The p53 and Calcium Regulated Actin Rearrangement in Model Cells
Abstract
:1. Introduction
2. Results
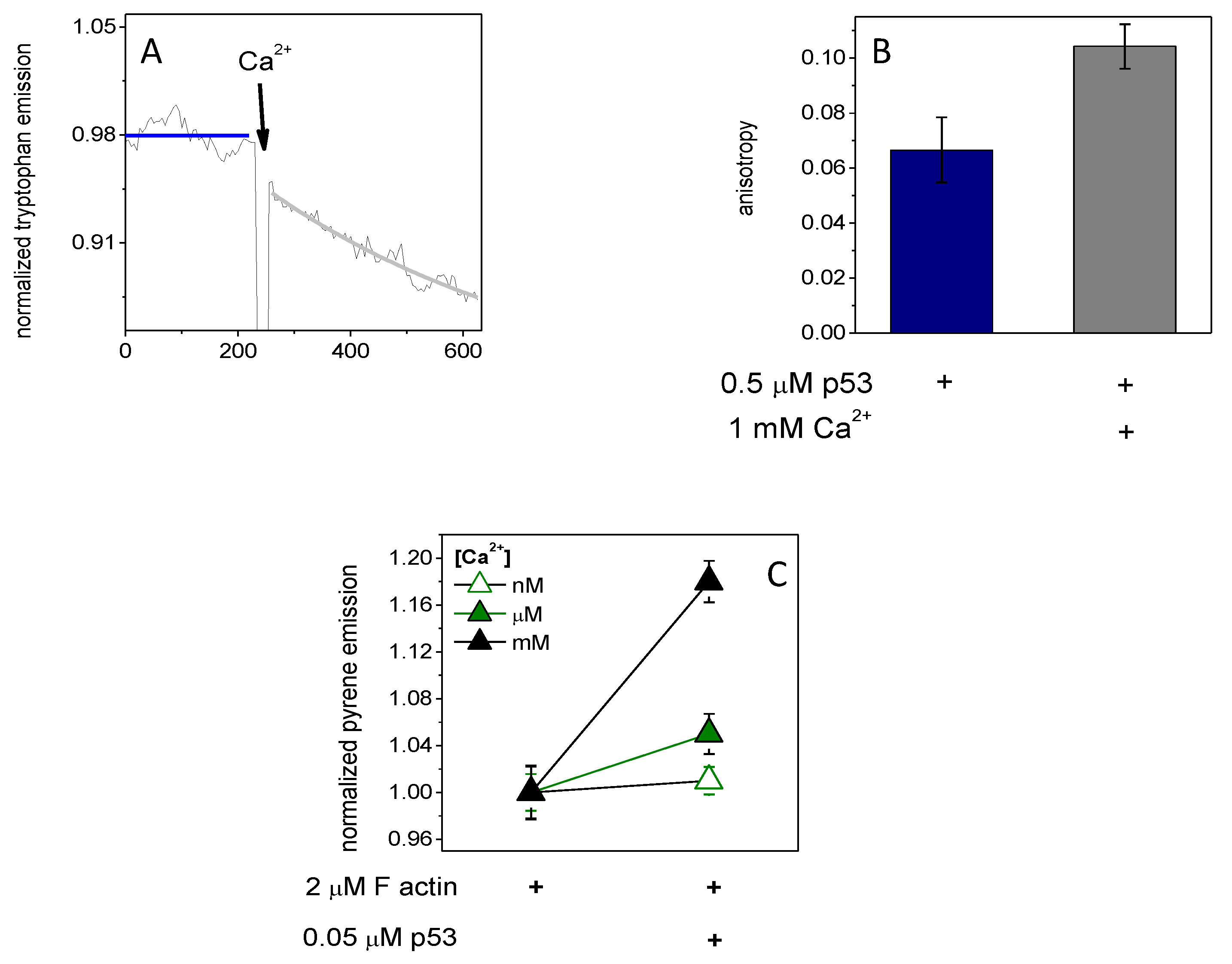
2.1. p53 Forms Dimers and Binds to Actin Monomers and Filaments
2.2. JMY Reorders Actin Filaments by Millimolar Calcium
2.3. Actin Competes with p53 on JMY and p53 Competes with Actin on GSN in a Calcium-Dependent Manner
2.4. Increased Cytoplasmic GSN Level in HeLa Cells
2.5. High Cytoplasmic GSN or JMY Affects the Division and Motility of HeLa Cells
3. Discussion
4. Materials and Methods
4.1. Proteins
4.2. Fluorescence Spectroscopy
4.3. Cosedimentation
4.4. Low-Voltage Electroporation
4.5. Cell Culture
4.6. Cell Fixation, Labeling
4.7. Microscopy, Confocal Microscopy
4.8. Scratching Method
4.9. Image Analysis
4.10. Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Doherty, G.J.; McMahon, H.T. Mediation, modulation, and consequences of membrane-cytoskeleton interactions. Annu. Rev. Biophys. 2008, 37, 65–95. [Google Scholar] [CrossRef] [PubMed]
- Welch, W.J. How cells respond to stress. Sci. Am. 1993, 268, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Atif, R.M.; Shahid, L.; Waqas, M.; Ali, B.; Rashid, M.A.R.; Azeem, F.; Nawaz, M.A.; Wani, S.H.; Chung, G. Insights on Calcium-Dependent Protein Kinases (CPKs) Signaling for Abiotic Stress Tolerance in Plants. Int. J. Mol. Sci. 2019, 20, 5298. [Google Scholar] [CrossRef] [PubMed]
- Ermak, G.; Davies, K.J. Calcium and oxidative stress: From cell signaling to cell death. Mol. Immunol. 2002, 38, 713–721. [Google Scholar] [CrossRef]
- Povea-Cabello, S.; Oropesa-Avila, M.; de la Cruz-Ojeda, P.; Villanueva-Paz, M.; de la Mata, M.; Suarez-Rivero, J.M.; Alvarez-Cordoba, M.; Villalon-Garcia, I.; Cotan, D.; Ybot-Gonzalez, P.; et al. Dynamic Reorganization of the Cytoskeleton during Apoptosis: The Two Coffins Hypothesis. Int. J. Mol. Sci. 2017, 18, 2393. [Google Scholar] [CrossRef]
- Desouza, M.; Gunning, P.W.; Stehn, J.R. The actin cytoskeleton as a sensor and mediator of apoptosis. Bioarchitecture 2012, 2, 75–87. [Google Scholar] [CrossRef]
- Leadsham, J.E.; Kotiadis, V.N.; Tarrant, D.J.; Gourlay, C.W. Apoptosis and the yeast actin cytoskeleton. Cell Death Differ. 2010, 17, 754–762. [Google Scholar] [CrossRef] [PubMed]
- Rodolfo, C.; Piacentini, M. Does cytoskeleton ‘Akt’ in apoptosis? Cell Death Differ. 2002, 9, 477–478. [Google Scholar] [CrossRef]
- Gourlay, C.W.; Ayscough, K.R. The actin cytoskeleton: A key regulator of apoptosis and ageing? Nat. Rev. Mol. Cell Biol. 2005, 6, 583–589. [Google Scholar] [CrossRef]
