Pro-Inflammatory Signalling PRRopels Cisplatin-Induced Toxicity
Abstract
1. Introduction
Inflammation and Pro-Inflammatory Signalling
2. Pattern Recognition Receptors (PRRs) in Cisplatin-Induced Toxicities
2.1. Toll-Like Receptors (TLRs)
2.2. NOD-Like Receptors (NLRs) and Inflammasomes
3. Pro-Inflammatory Messengers (Cytokines and Chemokines)
4. Oxidative Stress and Immunologic Regulation
5. Anti-Inflammatory Remedies
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rosenberg, B.; VanCamp, L. The Successful Regression of Large Solid Sarcoma 180 Tumors by Platinum Compounds. Cancer Res. 1970, 30, 1799–1802. [Google Scholar] [PubMed]
- Rosenberg, B.; VanCamp, L.; Krigas, T. Inhibition of Cell Division in Escherichia coli by Electrolysis Products from a Platinum Electrode. Nature 1965, 205, 698–699. [Google Scholar] [CrossRef] [PubMed]
- Peyrone, M. Ueber die Einwirkung des Ammoniaks auf Platinchlorur [On the Action of Ammonia on Platinum Chloride]. Ann. Chemi Pharm. 1845, 51, 1–29. [Google Scholar]
- Kelland, L. The resurgence of platinum-based cancer chemotherapy. Nat. Rev. Cancer 2007, 7, 573–584. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.J.; Thompson, A.J.; Cohen, W. The Mechanism of Action of Antitumor Platinum Compounds. Prog. Nucleic Acid Res. Mol. Biol. 1979, 22, 71–133. [Google Scholar] [PubMed]
- Pinto, A.L.; Lippard, S.J. Binding of the anti-tumor drug cisdiammine dichloroplatinum(ii) (cisplatin) to DNA. Biochim. Biophys. Acta 1985, 780, 167–180. [Google Scholar] [PubMed]
- Eastman, A. The formation, isolation and characterization of DNA adducts produced by anticancer platinum complexes. Pharmacol. Ther. 1987, 34, 155–166. [Google Scholar] [CrossRef]
- Kelland, L.R. New Platinum Antitumor Complexes. Crit. Rev. Oncol. /Hematol. 1993, 5, 91–93. [Google Scholar] [CrossRef]
- Nagasawa, S.; Takahashi, J.; Suzuki, G.; Hideya, Y.; Yamada, K. Why Concurrent CDDP and Radiotherapy Has Synergistic Antitumor Effects: A Review of In Vitro Experimental and Clinical-Based Studies. Int. J. Mol. Sci. 2021, 22, 3140. [Google Scholar] [CrossRef]
- Muzaffar, J.; Bari, S.; Kirtane, K.; Chung, C.H. Recent advances and future directions in clinical management of head and neck squamous cell carcinoma. Cancers 2021, 13, 1–16. [Google Scholar]
- Low, J.J.H.; Ilancheran, A.; Ng, J.S. Malignant Ovarian Germ-Cell Tumours. Best Pract. Res. Clin. Obstet. Gynaecol. 2012, 26, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Bookman, M.A. Optimal primary therapy of ovarian cancer. Ann. Oncol. 2016, 27, i58–i62. [Google Scholar] [CrossRef] [PubMed]
- Einhorn, L.H. Curing Metastatic Testicular Cancer. Proc. Natl. Acad. Sci. USA 2002, 99, 4592–4595. [Google Scholar] [CrossRef] [PubMed]
- de Vries, G.; Rosas-Plaza, X.; van Vugt, M.A.T.M.; Gietema, J.A.; de Jong, S. Testicular Cancer: Determinants of Cisplatin Sensitivity and Novel Therapeutic Opportunities. Cancer Treat. Rev. 2020, 88, 102054. [Google Scholar] [CrossRef]
- Dasari, S.; Tchounwou, P.B. Cisplatin in cancer therapy: Molecular mechanisms of action. Eur. J. Pharmacol. 2014, 740, 364–378. [Google Scholar] [CrossRef]
- Brown, A.; Kumar, S.; Tchounwou, P.B. Cisplatin-Based Chemotherapy of Human Cancers. J. Cancer Sci. Ther. 2019, 11, 97. [Google Scholar]
- Kamalakar, P.; Freeman, A.I.; Higby, D.J.; Wallace, H.J.; Sinks, L.F. Clinical response and toxicity with cisdichlorodiammineplatinum(II) in children. Cancer Treat. Rep. 1977, 61, 835–839. [Google Scholar]
- Meyers, P.A.; Schwartz, C.L.; Krailo, M.; Kleinerman, E.S.; Betcher, D.; Bernstein, M.L.; Conrad, E.; Ferguson, W.; Gebhardt, M.; Goorin, A.M.; et al. Osteosarcoma: A Randomized, Prospective Trial of the Addition of Ifosfamide and/or Muramyl Tripeptide to Cisplatin, Doxorubicin, and High-Dose Methotrexate. J. Clin. Oncol. 2005, 23, 2004–2011. [Google Scholar] [CrossRef]
- Schmid, I.; von Schweinitz, D. Pediatric hepato-cellular carcinoma: Challenges and solutions. J. Hepatocell. Carcinoma 2017, 4, 15–21. [Google Scholar] [CrossRef]
- Cushing, B.; Giller, R.; Cullen, J.W.; Marina, N.M.; Lauer, S.J.; Olson, T.A.; Castleberry, R.P. Randomized comparison of combination chemotherapy with etoposide, bleomycin, and either high-dose or standard-dose cis-platin in children and adolescents with high-risk malignant germ cell tumors: A pediatric intergroup study-Pediatric Oncology Group 9049 and Children’s Cancer Group 8882. J. Clin. Oncol. 2004, 22, 2691–2700. [Google Scholar]
- Perilongo, G.; Maibach, R.; Shafford, E.; Brugieres, L.; Brock, P.; Morland, B.; de Camargo, B.; Zsiros, J.; Roebuck, D.; Zimmermann, A.; et al. Cisplatin vs. Cisplatin-Doxorubicin in Hepatoblastoma. N. Engl. J. Med. 2009, 361, 1662–1670. [Google Scholar] [CrossRef]
- Tajbakhsh, M.; Houghton, P.J.; Morton, C.L.; Kolb, E.A.; Gorlick, R.; Maris, J.M.; Keir, S.T.; Wu, J.; Reynolds, C.P.; Smith, M.A.; et al. Initial testing of cisplatin by the pediatric preclinical testing program. Pediatr. Blood Cancer 2008, 50, 992–1000. [Google Scholar] [CrossRef] [PubMed]
- Kilday, J.-P.; Bouffet, E. Chemotherapy in Childhood Brain Tumors. Curr. Pediatr. Rep. 2013, 2, 38–49. [Google Scholar] [CrossRef][Green Version]
- Shahid, F.; Farooqui, Z.; Khan, F. Cisplatin-induced gastrointestinal toxicity: An update on possible mechanisms and on available gastroprotec-tive strategies. Eur. J. Pharmacol. 2018, 827, 49–57. [Google Scholar] [CrossRef] [PubMed]
- El-Awady, E.-S.E.; Moustafa, Y.M.; Abo-Elmatty, D.M.; Radwan, A. Cisplatin-induced cardiotoxicity: Mechanisms and cardioprotective strategies. Eur. J. Pharmacol. 2011, 650, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Tsang, R.Y.; Al-Fayea, T.; Au, H.-J. Cisplatin Overdose Toxicities and Management. Drug Saf. 2009, 32, 1109–1122. [Google Scholar] [CrossRef]
- Volarevic, V.; Djokovic, B.; Jankovic, M.G.; Harrell, C.R.; Fellabaum, C.; Djonov, V.; Arsenijevic, N. Molecular mechanisms of cis-platin-induced nephrotoxicity: A balance on the knife edge between renoprotection and tumor toxicity. J. Biomed. Sci. 2019, 26, 1–14. [Google Scholar] [CrossRef]
- Breglio, A.M.; Rusheen, A.E.; Shide, E.D.; Fernandez, K.A.; Spielbauer, K.K.; McLachlin, K.M.; Hall, M.D.; Amable, L.; Cunningham, L.L. Cisplatin is retained in the cochlea indefinitely following chemotherapy. Nat. Commun. 2017, 8, 1654. [Google Scholar] [CrossRef]
- Ruggiero, A.; Ferrara, P.; Attinà, G.; Rizzo, D.; Riccardi, R. Renal toxicity and chemotherapy in children with cancer. Br. J. Clin. Pharmacol. 2017, 83, 2605–2614. [Google Scholar] [CrossRef]
- Manohar, S.; Leung, N. Cisplatin nephrotoxici-ty: A review of the literature. J. Nephrol. 2018, 31, 15–25. [Google Scholar] [CrossRef]
- Vincenzi, B.; Armento, G.; Ceruso, M.S.; Catania, G.; Leakos, M.; Santini, D.; Minotti, G.; Tonini, G. Drug-induced hepatotoxicity in cancer patients-implication for treatment. Expert Opin. Drug Saf. 2016, 15, 1219–1238. [Google Scholar] [CrossRef] [PubMed]
- Yaegashi, A.; Yoshida, K.; Suzuki, N.; Shimada, I.; Tani, Y.; Saijo, Y.; Toyama, A. A case of severe hepatotoxicity induced by cisplatin and 5-fluorouracil. Int. Cancer Conf. J. 2019, 9, 24–27. [Google Scholar] [CrossRef] [PubMed]
- Chiorazzi, A.; Semperboni, S.; Marmiroli, P. Current View in Platinum Drug Mechanisms of Peripheral Neurotoxicity. Toxics 2015, 3, 304–321. [Google Scholar] [CrossRef] [PubMed]
- Staff, N.P.; Cavaletti, G.; Islam, B.; Lustberg, M.; Psimaras, D.; Tamburin, S. Platinum-induced peripheral neurotoxicity: From pathogenesis to treatment. J. Peripher. Nerv. Syst. 2019, 24, S26–S39. [Google Scholar] [CrossRef]
- Albers, J.W.; Chaudhry, V.; Cavaletti, G.; Donehower, R.C. Interventions for preventing neuropathy caused by cisplatin and related com-pounds. Cochrane Database Syst. Rev. 2014, 3, 1–79. [Google Scholar]
- Romano, A.; Capozza, M.A.; Mastrangelo, S.; Maurizi, P.; Triarico, S.; Rolesi, R.; Attinà, G.; Fetoni, A.R.; Ruggiero, A. Assessment and Management of Platinum-Related Ototoxicity in Children Treated for Cancer. Cancers 2020, 12, 1266. [Google Scholar] [CrossRef]
- van As, J.W.; van den Berg, H.; van Dalen, E.C. Platinum-induced hearing loss after treatment for childhood cancer. Cochrane Database Syst. Rev. 2016, 8, 1–72. [Google Scholar] [CrossRef]
- Callejo, A.; Sedó-Cabezón, L.; Juan, I.D.; Llorens, J. Cisplatin-Induced Ototoxicity: Effects, Mechanisms and Protection Strategies. Toxics 2015, 3, 268–293. [Google Scholar] [CrossRef]
- Marshall, J.S.; Warrington, R.; Watson, W.; Kim, H.L. An introduction to immunology and immunopathology. Allergy Asthma Clin. Immunol. 2018, 14, 1–10. [Google Scholar] [CrossRef]
- Chaplin, D.D. Overview of the immune response. J. Allergy Clin. Immunol. 2010, 125, S3–S23. [Google Scholar] [CrossRef]
- Takeuchi, O.; Akira, S. Pattern Recognition Receptors and Inflammation. Cell 2010, 140, 805–820. [Google Scholar] [CrossRef]
- Kawasaki, T.; Kawai, T. Toll-like receptor signaling pathways. Front. Immunol. 2014, 5, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Piccinini, A.M.; Midwood, K.S. DAMPening Inflammation by Modulating TLR Signalling. Mediat. Inflamm. 2010, 2010, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Raghavan, B.; Müller, V.; Vogl, T.; Fejer, G.; Tchaptchet, S.; Keck, S.; Kalis, C.; Nielsen, P.J.; Galanos, C.; et al. Crucial role for human Toll-like receptor 4 in the development of contact allergy to nickel. Nat. Immunol. 2010, 11, 814–819. [Google Scholar] [CrossRef] [PubMed]
- Raghavan, B.; Martin, S.F.; Esser, P.; Goebeler, M.; Schmidt, M. Metal allergens nickel and cobalt facilitate TLR4 homodimerization independently of MD2. EMBO Rep. 2012, 13, 1109–1115. [Google Scholar] [CrossRef] [PubMed]
- Oblak, A.; Pohar, J.; Jerala, R. MD-2 determinants of nickel and cobalt-mediated activation of human TLR4. PLoS ONE 2015, 10, e0120583. [Google Scholar] [CrossRef] [PubMed]
- Rachmawati, D.; Bontkes, H.J.; Verstege, M.I.; Muris, J.; von Blomberg, B.M.E.; Scheper, R.J.; van Hoogstraten, I.M.W. Transition metal sensing by Toll-like receptor-4: Next to nickel, cobalt and palladium are potent human dendritic cell stimulators. Contact Dermat. 2013, 68, 331–338. [Google Scholar] [CrossRef]
- Brodsky, I.E.; Monack, D. NLR-mediated control of inflammasome assembly in the host response against bacterial pathogens. Semin. Immunol. 2009, 21, 199–207. [Google Scholar] [CrossRef]
- Lupfer, C.; Kanneganti, T.-D. The expanding role of NLRs in antiviral immunity. Immunol. Rev. 2013, 255, 13–24. [Google Scholar] [CrossRef]
- Platnich, J.M.; Muruve, D.A. NOD-like receptors and inflammasomes: A review of their canonical and non-canonical signaling pathways. Arch. Biochem. Biophys. 2019, 670, 4–14. [Google Scholar] [CrossRef] [PubMed]
- Broz, P.; Dixit, V.M. Inflammasomes: Mechanism of assembly, regulation and signalling. Nat. Rev. Immunol. 2016, 16, 407–420. [Google Scholar] [CrossRef] [PubMed]
