The Role of SBI2/ALG12/EBS4 in the Regulation of Endoplasmic Reticulum-Associated Degradation (ERAD) Studied by a Null Allele
Abstract
1. Introduction
2. Results
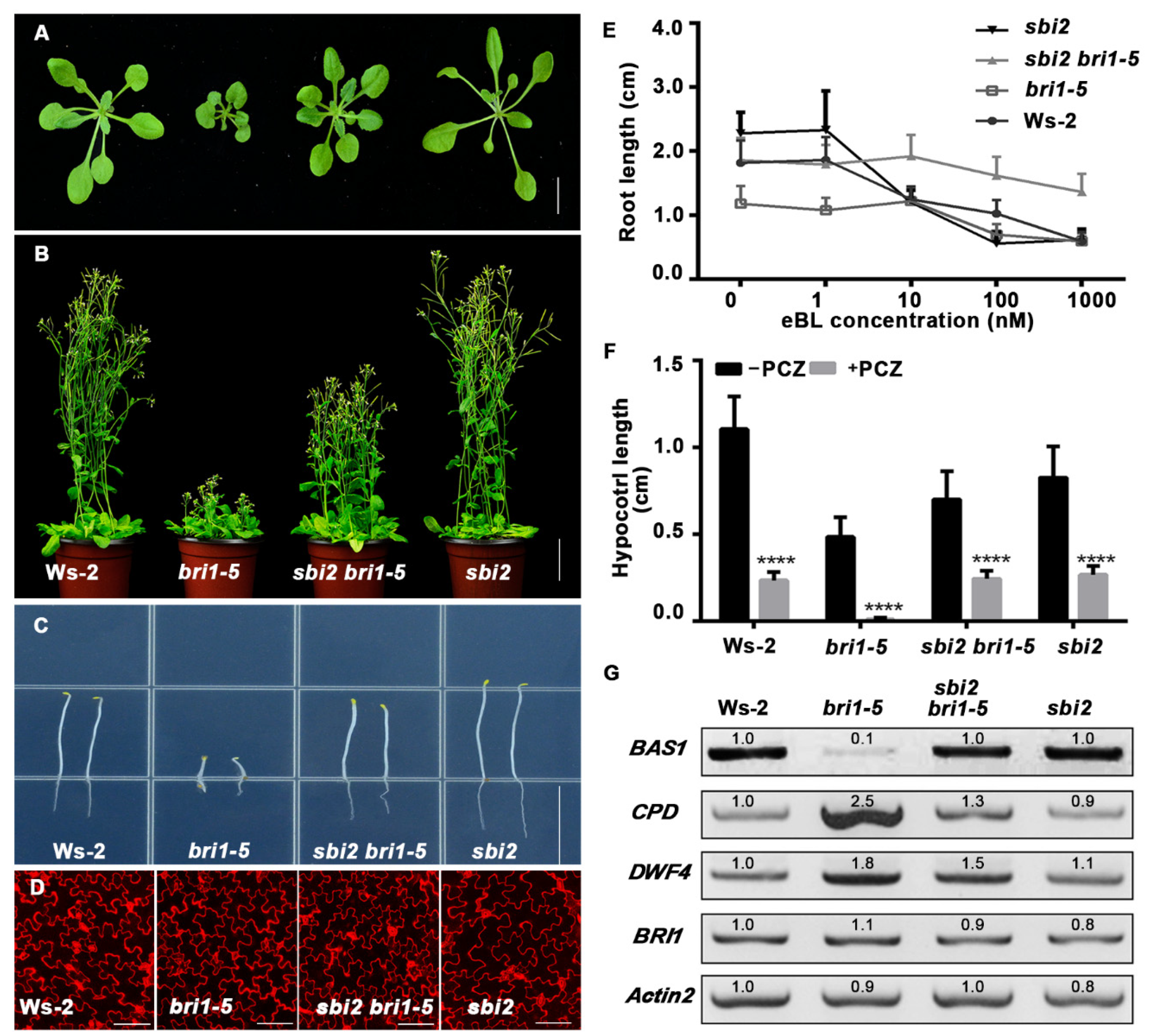
2.1. The sbi2 Mutation Partially Suppresses the Phenotypes of bri1-5, bri1-9, and bri1-235
2.2. A Single Base Substitution from G to A Results in a Null Allele of SBI2
2.3. The sbi2 Mutation Alters the Abundance and Localization of Glycoprotein
2.4. The sbi2 Mutation Cannot Regulate the ERAD of PM-Trapped BRI1
2.5. The sbi2 Mutation Involved in Plant Resistance to ER and Salt Stress
3. Discussion
4. Materials and Methods
4.1. Plant Materials and Growth Conditions
4.2. Plasmid Construction and Generation of Transgenic Plants
4.3. Protein Extraction and Endo H Treatment
4.4. Transcript Analysis by RT-PCR
4.5. Observation on Pavement Cell
4.6. Stress Treatment on ER and Salt Stress
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Helenius, A.; Aebi, M. Roles of N-linked glycans in the endoplasmic reticulum. Annu. Rev. Biochem. 2004, 73, 1019–1049. [Google Scholar] [CrossRef] [PubMed]
- Aebi, M.; Bernasconi, R.; Clerc, S.; Molinari, M. N-glycan structures: Recognition and processing in the ER. Trends Biochem. Sci. 2010, 35, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Hebert, D.N.; Lamriben, L.; Powers, E.T.; Kelly, J.W. The intrinsic and extrinsic effects of N-linked glycans on glycoproteostasis. Nat. Chem. Biol. 2014, 10, 902–910. [Google Scholar] [CrossRef] [PubMed]
- Helenius, A.; Aebi, M. Intracellular functions of N-linked glycans. Science 2001, 291, 2364–2369. [Google Scholar] [CrossRef] [PubMed]
- Moremen, K.W.; Tiemeyer, M.; Nairn, A.V. Vertebrate protein glycosylation: Diversity, synthesis and function. Nat. Rev. Mol. Cell Biol. 2012, 13, 448–462. [Google Scholar] [CrossRef]
- Strasser, R. Plant protein glycosylation. Glycobiology 2016, 26, 926–939. [Google Scholar] [CrossRef]
- Schoberer, J.; Shin, Y.J.; Vavra, U.; Veit, C.; Strasser, R. Analysis of Protein Glycosylation in the ER. Methods Mol. Biol. 2018, 1691, 205–222. [Google Scholar] [CrossRef]
- Hong, Z.; Jin, H.; Fitchette, A.C.; Xia, Y.; Monk, A.M.; Faye, L.; Li, J. Mutations of an alpha1,6 mannosyltransferase inhibit endoplasmic reticulum-associated degradation of defective brassinosteroid receptors in Arabidopsis. Plant Cell 2009, 21, 3792–3802. [Google Scholar] [CrossRef]
- Aebi, M. N-linked protein glycosylation in the ER. Biochim. Biophys. Acta 2013, 1833, 2430–2437. [Google Scholar] [CrossRef]
