Metabolome and Transcriptome Profiling Unveil the Mechanisms of Polyphenol Synthesis in the Developing Endopleura of Walnut (Juglans regia L.)
Abstract
:1. Introduction
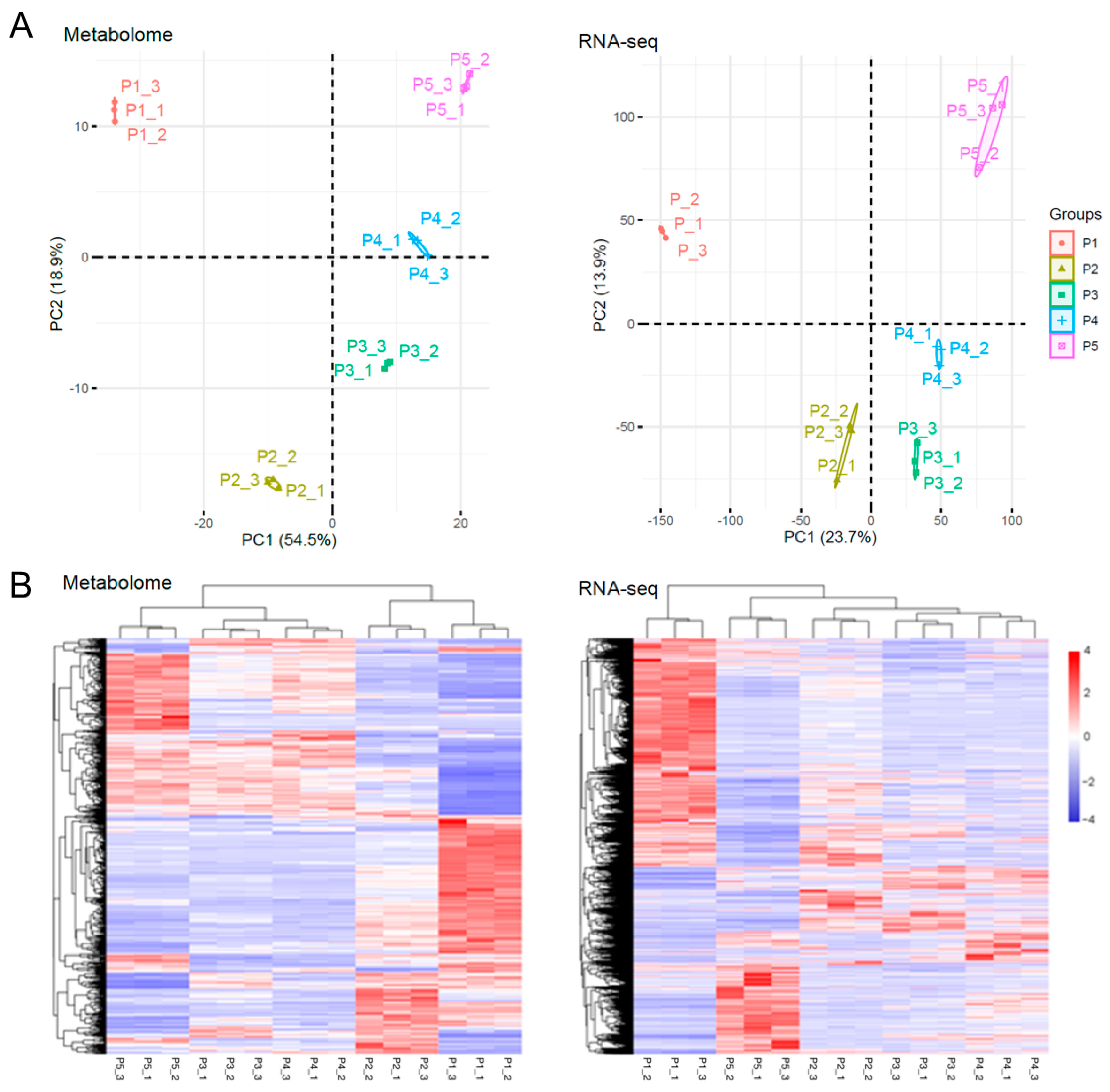
2. Results and Discussion
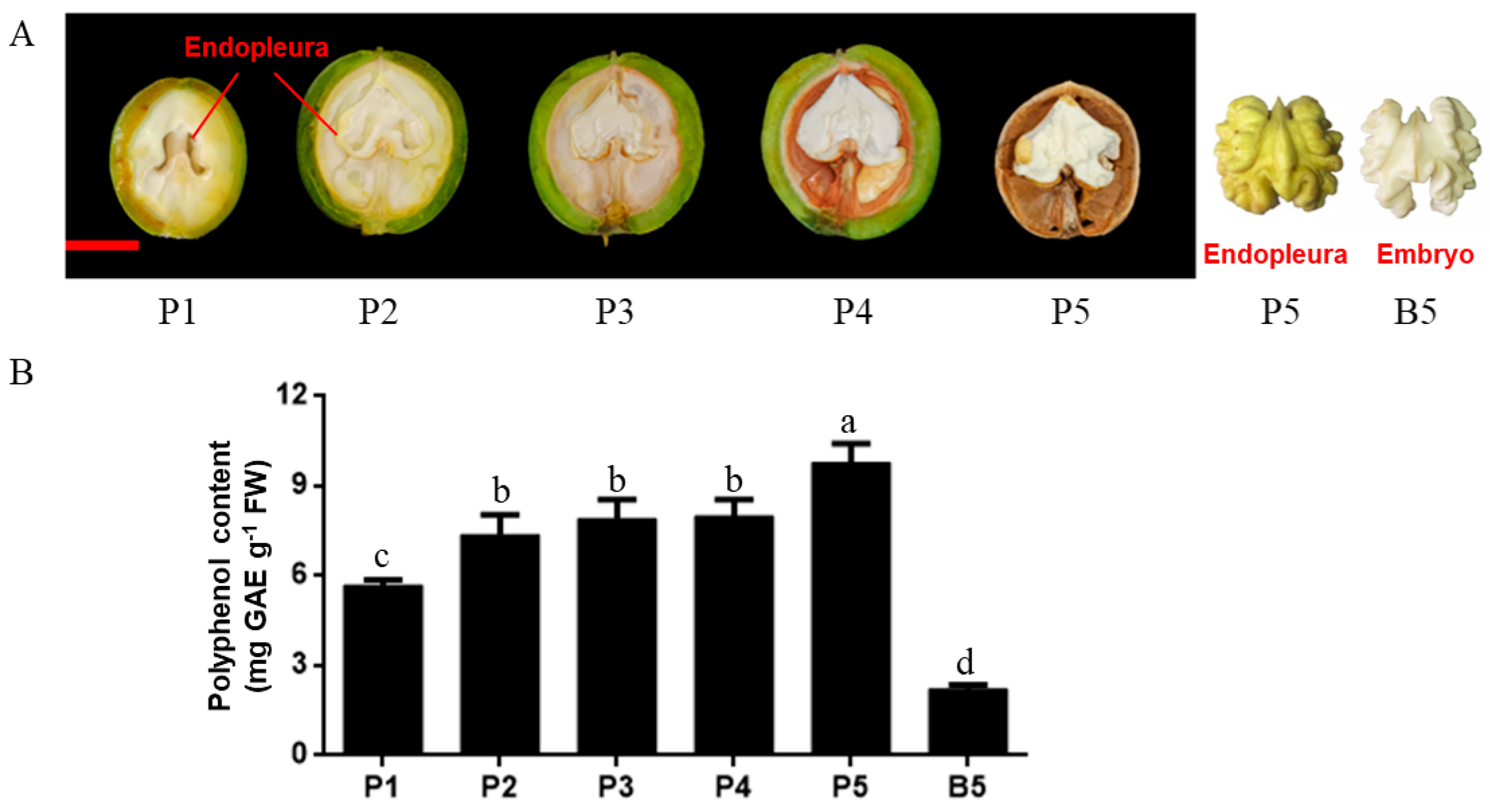
2.1. Morphological Characteristics and Polyphenol Content
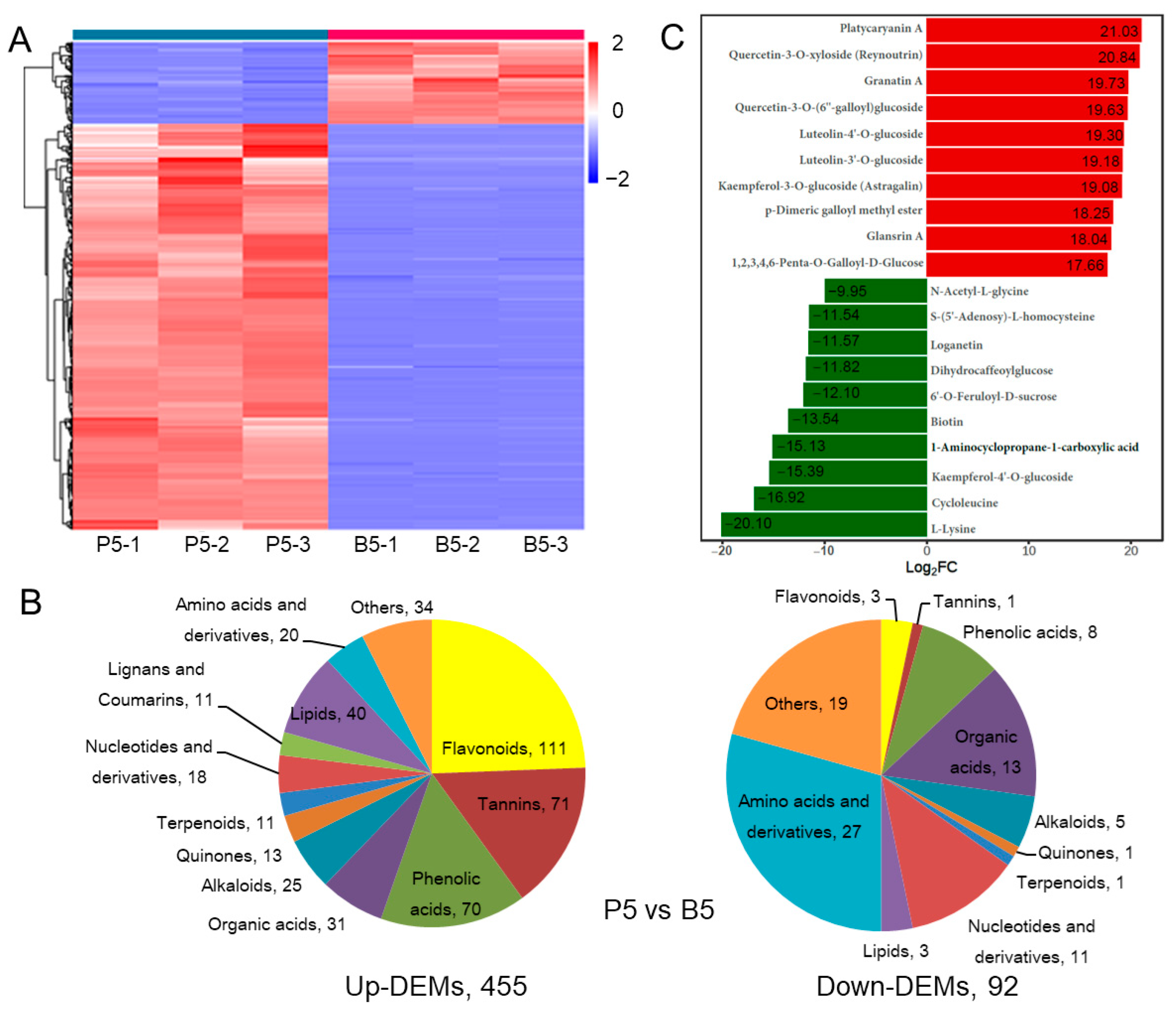
2.2. Comparison of DAMs between Endopleura and Embryo in the Mature Walnut Nut
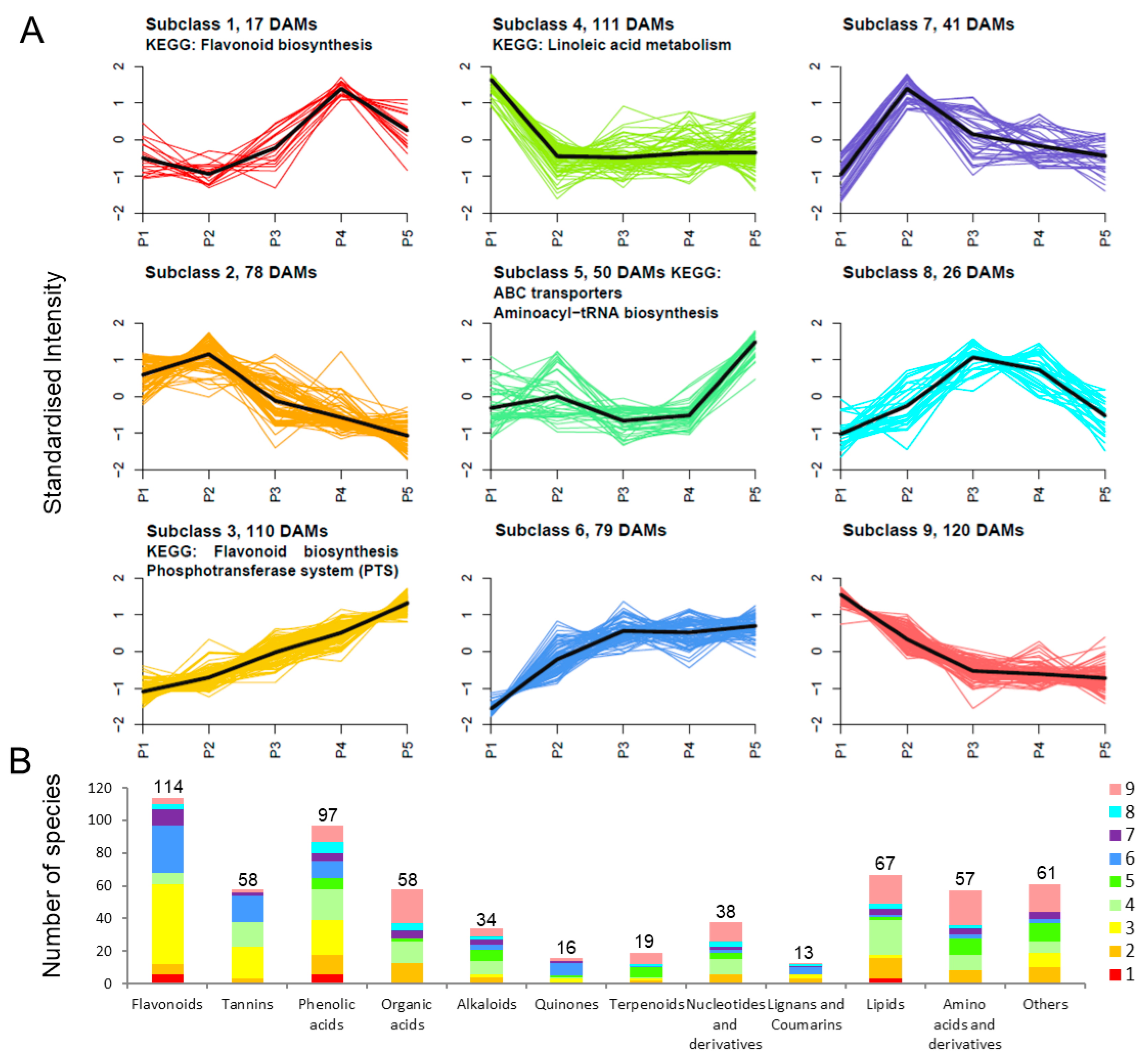
2.3. Trend Analysis of DAMs and DEGs in Endopleura
2.4. KEGG Enrichment Analysis of DAMs in Endopleura
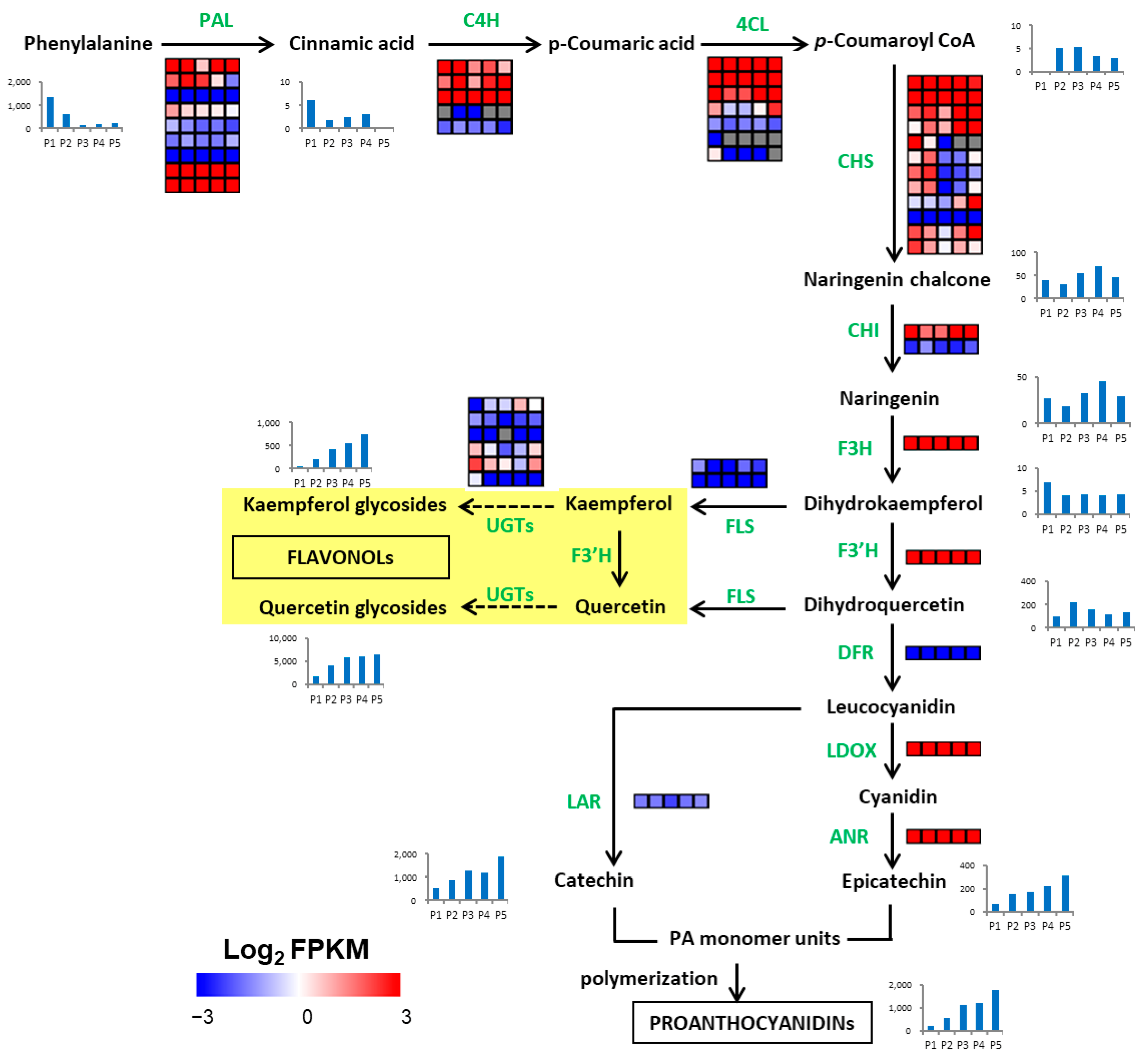
2.5. Metabolites Related to Polyphenol Synthesis in Endopleura
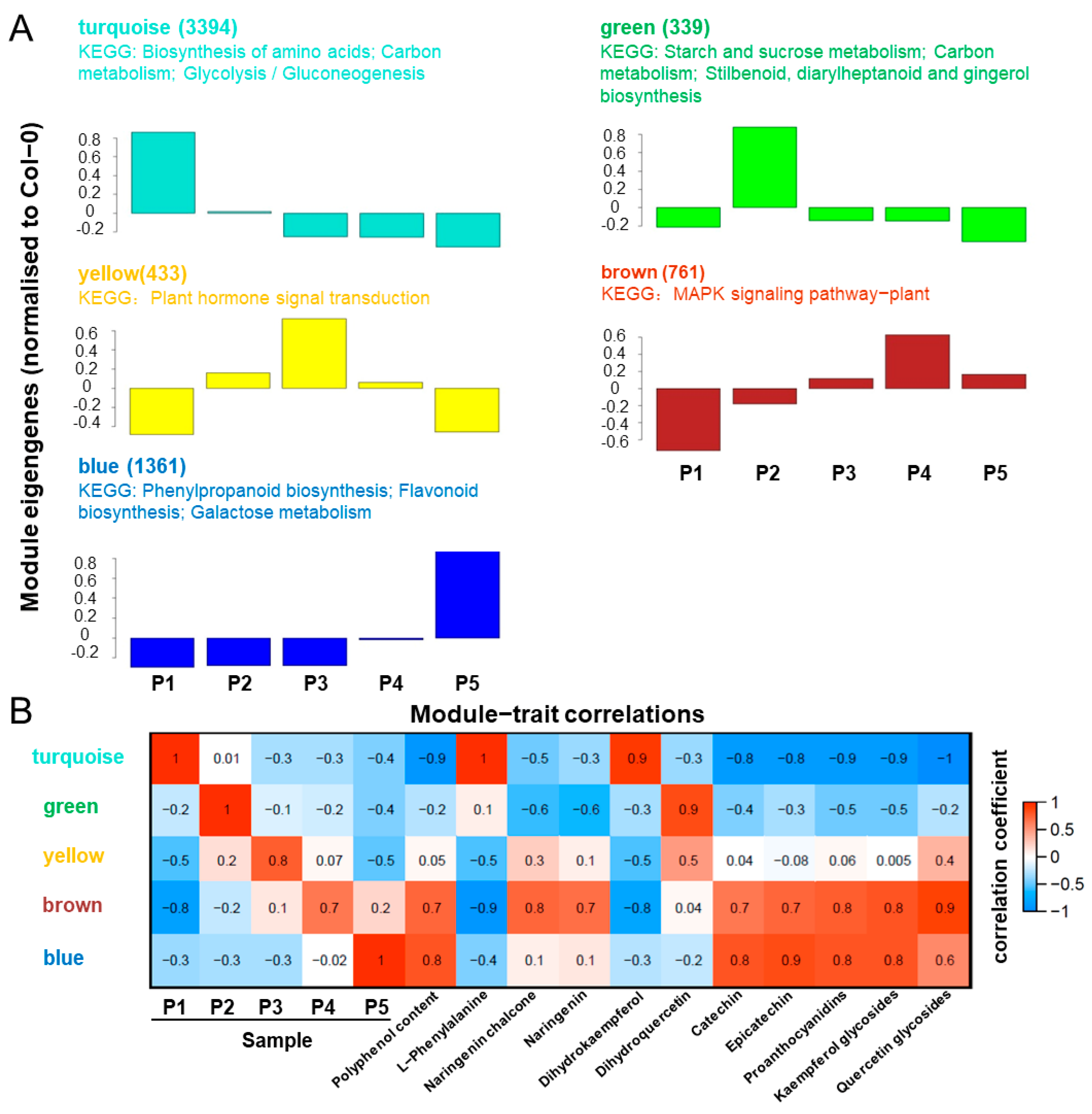
2.6. Trend Analysis of DEGs in Endopleura
2.7. Identification of Genes Related to Polyphenol Synthesis
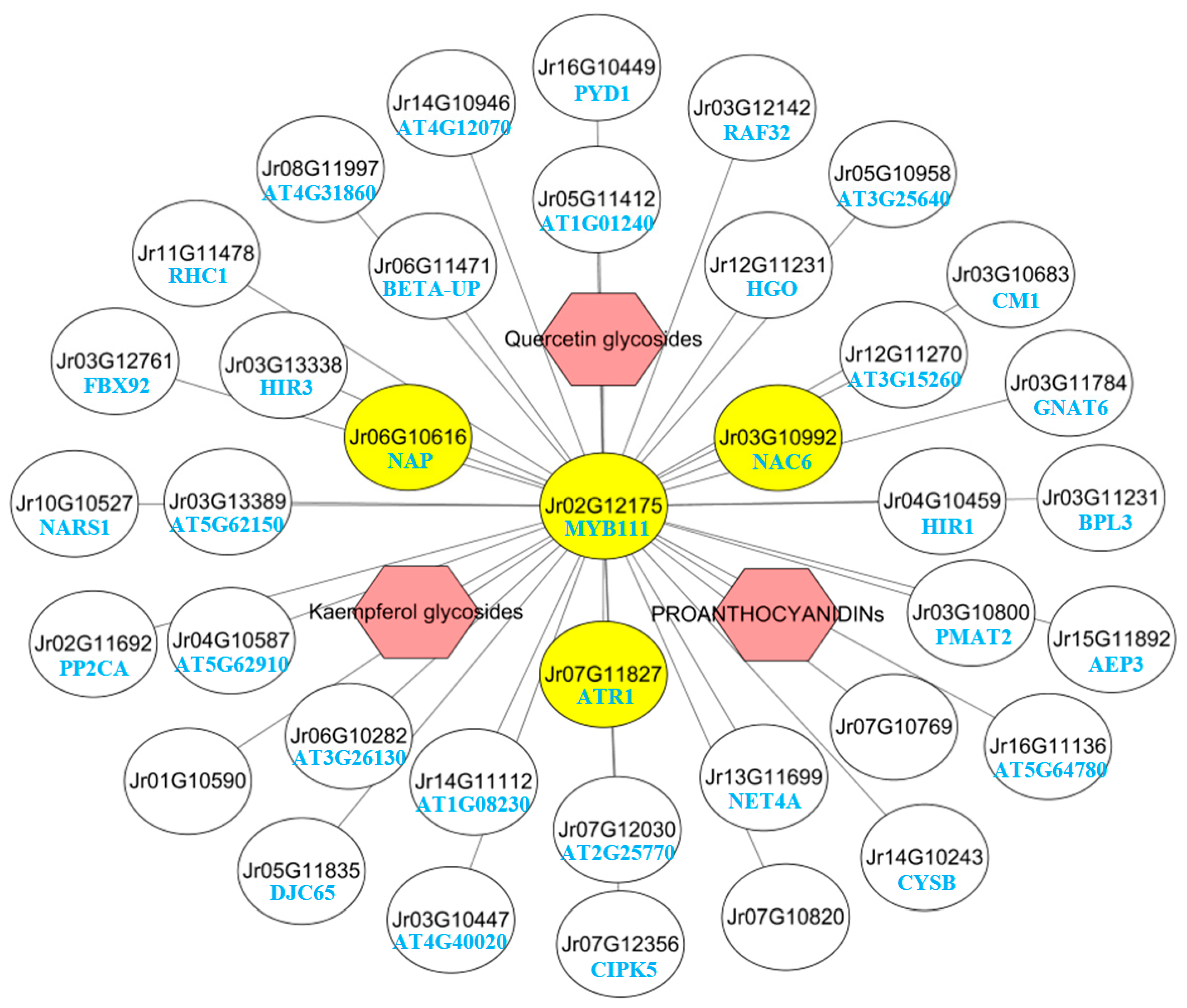
2.8. Transcription Factors Related to Polyphenol Synthesis
2.9. MYB111 Co-Expression Network Analysis
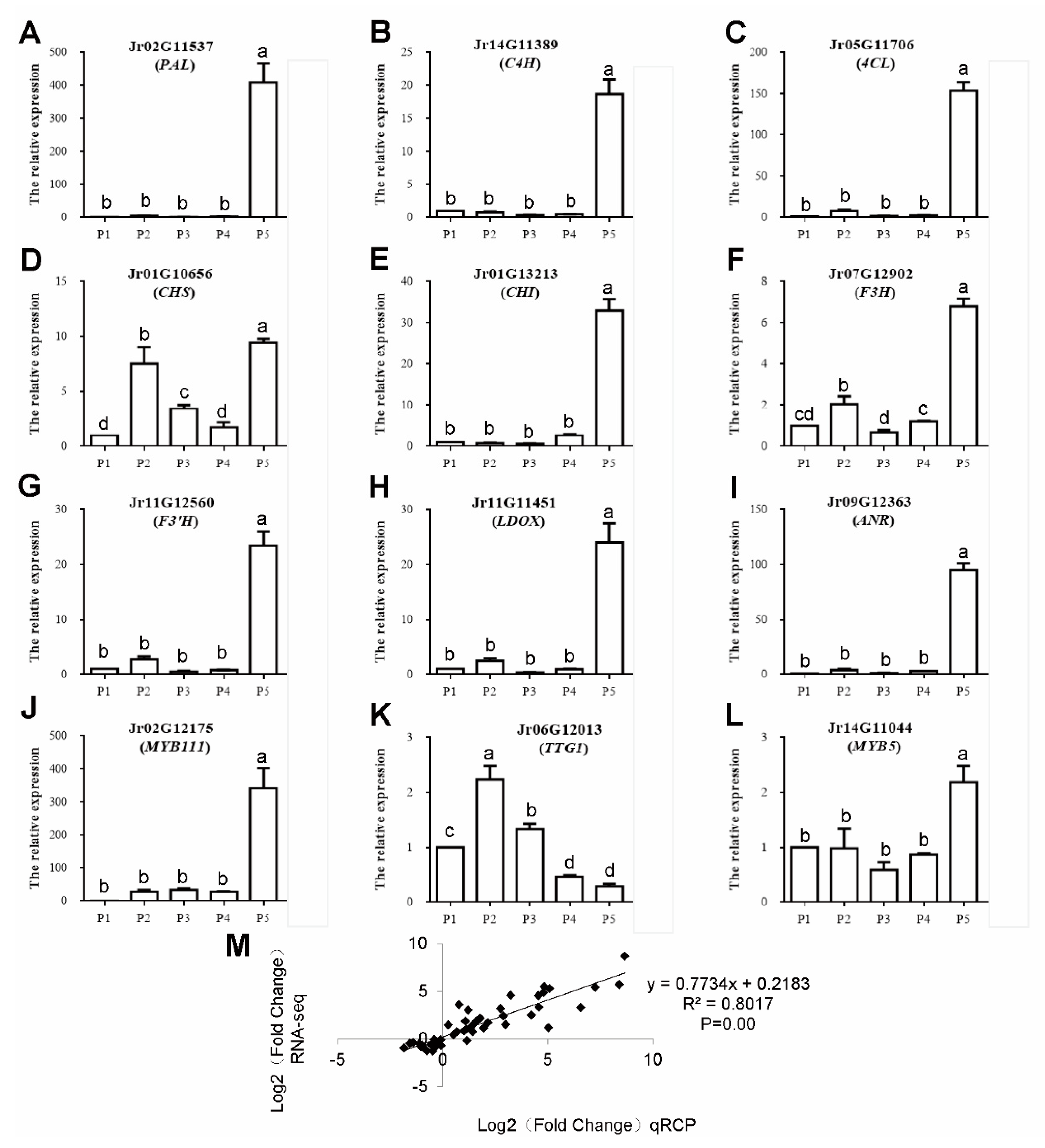
2.10. qPCR Analysis of Polyphenol-Related Genes
3. Materials and Methods
3.1. Plant Material
3.2. Determination of Polyphenol Content
3.3. Metabonomic Analysis
3.4. Transcriptome Sequencing
3.5. RNA-Seq Data Analysis
3.6. Co-Expression Network Analysis
3.7. qRT-PCR Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Ma, Q.; Bu, D.; Zhang, J.; Wu, Y.; Pei, D. The transcriptome landscape of walnut interspecies hybrid (Juglans hindsii × Juglans regia) and regulation of cambial activity in relation to grafting. Front. Genet. 2019, 10, 577. [Google Scholar] [CrossRef]
- Ros, E.; Izquierdo-Pulido, M.; Sala-Vila, A. Beneficial effects of walnut consumption on human health: Role of micronutrients. Curr. Opin. Clin. Nutr. Metab. Care 2018, 21, 498–504. [Google Scholar] [CrossRef]
- Abe, L.T.; Lajolo, F.M.; Genovese, M.I. Comparison of phenol content and antioxidant capacity of nuts. Food Sci. Technol. 2010, 30, 254–259. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Wang, K.; Lyu, S.; Huang, J.; Huang, C.; Xing, Y.; Wang, Y.; Xu, Y.; Li, P.; Hong, J. Genome-Wide Identification of Tannase Genes and Their Function of Wound Response and Astringent Substances Accumulation in Juglandaceae. Front. Plant Sci. 2021, 12, 739. [Google Scholar] [CrossRef]
- Zhang, Z.; Liao, L.; Moore, J.; Wu, T.; Wang, Z. Antioxidant phenolic compounds from walnut kernels (Juglans regia L.). Food Chem. 2009, 113, 160–165. [Google Scholar] [CrossRef]
- Choi, J.; Shin, P.-K.; Kim, Y.; Hong, C.P.; Choi, S.-W. Metabolic influence of walnut phenolic extract on mitochondria in a colon cancer stem cell model. Eur. J. Nutr. 2019, 58, 1635–1645. [Google Scholar] [CrossRef]
- Fukuda, T.; Ito, H.; Yoshida, T. Antioxidative polyphenols from walnuts (Juglans regia L.). Phytochemistry 2003, 63, 795–801. [Google Scholar] [CrossRef]
