Review about Powerful Combinations of Advanced and Hyphenated Sample Introduction Techniques with Inductively Coupled Plasma-Mass Spectrometry (ICP-MS) for Elucidating Trace Element Species in Pathologic Conditions on a Molecular Level
Abstract
:1. Introduction
2. Methods
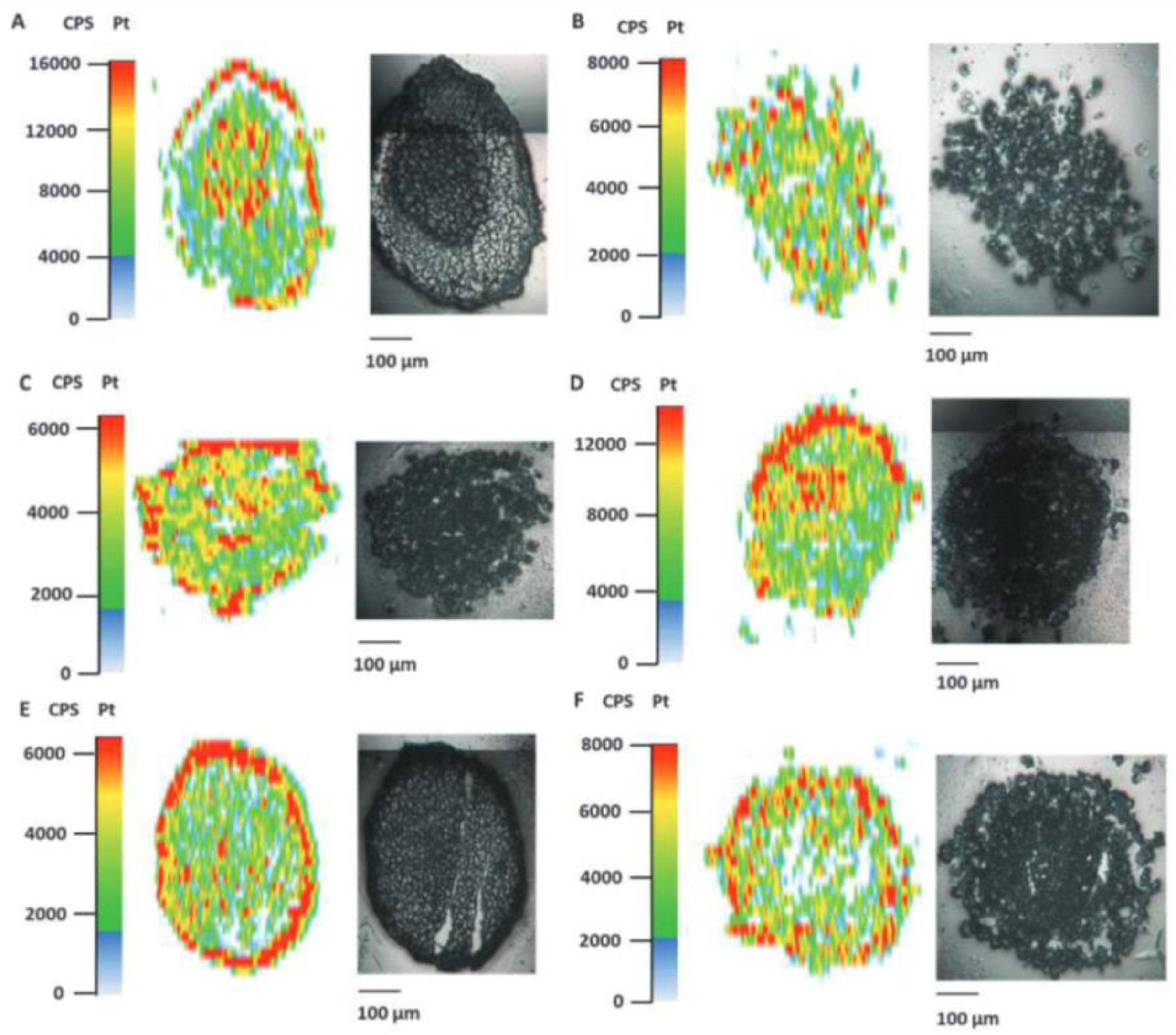
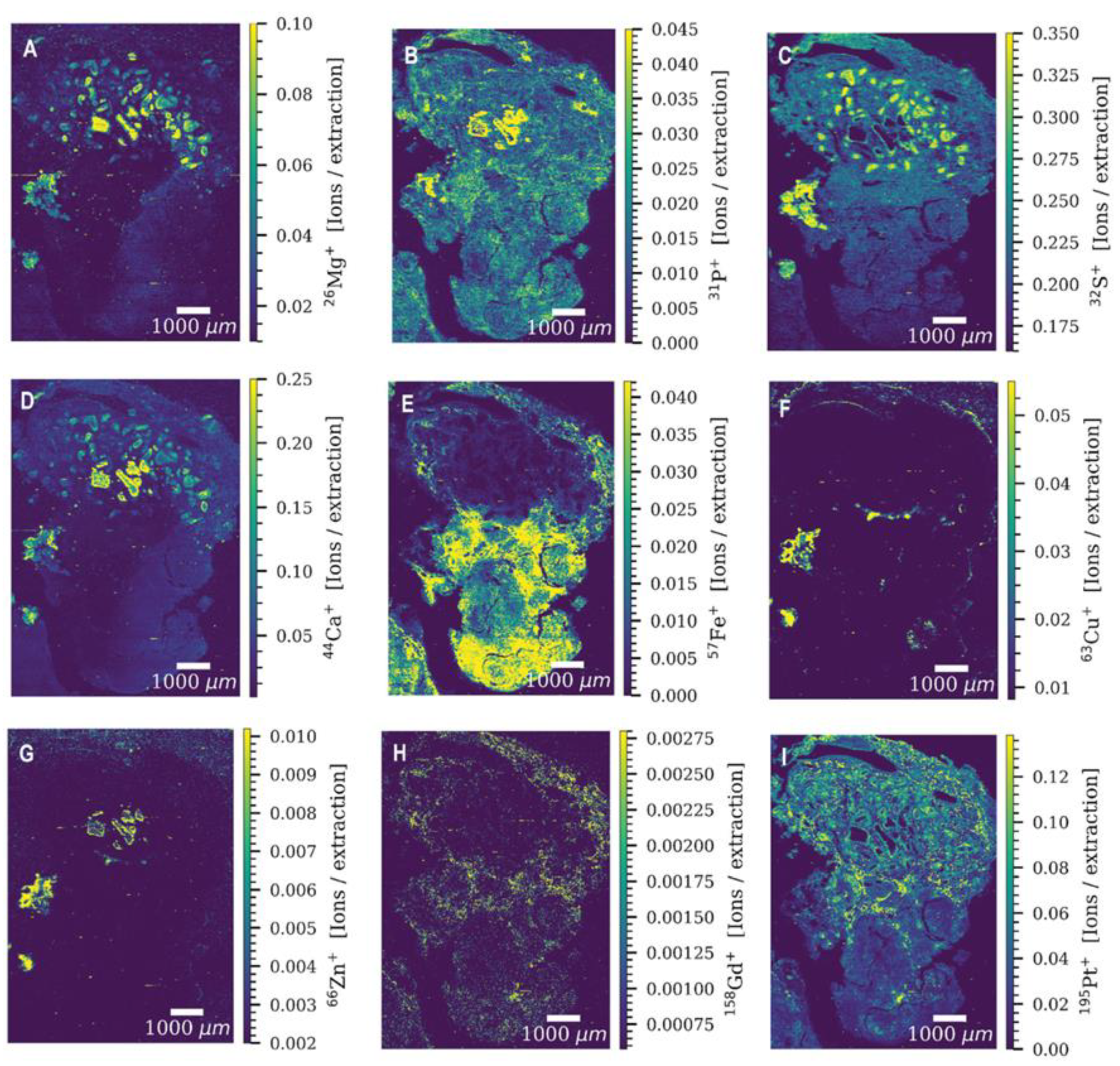
3. Laser Ablation Inductively Coupled Plasma Mass Spectrometry (LA-ICP-MS)—Spatial Element Mapping
4. Elemental Tagging in Inorganic Mass Spectrometric Bioanalysis
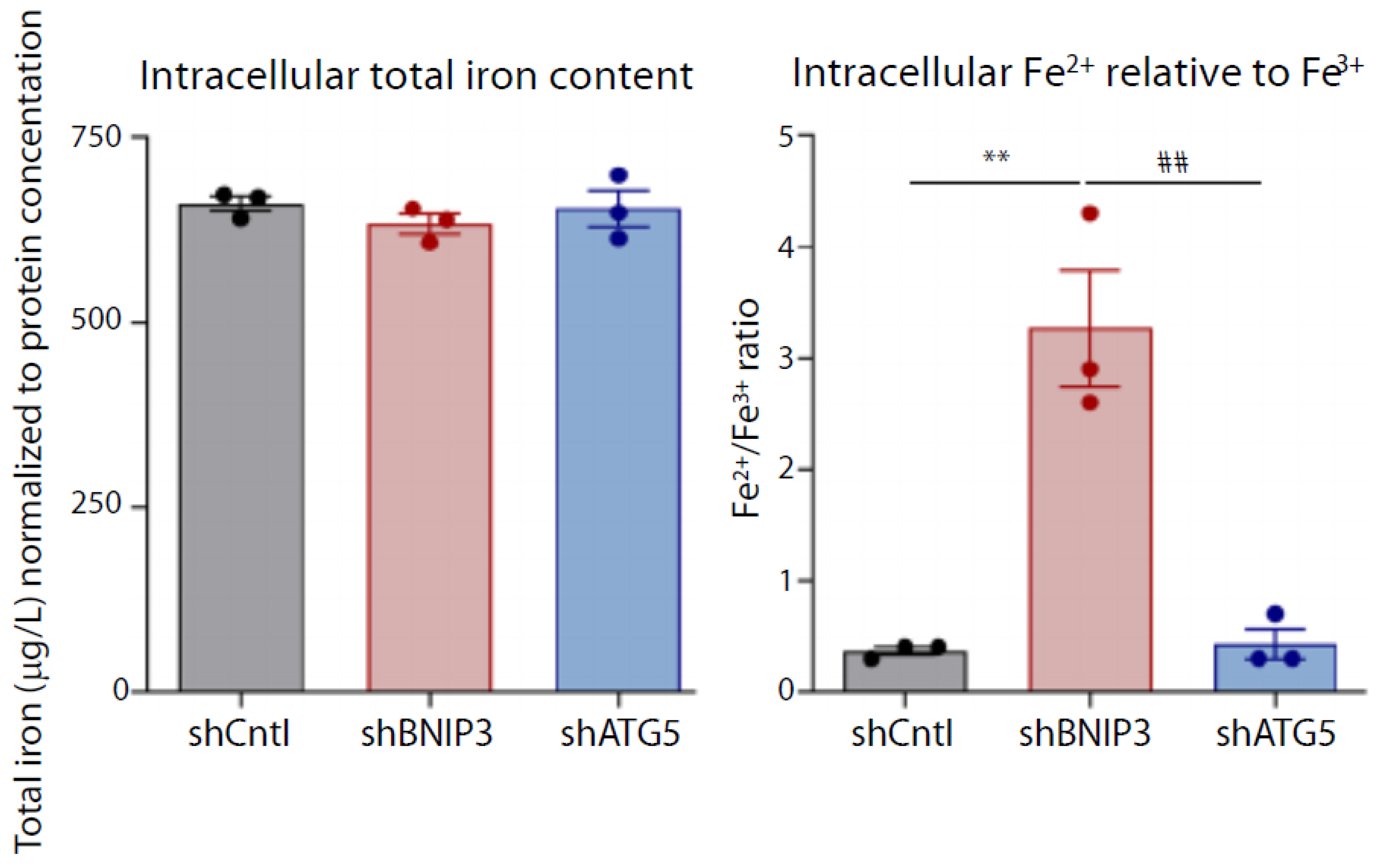
5. Iron Redox Speciation
6. Copper Speciation in the Frame of Cu-Related Diseases
7. Single Cell ICP-MS (SC-ICP-MS)
8. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cannas, D.; Loi, E.; Serra, M.; Firinu, D.; Valera, P.; Zavattari, P. Relevance of Essential Trace Elements in Nutrition and Drinking Water for Human Health and Autoimmune Disease Risk. Nutrients 2020, 12, 2074. [Google Scholar] [CrossRef] [PubMed]
- Aggett, P. Essential and Toxic Trace Elements in Human Health and Disease. Arch. Dis. Child. 1989, 64, 436. [Google Scholar] [CrossRef] [Green Version]
- Theiner, S.; Egger, A.; Keppler, B.; Heffeter, P.; Kornauth, C.; Theiner, S.; Heffeter, P.; Keppler, B. Bioimaging and quantification of metal-based anticancer drugs using LA-ICP-MS. J. Biol. Inorg. Chem. 2014, 19, S681. [Google Scholar]
- Timerbaev, A.R. Application of ICP-MS to the development of metal-based drugs and diagnostic agents: Where do we stand? J. Anal. At. Specetrom. 2021, 36, 254–266. [Google Scholar] [CrossRef]
- Jarosz, M.; Keppler, B.K.; Timerbaev, A.R. Current and emerging mass spectrometry methods for the preclinical development of metal-based drugs: A critical appraisal. Anal. Bioanal. Chem. 2022, 414, 95–102. [Google Scholar] [CrossRef]
- Linert, W.; Kozlowski, H. Metal Ions in Neurological Systems; Linert, W., Kozlowski, H., Eds.; Springer: Vienna, Austria, 2012. [Google Scholar]
- Kozlowski, H.; Luczkowski, M.; Remelli, M.; Valensin, D. Copper, zinc and iron in neurodegenerative diseases (Alzheimer’s, Parkinson’s and prion diseases). Coordin. Chem. Rev. 2012, 256, 2129–2141. [Google Scholar] [CrossRef]
- Leal, S.S.; Botelho, H.M.; Gomes, C.M. Metal ions as modulators of protein conformation and misfolding in neurodegeneration. Coordin. Chem. Rev. 2012, 256, 2253–2270. [Google Scholar] [CrossRef]
- Ashraf, A.; Clark, M.; So, P.W. The Aging of Iron Man. Front. Aging Neurosci. 2018, 10, 65. [Google Scholar] [CrossRef] [Green Version]
- Gaschler, M.M.; Stockwell, B.R. Lipid peroxidation in cell death. Biochem. Biophys. Res. Commun. 2017, 482, 419–425. [Google Scholar] [CrossRef]
- Schreiber-Brynzak, E.; Pichler, V.; Heffeter, P.; Hanson, B.; Theiner, S.; Lichtscheidl-Schultz, I.; Kornauth, C.; Bamonti, L.; Dhery, V.; Groza, D.; et al. Behavior of platinum(IV) complexes in models of tumor hypoxia: Cytotoxicity, compound distribution and accumulation. Metallomics 2016, 8, 422–433. [Google Scholar] [CrossRef] [Green Version]
- Profrock, D.; Prange, A. Inductively Coupled Plasma-Mass Spectrometry (ICP-MS) for Quantitative Analysis in Environmental and Life Sciences: A Review of Challenges, Solutions, and Trends. Appl. Spectrosc. 2012, 66, 843–868. [Google Scholar] [CrossRef] [PubMed]
- Michalke, B.; Berthele, A.; Venkataramani, V. Simultaneous Quantification and Speciation of Trace Metals in Paired Serum and CSF Samples by Size Exclusion Chromatography-Inductively Coupled Plasma-Dynamic Reaction Cell-Mass Spectrometry (SEC-DRC-ICP-MS). Int. J. Mol. Sci. 2021, 22, 8892. [Google Scholar] [CrossRef] [PubMed]
- Szpunar, J. Bio-inorganic speciation analysis by hyphenated techniques. Analyst 2000, 125, 963–988. [Google Scholar] [CrossRef] [PubMed]
- Szpunar, J.; Lobinski, R. Hyphenated Techniques in Speciation Analysis; The Royal Society of Chemistry: Cambridge, UK, 2003. [Google Scholar]
- Violi, F.; Solovyev, N.; Vinceti, M.; Mandrioli, J.; Lucio, M.; Michalke, B. The study of levels from redox-active elements in cerebrospinal fluid of amyotrophic lateral sclerosis patients carrying disease-related gene mutations shows potential copper dyshomeostasis. Metallomics 2020, 12, 668–681. [Google Scholar] [CrossRef]