- Li, S.T.; Lu, T.T.; Xu, X.X.; Ding, Y.; Li, Z.; Kitajima, T.; Dean, N.; Wang, N.; Gao, X.D. Reconstitution of the lipid-linked oligosaccharide pathway for assembly of high-mannose N-glycans. Nat. Commun. 2019, 10, 1813. [Google Scholar] [CrossRef]
- Strasser, R. Protein Quality Control in the Endoplasmic Reticulum of Plants. Annu. Rev. Plant Biol. 2018, 69, 147–172. [Google Scholar] [CrossRef] [PubMed]
- Henquet, M.; Lehle, L.; Schreuder, M.; Rouwendal, G.; Molthoff, J.; Helsper, J.; van der Krol, S.; Bosch, D. Identification of the gene encoding the alpha1,3-mannosyltransferase (ALG3) in Arabidopsis and characterization of downstream n-glycan processing. Plant Cell 2008, 20, 1652–1664. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kajiura, H.; Seki, T.; Fujiyama, K. Arabidopsis thaliana ALG3 mutant synthesizes immature oligosaccharides in the ER and accumulates unique N-glycans. Glycobiology 2010, 20, 736–751. [Google Scholar] [CrossRef] [PubMed]
- Hong, Z.; Kajiura, H.; Su, W.; Jin, H.; Kimura, A.; Fujiyama, K.; Li, J. Evolutionarily conserved glycan signal to degrade aberrant brassinosteroid receptors in Arabidopsis. Proc. Natl. Acad. Sci. USA 2012, 109, 11437–11442. [Google Scholar] [CrossRef] [PubMed]
- Trempel, F.; Eschen-Lippold, L.; Bauer, N.; Ranf, S.; Westphal, L.; Scheel, D.; Lee, J. A mutation in Asparagine-Linked Glycosylation 12 (ALG12) leads to receptor misglycosylation and attenuated responses to multiple microbial elicitors. FEBS Lett. 2020, 594, 2440–2451. [Google Scholar] [CrossRef]
- Farid, A.; Pabst, M.; Schoberer, J.; Altmann, F.; Glossl, J.; Strasser, R. Arabidopsis thaliana alpha1,2-glucosyltransferase (ALG10) is required for efficient N-glycosylation and leaf growth. Plant J. 2011, 68, 314–325. [Google Scholar] [CrossRef]
- Zhang, M.; Henquet, M.; Chen, Z.; Zhang, H.; Zhang, Y.; Ren, X.; van der Krol, S.; Gonneau, M.; Bosch, D.; Gong, Z. LEW3, encoding a putative alpha-1,2-mannosyltransferase (ALG11) in N-linked glycoprotein, plays vital roles in cell-wall biosynthesis and the abiotic stress response in Arabidopsis thaliana. Plant J. 2009, 60, 983–999. [Google Scholar] [CrossRef]
- Clouse, S.D.; Langford, M.; McMorris, T.C. A brassinosteroid-insensitive mutant in Arabidopsis thaliana exhibits multiple defects in growth and development. Plant Physiol. 1996, 111, 671–678. [Google Scholar] [CrossRef]
- Li, J.; Chory, J. A Putative Leucine-Rich Repeat Receptor Kinase Involved in Brassinosteroid Signal Transduction. Cell 1997, 90, 929–938. [Google Scholar] [CrossRef]
- Römisch, K. Endoplasmic reticulum-associated degradation. Annu. Rev. Cell. Dev. Biol. 2005, 21, 435–456. [Google Scholar] [CrossRef]