- Gramazio, P.; Prohens, J.; Plazas, M.; Andújar, I.; Herraiz, F.J.; Castillo, E.; Knapp, S.; Meyer, R.S.; Vilanova, S. Location of chlorogenic acid biosynthesis pathway and polyphenol oxidase genes in a new interspecific anchored linkage map of eggplant. BMC Plant Biol. 2014, 14, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Kocábek, T.; Mishra, A.K.; Matoušek, J.; Patzak, J.; Lomnická, A.; Khare, M.; Krofta, K. The R2R3 transcription factor HlMYB8 and its role in flavonoid biosynthesis in hop (Humulus lupulus L.). Plant Sci. 2018, 269, 32–46. [Google Scholar] [CrossRef]
- Akagi, T.; Katayama-Ikegami, A.; Yonemori, K. Proanthocyanidin biosynthesis of persimmon (Diospyros kaki Thunb.) fruit. Sci. Hortic. 2011, 130, 373–380. [Google Scholar] [CrossRef]
- Debeaujon, I.; Léon-Kloosterziel, K.M.; Koornneef, M. Influence of the testa on seed dormancy, germination, and longevity in Arabidopsis. Plant Physiol. 2000, 122, 403–414. [Google Scholar] [CrossRef] [Green Version]
- Auger, B.; Marnet, N.; Gautier, V.; Maia-Grondard, A.; Leprince, F.; Renard, M.; Guyot, S.; Nesi, N.; Routaboul, J.-M. A detailed survey of seed coat flavonoids in developing seeds of Brassica napus L. J. Agric. Food Chem. 2010, 58, 6246–6256. [Google Scholar] [CrossRef]
- Ikegami, A.; Akagi, T.; Potter, D.; Yamada, M.; Sato, A.; Yonemori, K.; Kitajima, A.; Inoue, K. Molecular identification of 1-Cys peroxiredoxin and anthocyanidin/flavonol 3-O-galactosyltransferase from proanthocyanidin-rich young fruits of persimmon (Diospyros kaki Thunb.). Planta 2009, 230, 841–855. [Google Scholar] [CrossRef] [Green Version]
- Wang, N.; Xu, H.; Jiang, S.; Zhang, Z.; Lu, N.; Qiu, H.; Qu, C.; Wang, Y.; Wu, S.; Chen, X. MYB12 and MYB22 play essential roles in proanthocyanidin and flavonol synthesis in red-fleshed apple (Malus sieversii f. niedzwetzkyana). Plant J. 2017, 90, 276–292. [Google Scholar] [CrossRef] [Green Version]
- Zhai, R.; Zhao, Y.; Wu, M.; Yang, J.; Li, X.; Liu, H.; Wu, T.; Liang, F.; Yang, C.; Wang, Z. The MYB transcription factor PbMYB12b positively regulates flavonol biosynthesis in pear fruit. BMC Plant Biol. 2019, 19, 1–11. [Google Scholar] [CrossRef]
- Lepiniec, L.; Debeaujon, I.; Routaboul, J.-M.; Baudry, A.; Pourcel, L.; Nesi, N.; Caboche, M. Genetics and biochemistry of seed flavonoids. Annu. Rev. Plant Biol. 2006, 57, 405–430. [Google Scholar] [CrossRef]
- Ravaglia, D.; Espley, R.V.; Henry-Kirk, R.A.; Andreotti, C.; Ziosi, V.; Hellens, R.P.; Costa, G.; Allan, A.C. Transcriptional regulation of flavonoid biosynthesis in nectarine (Prunus persica) by a set of R2R3 MYB transcription factors. BMC Plant Biol. 2013, 13, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Stracke, R.; Ishihara, H.; Huep, G.; Barsch, A.; Mehrtens, F.; Niehaus, K.; Weisshaar, B. Differential regulation of closely related R2R3-MYB transcription factors controls flavonol accumulation in different parts of the Arabidopsis thaliana seedling. Plant J. 2007, 50, 660–677. [Google Scholar] [CrossRef] [Green Version]