- Solovyev, N.; Vinceti, M.; Grill, P.; Mandrioli, J.; Michalke, B. Redox speciation of iron, manganese, and copper in cerebrospinal fluid by strong cation exchange chromatography-sector field inductively coupled plasma mass spectrometry. Anal. Chim. Acta 2017, 973, 25–33. [Google Scholar] [CrossRef]
- Solovyev, N.; Ala, A.; Schilsky, M.; Mills, C.; Willis, K.; Harrington, C.F. Biomedical copper speciation in relation to Wilson’s disease using strong anion exchange chromatography coupled to triple quadrupole inductively coupled plasma mass spectrometry. Anal. Chim. Acta 2020, 1098, 27–36. [Google Scholar] [CrossRef]
- Burger, M.; Gundlach-Graham, A.; Allner, S.; Schwarz, G.; Wang, H.A.O.; Gyr, L.; Burgener, S.; Hattendorf, B.; Grolimund, D.; Gunther, D. High-Speed, High-Resolution, Multielemental LA-ICP-TOFMS Imaging: Part II. Critical Evaluation of Quantitative Three-Dimensional Imaging of Major, Minor, and Trace Elements in Geological Samples. Anal. Chem. 2015, 87, 8259–8267. [Google Scholar] [CrossRef]
- Moreno-Gordaliza, E.; Giesen, C.; Lazaro, A.; Esteban-Fernandez, D.; Humanes, B.; Canas, B.; Panne, U.; Tejedor, A.; Jakubowski, N.; Gomez-Gomez, M.M. Elemental Bioimaging in Kidney by LA-ICP-MS as a Tool to Study Nephrotoxicity and Renal Protective Strategies in Cisplatin Therapies. Anal. Chem. 2011, 83, 7933–7940. [Google Scholar] [CrossRef]
- Theiner, S.; Schreiber-Brynzak, E.; Jakupec, M.A.; Galanski, M.; Koellensperger, G.; Keppler, B.K. LA-ICP-MS imaging in multicellular tumor spheroids—A novel tool in the preclinical development of metal-based anticancer drugs. Metallomics 2016, 8, 398–402. [Google Scholar] [CrossRef]
- Theiner, S.; Schweikert, A.; Haberler, C.; Peyrl, A.; Koellensperger, G. Laser ablation-ICP-TOFMS imaging of germ cell tumors of patients undergoing platinum-based chemotherapy. Metallomics 2020, 12, 1246–1252. [Google Scholar] [CrossRef]
- Neumann, B.; Hosl, S.; Schwab, K.; Theuring, F.; Jakubowski, N. Multiplex LA-ICP-MS bio-imaging of brain tissue of a parkinsonian mouse model stained with metal-coded affinity-tagged antibodies and coated with indium-spiked commercial inks as internal standards. J. Neurosci. Methods 2020, 334, 108591. [Google Scholar] [CrossRef] [PubMed]
- Baranov, V.I.; Quinn, Z.; Bandura, D.R.; Tanner, S.D. A sensitive and quantitative element-tagged immunoassay with ICPMS detection. Anal. Chem. 2002, 74, 1629–1636. [Google Scholar] [CrossRef] [PubMed]
- Giesen, C.; Mairinger, T.; Khoury, L.; Waentig, L.; Jakubowski, N.; Panne, U. Multiplexed immunohistochemical detection of tumor markers in breast cancer tissue using laser ablation inductively coupled plasma mass spectrometry. Anal. Chem. 2011, 83, 8177–8183. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Alonso, M.; Fernandez, B.; Alvarez, L.; Gonzalez-Iglesias, H.; Traub, H.; Jakubowski, N.; Pereiro, R. Bioimaging of metallothioneins in ocular tissue sections by laser ablation-ICP-MS using bioconjugated gold nanoclusters as specific tags. Mikrochim. Acta 2017, 185, 64. [Google Scholar] [CrossRef] [Green Version]
- Bettmer, J.; Jakubowski, N.; Prange, A. Elemental tagging in inorganic mass spectrometric bioanalysis. Anal. Bioanal. Chem. 2006, 386, 7–11. [Google Scholar] [CrossRef]
- Sanz-Medel, A.; Montes-Bayon, M.; Bettmer, J.; Fernandez-Sanchez, M.L.; Encinar, J.R. ICP-MS for absolute quantification of proteins for heteroatom-tagged, targeted proteomics. TrAC Trends Anal. Chem. 2012, 40, 52–63. [Google Scholar] [CrossRef]
- Weekley, C.M.; Harris, H.H. Which form is that? The importance of selenium speciation and metabolism in the prevention and treatment of disease. Chem. Soc. Rev. 2013, 42, 8870–8894. [Google Scholar] [CrossRef]
- Michalke, B.; Willkommen, D.; Drobyshev, E.; Solovyev, N. The importance of speciation analysis in neurodegeneration research. TrAC Trends Anal. Chem. 2018, 104, 160–170. [Google Scholar] [CrossRef]
- Vinceti, M.; Michalke, B.; Malagoli, C.; Eichmuller, M.; Filippini, T.; Tondelli, M.; Bargellini, A.; Vinceti, G.; Zamboni, G.; Chiari, A. Selenium and selenium species in the etiology of Alzheimer’s dementia: The potential for bias of the case-control study design. J. Trace Elem. Med. Biol. 2019, 53, 154–162. [Google Scholar] [CrossRef]
- Maass, F.; Michalke, B.; Willkommen, D.; Schulte, C.; Tonges, L.; Boerger, M.; Zerr, I.; Bahr, M.; Lingor, P. Selenium speciation analysis in the cerebrospinal fluid of patients with Parkinson’s disease. J. Trace Elem. Med. Biol. 2020, 57, 110–115. [Google Scholar] [CrossRef]