- Ebata, T.; Hirata, H.; Kawauchi, K. Functions of the Tumor Suppressors p53 and Rb in Actin Cytoskeleton Remodeling. BioMed Res. Int. 2016, 2016. [Google Scholar] [CrossRef]
- An, J.H.; Kim, J.W.; Jang, S.M.; Kim, C.H.; Kang, E.J.; Choi, K.H. Gelsolin negatively regulates the activity of tumor suppressor p53 through their physical interaction in hepatocarcinoma HepG2 cells. Biochem. Biophys. Res. Commun. 2011, 412, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Adighibe, O.; Pezzella, F. The Role of JMY in p53 Regulation. Cancers 2018, 10, 173. [Google Scholar] [CrossRef] [PubMed]
- Zuchero, J.B.; Coutts, A.S.; Quinlan, M.E.; Thangue, N.B.; Mullins, R.D. p53-cofactor JMY is a multifunctional actin nucleation factor. Nat. Cell Biol. 2009, 11, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Thompson, C.C.; Ashcroft, F.J.; Patel, S.; Saraga, G.; Vimalachandran, D.; Prime, W.; Campbell, F.; Dodson, A.; Jenkins, R.E.; Lemoine, N.R.; et al. Pancreatic cancer cells overexpress gelsolin family-capping proteins, which contribute to their cell motility. Gut 2007, 56, 95–106. [Google Scholar] [CrossRef] [PubMed]
- Shieh, D.B.; Godleski, J.; Herndon, J.E., 2nd; Azuma, T.; Mercer, H.; Sugarbaker, D.J.; Kwiatkowski, D.J. Cell motility as a prognostic factor in Stage I nonsmall cell lung carcinoma: The role of gelsolin expression. Cancer 1999, 85, 47–57. [Google Scholar] [CrossRef]
- Visapaa, H.; Bui, M.; Huang, Y.; Seligson, D.; Tsai, H.; Pantuck, A.; Figlin, R.; Rao, J.Y.; Belldegrun, A.; Horvath, S.; et al. Correlation of Ki-67 and gelsolin expression to clinical outcome in renal clear cell carcinoma. Urology 2003, 61, 845–850. [Google Scholar] [CrossRef]
- Li, W.X.; Yang, M.X.; Hong, X.Q.; Dong, T.G.; Yi, T.; Lin, S.L.; Qin, X.Y.; Niu, W.X. Overexpression of gelsolin reduces the proliferation and invasion of colon carcinoma cells. Mol. Med. Rep. 2016, 14, 3059–3065. [Google Scholar] [CrossRef]
- Stock, A.M.; Klee, F.; Edlund, K.; Grinberg, M.; Hammad, S.; Marchan, R.; Cadenas, C.; Niggemann, B.; Zanker, K.S.; Rahnenfuhrer, J.; et al. Gelsolin Is Associated with Longer Metastasis-free Survival and Reduced Cell Migration in Estrogen Receptor-positive Breast Cancer. Anticancer Res. 2015, 35, 5277–5285. [Google Scholar]
- Guo, Y.; Zhang, H.; Xing, X.; Wang, L.; Zhang, J.; Yan, L.; Zheng, X.; Zhang, M. Gelsolin regulates proliferation, apoptosis and invasion in natural killer/T-cell lymphoma cells. Biol. Open 2018, 7, bio027557. [Google Scholar]