- Regueiro, J.; Sánchez-González, C.; Vallverdú-Queralt, A.; Simal-Gándara, J.; Lamuela-Raventós, R.; Izquierdo-Pulido, M. Comprehensive identification of walnut polyphenols by liquid chromatography coupled to linear ion trap–Orbitrap mass spectrometry. Food Chem. 2014, 152, 340–348. [Google Scholar] [CrossRef]
- Wang, H.; Asker, K.; Zhan, C.; Wang, N. Transcriptomic and Metabolic Analysis of Fruit Development and Identification of Genes Involved in Raffinose and Hydrolysable Tannin Biosynthesis in Walnuts. J. Agric. Food Chem. 2021, 69, 8050–8062. [Google Scholar] [CrossRef]
- Jia, X.; Luo, H.; Xu, M.; Zhai, M.; Guo, Z.; Qiao, Y.; Wang, L. Dynamic changes in phenolics and antioxidant capacity during pecan (Carya illinoinensis) kernel ripening and its phenolics profiles. Molecules 2018, 23, 435. [Google Scholar] [CrossRef] [Green Version]
- Haramiishi, R.; Okuyama, S.; Yoshimura, M.; Nakajima, M.; Furukawa, Y.; Ito, H.; Amakura, Y. Identification of the characteristic components in walnut and anti-inflammatory effect of glansreginin A as an indicator for quality evaluation. Biosci. Biotechnol. Biochem. 2020, 84, 187–197. [Google Scholar] [CrossRef]
- Ingolia, N.T.; Ghaemmaghami, S.; Newman, J.R.; Weissman, J.S. Genome-wide analysis in vivo of translation with nucleotide resolution using ribosome profiling. Science 2009, 324, 218–223. [Google Scholar] [CrossRef] [Green Version]
- Pan, X.; Siloto, R.M.; Wickramarathna, A.D.; Mietkiewska, E.; Weselake, R.J. Identification of a pair of phospholipid: Diacylglycerol acyltransferases from developing flax (Linum usitatissimum L.) seed catalyzing the selective production of trilinolenin. J. Biol. Chem. 2013, 288, 24173–24188. [Google Scholar] [CrossRef] [Green Version]
- Arlotta, C.; Puglia, G.D.; Genovese, C.; Toscano, V.; Karlova, R.; Beekwilder, J.; De Vos, R.C.; Raccuia, S.A. MYB5-like and bHLH influence flavonoid composition in pomegranate. Plant Sci. 2020, 298, 110563. [Google Scholar] [CrossRef]
- Liu, C.; Jun, J.H.; Dixon, R.A. MYB5 and MYB14 play pivotal roles in seed coat polymer biosynthesis in Medicago truncatula. Plant Physiol. 2014, 165, 1424–1439. [Google Scholar] [CrossRef] [Green Version]
- Appelhagen, I.; Lu, G.H.; Huep, G.; Schmelzer, E.; Weisshaar, B.; Sagasser, M. TRANSPARENT TESTA1 interacts with R2R3-MYB factors and affects early and late steps of flavonoid biosynthesis in the endothelium of Arabidopsis thaliana seeds. Plant J. 2011, 67, 406–419. [Google Scholar] [CrossRef]
- Li, B.; Fan, R.; Guo, S.; Wang, P.; Zhu, X.; Fan, Y.; Chen, Y.; He, K.; Kumar, A.; Shi, J. The Arabidopsis MYB transcription factor, MYB111 modulates salt responses by regulating flavonoid biosynthesis. Environ. Exp. Bot. 2019, 166, 103807. [Google Scholar] [CrossRef]
