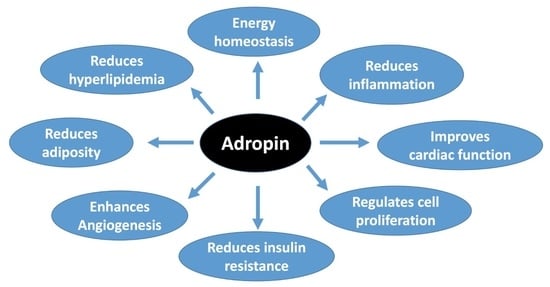
Adropin’s Role in Energy Homeostasis and Metabolic Disorders
Abstract
:1. Introduction
1.1. Discovery of Adropin
1.2. Enho Gene and Adropin Peptide Characteristics
2. Tissue Expression of Adropin
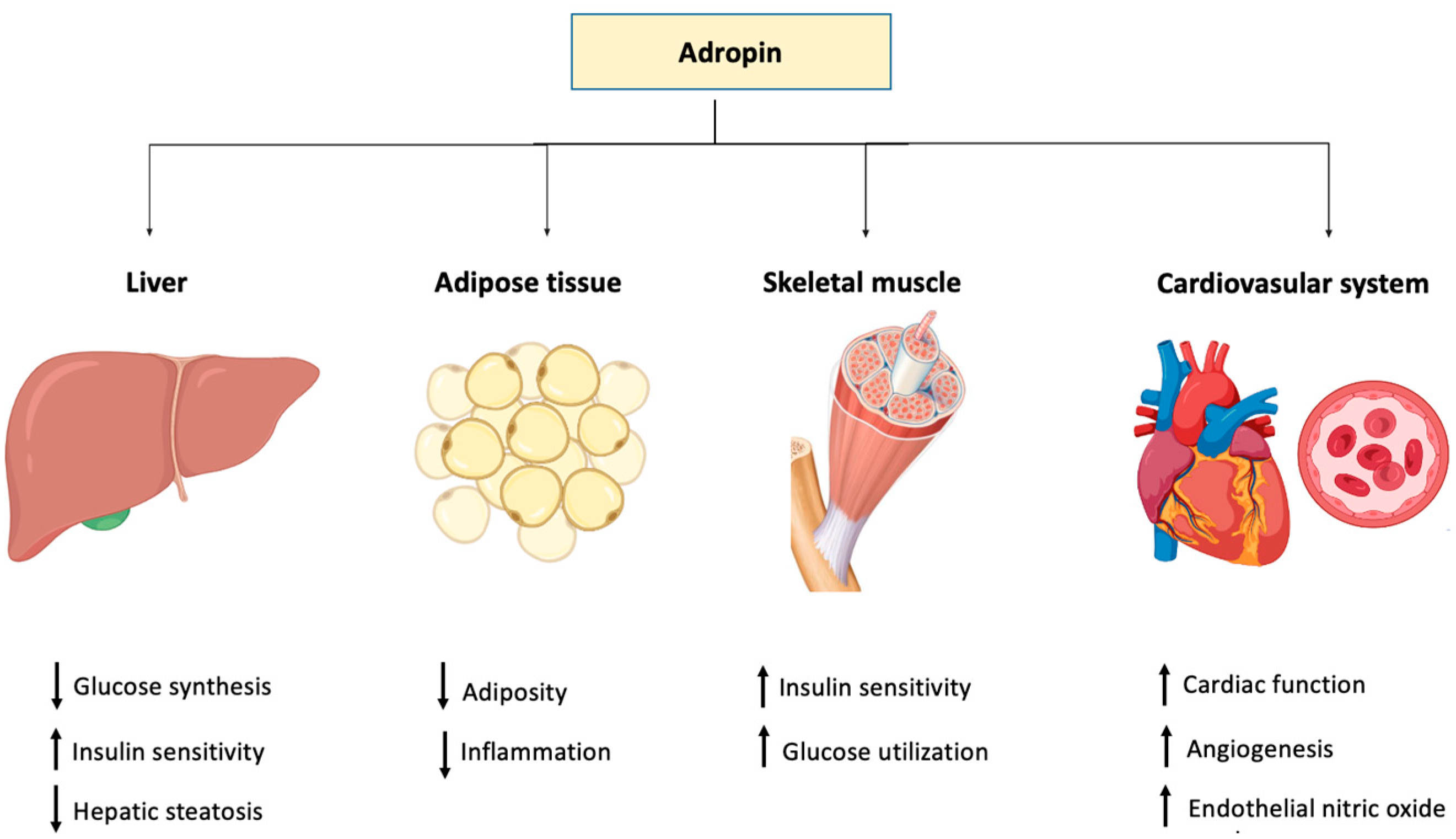
3. Adropin and Energy Homeostasis
4. The Role of Adropin in Health and Disease
4.1. Diabetes Mellitus
4.2. Obesity
4.3. Cardiovascular Diseases
4.4. Inflammation
4.5. Cell Proliferation and Differentiation
5. Characteristics of Adropin Knockout Mice
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hayashi, M.A.F.; Ducancel, F.; Konno, K. Natural peptides with potential applications in drug development, diagnosis, and/or biotechnology. Int. J. Pept. 2014, 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Elabadlah, H.; Hameed, R.; D’Souza, C.; Mohsin, S.; Adeghate, E.A. Exogenous Ghrelin Increases Plasma Insulin Level in Diabetic Rats. Biomolecules 2020, 10, 633. [Google Scholar] [CrossRef] [PubMed]
- Adeghate, E.; Lotfy, M.; D’Souza, C.; Alseiari, S.M.; Alsaadi, A.A.; Qahtan, S.A. Hypocretin/orexin modulates body weight and the metabolism of glucose and insulin. Diabetes/Metab. Res. Rev. 2020, 36, e3229. [Google Scholar] [CrossRef] [PubMed]
- Adeghate, E.; Fernandez-Cabezudo, M.; Hameed, R.; El-Hasasna, H.; El Wasila, M.; Abbas, T.; Al-Ramadi, B. Orexin-1 receptor co-localizes with pancreatic hormones in islet cells and modulates the outcome of streptozotocin-induced diabetes mellitus. PLoS ONE 2010, 5, e8587. [Google Scholar] [CrossRef] [Green Version]
- Mahgoub, M.O.; D’Souza, C.; Al Darmaki, R.; Baniyas, M.; Adeghate, E. An update on the role of irisin in the regulation of endocrine and metabolic functions. Peptides 2018, 104, 15–23. [Google Scholar] [CrossRef]
- Lotfy, M.; Singh, J.; Rashed, H.; Tariq, S.; Zilahi, E.; Adeghate, E. Mechanism of the beneficial and protective effects of exenatide in diabetic rats. J. Endocrinol. 2014, 220, 291–304. [Google Scholar] [CrossRef] [Green Version]
- Adeghate, E. Visfatin: Structure, function and relation to diabetes mellitus and other dysfunctions. Curr. Med. Chem. 2008, 15, 1851–1862. [Google Scholar] [CrossRef]
- Jaberi, S.A.; Cohen, A.; D’Souza, C.; Abdulrazzaq, Y.M.; Ojha, S.; Bastaki, S.; Adeghate, E.A. Lipocalin-2: Structure, function, distribution and role in metabolic disorders. Biomed. Pharm. 2021, 142, 112002. [Google Scholar] [CrossRef]
- Adeghate, E.; Mohsin, S.; Adi, F.; Ahmed, F.; Yahya, A.; Kalász, H.; Tekes, K.; Adeghate, E.A. An update of SGLT1 and SGLT2 inhibitors in early phase diabetes-type 2 clinical trials. Expert Opin. Investig. Drugs 2019, 28, 811–820. [Google Scholar] [CrossRef]
- Howarth, F.C.; Jacobson, M.; Shafiullah, M.; Adeghate, E. Effects of insulin treatment on heart rhythm, body temperature and physical activity in streptozotocin-induced diabetic rat. Clin. Exp. Pharmacol. Physiol. 2006, 33, 327–331. [Google Scholar] [CrossRef]
- Marqus, S.; Pirogova, E.; Piva, T.J. Evaluation of the use of therapeutic peptides for cancer treatment. J. Biomed. Sci. 2017, 24, 21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Craik, D.J.; Fairlie, D.P.; Liras, S.; Price, D. The Future of Peptide-based Drugs. Chem. Biol. Drug Design. 2013, 81, 136–147. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.F.G.; Fogacci, F.; Colletti, A. Potential role of bioactive peptides in prevention and treatment of chronic diseases: A narrative review. Br. J. Pharmacol. 2017, 174, 1378–1394. [Google Scholar] [CrossRef] [PubMed]