- Zheng, D.; Liwinski, T.; Elinav, E. Inflammasome activation and regulation: Toward a better understanding of complex mechanisms. Cell Discov. 2020, 36, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Chiu, Y.H.; MacMillan, J.B.; Chen, Z.J. RNA Polymerase III Detects Cytosolic DNA and Induces Type I Interferons through the RIG-I Pathway. Cell 2009, 138, 576–591. [Google Scholar] [CrossRef] [PubMed]
- Rehwinkel, J.; Gack, M.U. RIG-I-like receptors: Their regulation and roles in RNA sensing. Nat. Rev. Immunol. 2020, 20, 537–551. [Google Scholar] [CrossRef]
- Onomoto, K.; Onoguchi, K.; Yoneyama, M. Regulation of RIG-I-like receptor-mediated signaling: Interaction between host and viral factors. Cell. Mol. Immunol. 2021, 18, 539–555. [Google Scholar] [CrossRef]
- Drouin, M.; Saenz, J.; Chiffoleau, E. C-Type Lectin-Like Receptors: Head or Tail in Cell Death Immunity. Front. Immunol. 2020, 11, 251. [Google Scholar] [CrossRef]
- Brown, G.D.; Willment, J.; Whitehead, L. C-type lectins in immunity and homeostasis. Nat. Rev. Immunol. 2018, 18, 374–389. [Google Scholar] [CrossRef]
- Mogensen, T.H. Pathogen recognition and inflammatory signaling in innate immune defenses. Clin. Microbiol. Rev. 2009, 22, 240–273. [Google Scholar] [CrossRef]
- Shaulian, E.; Karin, M. AP-1 as a regulator of cell life and death. Nat. Cell Biol. 2002, 4, E131–E136. [Google Scholar] [CrossRef]
- Wagner, E.F.; Eferl, R. Fos/AP-1 proteins in bone and the immune system. Immunol. Rev. 2005, 208, 126–140. [Google Scholar] [CrossRef]
- Trop-Steinberg, S.; Azar, Y. AP-1 Expression and its Clinical Relevance in Immune Disorders and Cancer. Am. J. Med. Sci. 2017, 353, 474–483. [Google Scholar] [CrossRef] [PubMed]
- Kucharczak, J.; Simmons, M.J.; Fan, Y.; Gélinas, C. To be, or not to be: NF-κB is the answer-Role of Rel/NF-κB in the regulation of apoptosis. Oncogene 2003, 22, 8961–8982. [Google Scholar] [CrossRef]
- Sun, S.C. The non-canonical NF-κB pathway in immunity and inflammation. Nat. Rev. Immunol. 2017, 17, 545–558. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-κB signaling in inflammation. Signal Transduct. Target. Ther. 2017, 2, e17023. [Google Scholar] [CrossRef] [PubMed]
- Tamura, T.; Yanai, H.; Savitsky, D.; Taniguchi, T. The IRF Family Transcription Factors in Immunity and Oncogenesis. Annu. Rev. Immunol. 2008, 26, 535–584. [Google Scholar] [CrossRef]
- Jefferies, C.A. Regulating IRFs in IFN driven dis-ease. Front. Immunol. 2019, 10, 325. [Google Scholar] [CrossRef]
- Germolec, D.R.; Shipkowski, K.A.; Frawley, R.P.; Evans, E. Markers of Inflammation. In Immunotoxicity Testing—Methods in Molecular Biology; Springer: Berlin/Heidelberg, Germany, 2018; Volume 1803, pp. 57–79. [Google Scholar]
- Turner, M.D.; Nedjai, B.; Hurst, T.; Pennington, D.J. Cytokines and chemokines: At the crossroads of cell signalling and inflammatory disease. Biochim. Biophys. Acta 2014, 1843, 2563–2582. [Google Scholar] [CrossRef]
- Cray, C. Acute phase proteins in animals. Prog. Mol. Biol. Transl. Sci. 2012, 105, 113–150. [Google Scholar]
- Schrödl, W.; Büchler, R.; Wendler, S.; Reinhold, P.; Muckova, P.; Reindl, J.; Rhode, H. Acute phase proteins as promising biomarkers: Perspectives and limitations for human and veterinary medicine. Proteom.-Clin. Appl. 2016, 10, 1077–1092. [Google Scholar] [CrossRef]
- Kawai, T.; Akira, S. TLR signaling. Cell Death Differ. 2006, 13, 816–825. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Ramesh, G.; Uematsu, S.; Akira, S.; Reeves, W.B. TLR4 Signaling Mediates Inflammation and Tissue Injury in Nephrotoxicity. J. Am. Soc. Nephrol. 2008, 19, 923–932. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Fan, Y.; Zhong, J.; Huang, Z.; Huang, T.; Lin, S.; Chen, H. TAK1 mediates excessive autophagy via p38 and ERK in cisplatin-induced acute kidney injury. J. Cell. Mol. Med. 2018, 22, 2908–2921. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; An, C.; Jin, X.; Hu, Z.; Safirstein, R.L.; Wang, Y. TAK1 deficiency attenuates cisplatin-induced acute kidney injury. Am. J. Physiol. -Ren. Physiol. 2020, 318, F209–F215. [Google Scholar] [CrossRef] [PubMed]
- So, H.; Kim, H.; Kim, Y.; Kim, E.; Pae, H.O.; Chung, H.T.; Park, R. Evidence that cisplatin-induced auditory damage is attenuated by downregulation of pro-inflammatory cytokines via Nrf2/HO-1. JARO-J. Assoc. Res. Otolaryngol. 2008, 9, 290–306. [Google Scholar] [CrossRef]
- Oh, G.-S.; Kim, H.-J.; Choi, J.-H.; Shen, A.; Kim, C.-H.; Kim, S.-J.; Shin, S.-R.; Hong, S.-H.; Kim, Y.; Park, C.; et al. Activation of Lipopolysaccharide–TLR4 Signaling Accelerates the Ototoxic Potential of Cisplatin in Mice. J. Immunol. 2010, 186, 1140–1150. [Google Scholar] [CrossRef]
- Ramesh, G.; Zhang, B.; Uematsu, S.; Akira, S.; Reeves, W.B. Endotoxin and cisplatin synergistically induce renal dysfunction and cytokine production in mice. Am. J. Physiol.-Ren. Physiol. 2007, 293, 325–332. [Google Scholar] [CrossRef]
- Babolmorad, G.; Latif, A.; Domingo, I.K.; Pollock, N.M.; Delyea, C.; Rieger, A.M.; Bhavsar, A.P. Toll-like receptor 4 is activated by platinum and contributes to cisplatin-induced ototoxicity. EMBO Rep. 2021, 22, e51280. [Google Scholar] [CrossRef]
- Woller, S.A.; Corr, M.; Yaksh, T.L. Differences in cisplatin-induced mechanical allodynia in male and female mice. Eur. J. Pain 2015, 19, 1476–1485. [Google Scholar] [CrossRef]
- Park, H.J.; Stokes, J.A.; Corr, M.; Yaksh, T.L. Toll-like receptor signaling regulates cisplatin-induced mechanical allodynia in mice. Cancer Chemother. Pharmacol. 2013, 73, 25–34. [Google Scholar] [CrossRef]
- Hutchinson, M.R.; Zhang, Y.; Brown, K.; Coats, B.D.; Shridhar, M.; Sholar, P.W.; Watkins, L.R. Co-expression gene modules involved in cisplatin-induced peripheral neuropathy according to sensitivity, status, and severity. J. Peripher. Nerv. Syst. 2020, 25, 366–376. [Google Scholar]
- Fumagalli, G.; Monza, L.; Cavaletti, G.; Rigolio, R.; Meregalli, C. Neuroinflammatory Process Involved in Different Preclinical Models of Chemotherapy-Induced Peripheral Neuropathy. Front. Immunol. 2021, 11, 626687. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, M.R.; Zhang, Y.; Brown, K.; Coats, B.D.; Shridhar, M.; Sholar, P.W.; Patel, S.J.; Crysdale, N.Y.; Harrison, J.A.; Maier, S.F.; et al. Non-stereoselective reversal of neuropathic pain by naloxone and naltrexone: Involvement of toll-like receptor 4 (TLR4). Eur. J. Neurosci. 2008, 28, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Johnston, I.N.; Tan, M.; Cao, J.; Matsos, A.; Forrest, D.R.; Si, E.; Fardell, J.E.; Hutchinson, M. Ibudilast reduces oxaliplatin-induced tactile allodynia and cognitive impairments in rats. Behav. Brain Res. 2017, 334, 109–118. [Google Scholar] [CrossRef]
- Hutchinson, M.R.; Northcutt, A.L.; Hiranita, T.; Wang, X.; Lewis, S.S.; Thomas, J.; Van Steeg, K.; Kopajtic, T.A.; Loram, L.C.; Sfregola, C.; et al. Opioid Activation of Toll-Like Receptor 4 Contributes to Drug Reinforcement. J. Neurosci. 2012, 32, 11187–11200. [Google Scholar] [CrossRef]
- Oliveros, G.; Wallace, C.H.; Chaudry, O.; Liu, Q.; Qiu, Y.; Xie, L.; Serrano, P. A Repurposing ibudilast to mitigate Alzheimer’s disease by 1 targeting inflammation. Brain 2022. [Google Scholar] [CrossRef]
- Oliviera Nascimento, L.; Massari, P.; Wetzler, L.M. The Role of TLR2 in Infection and Immunity. Front. Immunol. 2012, 3, 79. [Google Scholar] [CrossRef]
- Andrade-Silva, M.; Cenedeze, M.A.; Perandini, L.A.; Felizardo, R.J.F.; Watanabe, I.K.M.; Agudelo, J.S.H.; de Almeida, D.C. TLR2 and TLR4 play opposite role in autophagy associated with cisplatin-induced acute kidney injury. Clin. Sci. 2018, 132, 1725–1739. [Google Scholar] [CrossRef]
- Shen, Q.; Zhang, X.; Li, Q.; Zhang, J.; Lai, H.; Gan, H.; Du, X.; Li, M. TLR2 protects cisplatin-induced acute kidney injury associated with autophagy via PI3K/Akt signaling pathway. J. Cell. Biochem. 2018, 120, 4366–4374. [Google Scholar] [CrossRef]
- Volarevic, V.; Markovic, B.S.; Jankovic, M.G.; Djokovic, B.; Jovicic, N.; Harrell, C.R.; Lukic, M.L. Galectin 3 protects from cispla-tin-induced acute kidney injury by promoting TLR-2-dependent activation of IDO1/Kynurenine pathway in renal DCs. Theranostics 2019, 9, 5976–6001. [Google Scholar] [CrossRef]
- Sallustio, F.; Costantino, V.; Cox, S.N.; Loverre, A.; Divella, C.; Rizzi, M.; Schena, F.P. Human renal stem/progenitor cells repair tubular epithelial cell injury through TLR2-driven inhibin-A and microvesicle-shuttled decorin. Kidney Int. 2013, 83, 392–403. [Google Scholar] [CrossRef] [PubMed]
- Chan, A.J.; Alikhan, M.A.; Odobasic, D.; Gan, P.Y.; Khouri, M.B.; Steinmetz, O.M.; Summers, S.A. Innate IL-17A-producing leuko-cytes promote acute kidney injury via inflammasome and toll-like receptor activation. Am. J. Pathol. 2014, 184, 1411–1418. [Google Scholar] [CrossRef] [PubMed]
- Awadalla, A.; Mahdi, M.R.; Zahran, M.H.; Abdelbaset-Ismail, A.; El-Dosoky, M.; Negm, A. Baicalein and Alpha-Tocopherol Inhibit Toll-like Receptor Pathways in Cisplatin-Induced Nephrotoxicity. Molecules 2022, 27, 2179. [Google Scholar] [CrossRef]
- Zhang, Q.; Raoof, M.; Chen, Y.; Sumi, Y.; Sursal, T.; Junger, W.; Brohi, K.; Itagaki, K.; Hauser, C.J. Circulating mitochondrial DAMPs cause inflammatory responses to injury. Nature 2010, 464, 104–107. [Google Scholar] [CrossRef] [PubMed]
- Alikhan, M.A.; Summers, S.A.; Gan, P.Y.; Chan, A.J.; Khouri, M.B.; Ooi, J.D.; Holdsworth, S.R. Endogenous Toll-like Recep-tor 9 Regulates AKI by Promoting Regulatory T-cell Recruitment. J. Am. Soc. Nephrol. 2016, 27, 706–714. [Google Scholar] [CrossRef]
- Luo, X.; Huh, Y.; Bang, S.; He, Q.; Zhang, L.; Matsuda, M.; Ji, R.-R. Macrophage Toll-like Receptor 9 Contributes to Chemotherapy-Induced Neuropathic Pain in Male Mice. J. Neurosci. 2019, 39, 6848–6864. [Google Scholar] [CrossRef]
- Ezhong, Y.; Ekinio, A.; Esaleh, M. Functions of NOD-Like Receptors in Human Diseases. Front. Immunol. 2013, 4, 333. [Google Scholar] [CrossRef]
- Akira, S.; Uematsu, S.; Takeuchi, O. Pathogen recognition and innate immunity. Cell 2006, 124, 783–801. [Google Scholar] [CrossRef]
- Swanson, K.V.; Deng, M.; Ting, J.P.-Y. The NLRP3 inflammasome: Molecular activation and regulation to therapeutics. Nat. Rev. Immunol. 2019, 19, 477–489. [Google Scholar] [CrossRef]
- Bauernfeind, F.G.; Horvath, G.; Stutz, A.; Alnemri, E.S.; MacDonald, K.; Speert, D.; Fernandes-Alnemri, T.; Wu, J.; Monks, B.G.; Fitzgerald, K.A.; et al. Cutting Edge: NF-κB Activating Pattern Recognition and Cytokine Receptors License NLRP3 Inflammasome Activation by Regulating NLRP3 Expression. J. Immunol. 2009, 183, 787–791. [Google Scholar] [CrossRef]
- Vajjhala, P.R.; Mirams, R.E.; Hill, J.M. Multiple binding sites on the pyrin domain of ASC protein allow self-association and interaction with NLRP3 protein. J. Biol. Chem. 2012, 287, 41732–41743. [Google Scholar] [CrossRef] [PubMed]
- Stutz, A.; Kolbe, C.-C.; Stahl, R.; Horvath, G.L.; Franklin, B.S.; Van Ray, O.; Brinkschulte, R.; Geyer, M.; Meissner, F.; Latz, E. NLRP3 inflammasome assembly is regulated by phosphorylation of the pyrin domain. J. Exp. Med. 2017, 214, 1725–1736. [Google Scholar] [CrossRef] [PubMed]
