Bioavailability Improvement Strategies for Icariin and Its Derivates: A Review
Abstract
:1. Introduction
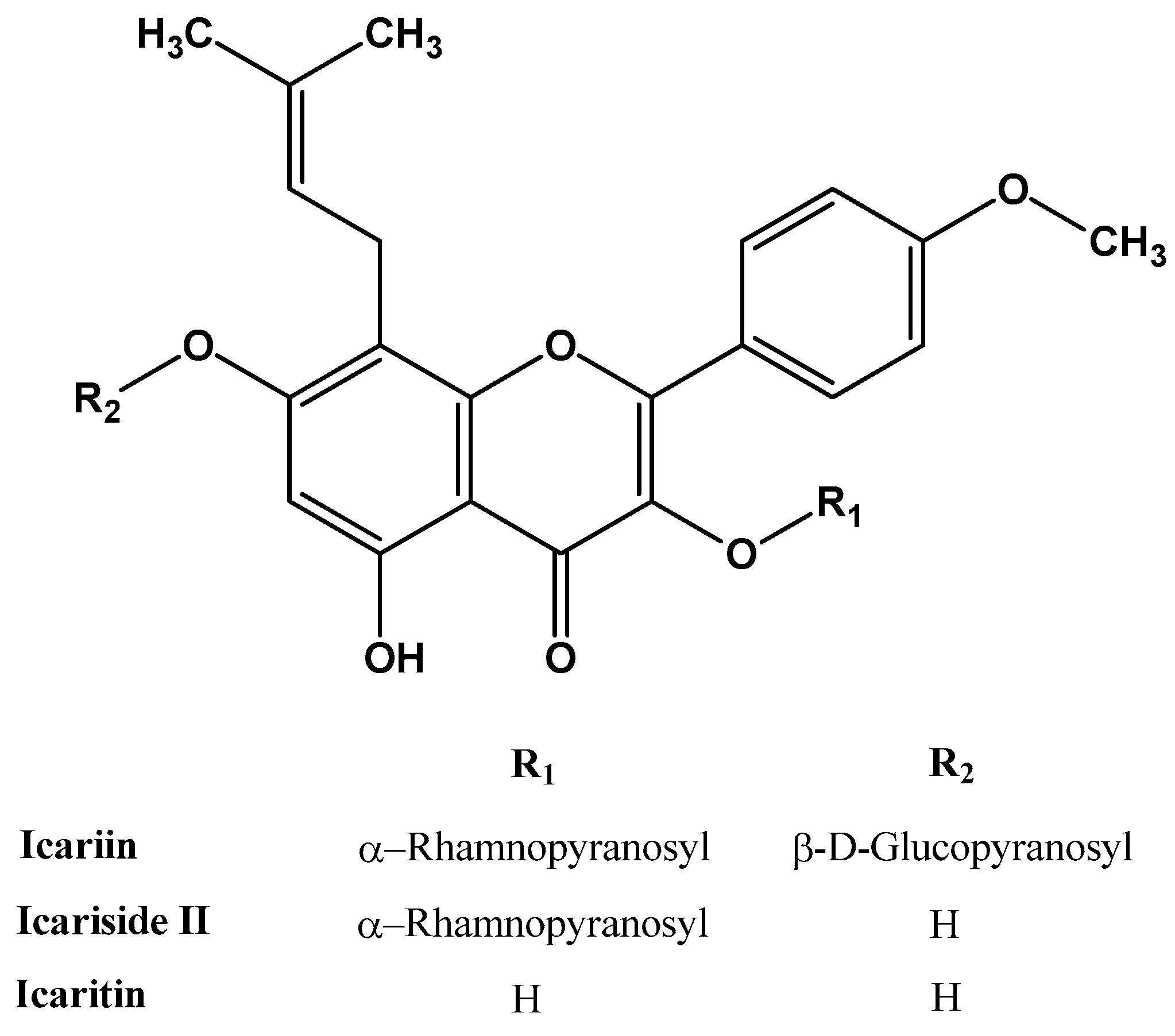
2. Icariin and Its Derivates
2.1. Extraction and Preparation
2.2. Medicinal Properties
3. Bioavailability Improvements
3.1. Pharmaceutical Technologies
3.1.1. Complex Formation with Phospholipids
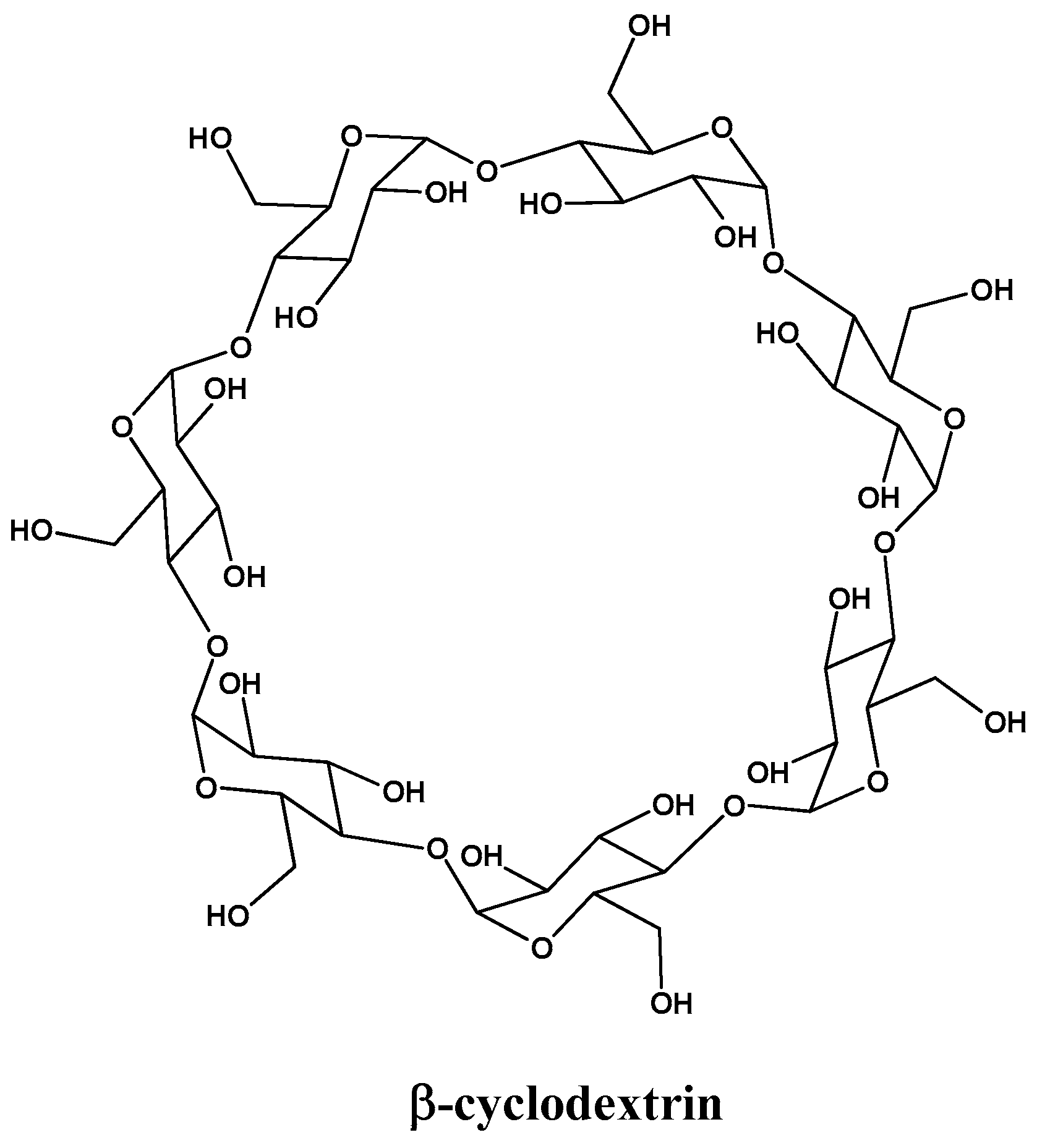
3.1.2. Complex Formation with Cyclodextrins
3.2. Nanotechnologies
3.2.1. Formation of Micelles
3.2.2. Nanocarriers
3.2.3. Nanogels
3.2.4. Nanocrystals
3.2.5. Microspheres
3.2.6. Extracellular Vesicles
3.2.7. Solid Lipid Nanoparticles
3.3. Structural Transformation
3.4. Absorption Enhancement
Colon-Specific Drug Delivery
4. Conclusions and Future Prospects
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Xiao, J. Dietary flavonoid aglycones and their glycosides: Which show better biological significance? Crit. Rev. Food Sci. Nutr. 2017, 57, 1874–1905. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, G.B.; Van Camp, J.; Smagghe, G.; Raes, K.; Mackie, A. Flavonoid–gastrointestinal mucus interaction and its potential role in regulating flavonoid bioavailability and mucosal biophysical properties. Food Res. Int. 2016, 88, 342–347. [Google Scholar] [CrossRef]
- Slámová, K.; Kapešová, J.; Valentová, K. “Sweet flavonoids”: Glycosidase-catalyzed modifications. Int. J. Mol. Sci. 2018, 19, 2126. [Google Scholar] [CrossRef] [Green Version]
- He, C.; Wang, Z.; Shi, J. Chapter Seven—Pharmacological effects of icariin. In Advances in Pharmacology; Du, G., Ed.; Academic Press: Cambridge, MA, USA, 2020; Volume 87, pp. 179–203. [Google Scholar]
- Wu, J.; Guan, M.; Wong, P.F.; Yu, H.; Dong, J.; Xu, J. Icariside II potentiates paclitaxel-induced apoptosis in human melanoma A375 cells by inhibiting TLR4 signaling pathway. Food Chem. Toxicol. 2012, 50, 3019–3024. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Shi, Y.-M.; Xia, Y.-Z.; Guo, C.; Yang, L.; Kong, L.-Y. Icariside II-induced mitochondrion and lysosome mediated apoptosis is counterbalanced by an autophagic salvage response in hepatoblastoma. Cancer Lett. 2015, 366, 19–31. [Google Scholar]
- Adlimoghaddam, A.; Neuendorff, M.; Roy, B.; Albensi, B.C. A review of clinical treatment considerations of donepezil in severe Alzheimer’s disease. CNS Neurosci. Ther. 2018, 24, 876–888. [Google Scholar] [CrossRef] [Green Version]
- Vazhappilly, C.G.; Amararathna, M.; Cyril, A.C.; Linger, R.; Matar, R.; Merheb, M.; Ramadan, W.S.; Radhakrishnan, R.; Rupasinghe, H.P.V. Current methodologies to refine bioavailability, delivery, and therapeutic efficacy of plant flavonoids in cancer treatment. J. Nutr. Biochem. 2021, 94, 108623. [Google Scholar] [CrossRef]
- Jin, J.; Wang, H.; Hua, X.; Chen, D.; Huang, C.; Chen, Z. An outline for the pharmacological effect of icariin in the nervous system. Eur. J. Pharmacol. 2019, 842, 20–32. [Google Scholar] [CrossRef]
- Zhao, J.; Yang, J.; Xie, Y. Improvement strategies for the oral bioavailability of poorly water-soluble flavonoids: An overview. Int. J. Pharm. 2019, 570, 118642. [Google Scholar] [CrossRef]
