Effect of Heat Shock Preconditioning on Pressure Injury Prevention via Hsp27 Upregulation in Rat Models
Abstract
:1. Introduction
2. Results
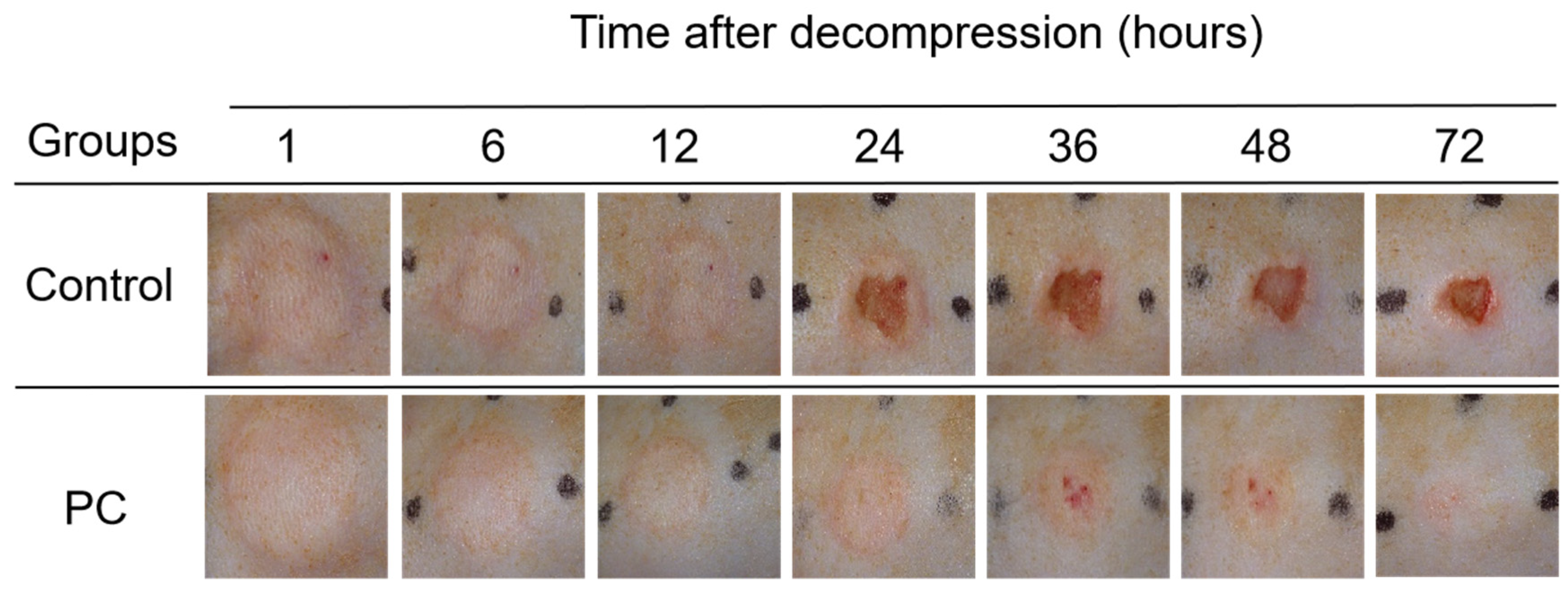
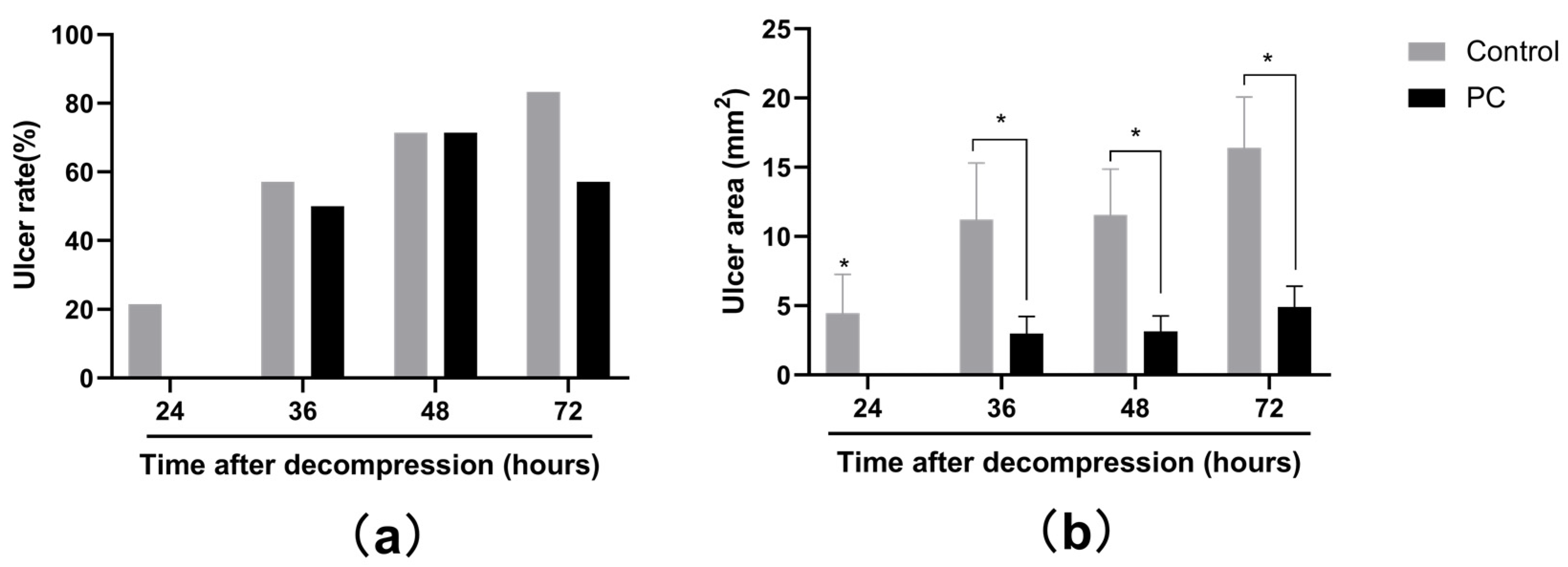
2.1. Macroscopic Observation of PI Wounds
2.2. Skin Temperature and Moisture of PI Wounds
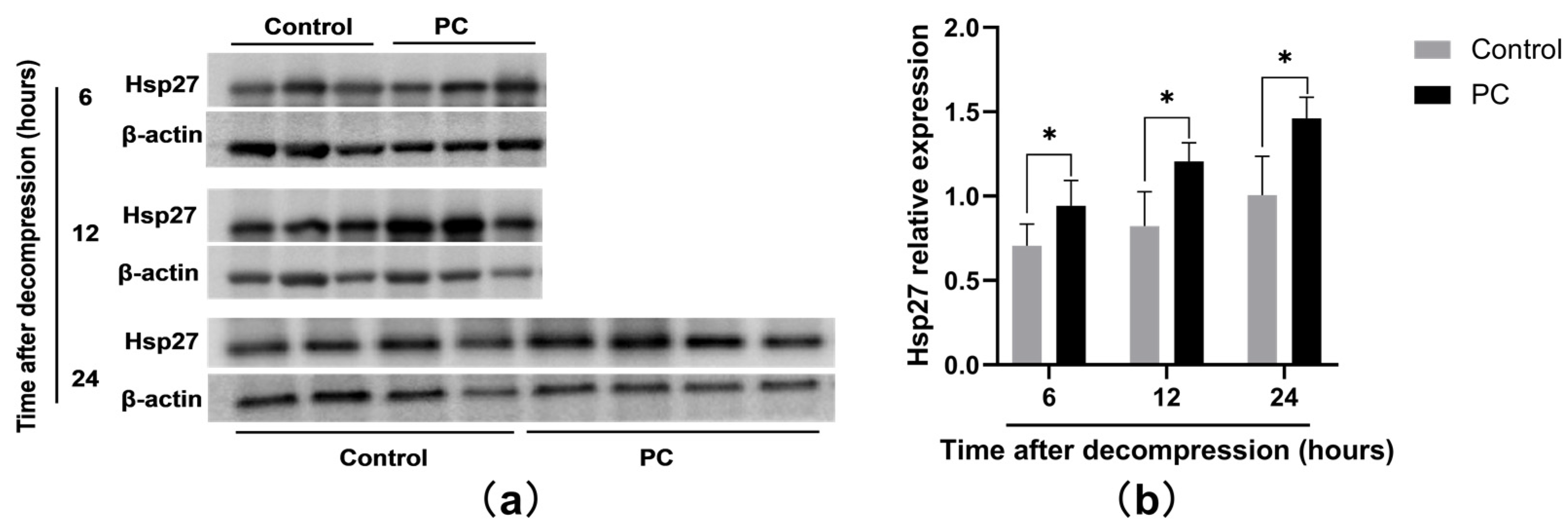
2.3. Hsp27 Expression of PI Wounds
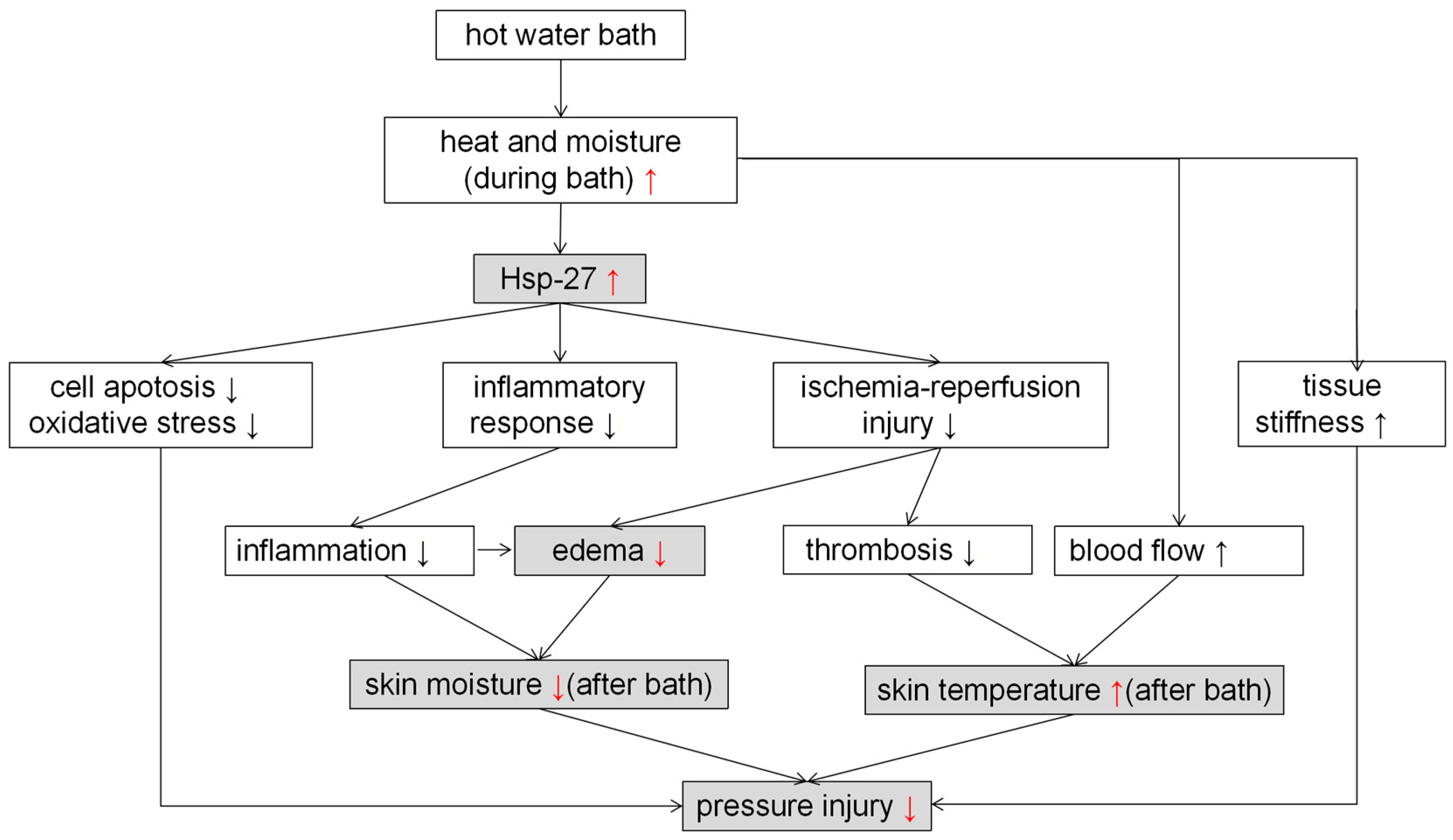
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Grouping
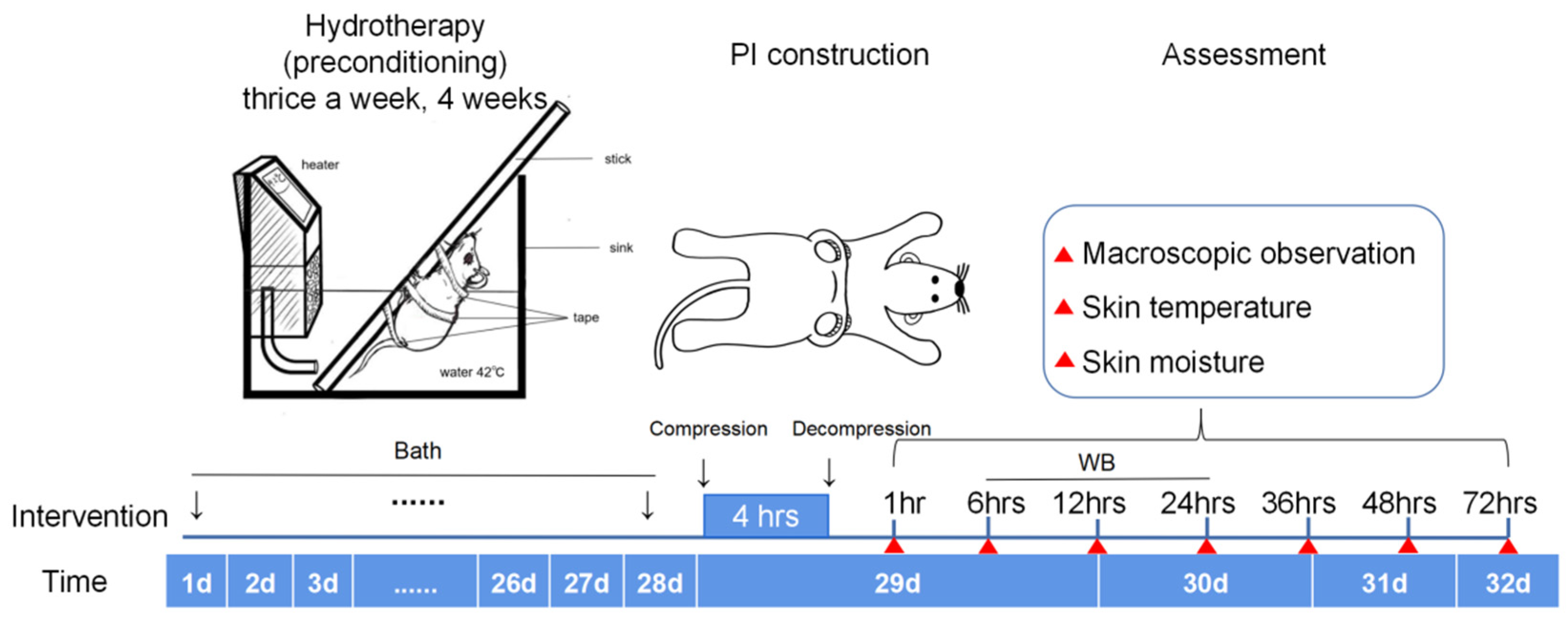