- Winston, J.S.; Asch, H.L.; Zhang, P.J.; Edge, S.B.; Hyland, A.; Asch, B.B. Downregulation of gelsolin correlates with the progression to breast carcinoma. Breast Cancer Res. Treat. 2001, 65, 11–21. [Google Scholar] [CrossRef]
- Dosaka-Akita, H.; Hommura, F.; Fujita, H.; Kinoshita, I.; Nishi, M.; Morikawa, T.; Katoh, H.; Kawakami, Y.; Kuzumaki, N. Frequent loss of gelsolin expression in non-small cell lung cancers of heavy smokers. Cancer Res. 1998, 58, 322–327. [Google Scholar]
- Tanaka, H.; Shirkoohi, R.; Nakagawa, K.; Qiao, H.; Fujita, H.; Okada, F.; Hamada, J.; Kuzumaki, S.; Takimoto, M.; Kuzumaki, N. siRNA gelsolin knockdown induces epithelial-mesenchymal transition with a cadherin switch in human mammary epithelial cells. Int. J. Cancer 2006, 118, 1680–1691. [Google Scholar] [CrossRef] [PubMed]
- Rao, J.; Seligson, D.; Visapaa, H.; Horvath, S.; Eeva, M.; Michel, K.; Pantuck, A.; Belldegrun, A.; Palotie, A. Tissue microarray analysis of cytoskeletal actin-associated biomarkers gelsolin and E-cadherin in urothelial carcinoma. Cancer 2002, 95, 1247–1257. [Google Scholar] [CrossRef] [PubMed]
- Shieh, D.B.; Chen, I.W.; Wei, T.Y.; Shao, C.Y.; Chang, H.J.; Chung, C.H.; Wong, T.Y.; Jin, Y.T. Tissue expression of gelsolin in oral carcinogenesis progression and its clinicopathological implications. Oral Oncol. 2006, 42, 599–606. [Google Scholar] [CrossRef] [PubMed]
- Coutts, A.S.; Weston, L.; La Thangue, N.B. A transcription co-factor integrates cell adhesion and motility with the p53 response. Proc. Natl. Acad. Sci. USA 2009, 106, 19872–19877. [Google Scholar] [CrossRef] [PubMed]
- D’Souza-Schorey, C. Disassembling adherens junctions: Breaking up is hard to do. Trends Cell Biol. 2005, 15, 19–26. [Google Scholar] [CrossRef]
- Jeanes, A.; Gottardi, C.J.; Yap, A.S. Cadherins and cancer: How does cadherin dysfunction promote tumor progression? Oncogene 2008, 27, 6920–6929. [Google Scholar] [CrossRef]
- Adighibe, O.; Turley, H.; Leek, R.; Harris, A.; Coutts, A.S.; La Thangue, N.; Gatter, K.; Pezzella, F. JMY protein, a regulator of P53 and cytoplasmic actin filaments, is expressed in normal and neoplastic tissues. Virchows Arch. 2014, 465, 715–722. [Google Scholar] [CrossRef]
- Saha, T.; Guha, D.; Manna, A.; Panda, A.K.; Bhat, J.; Chatterjee, S.; Sa, G. G-actin guides p53 nuclear transport: Potential contribution of monomeric actin in altered localization of mutant p53. Sci. Rep. 2016, 6, 32626. [Google Scholar] [CrossRef]
- Metcalfe, S.; Weeds, A.; Okorokov, A.L.; Milner, J.; Cockman, M.; Pope, B. Wild-type p53 protein shows calcium-dependent binding to F-actin. Oncogene 1999, 18, 2351–2355. [Google Scholar] [CrossRef]