- Wu, C.; Zhang, J.; Zhou, T.; Guo, B.; Wang, Y.; Hou, J. Simultaneous determination of seven flavonoids in dog plasma by ultra-performance liquid chromatography–tandem mass spectrometry and its application to a bioequivalence study of bioactive components in Herba Epimedii and Er-Xian Decoction. J. Pharm. Biomed. Anal. 2011, 54, 186–191. [Google Scholar] [CrossRef]
- Arief, Z.M.; Shawl, A.S.; Munshi, A.H. Altitudinal variation in pharmacologically active compounds of wild and cultivated populations of Epimedium elatum. J. Appl Res. Med. Aromat Plants 2016, 3, 48–51. [Google Scholar] [CrossRef]
- Wu, C.S.; Guo, B.L.; Sheng, Y.X.; Zhang, J.L. Simultaneous determination of seven flavonoids in Epimedium by liquid chromatography–tandem mass spectrometry method. Chin. Chem. Lett. 2008, 19, 329–332. [Google Scholar] [CrossRef]
- Wu, H.; Kim, M.; Han, J. Icariin metabolism by human intestinal microflora. Molecules 2016, 21, 1158. [Google Scholar] [CrossRef] [PubMed]
- Cheng, T.; Sheng, T.; Yi, Y.; Zhang, T.; Han, H. Metabolism profiles of icariin in rats using ultra-high performance liquid chromatography coupled with quadrupole time-of-flight tandem mass spectrometry and in vitro enzymatic study. J. Chromatogr. B 2016, 1033, 353–360. [Google Scholar] [CrossRef]
- Liu, R.; Li, A.; Sun, A.; Cui, J.; Kong, L. Preparative isolation and purification of three flavonoids from the Chinese medicinal plant Epimedium koreamum Nakai by high-speed counter-current chromatography. J. Chromatogr. A 2005, 1064, 53–57. [Google Scholar] [CrossRef]
- Xia, Q.; Xu, D.; Huang, Z.; Liu, J.; Wang, X.; Wang, X.; Liu, S. Preparation of icariside II from icariin by enzymatic hydrolysis method. Fitoterapia 2010, 81, 437–442. [Google Scholar] [CrossRef]
- Park, J.-S.; Park, H.-Y.; Rho, H.-S.; Ahn, S.-M.; Kim, D.-H.; Chang, I.-S. Statistically designed enzymatic hydrolysis for optimized production of icariside II as a novel melanogenesis inhibitor. J. Microbiol. Biotechnol. 2008, 18, 110–117. [Google Scholar]
- Shen, Y.; Wang, M.; Zhou, J.; Chen, Y.; Xu, L.; Wu, M.; Xia, G.; Tam, J.P.; Yu, J.; Teng, X. Eco-efficient biphasic enzymatic hydrolysis for the green production of rare baohuoside I. Enzyme Microb. Technol. 2019, 131, 109431. [Google Scholar] [CrossRef]
- Bi, Z.; Zhang, W.; Yan, X. Anti-inflammatory and immunoregulatory effects of icariin and icaritin. Biomed. Pharmacother. 2022, 151, 113180. [Google Scholar] [CrossRef]
- Li, Q.; Ge, L.; Zheng, D.; Zhang, X.; Zhao, L. Screening and characterization of a GH78 α-l-rhamnosidase from Aspergillus terreus and its application in the bioconversion of icariin to icaritin with recombinant β-glucosidase. Enzyme Microb. Technol. 2022, 153, 109940. [Google Scholar] [CrossRef]
- Yang, L.; Zhang, C.; Kong, L.-y. Blockade of epidermal growth factor receptor/mammalian target of rapamycin pathway by Icariside II results in reduced cell proliferation of osteosarcoma cells. Food Chem. Toxicol. 2014, 73, 7–16. [Google Scholar]