4.3. Protocol of The Study Design
4.4. Preconditioning Group
4.5. Control Group
4.6. Construction of PI Rat Models
4.7. Macroscopic Observation of PI
4.8. Assessment of Skin Temperature
4.9. Skin Moisture Measures
4.10. Protein Preparation and Western Blot Analysis
4.11. Statistical Analysis
5. Conclusions
6. Limitations
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- National Pressure Ulcer Advisory Panel; European Pressure Ulcer Advisory Panel. Prevention and Treatment of Pressure Ulcers: Clinical Practice Guideline; National Pressure Ulcer Advisory Panel: Washington, DC, USA, 2019. [Google Scholar]
- Oliveira, A.L.; Moore, Z.; Connor, T.O.; Patton, D. Accuracy of ultrasound, thermography and subepidermal moisture in predicting pressure ulcers: A systematic review. J. Wound Care 2017, 26, 199–215. [Google Scholar] [CrossRef] [PubMed]
- Labeau, S.O.; Afonso, E.; Benbenishty, J.; Blackwood, B.; Boulanger, C.; Brett, S.J.; Calvino-Gunther, S.; Chaboyer, W.; Coyer, F.; Deschepper, M.; et al. Prevalence, associated factors and outcomes of pressure injuries in adult intensive care unit patients: The DecubICUs study. Intensiv. Care Med. 2020, 47, 160–169. [Google Scholar] [CrossRef]
- Flock, P. Pilot Study to Determine the Effectiveness of Diamorphine Gel to Control Pressure Ulcer Pain. J. Pain Symptom Manag. 2003, 25, 547–554. [Google Scholar] [CrossRef]
- Healthcare Cost and Utilization Project (HCUP) Statistical Briefs; Agency for Health Care Policy and Research: Rockville, MD, USA, 2006.
