Role of Ion Channels in the Chemotransduction and Mechanotransduction in Digestive Function and Feeding Behavior
Abstract
:1. Introduction
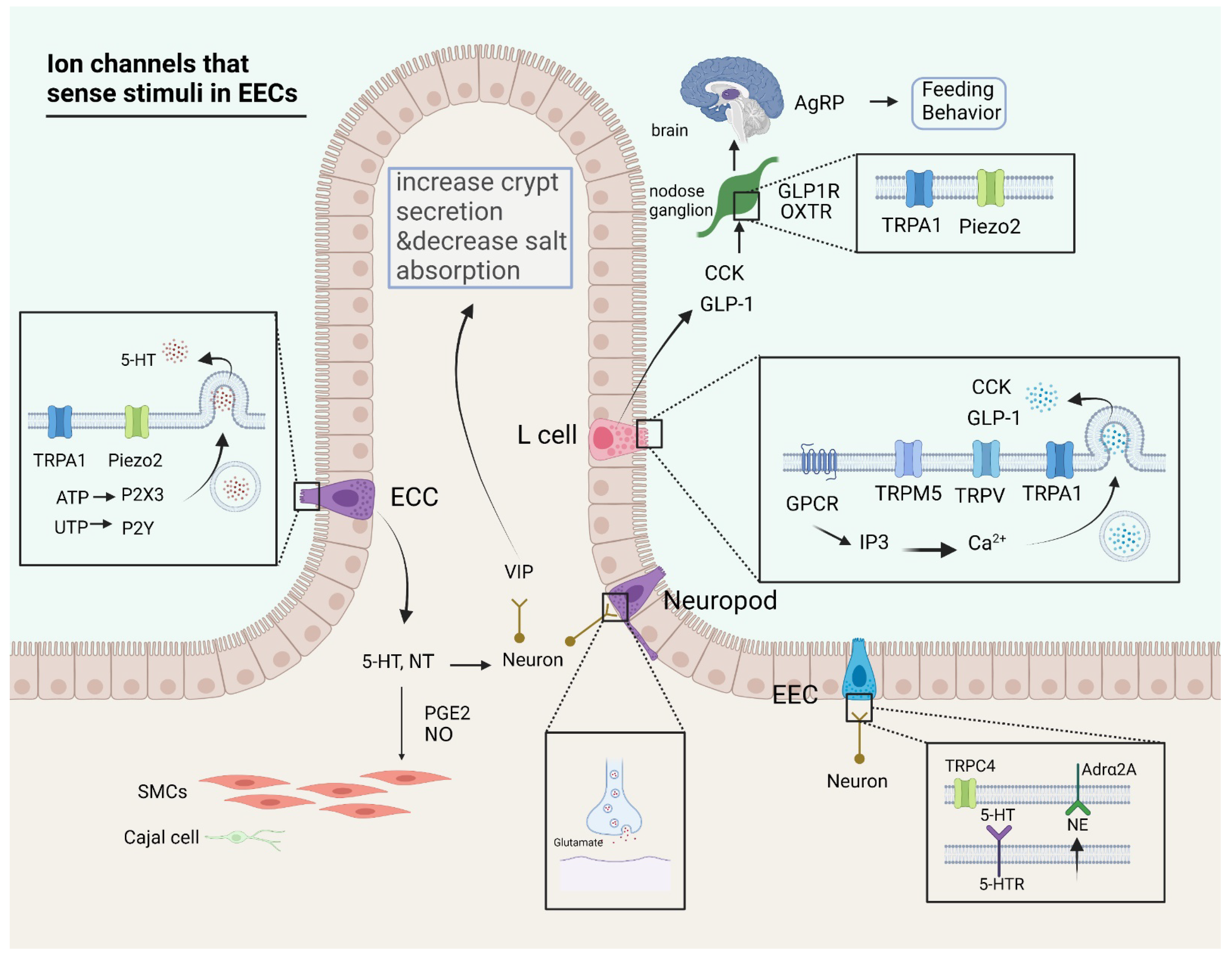
2. Ion Channels Linking Stimuli to Molecular Secretion and Intestinal Secretion
3. Ion Channels Involved in the Induction of Nutrition Response and Satiety
4. Ion Channels Modulating Gastrointestinal Motility
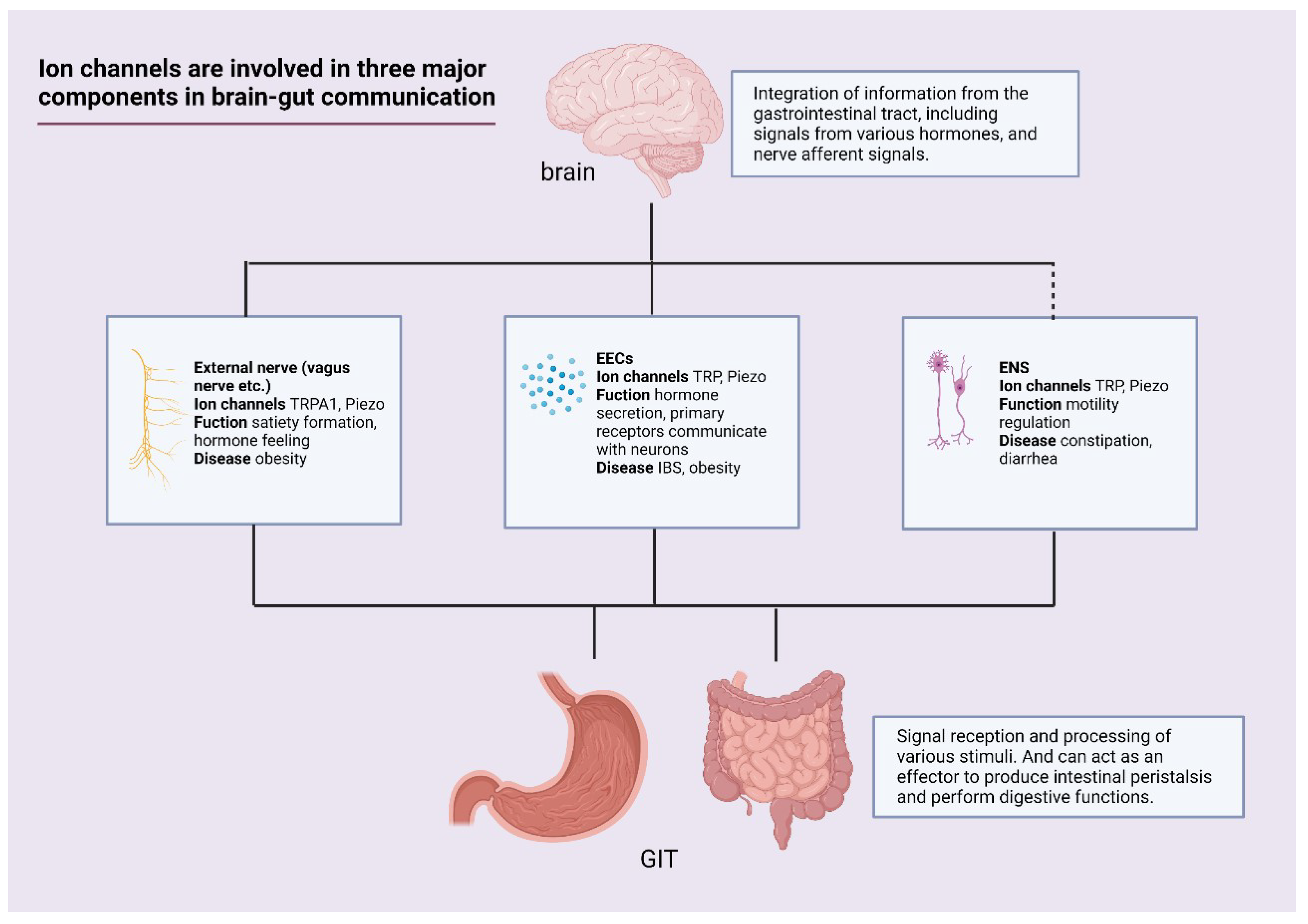
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Psichas, A.; Reimann, F.; Gribble, F.M. Gut chemosensing mechanisms. J. Clin. Investig. 2015, 125, 908–917. [Google Scholar] [CrossRef] [PubMed]
- Mercado-Perez, A.; Beyder, A. Gut feelings: Mechanosensing in the gastrointestinal tract. Nat. Rev. Gastroenterol. Hepatol. 2022, 19, 283–296. [Google Scholar] [CrossRef] [PubMed]
- Duca, F.A.; Waise, T.M.Z.; Peppler, W.T.; Lam, T.K.T. The metabolic impact of small intestinal nutrient sensing. Nat. Commun. 2021, 12, 903. [Google Scholar] [CrossRef] [PubMed]
- Clemmensen, C.; Müller, T.D.; Woods, S.C.; Berthoud, H.-R.; Seeley, R.J.; Tschöp, M.H. Gut-Brain Cross-Talk in Metabolic Control. Cell 2017, 168, 758–774. [Google Scholar] [CrossRef]