- Lee, K.-S.; Lee, H.-J.; Ahn, K.S.; Kim, S.-H.; Nam, D.; Kim, D.K.; Choi, D.-Y.; Ahn, K.-S.; Lu, J.; Kim, S.-H. Cyclooxygenase-2/prostaglandin E2 pathway mediates icariside II induced apoptosis in human PC-3 prostate cancer cells. Cancer Lett. 2009, 280, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.-S.; Thakur, K.; Hu, F.; Zhang, J.-G.; Wei, Z.-J. Icariside II inhibits tumorigenesis via inhibiting AKT/Cyclin E/CDK 2 pathway and activating mitochondria-dependent pathway. Pharmacol. Res. 2020, 152, 104616. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Wu, J.; Luo, Q.; Mo, S.; Lyu, Y.; Wei, Y.; Dong, J. The anticancer properties of Herba Epimedii and its main bioactive componentsicariin and icariside II. Nutrients 2016, 8, 563. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhang, H.; Dai, L.; Song, T.; Li, P.; Liu, Y.; Wang, L. Arsenic trioxide and icariin show synergistic anti-leukemic activity. Cell Biochem. Biophys. 2015, 73, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Lan, J.; Du, H.; Zhang, X.; Li, A.; Zhang, X.; Liu, Y.; Zhang, J.; Zhang, C.; Ding, Y. Icariside II induces cell cycle arrest and differentiation via TLR8/MyD88/p38 pathway in acute myeloid leukemia cells. Eur. J. Pharmacol. 2019, 846, 12–22. [Google Scholar] [CrossRef]
- Sun, Y.-S.; Thakur, K.; Hu, F.; Cespedes-Acuña, C.L.; Zhang, J.-G.; Wei, Z.-J. Icariside II suppresses cervical cancer cell migration through JNK modulated matrix metalloproteinase-2/9 inhibition in vitro and in vivo. Biomed. Pharmacother. 2020, 125, 110013. [Google Scholar] [CrossRef]
- Kim, S.-H.; Ahn, K.S.; Jeong, S.-J.; Kwon, T.-R.; Jung, J.H.; Yun, S.-M.; Han, I.; Lee, S.-G.; Kim, D.K.; Kang, M. Janus activated kinase 2/signal transducer and activator of transcription 3 pathway mediates icariside II-induced apoptosis in U266 multiple myeloma cells. Eur. J. Pharmacol. 2011, 654, 10–16. [Google Scholar] [CrossRef]
- Sharma, S.; Khan, V.; Dhyani, N.; Najmi, A.; Haque, S. Icariin attenuates isoproterenol-induced cardiac toxicity in Wistar rats via modulating cGMP level and NF-κB signaling cascade. Hum. Exp. Toxicol. 2020, 39, 117–126. [Google Scholar] [CrossRef]
- Ren, L.; Wang, Z.; Hua, Q.; Xie, H.; Tang, S. Icaritin prevents vascular calcification in mice. Zhong Nan Da Xue Xue Bao Yi Xue Ban 2019, 44, 35–39. [Google Scholar]
- Liu, X.-Y.; Liao, H.-h.; Feng, H.; Zhang, N.; Yang, J.-j.; Li, W.-j.; Chen, S.; Deng, W.; Tang, Q.-Z. Icariside II attenuates cardiac remodeling via AMPKα2/mTORC1 in vivo and in vitro. J. Pharmacol. Sci. 2018, 138, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Fu, S.; Li, Y.-L.; Wu, Y.-T.; Yue, Y.; Qian, Z.-Q.; Yang, D.-L. Icariside II attenuates myocardial fibrosis by inhibiting nuclear factor-κB and the TGF-β1/Smad2 signalling pathway in spontaneously hypertensive rats. Biomed. Pharmacother. 2018, 100, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Zheng, T.; Zhang, T.; Zhang, W.; Lv, K.; Jia, D.; Yang, F.; Sun, Y.; Lian, J.; Wang, R. Icariside II facilitates the differentiation of ADSCs to schwann cells and restores erectile dysfunction through regulation of miR-33/GDNF axis. Biomed. Pharmacother. 2020, 125, 109888. [Google Scholar] [CrossRef]
- Li, X.; Yang, H.F.; Chen, Y.; Pei, L.J.; Jiang, R. Effect of the icariin on endothelial microparticles, endothelial progenitor cells, platelets, and erectile function in spontaneously hypertensive rats. Andrology 2022, 10, 576–584. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.-B.; Zhou, C.-H. The effect of extracts from Herba Epimedii and Semen Cuscutae on the function of male reproduction. Chin. Pharm. J. 1994, 29, 89. [Google Scholar]
