an MMP-9/MMP-12 inhibitor on smoke-induced emphysema and airway remodelling in guinea pigs. Thorax 2007, 62, 706–713. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Janelle, M.F.; Doucet, A.; Bouchard, D.; Bourbonnais, Y.; Tremblay, G.M. Increased local levels of granulocyte colony-stimulating factor are associated with the beneficial effect of pre-elafin (SKALP/trappin-2/WAP3) in experimental emphysema. Biol. Chem. 2006, 387, 903–909. [Google Scholar] [CrossRef] [PubMed]
- Gehrig, S.; Duerr, J.; Weitnauer, M.; Wagner, C.J.; Graeber, S.Y.; Schatterny, J.; Hirtz, S.; Belaaouaj, A.; Dalpke, A.H.; Schultz, C.; et al. Lack of Neutrophil Elastase Reduces Inflammation, Mucus Hypersecretion, and Emphysema, but Not Mucus Obstruction, in Mice with Cystic Fibrosis–like Lung Disease. Am. J. Respir. Crit. Care Med. 2014, 189, 1082–1092. [Google Scholar] [CrossRef]
- Dirksen, A.; Piitulainen, E.; Parr, D.G.; Deng, C.; Wencker, M.; Shaker, S.B.; Stockleye, R.A. Exploring the role of CT densitometry: A randomised study of augmentation therapy in α1-antitrypsin deficiency. Eur. Respir. J. 2009, 33, 1345–1353. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chapman, K.R.; Burdon, J.G.W.; Piitulainen, E.; Sandhaus, R.A.; Seersholm, N.; Stocks, J.M.; Stoel, B.C.; Huang, L.; Yao, Z.; Edelman, J.M.; et al. Intravenous augmentation treatment and lung density in severe α1 antitrypsin deficiency (RAPID): A randomised, double-blind, placebo-controlled trial. Lancet 2015, 386, 360–368. [Google Scholar] [CrossRef]
- McElvaney, N.G.; Burdon, J.; Holmes, M.; Glanville, A.; Wark, P.A.B.; Thompson, P.J.; Hernandez, P.; Chlumsky, J.; Teschler, H.; Ficker, J.H.; et al. Long-term efficacy and safety of α1 proteinase inhibitor treatment for emphysema caused by severe α1 antitrypsin deficiency: An open-label extension trial (RAP-ID-OLE). Lancet Respir. Med. 2017, 5, 51–60. [Google Scholar] [CrossRef]
- Owen, C.A.; Campbell, M.A.; Sannes, P.L.; Boukedes, S.S.; Campbell, E.J. Cell surface-bound elastase and cathepsin G on human neutrophils: A novel, non-oxidative mechanism by which neutrophils focus and preserve catalytic activity of serine proteinases. J. Cell Biol. 1995, 131, 775–789. [Google Scholar] [CrossRef]
- Owen, C.A.; Hu, Z.; Barrick, B.; Shapiro, S.D. Inducible expression of tissue inhibitor of metalloproteinases-resistant matrix metal-loproteinase-9 on the cell surface of neutrophils. Am. J. Respir. Cell Mol. Biol. 2003, 29, 283–294. [Google Scholar] [CrossRef]
- Allan, E.R.O.; Yates, R.M. Redundancy between Cysteine Cathepsins in Murine Experimental Autoimmune Encephalomyelitis. PLoS ONE 2015, 10, e0128945. [Google Scholar] [CrossRef]
- Vandenbroucke, R.E.; Libert, C. Is there new hope for therapeutic matrix metalloproteinase inhibition? Nat. Rev. Drug Discov. 2014, 13, 904–927. [Google Scholar] [CrossRef]
- Griese, M.; Latzin, P.; Kappler, M.; Weckerle, K.; Heinzimaier, T.; Bernhardt, T.; Hartl, D. α1-Antitrypsin inhalation reduces airway inflammation in cystic fibrosis patients. Eur. Respir. J. 2007, 29, 240–250. [Google Scholar] [CrossRef] [Green Version]
- Phase II Study of the Safety and Efficacy of Inhaled Alpha-1 Antitrypsin (AAT) in Cystic Fibrosis Patients-Full Text View-ClinicalTrials.gov. Available online: https://www.clinicaltrials.gov/ct2/show/NCT00499837 (accessed on 4 January 2021).
