RNA Sequencing Reveals the Upregulation of FOXO Signaling Pathway in Porphyromonas gingivalis Persister-Treated Human Gingival Epithelial Cells
Abstract
:1. Introduction
2. Results
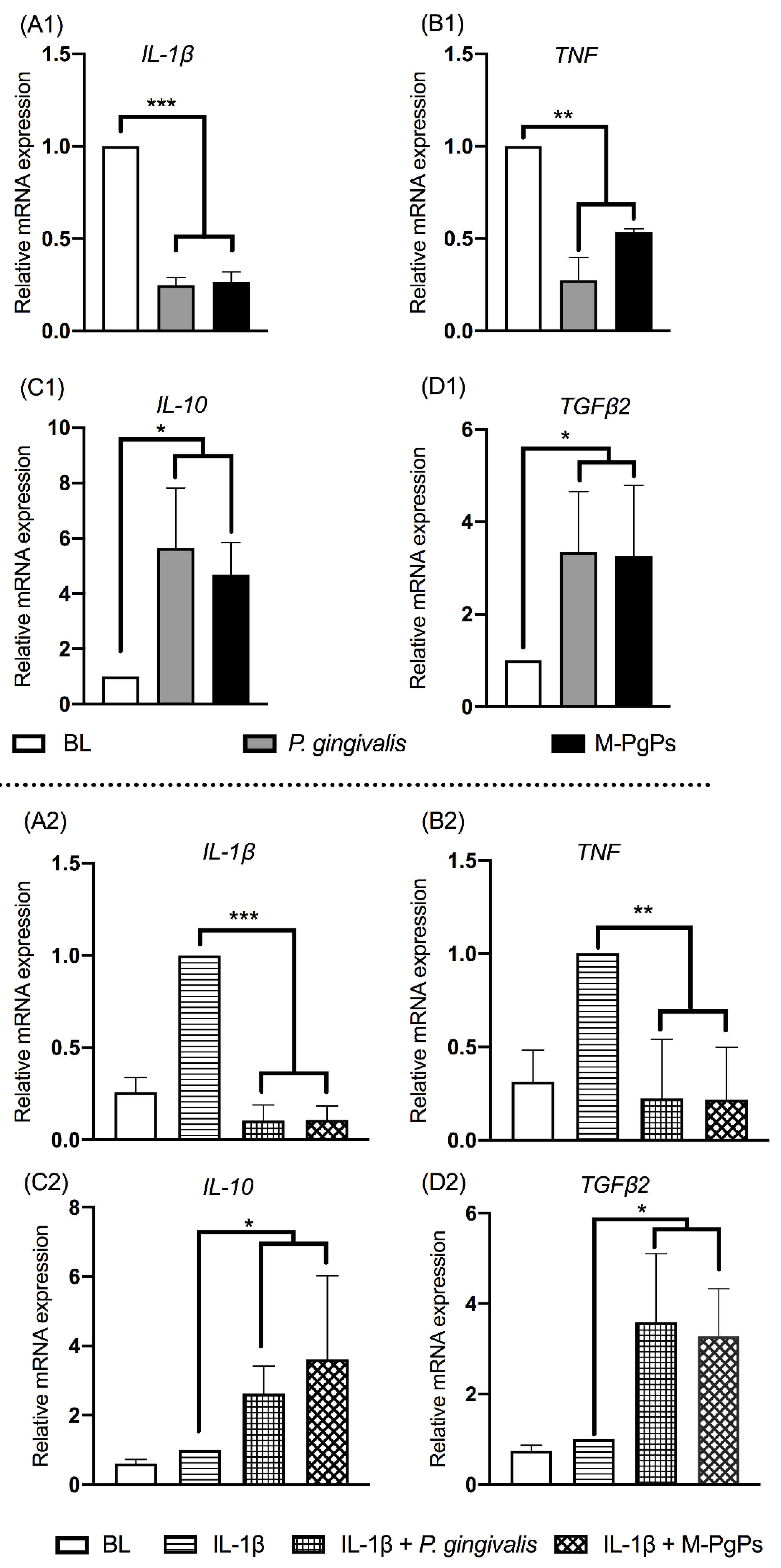
2.1. M-PgPs Inhibited the Innate Host Defense in HGECs
2.2. Differential Gene Expression
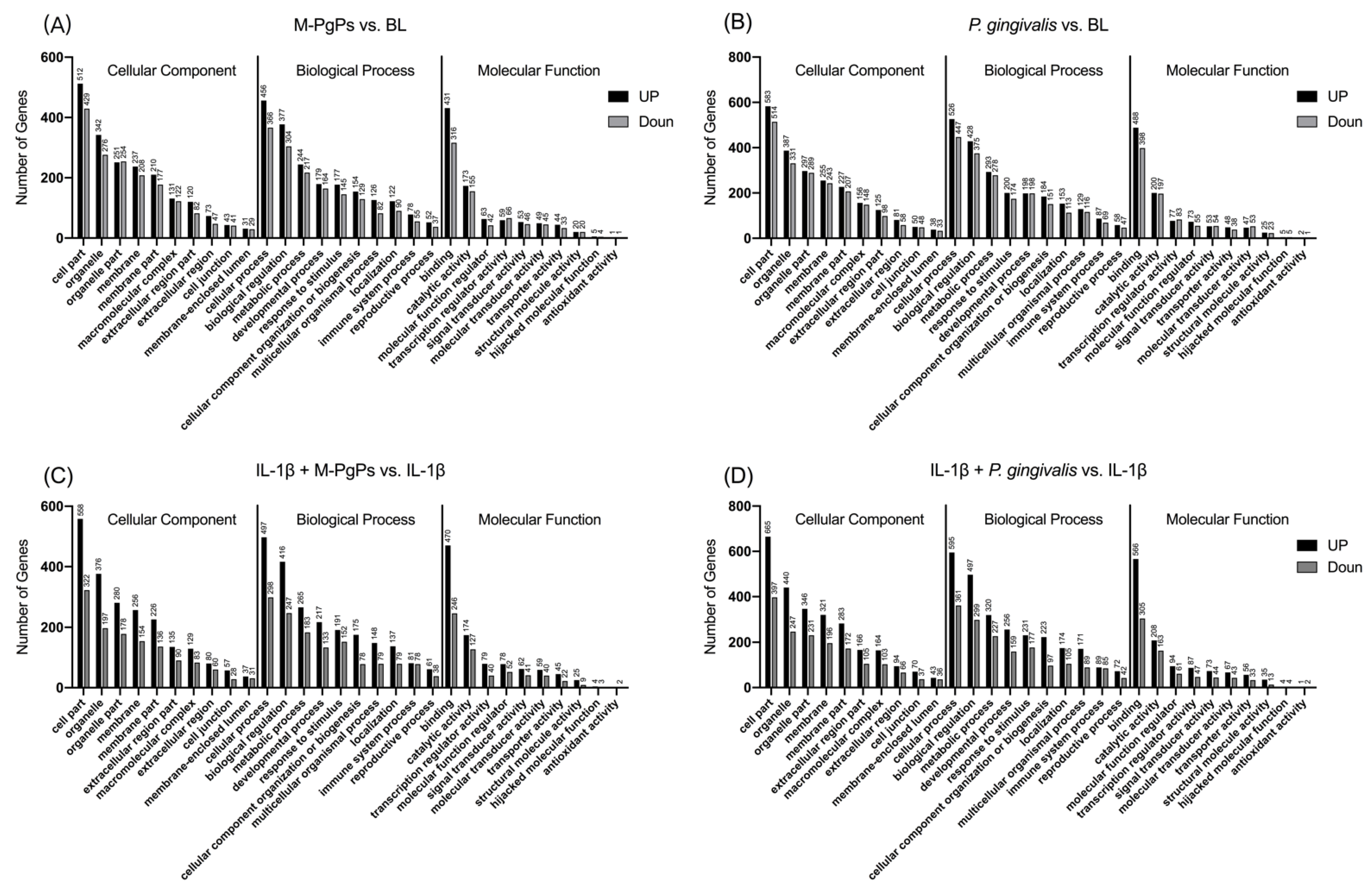
2.3. Gene Ontology (GO) Functional Annotation and Enrichment Analyses
2.4. KEGG Pathway Enrichment Analysis
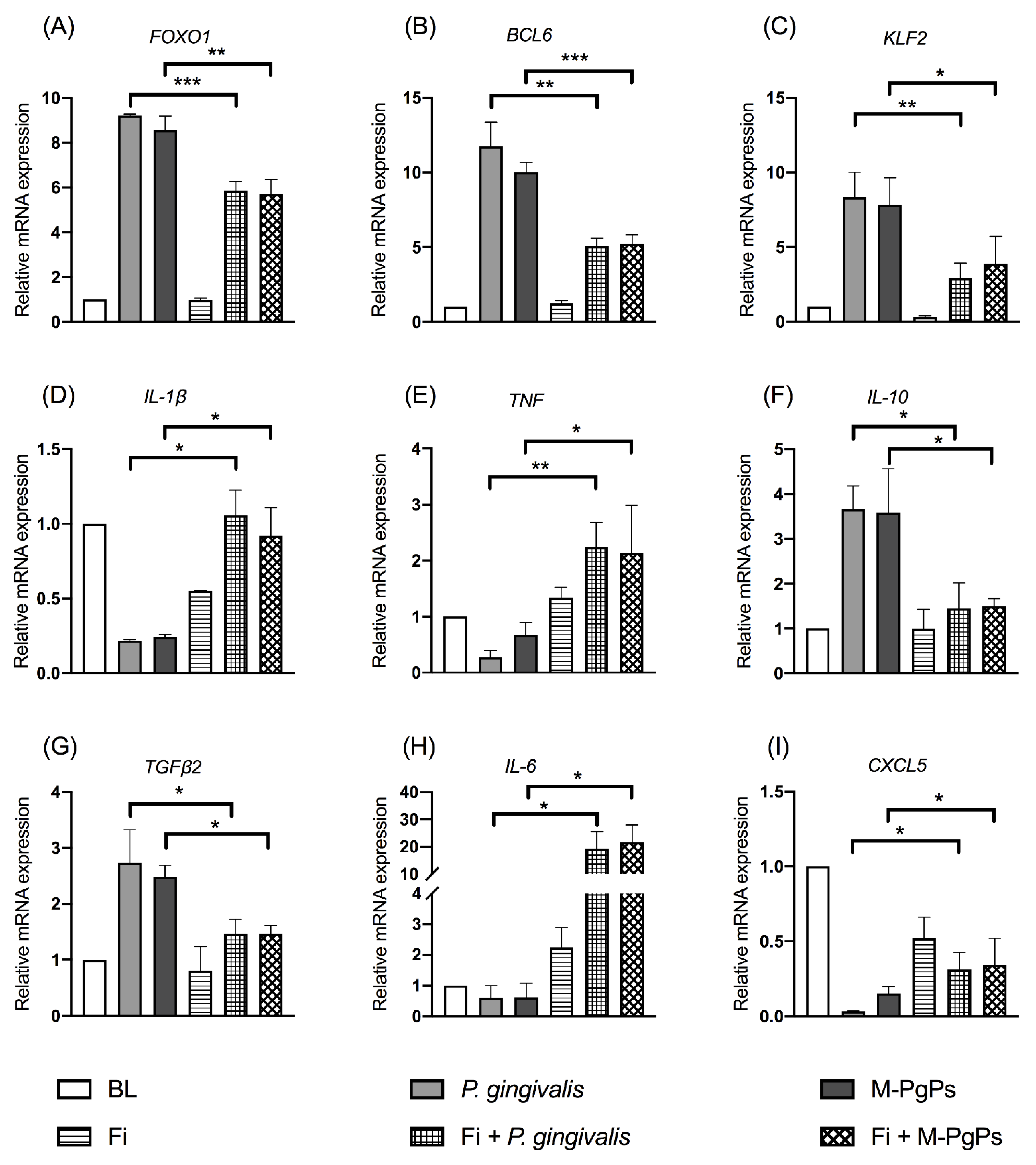
2.5. FOXO Signaling Pathway Was Involved in the Inhibitory Effects of M-PgPs on the Innate Host Defense in HGECs
3. Discussion
4. Materials and Methods
4.1. Culture of Cells
4.2. Bacterial Culture and M-PgPs Formation
4.3. Infection of HGECs with M-PgPs
4.4. RT-qPCR
4.5. Library Preparation and RNA Sequencing
4.6. Data Filtering and Alignment
4.7. Analysis of Differentially Expressed Genes
4.8. Functional Annotation and Enrichment Analysis
4.9. Cell Viability Test
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pihlstrom, B.L.; Michalowicz, B.S.; Johnson, N.W. Periodontal diseases. Lancet 2005, 366, 1809–1820. [Google Scholar] [CrossRef] [Green Version]
- Petersen, P.E.; Ogawa, H. The global burden of periodontal disease: Towards integration with chronic disease prevention and control. Periodontol. 2000 2012, 60, 15–39. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.J.; Armitage, G.C.; Klinge, B.; Lang, N.P.; Tonetti, M.; Williams, R.C. Global oral health inequalities: Task group—Periodontal disease. Adv. Dent. Res. 2011, 23, 221–226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kassebaum, N.J.; Bernabe, E.; Dahiya, M.; Bhandari, B.; Murray, C.J.; Marcenes, W. Global burden of severe periodontitis in 1990–2010: A systematic review and meta-regression. J. Dent. Res. 2014, 93, 1045–1053. [Google Scholar] [CrossRef]
- Listl, S.; Galloway, J.; Mossey, P.A.; Marcenes, W. Global economic impact of dental diseases. J. Dent. Res. 2015, 94, 1355–1361. [Google Scholar] [CrossRef] [PubMed]
- Herrera, D.; Meyle, J.; Renvert, S.; Jin, L. White Paper on Prevention and Management of Periodontal Diseases for Oral Health and General Health. Available online: https://www.fdiworlddental.org/sites/default/files/2020-11/gphp-2018-white_paper-en.pdf (accessed on 15 April 2022).