- Michalke, B.; Berthele, A. Contribution to selenium speciation in cerebrospinal fluid samples. J. Anal. At. Spectrom. 2011, 26, 165–170. [Google Scholar] [CrossRef]
- Tondo, M.; Moreno, J.; Casado, M.; Brandi, N.; Sierra, C.; Vilaseca, M.A.; Ormazabal, A.; Artuch, R. Selenium concentration in cerebrospinal fluid samples from a paediatric population. Neurochem. Res. 2010, 35, 1290–1293. [Google Scholar] [CrossRef] [PubMed]
- Solovyev, N.; Berthele, A.; Michalke, B. Selenium speciation in paired serum and cerebrospinal fluid samples. Anal. Bioanal. Chem. 2013, 405, 1875–1884. [Google Scholar] [CrossRef] [PubMed]
- Mandrioli, J.; Michalke, B.; Solovyev, N.; Grill, P.; Violi, F.; Lunetta, C.; Conte, A.; Sansone, V.A.; Sabatelli, M.; Vinceti, M. Elevated Levels of Selenium Species in Cerebrospinal Fluid of Amyotrophic Lateral Sclerosis Patients with Disease-Associated Gene Mutations. Neurodegener. Dis. 2017, 17, 171–180. [Google Scholar] [CrossRef]
- Solovyev, N. Selenoprotein P and its potential role in Alzheimer’s disease. Hormones 2020, 19, 73–79. [Google Scholar] [CrossRef]
- Solovyev, N.; Drobyshev, E.; Blume, B.; Michalke, B. Selenium at the Neural Barriers: A Review. Front. Neurosci. 2021, 15, 630016. [Google Scholar] [CrossRef]
- Sargent, M.; Goenaga-Infante, H.; Inagaki, K.; Ma, L.; Meija, J.; Pramann, A.; Rienitz, O.; Sturgeon, R.; Vogl, J.; Wang, J.; et al. The role of ICP-MS in inorganic chemical metrology. Metrologia 2019, 56, 034005. [Google Scholar] [CrossRef]
- Guo, Q.H.; Tong, Q.H.; Lu, N.; Cao, H.; Yang, L.; Zhang, Y.Q. Proteomic Analysis of the Hippocampus in Mouse Models of Trigeminal Neuralgia and Inescapable Shock-Induced Depression. Neurosci. Bull. 2018, 34, 74–84. [Google Scholar] [CrossRef]
- Vinceti, M.; Grill, P.; Malagoli, C.; Filippini, T.; Storani, S.; Malavolti, M.; Michalke, B. Selenium speciation in human serum and its implications for epidemiologic research: A cross-sectional study. J. Trace Elem. Med. Biol. 2015, 31, 1–10. [Google Scholar] [CrossRef]
- Heitland, P.; Koster, H.D. Fast determination of arsenic species and total arsenic in urine by HPLC-ICP-MS: Concentration ranges for unexposed german inhabitants and clinical case studies. J. Anal. Toxicol. 2008, 32, 308–314. [Google Scholar] [CrossRef] [Green Version]
- Lucio, M.; Barbir, R.; Lovrencic, M.V.; Varzic, S.C.; Ljubic, S.; Duvnjak, L.S.; Seric, V.; Milic, M.; Lovakovic, B.T.; Krivohlavek, A.; et al. Association between arsenic exposure and biomarkers of type 2 diabetes mellitus in a Croatian population: A comparative observational pilot study. Sci. Total Environ. 2020, 720, 137575. [Google Scholar] [CrossRef] [PubMed]
- Pei, K.L.; Gailer, J. Probing the interaction of arsenobetaine with blood plasma constituents in vitro: An SEC-ICP-AES study. Metallomics 2009, 1, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Siethoff, C.; Feldmann, I.; Jakubowski, N.; Linscheid, M. Quantitative determination of DNA adducts using liquid chromatography electrospray ionization mass spectrometry and liquid chromatography high-resolution inductively coupled plasma mass spectrometry. J. Mass Spectrom. 1999, 34, 421–426. [Google Scholar] [CrossRef]
- Edler, M.; Jakubowski, N.; Linscheid, M. Styrene oxide DNA adducts: Quantitative determination using P-31 monitoring. Anal. Bioanal. Chem. 2005, 381, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Edler, M.; Jakubowski, N.; Linscheid, M. Quantitative determination of melphalan DNA adducts using HPLC-inductively coupled mass spectrometry. J. Mass Spectrom. 2006, 41, 507–516. [Google Scholar] [CrossRef] [PubMed]
- Wind, M.; Wegener, A.; Eisenmenger, A.; Kellner, R.; Lehmann, W.D. Sulfur as the key element for quantitative protein analysis by capillary liquid chromatography coupled to element mass spectrometry. Angew. Chem. Int. Ed. 2003, 42, 3425–3427. [Google Scholar] [CrossRef] [PubMed]
- Wind, M.; Lehmann, W.D. Element and molecular mass spectrometry—An emerging analytical dream team in the life sciences. J. Anal. At. Spectrom. 2004, 19, 20–25. [Google Scholar] [CrossRef]