- Hinckley, W.E.; Brusslan, J.A. Gene expression changes occurring at bolting time are associated with leaf senescence in Arabidopsis. Plant Direct 2020, 4, e00279. [Google Scholar] [CrossRef]
- Sundström, L.; Larsson, S.; Jönsson, L.J. Identification of Saccharomyces cerevisiae genes involved in the resistance to phenolic fermentation inhibitors. Appl. Biochem. Biotechnol. 2010, 161, 106–115. [Google Scholar] [CrossRef]
- Taguchi, G.; Ubukata, T.; Nozue, H.; Kobayashi, Y.; Takahi, M.; Yamamoto, H.; Hayashida, N. Malonylation is a key reaction in the metabolism of xenobiotic phenolic glucosides in Arabidopsis and tobacco. Plant J. 2010, 63, 1031–1041. [Google Scholar] [CrossRef]
- Baute, J.; Polyn, S.; De Block, J.; Blomme, J.; Van Lijsebettens, M.; Inzé, D. F-box protein FBX92 affects leaf size in Arabidopsis thaliana. Plant Cell Physiol. 2017, 58, 962–975. [Google Scholar] [CrossRef] [Green Version]
- Golz, J.F.; Allen, P.J.; Li, S.F.; Parish, R.W.; Jayawardana, N.U.; Bacic, A.; Doblin, M.S. Layers of regulation–Insights into the role of transcription factors controlling mucilage production in the Arabidopsis seed coat. Plant Sci. 2018, 272, 179–192. [Google Scholar] [CrossRef]
- Havé, M.; Marmagne, A.; Chardon, F.; Masclaux-Daubresse, C. Nitrogen remobilization during leaf senescence: Lessons from Arabidopsis to crops. J. Exp. Bot. 2017, 68, 2513–2529. [Google Scholar]
- Wang, W.; Li, X.; Zhu, M.; Tang, X.; Wang, Z.; Guo, K.; Zhou, Y.; Sun, Y.; Zhang, W.; Li, X. Arabidopsis GAAP1 to GAAP3 play redundant role in cell death inhibition by suppressing the upregulation of salicylic acid pathway under endoplasmic reticulum stress. Front. Plant Sci. 2019, 10, 1032. [Google Scholar] [CrossRef]
- Huang, R.; Zhou, Y.; Zhang, J.; Ji, F.; Jin, F.; Fan, W.; Pei, D. Transcriptome Analysis of Walnut (Juglans regia L.) Embryos Reveals Key Developmental Stages and Genes Involved in Lipid Biosynthesis and Polyunsaturated Fatty Acid Metabolism. J. Agric. Food Chem. 2020, 69, 377–396. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. [14] Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar]
- Ji, F.; Ma, Q.; Zhang, W.; Liu, J.; Feng, Y.; Zhao, P.; Song, X.; Chen, J.; Zhang, J.; Wei, X. A genome variation map provides insights into the genetics of walnut adaptation and agronomic traits. Genome Biol. 2021, 22, 1–22. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, W.; Ji, F.; Qiu, J.; Song, X.; Bu, D.; Pan, G.; Ma, Q.; Chen, J.; Huang, R. A high-quality walnut genome assembly reveals extensive gene expression divergences after whole-genome duplication. Plant Biotechnol. J. 2020, 18, 1848. [Google Scholar] [CrossRef]

| KEGG Pathway | Number of DAMs | p-Value |
|---|---|---|
| Flavonoid biosynthesis | 28 | 0.0006 |