- Tonetti, M.S.; Kornman, K.S. Special Issue: Periodontitis and Systemic Diseases—Proceedings of a workshop jointly held by the European Federation of Periodontology and American Academy of Periodontology. J. Clin. Periodontol. 2013, 40, S1–S214. [Google Scholar]
- Jin, L.J.; Lamster, I.B.; Greenspan, J.S.; Pitts, N.B.; Scully, C.; Warnakulasuriya, S. Global burden of oral diseases: Emerging concepts, management and interplay with systemic health. Oral. Dis. 2016, 22, 609–619. [Google Scholar] [CrossRef]
- Monsarrat, P.; Blaizot, A.; Kemoun, P.; Ravaud, P.; Nabet, C.; Sixou, M.; Vergnes, J.N. Clinical research activity in periodontal medicine: A systematic mapping of trial registers. J. Clin. Periodontol. 2016, 43, 390–400. [Google Scholar] [CrossRef] [PubMed]
- Tonetti, M.S.; Jepsen, S.; Jin, L.; Otomo-Corgel, J. Impact of the global burden of periodontal diseases on health, nutrition and wellbeing of mankind: A call for global action. J. Clin. Periodontol. 2017, 44, 456–462. [Google Scholar] [CrossRef] [Green Version]
- Hajishengallis, G.; Darveau, R.P.; Curtis, M.A. The keystone-pathogen hypothesis. Nat. Rev. Microbiol. 2012, 10, 717–725. [Google Scholar] [CrossRef]
- Honda, K. Porphyromonas gingivalis sinks teeth into the oral microbiota and periodontal disease. Cell Host Microbe 2011, 10, 423–425. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hajishengallis, G.; Liang, S.; Payne, M.A.; Hashim, A.; Jotwani, R.; Eskan, M.A.; McIntosh, M.L.; Alsam, A.; Kirkwood, K.L.; Lambris, J.D.; et al. Low-abundance biofilm species orchestrates inflammatory periodontal disease through the commensal microbiota and complement. Cell Host Microbe 2011, 10, 497–506. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hajishengallis, G. Periodontitis: From microbial immune subversion to systemic inflammation. Nat. Rev. Immunol. 2015, 15, 30–44. [Google Scholar] [CrossRef] [PubMed]
- Hajishengallis, G.; Chavakis, T. Local and systemic mechanisms linking periodontal disease and inflammatory comorbidities. Nat. Rev. Immunol. 2021, 21, 426–440. [Google Scholar] [CrossRef] [PubMed]
- Uehara, A.; Muramoto, K.; Imamura, T.; Nakayama, K.; Potempa, J.; Travis, J.; Sugawara, S.; Takada, H. Arginine-specific gingipains from Porphyromonas gingivalis stimulate production of hepatocyte growth factor (scatter factor) through protease-activated receptors in human gingival fibroblasts in culture. J. Immunol. 2005, 175, 6076–6084. [Google Scholar] [CrossRef] [Green Version]