- Schaumloffel, D.; Prange, A.; Marx, G.; Heumann, K.G.; Bratter, P. Characterization and quantification of metallothionein isoforms by capillary electrophoresis-inductively coupled plasma-isotope-dilution mass spectrometry. Anal. Bioanal. Chem. 2002, 372, 155–163. [Google Scholar] [CrossRef]
- Huang, Z.L.; Li, Z.Y.; Jiang, M.; Liu, R.; Lv, Y. Homogeneous Multiplex Immunoassay for One-Step Pancreatic Cancer Biomarker Evaluation. Anal. Chem. 2020, 92, 16105–16112. [Google Scholar] [CrossRef]
- Li, X.T.; Chen, B.B.; He, M.; Xiao, G.Y.; Hu, B. Gold nanoparticle labeling with tyramide signal amplification for highly sensitive detection of alpha fetoprotein in human serum by ICP-MS. Talanta 2018, 176, 40–46. [Google Scholar] [CrossRef]
- Chen, X.; Song, H.; Li, Z.; Liu, R.; Lv, Y. Lanthanide Nanoprobes for the Multiplex Evaluation of Breast Cancer Biomarkers. Anal. Chem. 2021, 93, 13719–13726. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Alonso, M.; Fernandez, B.; Garcia, M.; Gonzalez-Iglesias, H.; Pereiro, R. Quantitative Imaging of Specific Proteins in the Human Retina by Laser Ablation ICPMS using Bioconjugated Metal Nanoclusters as Labels. Anal. Chem. 2018, 90, 12145–12151. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Alonso, M.; Fernandez, B.; Navarro, A.; Junceda, S.; Astudillo, A.; Pereiro, R. Laser ablation ICP-MS for simultaneous quantitative imaging of iron and ferroportin in hippocampus of human brain tissues with Alzheimer’s disease. Talanta 2019, 197, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Menero-Valdés, P.; Lores-Padin, A.; Fernández, B.; Gonzalez Igelsias, H.; Pereiro, R. Iridium nanoclusters as high sensitive-tunable elemental labels for immunoassays: Determination of IgE and APOE in aqueous humor by inductively coupled plasma-mass spectrometry. Talanta 2022, 244, 123424. [Google Scholar] [CrossRef] [PubMed]
- Nowak, A.; Rozpedek, W.; Cuchra, M.; Wojtczak, R.; Siwak, M.; Szymanek, K.; Szaflik, M.; Szaflik, J.; Szaflik, J.; Majsterek, I. Association of the expression level of the neurodegeneration-related proteins with the risk of development and progression of primary open-angle glaucoma. Acta Ophthalmol. 2018, 96, E97–E98. [Google Scholar] [CrossRef]
- Jonas, J.B.; Aung, T.; Bourne, R.R.; Bron, A.M.; Ritch, R.; Panda-Jonas, S. Glaucoma. Lancet 2017, 390, 2183–2193. [Google Scholar] [CrossRef]
- Shahidullah, M.; Al-Malki, W.H.; Delamere, N.A. Mechanism of Aqueous Humor Secretion, Its Regulation and Relevance to Glaucoma. In Glaucoma: Basic and Clinical Concepts; Rumelt, S., Ed.; IntechOpen: Rijeka, Croatia, 2011. [Google Scholar]
- Sacca, S.C.; Centofanti, M.; Izzotti, A. New proteins as vascular biomarkers in primary open angle glaucomatous aqueous humor. Investig. Ophthalmol. Vis. Sci. 2012, 53, 4242–4253. [Google Scholar] [CrossRef]
- Inoue, T.; Kawaji, T.; Tanihara, H. Elevated levels of multiple biomarkers of Alzheimer’s disease in the aqueous humor of eyes with open-angle glaucoma. Investig. Ophthalmol. Vis. Sci. 2013, 54, 5353–5358. [Google Scholar] [CrossRef] [Green Version]
- Neilsen, J.L.; Abildtrup, A.; Christensen, J.; Watson, P.; Cox, A.; McLeod, C.W. Laser ablation inductively coupled plasma-mass spectrometry in combination with gel electrophoresis: A new strategy for speciation of metal binding serum proteins. Spectrochim. Acta B 1998, 53, 339–345. [Google Scholar] [CrossRef]
- Marshall, P.; Heudi, O.; Bains, S.; Freeman, H.N.; Abou-Shakra, F.; Reardon, K. The determination of protein phosphorylation on electrophoresis gel blots by laser ablation inductively coupled plasma-mass spectrometry. Analyst 2002, 127, 459–461. [Google Scholar] [CrossRef]
- Wind, M.; Feldmann, I.; Jakubowski, N.; Lehmann, W.D. Spotting and quantification of phosphoproteins purified by gel electrophoresis and laser ablation-element mass spectrometry with phosphorus-31 detection. Electrophoresis 2003, 24, 1276–1280. [Google Scholar] [CrossRef] [PubMed]
- Becker, J.S.; Boulyga, S.F.; Becker, J.S.; Pickhardt, C.; Damoc, E.; Przybylski, M. Structural identification and quantification of protein phosphorylations after gel electrophoretic separation using Fourier transform ion cyclotron resonance mass spectrometry and laser ablation inductively coupled plasma mass spectrometry. Int. J. Mass Spectrom. 2003, 228, 985–997. [Google Scholar] [CrossRef]
