The Zebrafish, an Outstanding Model for Biomedical Research in the Field of Melatonin and Human Diseases
Abstract
:1. Zebrafish as a Model for Biomedical Research
1.1. General Features of Zebrafish
1.2. Advantages of Using Zebrafish as an Experimental Model
1.3. Circadian Rhythms in Zebrafish
2. Melatonin in Zebrafish
2.1. Synthesis and Metabolism of Melatonin
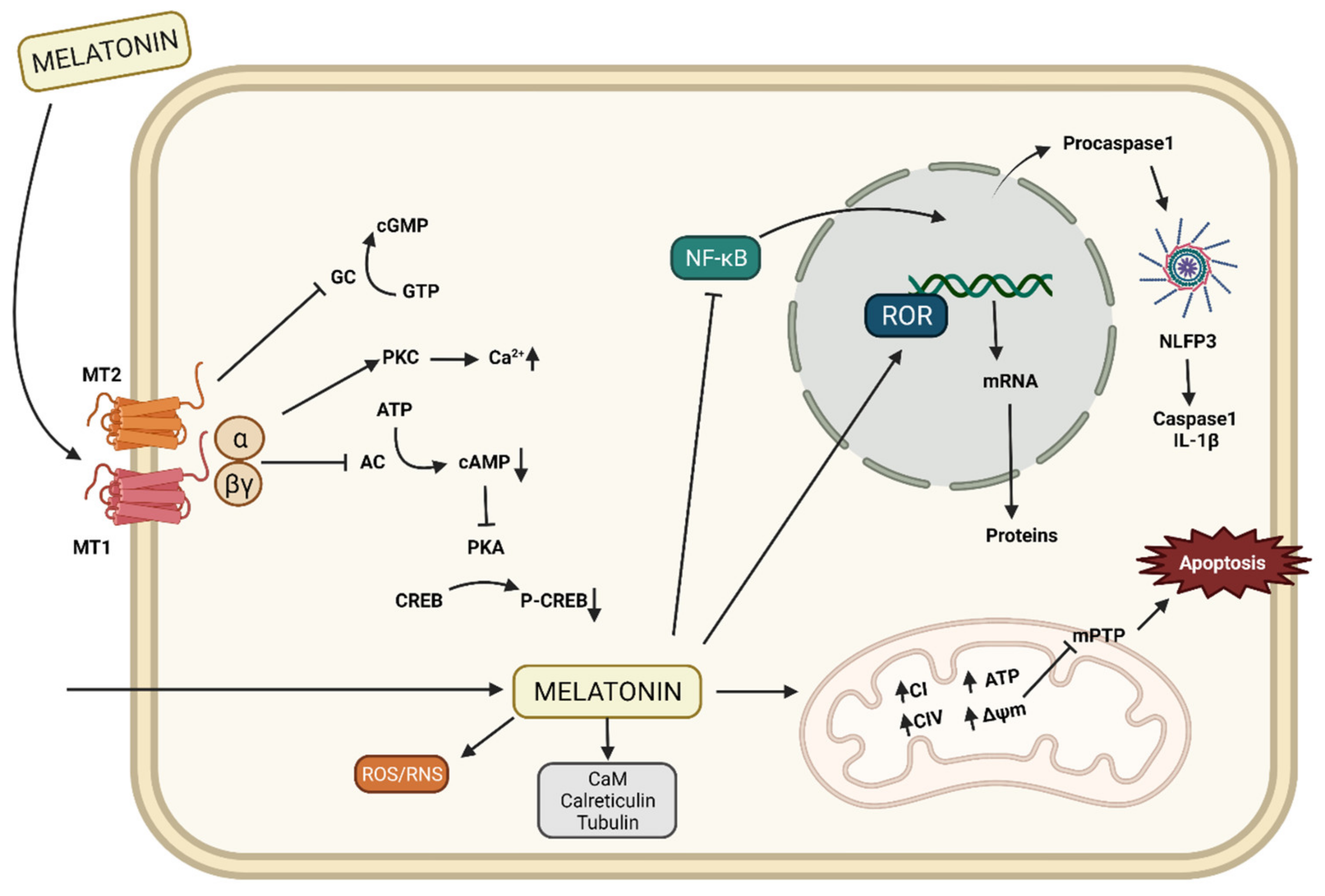
2.2. Mechanisms of Action of Melatonin
2.3. Pineal Melatonin Properties
2.4. Extrapineal Melatonin Properties
3. Therapeutic Properties of Melatonin in Zebrafish Models of Disease
3.1. Melatonin in Parkinsonian Zebrafish
3.2. Melatonin against Epilepsy in Zebrafish
3.3. Protective Effects of Melatonin against Cardiovascular Damage in Zebrafish
3.4. Melatonin and Obesity in Zebrafish
3.5. Regulation of Bone Metabolism by Melatonin in Zebrafish
3.6. Melatonin in Zebrafish Model of Psychiatric Diseases
3.7. Melatonin, Aging, and Chronodisruption in Zebrafish
4. Prospective Research
5. Concluding Remarks—Importance to Humans
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Spence, R.; Gerlach, G.; Lawrence, C.; Smith, C. The behaviour and ecology of the zebrafish, Danio rerio. Biol. Rev. Camb. Philos. Soc. 2008, 83, 13–34. [Google Scholar] [CrossRef] [PubMed]
- Parichy, D.M. Evolution of danio pigment pattern development. Heredity 2006, 97, 200–210. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kimmel, C.B.; Ballard, W.W.; Kimmel, S.R.; Ullmann, B.; Schilling, T.F. Stages of embryonic development of the zebrafish. Dev. Dyn. Off. Publ. Am. Assoc. Anat. 1995, 203, 253–310. [Google Scholar] [CrossRef] [PubMed]
- Streisinger, G.; Walker, C.; Dower, N.; Knauber, D.; Singer, F. Production of clones of homozygous diploid zebra fish (Brachydanio rerio). Nature 1981, 291, 293–296. [Google Scholar] [CrossRef]
- Dooley, K.; Zon, L.I. Zebrafish: A model system for the study of human disease. Curr. Opin. Genet. Dev. 2000, 10, 252–256. [Google Scholar] [CrossRef]
- Facchinello, N.; Laquatra, C.; Locatello, L.; Beffagna, G.; Brañas Casas, R.; Fornetto, C.; Dinarello, A.; Martorano, L.; Vettori, A.; Risato, G.; et al. Efficient clofilium tosylate-mediated rescue of POLG-related disease phenotypes in zebrafish. Cell Death Dis. 2021, 12, 100. [Google Scholar] [CrossRef]
- Hwang, W.Y.; Peterson, R.T.; Yeh, J.R. Methods for targeted mutagenesis in zebrafish using TALENs. Methods 2014, 69, 76–84. [Google Scholar] [CrossRef] [Green Version]
- Amsterdam, A.; Becker, T.S. Transgenes as screening tools to probe and manipulate the zebrafish genome. Dev. Dyn. Off. Publ. Am. Assoc. Anat. 2005, 234, 255–268. [Google Scholar] [CrossRef]
- Miyawaki, I. Application of zebrafish to safety evaluation in drug discovery. J. Toxicol. Pathol. 2020, 33, 197–210. [Google Scholar] [CrossRef]
- Díaz-Casado, M.E.; Rusanova, I.; Aranda, P.; Fernández-Ortiz, M.; Sayed, R.K.A.; Fernández-Gil, B.I.; Hidalgo-Gutiérrez, A.; Escames, G.; López, L.C.; Acuña-Castroviejo, D. In Vivo Determination of Mitochondrial Respiration in 1-Methyl-4-Phenyl-1,2,3,6-Tetrahydropyridine-Treated Zebrafish Reveals the Efficacy of Melatonin in Restoring Mitochondrial Normalcy. Zebrafish 2018, 15, 15–26. [Google Scholar] [CrossRef]
- Adusumilli, L.; Facchinello, N.; Teh, C.; Busolin, G.; Le, M.T.; Yang, H.; Beffagna, G.; Campanaro, S.; Tam, W.L.; Argenton, F.; et al. miR-7 Controls the Dopaminergic/Oligodendroglial Fate through Wnt/β-catenin Signaling Regulation. Cells 2020, 9, 711. [Google Scholar] [CrossRef] [Green Version]
- Howe, K.; Clark, M.D.; Torroja, C.F.; Torrance, J.; Berthelot, C.; Muffato, M.; Collins, J.E.; Humphray, S.; McLaren, K.; Matthews, L.; et al. The zebrafish reference genome sequence and its relationship to the human genome. Nature 2013, 496, 498–503. [Google Scholar] [CrossRef] [Green Version]
- Barbazuk, W.B.; Korf, I.; Kadavi, C.; Heyen, J.; Tate, S.; Wun, E.; Bedell, J.A.; McPherson, J.D.; Johnson, S.L. The syntenic relationship of the zebrafish and human genomes. Genome Res. 2000, 10, 1351–1358. [Google Scholar] [CrossRef] [Green Version]
- Tavares, B.; Santos Lopes, S. The importance of Zebrafish in biomedical research. Acta Med. Port. 2013, 26, 583–592. [Google Scholar] [CrossRef]
- Cahill, G.M. Clock mechanisms in zebrafish. Cell Tissue Res. 2002, 309, 27–34. [Google Scholar] [CrossRef]
- Vatine, G.; Vallone, D.; Gothilf, Y.; Foulkes, N.S. It’s time to swim! Zebrafish and the circadian clock. FEBS Lett. 2011, 585, 1485–1494. [Google Scholar] [CrossRef]
- Wang, H. Comparative genomic analysis of teleost fish bmal genes. Genetica 2009, 136, 149–161. [Google Scholar] [CrossRef]
- Wang, H. Comparative analysis of teleost fish genomes reveals preservation of different ancient clock duplicates in different fishes. Mar. Genom. 2008, 1, 69–78. [Google Scholar] [CrossRef]