- Kumar, K.G.; Trevaskis, J.L.; Lam, D.D.; Sutton, G.M.; Koza, R.A.; Chouljenko, V.N.; Kousoulas, K.G.; Rogers, P.M.; Kesterson, R.A.; Thearle, M.; et al. Identification of Adropin as a Secreted Factor Linking Dietary Macronutrient Intake with Energy Homeostasis and Lipid Metabolism. Cell Metab. 2008, 8, 468–481. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soltani, S.; Kolahdouz-Mohammadi, R.; Aydin, S.; Yosaee, S.; Clark, C.; Abdollahi, S. Circulating levels of adropin and overweight/obesity: A systematic review and meta-analysis of observational studies. Hormones 2022, 21, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Thapa, D.; Xie, B.; Manning, J.R.; Zhang, M.; Stoner, M.W.; Huckestein, B.R.; Edmunds, L.R.; Zhang, X.; Dedousis, N.L.; O’Doherty, R.M.; et al. Adropin reduces blood glucose levels in mice by limiting hepatic glucose production. Physiol. Rep. 2019, 7, e14043. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thapa, D.; Stoner, M.W.; Zhang, M.; Xie, B.; Manning, J.R.; Guimaraes, D.; Shiva, S.; Jurczak, M.J.; Scott, I. Adropin regulates pyruvate dehydrogenase in cardiac cells via a novel GPCR-MAPK-PDK4 signaling pathway. Redox Biol. 2018, 18, 25–32. [Google Scholar] [CrossRef]
- Butler, A.A.; Havel, P.J. Adropin and insulin resistance: Integration of endocrine, circadian, and stress signals regulating glucose metabolism. Obesity 2021, 29, 1799–1801. [Google Scholar] [CrossRef]
- Wong, C.M.; Wang, Y.; Lee, J.T.; Huang, Z.; Wu, D.; Xu, A.; Lam, K.S. Adropin is a brain membrane-bound protein regulating physical activity via the NB-3/notch signaling pathway in mice. J. Biol. Chem. 2014, 289, 25976–25986. [Google Scholar] [CrossRef] [Green Version]
- Gao, S.; McMillan, R.P.; Zhu, Q.; Lopaschuk, G.D.; Hulver, M.W.; Butler, A.A. Therapeutic effects of adropin on glucose tolerance and substrate utilization in diet-induced obese mice with insulin resistance. Mol. Metab. 2015, 4, 310–324. [Google Scholar] [CrossRef]
- Stein, L.M.; Yosten, G.L.C.; Samson, W.K. Adropin acts in brain to inhibit water drinking: Potential interaction with the orphan G protein-coupled receptor, GPR19. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2016, 310, R476–R480. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalaany, N.Y.; Mangelsdorf, D.J. LXRS AND FXR: The Yin and Yang of Cholesterol and Fat Metabolism. Annu. Rev. Physiol. 2006, 68, 159–191. [Google Scholar] [CrossRef] [PubMed]
- Lovren, F.; Pan, Y.; Quan, A.; Singh, K.K.; Shukla, P.C.; Gupta, M.; Al-Omran, M.; Teoh, H.; Verma, S. Adropin is a novel regulator of endothelial function. Circulation 2010, 11 (Suppl. S1), 185–192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aydin, S.; Kuloglu, T.; Aydin, S.; Eren, M.N.; Yilmaz, M.; Kalayci, M.; Sahin, I.; Kocaman, N.; Citil, C.; Kendir, Y. Expression of adropin in rat brain, cerebellum, kidneys, heart, liver, and pancreas in streptozotocin-induced diabetes. Mol. Cell. Biochem. 2013, 380, 73–81. [Google Scholar] [CrossRef]
- Aydin, S. Three new players in energy regulation: Preptin, adropin and irisin. Peptides 2014, 56, 94–110. [Google Scholar] [CrossRef]
- Kuloglu, T.; Aydin, S. Immunohistochemical expressions of adropin and inducible nitric oxide synthase in renal tissues of rats with streptozotocin-induced experimental diabetes. Biotech. Histochem. 2014, 89, 104–110. [Google Scholar] [CrossRef]
- Akkaya, H.; Güntürk, E.E.; Akkaya, F.; Karabıyık, U.; Güntürk, İ.; Yılmaz, S. Assessment of the Relatıonshıp between the Adropın Levels and the Coronary Collateral Cırculatıon in Patıents wıth Chronıc Coronary Syndrome. Avaliação da Relação entre Níveis de Adropina e Circulação Colateral Coronária em Pacientes com Síndrome Coronariana Crônica. Arq. Bras. De Cardiol. 2022. Online ahead of print. [Google Scholar] [CrossRef]
- Dodd, W.S.; Patel, D.; Lucke-Wold, B.; Hosaka, K.; Chalouhi, N.; Hoh, B.L. Adropin decreases endothelial monolayer permeability after cell-free hemoglobin exposure and reduces MCP-1-induced macrophage transmigration. Biochem. Biophys. Res. Commun. 2021, 582, 105–110. [Google Scholar] [CrossRef]