- Vembar, S.S.; Brodsky, J.L. One step at a time: Endoplasmic reticulum-associated degradation-b. Nat. Rev. Mol. Cell Biol. 2008, 9, 944–957. [Google Scholar] [CrossRef] [PubMed]
- Hong, Z.; Jin, H.; Tzfira, T.; Li, J. Multiple mechanism-mediated retention of a defective brassinosteroid receptor in the endoplasmic reticulum of Arabidopsis. Plant Cell 2008, 20, 3418–3429. [Google Scholar] [CrossRef] [PubMed]
- Hou, Q.; Saima, S.; Ren, H.; Ali, K.; Bai, C.; Wu, G.; Li, G. Less Conserved LRRs Is Important for BRI1 Folding. Front. Plant Sci. 2019, 10, 634. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; Yan, Z.; Nam, K.H.; Li, J. Allele-specific suppression of a defective brassinosteroid receptor reveals a essential role of UGGT in ER quality control. Mol. Cell 2007, 26, 821–830. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; Hong, Z.; Su, W.; Li, J. A plant-specific calreticulin is a key retention factor for a defective brassinosteroid receptor in the endoplasmic reticulum. Proc. Natl. Acad. Sci. USA 2009, 106, 13612–13617. [Google Scholar] [CrossRef]
- Su, W.; Liu, Y.; Xia, Y.; Hong, Z.; Li, J. Conserved endoplasmic reticulum-associated degradation system to eliminate mutated receptor-like kinases in Arabidopsis. Proc. Natl. Acad. Sci. USA 2011, 108, 870–875. [Google Scholar] [CrossRef]
- Su, W.; Liu, Y.; Xia, Y.; Hong, Z.; Li, J. The Arabidopsis homolog of the mammalian OS-9 protein plays a key role in the endoplasmic reticulum-associated degradation of misfolded receptor-like kinases. Mol. Plant 2012, 5, 929–940. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, C.; Wang, D.; Su, W.; Liu, L.; Wang, M.; Li, J. EBS7 is a plant-specific component of a highly conserved endoplasmic reticulum-associated degradation system in Arabidopsis. Proc. Natl. Acad. Sci. USA 2015, 112, 12205–12210. [Google Scholar] [CrossRef]
- Wu, G.; Wang, X.; Li, X.; Kamiya, Y.; Otegui, M.S.; Chory, J. Methylation of a Phosphatase Specifies Dephosphorylation and Degradation of Activated Brassinosteroid Receptors. Sci. Signal. 2011, 4, ra29. [Google Scholar] [CrossRef]
- Sun, X.; Guo, C.; Ali, K.; Zheng, Q.; Wei, Q.; Zhu, Y.; Wang, L.; Li, G.; Li, W.; Zheng, B.; et al. Non-redundant Function of MNS5: A Class I α-1, 2 Mannosidase, in the Regulation of Endoplasmic Reticulum-Associated Degradation of Misfolded Glycoproteins. Front. Plant Sci. 2022, 13, 873688. [Google Scholar] [CrossRef]