- Liu, C.; Hu, J.; Qu, C.; Wang, L.; Huang, G.; Niu, P.; Zhong, Z.; Hong, F.; Wang, G.; Postlethwait, J.H.; et al. Molecular evolution and functional divergence of zebrafish (Danio rerio) cryptochrome genes. Sci. Rep. 2015, 5, 8113. [Google Scholar] [CrossRef] [Green Version]
- Kobayashi, Y.; Ishikawa, T.; Hirayama, J.; Daiyasu, H.; Kanai, S.; Toh, H.; Fukuda, I.; Tsujimura, T.; Terada, N.; Kamei, Y.; et al. Molecular analysis of zebrafish photolyase/cryptochrome family: Two types of cryptochromes present in zebrafish. Genes Cells Devoted Mol. Cell. Mech. 2000, 5, 725–738. [Google Scholar] [CrossRef]
- Wang, H. Comparative analysis of period genes in teleost fish genomes. J. Mol. Evol. 2008, 67, 29–40. [Google Scholar] [CrossRef]
- Ben-Moshe, Z.; Foulkes, N.S.; Gothilf, Y. Functional development of the circadian clock in the zebrafish pineal gland. BioMed Res. Int. 2014, 2014, 235781. [Google Scholar] [CrossRef] [Green Version]
- Dekens, M.P.; Santoriello, C.; Vallone, D.; Grassi, G.; Whitmore, D.; Foulkes, N.S. Light regulates the cell cycle in zebrafish. Curr. Biol. 2003, 13, 2051–2057. [Google Scholar] [CrossRef] [Green Version]
- Elbaz, I.; Foulkes, N.S.; Gothilf, Y.; Appelbaum, L. Circadian clocks, rhythmic synaptic plasticity and the sleep-wake cycle in zebrafish. Front. Neural Circuits 2013, 7, 9. [Google Scholar] [CrossRef] [Green Version]
- Morbiato, E.; Frigato, E.; Dinarello, A.; Maradonna, F.; Facchinello, N.; Argenton, F.; Carnevali, O.; Dalla Valle, L.; Bertolucci, C. Feeding Entrainment of the Zebrafish Circadian Clock Is Regulated by the Glucocorticoid Receptor. Cells 2019, 8, 1342. [Google Scholar] [CrossRef] [Green Version]
- Sánchez-Vázquez, F.J.; López-Olmeda, J.F.; Vera, L.M.; Migaud, H.; López-Patiño, M.A.; Míguez, J.M. Environmental Cycles, Melatonin, and Circadian Control of Stress Response in Fish. Front. Endocrinol. 2019, 10, 279. [Google Scholar] [CrossRef]
- Korf, H.W.; Schomerus, C.; Stehle, J.H. The pineal organ, its hormone melatonin, and the photoneuroendocrine system. Adv. Anat. Embryol. Cell Biol. 1998, 146, 1–100. [Google Scholar] [CrossRef]
- Falcón, J.; Gothilf, Y.; Coon, S.L.; Boeuf, G.; Klein, D.C. Genetic, temporal and developmental differences between melatonin rhythm generating systems in the teleost fish pineal organ and retina. J. Neuroendocrinol. 2003, 15, 378–382. [Google Scholar] [CrossRef] [Green Version]
- Gothilf, Y.; Coon, S.L.; Toyama, R.; Chitnis, A.; Namboodiri, M.A.; Klein, D.C. Zebrafish serotonin N-acetyltransferase-2: Marker for development of pineal photoreceptors and circadian clock function. Endocrinology 1999, 140, 4895–4903. [Google Scholar] [CrossRef]
- Ziv, L.; Tovin, A.; Strasser, D.; Gothilf, Y. Spectral sensitivity of melatonin suppression in the zebrafish pineal gland. Exp. Eye Res. 2007, 84, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Ziv, L.; Levkovitz, S.; Toyama, R.; Falcon, J.; Gothilf, Y. Functional development of the zebrafish pineal gland: Light-induced expression of period2 is required for onset of the circadian clock. J. Neuroendocrinol. 2005, 17, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Ben-Moshe Livne, Z.; Alon, S.; Vallone, D.; Bayleyen, Y.; Tovin, A.; Shainer, I.; Nisembaum, L.G.; Aviram, I.; Smadja-Storz, S.; Fuentes, M.; et al. Genetically Blocking the Zebrafish Pineal Clock Affects Circadian Behavior. PLoS Genet. 2016, 12, e1006445. [Google Scholar] [CrossRef] [PubMed]
- Lima-Cabello, E.; Díaz-Casado, M.E.; Guerrero, J.A.; Otalora, B.B.; Escames, G.; López, L.C.; Reiter, R.J.; Acuña-Castroviejo, D. A review of the melatonin functions in zebrafish physiology. J. Pineal. Res. 2014, 57, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Lerner, A.; Case, J.; Mori, W.; Wright, M.R. Melatonin in Peripheral Nerve. Nature 1959, 183, 1821. [Google Scholar] [CrossRef]
- Quay, W.B.; Bagnara, J.T. Relative Potencies of Indolic and Related Compounds in The Body-Lightening Reaction of Larval Xenopus. Arch. Int. Pharmacodyn. Ther. 1964, 150, 137–143. [Google Scholar]
- Acuña-Castroviejo, D.; Escames, G.; Venegas, C.; Díaz-Casado, M.E.; Lima-Cabello, E.; López, L.C.; Rosales-Corral, S.; Tan, D.X.; Reiter, R.J. Extrapineal melatonin: Sources, regulation, and potential functions. Cell Mol. Life Sci. 2014, 71, 2997–3025. [Google Scholar] [CrossRef]
- Reiter, R.J. Pineal melatonin: Cell biology of its synthesis and of its physiological interactions. Endocr. Rev. 1991, 12, 151–180. [Google Scholar] [CrossRef] [Green Version]
- Venegas, C.; García, J.A.; Escames, G.; Ortiz, F.; López, A.; Doerrier, C.; García-Corzo, L.; López, L.C.; Reiter, R.J.; Acuña-Castroviejo, D. Extrapineal melatonin: Analysis of its subcellular distribution and daily fluctuations. J. Pineal. Res. 2012, 52, 217–227. [Google Scholar] [CrossRef]