- Bozic, J.; Kumric, M.; Ticinovic Kurir, T.; Males, I.; Borovac, J.A.; Martinovic, D.; Vilovic, M. Role of Adropin in Cardiometabolic Disorders: From Pathophysiological Mechanisms to Therapeutic Target. Biomedicines 2021, 9, 1407. [Google Scholar] [CrossRef]
- Li, B.; Li, N.; Guo, S.; Zhang, M.; Li, J.; Zhai, N.; Wang, H.; Zhang, Y. The changing features of serum adropin, copeptin, neprilysin and chitotriosidase which are associated with vascular endothelial function in type 2 diabetic retinopathy patients. J. Diabetes Its Complicat. 2020, 34, 107686. [Google Scholar] [CrossRef]
- Celikhisar, H.; Ilkhan, G.D. Alterations in Serum Adropin, Adiponectin, and Proinflammatory Cytokine Levels in OSAS. Can. Respir. J. 2020, 2020, 2571283. [Google Scholar] [CrossRef] [PubMed]
- Fan, Z.; Zhang, Y.; Zou, F.; Xu, T.; Pan, P.; Hu, C.; Su, X. Serum adropin level is associated with endothelial dysfunction in patients with obstructive sleep apnea and hypopnea syndrome. Sleep Breath. 2021, 25, 117–123. [Google Scholar] [CrossRef]
- Wu, L.; Fang, J.; Yuan, X.; Xiong, C.; Chen, L. Adropin reduces hypoxia/reoxygenation-induced myocardial injury via the reperfusion injury salvage kinase pathway. Exp. Ther. Med. 2019, 18, 3307–3314. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, F.; Fang, J.; Chen, F.; Wang, C.; Chen, S.; Zhang, S.; Lv, X.; Zhang, J.; He, Q.; Weng, S.; et al. Enho Mutations Causing Low Adropin: A Possible Pathomechanism of MPO-ANCA Associated Lung Injury. EBioMedicine 2016, 9, 324–335. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aydin, S. Presence of adropin, nesfatin-1, apelin-12, ghrelins and salusins peptides in the milk, cheese whey and plasma of dairy cows. Peptides 2013, 43, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.P.; You, T.; Chan, S.P.; Chen, J.C.; Xu, W.T. Adropin is associated with hyperhomocysteine and coronary atherosclerosis. Exp. Ther. Med. 2016, 11, 1065–1070. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Z.W.; Ren, Y.G.; Liu, J. Low Serum Adropin Levels are Associated with Coronary Slow Flow Phenomenon. Acta Cardiol. Sin. 2018, 34, 307–312. [Google Scholar] [CrossRef]
- Celik, H.T.; Bilen, M.; Kazancı, F.; Yildirim, M.E.; İncebay, İ.B.; Erdamar, H. Serum adropin as a predictive biomarker of erectile dysfunction in coronary artery disease patients. Cent. Eur. J. Urol. 2019, 72, 302–306. [Google Scholar] [CrossRef]
- Yang, M.; Pei, Q.; Zhang, J.; Weng, H.; Jing, F.; Yi, Q. Association between adropin and coronary artery lesions in children with Kawasaki disease. Eur. J. Pediatrics 2021, 180, 2253–2259. [Google Scholar] [CrossRef]
- Woods, S.C.; Seeley, R.J.; Porte, D.; Schwartz, M.W. Signals that regulate food intake and energy homeostasis. Science 1998, 280, 1378–1383. [Google Scholar] [CrossRef] [Green Version]
- Powley, T.L.; Keessey, R.E. Body energy homeostasis. Appetite 2008, 51, 442–445. [Google Scholar] [CrossRef]
- Renquist, B.J.; Lippert, R.; Sebag, J.A.; Ellacott, K.L.J.; Cone, R.D. Physiological roles of the melanocortin MC 3 receptor. Eur. J. Pharmacol. 2010, 660, 13–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jéquier, E. Leptin Signaling, Adiposity, and Energy Balance. Ann. N. Y. Acad. Sci. 2006, 967, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Jaberi, S.A.; Cohen, A.; Saeed, Z.; Ojha, S.; Singh, J.; Adeghate, E. Obesity: Molecular Mechanisms, Epidemiology, Complications and Pharmacotherapy. In Cellular and Biochemical Mechanisms of Obesity; Springer: Cham, Switzerland, 2021; pp. 249–266. [Google Scholar]