- Inhaled A1AT in Adult Stable Bronchiectasis-Full Text View-ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT03383939 (accessed on 25 March 2021).
- Campos, M.A.; Geraghty, P.; Holt, G.; Mendes, E.; Newby, P.R.; Ma, S.; Luna-Diaz, L.V.; Turino, G.M.; Stockley, R.A. The Biological Effects of Double-Dose Alpha-1 Antitrypsin Augmentation Therapy. A Pilot Clinical Trial. Am. J. Respir. Crit. Care Med. 2019, 200, 318–326. [Google Scholar] [CrossRef] [PubMed]
- Phase II, Safety and Efficacy Study of Kamada-alpha-1-antitrypsin (AAT) for Inhalation-Study Results-ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT02001688 (accessed on 25 March 2021).
- Stage 1 Study of ARALAST NP and GLASSIA in A1PI Deficiency-Study Results-ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT02722304 (accessed on 25 March 2021).
- Elborn, J.S.; Perrett, J.; Forsman-Semb, K.; Marks-Konczalik, J.; Gunawardena, K.; Entwistle, N. Efficacy, safety and effect on biomarkers of AZD9668 in cystic fibrosis. Eur. Respir. J. 2012, 40, 969–976. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuna, P.; Jenkins, M.; O’Brien, C.D.; Fahy, W.A. AZD9668, a neutrophil elastase inhibitor, plus ongoing budesonide/ formoterol in patients with COPD. Respir. Med. 2012, 106, 531–539. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vogelmeier, C.; Aquino, T.O.; O’Brien, C.D.; Perrett, J.; Gunawardena, K.A. A randomised, placebo-controlled, dose-finding study of AZD9668, an oral inhibitor of neutrophil elastase, in patients with chronic obstructive pulmonary disease treated with tiotropium. COPD J. Chronic Obstr. Pulm. Dis. 2012, 9, 111–120. [Google Scholar] [CrossRef]
- Stockley, R.; De Soyza, A.; Gunawardena, K.; Perrett, J.; Forsman-Semb, K.; Entwistle, N.; Snell, N. Phase II study of a neutrophil elastase inhibitor (AZD9668) in patients with bronchiectasis. Respir. Med. 2013, 107, 524–533. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- A Study to Assess Safety, Tolerability and Pharmacokinetics of of AZD6553 in Healthy Volunteers and Patients with Chronic Obstructive Pulmonary Disease (COPD)-Full Text View-ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT01068184 (accessed on 25 March 2021).
- A 12-Week Study Treating Participants Who Have Alpha1-Antitrypsin-Related COPD with Alvelestat (MPH966) or Placebo. Full Text View-ClinicalTrials.gov. Available online: https://www.clinicaltrials.gov/ct2/show/NCT03636347 (accessed on 25 March 2021).
- Barth, P.; Bruijnzeel, P.; Wach, A.; Kessler, O.S.; Hooftman, L.; Zimmermann, J.; Naue, N.; Huber, B.; Heimbeck, I.; Kappeler, D.; et al. Single dose escalation studies with inhaled POL6014, a potent novel selective reversible inhibitor of human neutrophil elastase, in healthy volunteers and subjects with cystic fibrosis. J. Cyst. Fibros. 2020, 19, 299–304. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watz, H.; Pedersen, F.; Kirsten, A.; Nagelschmitz, J.; Bandel, T.-J.; Schwers, S.; Rabe, K. Safety and tolerability of the NE inhibitor BAY 85-8501 in patients with non-CF bronchiectasis. Eur. Respir. J. 2016, 48, PA4088. [Google Scholar] [CrossRef]
- Clinical Study to Investigate Safety, Tolerability, Pharmacokinetics and Pharmacodynamics of POL6014 in Patients with CF Full Text View-ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT03748199 (accessed on 25 March 2021).