| Flavone and flavonol biosynthesis | 15 | 0.0338 |
| Arginine and proline metabolism | 14 | 0.0451 |
| Compounds | Molecular Weight (Da) | P1 | P2 | P3 | P4 | P5 | Subclass |
|---|---|---|---|---|---|---|---|
| ×104 | ×104 | ×104 | ×104 | ×104 | |||
| Phenylalanine | 165 | 1367.73 | 613.52 | 152.69 | 186.15 | 230.17 | 9 |
| Naringenin chalcone | 272 | 40.63 | 30.8 | 55.69 | 70.03 | 47.05 | 1 |
| Naringenin (5,7,4′-trihydroxyflavanone) | 272 | 27.55 | 19.02 | 32.15 | 45.58 | 29.86 | 1 |
| Aromadendrin (dihydrokaempferol) | 288 | 6.87 | 4.01 | 4.34 | 4.12 | 4.3 | |
| Dihydroquercetin(taxifolin) | 304 | 94.73 | 221.76 | 156.95 | 116.33 | 130.31 | |
| Catechin | 290 | 533.24 | 875.19 | 1288.7 | 1184.77 | 1861.27 | 3 |
| Epicatechin | 290 | 64.98 | 153.18 | 167.48 | 220.96 | 311.58 | 3 |
| PROANTHOCYANIDINs | |||||||
| Procyanidin B3 | 578 | 15.78 | 117.61 | 290.39 | 269.8 | 400.24 | 3 |
| (EC→EC)g(1) | 730 | 45.41 | 79.09 | 147.32 | 170.2 | 187.31 | 3 |
| Procyanidin B2 | 578 | 61.04 | 111.94 | 152.08 | 174.53 | 348.35 | 3 |
| Procyanidin B1 | 578 | 11.41 | 83.99 | 186.75 | 175.01 | 276.04 | 3 |
| Procyanidin B4 | 578 | 10.81 | 21.49 | 68.08 | 91.78 | 141.51 | 3 |
| Galloylprocyanidin B4 | 730 | 34.51 | 72.99 | 143.77 | 137.29 | 204.11 | |
| Procyanidin C1 3′-O-gallate | 1018 | 8.82 | 24.19 | 27.92 | 29.96 | 31.35 | |
| Pedunculagin | 784 | 1.83 | 8.78 | 32.76 | 62.05 | 64.32 | 3 |
| Procyanidin C2 | 866 | 7.31 | 13.29 | 22.66 | 30.32 | 44.74 | 3 |
| Procyanidin A6 | 592 | 5.33 | 5.56 | 13.58 | 13.34 | 25.18 | 3 |
| Galloylprocyanidin C2 | 1018 | 0.66 | 3.53 | 4.41 | 5.47 | 5.95 | 6 |
| Procyanidin C1 | 866 | 1.22 | 5.59 | 12.65 | 8.59 | 13.97 | 6 |
| (EC→EC→EC)g(4) | 1018 | 0.77 | 1.45 | 4.26 | 2.89 | 4.33 | |
| (EC→EC→EC)g(5) | 1018 | 0 | 3.12 | 4.4 | 3.02 | 4.09 | 6 |
| (EC→EC→EC)g(3) | 1018 | 0.79 | 2.12 | 3.11 | 5.09 | 3.54 | |
| Kaempferol glycosides | |||||||
| Kaempferol-4′-O-glucoside | 448 | 11.68 | 0 | 0 | 0 | 0 | |
| Kaempferol-3,7-O-dirhamnoside (kaempferitrin) | 578 | 17.14 | 47.25 | 102.78 | 120.58 | 203.56 | 3 |
| Kaempferol-3-O-(6″-acetyl)glucosyl-(1→3)-Galactoside | 652 | 0 | 1.85 | 3.93 | 4.13 | 10.45 | 3 |
| Kaempferol-3-O-rhamnoside (afzelin) (kaempferin) | 432 | 6.81 | 10.54 | 8.72 | 10.5 | 5.71 | |
| Kaempferol-3-O-glucoside (astragalin) | 448 | 11.93 | 134.91 | 284.47 | 385.36 | 497.94 | 3 |
| Kaempferol-3-O-(6″-p-coumaroyl)glucoside (Tiliroside) | 594 | 0 | 1.78 | 2.57 | 1.84 | 2.84 | 6 |
| Kaempferol-3-O-rutinoside (nicotiflorin) | 594 | 0 | 1.1 | 4.16 | 11.91 | 13.58 | 3 |
| Kaempferol-3,7-di-O-glucoside | 610 | 0 | 1.78 | 6.89 | 8.68 | 9.77 | 3 |
| Kaempferol-3-O-(6″-malonyl)glucoside | 534 | 0 | 0 | 1.13 | 1.68 | 1.54 | 3 |
| Kaempferol-3-O-sophoroside | 610 | 0.66 | 0.97 | 1.04 | 0.82 | 0.79 | |
| Quercetin glycosides | |||||||
| Avicularin(quercetin-3-O-α-L-arabinofuranoside) | 434 | 188.08 | 754.09 | 1127.83 | 1422.17 | 1467.53 | 6 |