- Brandeis, G.H.; Morris, J.N.; Nash, D.J.; Lipsitz, L.A. The epidemiology and natural history of pressure ulcers in elderly nursing home residents. JAMA 1990, 264, 2905–2909. [Google Scholar] [CrossRef] [PubMed]
- Berlowitz, D.R.; Brandeis, G.H.; Anderson, J.; Du, W.; Brand, H. Effect of Pressure Ulcers on the Survival of Long-Term Care Residents. J. Gerontol. A Biol. Sci. Med. Sci. 1997, 52, M106–M110. [Google Scholar] [CrossRef]
- Redelings, M.D.; Lee, N.E.; Sorvillo, F. Pressure ulcers: More lethal than we thought? Adv. Skin Wound Care 2005, 18, 367–372. [Google Scholar] [CrossRef]
- Suddaby, E.C.; Barnett, S.; Facteau, L. Skin breakdown in acute care pediatrics. Pediatr. Nurs. 2005, 31, 132–138, 148. [Google Scholar]
- Amlung, S.R.; Miller, W.L.; Bosley, L.M. The 1999 National Pressure Ulcer Prevalence Survey: A benchmarketing approach. Adv. Skin Wound Care 2001, 14, 297–301. [Google Scholar] [CrossRef]
- Calabrese, E.J.; Bachmann, K.A.; Bailer, A.J.; Bolger, P.M.; Borak, J.; Cai, L.; Cedergreen, N.; Cherian, M.G.; Chiueh, C.C.; Clarkson, T.W.; et al. Biological stress response terminology: Integrating the concepts of adaptive response and preconditioning stress within a hormetic dose–response framework. Toxicol. Appl. Pharmacol. 2007, 222, 122–128. [Google Scholar] [CrossRef]
- Mattson, M.P. Hormesis defifined. Ageing Res. Rev. 2008, 7, 1–7. [Google Scholar] [CrossRef]
- Calabrese, E.J.; Agathokleous, E. Building Biological Shields via Hormesis. Trends Pharmacol. Sci. 2019, 40, 8–10. [Google Scholar] [CrossRef] [PubMed]
- Othman, F.A.; Tan, S.C. Preconditioning Strategies to Enhance Neural Stem Cell-Based Therapy for Ischemic Stroke. Brain Sci. 2020, 10, 893. [Google Scholar] [CrossRef] [PubMed]
- Moseley, P.L. Heat shock proteins and heat adaptation of the whole organism. J. Appl. Physiol. 1997, 83, 1413–1417. [Google Scholar] [CrossRef] [PubMed]
- Schöffl, F.; Prändl, R.; Reindl, A. Regulation of the Heat-Shock Response. Plant Physiol. 1998, 117, 1135–1141. [Google Scholar] [CrossRef] [PubMed]
- Pincus, D. Regulation of Hsf1 and the Heat Shock Response. Adv. Exp. Med. Biol. 2020, 1243, 41–50. [Google Scholar] [CrossRef]
- Voellmy, R.; Boellmann, F. Chaperone Regulation of the Heat Shock Protein Response. Adv. Exp. Med. Biol. 2007, 594, 89–99. [Google Scholar] [CrossRef]
- Wang, Q.; Li, X.; Xie, J.; Xie, C.; Fu, X.; Wang, Q. Heat shock pretreatment improves mesenchymal stem cell viability by heat shock proteins and autophagy to prevent cisplatin-induced granulosa cell apoptosis. Stem Cell Res. Ther. 2019, 10, 1–12. [Google Scholar] [CrossRef]
- Loktionova, S.A.; Ilyinskaya, O.; Kabakov, A.E. Early and delayed tolerance to simulated ischemia in heat-preconditioned endothelial cells: A role for HSP27. Am. J. Physiol. Content 1998, 275, H2147–H2158. [Google Scholar] [CrossRef]
- Chen, L.H. Clinical study on heat-sensitive moxibustion in preventing bedsores after ischemic stroke. Shanghai J. Acupunct. Moxibustion 2018, 37, 262–265. [Google Scholar]
- Petrofsky, J.; Bains, G.; Prowse, M.; Gunda, S.; Berk, L.; Raju, C.; Ethiraju, G.; Vanarasa, D.; Madani, P. Dry heat, moist heat and body fat: Are heating modalities really effective in people who are overweight? J. Med. Eng. Technol. 2009, 33, 361–369. [Google Scholar] [CrossRef]
- Alderton, F.; Darroch, P.; Sambi, B.; McKie, A.; Ahmed, I.S.; Pyne, N.; Pyne, S. G-protein-coupled receptor stimulation of the p42/p44 mitogen-activated protein kinase pathway is attenuated by lipid phosphate phosphatases 1, 1a, and 2 in human embryonic kidney 293 cells. J. Biol. Chem. 2001, 276, 13452–13460. [Google Scholar] [CrossRef] [PubMed]
- Alderton, F.; Sambi, B.; Tate, R.; Pyne, N.J.; Pyne, S. Assessment of agonism at G-protein coupled receptors by phosphatidic acid and lysophosphatidic acid in human embryonic kidney 293 cells. Br. J. Pharmacol. 2001, 134, 6–9. [Google Scholar] [CrossRef] [PubMed]