- A Study to Investigate Safety, Tolerability and Pharmacokinetics of Single and Repeat Doses of CHF6333 in Healthy Subjects-Full Text View-ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT03056326 (accessed on 25 March 2021).
- A Study in Patients with Non-cystic Fibrosis Bronchiectasis to Test How Well Different Doses of BI 1323495 Are Tolerated and How BI 1323495 Affects Biomarkers of Inflammation-Full Text View-ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT04656275 (accessed on 25 March 2021).
- Watz, H.; Nagelschmitz, J.; Kirsten, A.; Pedersen, F.; van der Mey, D.; Schwers, S.; Bandel, T.-J.; Rabe, K.F. Safety and efficacy of the human neutrophil elastase inhibitor BAY 85-8501 for the treatment of non-cystic fibrosis bronchiectasis: A randomized controlled trial. Pulm. Pharmacol. Ther. 2019, 56, 86–93. [Google Scholar] [CrossRef]
- Dahl, R.; Titlestad, I.; Lindqvist, A.; Wielders, P.; Wray, H.; Wang, M.; Samuelsson, V.; Mo, J.; Holt, A. Effects of an oral MMP-9 and -12 inhibitor, AZD1236, on biomarkers in moderate/severe COPD: A randomised controlled trial. Pulm. Pharmacol. Ther. 2012, 25, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, J.D.; Haworth, C.S.; Metersky, M.L.; Loebinger, M.R.; Blasi, F.; Sibila, O.; O’Donnell, A.E.; Sullivan, E.J.; Mange, K.C.; Fernandez, C.; et al. Phase 2 Trial of the DPP-1 Inhibitor Brensocatib in Bronchiectasis. N. Engl. J. Med. 2020, 383, 2127–2137. [Google Scholar] [CrossRef] [PubMed]
- Geraghty, P.; Rogan, M.P.; Greene, C.M.; Boxio, R.M.M.; Poiriert, T.; O’Mahony, M.; Belaaouaj, A.; O’Neill, S.J.; Taggart, C.C.; McElvaney, N.G. Neutrophil elastase up-regulates cathepsin B and matrix metalloprotease-2 expression. J. Immunol. 2007, 178, 5871–5878. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Geraghty, P.; Rogan, M.P.; Greene, C.M.; Brantly, M.L.; O’Neill, S.J.; Taggart, C.C.; McElvaney, N.G. Alpha-1-antitrypsin aerosolised augmentation abrogates neutrophil elastase-induced expression of cathepsin B and matrix metalloprotease 2 in vivo and in vitro. Thorax 2008, 63, 621–626. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Donnell, A.E.; Barker, A.F.; Ilowite, J.S.; Fick, R.B. Treatment of Idiopathic Bronchiectasis with Aerosolized Recombinant Human DNase I. Chest 1998, 113, 1329–1334. [Google Scholar] [CrossRef] [Green Version]
- Fuchs, H.J.; Borowitz, D.S.; Christiansen, D.H.; Morris, E.M.; Nash, M.L.; Ramsey, B.W.; Rosenstein, B.J.; Smith, A.L.; Wohl, M.E. Effect of Aerosolized Recombinant Human DNase on Exacerbations of Respiratory Symptoms and on Pulmonary Function in Patients with Cystic Fibrosis. N. Engl. J. Med. 1994, 331, 637–642. [Google Scholar] [CrossRef]
- Decramer, M.; Rutten-Van Mölken, M.; Dekhuijzen, P.N.R.; Troosters, T.; Van Herwaarden, C.; Pellegrino, R.; Van Schayk, C.P.; Olivieri, D.; Del Donno, M.; De Backer, W.; et al. Effects of N-acetylcysteine on outcomes in chronic obstructive pulmonary disease (Bronchitis Randomized on NAC Cost-Utility Study, BRONCUS): A randomised placebo-controlled trial. Lancet 2005, 365, 1552–1560. [Google Scholar] [CrossRef]