- Ramesh, G.; Reeves, W.B. Inflammatory cytokines in acute renal failure. Kidney Int. 2004, 66 (Suppl. 91), S56–S61. [Google Scholar] [CrossRef] [PubMed]
- Devarajan, P. Update on Mechanisms of Ischemic Acute Kidney Injury. J. Am. Soc. Nephrol. 2006, 17, 1503–1520. [Google Scholar] [CrossRef]
- Zhang, Y.; Yuan, F.; Cao, X.; Zhai, Z.; Huang, G.; Du, X.; Wang, Y.; Zhang, J.; Huang, Y.; Zhao, J.; et al. P2X7 receptor blockade protects against cisplatin-induced nephrotoxicity in mice by decreasing the activities of inflammasome com-ponents, oxidative stress and caspase-3. Toxicol. Appl. Pharmacol. 2014, 281, 1–10. [Google Scholar] [CrossRef]
- Muñoz-Planillo, R.; Kuffa, P.; Martínez-Colón, G.; Smith, B.L.; Rajendiran, T.M.; Núñez, G. K+ Efflux Is the Common Trigger of NLRP3 Inflammasome Activation by Bacterial Toxins and Particulate Matter. Immunity 2013, 38, 1142–1153. [Google Scholar] [CrossRef]
- Suprenant, A.; Rassendren, F.; Kawashima, E.; North, R.; Buell, G. The Cytolytic P2z Receptor for Extracellular ATP Identified as a P2x Receptor (P2x7). Science 1996, 272, 735–738. [Google Scholar] [CrossRef]
- Yang, S.-K.; Han, Y.-C.; He, J.-R.; Yang, M.; Zhang, W.; Zhan, M.; Li, A.-M.; Li, L.; Song, N.; Liu, Y.-T.; et al. Mitochondria targeted peptide SS-31 prevent on cisplatin-induced acute kidney injury via regulating mitochondrial ROS-NLRP3 pathway. Biomed. Pharmacother. 2020, 130, 110521. [Google Scholar] [CrossRef]
- Gao, H.; Wang, X.; Qu, X.; Zhai, J.; Tao, L.; Zhang, Y.; Song, Y.; Zhang, W. Omeprazole attenuates cisplatin-induced kidney injury through suppression of the TLR4/NF-κB/NLRP3 signaling pathway. Toxicology 2020, 440, 152487. [Google Scholar] [CrossRef]
- Lee, D.W.; Faubel, S.; Edelstein, C.L. A pan caspase inhibitor decreases caspase-1, IL-1αand IL-1β, and protects against necrosis of cisplatin-treated freshly isolated proximal tubules. Ren. Fail. 2014, 37, 144–150. [Google Scholar] [CrossRef]
- Jiang, S.; Zhang, H.; Li, X.; Yi, B.; Huang, L.; Hu, L.; Li, A.; Du, J.; Li, Y.; Zhang, W. Vitamin D/VDR attenuate cisplatin-induced AKI by down-regulating NLRP3/Caspase-1/GSDMD pyroptosis pathway. J. Steroid Biochem. Mol. Biol. 2021, 206, 105789. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Sun, Q.; Tong, Y.; Bi, X.; Chen, L.; Lu, J.; Ding, W. Leonurine attenuates cisplatin nephrotoxicity by suppressing the NLRP3 inflammasome, mitochondrial dysfunction, and endoplasmic reticulum stress. Int. Urol. Nephrol. 2022, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Qu, X.; Gao, H.; Tao, L.; Zhang, Y.; Zhai, J.; Sun, J.; Song, Y.; Zhang, S. Astragaloside IV protects against cis-platin-induced liver and kidney injury via autophagy-mediated inhibition of NLRP3 in rats. J. Toxicol. Sci. 2019, 44, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Lin, Q.; Shao, X.; Mou, S.; Gu, L.; Wang, L.; Zhang, Z.; Shen, J.; Zhou, Y.; Qi, C.; et al. NLRP3 inflammasome inhibition attenuates cisplatin-induced renal fibrosis by decreasing oxidative stress and inflammation. Exp. Cell Res. 2019, 383, 111488. [Google Scholar] [CrossRef]
- Kim, H.-J.; Lee, D.W.; Ravichandran, K.; Keys, D.O.; Akcay, A.; Nguyen, Q.; He, Z.; Jani, A.; Ljubanovic, D.; Edelstein, C.L. NLRP3 Inflammasome Knockout Mice Are Protected against Ischemic but Not Cisplatin-Induced Acute Kidney Injury. J. Pharmacol. Exp. Ther. 2013, 346, 465–472. [Google Scholar] [CrossRef]
- Faubel, S.; Ljubanovic, D.; Reznikov, L.; Somerset, H.; Dinarello, C.A.; Edelstein, C.L. Caspase-1-deficient mice are protected against cisplatin-induced apoptosis and acute tubular necrosis. Kidney Int. 2004, 66, 2202–2213. [Google Scholar] [CrossRef]
- Vilaysane, A.; Chun, J.; Seamone, M.E.; Wang, W.; Chin, R.; Hirota, S.; Li, Y.; Clark, S.A.; Tschopp, J.; Trpkov, K.; et al. The NLRP3 Inflammasome Promotes Renal Inflammation and Contributes to CKD. J. Am. Soc. Nephrol. 2010, 21, 1732–1744. [Google Scholar] [CrossRef]
- Mulay, S.R.; Kulkarni, O.P.; Rupanagudi, K.V.; Migliorini, A.; Darisipudi, M.N.; Vilaysane, A.; Muruve, D.; Shi, Y.; Munro, F.; Liapis, H.; et al. Calcium oxalate crystals induce renal inflammation by NLRP3-mediated IL-1β secretion. J. Clin. Investig. 2012, 123, 236–246. [Google Scholar] [CrossRef]
- Xiong, J.; Wang, Y.; Shao, N.; Gao, P.; Tang, H.; Su, H.; Zhang, C.; Meng, X.F. The Expression and Significance of NLRP3 Inflammasome in Patients with Primary Glomerular Diseases. Kidney Blood Press. Res. 2015, 40, 344–354. [Google Scholar] [CrossRef]
- Eisa, N.H.; El-Sherbiny, M.; El-Magd, N.F.A. Betulin alleviates cisplatin-induced hepatic injury in rats: Targeting apoptosis and Nek7-independent NLRP3 inflammasome pathways. Int. Immunopharmacol. 2021, 99, 107925. [Google Scholar] [CrossRef]
- Shi, F.; Zhang, L.; Liu, X.; Wang, Y. Knock-down of microRNA miR-556-5p increases cisplatin-sensitivity in non-small cell lung cancer (NSCLC) via activating NLR family pyrin domain containing 3 (NLRP3)-mediated pyroptotic cell death. Bioengineered 2021, 12, 6332–6342. [Google Scholar] [CrossRef] [PubMed]
- So, H.; Kim, H.; Lee, J.H.; Park, C.; Kim, Y.; Kim, E.; Kim, J.K.; Yun, K.J.; Lee, K.M.; Lee, H.Y.; et al. Cisplatin cytotoxicity of auditory cells requires secretions of proinflammatory cytokines via activation of ERK and NF-κB. JARO-J. Assoc. Res. Otolaryngol. 2007, 8, 338–355. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Ramesh, G.; Norbury, C.; Reeves, W. Cisplatin-induced nephrotoxicity is mediated by tumor necrosis factor-α produced by renal parenchymal cells. Kidney Int. 2007, 72, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Faubel, S.; Lewis, E.C.; Reznikov, L.; Ljubanovic, D.; Hoke, T.S.; Somerset, H.; Oh, D.; Lu, L.; Klein, C.L.; Dinarello, C.A.; et al. Cisplatin-induced acute renal failure is associated with an increase in the cytokines interleukin (IL)-1β, IL-18, IL-6, and neutrophil in-filtration in the kidney. J. Pharmacol. Exp. Ther. 2007, 322, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Oh, G.S.; Lee, J.H.; Lyu, A.R.; Ji, H.M.; Lee, S.H.; Song, J.; Park, S.J.; You, Y.O.; Sul, J.D.; et al. Cisplatin ototoxicity involves cy-tokines and STAT6 signaling network. Cell Res. 2011, 21, 944–956. [Google Scholar] [CrossRef]
- Sommer, C.; Kress, M. Recent findings on how proinflammatory cytokines cause pain: Peripheral mechanisms in inflammatory and neuropathic hyperalgesia. Neurosci. Lett. 2004, 361, 184–187. [Google Scholar] [CrossRef]
- Wang, X.-M.; Lehky, T.J.; Brell, J.M.; Dorsey, S.G. Discovering cytokines as targets for chemotherapy-induced painful peripheral neuropathy. Cytokine 2012, 59, 3–9. [Google Scholar] [CrossRef]
- Rose-John, S.; Waetzig, G.H.; Cheller, J.; Grötzinger, J.; Seegert, D. The IL-6/sIL-6R complex as a novel target for therapeutic approaches. Expert Opin. Ther. Targets 2007, 11, 613–624. [Google Scholar] [CrossRef]
- Jones, S.A.; Jenkins, B.J. Recent insights into targeting the IL-6 cytokine family in inflammatory diseases and cancer. Nat. Rev. Immunol. 2018, 18, 773–789. [Google Scholar] [CrossRef]
- Didion, S.P. Cellular and Oxidative Mechanisms Associated with Interleukin-6 Signaling in the Vasculature. Int. J. Mol. Sci. 2017, 18, 2563. [Google Scholar] [CrossRef]
- Wassmann, S.; Stumpf, M.; Strehlow, K.; Schmid, A.; Schiefffer, B.; Böhm, M.; Nickenig, G. Interleukin-6 Induces Oxidative Stress and Endothelial Dysfunction by Overexpression of the Angiotensin II Type 1 Receptor. Circ. Res. 2004, 94, 534–541. [Google Scholar] [CrossRef] [PubMed]
- Zelová, H.; Hošek, J. TNF-α signalling and in-flammation: Interactions between old acquaintances. Inflamm. Res. 2013, 62, 641–651. [Google Scholar] [CrossRef]
- Tsuruya, K.; Ninomiya, T.; Tokumoto, M.; Hirakawa, M.; Masutani, K.; Taniguchi, M.; Fukuda, K.; Kanai, H.; Kishihara, K.; Hirakata, H.; et al. Direct involvement of the receptor-mediated apoptotic pathways in cisplatin-induced renal tubular cell death. Kidney Int. 2003, 63, 72–82. [Google Scholar] [CrossRef] [PubMed]
- Ramesh, G.; Reeves, W. TNFR2-mediated apoptosis and necrosis in cisplatin-induced acute renal failure. Am. J. Physiol. Physiol. 2003, 285, F610–F618. [Google Scholar] [CrossRef] [PubMed]
- Ramesh, G.; Brian Reeves, W. TNF-α mediates chemokine and cytokine expression and renal injury in cisplatin nephrotoxicity. J. Clin. Investig. 2002, 110, 835–842. [Google Scholar] [CrossRef]
- Kaur, T.; Mukherjea, D.; Sheehan, K.; Jajoo, S.; Rybak, L.P.; Ramkumar, V. Short interfering RNA against STAT1 attenuates cisplatin-induced ototoxicity in the rat by suppressing inflammation. Cell Death Dis. 2011, 2, e180. [Google Scholar] [CrossRef]
- Digby, J.L.M.; Vanichapol, T.; Przepiorski, A.; Davidson, A.J.; Sander, V. Evaluation of cisplatin-induced injury in human kidney organoids. Am. J. Physiol. Physiol. 2020, 318, F971–F978. [Google Scholar] [CrossRef]
- Li, J.; Tang, Y.; Tang, P.M.K.; Lv, J.; Huang, X.R.; Carlsson-Skwirut, C.; Da Costa, L.; Aspesi, A.; Frohlich, S.; Szcześniak, P.; et al. Blocking Macrophage Migration Inhibitory Factor Protects Against Cisplatin-Induced Acute Kidney Injury in Mice. Mol. Ther. 2018, 26, 2523–2532. [Google Scholar] [CrossRef]
- Liu, P.; Li, X.; Lv, W.; Xu, Z. Inhibition of CXCL1-CXCR2 axis ameliorates cisplatin-induced acute kidney injury by mediating inflammatory response. Biomed. Pharmacother. 2019, 122, 109693. [Google Scholar] [CrossRef]
- Dupre, T.; Doll, M.A.; Shah, P.P.; Sharp, C.N.; Siow, D.; Megyesi, J.; Shayman, J.; Bielawska, A.; Bielawski, J.; Beverly, L.J.; et al. Inhibiting glucosylceramide synthase exacerbates cisplatin-induced acute kidney injury. J. Lipid Res. 2017, 58, 1439–1452. [Google Scholar] [CrossRef]
- Ozkok, A.; Ravichandran, K.; Wang, Q.; Ljuba-novic, D.; Edelstein, C.L. NF-κB transcriptional inhibition ameliorates cisplatin-induced acute kidney injury (AKI). Toxicol. Lett. 2016, 240, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Feng, Y.; Chen, X.; Wang, G.; Nawaz, I.; Hu, L.; Zhao, L. Paracrine action of human placental trophoblast cells attenuates cisplatin-induced acute kidney injury. Life Sci. 2019, 230, 45–54. [Google Scholar] [CrossRef]
- Brandolini, L.; D’Angelo, M.; Antonosante, A.; Cimini, A.; Allegretti, M. Chemokine Signaling in Chemotherapy-Induced Neuropathic Pain. Int. J. Mol. Sci. 2019, 20, 2904. [Google Scholar] [CrossRef] [PubMed]
- Kawai, H.; Sato, W.; Yuzawa, Y.; Kosugi, T.; Matsuo, S.; Takei, Y.; Kadomatsu, K.; Muramatsu, T. Lack of the Growth Factor Midkine Enhances Survival against Cisplatin-Induced Renal Damage. Am. J. Pathol. 2004, 165, 1603–1612. [Google Scholar] [CrossRef]
- Schmitt, N.C.; Rubel, E.W.; Nathanson, N.M. Cisplatin-Induced Hair Cell Death Requires STAT1 and Is Attenuated by Epigallocatechin Gallate. J. Neurosci. 2009, 29, 3843–3851. [Google Scholar] [CrossRef]
- Wei, J.; Chen, X.; Li, Q.; Chen, J.; Khan, N.; Wang, B.; Cheng, J.W.; Gordon, J.R.; Li, F. ELR-CXC chemokine antagonism and cisplatin co-treatment additively reduce H22 hepatoma tumor progression and ameliorate cis-platin-induced nephrotoxicity. Oncol. Rep. 2014, 31, 1599–1604. [Google Scholar] [CrossRef][Green Version]