- Burke, D.T.; Ho, C.H.-K.; Saucier, M.A.; Stewart, G. Effects of hydrotherapy on pressure ulcer healing1. Am. J. Phys. Med. Rehabil. 1998, 77, 394–398. [Google Scholar] [CrossRef] [PubMed]
- Szekeres, M.; MacDermid, J.C.; Grewal, R.; Birmingham, T. The short-term effects of hot packs vs. therapeutic whirlpool on active wrist range of motion for patients with distal radius fracture: A randomized controlled trial. J. Hand Ther. 2018, 31, 276–281. [Google Scholar] [CrossRef] [PubMed]
- King, M.R. Principles and Application of Hydrotherapy for Equine Athletes. Vet. Clin. N. Am. Equine Pract. 2016, 32, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Nocera, T.; Jean-Decoster, C.; Georgescu, V.; Guerrero, D. Benefits of Avène thermal hydrotherapy in chronic skin diseases and dermatological conditions: An overview. J. Eur. Acad. Dermatol. Venereol. 2020, 34, 49–52. [Google Scholar] [CrossRef]
- Gálvez, I.; Torres-Piles, S.; Ortega-Rincón, E. Balneotherapy, Immune System, and Stress Response: A Hormetic Strategy? Int. J. Mol. Sci. 2018, 19, 1687. [Google Scholar] [CrossRef]
- Hu, S.; Hu, Y.Z.; Wang, X.P.; Teng, Y.; Wang, H.P. Enthesiopathy prevention by heat shock pretreatment. Chin. J. Trad. Med. Traum. Orthop. 2006, 14, 88–90. [Google Scholar]
- Yamashita, N.; Hoshida, S.; Taniguchi, N.; Kuzuya, T.; Hori, M. Whole-body hyperthermia provides biphasic cardioprotection against ischemia/reperfusion injury in the rat. Circulation 1998, 98, 1414–1421. [Google Scholar] [CrossRef]
- Kavanagh, K.; Davis, A.T.; Jenkins, K.A.; Flynn, D.M. Effects of heated hydrotherapy on muscle HSP70 and glucose metabolism in old and young vervet monkeys. Cell Stress Chaperones 2016, 21, 717–725. [Google Scholar] [CrossRef]
- Chang, S.W.; Miura, D.; Kitayama, A.; Isshi, H.; Liang, J.Y.; Chen, Q.; Shu, Y.S.; Takashi, E. Prevention and treatment effect of baxianxiaoyao decoction medicated bathing on rat pressure injury. Chin. J. Mod. Nurs. 2017, 23, 2714–2720. [Google Scholar]
- Sart, S.; Ma, T.; Li, Y. Preconditioning Stem Cells for In Vivo Delivery. BioRes. Open Access 2014, 3, 137–149. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.; Knapp, C.F.; Donofrio, J.C.; Salcido, R. Temperature effects on surface pressure-induced changes in rat skin perfu-sion: Implications in pressure ulcer development. J. Rehabil. Res. Dev. 1999, 36, 189–201. [Google Scholar] [PubMed]
- Petrofsky, J.; Berk, L.; Alshammari, F.; Lee, H.; Hamdan, A.; Yim, J.E.; Patel, D.; Kodawala, Y.; Shetye, G.; Chen, W.-T.; et al. The Effect of Moist Air on Skin Blood Flow and Temperature in Subjects With and Without Diabetes. Diabetes Technol. Ther. 2012, 14, 105–116. [Google Scholar] [CrossRef] [PubMed]
- Petrofsky, J.S.; Berk, L.; Alshammari, F.; Lee, H.; Hamdan, A.; Yim, J.E.; Kodawala, Y.; Patel, D.; Nevgi, B.; Shetye, G.; et al. The interrelationship between air temperature and humidity as applied locally to the skin: The resultant re-sponse on skin temperature and blood flow with age differences. Med. Sci. Monit. 2012, 18, CR201–CR208. [Google Scholar] [CrossRef]
- Tzen, Y.; Champagne, P.T.; Rao, V.; Wang, J.; Tan, W.; Klakeel, M.; Jain, N.B.; Wukich, D.K.; Koh, T.J. Utilisation of skin blood flow as a precursor for pressure injury development in persons with acute spinal cord injury: A proof of concept. Int. Wound J. 2022. online ahead of print. [Google Scholar] [CrossRef]
- Wang, Y.Y.; Zhang, C.Y.; Sun, Y.; Tu, Q.; Jiang, L.P. The role of oxidative stress and apoptosts in the progress of early stage pressure ulcer. Chin. J. Nurs. 2011, 46, 508–510. [Google Scholar]