- Hajishengallis, G.; Wang, M.; Liang, S.; Triantafilou, M.; Triantafilou, K. Pathogen induction of CXCR4/TLR2 cross-talk impairs host defense function. Proc. Natl. Acad. Sci. USA 2008, 105, 13532–13537. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, P.; Fung, Y.E.; Yin, X.; Seneviratne, C.J.; Che, C.M.; Jin, L. Controlled cellular redox, repressive hemin utilization and adaptive stress responses are crucial to metronidazole tolerance of Porphyromonas gingivalis persisters. J. Clin. Periodontol. 2018, 45, 1211–1221. [Google Scholar] [CrossRef]
- Wang, C.; Cheng, T.; Li, X.; Jin, L. Metronidazole-treated Porphyromonas gingivalis persisters invade human gingival epithelial cells and perturb innate responses. Antimicrob. Agents Chemother. 2020, 64, e02529-19. [Google Scholar] [CrossRef]
- Lewis, K. Persister cells. Annu. Rev. Microbiol. 2010, 64, 357–372. [Google Scholar] [CrossRef]
- Bigger, J. Treatment of Staphylococcal infections with penicillin by intermittent sterilisation. Lancet 1944, 244, 497–500. [Google Scholar] [CrossRef]
- Fisher, R.A.; Gollan, B.; Helaine, S. Persistent bacterial infections and persister cells. Nat. Rev. Microbiol. 2017, 15, 453–464. [Google Scholar] [CrossRef] [PubMed]
- Mulcahy, L.R.; Burns, J.L.; Lory, S.; Lewis, K. Emergence of Pseudomonas aeruginosa strains producing high levels of persister cells in patients with cystic fibrosis. J. Bacteriol. 2010, 192, 6191–6199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lafleur, M.D.; Qi, Q.; Lewis, K. Patients with long-term oral carriage harbor high-persister mutants of Candida albicans. Antimicrob. Agents Chemother. 2010, 54, 39–44. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goneau, L.W.; Yeoh, N.S.; MacDonald, K.W.; Cadieux, P.A.; Burton, J.P.; Razvi, H.; Reid, G. Selective target inactivation rather than global metabolic dormancy causes antibiotic tolerance in uropathogens. Antimicrob. Agents Chemother. 2014, 58, 2089–2097. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jain, P.; Weinrick, B.C.; Kalivoda, E.J.; Yang, H.; Munsamy, V.; Vilcheze, C.; Weisbrod, T.R.; Larsen, M.H.; O’Donnell, M.R.; Pym, A.; et al. Dual-reporter Mycobacteriophages (Phi2DRMs) reveal preexisting Mycobacterium tuberculosis persistent cells in human sputum. MBio 2016, 7, e01023-16. [Google Scholar] [CrossRef] [Green Version]