- Becker, J.S.; Zoriy, M.; Becker, J.S.; Pickhardt, C.; Przybylski, M. Determination of phosphorus and metals in human brain proteins after isolation by gel electrophoresis by laser ablation inductively coupled plasma source mass spectrometry. J. Anal. At. Spectrom. 2004, 19, 149–152. [Google Scholar] [CrossRef] [Green Version]
- Ma, R.L.; McLeod, C.W.; Tomlinson, K.; Poole, R.K. Speciation of protein-bound trace elements by gel electrophoresis and atomic spectrometry. Electrophoresis 2004, 25, 2469–2477. [Google Scholar] [CrossRef] [PubMed]
- Hare, D.J.; Arora, M.; Jenkins, N.L.; Finkelstein, D.I.; Doble, P.A.; Bush, A.I. Is early-life iron exposure critical in neurodegeneration? Nat. Rev. Neurol. 2015, 11, 536–544. [Google Scholar] [CrossRef] [PubMed]
- Hare, D.J.; Cardoso, B.R.; Szymlek-Gay, E.A.; Biggs, B.A. Neurological effects of iron supplementation in infancy: Finding the balance between health and harm in iron-replete infants. Lancet Child Adolesc. Health 2018, 2, 144–156. [Google Scholar] [CrossRef]
- Kehrer, J.P. The Haber-Weiss reaction and mechanisms of toxicity. Toxicology 2000, 149, 43–50. [Google Scholar] [CrossRef]
- Michalke, B.; Halbach, S.; Nischwitz, V. JEM spotlight: Metal speciation related to neurotoxicity in humans. J. Environ. Monit. 2009, 11, 939–954. [Google Scholar] [CrossRef]
- Solovyev, N.D. Importance of selenium and selenoprotein for brain function: From antioxidant protection to neuronal signalling. J. Inorg. Biochem. 2015, 153, 1–12. [Google Scholar] [CrossRef]
- Fernsebner, K.; Zorn, J.; Kanawati, B.; Walker, A.; Michalke, B. Manganese leads to an increase in markers of oxidative stress as well as to a shift in the ratio of Fe(II)/(III) in rat brain tissue. Metallomics 2014, 6, 921–931. [Google Scholar] [CrossRef] [Green Version]
- Venkataramani, V.; Doeppner, T.R.; Willkommen, D.; Cahill, C.M.; Xin, Y.; Ye, G.; Liu, Y.; Southon, A.; Aron, A.; Au-Yeung, H.Y.; et al. Manganese causes neurotoxic iron accumulation via translational repression of amyloid precursor protein and H-Ferritin. J. Neurochem. 2018, 147, 831–848. [Google Scholar] [CrossRef] [PubMed]
- Michalke, B.; Willkommen, D.; Venkataramani, V. Iron Redox Speciation Analysis Using Capillary Electrophoresis Coupled to Inductively Coupled Plasma Mass Spectrometry (CE-ICP-MS). Front. Chem. 2019, 7, 136. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vara-Perez, M.; Rossi, M.; Van den Haute, C.; Maes, H.; Sassano, M.L.; Venkataramani, V.; Michalke, B.; Romano, E.; Rillaerts, K.; Garg, A.D.; et al. BNIP3 promotes HIF-1alpha-driven melanoma growth by curbing intracellular iron homeostasis. EMBO J. 2021, 40, e106214. [Google Scholar] [CrossRef] [PubMed]
- Kruszewska, J.; Sikorski, J.; Sansonowicz-Gorski, J.; Matczuk, M. A CE-ICP-MS/MS method for the determination of superparamagnetic iron oxide nanoparticles under simulated physiological conditions. Anal. Bioanal. Chem. 2020, 412, 8145–8153. [Google Scholar] [CrossRef] [PubMed]
- Ansari, M.O.; Ahmad, M.; Parveen, N.; Ahmad, S.; Jameel, S.; Shadab, G.G.H.A. Iron oxide nanoparticles-synthesis, surface modification, applications and toxicity: A review. Mater. Focus 2017, 6, 269–279. [Google Scholar] [CrossRef]
- Zheng, J.; Ren, W.; Chen, T.; Jin, Y.; Li, A.; Yan, K.; Wu, Y.; Wu, A. Recent advances in superparamagnetic iron oxide based nanoprobes as multifunctional theranostic agents for breast cancer imaging and therapy. Curr. Med. Chem. 2017, 24, 1–16. [Google Scholar] [CrossRef]
- El Balkhi, S.; Poupon, J.; Trocello, J.M.; Leyendecker, A.; Massicot, F.; Galliot-Guilley, M.; Woimant, F. Determination of ultrafiltrable and exchangeable copper in plasma: Stability and reference values in healthy subjects. Anal. Bioanal. Chem. 2009, 394, 1477–1484. [Google Scholar] [CrossRef]
- Mohr, I.; Weiss, K.H. Biochemical Markers for the Diagnosis and Monitoring of Wilson Disease. Clin. Biochem. Rev. 2019, 40, 59–77. [Google Scholar] [CrossRef]
- Sies, H. Oxidative stress: A concept in redox biology and medicine. Redox Biol. 2015, 4, 180–183. [Google Scholar] [CrossRef] [Green Version]
- Roeser, H.P.; Lee, G.R.; Nacht, S.; Cartwright, G.E. The role of ceruloplasmin in iron metabolism. J. Clin. Investig. 1970, 49, 2408–2417. [Google Scholar] [CrossRef]