- Wachsmuth, H.R.; Weninger, S.N.; Duca, F.A. Role of the gut–brain axis in energy and glucose metabolism. Exp. Mol. Med. 2022, 54, 377–392. [Google Scholar] [CrossRef]
- Furness, J.B.; Rivera, L.R.; Cho, H.-J.; Bravo, D.M.; Callaghan, B. The gut as a sensory organ. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 729–740. [Google Scholar] [CrossRef]
- Capdevila, C.; Trifas, M.; Miller, J.; Anderson, T.; Sims, P.A.; Yan, K.S. Cellular origins and lineage relationships of the intestinal epithelium. Am. J. Physiol. Gastrointest. Liver Physiol. 2021, 321, G413–G425. [Google Scholar] [CrossRef]
- Allaire, J.M.; Crowley, S.M.; Law, H.T.; Chang, S.-Y.; Ko, H.-J.; Vallance, B.A. The Intestinal Epithelium: Central Coordinator of Mucosal Immunity. Trends Immunol. 2018, 39, 677–696. [Google Scholar] [CrossRef]
- Suzuki, T. Regulation of the intestinal barrier by nutrients: The role of tight junctions. Anim. Sci. J. 2020, 91, e13357. [Google Scholar] [CrossRef]
- Paone, P.; Cani, P.D. Mucus barrier, mucins and gut microbiota: The expected slimy partners? Gut 2020, 69, 2232–2243. [Google Scholar] [CrossRef]