- Huttner, S.; Veit, C.; Vavra, U.; Schoberer, J.; Liebminger, E.; Maresch, D.; Grass, J.; Altmann, F.; Mach, L.; Strasser, R. Arabidopsis Class I alpha-Mannosidases MNS4 and MNS5 Are Involved in Endoplasmic Reticulum-Associated Degradation of Misfolded Glycoproteins. Plant Cell 2014, 26, 1712–1728. [Google Scholar] [CrossRef] [PubMed]
- Aklilu, E. Review on forward and reverse genetics in plant breeding. All Life 2021, 14, 127–135. [Google Scholar] [CrossRef]
- Chen, J.G.; Ullah, H.; Young, J.C.; Sussman, M.R.; Jones, A.M. ABP1 is required for organized cell elongation and division in Arabidopsis embryogenesis. Genes Dev. 2001, 15, 902–911. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.; Zhang, Y.; Zhang, D.; Chen, J.; Gao, X.; Estelle, M.; Zhao, Y. Embryonic lethality of Arabidopsis abp1-1 is caused by deletion of the adjacent BSM gene. Nat. Plants 2015, 1, 15183. [Google Scholar] [CrossRef]
- Gao, Y.; Zhang, Y.; Zhang, D.; Dai, X.; Estelle, M.; Zhao, Y. Auxin binding protein 1 (ABP1) is not required for either auxin signaling or Arabidopsis development. Proc. Natl. Acad. Sci. USA 2015, 112, 2275–2280. [Google Scholar] [CrossRef]
- Braun, N.; Wyrzykowska, J.; Muller, P.; David, K.; Couch, D.; Perrot-Rechenmann, C.; Fleming, A.J. Conditional repression of AUXIN BINDING PROTEIN1 reveals that it coordinates cell division and cell expansion during postembryonic shoot development in Arabidopsis and tobacco. Plant Cell 2008, 20, 2746–2762. [Google Scholar] [CrossRef]
- Paque, S.; Mouille, G.; Grandont, L.; Alabadi, D.; Gaertner, C.; Goyallon, A.; Muller, P.; Primard-Brisset, C.; Sormani, R.; Blazquez, M.A.; et al. AUXIN BINDING PROTEIN1 links cell wall remodeling, auxin signaling, and cell expansion in arabidopsis. Plant Cell 2014, 26, 280–295. [Google Scholar] [CrossRef]
- Chen, X.; Grandont, L.; Li, H.; Hauschild, R.; Paque, S.; Abuzeineh, A.; Rakusova, H.; Benkova, E.; Perrot-Rechenmann, C.; Friml, J. Inhibition of cell expansion by rapid ABP1-mediated auxin effect on microtubules. Nature 2014, 516, 90–93. [Google Scholar] [CrossRef]
- Liu, C.M. Auxin Binding Protein 1 (ABP1): A matter of fact. J. Integr. Plant Biol. 2015, 57, 234–235. [Google Scholar] [CrossRef]
- Lin, L.; Zhang, C.; Chen, Y.; Wang, Y.; Wang, D.; Liu, X.; Wang, M.; Mao, J.; Zhang, J.; Xing, W.; et al. PAWH1 and PAWH2 are plant-specific components of an Arabidopsis endoplasmic reticulum-associated degradation complex. Nat. Commun. 2019, 10, 3492. [Google Scholar] [CrossRef]
- El-Brolosy, M.A.; Stainier, D.Y.R. Genetic compensation: A phenomenon in search of mechanisms. PLoS Genet. 2017, 13, e1006780. [Google Scholar] [CrossRef] [PubMed]