- Stefulj, J.; Hörtner, M.; Ghosh, M.; Schauenstein, K.; Rinner, I.; Wölfler, A.; Semmler, J.; Liebmann, P.M. Gene expression of the key enzymes of melatonin synthesis in extrapineal tissues of the rat. J. Pineal. Res. 2001, 30, 243–247. [Google Scholar] [CrossRef]
- Klein, D.C.; Schaad, N.L.; Namboordiri, M.A.; Yu, L.; Weller, J.L. Regulation of pineal serotonin N-acetyltransferase activity. Biochem Soc. Trans. 1992, 20, 299–304. [Google Scholar] [CrossRef]
- Ma, X.; Idle, J.R.; Krausz, K.W.; Gonzalez, F.J. Metabolism of melatonin by human cytochromes p450. Drug Metab. Dispos. Biol. Fate Chem. 2005, 33, 489–494. [Google Scholar] [CrossRef]
- Semak, I.; Korik, E.; Antonova, M.; Wortsman, J.; Slominski, A. Metabolism of melatonin by cytochrome P450s in rat liver mitochondria and microsomes. J. Pineal Res. 2008, 45, 515–523. [Google Scholar] [CrossRef] [Green Version]
- Hirata, F.; Hayaishi, O.; Tokuyama, T.; Seno, S. In vitro and in vivo formation of two new metabolites of melatonin. J. Biol. Chem. 1974, 249, 1311–1313. [Google Scholar] [CrossRef]
- Hardeland, R.; Pandi-Perumal, S.R. Melatonin, a potent agent in antioxidative defense: Actions as a natural food constituent, gastrointestinal factor, drug and prodrug. Nutr. Metab. 2005, 2, 22. [Google Scholar] [CrossRef] [Green Version]
- León, J.; Escames, G.; Rodríguez, M.I.; López, L.C.; Tapias, V.; Entrena, A.; Camacho, E.; Carrión, M.D.; Gallo, M.A.; Espinosa, A.; et al. Inhibition of neuronal nitric oxide synthase activity by N1-acetyl-5-methoxykynuramine, a brain metabolite of melatonin. J. Neurochem. 2006, 98, 2023–2033. [Google Scholar] [CrossRef]
- Nikolaev, G.; Robeva, R.; Konakchieva, R. Membrane Melatonin Receptors Activated Cell Signaling in Physiology and Disease. Int. J. Mol. Sci. 2021, 23, 471. [Google Scholar] [CrossRef]
- Acuña-Castroviejo, D.; Pablos, M.I.; Menendez-Pelaez, A.; Reiter, R.J. Melatonin receptors in purified cell nuclei of liver. Res. Commun. Chem. Pathol. Pharmacol. 1993, 82, 253–256. [Google Scholar]
- Finocchiaro, L.M.; Glikin, G.C. Intracellular melatonin distribution in cultured cell lines. J. Pineal. Res. 1998, 24, 22–34. [Google Scholar] [CrossRef]
- Macías, M.; Escames, G.; Leon, J.; Coto, A.; Sbihi, Y.; Osuna, A.; Acuña-Castroviejo, D. Calreticulin-melatonin. An unexpected relationship. Eur. J. Biochem. 2003, 270, 832–840. [Google Scholar] [CrossRef] [Green Version]
- Tan, D.X.; Manchester, L.C.; Esteban-Zubero, E.; Zhou, Z.; Reiter, R.J. Melatonin as a Potent and Inducible Endogenous Antioxidant: Synthesis and Metabolism. Molecules 2015, 20, 18886–18906. [Google Scholar] [CrossRef] [Green Version]
- Reiter, R.J.; Tan, D.X.; Manchester, L.C.; Pilar Terron, M.; Flores, L.J.; Koppisepi, S. Medical implications of melatonin: Receptor-mediated and receptor-independent actions. Adv. Med. Sci. 2007, 52, 11–28. [Google Scholar] [PubMed]
- Denker, E.; Ebbesson, L.O.E.; Hazlerigg, D.G.; Macqueen, D.J. Phylogenetic Reclassification of Vertebrate Melatonin Receptors To Include Mel1d. G3 2019, 9, 3225–3238. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lutfi, E.; Basili, D.; Falcinelli, S.; Morillas, L.; Carnevali, O.; Capilla, E.; Navarro, I. The probiotic Lactobacillus rhamnosus mimics the dark-driven regulation of appetite markers and melatonin receptors’ expression in zebrafish (Danio rerio) larvae: Understanding the role of the gut microbiome. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2021, 256, 110634. [Google Scholar] [CrossRef] [PubMed]
- Danilova, N.; Krupnik, V.E.; Sugden, D.; Zhdanova, I.V. Melatonin stimulates cell proliferation in zebrafish embryo and accelerates its development. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2004, 18, 751–753. [Google Scholar] [CrossRef] [PubMed]
- Sakai, K.; Yamamoto, Y.; Ikeuchi, T. Vertebrates originally possess four functional subtypes of G protein-coupled melatonin receptor. Sci. Rep. 2019, 9, 9465. [Google Scholar] [CrossRef]
- Gilman, A.G. Nobel Lecture. G proteins and regulation of adenylyl cyclase. Biosci. Rep. 1995, 15, 65–97. [Google Scholar] [CrossRef]
- Von Gall, C.; Stehle, J.H.; Weaver, D.R. Mammalian melatonin receptors: Molecular biology and signal transduction. Cell Tissue Res. 2002, 309, 151–162. [Google Scholar] [CrossRef]
- Flores, M.V.; Hall, C.; Jury, A.; Crosier, K.; Crosier, P. The zebrafish retinoid-related orphan receptor (ror) gene family. Gene Expr. Patterns GEP 2007, 7, 535–543. [Google Scholar] [CrossRef]
- Delerive, P.; Monté, D.; Dubois, G.; Trottein, F.; Fruchart-Najib, J.; Mariani, J.; Fruchart, J.C.; Staels, B. The orphan nuclear receptor ROR alpha is a negative regulator of the inflammatory response. EMBO Rep. 2001, 2, 42–48. [Google Scholar] [CrossRef]