- Gao, S.; Ghoshal, S.; Zhang, L.; Stevens, J.R.; McCommis, K.S.; Finck, B.N.; Lopaschuk, G.D.; Butler, A.A. The peptide hormone adropin regulates signal transduction pathways controlling hepatic glucose metabolism in a mouse model of diet-induced obesity. J. Biol. Chem. 2019, 294, 13366–13377. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhang, Q.; Huang, Z.; Jiang, Q. Adropin inhibited tilapia hepatic glucose output and triglyceride accumulation via AMPK activation. J. Endocrinol. 2020, 246, 109–122. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Chen, S.; Shen, T.; Yang, W.; Chen, Q.; Zhang, P.; You, Y.; Sun, X.; Xu, H.; Tang, Y.; et al. Adropin regulates hepatic glucose production via PP2A/AMPK pathway in insulin-resistant hepatocytes. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2020, 34, 10056–10072. [Google Scholar] [CrossRef]
- Banerjee, S.; Ghoshal, S.; Stevens, J.R.; McCommis, K.S.; Gao, S.; Castro-Sepulveda, M.; Mizgier, M.L.; Girardet, C.; Kumar, K.G.; Galgani, J.E.; et al. Hepatocyte expression of the micropeptide adropin regulates the liver fasting response and is enhanced by caloric restriction. J. Biol. Chem. 2020, 295, 13753–13768. [Google Scholar] [CrossRef]
- Demirdöğen, F.; Akdağ, T.; Gündüz, Z.B.; Odabaş, F.Ö. Investigation of serum adropin levels and its relationship with hypothalamic atrophy in patients with multiple sclerosis. Mult. Scler. Relat. Disord. 2022, 66, 103948. [Google Scholar] [CrossRef]
- Aydın, P.; Uzunçakmak, S.K.; Tör, İ.H.; Bilen, A.; Özden, A. Comparison of Serum Adropin Levels in Patients with Diabetes Mellitus, COVID-19, and COVID-19 with Diabetes Mellitus. Eurasian J. Med. 2022, 54, 197–201. [Google Scholar] [CrossRef]
- Ruszała, M.; Pilszyk, A.; Niebrzydowska, M.; Kimber-Trojnar, Ż.; Trojnar, M.; Leszczyńska-Gorzelak, B. Novel Biomolecules in the Pathogenesis of Gestational Diabetes Mellitus 2.0. Int. J. Mol. Sci. 2022, 23, 4364. [Google Scholar] [CrossRef]
- Kong, Z.; Liu, Y. Soluble Vascular Adhesion Protein-1 Level Correlates With Adropin and Inflammatory Biomarkers in Patients With Obstructive Sleep Apnea. Ear Nose Throat J. 2022. Online ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Simac, P.; Perkovic, D.; Bozic, I.; Bilopavlovic, N.; Martinovic, D.; Bozic, J. Serum Adropin Levels in Patients with Rheumatoid Arthritis. Life 2022, 12, 169. [Google Scholar] [CrossRef] [PubMed]
- Uysal, B.A.; Kuyumcu, M.S. Serum irisin and adropin levels may be predictors for coronary artery ectasia. Clin. Exp. Hypertens. 2022, 44, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Kurt, E.; Tekin, E.; Kurt, N.; Bayramoglu, A. The role of adropin, HIF-1α and apelin biomarkers in the diagnosis of acute mesentaric ischemia. Am. J. Emerg. Med. 2022, 51, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Es-Haghi, A.; Al-Abyadh, T.; Mehrad-Majd, H. The Clinical Value of Serum Adropin Level in Early Detection of Diabetic Nephropathy. Kidney Blood Press. Res. 2021, 46, 734–740. [Google Scholar] [CrossRef] [PubMed]
- Zang, H.; Jiang, F.; Cheng, X.; Xu, H.; Hu, X. Serum adropin levels are decreased in chinese type 2 diabetic patients and negatively correlated with body mass index. Endocr. J. 2018, 65, 685–691. [Google Scholar] [CrossRef] [Green Version]
- Chen, S.; Zeng, K.; Liu, Q.C.; Guo, Z.; Zhang, S.; Chen, X.R.; Lin, J.H.; Wen, J.P.; Zhao, C.F.; Lin, X.H.; et al. Adropin deficiency worsens HFD-induced metabolic defects. Cell Death Dis. 2017, 8, e3008. [Google Scholar] [CrossRef] [Green Version]
- Li, N.; Xie, G.; Zhou, B.; Qu, A.; Meng, H.; Liu, J.; Wang, G. Serum Adropin as a Potential Biomarker for Predicting the Development of Type 2 Diabetes Mellitus in Individuals With Metabolic Dysfunction-Associated Fatty Liver Disease. Front. Physiol. 2021, 12, 696163. [Google Scholar] [CrossRef]
- Polkowska, A.; Pasierowska, I.E.; Pasławska, M.; Pawluczuk, E.; Bossowski, A. Assessment of Serum Concentrations of Adropin, Afamin, and Neudesin in Children with Type 1 Diabetes. BioMed Res. Int. 2019, 2019, 6128410. [Google Scholar] [CrossRef] [Green Version]