- Stevanović, M.; Lazic, A.; Popović, J.; Paunesku, T.; Woloschak, G.E. Insights into platinum-induced peripheral neuropathy–current perspective. Neural Regen. Res. 2020, 15, 1623. [Google Scholar] [CrossRef]
- Deng, J.; Kohda, Y.; Chiao, H.; Wang, Y.; Hu, X.; Hewitt, S.M.; Miyaji, T.; Mcleroy, P.; Nibhanupudy, B.; Li, S.; et al. Interleukin-10 inhibits ischemic and cisplatin-induced acute renal injury. Kidney Int. 2001, 60, 2118–2128. [Google Scholar] [CrossRef]
- Moon, S.K.; Woo, J.I.; Lim, D.J. Involvement of TNF-α and IFN-γ in Inflammation-Mediated Cochlear Injury. Ann. Otol. Rhinol. Laryngol. 2019, 128, 8S–15S. [Google Scholar] [CrossRef]
- Tadagavadi, R.K.; Reeves, W.B. Endogenous IL-10 Attenuates Cisplatin Nephrotoxicity: Role of Dendritic Cells. J. Immunol. 2010, 185, 4904–4911. [Google Scholar] [CrossRef] [PubMed]
- Kimura, A.; Ishida, Y.; Inagaki, M.; Nakamura, Y.; Sanke, T.; Mukaida, N.; Kondo, T. Interferon-γ is protective in cis-platin-induced renal injury by enhancing autophagic flux. Kidney Int. 2012, 82, 1093–1104. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.E.; Nakagawa, T.; Kita, T.; Kim, T.S.; Iguchi, F.; Endo, T.; Shiga, A.; Lee, S.H.; Ito, J. Mechanisms of Apoptosis Induced by Cisplatin in Marginal Cells in Mouse Stria Vascularis. ORL 2004, 66, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Rybak, L.P.; Mukherjea, D.; Jajoo, S.; Ramkumar, V. Cisplatin Ototoxicity and Protection: Clinical and Experimental Studies. Tohoku J. Exp. Med. 2009, 219, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Gentilin, E.; Simoni, E.; Candito, M.; Cazzador, D.; Astolfi, L. Cisplatin-Induced Ototoxicity: Updates on Molecular Targets. Trends Mol. Med. 2019, 25, 1123–1132. [Google Scholar] [CrossRef]
- Rybak, L.P.; Whitworth, C.; Somani, S. Application of Antioxidants and Other Agents to Prevent Cisplatin Ototoxicity from the Departments of Surgery. Laryngoscope 1999, 109, 1740–1744. [Google Scholar] [CrossRef]
- Kong, X.; Thimmulappa, R.; Kombairaju, P.; Biswal, S. NADPH Oxidase-Dependent Reactive Oxygen Species Mediate Amplified TLR4 Signaling and Sepsis-Induced Mortality in Nrf2-Deficient Mice. J. Immunol. 2010, 185, 569–577. [Google Scholar] [CrossRef]
- Hato, S.V.; Khong, A.; de Vries, I.J.M.; Lesterhuis, W.J. Molecular Pathways: The Immunogenic Effects of Platinum-Based Chemotherapeutics. Clin. Cancer Res. 2014, 20, 2831–2837. [Google Scholar] [CrossRef] [PubMed]
- Xue, D.-F.; Pan, S.-T.; Huang, G.; Qiu, J.-X. ROS enhances the cytotoxicity of cisplatin by inducing apoptosis and autophagy in tongue squamous cell carcinoma cells. Int. J. Biochem. Cell Biol. 2020, 122, 105732. [Google Scholar] [CrossRef]
- Kim, H.J.; Lee, J.H.; Kim, S.J.; Oh, G.S.; Moon, H.D.; Kwon, K.B.; Park, C.; Park, B.H.; Lee, H.K.; Chung, S.Y.; et al. Roles of NADPH oxidases in cis-platin-induced reactive oxygen species generation and ototoxicity. J. Neurosci. 2010, 30, 3933–3946. [Google Scholar] [CrossRef]
- Ohlemiller, K.K.; Wright, J.S.; Dugan, L.L. Early Elevation of Cochlear Reactive Oxygen Species following Noise Exposure. Audiol. Neurotol. 1999, 4, 229–236. [Google Scholar] [CrossRef]
- Henderson, D.; McFadden, S.L.; Liu, C.C.; Hight, N.; Zheng, X.Y. The role of antioxidants in protection from impulse noise. Ann. N. Y. Acad. Sci. 1999, 884, 368–380. [Google Scholar] [CrossRef]
- Kim, S.-J.; Park, C.; Han, A.L.; Youn, M.-J.; Lee, J.-H.; Kim, Y.; Kim, E.-S.; Kim, H.-J.; Kim, J.-K.; Lee, H.-K.; et al. Ebselen attenuates cisplatin-induced ROS generation through Nrf2 activation in auditory cells. Heart Res. 2009, 251, 70–82. [Google Scholar] [CrossRef]
- Zou, J.; Wang, S.-P.; Wang, Y.-T.; Wan, J.-B. Regulation of the NLRP3 inflammasome with natural products against chemical-induced liver injury. Pharmacol. Res. 2020, 164, 105388. [Google Scholar] [CrossRef]
- Choi, Y.-M.; Kim, H.; Shim, W.; Anwar, M.A.; Kwon, J.-W.; Kwon, H.-K.; Kim, H.J.; Jeong, H.; Kim, H.M.; Hwang, D.; et al. Mechanism of Cisplatin-Induced Cytotoxicity Is Correlated to Impaired Metabolism Due to Mitochondrial ROS Generation. PLoS ONE 2015, 10, e0135083. [Google Scholar] [CrossRef]
- De Sá Coutinho, D.; Pacheco, M.T.; Frozza, R.L.; Bernardi, A. Anti-Inflammatory Effects of Resveratrol: Mechanistic Insights. Int. J. Mol. Sci. 2018, 19, 1812. [Google Scholar] [CrossRef]
- Salehi, B.; Mishra, A.P.; Nigam, M.; Sener, B.; Kilic, M.; Sharifi-Rad, M.; Fokou, P.V.T.; Martins, N.; Sharifi-Rad, J. Resveratrol: A Double-Edged Sword in Health Benefits. Biomedicines 2018, 6, 91. [Google Scholar] [CrossRef]
- Meng, T.; Xiao, D.; Muhammed, A.; Deng, J.; Chen, L.; He, J. Anti-Inflammatory Action and Mechanisms of Resveratrol. Molecules 2021, 26, 229. [Google Scholar] [CrossRef]
- Do Amaral, C.L.; Francescato, H.D.C.; Coimbra, T.M.; Costa, R.S.; Darin, J.D.C.; Antunes, L.M.G.; Bianchi, M.D.L.P. Resveratrol attenuates cis-platin-induced nephrotoxicity in rats. Arch. Toxicol. 2008, 82, 363–370. [Google Scholar] [CrossRef]
- Olgun, Y.; Kırkım, G.; Kolatan, E.; Kıray, M.; Bagrıyanık, A.; Olgun, A.; Kızmazoglu, D.C.; Ellıdokuz, H.; Serbetcıoglu, B.; Altun, Z.; et al. Friend or foe? Effect of oral resveratrol on cisplatin ototoxicity. Laryngoscope 2013, 124, 760–766. [Google Scholar] [CrossRef]
- Ibrahim, M.A.; Albahlol, I.A.; Wani, F.A.; Tammam, A.A.-E.; Kelleni, M.T.; Sayeed, M.U.; El-Fadeal, N.M.A.; Mohamed, A.A. Resveratrol protects against cisplatin-induced ovarian and uterine toxicity in female rats by attenuating oxidative stress, inflammation and apoptosis: Resveratrol protects against cisplatin toxicity in rats. Chem. Interact. 2021, 338, 109402. [Google Scholar] [CrossRef]
- Lee, C.H.; Kim, K.W.; Lee, S.M.; Kim, S.Y. Dose-Dependent Effects of Resveratrol on Cisplatin-Induced Hearing Loss. Int. J. Mol. Sci. 2020, 22, 113. [Google Scholar] [CrossRef]
- Darwish, M.A.; Abo-Youssef, A.M.; Khalaf, M.M.; Abo-Saif, A.A.; Saleh, I.G.; Abdelghany, T.M. Resveratrol influences platinum pharmacokinetics: A novel mechanism in protection against cisplatin-induced nephrotoxicity. Toxicol. Lett. 2018, 290, 73–82. [Google Scholar] [CrossRef]
- Kim, D.H.; Jung, Y.J.; Lee, J.E.; Lee, A.E.; Kang, K.P.; Lee, S.; Park, S.K.; Han, M.K.; Lee, S.Y.; Ramkumar, K.M.; et al. SIRT1 activation by resveratrol ameliorates cisplatin-induced renal injury through deacetylation of p53. Am. J. Physiol.-Ren. Physiol. 2011, 301, 427–435. [Google Scholar] [CrossRef]
- Liu, Y.; Wu, H.; Zhang, F.; Yang, J.; He, J. Resveratrol upregulates miR-455-5p to antagonize cisplatin ototoxicity via modulating the PTEN–PI3K–AKT axis. Biochem. Cell Biol. 2021, 99, 385–395. [Google Scholar] [CrossRef]
- Hao, Q.; Xiao, X.; Zhen, J.; Feng, J.; Song, C.; Jiang, B.; Hu, Z. Resveratrol attenuates acute kidney injury by inhibiting death receptor-mediated apoptotic pathways in a cisplatin-induced rat model. Mol. Med. Rep. 2016, 14, 3683–3689. [Google Scholar] [CrossRef]
- Said, R.S.; Mantawy, E.M.; El-Demerdash, E. Mechanistic perspective of protective effects of resveratrol against cisplatin-induced ovarian injury in rats: Emphasis on anti-inflammatory and anti-apoptotic effects. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2019, 392, 1225–1238. [Google Scholar] [CrossRef]
- Hamad, R.; Jayakumar, C.; Ranganathan, P.; Mohamed, R.; El-Hamamy, M.M.I.; Dessouki, A.A.; Ibrahim, A.; Ramesh, G.; El-Hamamy, M.M.I. Honey feeding protects kidney against cisplatin nephrotoxicity through suppression of inflammation. Clin. Exp. Pharmacol. Physiol. 2015, 42, 843–848. [Google Scholar] [CrossRef]
- Choi, J.; Kim, S.H.; Rah, Y.C.; Chae, S.W.; Lee, J.D.; Lee, B.D.; Park, M.K. Effects of caffeic acid on cisplatin-induced hair cell damage in HEI-OC1 auditory cells. Int. J. Pediatr. Otorhinolaryngol. 2014, 78, 2198–2204. [Google Scholar] [CrossRef]
- Huang, H.; Shen, Z.; Geng, Q.; Wu, Z.; Shi, P.; Miao, X. Protective effect of Schisandra chinensis bee pollen extract on liver and kidney injury induced by cisplatin in rats. Biomed. Pharmacother. 2017, 95, 1765–1776. [Google Scholar] [CrossRef]
- Tender, T.; Rahangdale, R.R.; Balireddy, S.; Nampoothiri, M.; Sharma, K.K.; Chandrashekar, H.R. Melittin, a honeybee venom de-rived peptide for the treatment of chemotherapy-induced peripheral neuropathy. Med. Oncol. 2021, 38, 52. [Google Scholar] [CrossRef]
- Kim, H.; Hong, J.Y.; Jeon, W.-J.; Baek, S.H.; Ha, I.-H. Bee Venom Melittin Protects against Cisplatin-Induced Acute Kidney Injury in Mice via the Regulation of M2 Macrophage Activation. Toxins 2020, 12, 574. [Google Scholar] [CrossRef]
- Kim, H.; Lee, G.; Park, S.; Chung, H.-S.; Lee, H.; Kim, J.-Y.; Nam, S.; Kim, S.K.; Bae, H. Bee Venom Mitigates Cisplatin-Induced Nephrotoxicity by Regulating CD4+CD25+Foxp3+Regulatory T Cells in Mice. Evid.-Based Complement. Altern. Med. 2013, 2013, 879845. [Google Scholar] [CrossRef]
- Rezaee, R.; Momtazi, A.A.; Monemi, A.; Sahebkar, A. Curcumin: A potentially powerful tool to reverse cisplatin-induced toxicity. Pharmacol. Res. 2017, 117, 218–227. [Google Scholar] [CrossRef]
- Mercantepe, F.; Mercantepe, T.; Topcu, A.; Yılmaz, A.; Tumkaya, L. Protective effects of amifostine, curcumin, and melatonin against cispla-tin-induced acute kidney injury. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2018, 391, 915–931. [Google Scholar] [CrossRef]
- El-Gizawy, M.M.; Hosny, E.N.; Mourad, H.H.; Abd-El Razik, A.N. Curcumin nanoparticles ameliorate hepatotoxicity and nephrotoxicity in-duced by cisplatin in rats. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2020, 393, 1941–1953. [Google Scholar] [CrossRef]
- Khadrawy, Y.A.; El-Gizawy, M.M.; Sorour, S.M.; Sawie, H.G.; Hosny, E.N. Effect of curcumin nanoparticles on the cisplatin-induced neurotoxicity in rat. Drug Chem. Toxicol. 2018, 42, 194–202. [Google Scholar] [CrossRef]
- Ueki, M.; Ueno, M.; Morishita, J.; Maekawa, N. Curcumin ameliorates cisplatin-induced nephrotoxicity by inhibiting renal inflammation in mice. J. Biosci. Bioeng. 2012, 115, 547–551. [Google Scholar] [CrossRef]
- Topcu-Tarladacalisir, Y.; Sapmaz-Metin, M.; Karaca, T. Curcumin counteracts cisplatin-induced nephrotoxicity by preventing renal tubular cell apoptosis. Ren. Fail. 2016, 38, 1741–1748. [Google Scholar] [CrossRef]
- Ali, B.H.; Abdelrahman, A.; Al Suleimani, Y.; Manoj, P.; Ali, H.; Nemmar, A.; Al Za’Abi, M. Effect of concomitant treatment of curcumin and melatonin on cisplatin-induced nephrotoxicity in rats. Biomed. Pharmacother. 2020, 131, 110761. [Google Scholar] [CrossRef]
- El-Kader, M.A.; Taha, R.I. Comparative nephroprotective effects of curcumin and etoricoxib against cisplatin-induced acute kidney injury in rats. Acta Histochem. 2020, 122, 151534. [Google Scholar] [CrossRef]
- al Fayi, M.; Otifi, H.; Alshyarba, M.; Dera, A.A.; Rajagopalan, P. Thymoquinone and curcumin combination protects cisplatin-induced kidney inju-ry, nephrotoxicity by attenuating NFκB, KIM-1 and ameliorating Nrf2/HO-1 signalling. J. Drug Target. 2020, 28, 913–922. [Google Scholar] [CrossRef]