- Garcia, J.A.; Volt, H.; Venegas, C.; Doerrier, C.; Escames, G.; Lopez, L.C.; Acuna-Castroviejo, D. Disruption of the NF-kappaB/NLRP3 connection by melatonin requires retinoid-related orphan receptor-alpha and blocks the septic response in mice. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2015, 29, 3863–3875. [Google Scholar] [CrossRef]
- Venegas, C.; García, J.A.; Doerrier, C.; Volt, H.; Escames, G.; López, L.C.; Reiter, R.J.; Acuña-Castroviejo, D. Analysis of the daily changes of melatonin receptors in the rat liver. J. Pineal Res. 2013, 54, 313–321. [Google Scholar] [CrossRef]
- Loganathan, K.; Moriya, S.; Parhar, I.S. Diurnal Rhythm of trek2a Expression is Associated with Diurnal Rhythm of gnrh3 Expression in Zebrafish. Zool. Sci. 2019, 36, 167–171. [Google Scholar] [CrossRef]
- Benítez-King, G.; Antón-Tay, F. Calmodulin mediates melatonin cytoskeletal effects. Experientia 1993, 49, 635–641. [Google Scholar] [CrossRef]
- Cardinali, D.P.; Freire, F. Melatonin effects on brain. Interaction with microtubule protein, inhibition of fast axoplasmic flow and induction of crystaloid and tubular formations in the hypothalamus. Mol. Cell. Endocrinol. 1975, 2, 317–330. [Google Scholar] [CrossRef]
- Benítez-King, G.; Ríos, A.; Martínez, A.; Antón-Tay, F. In vitro inhibition of Ca2+/calmodulin-dependent kinase II activity by melatonin. Biochim. Biophys. Acta 1996, 1290, 191–196. [Google Scholar] [CrossRef]
- Benítez-King, G. Melatonin as a cytoskeletal modulator: Implications for cell physiology and disease. J. Pineal Res. 2006, 40, 1–9. [Google Scholar] [CrossRef]
- Reiter, R.J. The melatonin rhythm: Both a clock and a calendar. Experientia 1993, 49, 654–664. [Google Scholar] [CrossRef]
- De Borsetti, N.H.; Dean, B.J.; Bain, E.J.; Clanton, J.A.; Taylor, R.W.; Gamse, J.T. Light and melatonin schedule neuronal differentiation in the habenular nuclei. Dev. Biol. 2011, 358, 251–261. [Google Scholar] [CrossRef] [Green Version]
- Chiu, C.N.; Prober, D.A. Regulation of zebrafish sleep and arousal states: Current and prospective approaches. Front. Neural Circuits 2013, 7, 58. [Google Scholar] [CrossRef] [Green Version]
- Zhdanova, I.V.; Wang, S.Y.; Leclair, O.U.; Danilova, N.P. Melatonin promotes sleep-like state in zebrafish. Brain Res. 2001, 903, 263–268. [Google Scholar] [CrossRef]
- Zhdanova, I.V.; Yu, L.; Lopez-Patino, M.; Shang, E.; Kishi, S.; Guelin, E. Aging of the circadian system in zebrafish and the effects of melatonin on sleep and cognitive performance. Brain Res. Bull. 2008, 75, 433–441. [Google Scholar] [CrossRef] [Green Version]
- Appelbaum, L.; Wang, G.X.; Maro, G.S.; Mori, R.; Tovin, A.; Marin, W.; Yokogawa, T.; Kawakami, K.; Smith, S.J.; Gothilf, Y.; et al. Sleep-wake regulation and hypocretin-melatonin interaction in zebrafish. Proc. Natl. Acad. Sci. USA 2009, 106, 21942–21947. [Google Scholar] [CrossRef] [Green Version]
- Piccinetti, C.C.; Migliarini, B.; Olivotto, I.; Coletti, G.; Amici, A.; Carnevali, O. Appetite regulation: The central role of melatonin in Danio rerio. Horm. Behav. 2010, 58, 780–785. [Google Scholar] [CrossRef]
- Piccinetti, C.C.; Migliarini, B.; Olivotto, I.; Simoniello, M.P.; Giorgini, E.; Carnevali, O. Melatonin and peripheral circuitries: Insights on appetite and metabolism in Danio rerio. Zebrafish 2013, 10, 275–282. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blanco-Vives, B.; Sánchez-Vázquez, F.J. Synchronisation to light and feeding time of circadian rhythms of spawning and locomotor activity in zebrafish. Physiol. Behav. 2009, 98, 268–275. [Google Scholar] [CrossRef] [PubMed]
- Carnevali, O.; Gioacchini, G.; Maradonna, F.; Olivotto, I.; Migliarini, B. Melatonin induces follicle maturation in Danio rerio. PLoS ONE 2011, 6, e19978. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Ge, W. Developmental profiles of activin betaA, betaB, and follistatin expression in the zebrafish ovary: Evidence for their differential roles during sexual maturation and ovulatory cycle. Biol. Reprod. 2004, 71, 2056–2064. [Google Scholar] [CrossRef] [PubMed]
- So, W.K.; Kwok, H.F.; Ge, W. Zebrafish gonadotropins and their receptors: II. Cloning and characterization of zebrafish follicle-stimulating hormone and luteinizing hormone subunits—Their spatial-temporal expression patterns and receptor specificity. Biol. Reprod. 2005, 72, 1382–1396. [Google Scholar] [CrossRef] [Green Version]
- Falcón, J.; Migaud, H.; Muñoz-Cueto, J.A.; Carrillo, M. Current knowledge on the melatonin system in teleost fish. Gen. Comp. Endocrinol. 2010, 165, 469–482. [Google Scholar] [CrossRef] [Green Version]
- Lahiri, K.; Vallone, D.; Gondi, S.B.; Santoriello, C.; Dickmeis, T.; Foulkes, N.S. Temperature regulates transcription in the zebrafish circadian clock. PLoS Biol. 2005, 3, e351. [Google Scholar] [CrossRef] [Green Version]