- Stapels, D.A.C.; Hill, P.W.S.; Westermann, A.J.; Fisher, R.A.; Thurston, T.L.; Saliba, A.E.; Blommestein, I.; Vogel, J.; Helaine, S. Salmonella persisters undermine host immune defenses during antibiotic treatment. Science 2018, 362, 1156–1160. [Google Scholar] [CrossRef] [Green Version]
- Bakkeren, E.; Huisman, J.S.; Fattinger, S.A.; Hausmann, A.; Furter, M.; Egli, A.; Slack, E.; Sellin, M.E.; Bonhoeffer, S.; Regoes, R.R.; et al. Salmonella persisters promote the spread of antibiotic resistance plasmids in the gut. Nature 2019, 573, 276–280. [Google Scholar] [CrossRef]
- Gunther, J.; Seyfert, H.M. The first line of defence: Insights into mechanisms and relevance of phagocytosis in epithelial cells. Semin. Immunopathol. 2018, 40, 555–565. [Google Scholar] [CrossRef] [Green Version]
- Ji, S.; Choi, Y. Innate immune response to oral bacteria and the immune evasive characteristics of periodontal pathogens. J. Periodontal. Implant Sci. 2013, 43, 3–11. [Google Scholar] [CrossRef]
- Ramage, G.; Lappin, D.F.; Millhouse, E.; Malcolm, J.; Jose, A.; Yang, J.; Bradshaw, D.J.; Pratten, J.R.; Culshaw, S. The epithelial cell response to health and disease associated oral biofilm models. J. Periodontal. Res. 2017, 52, 325–333. [Google Scholar] [CrossRef] [Green Version]
- Rath-Deschner, B.; Memmert, S.; Damanaki, A.; de Molon, R.S.; Nokhbehsaim, M.; Eick, S.; Kirschneck, C.; Cirelli, J.A.; Deschner, J.; Jager, A.; et al. CXCL5, CXCL8, and CXCL10 regulation by bacteria and mechanical forces in periodontium. Ann. Anat. 2021, 234, 151648. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Zhang, X.; Xiang, Y.; Qu, X.; Liu, H.; Liu, C.; Tan, M.; Jiang, J.; Qin, X. Role of epithelial chemokines in the pathogenesis of airway inflammation in asthma (Review). Mol. Med. Rep. 2018, 17, 6935–6941. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elkaim, R.; Bugueno-Valdebenito, I.M.; Benkirane-Jessel, N.; Tenenbaum, H. Porphyromonas gingivalis and its lipopolysaccharide differently modulate epidermal growth factor-dependent signaling in human gingival epithelial cells. J. Oral. Microbiol. 2017, 9, 1334503. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Albuquerque-Souza, E.; Balzarini, D.; Ando-Suguimoto, E.S.; Ishikawa, K.H.; Simionato, M.R.L.; Holzhausen, M.; Mayer, M.P.A. Probiotics alter the immune response of gingival epithelial cells challenged by Porphyromonas gingivalis. J. Periodontal. Res. 2019, 54, 115–127. [Google Scholar] [CrossRef]