| Quercetin-3-O-(6″-malonyl)glucoside | 550 | 0 | 0 | 0 | 1.69 | 1.47 | 1 |
| Quercetin-4′-O-glucoside (spiraeoside) | 464 | 26.09 | 56.66 | 109.49 | 164.76 | 204.97 | 3 |
| Quercetin-7-O-glucoside | 464 | 18.68 | 44.89 | 86.32 | 117.17 | 141.43 | 3 |
| Quercetin-3-O-glucoside (isoquercitrin) | 464 | 28.33 | 64.87 | 95.64 | 127.38 | 103.48 | 6 |
| Quercetin-3-O-rhamnoside (quercitrin) | 448 | 607.33 | 1176.8 | 1408.7 | 880.52 | 984.4 | |
| Quercetin-3-O-galactoside (hyperin) | 464 | 34.74 | 94.1 | 135.54 | 162.06 | 138.69 | 6 |
| Quercetin-3-O-(6″-galloyl)galactoside | 616 | 48.03 | 346.3 | 630.69 | 649.71 | 805.03 | 6 |
| Quercetin-3-O-(2″,3″-digalloyl)-glucoside | 768 | 366.24 | 258.37 | 230.15 | 235.74 | 148.62 | 9 |
| Quercetin-7-O-rutinoside | 610 | 6.92 | 1.83 | 4.83 | 5.15 | 6.42 | 4 |
| Quercetin-3-O-xyloside (reynoutrin) | 434 | 223.32 | 865.72 | 1326.27 | 1676.7 | 1694.33 | 6 |
| Quercetin-3-O-(6″-galloyl)glucoside | 616 | 45.85 | 327.28 | 600.16 | 667.84 | 727.71 | 6 |
| Quercetin-3-O-(2″-O-rhamnosyl)galactoside | 610 | 0 | 2.17 | 1.45 | 1.57 | 2.07 | 6 |
| Quercetin-5-O-glucuronide | 478 | 4.35 | 5.91 | 16.55 | 8.05 | 10.25 | |
| Quercetin-3-O-sambubioside | 596 | 1.28 | 0 | 0 | 4.41 | 6.23 | 3 |
| Quercetin-3-O-(6″-p-coumaroyl)glucoside | 610 | 0 | 2.25 | 2.91 | 1.99 | 4.16 | 6 |
| Quercetin-7-O-(6″-malonyl)glucoside | 550 | 1.21 | 4.2 | 4.03 | 3.04 | 2.21 | 7 |
| Quercetin-3-O-glucosyl(1→4)rhamnoside-7-O-rutinoside | 918 | 15.81 | 17.68 | 12.23 | 9.29 | 7.12 | 2 |
| Quercetin-3-O-(6″-malonyl)glucosyl-5-O-glucoside | 712 | 0 | 0.51 | 0.96 | 0.64 | 0.85 | 6 |
| Quercetin-3,4′-O-di-glucoside | 626 | 0 | 1.29 | 5.57 | 5.9 | 8.48 | 3 |
| Quercetin-3-O-(2″-O-glucosyl)glucuronide | 640 | 1.84 | 2.71 | 2.04 | 0 | 0 | 2 |
| Enzyme | KEGG Annotation | Gene ID | P1 | P2 | P3 | P4 | P5 | Model |
|---|---|---|---|---|---|---|---|---|
| PAL | Phenylalanine ammonia-lyase (EC:4.3.1.24) | Jr01G13110 | 11.28 | 15.65 | 1.63 | 15.62 | 50.56 | Blue |
| Jr04G10983 | 3.65 | 6.69 | 5 | 1.31 | 0.35 | Turquoise | ||
| Jr04G10985 | 0.02 | 0.04 | 0.04 | 0.03 | 0.04 | |||
| Jr04G10989 | 2.25 | 1.45 | 1.3 | 1.19 | 0.91 | |||
| Jr04G10993 | 0.55 | 0.39 | 0.28 | 0.32 | 0.23 | |||
| Jr04G10994 | 0.32 | 0.45 | 0.34 | 0.34 | 0.52 | |||
| Jr05G10885 | 0.08 | 0.06 | 0.03 | 0.03 | 0.02 | |||
| Jr02G11537 | 18.64 | 62.06 | 21.79 | 68.69 | 194.65 | Blue | ||
| Jr09G11096 | 12.2 | 15.21 | 18.93 | 24.19 | 13.33 | |||
| C4H | Trans-cinnamate 4-monooxygenase (EC:1.14.14.91) | Jr13G11700 | 103.25 | 44.02 | 2.61 | 4.1 | 1.73 | Turquoise |
| Jr13G11701 | 3.12 | 7.9 | 2.1 | 5.84 | 15.49 | Blue | ||
| Jr14G11389 | 37.81 | 29.37 | 27.36 | 65.56 | 213.3 | Blue | ||
| Jr15G11420 | 0 | 0.05 | 0.01 | 0 | 0 | |||
| Jr16G10525 | 0.28 | 0.39 | 0.32 | 0.35 | 0.18 | |||