- Chene, P. The role of tetramerization in p53 function. Oncogene 2001, 20, 2611–2617. [Google Scholar] [CrossRef] [PubMed]
- Ho, W.C.; Fitzgerald, M.X.; Marmorstein, R. Structure of the p53 core domain dimer bound to DNA. J. Biol. Chem. 2006, 281, 20494–20502. [Google Scholar] [CrossRef] [PubMed]
- Madhumalar, A.; Jun, L.H.; Lane, D.P.; Verma, C.S. Dimerization of the core domain of the p53 family: A computational study. Cell Cycle 2009, 8, 137–148. [Google Scholar] [CrossRef] [PubMed]
- Green, D.R.; Kroemer, G. Cytoplasmic functions of the tumour suppressor p53. Nature 2009, 458, 1127–1130. [Google Scholar] [CrossRef]
- Baptiste, N.; Prives, C. p53 in the cytoplasm: A question of overkill? Cell 2004, 116, 487–489. [Google Scholar] [CrossRef]
- Morselli, E.; Tasdemir, E.; Maiuri, M.C.; Galluzzi, L.; Kepp, O.; Criollo, A.; Vicencio, J.M.; Soussi, T.; Kroemer, G. Mutant p53 protein localized in the cytoplasm inhibits autophagy. Cell Cycle 2008, 7, 3056–3061. [Google Scholar] [CrossRef] [PubMed]
- Rajagopalan, S.; Jaulent, A.M.; Wells, M.; Veprintsev, D.B.; Fersht, A.R. 14-3-3 activation of DNA binding of p53 by enhancing its association into tetramers. Nucleic Acids Res. 2008, 36, 5983–5991. [Google Scholar] [CrossRef] [PubMed]
- Mattila, P.K.; Salminen, M.; Yamashiro, T.; Lappalainen, P. Mouse MIM, a tissue-specific regulator of cytoskeletal dynamics, interacts with ATP-actin monomers through its C-terminal WH2 domain. J. Biol. Chem. 2003, 278, 8452–8459. [Google Scholar] [CrossRef]
- Hou, W.; Izadi, M.; Nemitz, S.; Haag, N.; Kessels, M.M.; Qualmann, B. The Actin Nucleator Cobl Is Controlled by Calcium and Calmodulin. PLoS Biol. 2015, 13, e1002233. [Google Scholar] [CrossRef]
- Zuchero, J.B.; Belin, B.; Mullins, R.D. Actin binding to WH2 domains regulates nuclear import of the multifunctional actin regulator JMY. Mol. Biol. Cell 2012, 23, 853–863. [Google Scholar] [CrossRef]
- Rottner, K.; Hanisch, J.; Campellone, K.G. WASH, WHAMM and JMY: Regulation of Arp2/3 complex and beyond. Trends Cell Biol. 2010, 20, 650–661. [Google Scholar] [CrossRef] [PubMed]
- Ahuja, R.; Pinyol, R.; Reichenbach, N.; Custer, L.; Klingensmith, J.; Kessels, M.M.; Qualmann, B. Cordon-bleu is an actin nucleation factor and controls neuronal morphology. Cell 2007, 131, 337–350. [Google Scholar] [CrossRef] [PubMed]
- Shikama, N.; Lee, C.W.; France, S.; Delavaine, L.; Lyon, J.; Krstic-Demonacos, M.; La Thangue, N.B. A novel cofactor for p300 that regulates the p53 response. Mol. Cell 1999, 4, 365–376. [Google Scholar] [CrossRef]