- Chen, M.; Hao, J.; Yang, Q.; Li, G. Effects of icariin on reproductive functions in male rats. Molecules 2014, 19, 9502–9514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, L.; Li, D.; Chen, C.; Wang, G.; Shi, J.; Zhang, F. Activation of Nrf2 signaling by Icariin protects against 6-OHDA-induced neurotoxicity. Biotechnol. Appl. Biochem. 2019, 66, 465–471. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Deng, Y.; Gao, J.; Zeng, L.; Gong, Q. Icariside II ameliorates ibotenic acid-induced cognitive impairment and apoptotic response via modulation of MAPK pathway in rats. Phytomedicine 2018, 41, 74–81. [Google Scholar] [CrossRef]
- Yan, B.-Y.; Pan, C.-S.; Mao, X.-W.; Yang, L.; Liu, Y.-Y.; Yan, L.; Mu, H.-N.; Wang, C.-S.; Sun, K.; Liao, F.-L. Icariside II improves cerebral microcirculatory disturbance and alleviates hippocampal injury in gerbils after ischemia–reperfusion. Brain Res. 2014, 1573, 63–73. [Google Scholar] [CrossRef]
- Deng, Y.; Xiong, D.; Yin, C.; Liu, B.; Shi, J.; Gong, Q. Icariside II protects against cerebral ischemia–reperfusion injury in rats via nuclear factor-κB inhibition and peroxisome proliferator-activated receptor up-regulation. Neurochem. Int. 2016, 96, 56–61. [Google Scholar] [CrossRef]
- Zhou, J.; Deng, Y.; Li, F.; Yin, C.; Shi, J.; Gong, Q. Icariside II attenuates lipopolysaccharide-induced neuroinflammation through inhibiting TLR4/MyD88/NF-κB pathway in rats. Biomed. Pharmacother. 2019, 111, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Eastell, R.; O’Neill, T.W.; Hofbauer, L.C.; Langdahl, B.; Reid, I.R.; Gold, D.T.; Cummings, S.R. Postmenopausal osteoporosis. Nat. Rev. Dis. Primers 2016, 2, 16069. [Google Scholar] [CrossRef] [PubMed]
- Xi, Y.; Jiang, T.; Yu, J.; Xue, M.; Xu, N.; Wen, J.; Wang, W.; He, H.; Ye, X. Preliminary studies on the anti-osteoporosis activity of Baohuoside I. Biomed. Pharmacother. 2019, 115, 108850. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Han, B.; Wei, Y.; Jing, J.; Li, J. Icariin promotes fracture healing in ovariectomized rats. Med. Sci. Monit. 2020, 26, e924554-1–e924554-8. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-Y.; Chen, Y.-P.; Zhang, C.; Zhang, X.; Xia, T.; Han, J.; Yang, N.; Song, S.-L.; Xu, C.-H. Icariin accelerates fracture healing via activation of the WNT1/β-catenin osteogenic signaling pathway. Curr. Pharm. Biotechnol. 2020, 21, 1645–1653. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, Y.; Zhang, C.; Zhang, X.; Xia, T.; Han, J.; Song, S.; Xu, C.; Chen, F. Effects of icariin on the fracture healing in young and old rats and its mechanism. Pharm. Biol. 2021, 59, 1243–1253. [Google Scholar] [CrossRef]
- Xu, F.; Wu, Q.; Li, L.; Gong, J.; Huo, R.; Cui, W. Icariside II: Anticancer Potential and Molecular Targets in Solid Cancers. Front. Pharmacol. 2021, 12, 663776. [Google Scholar] [CrossRef]
- Epimedium Grandiflorum. Available online: https://commons.wikimedia.org/wiki/File:Epimedium_grandiflorum.JPG (accessed on 7 June 2022).