- Bost, M.; Houdart, S.; Oberli, M.; Kalonji, E.; Huneau, J.F.; Margaritis, I. Dietary copper and human health: Current evidence and unresolved issues. J. Trace Elem. Med. Biol. 2016, 35, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Zischka, H.; Einer, C. Mitochondrial copper homeostasis and its derailment in Wilson disease. Int. J. Biochem. Cell Biol. 2018, 102, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Saporito-Magrina, C.M.; Musacco-Sebio, R.N.; Andrieux, G.; Kook, L.; Orrego, M.T.; Tuttolomondo, M.V.; Desimone, M.F.; Boerries, M.; Borner, C.; Repetto, M.G. Copper-induced cell death and the protective role of glutathione: The implication of impaired protein folding rather than oxidative stress. Metallomics 2018, 10, 1743–1754. [Google Scholar] [CrossRef] [PubMed]
- Poujois, A.; Trocello, J.M.; Djebrani-Oussedik, N.; Poupon, J.; Collet, C.; Girardot-Tinant, N.; Sobesky, R.; Habes, D.; Debray, D.; Vanlemmens, C.; et al. Exchangeable copper: A reflection of the neurological severity in Wilson’s disease. Eur. J. Neurol. 2017, 24, 154–160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Woimant, F.; Djebrani-Oussedik, N.; Poujois, A. New tools for Wilson’s disease diagnosis: Exchangeable copper fraction. Ann. Transl. Med. 2019, 7, S70. [Google Scholar] [CrossRef]
- Poujois, A.; Woimant, F. Challenges in the diagnosis of Wilson disease. Ann. Transl. Med. 2019, 7, S67. [Google Scholar] [CrossRef]
- Twomey, P.J.; Vijoen, A.; House, I.M.; Reynolds, T.M.; Wierzbicki, A.S. Relationship between serum copper, ceruloplasmin, and non-ceruloplasmin-bound copper in routine clinical practice. Clin. Chem. 2005, 51, 1558–1559. [Google Scholar] [CrossRef]
- Viljoen, A.; Twomey, P.J. Non-caeruloplasmin bound copper in clinical practice. Clin. Chim. Acta 2005, 355, S214. [Google Scholar]
- Walshe, J.M. Wilson’s disease: The importance of measuring serum caeruloplasmin non-immunologically. Ann. Clin. Biochem. 2003, 40, 115–121. [Google Scholar] [CrossRef] [Green Version]
- Huang, X.; Atwood, C.S.; Hartshorn, M.A.; Multhaup, G.; Goldstein, L.E.; Scarpa, R.C.; Cuajungco, M.P.; Gray, D.N.; Lim, J.; Moir, R.D.; et al. The A beta peptide of Alzheimer’s disease directly produces hydrogen peroxide through metal ion reduction. Biochemistry 1999, 38, 7609–7616. [Google Scholar] [CrossRef]
- Roberts, E.A.; Schilsky, M.L.; American Association for Study of Liver Diseases. Diagnosis and treatment of Wilson disease: An update. Hepatology 2008, 47, 2089–2111. [Google Scholar] [CrossRef] [PubMed]
- Wirth, P.L.; Linder, M.C. Distribution of copper among components of human serum. J. Natl. Cancer Inst. 1985, 75, 277–284. [Google Scholar] [PubMed]
- Lopez-Avila, V.; Sharpe, O.; Robinson, W.H. Determination of ceruloplasmin in human serum by SEC-ICPMS. Anal. Bioanal. Chem. 2006, 386, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Gray, L.W.; Kidane, T.Z.; Nguyen, A.; Akagi, S.; Petrasek, K.; Chu, Y.L.; Cabrera, A.; Kantardjieff, K.; Mason, A.Z.; Linder, M.C. Copper proteins and ferroxidases in human plasma and that of wild-type and ceruloplasmin knockout mice. Biochem. J. 2009, 419, 237–245. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Inagaki, K.; Mikuriya, N.; Morita, S.; Haraguchi, H.; Nakahara, Y.; Hattori, M.; Kinosita, T.; Saito, H. Speciation of protein-binding zinc and copper in human blood serum by chelating resin pre-treatment and inductively coupled plasma mass spectrometry. Analyst 2000, 125, 197–203. [Google Scholar] [CrossRef]
- Muniz, C.S.; Gayon, J.M.M.; Alonso, J.I.G.; Sanz-Medel, A. Speciation of essential elements in human serum using anion-exchange chromatography coupled to post-column isotope dilution analysis with double focusing ICP-MS. J. Anal. At. Spectrom. 2001, 16, 587–592. [Google Scholar] [CrossRef]
- Quarles, C.D.; Macke, M.; Michalke, B.; Zischka, H.; Karst, U.; Sullivan, P.; Field, M.P. LC-ICP-MS method for the determination of “extractable copper” in serum. Metallomics 2020, 12, 1348–1355. [Google Scholar] [CrossRef]
- del Castillo Busto, M.E.D.; Cuello-Nunez, S.; Ward-Deitrich, C.; Morley, T.; Goenaga-Infante, H. A fit-for-purpose copper speciation method for the determination of exchangeable copper relevant to Wilson’s disease. Anal. Bioanal. Chem. 2022, 414, 561–573. [Google Scholar] [CrossRef]
- Trouillon, R.; Passarelli, M.K.; Wang, J.; Kurczy, M.E.; Ewing, A.G. Chemical analysis of single cells. Anal. Chem. 2013, 85, 522–542. [Google Scholar] [CrossRef]