- Hosseini, A.; Shanaki, M.; Emamgholipour, S.; Nakhjavani, M.; Razi, F.; Golmohammadi, T. Elevated serum levels of adropin in patients with type 2 diabetes mellitus and its association with insulin resistance. J. Biol. Today’s World 2016, 5, 44–49. [Google Scholar] [CrossRef]
- Li, S.; Sun, J.; Hu, W.; Liu, Y.; Lin, D.; Duan, H.; Liu, F. The association of serum and vitreous adropin concentrations with diabetic retinopathy. Ann. Clin. Biochem. Int. J. Lab. Med. 2019, 56, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Hu, W.; Chen, L. Association of Serum Adropin Concentrations with Diabetic Nephropathy. Mediat. Inflamm. 2016, 2016, 6038261. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beigi, A.; Shirzad, N.; Nikpour, F.; Nasli Esfahani, E.; Emamgholipour, S.; Bandarian, F. Association between serum adropin levels and gestational diabetes mellitus; a case–control study. Gynecol. Endocrinol. 2015, 31, 939–941. [Google Scholar] [CrossRef] [PubMed]
- Kuo, F.Y.; Cheng, K.C.; Li, Y.; Cheng, J.T.; Tsai, C.C. Promotion of adropin expression by hyperglycemia is associated with STAT3 activation in diabetic rats. Diabetes Metab. Syndr. Obes. Targets Ther. 2020, 13, 2269–2277. [Google Scholar] [CrossRef]
- Ozkan, A.; Aslan, M.A.; Sinen, O.; Munzuroglu, M.; Derin, N.; Parlak, H.; Bulbul, M.; Agar, A. Effects of adropin on learning and memory in rats tested in the Morris water maze. Hippocampus 2022, 32, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Puigserver, P.; Rhee, J.; Donovan, J.; Walkey, C.J.; Yoon, J.C.; Oriente, F.; Kitamura, Y.; Altomonte, J.; Dong, H.H.; Accili, D.; et al. Insulin-regulated hepatic gluconeogenesis through FOXO1–PGC-1a interaction. Nature 2003, 423, 545–550. [Google Scholar] [CrossRef] [PubMed]
- Hill, M.J.; Suzuki, S.; Segars, J.H.; Kino, T. CRTC2 is a coactivator of GR and couples GR and CREB in the regulation of hepatic gluconeogenesis. Mol. Endocrinol. 2016, 30, 104–117. [Google Scholar] [CrossRef] [Green Version]
- Kurir, T.T.; Miličević, T.; Novak, A.; Vilović, M.; Božić, J. Adropin—potential link in cardiovascular protection for obese male type 2 diabetes mellitus patients treated with liraglutide. Acta Clin. Croat. 2020, 59, 344–350. [Google Scholar] [CrossRef]
- Tasyurek, H.M.; Altunbas, H.A.; Balci, M.K.; Sanlioglu, S. Incretins: Their physiology and application in the treatment of diabetes mellitus. Diabetes/Metab. Res. Rev. 2014, 30, 354–371. [Google Scholar] [CrossRef]
- Lotfy, M.; Singh, J.; Kalász, H.; Tekes, K.; Adeghate, E. Medicinal Chemistry and Applications of Incretins and DPP-4 Inhibitors in the Treatment of Type 2 Diabetes Mellitus. Open Med. Chem. J. 2011, 5, 82–92. [Google Scholar] [CrossRef]
- Adeghate, J.O.; D’Souza, C.; Kántor, O.; Tariq, S.; Souid, A.K.; Adeghate, E. Early (5-Day) Onset of Diabetes Mellitus Causes Degeneration of Photoreceptor Cells, Overexpression of Incretins, and Increased Cellular Bioenergetics in Rat Retina. Cells 2021, 10, 1981. [Google Scholar] [CrossRef] [PubMed]
- Lotfy, M.; Singh, J.; Rashed, H.; Tariq, S.; Zilahi, E.; Adeghate, E. The effect of glucagon-like peptide-1 in the management of diabetes mellitus: Cellular and molecular mechanisms. Cell Tissue Res. 2014, 358, 343–358. [Google Scholar] [CrossRef] [PubMed]
- Behera, J.; Ison, J.; Voor, M.J.; Tyagi, N. Exercise-Linked Skeletal Irisin Ameliorates Diabetes-Associated Osteoporosis by Inhibiting the Oxidative Damage-Dependent miR-150-FNDC5/Pyroptosis Axis. Diabetes 2022, db210573. [Google Scholar] [CrossRef] [PubMed]
- Marrano, N.; Biondi, G.; Borrelli, A.; Cignarelli, A.; Perrini, S.; Laviola, L.; Giorgino, F.; Natalicchio, A. Irisin and incretin hormones: Similarities, differences, and implications in type 2 diabetes and obesity. Biomolecules 2021, 11, 286. [Google Scholar] [CrossRef]