- Tan, O.J.; Loo, H.L.; Thiagarajah, G.; Palanisamy, U.D.; Sundralingam, U. Improving oral bioavailability of medicinal herbal compounds through lipid-based formulations–A Scoping Review. Phytomedicine 2021, 153651. [Google Scholar] [CrossRef]
- Xie, J.; Zhang, K.; Wang, H.; Yang, X.; Mao, W.; Liu, C.; Wang, N. Determination of the Solubility, Dissolution Enthalpy, and Entropy of Icariin in Acetone, Acetoacetate, Chloroform, and Light Petroleum. Nat. Prod. Commun. 2020, 15, 1934578X20971491. [Google Scholar] [CrossRef]
- Dong, M.; Wu, S.; Xu, H.; Yu, X.; Wang, L.; Bai, H.; Niu, W. FBS-Derived Exosomes as a Natural Nano-Scale Carrier for Icariin Promote Osteoblast Proliferation. Front. Bioeng. Biotechnol. 2021, 9, 146. [Google Scholar] [CrossRef]
- Li, C.; Chen, F.-F.; Jia, X.-B.; Feng, L.; Tan, X.-B. Effects and mechanism of epimedium polysaccharide on solubility of icariin and baohuoside I. China J. Chin. Mater. Med. 2021, 46, 5825–5831. [Google Scholar]
- Li, Y.; Sun, S.; Chang, Q.; Zhang, L.; Wang, G.; Chen, W.; Miao, X.; Zheng, Y. A strategy for the improvement of the bioavailability and antiosteoporosis activity of BCS IV flavonoid glycosides through the formulation of their lipophilic aglycone into nanocrystals. Mol. Pharm. 2013, 10, 2534–2542. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.; Chen, J.; Liu, S. Pharmacokinetics of icariin in rats. Chin. Pharm. J. 1999, 34, 33–36. [Google Scholar]
- Xin, X.; Fan, G.-J.; Sun, Z.; Zhang, N.; Li, Y.; Lan, R.; Chen, L.; Dong, P. Biotransformation of major flavonoid glycosides in herb epimedii by the fungus Cunninghamella blakesleana. J. Mol. Catal. B Enzym. 2015, 122, 141–146. [Google Scholar] [CrossRef]
- Storm, G.; Belliot, S.O.; Daemen, T.; Lasic, D.D. Surface modification of nanoparticles to oppose uptake by the mononuclear phagocyte system. Adv. Drug Del. Rev. 1995, 17, 31–48. [Google Scholar] [CrossRef]
- Loftsson, T. Drug solubilization by complexation. Int. J. Pharm. 2017, 531, 276–280. [Google Scholar] [CrossRef]
- Kuche, K.; Bhargavi, N.; Dora, C.P.; Jain, S. Drug-phospholipid complex—A go through strategy for enhanced oral bioavailability. AAPS PharmSciTech 2019, 20, 43. [Google Scholar] [CrossRef]
- Jin, X.; Zhang, Z.-h.; Sun, E.; Qian, Q.; Tan, X.-b.; Jia, X.-b. Preparation of a nanoscale baohuoside I-phospholipid complex and determination of its absorption: In vivo and in vitro evaluations. Int. J. Nanomed. 2012, 7, 4907. [Google Scholar]
- Song, J.; Huang, H.; Xia, Z.; Wei, Y.; Yao, N.; Zhang, L.; Yan, H.; Jia, X.; Zhang, Z. TPGS/phospholipids mixed micelles for delivery of icariside II to multidrug-resistant breast cancer. Integr. Cancer Ther. 2016, 15, 390–399. [Google Scholar] [CrossRef] [Green Version]
- Mu, L.; Feng, S.-S. PLGA/TPGS Nanoparticles for Controlled Release of Paclitaxel: Effects of the Emulsifier and Drug Loading Ratio. Pharm. Res. 2003, 20, 1864–1872. [Google Scholar] [CrossRef]
- Jin, X.; Zhang, Z.-H.; Sun, E.; Tan, X.-B.; Zhu, F.-X.; Jia, X.-B. A novel drug–phospholipid complex loaded micelle for baohuoside I enhanced oral absorption: In vivo and in vivo evaluations. Drug Dev. Ind. Pharm. 2013, 39, 1421–1430. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Shen, J.; Yang, X.; Jin, Y.; Yang, Z.; Wang, R.; Zhang, F.; Linhardt, R.J. Mechanism of enhanced oral absorption of akebia saponin D by a self-nanoemulsifying drug delivery system loaded with phospholipid complex. Drug Dev. Ind. Pharm. 2019, 45, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Pan, C.; Cao, X.; Tang, L.; Zhang, Y.; He, H.; Yin, T.; Piao, H.; Tang, X. Phospholipid Complex of ICA and ICA II Prepared by Wet Media Milling for Improving Bioavailability. Eur. J. Lipid Sci. Technol. 2018, 120, 1700317. [Google Scholar] [CrossRef]
- Jacob, S.; Nair, A.B. Cyclodextrin complexes: Perspective from drug delivery and formulation. Drug Dev. Res. 2018, 79, 201–217. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, Q.-S.; Cui, Y.-L.; Meng, F.-C.; Lin, K.-M. Changes in the intestinal absorption mechanism of icariin in the nanocavities of cyclodextrins. Int. J. Nanomed. 2012, 7, 4239. [Google Scholar]
- Jin, X.; Zhang, Z.-h.; Sun, E.; Jia, X.-B. β-cyclodextrin assistant flavonoid glycosides enzymatic hydrolysis. Pharmacogn. Mag. 2013, 9, S11. [Google Scholar]