- Kleparnik, K.; Foret, F. Recent advances in the development of single cell analysis—A review. Anal. Chim. Acta 2013, 800, 12–21. [Google Scholar] [CrossRef]
- Lanni, E.J.; Rubakhin, S.S.; Sweedler, J.V. Mass spectrometry imaging and profiling of single cells. J. Proteom. 2012, 75, 5036–5051. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Theiner, S.; Schoeberl, A.; Schweikert, A.; Keppler, B.K.; Koellensperger, G. Mass spectrometry techniques for imaging and detection of metallodrugs. Curr. Opin. Chem. Biol. 2021, 61, 123–134. [Google Scholar] [CrossRef] [PubMed]
- Mueller, L.; Traub, H.; Jakubowski, N.; Drescher, D.; Baranov, V.I.; Kneipp, J. Trends in single-cell analysis by use of ICP-MS. Anal. Bioanal. Chem. 2014, 406, 6963–6977. [Google Scholar] [CrossRef] [PubMed]
- Amable, L.; Stephan, C.; Smith, S.; Merrifield, R.B. An Introduction to Single Cell ICP-MS Analysis. White Paper 2017. Available online: https://www.semanticscholar.org/paper/An-Introduction-to-Single-Cell-ICP-MS-Analysis-Amable-Stephan/83b07090a415b10c57d16f622348d383daef03f5 (accessed on 29 March 2022).
- Meyer, S.; Lopez-Serrano, A.; Mitze, H.; Jakubowski, N.; Schwerdtle, T. Single-cell analysis by ICP-MS/MS as a fast tool for cellular bioavailability studies of arsenite. Metallomics 2018, 10, 73–76. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Houk, R.S. Behavior of bacteria in the inductively coupled plasma: Atomization and production of atomic ions for mass spectrometry. Anal. Chem. 2005, 77, 1407–1413. [Google Scholar] [CrossRef]
- Verboket, P.E.; Borovinskaya, O.; Meyer, N.; Gunther, D.; Dittrich, P.S. A New Microfluidics-Based Droplet Dispenser for ICPMS. Anal. Chem. 2014, 86, 6012–6018. [Google Scholar] [CrossRef]
- Giesen, C.; Wang, H.A.O.; Schapiro, D.; Zivanovic, N.; Jacobs, A.; Hattendorf, B.; Schuffler, P.J.; Grolimund, D.; Buhmann, J.M.; Brandt, S.; et al. Highly multiplexed imaging of tumor tissues with subcellular resolution by mass cytometry. Nat. Methods 2014, 11, 417. [Google Scholar] [CrossRef]
- Giesen, C.; Wang, H.A.O.; Schapiro, D.; Hattendorf, B.; Wild, P.J.; Varga, Z.; Gunther, D.; Bodenmiller, B. Highly multiplexed tissue imaging of breast tumors and their microenvironment by mass cytometry. FEBS J. 2014, 281, 55. [Google Scholar]
- Bodenmiller, B.; Zunder, E.R.; Finck, R.; Chen, T.J.; Savig, E.S.; Bruggner, R.V.; Simonds, E.F.; Bendall, S.C.; Sachs, K.; Krutzik, P.O.; et al. Multiplexed mass cytometry profiling of cellular states perturbed by small-molecule regulators. Nat. Biotechnol. 2012, 30, 858–867. [Google Scholar] [CrossRef]
- Zivanovic, N.; Jacobs, A.; Bodenmiller, B. A Practical Guide to Multiplexed Mass Cytometry. Curr. Top. Microbiol. 2014, 377, 95–109. [Google Scholar] [CrossRef]
- Amable, L. Cisplatin resistance and opportunities for precision medicine. Pharmacol. Res. 2016, 106, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Gale, A.; Hofmann, L.; Ludi, N.; Hungerbuhler, M.N.; Kempf, C.; Heverhagen, J.T.; Von Tengg-Kobligk, H.; Broekmann, P.; Ruprecht, N. Beyond Single-Cell Analysis of Metallodrugs by ICP-MS: Targeting Cellular Substructures. Int. J. Mol. Sci. 2021, 22, 9468. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.-N.; Wang, M.; Zhao, L.-C.; Sun, B.-Y.; Wang, B.; Chen, H.-Q.; Zhao, Y.-L.; Chai, Z.-F.; Feng, W.-Y. Quantitative analyisof Gd@C82(OH)22 and cisplatin uptake in single cells by inductively coupled plasma mass spectrometry. Anal. Bioanal. Chem. 2015, 407, 2383–2391. [Google Scholar] [CrossRef]
- Hu, K.; Nguyen, T.D.K.; Rabasco, S.; Oomen, P.E.; Ewing, A.G. Chemical Analysis of Single Cells and Organelles. Anal. Chem. 2021, 93, 41–71. [Google Scholar] [CrossRef] [PubMed]
- Lohr, K.; Borovinskaya, O.; Tourniaire, G.; Panne, U.; Jakubowski, N. Arraying of Single Cells for Quantitative High Throughput Laser Ablation ICP-TOF-MS. Anal. Chem. 2019, 91, 11520–11528. [Google Scholar] [CrossRef]
- Pisonero, J.; Bouzas-Ramos, D.; Traub, H.; Cappella, B.; Álvarez-Llamas, C.; Richter, S.; Mayo, J.C.; Costa-Fernandez, J.M.; Bordela, N.; Jakubowski, N. Critical evaluation of fast and highly resolved elemental distribution in single cells using LA-ICP-SFMS. J. Anal. At. Spectrom. 2019, 34, 655–663. [Google Scholar] [CrossRef]