- Coutts, A.S.; Boulahbel, H.; Graham, A.; La Thangue, N.B. Mdm2 targets the p53 transcription cofactor JMY for degradation. EMBO Rep. 2007, 8, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Szatmari, D.; Xue, B.; Kannan, B.; Burtnick, L.D.; Bugyi, B.; Nyitrai, M.; Robinson, R.C. ATP competes with PIP2 for binding to gelsolin. PLoS ONE 2018, 13. [Google Scholar] [CrossRef]
- Burtnick, L.D.; Koepf, E.K.; Grimes, J.; Jones, E.Y.; Stuart, D.I.; McLaughlin, P.J.; Robinson, R.C. The crystal structure of plasma gelsolin: Implications for actin severing, capping, and nucleation. Cell 1997, 90, 661–670. [Google Scholar] [CrossRef]
- Singaravelu, P.; Lee, W.L.; Wee, S.; Ghoshdastider, U.; Ding, K.; Gunaratne, J.; Grimes, J.M.; Swaminathan, K.; Robinson, R.C. Yersinia effector protein (YopO)-mediated phosphorylation of host gelsolin causes calcium-independent activation leading to disruption of actin dynamics. J. Biol. Chem. 2017, 292, 8092–8100. [Google Scholar] [CrossRef]
- Choe, H.; Burtnick, L.D.; Mejillano, M.; Yin, H.L.; Robinson, R.C.; Choe, S. The calcium activation of gelsolin: Insights from the 3 angstrom structure of the G4-G6/actin complex. J. Mol. Biol. 2002, 324, 691–702. [Google Scholar] [CrossRef]
- Burtnick, L.D.; Urosev, D.; Irobi, E.; Narayan, K.; Robinson, R.C. Structure of the N-terminal half of gelsolin bound to actin: Roles in severing, apoptosis and FAF. Embo J. 2004, 23, 2713–2722. [Google Scholar] [CrossRef]
- Kinosian, H.J.; Newman, J.; Lincoln, B.; Selden, L.A.; Gershman, L.C.; Estes, J.E. Ca2+ regulation of gelsolin activity: Binding and severing of F-actin. Biophys. J. 1998, 75, 3101–3109. [Google Scholar] [CrossRef]
- Ashish; Paine, M.S.; Perryman, P.B.; Yang, L.; Yin, H.L.; Krueger, J.K. Global structure changes associated with Ca2+ activation of full-length human plasma gelsolin. J. Biol. Chem. 2007, 282, 25884–25892. [Google Scholar] [CrossRef] [PubMed]
- Sugar, I.P.; Lindesay, J.; Schmukler, R.E. Phenomenological theory of low-voltage electroporation. Electric field calculations. J. Phys. Chem. B 2003, 107, 3862–3870. [Google Scholar] [CrossRef]
- Kurosawa, O.; Oana, H.; Matsuoka, S.; Noma, A.; Kotera, H.; Washizu, M. Electroporation through a micro-fabricated orifice and its application to the measurement of cell response to external stimuli. Meas. Sci. Technol. 2006, 17, 3127–3133. [Google Scholar] [CrossRef]
- Zhou, W.; Xiong, Z.G.; Liu, Y.; Yao, C.G.; Li, C.X. Low voltage irreversible electroporation induced apoptosis in HeLa cells. J. Cancer Res. Ther. 2012, 8, 80–85. [Google Scholar] [CrossRef]