- Wattez, J.S.; Ravallec, R.; Cudennec, B.; Knauf, C.; Dhulster, P.; Valet, P.; Breton, C.; Vieau, D.; Lesage, J. Apelin stimulates both cholecystokinin and glucagon-like peptide 1 secretions in vitro and in vivo in rodents. Peptides 2013, 48, 134–136. [Google Scholar] [CrossRef]
- Howarth, F.C.; Jacobson, M.; Shafiullah, M.; Adeghate, E. Long-term effects of type 2 diabetes mellitus on heart rhythm in the Goto-Kakizaki rat. Exp. Physiol. 2008, 93, 362–369. [Google Scholar] [CrossRef]
- Marcolin, É.; Forgiarini, L.F.; Tieppo, J.; Dias, A.S.; de Freitas, L.A.R.; Marroni, N.P. Methionine- and choline-deficient diet induces hepatic changes characteristic of non-alcoholic steatohepatitis. Arq. De Gastroenterol. 2011, 48, 72–79. [Google Scholar] [CrossRef] [Green Version]
- Jasaszwili, M.; Wojciechowicz, T.; Billert, M.; Strowski, M.Z.; Nowak, K.W.; Skrzypski, M. Effects of adropin on proliferation and differentiation of 3T3-L1 cells and rat primary preadipocytes. Mol. Cell. Endocrinol. 2019, 496, 110532. [Google Scholar] [CrossRef]
- Hansson, G.K.; Libby, P. The immune response in atherosclerosis: A double-edged sword. Nat. Rev. Immunol. 2006, 6, 508–519. [Google Scholar] [CrossRef]
- Hart, D.A. Obesity, the Obesity Epidemic, and Metabolic Dysfunction: The Conundrum Presented by the Disconnect between Evolution and Modern Societies. J. Biomed. Sci. Eng. 2021, 14, 203–211. [Google Scholar] [CrossRef]
- Ganesh-Kumar, K.; Zhang, J.; Gao, S.; Rossi, J.; McGuinness, O.P.; Halem, H.H.; Culler, M.D.; Mynatt, R.L.; Butler, A.A. Adropin deficiency is associated with increased adiposity and insulin resistance. Obesity 2012, 20, 1394–1402. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Erman, H.; Ozdemir, A.; Sitar, M.E.; Cetin, S.I.; Boyuk, B. Role of serum adropin measurement in the assessment of insulin resistance in obesity. J. Investig. Med. Off. Publ. Am. Fed. Clin. Res. 2021, 69, 1318–1323. [Google Scholar] [CrossRef] [PubMed]
- Butler, A.A.; Tam, C.S.; Stanhope, K.L.; Wolfe, B.M.; Ali, M.R.; O’Keeffe, M.; St-Onge, M.P.; Ravussin, E.; Havel, P.J. Low Circulating Adropin Concentrations with Obesity and Aging Correlate with Risk Factors for Metabolic Disease and Increase after Gastric Bypass Surgery in Humans. J. Clin. Endocrinol. Metab. 2012, 97, 3783–3791. [Google Scholar] [CrossRef] [Green Version]
- Glück, M.; Glück, J.; Wiewióra, M.; Rogala, B.; Piecuch, J. Serum Irisin, Adropin, and Preptin in Obese Patients 6 Months After Bariatric Surgery. Obes. Surg. 2019, 29, 3334–3341. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kocaoglu, C.; Buyukinan, M.; Erdem, S.S.; Ozel, A. Are obesity and metabolic syndrome associated with plasma adropin levels in children? J. Pediatric Endocrinol. Metab. 2015, 28, 1293–1297. [Google Scholar] [CrossRef] [PubMed]
- Adeghate, E.A.; Kalász, H.; Al Jaberi, S.; Adeghate, J.; Tekes, K. Tackling type 2 diabetes-associated cardiovascular and renal comorbidities: A key challenge for drug development. Expert Opin. Investig. Drugs 2021, 30, 85–93. [Google Scholar] [CrossRef]
- Singh, R.M.; Waqar, T.; Howarth, F.C.; Adeghate, E.; Bidasee, K.; Singh, J. Hyperglycemia-induced cardiac contractile dysfunction in the diabetic heart. Heart Fail. Rev. 2018, 23, 37–54. [Google Scholar] [CrossRef]
- Lotfy, M.; Adeghate, J.; Kalasz, H.; Singh, J.; Adeghate, E. Chronic Complications of Diabetes Mellitus: A Mini Review. Curr. Diabetes Rev. 2017, 13, 3–10. [Google Scholar] [CrossRef] [Green Version]
- Adeghate, E.; Singh, J. Structural changes in the myocardium during diabetes-induced cardiomyopathy. Heart Fail. Rev. 2014, 19, 15–23. [Google Scholar] [CrossRef]
- Adeghate, E.; Schattner, P.; Dunn, E. An update on the etiology and epidemiology of diabetes mellitus. Ann. N. Y. Acad. Sci. 2006, 1084, 1–29. [Google Scholar] [CrossRef]