- Zheng, L.-N.; Sang, Y.-B.; Luo, R.-P.; Wang, B.; Yi, F.-T.; Wang, M.; Feng, W.-Y. Determination of silver nanoparticles in single cells by microwell trapping and laser ablation ICP-MS determination. J. Anal. At. Spectrom. 2019, 34, 915–921. [Google Scholar] [CrossRef]
- Van Acker, T.; Buckle, T.; Van Malderen, S.J.M.; van Willigen, D.M.; van Unen, V.; van Leeuwen, F.W.B.; Vanhaecke, F. High-resolution imaging and single-cell analysis via laser ablation-inductively coupled plasma-mass spectrometry for the determination of membranous receptor expression levels in breast cancer cell lines using receptor-specific hybrid tracers. Anal. Chim. Acta 2019, 1074, 43–53. [Google Scholar] [CrossRef]
- Lee, S.Y.; Kim, C.Y.; Nam, T.-G. Ruthenium complexes as anticancer agents: A brief history and perspectives. Drug Des. Dev. Ther. 2020, 14, 5375–5392. [Google Scholar] [CrossRef]
- Zhang, S.Q.; Gao, L.-H.; Zhao, H.; Wang, K.-Z. Recent progress in polynuclear ruthenium complex-based DNA binders/structural probes and anticancer agents. Curr. Med. Chem. 2020, 27, 3735–3752. [Google Scholar] [CrossRef]
- Mora, M.; Gimeno, M.C.; Visbal, R. Recent advances in gold-NHC complexes with biological properties. Chem. Soc. Rev. 2019, 48, 447–462. [Google Scholar] [CrossRef] [PubMed]
- Bayrakdar, T.A.C.A.; Scattolin, T.; Ma, X.; Nolan, S.P. Dinuclear gold(I) complexes: From bonding to applications. Chem. Soc. Rev. 2020, 49, 7044–7100. [Google Scholar] [CrossRef] [PubMed]
- Casini, A.; Vessières, A.; Meier-Menches, S.M. Metal-Based Anticancer Agents; Royal Society of Chemistry: London, UK, 2019; ISBN 978-1-78801-645-2. [Google Scholar]
- Klose, M.H.M.; Theiner, S.; Kornauth, C.; Meier-Menches, S.M.; Heffeter, P.; Berger, W.; Koellensperger, G.; Keppler, B.K. Bioimaging of isosteric osmium and ruthenium anticancer agents by LA-ICP-MS. Metallomic 2018, 10, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Greenhalgh, C.J.; Karekla, E.; Miles, G.J.; Powley, I.R.; Costa, C.; de Jesus, J.; Bailey, M.J.; Pritchard, C.; MacFarlane, M.; Pringle, J.H.; et al. Exploration of matrix effects in laser ablation inductively coupled plasma mass spectrometry imaging of cisplatin-treated tumors. Anal. Chem. 2020, 92, 9847–9855. [Google Scholar] [CrossRef]
- Theiner, S.; Kornauth, C.; Varbanov, H.P.; Galanski, M.; Van Schoonhoven, S.; Heffeter, P.; Berger, W.; Egger, A.E.; Keppler, B.K. Tumor microenvironment in focus: LA-ICP-MS bioimaging of a preclinical tumor model upon treatment with platinum(iv)-based anticancer agents. Metallomics 2015, 7, 1256–1264. [Google Scholar] [CrossRef]
- Scattolin, T.; Voloshkin, V.A.; Visentin, F.; Nolan, S.P. A critical review of palladium organometallic anticancer agents. Cell Rep. Phys. Sci. 2021, 2, 100446. [Google Scholar] [CrossRef]
- Niehoff, A.-C.; Moosmann, A.; Sobbing, J.; Wiehe, A.; Mulac, D.; Wehe, C.A.; Reifschneider, O.; Blaske, F.; Wagner, S.; Sperling, M.; et al. A palladium label to monitor nanoparticle-assisted drug delivery of a photosensitizer into tumor spheroids by elemental bioimaging. Metallomics 2014, 6, 77–81. [Google Scholar] [CrossRef]
- Bouzekri, A.; Esch, A.; Ornatsk, O. Multidimensional profiling of drug-treated cells by Imaging Mass Cytometry. FEBS Open Bio 2019, 9, 1652–1669. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Michalke, B. Review about Powerful Combinations of Advanced and Hyphenated Sample Introduction Techniques with Inductively Coupled Plasma-Mass Spectrometry (ICP-MS) for Elucidating Trace Element Species in Pathologic Conditions on a Molecular Level. Int. J. Mol. Sci. 2022, 23, 6109. https://doi.org/10.3390/ijms23116109
Michalke B. Review about Powerful Combinations of Advanced and Hyphenated Sample Introduction Techniques with Inductively Coupled Plasma-Mass Spectrometry (ICP-MS) for Elucidating Trace Element Species in Pathologic Conditions on a Molecular Level. International Journal of Molecular Sciences. 2022; 23(11):6109. https://doi.org/10.3390/ijms23116109
Chicago/Turabian StyleMichalke, Bernhard. 2022. "Review about Powerful Combinations of Advanced and Hyphenated Sample Introduction Techniques with Inductively Coupled Plasma-Mass Spectrometry (ICP-MS) for Elucidating Trace Element Species in Pathologic Conditions on a Molecular Level" International Journal of Molecular Sciences 23, no. 11: 6109. https://doi.org/10.3390/ijms23116109