- Guido, I.; Xiong, C.Y.; Fang, J. Low-voltage electroporation with fluidic microelectrodes. Microelectron. Eng. 2012, 98, 707–710. [Google Scholar] [CrossRef]
- Wang, Y. JimMY on the stage: Linking DNA damage with cell adhesion and motility. Cell Adhes. Migr. 2010, 4, 166–168. [Google Scholar] [CrossRef]
- Coutts, A.S.; Weston, L.; La Thangue, N.B. Actin nucleation by a transcription co-factor that links cytoskeletal events with the p53 response. Cell Cycle 2010, 9, 1511–1515. [Google Scholar] [CrossRef]
- Spudich, J.A.; Watt, S. The regulation of rabbit skeletal muscle contraction. I. Biochemical studies of the interaction of the tropomyosin-troponin complex with actin and the proteolytic fragments of myosin. J. Biol. Chem. 1971, 246, 4866–4871. [Google Scholar] [CrossRef]
- Wang, H.; Robinson, R.C.; Burtnick, L.D. The structure of native G-actin. Cytoskeleton 2010, 67, 456–465. [Google Scholar] [CrossRef] [PubMed]
- Qing, Y.; Yang, X.Q.; Zhong, Z.Y.; Lei, X.; Xie, J.Y.; Li, M.X.; Xiang, D.B.; Li, Z.P.; Yang, Z.Z.; Wang, G.; et al. Microarray analysis of DNA damage repair gene expression profiles in cervical cancer cells radioresistant to 252Cf neutron and X-rays. BMC Cancer 2010, 10, 71. [Google Scholar] [CrossRef]
- Kumei, Y.; Nakajima, T.; Sato, A.; Kamata, N.; Enomoto, S. Reduction of G1 phase duration and enhancement of c-myc gene expression in HeLa cells at hypergravity. J. Cell Sci. 1989, 93 Pt 2, 221–226. [Google Scholar] [CrossRef]
- Meck, R.A.; Carsten, A.L.; Kelsch, J.J. Growth of HeLa cells in diffusion chamber cultures in vivo. Cancer Res. 1976, 36, 2317–2320. [Google Scholar] [PubMed]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hencz, A.; Szabó-Meleg, E.; Dayo, M.Y.; Bilibani, A.; Barkó, S.; Nyitrai, M.; Szatmári, D. The p53 and Calcium Regulated Actin Rearrangement in Model Cells. Int. J. Mol. Sci. 2022, 23, 9078. https://doi.org/10.3390/ijms23169078
Hencz A, Szabó-Meleg E, Dayo MY, Bilibani A, Barkó S, Nyitrai M, Szatmári D. The p53 and Calcium Regulated Actin Rearrangement in Model Cells. International Journal of Molecular Sciences. 2022; 23(16):9078. https://doi.org/10.3390/ijms23169078
Chicago/Turabian StyleHencz, Alexandra, Edina Szabó-Meleg, Muhammad Yaqoob Dayo, Ardora Bilibani, Szilvia Barkó, Miklós Nyitrai, and Dávid Szatmári. 2022. "The p53 and Calcium Regulated Actin Rearrangement in Model Cells" International Journal of Molecular Sciences 23, no. 16: 9078. https://doi.org/10.3390/ijms23169078