- Acuna-Castroviejo, D.; Escames, G.; Rodriguez, M.I.; Lopez, L.C. Melatonin role in the mitochondrial function. Front. Biosci. A J. Virtual Libr. 2007, 12, 947–963. [Google Scholar] [CrossRef] [Green Version]
- Acuña-Castroviejo, D.; Martín, M.; Macías, M.; Escames, G.; León, J.; Khaldy, H.; Reiter, R.J. Melatonin, mitochondria, and cellular bioenergetics. J. Pineal Res. 2001, 30, 65–74. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Squadrito, G.L.; Uppu, R.; Pryor, W.A. Reaction of peroxynitrite with melatonin: A mechanistic study. Chem. Res. Toxicol. 1999, 12, 526–534. [Google Scholar] [CrossRef]
- Han, J.; Ji, C.; Guo, Y.; Yan, R.; Hong, T.; Dou, Y.; An, Y.; Tao, S.; Qin, F.; Nie, J.; et al. Mechanisms underlying melatonin-mediated prevention of fenvalerate-induced behavioral and oxidative toxicity in zebrafish. J. Toxicol. Environ. Health. Part A 2017, 80, 1331–1341. [Google Scholar] [CrossRef]
- Escames, G.; López, L.C.; Tapias, V.; Utrilla, P.; Reiter, R.J.; Hitos, A.B.; León, J.; Rodríguez, M.I.; Acuña-Castroviejo, D. Melatonin counteracts inducible mitochondrial nitric oxide synthase-dependent mitochondrial dysfunction in skeletal muscle of septic mice. J. Pineal Res. 2006, 40, 71–78. [Google Scholar] [CrossRef]
- Ortiz, F.; García, J.A.; Acuña-Castroviejo, D.; Doerrier, C.; López, A.; Venegas, C.; Volt, H.; Luna-Sánchez, M.; López, L.C.; Escames, G. The beneficial effects of melatonin against heart mitochondrial impairment during sepsis: Inhibition of iNOS and preservation of nNOS. J. Pineal Res. 2014, 56, 71–81. [Google Scholar] [CrossRef]
- Urata, Y.; Honma, S.; Goto, S.; Todoroki, S.; Iida, T.; Cho, S.; Honma, K.; Kondo, T. Melatonin induces gamma-glutamylcysteine synthetase mediated by activator protein-1 in human vascular endothelial cells. Free Radic. Biol. Med. 1999, 27, 838–847. [Google Scholar] [CrossRef]
- Pierrefiche, G.; Laborit, H. Oxygen free radicals, melatonin, and aging. Exp. Gerontol 1995, 30, 213–227. [Google Scholar] [CrossRef]
- Leon, J.; Acuña-Castroviejo, D.; Sainz, R.M.; Mayo, J.C.; Tan, D.X.; Reiter, R.J. Melatonin and mitochondrial function. Life Sci. 2004, 75, 765–790. [Google Scholar] [CrossRef]
- Martín, M.; Macías, M.; Escames, G.; León, J.; Acuña-Castroviejo, D. Melatonin but not vitamins C and E maintains glutathione homeostasis in t-butyl hydroperoxide-induced mitochondrial oxidative stress. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2000, 14, 1677–1679. [Google Scholar] [CrossRef]
- Acuña Castroviejo, D.; López, L.C.; Escames, G.; López, A.; García, J.A.; Reiter, R.J. Melatonin-mitochondria interplay in health and disease. Curr. Top. Med. Chem. 2011, 11, 221–240. [Google Scholar] [CrossRef]
- García, J.J.; Piñol-Ripoll, G.; Martínez-Ballarín, E.; Fuentes-Broto, L.; Miana-Mena, F.J.; Venegas, C.; Caballero, B.; Escames, G.; Coto-Montes, A.; Acuña-Castroviejo, D. Melatonin reduces membrane rigidity and oxidative damage in the brain of SAMP8 mice. Neurobiol. Aging 2011, 32, 2045–2054. [Google Scholar] [CrossRef]
- Acuña-Castroviejo, D.; Rahim, I.; Acuña-Fernández, C.; Fernández-Ortiz, M.; Solera-Marín, J.; Sayed, R.K.A.; Díaz-Casado, M.E.; Rusanova, I.; López, L.C.; Escames, G. Melatonin, clock genes and mitochondria in sepsis. Cell Mol. Life Sci. 2017, 74, 3965–3987. [Google Scholar] [CrossRef]
- Deng, W.G.; Tang, S.T.; Tseng, H.P.; Wu, K.K. Melatonin suppresses macrophage cyclooxygenase-2 and inducible nitric oxide synthase expression by inhibiting p52 acetylation and binding. Blood 2006, 108, 518–524. [Google Scholar] [CrossRef]
- Díaz-Casado, M.E.; Lima, E.; García, J.A.; Doerrier, C.; Aranda, P.; Sayed, R.K.; Guerra-Librero, A.; Escames, G.; López, L.C.; Acuña-Castroviejo, D. Melatonin rescues zebrafish embryos from the parkinsonian phenotype restoring the parkin/PINK1/DJ-1/MUL1 network. J. Pineal Res. 2016, 61, 96–107. [Google Scholar] [CrossRef]
- Ren, D.L.; Li, Y.J.; Hu, B.B.; Wang, H.; Hu, B. Melatonin regulates the rhythmic migration of neutrophils in live zebrafish. J. Pineal Res. 2015, 58, 452–460. [Google Scholar] [CrossRef]
- Ren, D.L.; Sun, A.A.; Li, Y.J.; Chen, M.; Ge, S.C.; Hu, B. Exogenous melatonin inhibits neutrophil migration through suppression of ERK activation. J. Endocrinol. 2015, 227, 49–60. [Google Scholar] [CrossRef] [Green Version]
- Hornykiewicz, O. The tropical localization and content of noradrenalin and dopamine (3-hydroxytyramine) in the substantia nigra of normal persons and patients with Parkinson’s disease. Wien. Klin. Wochenschr. 1963, 75, 309–312. [Google Scholar] [PubMed]
- Braak, H.; Sandmann-Keil, D.; Gai, W.; Braak, E. Extensive axonal Lewy neurites in Parkinson’s disease: A novel pathological feature revealed by alpha-synuclein immunocytochemistry. Neurosci. Lett. 1999, 265, 67–69. [Google Scholar] [CrossRef]