- Adeghate, E. Molecular and cellular basis of the aetiology and management of diabetic cardiomyopathy: A short review. Mol. Cell. Biochem. 2004, 261, 187–191. [Google Scholar] [CrossRef] [PubMed]
- Fillmore, N.; Mori, J.; Lopaschuk, G.D. Mitochondrial fatty acid oxidation alterations in heart failure, ischaemic heart disease and diabetic cardiomyopathy. Br. J. Pharmacol. 2014, 171, 2080–2090. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Altamimi, T.R.; Gao, S.; Karwi, Q.G.; Fukushima, A.; Rawat, S.; Wagg, C.S.; Zhang, L.; Lopaschuk, G.D. Adropin regulates cardiac energy metabolism and improves cardiac function and efficiency. Metab. Clin. Exp. 2019, 98, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Thapa, D.; Xie, B.; Zhang, M.; Stoner, M.W.; Manning, J.R.; Huckestein, B.R.; Edmunds, L.R.; Mullett, S.J.; McTiernan, C.F.; Wendell, S.G.; et al. Adropin treatment restores cardiac glucose oxidation in pre-diabetic obese mice. J. Mol. Cell. Cardiol. 2019, 129, 174–178. [Google Scholar] [CrossRef]
- Arkadievich, O.D. Metabolic markers of myocardium insulin resistance in dogs with heart failure. Open Vet. J. 2021, 10, 363–370. [Google Scholar] [CrossRef]
- Thapa, D.; Xie, B.; Mushala, B.; Zhang, M.; Manning, J.R.; Bugga, P.; Stoner, M.W.; Jurczak, M.J.; Scott, I. Diet-induced obese mice are resistant to improvements in cardiac function resulting from short-term adropin treatment. Curr. Res. Physiol. 2022, 5, 55–62. [Google Scholar] [CrossRef]
- Mushala, B.A.S.; Scott, I. Adropin: A hepatokine modulator of vascular function and cardiac fuel metabolism. Am. J. Physiol. Circ. Physiol. 2021, 320, H238–H244. [Google Scholar] [CrossRef]
- Poss, J.; Custodis, F.; Werner, C.; Weingartner, O.; Bohm, M.; Laufs, U. Cardiovascular Disease and Dyslipidemia: Beyond LDL. Curr. Pharm. Des. 2011, 17, 861–870. [Google Scholar] [CrossRef]
- Akcılar, R.; Emel Koçak, F.; Şimşek, H.; Akcılar, A.; Bayat, Z.; Ece, E.; Kökdaşgil, H. The effect of adropin on lipid and glucose metabolism in rats with hyperlipidemia. Iran. J. Basic Med. Sci. 2016, 19, 245–251. [Google Scholar] [CrossRef]
- Zheng, J.; Liu, M.; Chen, L.; Yin, F.; Zhu, X.; Gou, J.; Zeng, W.; Lv, Z. Association between serum adropin level and coronary artery disease: A systematic review and meta-analysis. Cardiovasc. Diagn. Ther. 2019, 9, 1–7. [Google Scholar] [CrossRef]
- Nattel, S. New ideas about atrial fibrillation 50 years on. Nature 2002, 415, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Sultan, A.; Jacobson, M.; Adeghate, E.; Oulhaj, A.; Shafiullah, M.; Qureshi, A.; Howarth, F.C. Effects of obesity and diabesity on heart rhythm in the Zucker rat. Clin. Exp. Pharmacol. Physiol. 2021, 48, 735–747. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Xue, Y.; Shang, F.; Ni, S.; Liu, X.; Fan, B.; Wang, H. Association of serum adropin with the presence of atrial fibrillation and atrial remodeling. J. Clin. Lab. Anal. 2019, 33, e22672. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meeran, M.; Al Taee, H.; Azimullah, S.; Tariq, S.; Adeghate, E.; Ojha, S. β-Caryophyllene, a natural bicyclic sesquiterpene attenuates doxorubicin-induced chronic cardiotoxicity via activation of myocardial cannabinoid type-2 (CB2) receptors in rats. Chem. Biol. Interact. 2019, 304, 158–167. [Google Scholar] [CrossRef] [PubMed]
- Al-Taee, H.; Azimullah, S.; Meeran, M.; Alaraj Almheiri, M.K.; Al Jasmi, R.A.; Tariq, S.; Ab Khan, M.; Adeghate, E.; Ojha, S. β-caryophyllene, a dietary phytocannabinoid attenuates oxidative stress, inflammation, apoptosis and prevents structural alterations of the myocardium against doxorubicin-induced acute cardiotoxicity in rats: An in vitro and in vivo study. Eur. J. Pharmacol. 2019, 858, 172467. [Google Scholar] [CrossRef] [PubMed]