- Lee, K.; Roberts, J.S.; Choi, C.H.; Atanasova, K.R.; Yilmaz, O. Porphyromonas gingivalis traffics into endoplasmic reticulum-rich-autophagosomes for successful survival in human gingival epithelial cells. Virulence 2018, 9, 845–859. [Google Scholar] [CrossRef] [Green Version]
- Eijkelenboom, A.; Burgering, B.M. FOXOs: Signalling integrators for homeostasis maintenance. Nat. Rev. Mol. Cell Biol. 2013, 14, 83–97. [Google Scholar] [CrossRef]
- Peng, S.L. Immune regulation by Foxo transcription factors. Autoimmunity 2007, 40, 462–469. [Google Scholar] [CrossRef]
- Peng, S.L. Foxo in the immune system. Oncogene 2008, 27, 2337–2344. [Google Scholar] [CrossRef] [Green Version]
- Kandula, V.; Kosuru, R.; Li, H.; Yan, D.; Zhu, Q.; Lian, Q.; Ge, R.S.; Xia, Z.; Irwin, M.G. Forkhead box transcription factor 1: Role in the pathogenesis of diabetic cardiomyopathy. Cardiovasc. Diabetol. 2016, 15, 44. [Google Scholar] [CrossRef] [Green Version]
- Dong, G.; Song, L.; Tian, C.; Wang, Y.; Miao, F.; Zheng, J.; Lu, C.; Alsadun, S.; Graves, D.T. FOXO1 Regulates Bacteria-Induced Neutrophil Activity. Front. Immunol. 2017, 8, 1088. [Google Scholar] [CrossRef] [Green Version]
- Xing, Y.Q.; Li, A.; Yang, Y.; Li, X.X.; Zhang, L.N.; Guo, H.C. The regulation of FOXO1 and its role in disease progression. Life Sci. 2018, 193, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Graves, D.T.; Milovanova, T.N. Mucosal Immunity and the FOXO1 Transcription Factors. Front. Immunol. 2019, 10, 2530. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhou, Y.; Graves, D.T. FOXO transcription factors: Their clinical significance and regulation. Biomed Res. Int. 2014, 2014, 925350. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Q.; Sztukowska, M.; Ojo, A.; Scott, D.A.; Wang, H.; Lamont, R.J. FOXO responses to Porphyromonas gingivalis in epithelial cells. Cell. Microbiol. 2015, 17, 1605–1617. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nagashima, T.; Shigematsu, N.; Maruki, R.; Urano, Y.; Tanaka, H.; Shimaya, A.; Shimokawa, T.; Shibasaki, M. Discovery of novel forkhead box O1 inhibitors for treating type 2 diabetes: Improvement of fasting glycemia in diabetic db/db mice. Mol. Pharmacol. 2010, 78, 961–970. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gopal, K.; Al Batran, R.; Altamimi, T.R.; Greenwell, A.A.; Saed, C.T.; Tabatabaei Dakhili, S.A.; Dimaano, M.T.E.; Zhang, Y.; Eaton, F.; Sutendra, G.; et al. FoxO1 inhibition alleviates type 2 diabetes-related diastolic dysfunction by increasing myocardial pyruvate dehydrogenase activity. Cell Rep. 2021, 35, 108935. [Google Scholar] [CrossRef]
- Jeon, H.H.; Yu, Q.; Witek, L.; Lu, Y.; Zhang, T.; Stepanchenko, O.; Son, V.J.; Spencer, E.; Oshilaja, T.; Shin, M.K.; et al. Clinical application of a FOXO1 inhibitor improves connective tissue healing in a diabetic minipig model. Am. J. Transl. Res. 2021, 13, 781–791. [Google Scholar]
- Lee, Y.K.; Diaz, B.; Deroose, M.; Lee, S.X.; Belvedere, S.; Accili, D.; Leibel, R.L.; Lin, H.V. FOXO1 inhibition synergizes with FGF21 to normalize glucose control in diabetic mice. Mol. Metab. 2021, 49, 101187. [Google Scholar] [CrossRef]
- Cheng, T.; Lai, Y.T.; Wang, C.; Wang, Y.; Jiang, N.; Li, H.; Sun, H.; Jin, L. Bismuth drugs tackle Porphyromonas gingivalis and attune cytokine response in human cells. Metallomics 2019, 11, 1207–1218. [Google Scholar] [CrossRef]
- Wang, C.; Li, X.; Cheng, T.; Sun, H.; Jin, L. Eradication of Porphyromonas gingivalis persisters through colloidal bismuth subcitrate synergistically combined with metronidazole. Front. Microbiol. 2021, 12, 748121. [Google Scholar] [CrossRef]
- Van Acker, H.; Sass, A.; Bazzini, S.; De Roy, K.; Udine, C.; Messiaen, T.; Riccardi, G.; Boon, N.; Nelis, H.J.; Mahenthiralingam, E.; et al. Biofilm-grown Burkholderia cepacia complex cells survive antibiotic treatment by avoiding production of reactive oxygen species. PLoS ONE 2013, 8, e58943. [Google Scholar] [CrossRef] [Green Version]
- Helaine, S.; Kugelberg, E. Bacterial persisters: Formation, eradication, and experimental systems. Trends Microbiol. 2014, 22, 417–424. [Google Scholar] [CrossRef] [PubMed]