- Fetoni, A.R.; Paciello, F.; Mezzogori, D.; Rolesi, R.; Eramo, S.L.M.; Paludetti, G.; Troiani, D. Molecular targets for anti-cancer redox chemotherapy and cisplatin-induced ototoxicity: The role of curcumin on pSTAT3 and Nrf-2 signalling. Br. J. Cancer 2015, 113, 1434–1444. [Google Scholar] [CrossRef]
- Kumar, P.; Sulakhiya, K.; Barua, C.C.; Mundhe, N. TNF-α, IL-6 and IL-10 expressions, responsible for disparity in action of curcumin against cispla-tin-induced nephrotoxicity in rats. Mol. Cell. Biochem. 2017, 431, 113–122. [Google Scholar] [CrossRef]
- Borse, V.; Aameri, R.F.H.A.; Sheehan, K.; Sheth, S.; Kaur, T.; Mukherjea, D.; Tupal, S.; Lowy, M.; Ghosh, S.; Dhukwa, A.; et al. Epigallocatechin-3-gallate, a proto-typic chemopreventative agent for protection against cisplatin-based ototoxicity. Cell Death Dis. 2017, 8, e2921. [Google Scholar] [CrossRef]
- Yang, D.; Cao, G.; Ba, X.; Jiang, H. Epigallocat-echin-3-O-gallate promotes extracellular matrix and inhibits inflammation in IL-1β stimulated chon-drocytes by the PTEN/miRNA-29b pathway. Pharm. Biol. 2022, 60, 589–599. [Google Scholar] [CrossRef]
- Fatima, S.; Suhail, N.; Alrashed, M.; Wasi, S.; Aljaser, F.S.; AlSubki, R.A.; Alsharidah, A.S.; Banu, N. Epigallocatechin gallate and coen-zyme Q10 attenuate cisplatin-induced hepatotoxicity in rats via targeting mitochondrial stress and apoptosis. J. Biochem. Mol. Toxicol. 2021, 35, e22701. [Google Scholar] [CrossRef]
- Arafa, M.H.; Atteia, H.H. Protective Role of Epigallocatechin Gallate in a Rat Model of Cisplatin-Induced Cerebral Inflammation and Oxidative Damage: Impact of Modulating NF-κB and Nrf2. Neurotox. Res. 2020, 37, 380–396. [Google Scholar] [CrossRef]
- Bae, K.H.; Tan, S.; Yamashita, A.; Ang, W.X.; Gao, S.J.; Wang, S.; Chung, J.E.; Kurisawa, M. Hyaluronic acid-green tea cate-chin micellar nanocomplexes: Fail-safe cisplatin nanomedicine for the treatment of ovarian cancer without off-target toxicity. Biomaterials 2017, 148, 41–53. [Google Scholar] [CrossRef]
- Malik, S.; Suchal, K.; Bhatia, J.; Gamad, N.; Dinda, A.K.; Gupta, Y.K.; Arya, D.S. Molecular mechanisms underlying attenuation of cisplatin-induced acute kidney injury by epicatechin gallate. Lab. Investig. 2016, 96, 853–861. [Google Scholar] [CrossRef]
- Gündoğdu, R.; Erkan, M.; Kökoğlu, K. Assessment of the effectiveness of quercetin on cisplatin-induced ototoxicity in rats. J. Int. Adv. Otol. 2019, 15, 229–236. [Google Scholar] [CrossRef]
- Lee, S.K.; Oh, K.H.; Chung, A.Y.; Park, H.C.; Lee, S.H.; Kwon, S.Y.; Choi, J. Protective role of quercetin against cisplatin-induced hair cell damage in zebrafish embryos. Hum. Exp. Toxicol. 2015, 34, 1043–1052. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-González, P.D.; López-Hernández, F.J.; Dueñas, M.; Prieto, M.; Sanchez-Lopez, E.; Thomale, J.; Ruiz-Ortega, M.; López-Novoa, J.M.; Morales, A.I. Differential effect of quercetin on cisplatin-induced toxicity in kidney and tumor tissues. Food Chem. Toxicol. 2017, 107, 226–236. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Reyes, D.; Casanova, A.G.; González-Paramás, A.M.; Martín, Á.; Santos-Buelga, C.; Morales, A.I.; López-Hernández, F.J.; Prieto, M. Protective Effect of Quercetin 3-O-Glucuronide against Cisplatin Cytotoxicity in Renal Tubular Cells. Molecules 2022, 27, 1319. [Google Scholar] [CrossRef] [PubMed]
- Azevedo, M.I.; Pereira, A.F.; Nogueira, R.B.; Rolim, F.E.; Brito, G.A.; Wong, D.V.T.; Lima-Júnior, R.C.; Ribeiro, R.D.A.; Vale, M.L. The Antioxidant Effects of the Flavonoids Rutin and Quercetin Inhibit Oxaliplatin-Induced Chronic Painful Peripheral Neuropathy. Mol. Pain 2013, 9, 53. [Google Scholar] [CrossRef]
- Warren, C.A.; Paulhill, K.J.; Davidson, L.A.; Lupton, J.R.; Taddeo, S.S.; Hong, M.Y.; Carroll, R.J.; Chapkin, R.S.; Turner, N.D. Quercetin May Suppress Rat Aberrant Crypt Foci Formation by Suppressing Inflammatory Mediators That Influence Proliferation and Apoptosis. J. Nutr. 2008, 139, 101–105. [Google Scholar] [CrossRef]
- Sahu, B.D.; Kumar, J.M.; Sistla, R. Baicalein, a bioflavonoid, prevents cisplatin- Induced acute kidney injury by up- regulating antioxidant defenses and down- Regulating the MAPKs and NF-κB Pathways. PLoS ONE 2015, 10, e0134139. [Google Scholar] [CrossRef]
- Huynh, D.L.; Ngau, T.H.; Nguyen, N.H.; Tran, G.B.; Nguyen, C.T. Potential therapeutic and pharmacological effects of Wogonin: An updated re-view. Mol. Biol. Rep. 2020, 47, 9779–9789. [Google Scholar] [CrossRef]
- Meng, X.M.; Li, H.D.; Wu, W.F.; Tang, P.M.K.; Ren, G.L.; Li, X.F.; Yang, Y.; Xu, T.; Ma, T.T. Wogonin protects against cis-platin-induced acute kidney injury by targeting RIPK1-mediated necroptosis. Lab. Investig. 2018, 98, 79–94. [Google Scholar] [CrossRef]
- Chi, Y.S.; Cheon, B.S.; Kim, H.P. Effect of wogonin, a plant flavone from Scutellaria radix, on the suppression of cyclooxygenase-2 and the in-duction of inducible nitric oxide synthase in lipopolysaccharide-treated RAW 264.7 cell. Biochem. Pharmacol. 2001, 61, 1195–1203. [Google Scholar]
- Man, Q.; Deng, Y.; Li, P.; Ma, J.; Yang, Z.; Yang, X.; Zhou, Y.; Yan, X. Licorice Ameliorates Cisplatin-Induced Hepatotoxicity Through Antiapoptosis, Antioxidative Stress, Anti-Inflammation, and Acceleration of Metabolism. Front. Pharmacol. 2020, 11, 563750. [Google Scholar] [CrossRef]
- Richard, S.A. Exploring the Pivotal Immunomodulatory and Anti-Inflammatory Potentials of Glycyrrhizic and Glycyrrhetinic Acids. Mediat. Inflamm. 2021, 2021, 6699560. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-H.; Chen, A.-Z.; Yen, G.-C. Protective Effects of Glycyrrhizic Acid and 18β-Glycyrrhetinic Acid against Cisplatin-Induced Nephrotoxicity in BALB/c Mice. J. Agric. Food Chem. 2015, 63, 1200–1209. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wei, W.; Li, Y.; Huang, J.; Ci, X. Hesperetin relieves cisplatin-induced acute kidney injury by mitigating oxidative stress, inflammation and apoptosis. Chem.-Biol. Interact. 2019, 308, 269–278. [Google Scholar] [CrossRef]
- Vasaikar, N.; Mahajan, U.; Patil, K.R.; Suchal, K.; Patil, C.R.; Ojha, S.; Goyal, S.N. D-pinitol attenuates cisplatin-induced nephrotoxicity in rats: Impact on pro-inflammatory cytokines. Chem. Interact. 2018, 290, 6–11. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Chen, Z.; Yang, L.; Ding, L. Sappanone A Protects Against Inflammation, Oxidative Stress and Apoptosis in Cerebral Ischemia-Reperfusion Injury by Alleviating Endoplasmic Reticulum Stress. Inflammation 2021, 44, 934–945. [Google Scholar] [CrossRef]
- Kang, L.; Zhao, H.; Chen, C.; Zhang, X.; Xu, M.; Duan, H. Sappanone A protects mice against cisplatin-induced kidney injury. Int. Immunopharmacol. 2016, 38, 246–251. [Google Scholar] [CrossRef]
- Lee, S.; Choi, S.Y.; Choo, Y.Y.; Kim, O.; Tran, P.T.; Dao, C.T.; Min, B.S.; Lee, J.H. Sappanone A exhibits anti-inflammatory effects via modulation of Nrf2 and NF-κB. Int. Immunopharmacol. 2015, 28, 328–336. [Google Scholar] [CrossRef]
- Li, F.; Yao, Y.; Huang, H.; Hao, H.; Ying, M. Xanthohumol attenuates cisplatin-induced nephrotoxicity through inhibiting NF-κB and activating Nrf2 signaling pathways. Int. Immunopharmacol. 2018, 61, 277–282. [Google Scholar] [CrossRef]
- Ma, X.; Yan, L.; Zhu, Q.; Shao, F. Puerarin attenuates cisplatin-induced rat nephrotoxicity: The involvement of TLR4/NF-KB signaling pathway. PLoS ONE 2017, 12, e0171612. [Google Scholar] [CrossRef]
- Xu, B.; Li, J.; Chen, X.; Kou, M. Puerarin atten-uates cisplatin-induced apoptosis of hair cells through the mitochondrial apoptotic pathway. Biochim. Biophys. Acta-Mol. Cell Res. 1869, 4, 119208. [Google Scholar]
- Zhou, Y.-D.; Hou, J.-G.; Yang, G.; Jiang, S.; Chen, C.; Wang, Z.; Liu, Y.-Y.; Ren, S.; Li, W. Icariin ameliorates cisplatin-induced cytotoxicity in human embryonic kidney 293 cells by suppressing ROS-mediated PI3K/Akt pathway. Biomed. Pharmacother. 2018, 109, 2309–2317. [Google Scholar] [CrossRef] [PubMed]
- Ma, P.; Zhang, S.; Su, X.; Qiu, G.; Wu, Z. Protective effects of icariin on cisplatin-induced acute renal injury in mice. Am. J. Transl. Res. 2015, 7, 2105–2114. [Google Scholar] [PubMed]
- Sung, M.J.; Kim, D.H.; Jung, Y.J.; Kang, K.P.; Lee, A.S.; Lee, S.; Kim, W.; Davaatseren, M.; Hwang, J.-T.; Kim, H.-J.; et al. Genistein protects the kidney from cisplatin-induced injury. Kidney Int. 2008, 74, 1538–1547. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Toplu, Y.; Varan, E.; Sapmaz, E.; Özhan, O.; Parlakpınar, H.; Polat, A. The effect of genistein on cisplatin induced ototoxicity and oxidative stress. Braz. J. Otorhinolaryngol. 2021, 88, 105–111. [Google Scholar] [CrossRef]
- Huang, Y.-C.; Tsai, M.-S.; Hsieh, P.-C.; Shih, J.-H.; Wang, T.-S.; Wang, Y.-C.; Lin, T.-H.; Wang, S.-H. Galangin ameliorates cisplatin-induced nephrotoxicity by attenuating oxidative stress, inflammation and cell death in mice through inhibition of ERK and NF-kappaB signaling. Toxicol. Appl. Pharmacol. 2017, 329, 128–139. [Google Scholar] [CrossRef]
- Tomar, A.; Vasisth, S.; Khan, S.I.; Malik, S.; Nag, T.C.; Arya, D.S.; Bhatia, J. Galangin ameliorates cisplatin induced nephrotoxicity in vivo by modulation of oxidative stress, apoptosis and inflammation through interplay of MAPK signaling cascade. Phytomedicine 2017, 34, 154–161. [Google Scholar] [CrossRef]
- Domitrović, R.; Cvijanović, O.; Pugel, E.P.; Zagorac, G.B.; Mahmutefendić, H.; Škoda, M. Luteolin ameliorates cisplatin-induced nephrotoxicity in mice through inhibition of platinum accumulation, inflammation and apoptosis in the kidney. Toxicology 2013, 310, 115–123. [Google Scholar] [CrossRef]
- Li, W.; Yan, M.H.; Liu, Y.; Liu, Z.; Wang, Z.; Chen, C.; Zhang, J.; Sun, Y.S. Ginsenoside Rg5 ameliorates cisplatin-induced nephrotoxicity in mice through inhibition of inflammation, oxidative stress, and apoptosis. Nutrients 2016, 8, 566. [Google Scholar] [CrossRef]
- Zhang, W.; Hou, J.; Yan, X.; Leng, J.; Li, R.; Zhang, J.; Xing, J.; Chen, C.; Wang, Z.; Li, W. Platycodon grandiflorum saponins ameliorate cisplatin-induced acute nephrotoxicity through the NF-κB-mediated inflammation and PI3K/Akt/apoptosis signaling pathways. Nutrients 2018, 10, 1328. [Google Scholar] [CrossRef]
- Wan, Y.; Wang, J.; Xu, J.F.; Tang, F.; Chen, L.; Tan, Y.-Z.; Rao, C.L.; Ao, H.; Peng, C. Panax ginseng and its ginsenosides: Potential candidates for the prevention and treatment of chemotherapy-induced side effects. J. Ginseng Res. 2021, 45, 617–630. [Google Scholar] [CrossRef]
- Olgun, Y.; Kirkim, G.; Altun, Z.; Aktaş, S.; Kolatan, E.; Kiray, M.; Bağriyanik, A.; Olgun, A.; Kizmazoğlu, D.C.; Özoğul, C.; et al. Protective effect of Korean Red Ginseng on cisplatin ototoxicity: Is it effective enough? J. Int. Adv. Otol. 2016, 12, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Kwak, H.J.; Kim, D.S.; Choi, H.M.; Sim, J.E.; Kim, S.H.; Um, J.Y.; Hong, S.H. Protective mechanism of Korean Red Ginseng in cisplatin-induced ototoxicity through attenuation of nuclear factor-κB and caspase-1 activation. Mol. Med. Rep. 2015, 12, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Dang, C.; Kang, H.; Dai, Z.; Lin, S.; Guan, H.; Liu, X.; Wang, X.; Hui, W. Saikosaponin-D reduces cisplatin-induced nephrotoxicity by repressing ROS-mediated activation of MAPK and NF-κB signalling pathways. Int. Immunopharmacol. 2015, 28, 399–408. [Google Scholar] [CrossRef]