- Sun, Z.; Gitler, A.D. Discovery and characterization of three novel synuclein genes in zebrafish. Dev. Dyn. Off. Publ. Am. Assoc. Anat. 2008, 237, 2490–2495. [Google Scholar] [CrossRef]
- Flinn, L.; Mortiboys, H.; Volkmann, K.; Köster, R.W.; Ingham, P.W.; Bandmann, O. Complex I deficiency and dopaminergic neuronal cell loss in parkin-deficient zebrafish (Danio rerio). Brain A J. Neurol. 2009, 132, 1613–1623. [Google Scholar] [CrossRef] [Green Version]
- Kane, L.A.; Lazarou, M.; Fogel, A.I.; Li, Y.; Yamano, K.; Sarraf, S.A.; Banerjee, S.; Youle, R.J. PINK1 phosphorylates ubiquitin to activate Parkin E3 ubiquitin ligase activity. J. Cell Biol. 2014, 205, 143–153. [Google Scholar] [CrossRef]
- Ariga, H.; Takahashi-Niki, K.; Kato, I.; Maita, H.; Niki, T.; Iguchi-Ariga, S.M. Neuroprotective function of DJ-1 in Parkinson’s disease. Oxidative Med. Cell. Longev. 2013, 2013, 683920. [Google Scholar] [CrossRef] [Green Version]
- Ishikawa, S.; Taira, T.; Takahashi-Niki, K.; Niki, T.; Ariga, H.; Iguchi-Ariga, S.M. Human DJ-1-specific transcriptional activation of tyrosine hydroxylase gene. J. Biol. Chem. 2010, 285, 39718–39731. [Google Scholar] [CrossRef] [Green Version]
- Blesa, J.; Trigo-Damas, I.; Quiroga-Varela, A.; Jackson-Lewis, V.R. Oxidative stress and Parkinson’s disease. Front. Neuroanat. 2015, 9, 91. [Google Scholar] [CrossRef] [Green Version]
- Ryan, B.J.; Hoek, S.; Fon, E.A.; Wade-Martins, R. Mitochondrial dysfunction and mitophagy in Parkinson’s: From familial to sporadic disease. Trends Biochem. Sci. 2015, 40, 200–210. [Google Scholar] [CrossRef]
- Qian, L.; Flood, P.M.; Hong, J.S. Neuroinflammation is a key player in Parkinson’s disease and a prime target for therapy. J. Neural Transm. 2010, 117, 971–979. [Google Scholar] [CrossRef] [Green Version]
- Wen, L.; Wei, W.; Gu, W.; Huang, P.; Ren, X.; Zhang, Z.; Zhu, Z.; Lin, S.; Zhang, B. Visualization of monoaminergic neurons and neurotoxicity of MPTP in live transgenic zebrafish. Dev. Biol. 2008, 314, 84–92. [Google Scholar] [CrossRef] [Green Version]
- Richardson, J.R.; Caudle, W.M.; Guillot, T.S.; Watson, J.L.; Nakamaru-Ogiso, E.; Seo, B.B.; Sherer, T.B.; Greenamyre, J.T.; Yagi, T.; Matsuno-Yagi, A.; et al. Obligatory role for complex I inhibition in the dopaminergic neurotoxicity of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP). Toxicol. Sci. Off. J. Soc. Toxicol. 2007, 95, 196–204. [Google Scholar] [CrossRef] [Green Version]
- Ertem, D.H.; Dirican, A.C.; Aydın, A.; Baybas, S.; Sözmen, V.; Ozturk, M.; Altunkaynak, Y. Exploring psychiatric comorbidities and their effects on quality of life in patients with temporal lobe epilepsy and juvenile myoclonic epilepsy. Psychiatry Clin. Neurosci. 2017, 71, 280–288. [Google Scholar] [CrossRef]
- Hauser, R.M.; Henshall, D.C.; Lubin, F.D. The Epigenetics of Epilepsy and Its Progression. Neurosci. A Rev. J. Bringing Neurobiol. Neurol. Psychiatry 2018, 24, 186–200. [Google Scholar] [CrossRef]
- Kanner, A.M. Management of psychiatric and neurological comorbidities in epilepsy. Nat. Rev. Neurol. 2016, 12, 106–116. [Google Scholar] [CrossRef]
- Moavero, R.; Santarone, M.E.; Galasso, C.; Curatolo, P. Cognitive and behavioral effects of new antiepileptic drugs in pediatric epilepsy. Brain Dev. 2017, 39, 464–469. [Google Scholar] [CrossRef]
- Dabak, O.; Altun, D.; Arslan, M.; Yaman, H.; Vurucu, S.; Yesilkaya, E.; Unay, B. Evaluation of Plasma Melatonin Levels in Children With Afebrile and Febrile Seizures. Pediatric Neurol. 2016, 57, 51–55. [Google Scholar] [CrossRef]
- Escames, G.; Acuña Castroviejo, D.; Vives, F. Melatonin-dopamine interaction in the striatal projection area of sensorimotor cortex in the rat. Neuroreport 1996, 7, 597–600. [Google Scholar] [CrossRef]
- León, J.; Macías, M.; Escames, G.; Camacho, E.; Khaldy, H.; Martín, M.; Espinosa, A.; Gallo, M.A.; Acuña-Castroviejo, D. Structure-related inhibition of calmodulin-dependent neuronal nitric-oxide synthase activity by melatonin and synthetic kynurenines. Mol. Pharmacol. 2000, 58, 967–975. [Google Scholar] [CrossRef]
- Khan, S.; Khurana, M.; Vyas, P.; Vohora, D. The role of melatonin and its analogues in epilepsy. Rev. Neurosci. 2021, 32, 49–67. [Google Scholar] [CrossRef] [PubMed]
- Bikjdaouene, L.; Escames, G.; León, J.; Ferrer, J.M.; Khaldy, H.; Vives, F.; Acuña-Castroviejo, D. Changes in brain amino acids and nitric oxide after melatonin administration in rats with pentylenetetrazole-induced seizures. J. Pineal. Res. 2003, 35, 54–60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ren, Q.; Gao, D.; Mou, L.; Zhang, S.; Zhang, M.; Li, N.; Sik, A.; Jin, M.; Liu, K. Anticonvulsant activity of melatonin and its success in ameliorating epileptic comorbidity-like symptoms in zebrafish. Eur. J. Pharmacol. 2021, 912, 174589. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.R.; Samsa, L.A.; Qian, L.; Liu, J. Advances in the Study of Heart Development and Disease Using Zebrafish. J. Cardiovasc. Dev. Dis. 2016, 3, 13. [Google Scholar] [CrossRef]