- Mahgoub, E.; Kumaraswamy, S.M.; Kader, K.H.; Venkataraman, B.; Ojha, S.; Adeghate, E.; Rajesh, M. Genipin attenuates cisplatin-induced nephrotoxicity by counteracting oxidative stress, inflammation, and apoptosis. Biomed. Pharmacother. 2017, 93, 1083–1097. [Google Scholar] [CrossRef]
- Sato, K.; Yamashita, T.; Shirai, R.; Shibata, K.; Okano, T.; Yamaguchi, M.; Mori, Y.; Hirano, T.; Watanabe, T. Adropin contributes to anti-atherosclerosis by suppressing monocyte-endothelial cell adhesion and smooth muscle cell proliferation. Int. J. Mol. Sci. 2018, 19, 1293. [Google Scholar] [CrossRef] [Green Version]
- Jasaszwili, M.; Billert, M.; Strowski, M.Z.; Nowak, K.W.; Skrzypski, M. Adropin as A Fat-Burning Hormone with Multiple Functions-Review of a Decade of Research. Molecules 2020, 25, 549. [Google Scholar] [CrossRef] [Green Version]
- Bozic, J.; Borovac, J.A.; Galic, T.; Kurir, T.T.; Supe-Domic, D.; Dogas, Z. Adropin and inflammation biomarker levels in male patients with obstructive sleep apnea: A link with glucose metabolism and sleep parameters. J. Clin. Sleep Med. 2018, 14, 1109–1118. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.; Xue, H.; Fang, W.; Chen, K.; Chen, S.; Yang, W.; Shen, T.; Chen, X.; Zhang, P.; Ling, W. Adropin protects against liver injury in nonalcoholic steatohepatitis via the Nrf2 mediated antioxidant capacity. Redox Biol. 2019, 21, 101068. [Google Scholar] [CrossRef]
- Jasaszwili, M.; Wojciechowicz, T.; Strowski, M.Z.; Nowak, K.W.; Skrzypski, M. Adropin stimulates proliferation but suppresses differentiation in rat primary brown preadipocytes. Arch. Biochem. Biophys. 2020, 692, 108536. [Google Scholar] [CrossRef] [PubMed]
- Kondělková, K.; Vokurková, D.; Krejsek, J.; Borská, L.; Fiala, Z.; Ctirad, A. Regulatory T cells (TREG) and their roles in immune system with respect to immunopathological disorders. Acta Med. (Hradec Králové)/Univ. Carol. Fac. Med. Hradec Králové 2010, 53, 73–77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adeghate, E.; al-Ramadi, B.; Saleh, A.M.; Vijayarasathy, C.; Ponery, A.S.; Arfat, K.; Howarth, F.C.; El-Sharkawy, T. Increase in neuronal nitric oxide synthase content of the gastroduodenal tract of diabetic rats. Cell. Mol. Life Sci. 2003, 60, 1172–1179. [Google Scholar] [CrossRef] [PubMed]
- Emerald, B.S.; Mohsin, S.; D’Souza, C.; John, A.; El-Hasasna, H.; Ojha, S.; Raza, H.; Al-Ramadi, B.; Adeghate, E. Diabetes Mellitus Alters the Immuno-Expression of Neuronal Nitric Oxide Synthase in the Rat Pancreas. Int. J. Mol. Sci. 2022, 23, 4974. [Google Scholar] [CrossRef] [PubMed]

| Organ/Systems/Condition | Effect | Reference |
|---|---|---|
| Liver | Increases the expression of hepatic lipogenic genes and PPARγ | [14] |
| Cardiovascular system | Increases angiogenesis, blood flow, capillary density, and protects endothelial cells | [23] |
| Improves cardiac function and coronary flow | [77] | |
| Diabetes mellitus | Low adropin level increases the risk of chronic complications of diabetes | [63,64,65] |
| Stimulates insulin signal pathways by promoting Akt phosphorylation | [20] | |
| Fat | Suppresses lipid accumulation | [78,79] |
| Inflammation | Inhibits TNF-α | [80] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ali, I.I.; D’Souza, C.; Singh, J.; Adeghate, E. Adropin’s Role in Energy Homeostasis and Metabolic Disorders. Int. J. Mol. Sci. 2022, 23, 8318. https://doi.org/10.3390/ijms23158318
Ali II, D’Souza C, Singh J, Adeghate E. Adropin’s Role in Energy Homeostasis and Metabolic Disorders. International Journal of Molecular Sciences. 2022; 23(15):8318. https://doi.org/10.3390/ijms23158318
Chicago/Turabian StyleAli, Ifrah Ismail, Crystal D’Souza, Jaipaul Singh, and Ernest Adeghate. 2022. "Adropin’s Role in Energy Homeostasis and Metabolic Disorders" International Journal of Molecular Sciences 23, no. 15: 8318. https://doi.org/10.3390/ijms23158318