- Camandona, V.D.L.; Rios-Anjos, R.M.; Alegria, T.G.P.; Pereira, F.; Bicev, R.N.; da Cunha, F.M.; Digiampietri, L.A.; de Barros, M.H.; Netto, L.E.S.; Ferreira-Junior, J.R. Expression of human HSP27 in yeast extends replicative lifespan and uncovers a hormetic response. Biogerontology 2020, 21, 559–575. [Google Scholar] [CrossRef]
- Mao, J.; Li, G.Q. Expression of heat shock protein 17 in tongue squamous cell carcinoma cells after hyperthermia. Beijing J. Stomatol. 2015, 23, 33–35. [Google Scholar]
- Moloney, T.C.; Hoban, D.B.; Barry, F.P.; Howard, L.; Dowd, E. Kinetics of thermally induced heat shock protein 27 and 70 expression by bone marrow-derived mesenchymal stem cells. Protein Sci. 2012, 21, 904–909. [Google Scholar] [CrossRef]
- Geum, D.; Son, G.H.; Kim, K. Phosphorylation-dependent Cellular Localization and Thermoprotective Role of Heat Shock Protein 25 in Hippocampal Progenitor Cells. J. Biol. Chem. 2002, 277, 19913–19921. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Huang, W.; Meng, W.; Jegga, A.G.; Wang, Y.; Cai, W.; Kim, H.W.; Pasha, Z.; Wen, Z.; Rao, F.; et al. Heat shock improves Sca-1+ stem cell survival and directs ischemic cardiomyocytes toward a prosurvival phe-notype via exosomal transfer: A critical role for HSF1/miR-34a/HSP70 pathway. Stem Cells 2014, 32, 462–472. [Google Scholar] [CrossRef] [PubMed]
- Kostenko, S.; Moens, U. Heat shock protein 27 phosphorylation: Kinases, phosphatases, functions and pathology. Cell. Mol. Life Sci. 2009, 66, 3289–3307. [Google Scholar] [CrossRef] [PubMed]
- Zou, S.; Liao, M.; Yang, J.; Huang, T.; Green, M.; Wu, J.; Qu, L. Heat shock protein 27 plays a protective role in thoracic aortic dissection by promoting cell proliferation and inhibiting apoptosis. Cell. Mol. Biol. Lett. 2017, 22, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Peirce, S.M.; Skalak, T.C.; Rodeheaver, G.T. Ischemia-reperfusion injury in chronic pressure ulcer formation: A skin model in the rat. Wound Repair Regen. 2000, 8, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Xue, Y.; Hou, R.; Wang, H.F. Effects of Different Pressure Forms on Pressure Ulcer Ischemia-reperfusion Injury. Nurs. J. Chin. PLA 2012, 29, 5–7, 11. [Google Scholar] [CrossRef]
- Sekiguchi, A.; Motegi, S.I.; Uchiyama, A.; Uehara, A.; Fujiwara, C.; Yamazaki, S.; Perera, B.; Nakamura, H.; Ogino, S.; Yoko-yama, Y.; et al. Botulinum toxin B suppresses the pressure ulcer formation in cutaneous ische-mia-reperfusion injury mouse model: Possible regulation of oxidative and endoplasmic reticulum stress. J. Dermatol. Sci. 2018, 90, 144–153. [Google Scholar] [CrossRef]
- Chen, S.W.; Park, S.W.; Kim, M.; Brown, K.M.; D’Agati, V.D.; Lee, H.T. Human Heat Shock Protein 27 Overexpressing Mice Are Protected Against Hepatic Ischemia and Reperfusion Injury. Transplantation 2009, 87, 1478–1487. [Google Scholar] [CrossRef]
- Joo, J.D.; Kim, M.; Horst, P.; Kim, J.; D’Agati, V.D.; Emala, C.W., Sr.; Lee, H.T. Acute and delayed renal protection against renal ischemia and reperfusion injury with A1 adenosine receptors. Am. J. Physiol.-Renal Physiol. 2007, 293, F1847–F1857. [Google Scholar] [CrossRef]
- Efthymiou, C.A.; Mocanu, M.M.; de Belleroche, J.; Wells, D.J.; Latchmann, D.S.; Yellon, D.M. Heat shock protein 27 protects the heart against myocardial infarction. Basic Res. Cardiol. 2004, 99, 392–394. [Google Scholar] [CrossRef]
- Bruey, J.-M.; Ducasse, C.; Bonniaud, P.; Ravagnan, L.; Susin, S.A.; Diaz-Latoud, C.; Gurbuxani, S.; Arrigo, A.-P.; Kroemer, G.; Solary, E.; et al. Hsp27 negatively regulates cell death by interacting with cytochrome c. Nat. Cell Biol. 2000, 2, 645–652. [Google Scholar] [CrossRef] [PubMed]
- He, Z.Y.; Ge, C.L.; Guo, K.J.; Guo, R.X. Effects of HSP27 expression on the adhesion junction of cells during acute pancreatitis in rats. Chin. J. General. Surg. 2007, 16, 429–432. [Google Scholar]
- Qi, X.Q.; Yu, M.K. Inflammatory response and cerebral edema after cerebral trauma. Chin. J. Neuromed. 2007, 6, 850–852. [Google Scholar]