- Cui, L.; Zhang, Z.; Sun, E.; Jia, X.; Qian, Q. Effect of β-cyclodextrin complexation on solubility and enzymatic hydrolysis rate of icariin. J. Nat. Sci. Biol. Med. 2013, 4, 201. [Google Scholar] [PubMed] [Green Version]
- Choi, S.; Lee, Y.S.; Jo, H.-S.; Jeong, W.K.; Kim, H.-J.; Song, M.H.; Park, K.; Kim, S.E. Investigating the in vitro osteogenic properties of the inclusion nanocarrier of icariin with beta-cyclodextrin-alginate. Appl. Sci. 2020, 10, 4137. [Google Scholar] [CrossRef]
- Zhang, Y.; Cui, Y.-L.; Qi, A.-D.; Yao, K.-D. Characterization of Icarrin and Icariin-2-Hydroxypropyl-beta-Cyclodextrin Inclusion Complex-Loading Poly (L-Lactic Acid) Scaffolds. In Proceedings of the 2009 3rd International Conference on Bioinformatics and Biomedical Engineering, Beijing, China, 11–13 June 2009; pp. 1–4. [Google Scholar]
- Mensah, A.; Chen, Y.; Asinyo, B.K.; Howard, E.K.; Narh, C.; Huang, J.; Wei, Q. Bioactive Icariin/β-CD-IC/Bacterial Cellulose with Enhanced Biomedical Potential. Nanomaterials 2021, 11, 387. [Google Scholar] [CrossRef]
- Saokham, P.; Muankaew, C.; Jansook, P.; Loftsson, T. Solubility of cyclodextrins and drug/cyclodextrin complexes. Molecules 2018, 23, 1161. [Google Scholar] [CrossRef] [Green Version]
- Suvarna, V.; Gujar, P.; Murahari, M. Complexation of phytochemicals with cyclodextrin derivatives–An insight. Biomed. Pharmacother. 2017, 88, 1122–1144. [Google Scholar] [CrossRef] [PubMed]
- Mei, H.; Cai, S.; Huang, D.; Gao, H.; Cao, J.; He, B. Carrier-free nanodrugs with efficient drug delivery and release for cancer therapy: From intrinsic physicochemical properties to external modification. Bioact. Mater. 2022, 8, 220–240. [Google Scholar] [CrossRef] [PubMed]
- Moradi, S.; Taran, M.; Mohajeri, P.; Sadrjavadi, K.; Sarrami, F.; Karton, A.; Shahlaei, M. Study of dual encapsulation possibility of hydrophobic and hydrophilic drugs into a nanocarrier based on bio-polymer coated graphene oxide using density functional theory, molecular dynamics simulation and experimental methods. J. Mol. Liq. 2018, 262, 204–217. [Google Scholar] [CrossRef]
- Zhang, A.; Meng, K.; Liu, Y.; Pan, Y.; Qu, W.; Chen, D.; Xie, S. Absorption, distribution, metabolism, and excretion of nanocarriers in vivo and their influences. Adv. Colloid Interface Sci. 2020, 284, 102261. [Google Scholar] [CrossRef]
- Luong, D.; Kesharwani, P.; Killinger, B.A.; Moszczynska, A.; Sarkar, F.H.; Padhye, S.; Rishi, A.K.; Iyer, A.K. Solubility enhancement and targeted delivery of a potent anticancer flavonoid analogue to cancer cells using ligand decorated dendrimer nano-architectures. J. Colloid Interface Sci. 2016, 484, 33–43. [Google Scholar] [CrossRef]
- Wicki, A.; Witzigmann, D.; Balasubramanian, V.; Huwyler, J. Nanomedicine in cancer therapy: Challenges, opportunities, and clinical applications. J. Controlled Release 2015, 200, 138–157. [Google Scholar] [CrossRef]
- Lu, Y.; Zhang, E.; Yang, J.; Cao, Z. Strategies to improve micelle stability for drug delivery. Nano Res. 2018, 11, 4985–4998. [Google Scholar] [CrossRef]
- Rahdar, A.; Hajinezhad, M.R.; Sargazi, S.; Barani, M.; Karimi, P.; Velasco, B.; Taboada, P.; Pandey, S.; Bameri, Z.; Zarei, S. Pluronic F127/carfilzomib-based nanomicelles as promising nanocarriers: Synthesis, characterization, biological, and in silico evaluations. J. Mol. Liq. 2022, 346, 118271. [Google Scholar] [CrossRef]
- Shen, H.; Liu, Y.; Zhang, H.; Ding, P.; Zhang, L.; Zhang, L.; Ju, J. Enhancing the oral bioavailability of baicalein via Solutol® HS15 and Poloxamer 188 mixed micelles system. J. Pharm. Pharmacol. 2019, 71, 765–773. [Google Scholar] [CrossRef]
- Hou, J.; Wang, J.; Sun, E.; Yang, L.; Yan, H.-M.; Jia, X.-B.; Zhang, Z.-H. Preparation and evaluation of icariside II-loaded binary mixed micelles using Solutol HS15 and Pluronic F127 as carriers. Drug Deliv. Lett. 2016, 23, 3248–3256. [Google Scholar] [CrossRef] [Green Version]
- Yan, H.; Song, J.; Jia, X.; Zhang, Z. Hyaluronic acid-modified didecyldimethylammonium bromide/da-tocopheryl polyethylene glycol succinate mixed micelles for delivery of baohuoside I against non-small cell lung cancer: In vitro and in vivo evaluation. Drug Deliv. Lett. 2017, 24, 30–39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nocito, M.C.; De Luca, A.; Prestia, F.; Avena, P.; La Padula, D.; Zavaglia, L.; Sirianni, R.; Casaburi, I.; Puoci, F.; Chimento, A. Antitumoral Activities of Curcumin and Recent Advances to ImProve Its Oral Bioavailability. Biomedicines 2021, 9, 1476. [Google Scholar] [CrossRef] [PubMed]