- Pandi-Perumal, S.R.; BaHammam, A.S.; Ojike, N.I.; Akinseye, O.A.; Kendzerska, T.; Buttoo, K.; Dhandapany, P.S.; Brown, G.M.; Cardinali, D.P. Melatonin and Human Cardiovascular Disease. J. Cardiovasc. Pharmacol. Ther. 2017, 22, 122–132. [Google Scholar] [CrossRef]
- Liu, X.; Gao, Q.; Feng, Z.; Tang, Y.; Zhao, X.; Chen, D.; Feng, X. Protective Effects of Spermidine and Melatonin on Deltamethrin-Induced Cardiotoxicity and Neurotoxicity in Zebrafish. Cardiovasc. Toxicol. 2021, 21, 29–41. [Google Scholar] [CrossRef]
- Yang, M.; Huang, J.; Zhang, S.; Zhao, X.; Feng, D.; Feng, X. Melatonin mitigated circadian disruption and cardiovascular toxicity caused by 6-benzylaminopurine exposure in zebrafish. Ecotoxicol. Environ. Saf. 2021, 223, 112555. [Google Scholar] [CrossRef]
- Haslam, D.W.; James, W.P. Obesity. Lancet 2005, 366, 1197–1209. [Google Scholar] [CrossRef]
- Speakman, J.R. Functional analysis of seven genes linked to body mass index and adiposity by genome-wide association studies: A review. Hum. Hered. 2013, 75, 57–79. [Google Scholar] [CrossRef]
- De Luxán-Delgado, B.; Caballero, B.; Potes, Y.; Rubio-González, A.; Rodríguez, I.; Gutiérrez-Rodríguez, J.; Solano, J.J.; Coto-Montes, A. Melatonin administration decreases adipogenesis in the liver of ob/ob mice through autophagy modulation. J. Pineal Res. 2014, 56, 126–133. [Google Scholar] [CrossRef]
- Oka, T.; Nishimura, Y.; Zang, L.; Hirano, M.; Shimada, Y.; Wang, Z.; Umemoto, N.; Kuroyanagi, J.; Nishimura, N.; Tanaka, T. Diet-induced obesity in zebrafish shares common pathophysiological pathways with mammalian obesity. BMC Physiol. 2010, 10, 21. [Google Scholar] [CrossRef] [Green Version]
- Montalbano, G.; Mania, M.; Abbate, F.; Navarra, M.; Guerrera, M.C.; Laura, R.; Vega, J.A.; Levanti, M.; Germanà, A. Melatonin treatment suppresses appetite genes and improves adipose tissue plasticity in diet-induced obese zebrafish. Endocrine 2018, 62, 381–393. [Google Scholar] [CrossRef]
- Baht, G.S.; Vi, L.; Alman, B.A. The Role of the Immune Cells in Fracture Healing. Curr. Osteoporos Rep. 2018, 16, 138–145. [Google Scholar] [CrossRef] [Green Version]
- Histing, T.; Anton, C.; Scheuer, C.; Garcia, P.; Holstein, J.H.; Klein, M.; Matthys, R.; Pohlemann, T.; Menger, M.D. Melatonin impairs fracture healing by suppressing RANKL-mediated bone remodeling. J. Surg. Res. 2012, 173, 83–90. [Google Scholar] [CrossRef]
- Ladizesky, M.G.; Boggio, V.; Albornoz, L.E.; Castrillón, P.O.; Mautalen, C.; Cardinali, D.P. Melatonin increases oestradiol-induced bone formation in ovariectomized rats. J. Pineal Res. 2003, 34, 143–151. [Google Scholar] [CrossRef]
- Liu, L.; Xu, Y.; Reiter, R.J.; Pan, Y.; Chen, D.; Liu, Y.; Pu, X.; Jiang, L.; Li, Z. Inhibition of ERK1/2 Signaling Pathway is Involved in Melatonin’s Antiproliferative Effect on Human MG-63 Osteosarcoma Cells. Cell. Physiol. Biochem. Int. J. Exp. Cell. Physiol. Biochem. Pharmacol. 2016, 39, 2297–2307. [Google Scholar] [CrossRef]
- Li, T.; Jiang, S.; Lu, C.; Yang, W.; Yang, Z.; Hu, W.; Xin, Z.; Yang, Y. Melatonin: Another avenue for treating osteoporosis? J. Pineal Res. 2019, 66, e12548. [Google Scholar] [CrossRef] [Green Version]
- Kim, H.J.; Kim, H.J.; Bae, M.K.; Kim, Y.D. Suppression of Osteoclastogenesis by Melatonin: A Melatonin Receptor-Independent Action. Int. J. Mol. Sci. 2017, 18, 1142. [Google Scholar] [CrossRef] [Green Version]
- Einhorn, T.A.; Gerstenfeld, L.C. Fracture healing: Mechanisms and interventions. Nat. Rev. Rheumatol. 2015, 11, 45–54. [Google Scholar] [CrossRef] [Green Version]
- Mariotti, M.; Carnovali, M.; Banfi, G. Danio rerio: The Janus of the bone from embryo to scale. Clin. Cases Miner. Bone Metab. Off. J. Ital. Soc. Osteoporos. Miner. Metab. Skelet. Dis. 2015, 12, 188–194. [Google Scholar] [CrossRef]
- Sire, J.Y.; Allizard, F.; Babiar, O.; Bourguignon, J.; Quilhac, A. Scale development in zebrafish (Danio rerio). J. Anat. 1997, 190, 545–561. [Google Scholar] [CrossRef]
- Kobayashi-Sun, J.; Suzuki, N.; Hattori, A.; Yamaguchi, M.; Kobayashi, I. Melatonin suppresses both osteoblast and osteoclast differentiation through repression of epidermal Erk signaling in the zebrafish scale. Biochem. Biophys. Res. Commun. 2020, 530, 644–650. [Google Scholar] [CrossRef] [PubMed]