- Meng, J.; Qiu, S.; Zhang, L.; You, M.; Xing, H.; Zhu, J. Berberine Alleviate Cisplatin-Induced Peripheral Neuropathy by Modulating Inflammation Signal via TRPV1. Front. Pharmacol. 2022, 12, 774795. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-H.; Baek, J.-I.; Lee, I.-K.; Kim, U.-K.; Kim, Y.-R.; Lee, K.-Y. Protective effect of berberine chloride against cisplatin-induced ototoxicity. Genes Genom. 2021, 44, 1–7. [Google Scholar] [CrossRef]
- Hagar, H.; El Medany, A.; Salam, R.; El Medany, G.; Nayal, O.A. Betaine supplementation mitigates cisplatin-induced nephrotoxicity by abrogation of oxidative/nitrosative stress and suppression of inflammation and apoptosis in rats. Exp. Toxicol. Pathol. 2015, 67, 133–141. [Google Scholar] [CrossRef]
- al Za’abi, M.; Ali, H.; al Sabahi, M.; Ali, B.H. The salutary action of melatonin and betaine, given singly or concomitantly, on cisplatin-induced nephrotoxicity in mice. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2021, 394, 1693–1701. [Google Scholar] [CrossRef]
- Michel, H.E.; Menze, E.T. Tetramethylpyra-zine guards against cisplatin-induced nephrotoxicity in rats through inhibiting HMGB1/TLR4/NF-κB and activating Nrf2 and PPAR-γ signaling pathways. Eur. J. Pharmacol. 2019, 857, 172422. [Google Scholar] [CrossRef]
- Bayram, A.; Kaya, A.; Akay, E.; Hıra, İ.; Özcan, İ. The protective role of tetramethylpyrazine against cisplatin-induced ototoxicity. Int. J. Pediatric Otorhinolaryngol. 2017, 94, 1–7. [Google Scholar] [CrossRef]
- Yan, W.; Xu, Y.; Yuan, Y.; Tian, L.; Wang, Q.; Xie, Y.; Shao, X.; Zhang, M.; Ni, Z.; Mou, S. Renoprotective mechanisms of Astragaloside IV in cisplatin-induced acute kidney injury. Free Radic. Res. 2017, 51, 669–683. [Google Scholar] [CrossRef]
- Singh, T.G.; Singh, H.P.; Singh, R. Sinapic acid attenuates cisplatin-induced nephrotoxicity through peroxisome proliferator-activated receptor gamma agonism in rats. J. Pharm. Bioallied Sci. 2020, 12, 146–154. [Google Scholar] [CrossRef] [PubMed]
- Ansari, M.A. Sinapic acid modulates Nrf2/HO-1 signaling pathway in cisplatin-induced nephrotoxicity in rats. Biomed. Pharmacother. 2017, 93, 646–653. [Google Scholar] [CrossRef] [PubMed]
- Fouad, A.A.; Al-Melhim, W.N. Vanillin mitigates the adverse impact of cisplatin and methotrexate on rat kidneys. Hum. Exp. Toxicol. 2018, 37, 937–943. [Google Scholar] [CrossRef] [PubMed]
- Younis, N.N.; Elsherbiny, N.M.; Shaheen, M.A.; Elseweidy, M.M. Modulation of NADPH oxidase and Nrf2/HO-1 pathway by vanillin in cisplatin-induced nephrotoxicity in rats. J. Pharm. Pharmacol. 2020, 72, 1546–1555. [Google Scholar] [CrossRef]
- Potočnjak, I.; Marinić, J.; Batičić, L.; Šimić, L.; Broznić, D.; Domitrović, R. Aucubin administered by either oral or parenteral route protects against cisplatin-induced acute kidney injury in mice. Food Chem. Toxicol. 2020, 142, 111472. [Google Scholar] [CrossRef]
- Kadir, A.; Siddiqui, R.A.; Mirza, T. Nephroprotective role of eugenol against cisplatin-induced acute kidney injury in mice Dectection of Xeno-tropic murine leukemia virus related virus (XMRV) in Prostate Adenocarcinoma Biopsy Specimens View project Protective effects of different compounds on acetaminophen-induced hepatic damage in mice and rats. View Proj. Pak. J. Pharm. Sci. 2020, 33, 1281–1287. [Google Scholar] [CrossRef]
- Yogalakshmi, B.; Viswanathan, P.; Anuradha, C.V. Investigation of antioxidant, anti-inflammatory and DNA-protective properties of eugenol in thi-oacetamide-induced liver injury in rats. Toxicology 2010, 268, 204–212. [Google Scholar] [CrossRef]
- Sakat, M.S.; Kilic, K.; Akdemir, F.N.E.; Yildirim, S.; Eser, G.; Kiziltunc, A. The effectiveness of eugenol against cisplatin-induced ototoxicity. Braz. J. Otorhinolaryngol. 2018, 85, 766–773. [Google Scholar] [CrossRef]
- Akdemir, F.N.E.; Yildirim, S.; Kandemir, F.M.; Aksu, E.H.; Guler, M.C.; Ozmen, H.K.; Kucukler, S.; Eser, G. The antiapoptotic and antioxidant effects of eugenol against cisplatin-induced testicular damage in the experimental model. Andrologia 2019, 51, e13353. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, Y.; Li, B.; Ding, P.; Jin, D.; Hou, S.; Cai, X.; Sheng, X. The effect of monotropein on al-leviating cisplatin-induced acute kidney injury by inhibiting oxidative damage, inflammation and apoptosis. Biomed. Pharmacother. 2020, 129, 110408. [Google Scholar] [CrossRef]
- Shin, J.-S.; Yun, K.-J.; Chung, K.-S.; Seo, K.-H.; Park, H.-J.; Cho, Y.-W.; Baek, N.-I.; Jang, D.; Lee, K.-T. Monotropein isolated from the roots of Morinda officinalis ameliorates proinflammatory mediators in RAW 264.7 macrophages and dextran sulfate sodium (DSS)-induced colitis via NF-κB inactivation. Food Chem. Toxicol. 2013, 53, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.B.; Wang, J.Y.; Zhang, Y.L.; Hu, Y.G.; Yue, Z.S.; Zeng, L.R.; Zheng, W.J.; Hou, Q.; Yan, S.G.; Quan, R.F. Mechanisms underlying the antiapoptotic and anti-inflammatory effects of monotropein in hydrogen peroxide-treated osteo-blasts. Mol. Med. Rep. 2016, 14, 5377–5384. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fan, X.; Wei, W.; Huang, J.; Liu, X.; Ci, X. Isoorientin Attenuates Cisplatin-Induced Nephrotoxicity Through the Inhibition of Oxidative Stress and Apoptosis via Activating the SIRT1/SIRT6/Nrf-2 Pathway. Front. Pharmacol. 2020, 11, 264. [Google Scholar] [CrossRef] [PubMed]
- Muthuraman, A.; Sood, S.; Singla, S.K.; Singh, A.; Singh, A.; Singh, J. Ameliorative effect of flunarizine in cisplatin-induced acute renal failure via mitochondrial permeability transition pore in-activation in rats. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2011, 383, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Muthuraman, A.; Singla, S.K.; Peters, A. Exploring the potential of flunarizine for cisplatin-induced painful uremic neuropathy in rats. Int. Neurourol. J. 2011, 15, 127–134. [Google Scholar] [CrossRef][Green Version]
- So, H.-S.; Kim, H.-J.; Lee, J.-H.; Park, S.-Y.; Park, C.; Kim, Y.-H.; Kim, J.-K.; Lee, K.-M.; Kim, K.-S.; Chung, S.-Y.; et al. Flunarizine induces Nrf2-mediated transcriptional activation of heme oxygenase-1 in protection of auditory cells from cisplatin. Cell Death Differ. 2006, 13, 1763–1775. [Google Scholar] [CrossRef]
- Kaur, T.; Borse, V.; Sheth, S.; Sheehan, K.; Ghosh, S.; Tupal, S.; Jajoo, S.; Mukherjea, D.; Rybak, L.P.; Ramkumar, V. Adenosine A1 receptor protects against cisplatin ototoxicity by suppressing the NOX3/STAT1 inflammatory pathway in the cochlea. J. Neurosci. 2016, 36, 3962–3977. [Google Scholar] [CrossRef]
- Nishida, T.; Tsubota, M.; Kawaishi, Y.; Yamanishi, H.; Kamitani, N.; Sekiguchi, F.; Ishikura, H.; Liu, K.; Nishibori, M.; Kawabata, A. Involvement of high mobility group box 1 in the development and maintenance of chemotherapy-induced peripheral neuropathy in rats. Toxicology 2016, 365, 48–58. [Google Scholar] [CrossRef]
- Tsubota, M.; Fukuda, R.; Hayashi, Y.; Miyazaki, T.; Ueda, S.; Yamashita, R.; Koike, N.; Sekiguchi, F.; Wake, H.; Wakatsuki, S.; et al. Role of non-macrophage cell-derived HMGB1 in oxaliplatin-induced peripheral neuropathy and its prevention by the throm-bin/thrombomodulin system in rodents: Negative impact of anticoagulants. J. Neuroinflamm. 2019, 16, 199. [Google Scholar] [CrossRef]
- Sekiguchi, F.; Kawabata, A. Role of HMGB1 in Chemotherapy-Induced Peripheral Neuropathy. Int. J. Mol. Sci. 2020, 22, 367. [Google Scholar] [CrossRef]
- Miyagi, A.; Kawashiri, T.; Shimizu, S.; Shigematsu, N.; Kobayashi, D.; Shimazoe, T. Dimethyl Fumarate Attenuates Oxaliplatin-Induced Peripheral Neuropathy without Affecting the Anti-tumor Activity of Oxaliplatin in Rodents. Biol. Pharm. Bull. 2019, 42, 638–644. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, A.; Koike, N.; Murakami, T.; Suzuki, K. Dimethyl fumarate ameliorates cisplatin-induced renal tubulointerstitial lesions. J. Toxicol. Pathol. 2019, 32, 79–89. [Google Scholar] [CrossRef] [PubMed]
- US National Library of Medicine. Cisplatin-Induced Ototoxicity. Clinical Trials. 2022. Available online: https://www.clinicaltrials.gov/ct2/results?cond=Cisplatin-in-duced+ototoxicity&age_v=&gndr=&type=&rslt=&phase=0&phase=1&phase=2&phase=3&Search=Apply (accessed on 24 May 2022).
- US National Library of Medicine. Cisplatin-Induced Nephrotoxicity. Clinical Trials. 2022. Available online: https://www.clinicaltrials.gov/ct2/results?cond=cisplatin-in-duced+nephrotoxicity&age_v=&gndr=&type=&rslt=&phase=0&phase=1&phase=2&phase=3&Search=Apply (accessed on 24 May 2022).
- Kawashiri, T.; Miyagi, A.; Shimizu, S.; Shigematsu, N.; Kobayashi, D.; Shimazoe, T. Dimethyl fumarate ameliorates chemotherapy agent-induced neurotoxicity in vitro. J. Pharmacol. Sci. 2018, 137, 202–211. [Google Scholar] [CrossRef] [PubMed]
- Tenório, M.C.d.S.; Graciliano, N.G.; Moura, F.; de Oliveira, A.C.M.; Goulart, M.O.F. N-Acetylcysteine (NAC): Impacts on Human Health. Antioxidants 2021, 10, 967. [Google Scholar] [CrossRef]
- Wang, W.; Chen, E.; Ding, X.; Lu, P.; Chen, J.; Ma, P.; Lu, L. N-acetylcysteine protect inner hair cells from cisplatin by alleviated celluar oxidative stress and apoptosis. Toxicol. Vitr. 2022, 81, 105354. [Google Scholar] [CrossRef]
- Riga, M.G.; Chelis, L.; Kakolyris, S.; Papadopoulos, S.; Stathakidou, S.; Chamalidou, E.; Xenidis, N.; Amarantidis, K.; Dimopoulos, P.; Danielides, V. Transtympanic injections of N-acetylcysteine for the prevention of cisplatin-induced ototoxicity: A feasible method with promising efficacy. Am. J. Clin. Oncol. Cancer Clin. Trials 2013, 36, 1–6. [Google Scholar] [CrossRef]
- Abdel-Wahab, W.M.; Moussa, F.I.; Saad, N.A. Synergistic protective effect of N-acetylcysteine and taurine against cisplatin-induced nephrotoxicity in rats. Drug Des. Dev. Ther. 2017, 11, 901–908. [Google Scholar] [CrossRef]
- Chen, C.-H.; Huang, C.-Y.; Lin, H.-Y.H.; Wang, M.-C.; Chang, C.-Y.; Cheng, Y.-F. Association of Sodium Thiosulfate with Risk of Ototoxic Effects From Platinum-Based Chemotherapy. JAMA Netw. Open 2021, 4, e2118895. [Google Scholar] [CrossRef]
- Zhang, M.Y.; Dugbartey, G.J.; Juriasingani, S.; Sener, A. Hydrogen sulfide metabolite, sodium thiosulfate: Clinical applications and underlying molecular mechanisms. Int. J. Mol. Sci. 2021, 22, 6452. [Google Scholar] [CrossRef]
- Diamond, B.J.; Shiflett, S.C.; Feiwel, N.; Matheis, R.J.; Noskin, O.; Richards, J.A.; Schoenberger, N.E. Ginkgo biloba extract: Mechanisms and clinical indications. Arch. Phys. Med. Rehabil. 2000, 81, 668–678. [Google Scholar] [CrossRef]