- Dal Negro, R.W.; Wedzicha, J.A.; Iversen, M.; Fontana, G.; Page, C.; Cicero, A.F.; Pozzi, E.; Calverley, P.M.A. Effect of erdosteine on the rate and duration of COPD exacerbations: The RESTORE study. Eur. Respir. J. 2017, 50, 1700711. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aaron, S.D. Mucolytics for COPD: Negotiating a slippery slope towards proof of efficacy. Eur. Respir. J. 2017, 50, 1701465. [Google Scholar] [CrossRef] [PubMed]
- Leal, J.; Smyth, H.D.; Ghosh, D. Physicochemical properties of mucus and their impact on transmucosal drug delivery. Int. J. Pharm. 2017, 532, 555–572. [Google Scholar] [CrossRef]
| Target Protease | Inhibitor | Disease | Stage | Outcome |
|---|---|---|---|---|
| NE | A1-AT | CF | II | Reduced inflammation, no effect on lung function [189,190] |
| Bronchiectasis | I | Results unpublished [191] | ||
| COPD/A1-AT deficiency | II/III | Reduced serine protease levels, reduces elastin degradation in the lung, reduced inflammation [192,193,194] | ||
| AZD9668 | CF | II | Reduced inflammation, no effect on lung function [195] | |
| COPD | II | No changes in lung function or inflammation [196,197] | ||
| Bronchiectasis | II | Improved lung function and reduced sputum inflammatory biomarkers [198] | ||
| AZD6553 | COPD | I | Terminated due to emerging PK profile that could not be aligned to the known pharmaceutical properties of the IMP [199] | |
| Alvelestat | COPD | II | Currently recruiting [200] | |
| POL6014 | CF | I | No serious adverse effects noted [201,202] | |
| CHF6333 | CF + non-CF bronchiectasis | I | Results unpublished [203] | |
| BI 1323495 | Bronchiectasis | I | Currently recruiting [204] | |
| BAY85-8501 | Bronchiectasis | II | No changes in lung function or inflammation [205,206] | |
| MMP-9/-12 | AZD1236 | COPD | II | No clinical efficacy observed [207] |
| Cathepsin C | Brensocatib | Bronchiectasis | II | Improved clinical outcomes with reduced NE activity, reduced time to first exacerbation [208] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
McKelvey, M.C.; Brown, R.; Ryan, S.; Mall, M.A.; Weldon, S.; Taggart, C.C. Proteases, Mucus, and Mucosal Immunity in Chronic Lung Disease. Int. J. Mol. Sci. 2021, 22, 5018. https://doi.org/10.3390/ijms22095018
McKelvey MC, Brown R, Ryan S, Mall MA, Weldon S, Taggart CC. Proteases, Mucus, and Mucosal Immunity in Chronic Lung Disease. International Journal of Molecular Sciences. 2021; 22(9):5018. https://doi.org/10.3390/ijms22095018
Chicago/Turabian StyleMcKelvey, Michael C., Ryan Brown, Sinéad Ryan, Marcus A. Mall, Sinéad Weldon, and Clifford C. Taggart. 2021. "Proteases, Mucus, and Mucosal Immunity in Chronic Lung Disease" International Journal of Molecular Sciences 22, no. 9: 5018. https://doi.org/10.3390/ijms22095018
APA StyleMcKelvey, M. C., Brown, R., Ryan, S., Mall, M. A., Weldon, S., & Taggart, C. C. (2021). Proteases, Mucus, and Mucosal Immunity in Chronic Lung Disease. International Journal of Molecular Sciences, 22(9), 5018. https://doi.org/10.3390/ijms22095018