- El-Brolosy, M.A.; Kontarakis, Z.; Rossi, A.; Kuenne, C.; Gunther, S.; Fukuda, N.; Kikhi, K.; Boezio, G.L.M.; Takacs, C.M.; Lai, S.L.; et al. Genetic compensation triggered by mutant mRNA degradation. Nature 2019, 568, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Zhu, P.; Shi, H.; Guo, L.; Zhang, Q.; Chen, Y.; Chen, S.; Zhang, Z.; Peng, J.; Chen, J. PTC-bearing mRNA elicits a genetic compensation response via Upf3a and COMPASS components. Nature 2019, 568, 259–263. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, M.F. Genetic paradox explained by nonsense. Nature 2019, 568, 179–180. [Google Scholar] [CrossRef]
- Li, J.; Nam, K.H. Regulation of brassinosteroid signaling by a GSK3/SHAGGY-like kinase. Science 2002, 295, 1299–1301. [Google Scholar] [CrossRef]
- Howell, S.H. Endoplasmic reticulum stress responses in plants. Annu. Rev. Plant Biol. 2013, 64, 477–499. [Google Scholar] [CrossRef]
- Tanaka, K.; Asami, T.; Yoshida, S.; Nakamura, Y.; Matsuo, T.; Okamoto, S. Brassinosteroid homeostasis in Arabidopsis is ensured by feedback expressions of multiple genes involved in its metabolism. Plant Physiol. 2005, 138, 1117–1125. [Google Scholar] [CrossRef]
- Zhang, C.; Halsey, L.E.; Szymanski, D.B. The development and geometry of shape change in Arabidopsis thaliana cotyledon pavement cells. BMC Plant Biol. 2011, 11, 27. [Google Scholar] [CrossRef]







Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, X.; Zhao, D.; Ali, K.; Zhu, Y.; Wu, G.; Li, G. The Role of SBI2/ALG12/EBS4 in the Regulation of Endoplasmic Reticulum-Associated Degradation (ERAD) Studied by a Null Allele. Int. J. Mol. Sci. 2022, 23, 5811. https://doi.org/10.3390/ijms23105811
Sun X, Zhao D, Ali K, Zhu Y, Wu G, Li G. The Role of SBI2/ALG12/EBS4 in the Regulation of Endoplasmic Reticulum-Associated Degradation (ERAD) Studied by a Null Allele. International Journal of Molecular Sciences. 2022; 23(10):5811. https://doi.org/10.3390/ijms23105811
Chicago/Turabian StyleSun, Xiaoxia, Di Zhao, Khawar Ali, Yumeng Zhu, Guang Wu, and Guishuang Li. 2022. "The Role of SBI2/ALG12/EBS4 in the Regulation of Endoplasmic Reticulum-Associated Degradation (ERAD) Studied by a Null Allele" International Journal of Molecular Sciences 23, no. 10: 5811. https://doi.org/10.3390/ijms23105811
APA StyleSun, X., Zhao, D., Ali, K., Zhu, Y., Wu, G., & Li, G. (2022). The Role of SBI2/ALG12/EBS4 in the Regulation of Endoplasmic Reticulum-Associated Degradation (ERAD) Studied by a Null Allele. International Journal of Molecular Sciences, 23(10), 5811. https://doi.org/10.3390/ijms23105811