| 4CL | 4-coumarate--CoA ligase (EC:6.2.1.12) | Jr10G10964 | 7.67 | 9.57 | 19.39 | 14.77 | 6.07 | Yellow |
| Jr13G10323 | 22.95 | 28.85 | 44.82 | 27 | 56.86 | Blue | ||
| Jr14G10240 | 3.71 | 5.53 | 2.07 | 8.09 | 7.01 | |||
| Jr05G11706 | 1.11 | 3.23 | 1.89 | 9.06 | 47.92 | Blue | ||
| Jr05G12962 | 48.67 | 1.18 | 0.06 | 0 | 0 | Turquoise | ||
| Jr02G11900 | 1.2 | 3.48 | 0.31 | 0.33 | 1.11 | |||
| Jr01G13626 | 3.46 | 5.47 | 0.18 | 0.24 | 0.47 | Turquoise | ||
| Jr02G11899 | 1.98 | 3.33 | 0.01 | 0.3 | 1.1 | |||
| Jr07G11893 | 0.74 | 0.67 | 0.45 | 1.88 | 8.01 | Blue | ||
| Jr07G11894 | 0.12 | 0.01 | 0.01 | 0.03 | 0.09 | |||
| Jr07G11896 | 4.26 | 2.44 | 0.78 | 2.9 | 9.96 | Blue | ||
| Jr11G10952 | 5.01 | 2.19 | 0.93 | 1.95 | 1.19 | Turquoise | ||
| CHS | Chalcone synthase (EC:2.3.1.74) | Jr01G10656 | 39.87 | 211.03 | 179.28 | 478.44 | 965.23 | Blue |
| Jr02G10304 | 28.19 | 98.02 | 46.99 | 187.99 | 330.66 | Blue | ||
| Jr03G13232 | 10.32 | 4.12 | 4.15 | 13.14 | 8.52 | |||
| Jr07G12835 | 2.25 | 0.64 | 0.55 | 1.11 | 5.33 | Blue | ||
| Jr15G11549 | 0.41 | 0.28 | 0.35 | 0.4 | 0.26 | |||
| Jr16G11310 | 0.01 | 0 | 0 | 0 | 0 | |||
| Jr16G11311 | 1.21 | 0.07 | 0.03 | 0.03 | 0 | |||
| CHI | Chalcone isomerase (EC:5.5.1.6) | Jr01G13213 | 7.66 | 3.23 | 3.29 | 12.99 | 17.29 | Blue |
| Jr02G11831 | 0.18 | 0.4 | 0.19 | 0.17 | 0.26 | |||
| F3H | Naringenin 3-dioxygenase (EC:1.14.11.9) | Jr07G12902 | 83.89 | 151.38 | 55.6 | 235.15 | 755.52 | Blue |
| FLS | Flavonol synthase (EC:1.14.20.6) | Jr04G11647 | 0.4 | 0.03 | 0.04 | 0.3 | 0.2 | |
| Jr05G11778 | 0.13 | 0.03 | 0.01 | 0.01 | 0.02 | |||
| F3′H | Flavonoid 3′-monooxygenase (EC:1.14.14.82) | Jr11G12560 | 191.32 | 204.87 | 122.11 | 109.43 | 199.86 | |
| DFR | Dihydroflavonol 4-reductase (EC:1.1.1.2341.1.1.219) | Jr07G11524 | 0.11 | 0.07 | 0.04 | 0.12 | 0.06 | |
| LDOX | Anthocyanidin synthase (EC:1.14.20.4) | Jr11G11451 | 26.04 | 57.19 | 20.47 | 79.76 | 263.77 | Blue |
| ANR | Anthocyanidin reductase (EC:1.3.1.77) | Jr09G12363 | 36.21 | 80.56 | 49.02 | 132.8 | 358.59 | Blue |
| LAR | Leucoanthocyanidin reductase (EC:1.17.1.3) | Jr16G10851 | 0.34 | 0.34 | 0.22 | 0.32 | 0.39 | |
| UGTs | Flavonol-3-O-L-rhamnoside-7-O-glucosyltransferase (EC:2.4.1.-) | Jr09G11788 | 0.05 | 0.63 | 0.69 | 1.81 | 1 | |
| Jr09G11865 | 0.42 | 0.29 | 0.13 | 0.22 | 0.28 | |||
| Flavonol 3-O-glucosyltransferase (EC:2.4.1.91) | Jr04G11320 | 0.1 | 0.01 | 0 | 0.01 | 0.03 | ||
| Jr04G11321 | 1.74 | 0.84 | 0.26 | 0.54 | 1.4 | |||
| Jr04G11325 | 4.67 | 1.76 | 1.18 | 0.57 | 2.51 | |||
| Jr08G11705 | 0.84 | 0.11 | 0.11 | 0.13 | 0.06 |
| Enzyme | KEGG Annotation | Gene ID | P1 | P2 | P3 | P4 | P5 | Model |
|---|---|---|---|---|---|---|---|---|
| MYB111 | myb domain protein 111 | Jr01G10083 | 0.57 | 0.38 | 0.1 | 0.72 | 0.62 | |
| Jr02G10456 | 0 | 0 | 0 | 0.02 | 0 | |||
| Jr02G12175 | 1.89 | 56.08 | 73.12 | 83.57 | 97.4 | Brown | ||