- Stathopoulou, P.G.; Benakanakere, M.R.; Galicia, J.C.; Kinane, D.F. The host cytokine response to Porphyromonas gingivalis is modified by gingipains. Oral Microbiol. Immunol. 2009, 24, 11–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stathopoulou, P.G.; Galicia, J.C.; Benakanakere, M.R.; Garcia, C.A.; Potempa, J.; Kinane, D.F. Porphyromonas gingivalis induce apoptosis in human gingival epithelial cells through a gingipain-dependent mechanism. BMC Microbiol. 2009, 9, 107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Groeger, S.; Jarzina, F.; Domann, E.; Meyle, J. Porphyromonas gingivalis activates NFkappaB and MAPK pathways in human oral epithelial cells. BMC Immunol. 2017, 18, 1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, J.; Roberts, J.S.; Atanasova, K.R.; Chowdhury, N.; Yilmaz, O. A novel kinase function of a nucleoside-diphosphate-kinase homologue in Porphyromonas gingivalis is critical in subversion of host cell apoptosis by targeting heat-shock protein 27. Cell. Microbiol. 2018, 20, e12825. [Google Scholar] [CrossRef]
- Miller, M.R.; Brunelli, J.P.; Wheeler, P.A.; Liu, S.; Rexroad, C.E., 3rd; Palti, Y.; Doe, C.Q.; Thorgaard, G.H. A conserved haplotype controls parallel adaptation in geographically distant salmonid populations. Mol. Ecol. 2012, 21, 237–249. [Google Scholar] [CrossRef] [Green Version]
- Kim, D.; Langmead, B.; Salzberg, S.L. HISAT: A fast spliced aligner with low memory requirements. Nat. Methods 2015, 12, 357–360. [Google Scholar] [CrossRef] [Green Version]
- Langmead, B.; Salzberg, S.L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 2012, 9, 357–359. [Google Scholar] [CrossRef] [Green Version]
- Anders, S.; Pyl, P.T.; Huber, W. HTSeq—A Python framework to work with high-throughput sequencing data. Bioinformatics 2015, 31, 166–169. [Google Scholar] [CrossRef]
- Li, X.; Cooper, N.G.F.; O’Toole, T.E.; Rouchka, E.C. Choice of library size normalization and statistical methods for differential gene expression analysis in balanced two-group comparisons for RNA-seq studies. BMC Genom. 2020, 21, 75. [Google Scholar] [CrossRef] [PubMed]
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B Methodol. 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Anders, S.; Huber, W. Differential expression analysis for sequence count data. Genome Biol. 2010, 11, R106. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| Gene Name | Primer Direction | Primer Sequence (5′ to 3′) |
|---|---|---|
| β-actin | Forward | TTGGCAATGAGCGGTT |
| Reverse | AGTTGAAGGTAGTTTCGTGGAT | |