- Kusumoto, K.-i.; Ishikawa, T. Didodecyldimethylammonium bromide (DDAB) induces caspase-mediated apoptosis in human leukemia HL-60 cells. J. Controlled Release 2010, 147, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Kumar, C.S.; Raja, M.; Sundar, D.S.; Antoniraj, M.G.; Ruckmani, K. Hyaluronic acid co-functionalized gold nanoparticle complex for the targeted delivery of metformin in the treatment of liver cancer (HepG2 cells). Carbohydr. Polym. 2015, 128, 63–74. [Google Scholar] [CrossRef]
- Han, L.-Y.; Wu, Y.-L.; Zhu, C.-Y.; Wu, C.-S.; Yang, C.-R. Improved pharmacokinetics of icariin (ICA) within formulation of PEG-PLLA/PDLA-PNIPAM polymeric micelles. Pharmaceutics 2019, 11, 51. [Google Scholar] [CrossRef] [Green Version]
- Vashist, A.; Kaushik, A.; Vashist, A.; Bala, J.; Nikkhah-Moshaie, R.; Sagar, V.; Nair, M. Nanogels as potential drug nanocarriers for CNS drug delivery. Drug Discov. Today 2018, 23, 1436–1443. [Google Scholar] [CrossRef]
- Raemdonck, K.; Demeester, J.; De Smedt, S. Advanced nanogel engineering for drug delivery. Soft Matter 2009, 5, 707–715. [Google Scholar] [CrossRef]
- Xu, D.; Lu, Y.-R.; Kou, N.; Hu, M.-J.; Wang, Q.-S.; Cui, Y.-L. Intranasal delivery of icariin via a nanogel-thermoresponsive hydrogel compound system to improve its antidepressant-like activity. Int. J. Pharm. 2020, 586, 119550. [Google Scholar] [CrossRef]
- He, L.; Yang, J.; Lu, J.; Xiao, Y.; Fan, Y.; Zhang, X. Preparation and characterization of a novel hyaluronic acid–icariin conjugate hydrogel. Mater. Lett. 2014, 136, 41–44. [Google Scholar] [CrossRef]
- Yan, H.; Zhou, Z.; Huang, T.; Peng, C.; Liu, Q.; Zhou, H.; Zeng, W.; Liu, L.; Ou, B.; He, S. Controlled release in vitro of icariin from gelatin/hyaluronic acid composite microspheres. Polym. Bull. 2016, 73, 1055–1066. [Google Scholar] [CrossRef]
- Somiya, M.; Yoshioka, Y.; Ochiya, T. Biocompatibility of highly purified bovine milk-derived extracellular vesicles. J Extracell Vesicles 2018, 7, 1440132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moloudizargari, M.; Asghari, M.H.; Goel, A. The therapeutic triad of extracellular vesicles: As drug targets, as drugs, and as drug carriers. Biochem. Pharmacol. 2021, 192, 114714. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.-H.; Fang, D.; Xu, Y.-D.; Nan, T.-G.; Song, W.-P.; Gu, Y.-Y.; Gu, S.-J.; Yuan, Y.-M.; Xin, Z.-C.; Zhou, L.-Q. Skimmed bovine milk-derived extracellular vesicles isolated via “salting-out”: Characterizations and potential functions as nanocarriers. Front. Nutr. 2021, 8, 769223. [Google Scholar] [CrossRef]
- Becker Peres, L.; Becker Peres, L.; de Araújo, P.H.H.; Sayer, C. Solid lipid nanoparticles for encapsulation of hydrophilic drugs by an organic solvent free double emulsion technique. Colloids Surf. B. Biointerfaces 2016, 140, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Mirchandani, Y.; Patravale, V.B. Solid lipid nanoparticles for hydrophilic drugs. J. Controlled Release 2021, 335, 457–464. [Google Scholar] [CrossRef]
- LIU, K.-p.; WANG, L.-f.; Yang, L.; Bin, Y.; Chao, D.; Yang, W. Preparation, pharmacokinetics, and tissue distribution properties of icariin-loaded stealth solid lipid nanoparticles in mice. Chin. Herb. Med. 2012, 4, 170–174. [Google Scholar]
- Zhou, Y.; Wang, J.; Xiao, Y.; Wang, T.; Huang, X. The effects of polymorphism on physicochemical properties and pharmacodynamics of solid drugs. Curr. Pharm. Des. 2018, 24, 2375–2382. [Google Scholar] [CrossRef]
- Suresh, K.; Khandavilli, U.R.; Gunnam, A.; Nangia, A. Polymorphism, isostructurality and physicochemical properties of glibenclamide salts. CrystEngComm 2017, 19, 918–929. [Google Scholar] [CrossRef]
- Jia, L.; Zhang, Q.; Wang, J.-R.; Mei, X. Versatile solid modifications of icariin: Structure, properties and form transformation. CrystEngComm 2015, 17, 7500–7509. [Google Scholar] [CrossRef]
- Premathilaka, R.; Rashidinejad, A.; Golding, M.; Singh, J. Oral delivery of hydrophobic flavonoids and their incorporation into functional foods: Opportunities and challenges. Food Hydrocoll. 2022, 128, 107567. [Google Scholar] [CrossRef]
- Liu, C.; Gao, X.; Liu, Y.; Huang, M.; Qu, D.; Chen, Y. Icariin combined with snailase shows improved intestinal hydrolysis and absorption in osteoporosis rats. Biomed. Pharmacother. 2017, 94, 1048–1056. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Li, R.; Peng, J.; Qu, D.; Huang, M.; Chen, Y. Enhanced hydrolysis and antitumor efficacy of Epimedium flavonoids mediated by immobilized snailase on silica. Process Biochem. 2019, 86, 80–88. [Google Scholar] [CrossRef]