| Jr07G11017 | 0.01 | 0.7 | 1.11 | 0.64 | 0.01 | |||
| Jr08G10063 | 0.83 | 4.93 | 1.53 | 0.35 | 0.01 | Green | ||
| TTG1 | Transducin/WD40 repeat-like | Jr06G12013 | 74.18 | 65.96 | 71.07 | 52.08 | 38.58 | |
| TT8 | Basic helix-loop-helix (bHLH) | Jr07G10705 | 15.61 | 10.83 | 13.3 | 14.57 | 11.47 | |
| Jr08G10317 | 0.03 | 0.01 | 0.01 | 0.01 | 0.01 | |||
| TT2 | Duplicated homeodomain-like superfamily | Jr01G12462 | 0 | 0 | 0.01 | 0.12 | 0 | |
| Jr02G11138 | 0 | 0 | 0 | 0 | 0.01 | |||
| Jr03G11815 | 0.18 | 0.02 | 0 | 0 | 0 | |||
| Jr03G11816 | 0 | 0.03 | 0 | 0 | 0 | |||
| Jr03G11818 | 0.05 | 0.01 | 0 | 0 | 0 | |||
| Jr03G11824 | 0.07 | 0.01 | 0 | 0 | 0 | |||
| Jr03G11832 | 8.17 | 5.69 | 0.23 | 0.47 | 0.21 | Turquoise | ||
| Jr03G11834 | 5.95 | 4.7 | 3.57 | 5.23 | 0.47 | Turquoise | ||
| Jr04G10961 | 0.55 | 0.34 | 0.12 | 0.09 | 0.06 | |||
| Jr08G11467 | 0.01 | 0 | 0 | 0 | 0 | |||
| Jr08G11642 | 0.01 | 0 | 0 | 0 | 0 | |||
| MYB5 | myb domain protein 5 | Jr14G11044 | 12.12 | 7.57 | 5.49 | 10.61 | 24.69 | Blue |
| EGL3 | Basic helix-loop-helix (bHLH) | Jr11G11341 | 0.01 | 0.01 | 0 | 0.01 | 0.01 | |
| Jr12G10985 | 0.33 | 0.11 | 0.09 | 0.08 | 0.12 | |||
| TT1 | C2H2 and C2HC zinc fingers | Jr08G11835 | 5.7 | 1.42 | 0.91 | 1.47 | 6.41 | Blue |
| TT16 | K-box region and MADS-box | Jr05G10903 | 42.69 | 8.38 | 2.85 | 2.4 | 0.3 | Turquoise |
| Jr06G10192 | 3.56 | 0.39 | 0.94 | 0.17 | 0.31 | |||
| TTG2 | WRKY family | Jr09G11240 | 0.09 | 0.24 | 0.09 | 0.08 | 0.17 | |
| Jr10G11149 | 0.23 | 0.28 | 0.4 | 0.2 | 0.22 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, R.; Zhou, Y.; Jin, F.; Zhang, J.; Ji, F.; Bai, Y.; Pei, D. Metabolome and Transcriptome Profiling Unveil the Mechanisms of Polyphenol Synthesis in the Developing Endopleura of Walnut (Juglans regia L.). Int. J. Mol. Sci. 2022, 23, 6623. https://doi.org/10.3390/ijms23126623
Huang R, Zhou Y, Jin F, Zhang J, Ji F, Bai Y, Pei D. Metabolome and Transcriptome Profiling Unveil the Mechanisms of Polyphenol Synthesis in the Developing Endopleura of Walnut (Juglans regia L.). International Journal of Molecular Sciences. 2022; 23(12):6623. https://doi.org/10.3390/ijms23126623
Chicago/Turabian StyleHuang, Ruimin, Ye Zhou, Feng Jin, Junpei Zhang, Feiyang Ji, Yongchao Bai, and Dong Pei. 2022. "Metabolome and Transcriptome Profiling Unveil the Mechanisms of Polyphenol Synthesis in the Developing Endopleura of Walnut (Juglans regia L.)" International Journal of Molecular Sciences 23, no. 12: 6623. https://doi.org/10.3390/ijms23126623
APA StyleHuang, R., Zhou, Y., Jin, F., Zhang, J., Ji, F., Bai, Y., & Pei, D. (2022). Metabolome and Transcriptome Profiling Unveil the Mechanisms of Polyphenol Synthesis in the Developing Endopleura of Walnut (Juglans regia L.). International Journal of Molecular Sciences, 23(12), 6623. https://doi.org/10.3390/ijms23126623