- Huang, X.; Whitworth, C.A.; Rybak, L.P. Ginkgo Biloba Extract (EGb 761) Protects Against Cisplatin-Induced Ototoxicity in Rats. Otol. Neurotol. 2007, 28, 828–833. [Google Scholar] [CrossRef] [PubMed]
- Cakil, B.; Basar, F.S.; Atmaca, S.; Cengel, S.K.; Tekat, A.; Tanyeri, Y. The protective effect of Ginkgo biloba extract against experimental cisplatin ototoxicity: Animal research using distortion product otoacoustic emissions. J. Laryngol. Otol. 2012, 126, 1097–1101. [Google Scholar] [CrossRef] [PubMed]
- Esen, E.; Özdoğan, F.; Gürgen, S.G.; Özel, H.E.; Baser, S.; Genҫ, S.; Selҫuk, A. Ginkgo biloba and lycopene are effective on cisplatin induced ototoxicity? J. Int. Adv. Otol. 2018, 14, 23–27. [Google Scholar] [CrossRef] [PubMed]
- Grosser, N.; Erdmann, K.; Hemmerle, A.; Berndt, G.; Hinkelmann, U.; Smith, G.; Schröder, H. Rosuvastatin upregulates the antioxidant defense protein heme oxygenase-1. Biochem. Biophys. Res. Commun. 2004, 325, 871–876. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Luo, L.; Shen, Q.; Shi, G.; Mohammed, A.; Ni, S.; Wu, X. Rosuvastatin improves myocardial hypertrophy after hemodynamic pressure overload via regulating the crosstalk of Nrf2/ARE and TGF-β/smads pathways in rat heart. Eur. J. Pharmacol. 2018, 820, 173–182. [Google Scholar] [CrossRef]
- Mostafa, R.E.; Saleh, D.O.; Mansour, D.F. Cisplatin-induced nephrotoxicity in rats: Modulatory role of simvastatin and rosuvastatin against apoptosis and inflammation. J. Appl. Pharm. Sci. 2018, 8, 43–50. [Google Scholar]
- Kavalipati, N.; Shah, J.; Ramakrishan, A.; Vasnawala, H. Pleiotropic effects of statins. Indian J. Endocrinol. Metab. 2015, 19, 554–562. [Google Scholar]
- Bedi, O.; Dhawan, V.; Sharma, P.L.; Kumar, P. Pleiotropic effects of statins: New therapeutic targets in drug design. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2016, 389, 695–712. [Google Scholar] [CrossRef]
- Maheshwari, R.A.; Sailor, G.U.; Patel, L.; Balaraman, R. Amelioration of cisplatin-induced nephrotoxicity by statins. Indian J. Pharmacol. 2013, 45, 354–358. [Google Scholar] [CrossRef]
- Kim, S.Y.; Lee, C.H.; Min, C.; Yoo, D.M.; Choi, H.G. Association between statin medication and hearing impairment in a national health screening cohort. Sci. Rep. 2021, 11, 14388. [Google Scholar] [CrossRef]
- Marshak, T.; Steiner, M.; Kaminer, M.; Levy, L.; Shupak, A. Prevention of cisplatin-induced hearing loss by intratympanic dexamethasone: A ran-domized controlled study. Otolaryngol.-Head Neck Surg. 2014, 150, 983–990. [Google Scholar] [CrossRef] [PubMed]
- Waissbluth, S.; Salehi, P.; He, X.; Daniel, S.J. Systemic dexamethasone for the prevention of cisplatin-induced ototoxicity. Eur. Arch. Oto-Rhino-Laryngol. 2012, 270, 1597–1605. [Google Scholar] [CrossRef] [PubMed]
- Simsek, G.; Taş, B.M.; Muluk, N.B.; Azman, M.; Kılıç, R. Comparison of the protective efficacy between intratympanic dexamethasone and resveratrol treatments against cisplatin-induced ototoxicity: An experimental study. Eur. Arch. Oto-Rhino-Laryngol. 2019, 276, 3287–3293. [Google Scholar] [CrossRef] [PubMed]
- Özel, H.E.; Özdoğan, F.; Gürgen, S.G.; Esen, E.; Genҫ, S.; Selҫuk, A. Comparison of the protective effects of intratympanic dexamethasone and methylprednisolone against cisplatin-induced ototoxicity. J. Laryngol. Otol. 2016, 130, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Haake, S.M.; Dinh, C.T.; Chen, S.; Eshraghi, A.A.; van de Water, T.R. Dexamethasone protects auditory hair cells against TNFα-initiated apopto-sis via activation of PI3K/Akt and NFκB signaling. Hear. Res. 2009, 255, 22–32. [Google Scholar] [CrossRef] [PubMed]
- Dinh, C.T.; Chen, S.; Bas, E.; Dinh, J.; Goncalves, S.; Telischi, F.; Angeli, S.; Eshraghi, A.A.; Van De Water, T. Dexamethasone Protects Against Apoptotic Cell Death of Cisplatin-exposed Auditory Hair Cells In Vitro. Otol. Neurotol. 2015, 36, 1566–1571. [Google Scholar] [CrossRef]
- Fernandez, R.; Harrop-Jones, A.; Wang, X.; Dellamary, L.; LeBel, C.; Piu, F. The sustained-exposure dexamethasone formulation OTO-104 offers effective protection against cisplatin-induced hearing loss. Audiol. Neurotol. 2016, 21, 22–29. [Google Scholar] [CrossRef]
- Ahmed, L.A.; Shehata, N.I.; Abdelkader, N.F.; Khattab, M.M. Tempol, a superoxide dismutase mimetic agent, ameliorates cisplatin-induced ne-phrotoxicity through alleviation of mitochondrial dysfunction in mice. PLoS ONE 2014, 9, e115983. [Google Scholar] [CrossRef]
- Youn, C.K.; Kim, J.; Jo, E.-R.; Oh, J.; Do, N.Y.; Cho, S.I. Protective Effect of Tempol against Cisplatin-Induced Ototoxicity. Int. J. Mol. Sci. 2016, 17, 1931. [Google Scholar] [CrossRef]
- Ewees, M.G.; Messiha, B.A.S.; Abdel-Bakky, M.S.; Bayoumi, A.M.A.; Abo-Saif, A.A. Tempol, a superoxide dismutase mimetic agent, reduces cisplatin-induced nephrotoxicity in rats. Drug Chem. Toxicol. 2018, 42, 657–664. [Google Scholar] [CrossRef]
- Afjal, M.A.; Goswami, P.; Ahmad, S.; Dabeer, S.; Akhter, J.; Salman, M.; Mangla, A.; Raisuddin, S. Tempol (4-hydroxy tempo) protects mice from cisplatin-induced acute kidney injury via modulation of expression of aquaporins and kidney injury molecule-1. Drug Chem. Toxicol. 2022, 45, 1355–1363. [Google Scholar] [CrossRef] [PubMed]
- Surai, P.F. Silymarin as a Natural Antioxidant: An Overview of the Current Evidence and Perspectives. Antioxidants 2015, 4, 204–247. [Google Scholar] [CrossRef] [PubMed]
- Fanoudi, S.; Alavi, M.S.; Karimi, G.; Hosseinzadeh, H. Milk thistle (Silybum Marianum) as an antidote or a protective agent against natural or chemical toxicities: A review. Drug Chem. Toxicol. 2018, 43, 240–254. [Google Scholar] [CrossRef] [PubMed]
- Esmaeil, N.; Anaraki, S.B.; Gharagozloo, M.; Moayedi, B. Silymarin impacts on immune system as an immunomodulator: One key for many locks. Int. Immunopharmacol. 2017, 50, 194–201. [Google Scholar] [CrossRef]
- Hayes, D.M.; Cvitkovic, E.; Golbey, R.B.; Scheiner, E.; Helson, L.; Krakoff, I.H. High Dose Cis-Platinum Di-ammine Dichloride Amelioration of Renal Toxicity by Mannitol Diuresis. Cancer 1977, 39, 1372–1381. [Google Scholar] [CrossRef]
- Williams, R.P.; Ferlas, B.W.; Morales, P.C.; Kurtzweil, A.J. Mannitol for the prevention of cisplatin-induced nephrotoxicity: A retrospective comparison of hydration plus mannitol versus hydration alone in inpatient and outpatient regimens at a large academic medical center. J. Oncol. Pharm. Pract. 2016, 23, 422–428. [Google Scholar] [CrossRef]
- Ruggiero, A.; Ariano, A.; Triarico, S.; Capozza, M.A.; Romano, A.; Maurizi, P.; Mastrangelo, S.; Attiná, G. Cisplatin-induced nephrotoxi-city in children: What is the best protective strategy? J. Oncol. Pharm. Pract. 2021, 27, 180–186. [Google Scholar] [CrossRef]
- Sainamthip, P.; Saichaemchan, S.; Satirapoj, B.; Prasongsook, N. The Effect of Intravenous Mannitol Combined with Normal Saline in Preventing Cisplatin-Induced Nephrotoxicity: A Randomized, Double-Blind, Placebo-Controlled Trial. JCO Glob. Oncol. 2022, 8, 275. [Google Scholar] [CrossRef]
- Solanki, M.H.; Chatterjee, P.K.; Xue, X.; Gupta, M.; Rosales, I.; Yeboah, M.M.; Kohn, N.; Metz, C.N. Magnesium protects against cisplatin-induced acute kidney injury without compromising cisplatin-mediated killing of an ovarian tumor xenograft in mice. Am. J. Physiol. Renal Physiol. 2015, 309, F35–F47. [Google Scholar] [CrossRef]
- Solanki, M.H.; Chatterjee, P.K.; Gupta, M.; Xue, X.; Plagov, A.; Metz, M.H.; Mintz, R.; Singhal, P.; Metz, C.N. Magnesium protects against cisplatin-induced acute kidney injury by regulating platinum accumulation. Am. J. Physiol. Physiol. 2014, 307, F369–F384. [Google Scholar] [CrossRef]
- Saito, Y.; Okamoto, K.; Kobayashi, M.; Narumi, K.; Yamada, T.; Iseki, K. Magnesium attenuates cisplatin-induced nephrotoxicity by regulating the expression of renal transporters. Eur. J. Pharmacol. 2017, 811, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Kubo, Y.; Miyata, H.; Sugimura, K.; Shinno, N.; Ushigome, H.; Yanagimoto, Y.; Takahashi, Y.; Yamamoto, K.; Nishimura, J.; Wada, H.; et al. Prophylactic Effect of Premedication with Intravenous Magnesium on Renal Dysfunction in Preoperative Cisplatin-Based Chemotherapy for Esophageal Cancer. Oncology 2019, 97, 319–326. [Google Scholar] [CrossRef] [PubMed]
- Hamroun, A.; Lenain, R.; Bigna, J.J.; Speyer, E.; Bui, L.; Chamley, P.; Pottier, N.; Cauffiez, C.; Dewaeles, E.; Dhalluin, X.; et al. Prevention of Cisplatin-Induced Acute Kidney Injury: A Systematic Review and Meta-Analysis. Drugs 2019, 79, 1567–1582. [Google Scholar] [CrossRef]
- Danwilai, K.; Lohitnavy, O.; Sakunrag, I.; Dilokthornsakul, P. The effect of magnesium supplementation on cisplatin induced nephrotoxicity: A sys-tematic review and meta-analysis. Pharm. Sci. Asia 2021, 48, 25–36. [Google Scholar] [CrossRef]
- Osman, N.M.; Copley, M.P.; Litterst, C.L. Amelioration of cisplatin-induced nephrotoxicity by the diuretic acetazolamide in F344 rats. Cancer Treat. Rep. 1984, 68, 999–1004. [Google Scholar]
- el Hamamsy, M.; Kamal, N.; Bazan, N.S.; el Haddad, M. Evaluation of the effect of acetazolamide versus mannitol on cisplatin-induced ne-phrotoxicity, a pilot study. Int. J. Clin. Pharm. 2018, 40, 1539–1547. [Google Scholar] [CrossRef] [PubMed]
- Ismail, R.S.; El-Awady, M.S.; Hassan, M.H. Pantoprazole abrogated cisplatin-induced nephrotoxicity in mice via suppression of inflammation, apoptosis, and oxidative stress. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2020, 393, 1161–1171. [Google Scholar] [CrossRef]
- Ghonaim, E.; El-Haggar, S.; Gohar, S. Possible protective effect of pantoprazole against cisplatin-induced nephrotoxicity in head and neck cancer patients: A randomized controlled trial. Med. Oncol. 2021, 38, 1–12. [Google Scholar] [CrossRef]
- Okamoto, K.; Saito, Y.; Narumi, K.; Furugen, A.; Iseki, K.; Kobayashi, M. Non-steroidal anti-inflammatory drugs are a risk factor for cisplatin-induced nephrotoxicity: A meta-analysis of retrospective studies. Anticancer. Res. 2020, 40, 1747–1751. [Google Scholar] [CrossRef]
- Ramos-Inza, S.; Ruberte, A.C.; Sanmartín, C.; Sharma, A.K.; Plano, D. NSAIDs: Old Acquaintance in the Pipeline for Cancer Treatment and Pre-vention-Structural Modulation, Mechanisms of Action, and Bright Future. J. Med. Chem. 2021, 64, 16380–16421. [Google Scholar] [CrossRef]
- Sidhu, H.; Capalash, N. Synergistic anti-cancer action of salicylic acid and cisplatin on HeLa cells elucidated by network pharmacology and in vitro analysis. Life Sci. 2021, 282, 119802. [Google Scholar] [CrossRef] [PubMed]
- Khan, P.; Bhattacharya, A.; Sengupta, D.; Banerjee, S.; Adhikary, A.; Das, T. Aspirin enhances cisplatin sensitivity of resistant non-small cell lung carcinoma stem-like cells by targeting mTOR-Akt axis to repress migration. Sci. Rep. 2019, 9, 16913. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Zhu, Y.; Yu, L.; Li, Y.; Guo, J.; Cai, J.; Liu, L.; Wang, Z. Aspirin inhibits tumor progression and enhances cisplatin sensitivity in epithelial ovarian cancer. PeerJ 2021, 9, e11591. [Google Scholar] [CrossRef]