| IL-1β | Forward | GCACGATGCACCTGTACGAT |
| Reverse | TGGAGAACACCACTTGTTGC | |
| IL-10 | Forward | TCAAGGCGCATGTGAACTCC |
| Reverse | GATGTCAAACTCACTCATGGCT | |
| TNF | Forward | GCTGCACTTTGGAGTGATCG |
| Reverse | GGGTTTGCTACAACATGGGC | |
| TGF-β2 | Forward | CAGCACACTCGATATGGACCA |
| Reverse | CCTCGGGCTCAGGATAGTCT | |
| IL-6 | forward | AATCATCACTGGTCTTTTGGAG |
| reverse | GCATTTGTGGTTGGGTCA | |
| IL-8 | forward | GACATACTCCAAACCTTTCCACC |
| reverse | AACTTCTCCACAACCCTCTGC | |
| CXCL5 | forward | AGCTGCGTTGCGTTTGTTTAC |
| reverse | TGGCGAACACTTGCAGATTAC | |
| FOXO1 | Forward | TTATGACCGAACAGGATGATCTTG |
| Reverse | TGTTGGTGATGAGAGAAGGTTGAG | |
| BCL2L11 | Forward | TAAGTTCTGAGTGTGACCGAGA |
| Reverse | GCTCTGTCTGTAGGGAGGTAGG | |
| BCL6 | Forward | GGAGTCGAGACATCTTGACTGA |
| Reverse | ATGAGGACCGTTTTATGGGCT | |
| S1PR1 | Forward | GCCTCTTCCTGCTAATCAGCG |
| Reverse | GCAGTACAGAATGACGATGGAG | |
| TRAIL | Forward | TGCGTGCTGATCGTGATCTTC |
| Reverse | GCTCGTTGGTAAAGTACACGTA | |
| KLF2 | Forward | TTCGGTCTCTTCGACGACG |
| Reverse | TGCGAACTCTTGGTGTAGGTC |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, C.; Li, X.; Cheng, T.; Wang, L.; Jin, L. RNA Sequencing Reveals the Upregulation of FOXO Signaling Pathway in Porphyromonas gingivalis Persister-Treated Human Gingival Epithelial Cells. Int. J. Mol. Sci. 2022, 23, 5728. https://doi.org/10.3390/ijms23105728
Wang C, Li X, Cheng T, Wang L, Jin L. RNA Sequencing Reveals the Upregulation of FOXO Signaling Pathway in Porphyromonas gingivalis Persister-Treated Human Gingival Epithelial Cells. International Journal of Molecular Sciences. 2022; 23(10):5728. https://doi.org/10.3390/ijms23105728
Chicago/Turabian StyleWang, Chuan, Xuan Li, Tianfan Cheng, Leilei Wang, and Lijian Jin. 2022. "RNA Sequencing Reveals the Upregulation of FOXO Signaling Pathway in Porphyromonas gingivalis Persister-Treated Human Gingival Epithelial Cells" International Journal of Molecular Sciences 23, no. 10: 5728. https://doi.org/10.3390/ijms23105728
APA StyleWang, C., Li, X., Cheng, T., Wang, L., & Jin, L. (2022). RNA Sequencing Reveals the Upregulation of FOXO Signaling Pathway in Porphyromonas gingivalis Persister-Treated Human Gingival Epithelial Cells. International Journal of Molecular Sciences, 23(10), 5728. https://doi.org/10.3390/ijms23105728