- Zhou, J.; Ma, Y.H.; Zhou, Z.; Chen, Y.; Wang, Y.; Gao, X. Intestinal absorption and metabolism of epimedium flavonoids in osteoporosis rats. Drug Metab. Disposition 2015, 43, 1590–1600. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kurakula, M.; Gorityala, S.; Moharir, K. Recent trends in design and evaluation of chitosan-based colon targeted drug delivery systems: Update 2020. J. Drug Deliv. Sci. Technol. 2021, 64, 102579. [Google Scholar] [CrossRef]
- Kaur, A.; Kaur, A.; P Kaur, V.; Kaur, M.; Murthy, R. Polymeric drug delivery approaches for colon targeting: A review. Drug Deliv. Lett. 2014, 4, 38–48. [Google Scholar] [CrossRef]
- Wang, Q.-S.; Wang, G.-F.; Zhou, J.; Gao, L.-N.; Cui, Y.-L. Colon targeted oral drug delivery system based on alginate-chitosan microspheres loaded with icariin in the treatment of ulcerative colitis. Int. J. Pharm. 2016, 515, 176–185. [Google Scholar] [CrossRef]

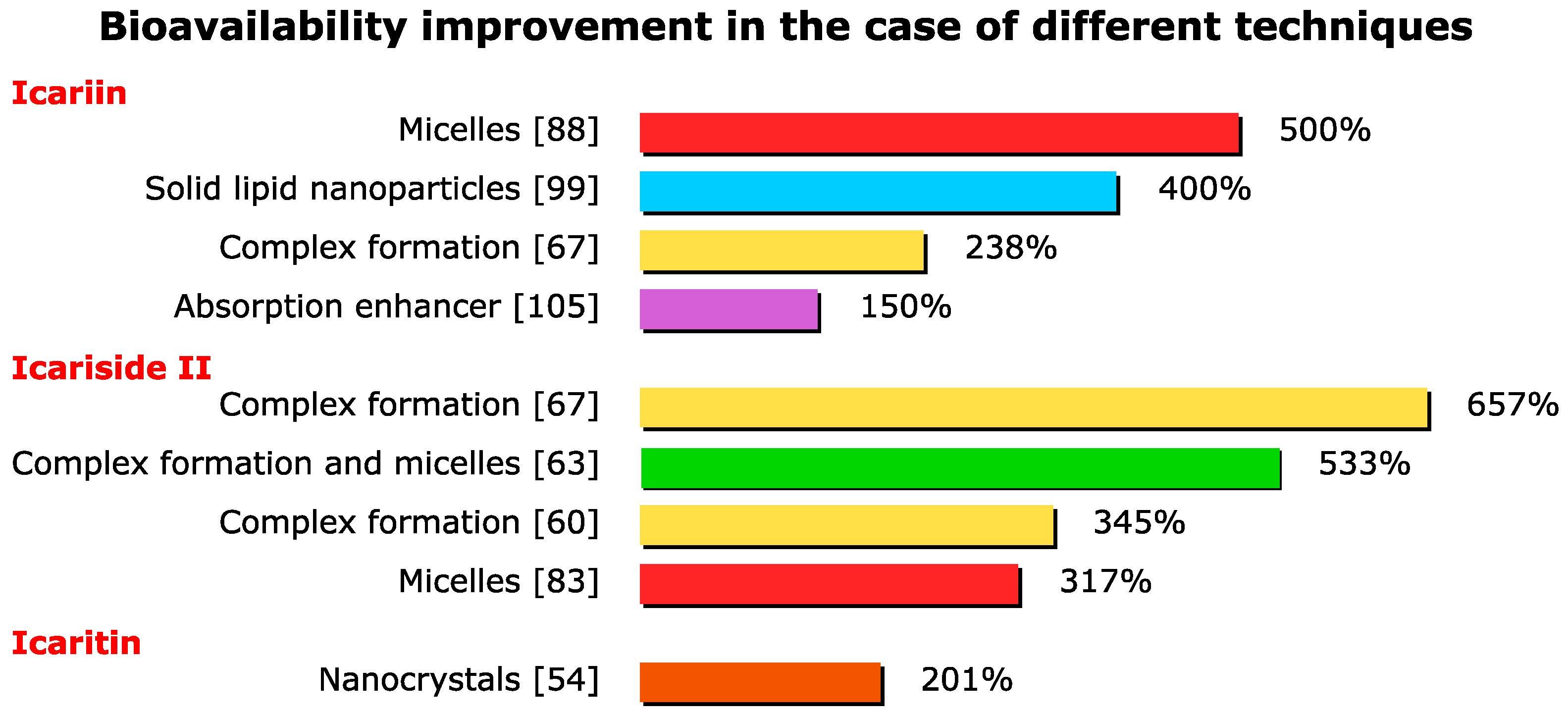
| Flavonol | Technique | Preparation Method | Carrier | Efficiency Improvement | Reference |
|---|---|---|---|---|---|
| Icariin | Complex formation | Lyophilization | β-Cyclodextrin | Cumulative drug release: 62% | [72] |
| Complex formation | Lyophilization | β-Cyclodextrin | Solubility (water, 25 °C): 1.68-fold Absorption rate: 2.32-fold Permeability rate: 3.46-fold | [67] | |
| Complex formation | Saturated solution method | β-Cyclodextrin | Solubility (water, 37 °C): 36-fold | [69] | |
| Microspheres | Emulsion and coagulation | Gelatin and hyaluronic acid | Controlled release | [93] | |
| Nanocarriers | Mixing and centrifuging | Fetal bovine serum exosomes | Cell proliferation significantly increased (p < 0.001) | [52] | |
| Micelle | Mixing and vacuum-drying | PEG-PLLA/PDLA-PNIPAM polymers | Bioavailability: 5-fold | [88] | |
| Solid modification | Heating | - | Solubility (water, 25 °C): 1.5-fold | [102] | |
| Hydrogel formation | Photopolymerization | Hyaluronic acid | Controlled release | [92] | |
| Hydrogel formation | Reverse microemulsion method | Span 80 and Tween 80 | - | [91] | |
| Solid lipid nanoparticles | High temperature melt-cool solidification method | Liposomal vesicles | Bioavailability: 4-fold | [99] | |
| Absorption enhancer | Mixing | Snailase | Bioavailability: 1.5-fold | [104] | |
| Icariside II and icariin mixture | Complex formation | Wet media milling | Soybean phospholipids | ICA: Dissolution: 1.39-fold (2 h) Bioavailability: 2.38-fold ICS: Dissolution: 1.24-fold (2 h) Bioavailability: 6.57-fold | [65] |
| Icariside II | Complex formation | Reduction vaporization | Phospholipid | Bioavailability: 3.45-fold | [60] |
| Complex formation and micelles | Solvent evaporation | Phospholipid and vitamin E TPGS 1000 | Bioavailability: 5.33-fold | [63] | |
| Micelles | Thin film hydration | DDAB and TPGS with hyaluronic acid | Controlled release | [84] | |
| Micelles | Solvent evaporation | Solutol® HS15 and Pluronic F127 | Solubility (water, temperature not mentioned) 900-fold Bioavailability: 3.17-fold | [83] | |
| Nanocarriers | Incubation and sonification | Bovine milk as extracellular vesicles | - | [96] | |
| Icaritin | Nanocrystal | Antisolvent-precipitation | Hydroxypropyl methylcellulose as stabilizer | Bioavailability: 2.01-fold | [54] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szabó, R.; Rácz, C.P.; Dulf, F.V. Bioavailability Improvement Strategies for Icariin and Its Derivates: A Review. Int. J. Mol. Sci. 2022, 23, 7519. https://doi.org/10.3390/ijms23147519
Szabó R, Rácz CP, Dulf FV. Bioavailability Improvement Strategies for Icariin and Its Derivates: A Review. International Journal of Molecular Sciences. 2022; 23(14):7519. https://doi.org/10.3390/ijms23147519
Chicago/Turabian StyleSzabó, Róbert, Csaba Pál Rácz, and Francisc Vasile Dulf. 2022. "Bioavailability Improvement Strategies for Icariin and Its Derivates: A Review" International Journal of Molecular Sciences 23, no. 14: 7519. https://doi.org/10.3390/ijms23147519
APA StyleSzabó, R., Rácz, C. P., & Dulf, F. V. (2022). Bioavailability Improvement Strategies for Icariin and Its Derivates: A Review. International Journal of Molecular Sciences, 23(14), 7519. https://doi.org/10.3390/ijms23147519