- Guan, H.; Kong, N.; Tian, R.; Cao, R.; Liu, G.; Li, Y.; Wei, Q.; Jiao, M.; Lei, Y.; Xing, F.; et al. Melatonin increases bone mass in normal, perimenopausal, and postmenopausal osteoporotic rats via the promotion of osteogenesis. J. Transl. Med. 2022, 20, 132. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Hattori, A. Melatonin suppresses osteoclastic and osteoblastic activities in the scales of goldfish. J. Pineal Res. 2002, 33, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Han, N.; Li, F.; Wang, L.; Liu, G.; Hu, M.; Wang, S.; Wei, X.; Guo, J.; Jiang, H.; et al. Melatonin enhances osteoblastogenesis of senescent bone marrow stromal cells through NSD2-mediated chromatin remodelling. Clin. Transl. Med. 2022, 12, e746. [Google Scholar] [CrossRef]
- Impellizzeri, P.; Vinci, E.; Gugliandolo, M.C.; Cuzzocrea, F.; Larcan, R.; Russo, T.; Gravina, M.R.; Arena, S.; D’Angelo, G.; Gitto, E.; et al. Premedication with melatonin vs midazolam: Efficacy on anxiety and compliance in paediatric surgical patients. Eur. J. Pediatrics 2017, 176, 947–953. [Google Scholar] [CrossRef]
- Marseglia, L.; D’Angelo, G.; Manti, S.; Aversa, S.; Arrigo, T.; Reiter, R.J.; Gitto, E. Analgesic, anxiolytic and anaesthetic effects of melatonin: New potential uses in pediatrics. Int. J. Mol. Sci. 2015, 16, 1209–1220. [Google Scholar] [CrossRef] [Green Version]
- Patel, T.; Kurdi, M.S. A comparative study between oral melatonin and oral midazolam on preoperative anxiety, cognitive, and psychomotor functions. J. Anaesthesiol. Clin. Pharmacol. 2015, 31, 37–43. [Google Scholar] [CrossRef]
- Acuña Castroviejo, D.; Rosenstein, R.E.; Romeo, H.E.; Cardinali, D.P. Changes in gamma-aminobutyric acid high affinity binding to cerebral cortex membranes after pinealectomy or melatonin administration to rats. Neuroendocrinology 1986, 43, 24–31. [Google Scholar] [CrossRef]
- Acuña-Castroviejo, D.; Lowenstein, P.R.; Rosenstein, R.; Cardinali, D.P. Diurnal variations of benzodiazepine binding in rat cerebral cortex: Disruption by pinealectomy. J. Pineal. Res. 1986, 3, 101–109. [Google Scholar] [CrossRef]
- Gomar, M.D.; Castillo, J.L.; del Aguila, C.M.; Fernández, B.; Acuña-Castroviejo, D. Intracerebroventricular injection of naloxone blocks melatonin-dependent brain [3H]flunitrazepam binding. Neuroreport 1993, 4, 987–990. [Google Scholar] [CrossRef]
- Dereje, S.; Sawyer, S.; Oxendine, S.E.; Zhou, L.; Kezios, Z.D.; Wong, R.Y.; Godwin, J.; Perrin, F. Comparing behavioral responses across multiple assays of stress and anxiety in zebrafish (Danio rerio). Behaviour 2012, 149, 1205–1240. [Google Scholar] [CrossRef] [Green Version]
- Philpott, C.; Donack, C.J.; Cousin, M.A.; Pierret, C. Reducing the noise in behavioral assays: Sex and age in adult zebrafish locomotion. Zebrafish 2012, 9, 191–194. [Google Scholar] [CrossRef] [Green Version]
- Reolon, G.K.; de Melo, G.M.; da Rosa, J.; Barcellos, L.J.G.; Bonan, C.D. Sex and the housing: Effects on behavior, cortisol levels and weight in zebrafish. Behav. Brain Res. 2018, 336, 85–92. [Google Scholar] [CrossRef]
- Genario, R.; Giacomini, A.; de Abreu, M.S.; Marcon, L.; Demin, K.A.; Kalueff, A.V. Sex differences in adult zebrafish anxiolytic-like responses to diazepam and melatonin. Neurosci. Lett. 2020, 714, 134548. [Google Scholar] [CrossRef]
- Lunkes, L.C.; Paiva, I.M.; Egger, R.C.; Braga, W.F.; Alvarez-Leite, J.I.; da Cunha Barreto-Vianna, A.R.; Murgas, L.D.S. Melatonin administration attenuates acute stress by inducing sleep state in zebrafish (Danio rerio). Comp. Biochem. Physiol. Toxicol. Pharmacol. 2021, 246, 109044. [Google Scholar] [CrossRef]
- Kondratov, R.V. A role of the circadian system and circadian proteins in aging. Ageing Res. Rev. 2007, 6, 12–27. [Google Scholar] [CrossRef]
- Yu, L.; Tucci, V.; Kishi, S.; Zhdanova, I.V. Cognitive aging in zebrafish. PLoS ONE 2006, 1, e14. [Google Scholar] [CrossRef]
- Keller, E.T.; Murtha, J.M. The use of mature zebrafish (Danio rerio) as a model for human aging and disease. Comp. Biochem. Physiol. Toxicol. Pharmacol. 2004, 138, 335–341. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aranda-Martínez, P.; Fernández-Martínez, J.; Ramírez-Casas, Y.; Guerra-Librero, A.; Rodríguez-Santana, C.; Escames, G.; Acuña-Castroviejo, D. The Zebrafish, an Outstanding Model for Biomedical Research in the Field of Melatonin and Human Diseases. Int. J. Mol. Sci. 2022, 23, 7438. https://doi.org/10.3390/ijms23137438
Aranda-Martínez P, Fernández-Martínez J, Ramírez-Casas Y, Guerra-Librero A, Rodríguez-Santana C, Escames G, Acuña-Castroviejo D. The Zebrafish, an Outstanding Model for Biomedical Research in the Field of Melatonin and Human Diseases. International Journal of Molecular Sciences. 2022; 23(13):7438. https://doi.org/10.3390/ijms23137438
Chicago/Turabian StyleAranda-Martínez, Paula, José Fernández-Martínez, Yolanda Ramírez-Casas, Ana Guerra-Librero, César Rodríguez-Santana, Germaine Escames, and Darío Acuña-Castroviejo. 2022. "The Zebrafish, an Outstanding Model for Biomedical Research in the Field of Melatonin and Human Diseases" International Journal of Molecular Sciences 23, no. 13: 7438. https://doi.org/10.3390/ijms23137438