- Wang, Y.; Zheng, G.R.; Gao, J.H.; Yan, Z.; Diao, B. Influence of fish oil on immune function, inflammatory response and cell apoptosis in stage I elderly pressure sores model rats. Chin. Med. Herald 2016, 10, 10–12. [Google Scholar]
- Miller-Graziano, C.L.; De, A.; Laudanski, K.; Herrmann, T.; Bandyopadhyay, S. HSP27: An anti-inflammatory and im-munomodulatory stress protein acting to dampen immune function. Novartis Found. Symp. 2008, 291, 196–224. [Google Scholar] [CrossRef]
- Allman, R.M. Pressure ulcers: Using what we know to improve quality of care. J. Am. Geriatr. Soc. 2001, 49, 996–997. [Google Scholar] [CrossRef]
- Kim, C.-G.; Park, S.; Ko, J.W.; Jo, S. The relationship of subepidermal moisture and early stage pressure injury by visual skin assessment. J. Tissue Viability 2018, 27, 130–134. [Google Scholar] [CrossRef]
- Bates-Jensen, B.M.; McCreath, H.E.; Kono, A.; Apeles, N.C.R.; Alessi, C. Subepidermal Moisture Predicts Erythema and Stage 1 Pressure Ulcers in Nursing Home Residents: A Pilot Study. J. Am. Geriatr. Soc. 2007, 55, 1199–1205. [Google Scholar] [CrossRef]
- Bates-Jensen, B.M.; McCreath, H.E.; Pongquan, V.; Apeles, N.C.R. Subepidermal moisture differentiates erythema and stage I pressure ulcers in nursing home residents. Wound Repair Regen. 2008, 16, 189–197. [Google Scholar] [CrossRef]
- Moore, Z.; Patton, D.; Rhodes, S.L.; O’Connor, T. Subepidermal moisture (SEM) and bioimpedance: A literature review of a novel method for early detection of pressure-induced tissue damage (pressure ulcers). Int. Wound J. 2017, 14, 331–337. [Google Scholar] [CrossRef]
- Newman, D.K.; Preston, A.M.; Salazar, S. Moisture control, urinary and fecal incontinence, and perineal skin management. In Chronic Wound Care: A Clinical Source Book for Healthcare Professionals, 4th ed.; Krasner, D.L., Rodeheaver, G.T., Sibbald, R.G., Eds.; HMP Communications: Malvern, UK, 2007; pp. 609–627. [Google Scholar]
- Reger, S.I.; Ranganathan, V.K.; Sahgal, V. Support surface, interface pressure, microenvironment and the prevalence of pressure ulcer: Analysis of the literature. Ostomy Wound Manag. 2007, 53, 50–58. [Google Scholar]
- Zhang, Z.; Luo, X.Z.; Cao, F. The risk factors of thrombosis after ischemia-reperfusion of skeletal muscle. Jiangsu Med. J. 2008, 34, 1231–1232. [Google Scholar]
- Liu, T.; Wu, X.R. Research progress on relationship between skin temperature changes and pressure injuries at compression interface. J. Nurs. Sci. 2019, 34, 99–102. [Google Scholar]
- Sprigle, S.; Linden, M.; McKenna, D.; Davis, K.; Riordan, B. Clinical Skin Temperature Measurement to Predict Incipient Pressure Ulcers. Adv. Ski. Wound Care 2001, 14, 133–137. [Google Scholar] [CrossRef]
- Xu, H.; Wang, Y.; Takashi, E.; Kamijo, A.; Miura, D.; Karasawa, K.; Kitayama, A.; Lu, J.; Zhang, L. Predicting the different progressions of early pressure injury by ultraviolet photography in rat models. Int. Wound J. 2021, 19, 834–844. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, H.; Takashi, E.; Liang, J.; Chen, Y.; Yuan, Y.; Fan, J. Effect of Heat Shock Preconditioning on Pressure Injury Prevention via Hsp27 Upregulation in Rat Models. Int. J. Mol. Sci. 2022, 23, 8955. https://doi.org/10.3390/ijms23168955
Xu H, Takashi E, Liang J, Chen Y, Yuan Y, Fan J. Effect of Heat Shock Preconditioning on Pressure Injury Prevention via Hsp27 Upregulation in Rat Models. International Journal of Molecular Sciences. 2022; 23(16):8955. https://doi.org/10.3390/ijms23168955
Chicago/Turabian StyleXu, Huiwen, En Takashi, Jingyan Liang, Yajie Chen, Yuan Yuan, and Jianglin Fan. 2022. "Effect of Heat Shock Preconditioning on Pressure Injury Prevention via Hsp27 Upregulation in Rat Models" International Journal of Molecular Sciences 23, no. 16: 8955. https://doi.org/10.3390/ijms23168955