- Jiang, W.; Yan, Y.; Chen, M.; Luo, G.; Hao, J.; Pan, J.; Hu, S.; Guo, P.; Li, W.; Wang, R.; et al. Aspirin enhances the sensitivity of colon cancer cells to cisplatin by abrogating the binding of NF-κB to the COX-2 promoter. Aging 2020, 12, 611–627. [Google Scholar] [CrossRef] [PubMed]
- Endo, H.; Yano, M.; Okumura, Y.; Kido, H. Ibuprofen enhances the anticancer activity of cisplatin in lung cancer cells by inhibiting the heat shock protein 70. Cell Death Dis. 2014, 5, e1027. [Google Scholar] [CrossRef]
- Neumann, W.; Crews, B.C.; Sárosi, M.B.; Daniel, C.M.; Ghebreselasie, K.; Scholz, M.S.; Marnett, L.J.; Hey-Hawkins, E. Conjugation of cisplatin analogues and cyclooxygenase inhibitors to overcome cisplatin resistance. ChemMedChem 2015, 10, 183–192. [Google Scholar] [CrossRef]
- Neumann, W.; Crews, B.C.; Marnett, L.J.; Hey-Hawkins, E. Conjugates of Cisplatin and Cyclooxygenase Inhibitors as Potent Antitumor Agents Overcoming Cisplatin Resistance. ChemMedChem 2014, 9, 1150–1153. [Google Scholar] [CrossRef]
- Intini, F.P.; Zajac, J.; Novohradsky, V.; Saltarella, T.; Pacifico, C.; Brabec, V.; Natile, G.; Kasparkova, J. Novel Antitumor Platinum(II) Conjugates Containing the Nonsteroidal Anti-inflammatory Agent Diclofenac: Synthesis and Dual Mechanisms of Antiproliferative Effects. Inorg. Chem. 2017, 56, 1483–1497. [Google Scholar] [CrossRef]
- Hou, J.; Karin, M.; Sun, B. Targeting cancer-promoting inflammation—Have anti-inflammatory therapies come of age? Nat. Rev. Clin. Oncol. 2021, 18, 261–279. [Google Scholar] [CrossRef]
- Paramanantham, A.; Kim, M.J.; Jung, E.J.; Kim, H.J.; Chang, S.H.; Jung, J.M.; Hong, S.C.; Shin, S.C.; Kim, G.S.; Lee, W.S. Anthocyanins isolated from vitis coignetiae pulliat enhances cisplatin sensitivity in MCF-7 human breast cancer cells through inhibition of Akt and NF-κB activation. Molecules 2020, 25, 3623. [Google Scholar] [CrossRef]
- Sun, C.-Y.; Zhang, Q.-Y.; Zheng, G.-J.; Feng, B. Phytochemicals: Current strategy to sensitize cancer cells to cisplatin. Biomed. Pharmacother. 2018, 110, 518–527. [Google Scholar] [CrossRef] [PubMed]
- Cheuk, I.W.; Chen, J.; Siu, M.; Ho, J.C.; Lam, S.S.; Shin, V.Y.; Kwong, A. Resveratrol enhanced chemo-sensitivity by reversing macrophage polarization in breast cancer. Clin. Transl. Oncol. 2022, 24, 854–863. [Google Scholar] [CrossRef] [PubMed]
- Chang, W.S.; Tsai, C.W.; Yang, J.S.; Hsu, Y.M.; Shih, L.C.; Chiu, H.Y.; Bau, D.T.; Tsai, F.J. Resveratrol inhibited the metastatic behaviors of cisplatin-resistant human oral cancer cells via phosphorylation of ERK/p-38 and suppression of MMP-2/9. J. Food Biochem. 2021, 45, e13666. [Google Scholar] [CrossRef] [PubMed]
- Gajski, G.; Čimbora-Zovko, T.; Rak, S.; Rožman, M.; Osmak, M.; Garaj-Vrhovac, V. Combined antitumor effects of bee venom and cisplatin on human cervical and laryngeal carcinoma cells and their drug resistant sublines. J. Appl. Toxicol. 2013, 34, 1332–1341. [Google Scholar] [CrossRef]
- Paciello, F.; Fetoni, A.R.; Mezzogori, D.; Rolesi, R.; Di Pino, A.; Paludetti, G.; Grassi, C.; Troiani, D. The dual role of curcumin and ferulic acid in counteracting chemoresistance and cisplatin-induced ototoxicity. Sci. Rep. 2020, 10, 1063. [Google Scholar] [CrossRef]

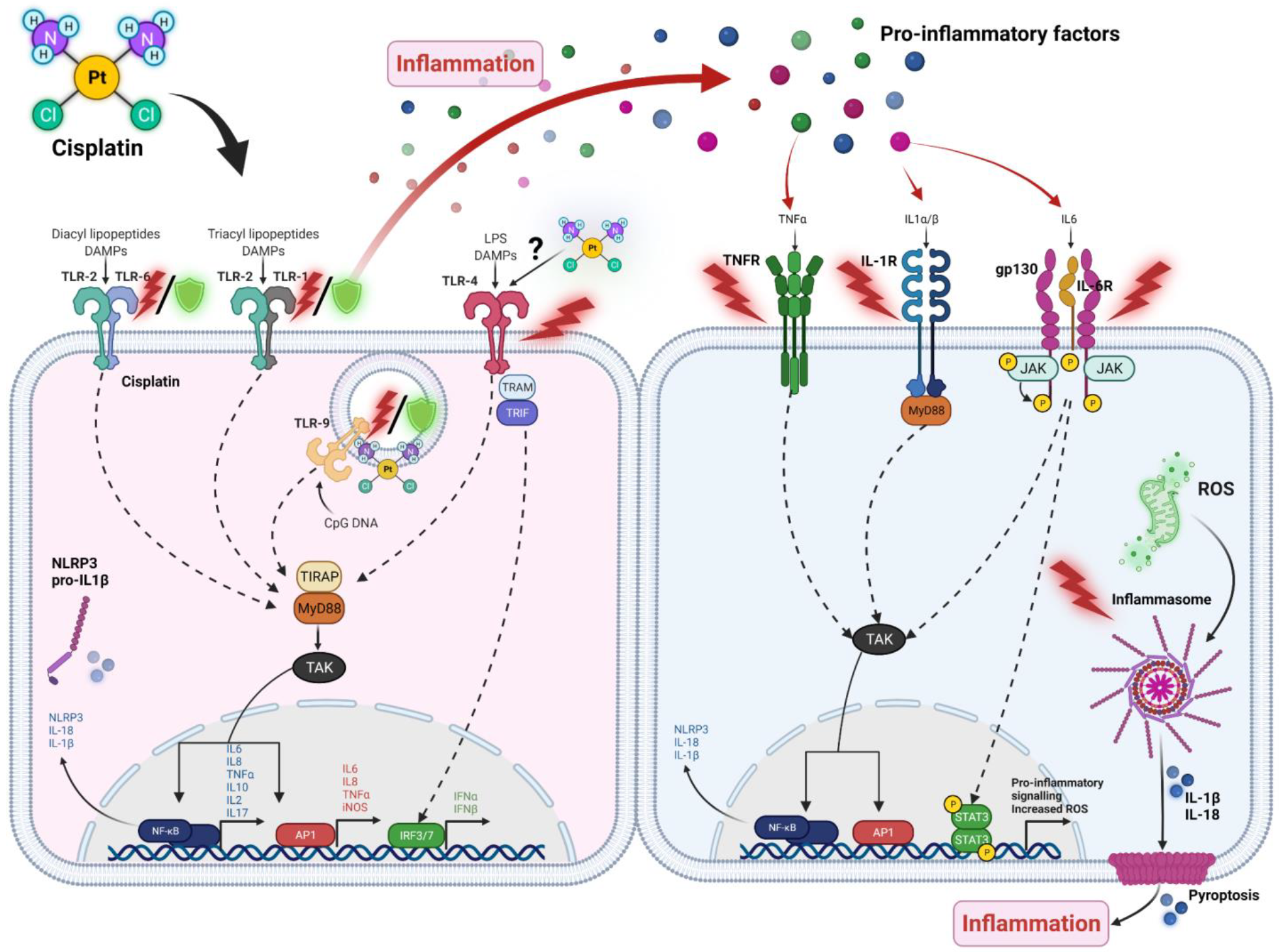

| Classification | Name | Anti-Inflammatory Mechanism(s) of Action | Targeted CITs | Reference(s) |
|---|---|---|---|---|
| Non- Flavonoid Polyphenol Phytoalexin | Resveratrol | Inhibits TLR4 Signalling Inhibits IL-6, TNF-α, Inhibits NF-κB Signalling Radical Scavenging Metal Chelation | CIO CIOV (Ovarian) CIU (Uterine) CIN | [165,166,167,168,169,170,171,172,173,174,175,176] |
| Bee Products | Honey | Inhibits IL6, TNF-α, MCP-1, ICAM1 | CIN | [177] |
| Caffeic Acid | Radical Scavenging | CIO | [178] | |
| Bee Pollen Extract | Inhibits IL6, IL1β Inhibits NF-κB Signalling | CIN CIH | [179] | |
| Mellit (Bee Venom) | Inhibits IL-6, IL-1β, TNF-α Upregulates IL-10 Alters/Inhibits Mθ Invasion Enhanced T-Reg Activity Inhibits COX2 | CIN CIPN (Potential) | [180,181,182] | |
| Flavonoid | Curcumin | Inhibits IL-6, IL-1β, TNF-α, MCP-1 Upregulates/maintains IL-10 Free Radical Scavenging Inhibits COX Inhibits JAK/STAT Signalling Inhibits NF-κB Signalling | CIN CIO CIPN CIH | [183,184,185,186,187,188,189,190,191,192,193] |
| Epigallocatechin Gallate (EGCG) | Inhibits MAPK Signalling Dis-regulates STAT1/STAT3 Signalling Inhibits IL-6, IL-1β, TNF-α Inhibits NF-κB Signalling Upregulates Nrf2 | CIN CIH CICN (Cerebral Neurotoxicity) | [194,195,196,197,198,199] | |
| Quercetin | Inhibits NF-κB Signalling Potential COX Inhibition Potential iNOS Inhibition | CIO CIN | [200,201,202,203,204,205] | |
| Baicalein | Inhibits IL-6, TNF-α Inhibits NF-κB Inhibits TLR-2, TLR-4 Signalling Upregulates Nrf2 Upregulates HO-1 | CIN | [93,206] | |
| Wogonin | Inhibits IL-6, IL-8, IL-1β, MCP-1, TNF-α Inhibits NF-κB Signalling Inhibit COX2 | CIN | [207,208,209] | |
| Glycyrrhizic Acid (Licorice) | Inhibits IL-1β, TNF-α Inhibits NF-κB Signalling Reduces DAMP Release | CIN CIH | [210,211,212] | |
| Hesperetin | Inhibits MAPK Signalling Upregulates TNF-α Signal Inhibitor Upregulates Nrf2 Upregulates HO-1 | CIN | [213] | |
| D-Pinitol | Inhibits IL-6, IL-1β, TNF-α | CIN | [214] | |
| Sappanone A | Inhibits IL-6, IL-1β, TNF-α Inhibits NF-κB Signalling Inhibits COX2 Upregulates Nrf2 | CIN | [215,216,217] | |
| Xanthohumol | Inhibits IL-6, IL-1β, TNF-α Inhibits TLR4 Expression Inhibits NF-κB Signalling | CIN | [218] | |
| Puerarin | Inhibits IL-6, TNF-α Inhibits TLR4 Signalling Inhibits ΝF-κB | CIN CIO | [219,220] | |
| Icariin | Inhibits IL-1β, TNF-α Inhibits NF-κΒ Signalling Inhibits iNOS | CIN | [221,222] | |
| Genistein | Inhibits ICAM-1, MCP-1 Inhibits Mθ Infiltration Inhibits NF-κB Signalling | CIN CIO | [223,224] | |
| Galangin | Inhibits IL-6, IL-1β, TNF-α Inhibits MAPK Signalling Inhibits NF-κΒ Signalling | CIN | [225,226] | |
| Luteolin | Inhibits TNF-α Inhibits NF-κB Signalling Inhibits COX2 | CIN | [227] | |
| Saponins | Ginsenosides (Several) | Inhibit IL-1β, TNF-α Inhibit NF-κB Signalling Inhibit COX2 Inhibit iNOS | CIN CIO | [228,229,230,231,232,233] |
| Alkaloids | Berberine | Inhibits IL-6, IL-1β, TNF-α Inhibits MAPK Signalling Inhibits NF-κB Signalling Upregulates IL-10 s Antioxidant Effect (Reduced ROS Generation) | CIPN CIO | [234,235] |
| Betaine | Inhibits IL-6, IL-1β, TNF-α Inhibits NF-κB Signalling | CIN | [236,237] | |
| Tetramethylpyrazine | Inhibits IL-1β, TNF-α Inhibits TLR4 Signalling Reduces DAMP Release Upregulates Nrf2s | CIN CIO (Potential) | [238,239] | |
| Other Natural Compounds | Astragaloside | Inhibits IL-1β, TNF-α Inhibits Caspase-1 & NLRP3 Inhibits NF-κB Signalling Upregulates Nrf2 | CIN | [113,240] |
| Sinapic Acid | Inhibits IL-6, IL-1β, TNF-α Inhibits NF-κB Signalling Upregulates Nrf2 Upregulates HO-1 | CIN | [241,242] | |
| Vanillin | Inhibits IL-18, TNF-α Inhibits NF-κB Signalling Inhibits iNOS Upregulates IL-10 Upregulates Nrf2 | CIN | [243,244] |
| Classification | Name | Anti-Inflammatory Mechanism(s) of Action | Targeted CITs | Reference(s) |
|---|---|---|---|---|
| Anti-Inflammatory (Iridoid Glycoside) | Aucubin | Inhibits TNF-α Inhibits STAT3/MAPK Signalling Inhibits NF-κB Signalling Upregulates HO-1 | CIN | [245] |
| Anti-Inflammatory (COX2 Inhibitor) | Etoricoxib | Inhibits TNF-α Inhibits iNOS Inhibits COX2 | CIN | [173] |
| Anti-Inflammatory (Clove Oil) | Eugenol | Inhibits IL-6, TNF-α Inhibits COX2 | CIO CIN CITT (Testicular) | [246,247,248,249] |
| Anti-Inflammatory (Iridoid Monoterpinoid) | Monotropein | Upregulated HO-1 Upregulated Nrf2 | CIN | [250,251,252] |
| Anti-Inflammatory (Synthetic Luteolin) | Isoorientin | Inhibits MAPK Signalling Reduces DAMP Release Inhibits NF-κB Signalling Upregulated HO-1 Upregulates Nrf2 | CIN | [253] |
| Anti-Inflammatory (Calcium Antagonist) | Flunarizine | Inhibits IL-6, IL-1β, TNF-α Inhibits MAPK Signalling Inhibits NF-κB Signalling Upregulates Nrf2 Upregulates HO-1 | CIO CIUN (Uremic Neuropathy) | [75,254,255,256] |
| Anti-Inflammatory | R-Phenylisopropyladenosine (R-PIA) | Inhibits TNF-α Inhibits MAPK Signalling Inhibits STAT1 Signalling Inhibits iNOS Inhibits COX2 | CIO | [257] |
| Solubilised Membrane Protein | Thrombomodulin Alfa | Thrombin-Mediated DAMP Degradation | Chemotherapy-IPN | [258,259,260] |
| Anti-Inflammatory (Nrf2 Activator) | Dimethyl Fumarate | Inhibits Immune Cell Infiltrations Upregulation of Nrf2s Inhibits IL-6, IL4, IL-10, TNF-α | Chemotherapy-IPN CIPNs CIN | [261,262,263] |
| Clinical Trial Status | Intervention (Name) | Mechanism(s) of Action | Listed Target Condition (CIT) | Additional Reference(s) |
|---|---|---|---|---|
| Recruiting | N-Acetylcysteine | Antioxidant | CIO (CIN) | [266,267,268,269] |
| Terminated | Sodium Thiosulfate (Trans-Tympanic Gel) | Antioxidant | CIO | [270,271] |
| Completed | Ginkgo Biloba Extract (GBE761) | Antioxidant Anti-Inflammatory | CIO | [272,273,274,275] |
| Completed | Sodium Thiosulfate | Antioxidant | CIO | [270,271] |
| Not Recruiting | Sodium Thiosulfate (+Mannitol) | Antioxidant | CIO | [270,271] |
| Recruiting | Rosuvastatin | Cholesterol Reduction (HMG-CoA Inhibition) Antioxidant Anti-Inflammatory | CIO CIN | [276,277,278,279,280,281,282] |
| Completed | Dexamethasone (Synthetic Corticosteroid) | Anti-Inflammatory Antioxidant | CIO | [283,284,285,286,287,288] |
| Terminated | OTO-104 (Dexamethasone Hydrogel) | Anti-Inflammatory Antioxidant | CIO | [289] |
| Recruiting | Tempol (SOD and Catalase Mimetic) | Antioxidant | CIN CIO | [290,291,292,293] |
| Completed | Silymarin | Antioxidant Anti-Inflammatory | CIN | [294,295,296] |
| Completed | Mannitol (Intravenous) | Osmotic Diuretic | CIN | [297,298,299,300] |
| Completed | Preloaded Magnesium | Homeostatic Cisplatin Efflux Regulation (Downregulated Transporters) | CIN | [301,302,303,304,305,306] |
| Completed | Acetazolamide + Mannitol, (+N-Acetylcysteine) | Alkaline Diuretic (Carbonic Anhydrase Inhibitor) | CIN | [307,308] |
| Recruiting | Pantoprazole | Cisplatin Influx Regulation (OCT2 Inhibitor) Anti-Inflammatory | CIN | [309,310] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Domingo, I.K.; Latif, A.; Bhavsar, A.P. Pro-Inflammatory Signalling PRRopels Cisplatin-Induced Toxicity. Int. J. Mol. Sci. 2022, 23, 7227. https://doi.org/10.3390/ijms23137227
Domingo IK, Latif A, Bhavsar AP. Pro-Inflammatory Signalling PRRopels Cisplatin-Induced Toxicity. International Journal of Molecular Sciences. 2022; 23(13):7227. https://doi.org/10.3390/ijms23137227
Chicago/Turabian StyleDomingo, Ivan K., Asna Latif, and Amit P. Bhavsar. 2022. "Pro-Inflammatory Signalling PRRopels Cisplatin-Induced Toxicity" International Journal of Molecular Sciences 23, no. 13: 7227. https://doi.org/10.3390/ijms23137227
APA StyleDomingo, I. K., Latif, A., & Bhavsar, A. P. (2022). Pro-Inflammatory Signalling PRRopels Cisplatin-Induced Toxicity. International Journal of Molecular Sciences, 23(13), 7227. https://doi.org/10.3390/ijms23137227