- Bellono, N.W.; Bayrer, J.R.; Leitch, D.B.; Castro, J.; Zhang, C.; O’Donnell, T.A.; Brierley, S.M.; Ingraham, H.A.; Julius, D. Enterochromaffin Cells Are Gut Chemosensors that Couple to Sensory Neural Pathways. Cell 2017, 170, 185–198.e16. [Google Scholar] [CrossRef]
- Worthington, J.J.; Reimann, F.; Gribble, F.M. Enteroendocrine cells-sensory sentinels of the intestinal environment and orchestrators of mucosal immunity. Mucosal Immunol. 2017, 11, 3–20. [Google Scholar] [CrossRef]
- Kaelberer, M.M.; Rupprecht, L.E.; Liu, W.W.; Weng, P.; Bohórquez, D.V. Neuropod Cells: The Emerging Biology of Gut-Brain Sensory Transduction. Annu. Rev. Neurosci. 2020, 43, 337–353. [Google Scholar] [CrossRef]
- Spencer, N.J.; Hu, H. Enteric nervous system: Sensory transduction, neural circuits and gastrointestinal motility. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 338–351. [Google Scholar] [CrossRef]
- Kim, M.; Heo, G.; Kim, S.-Y. Neural signalling of gut mechanosensation in ingestive and digestive processes. Nat. Rev. Neurosci. 2022, 23, 135–156. [Google Scholar] [CrossRef]
- Wouters, M.M.; Farrugia, G.; Schemann, M. 5-HT receptors on interstitial cells of Cajal, smooth muscle and enteric nerves. Neurogastroenterol. Motil. 2007, 19 (Suppl. 2), 5–12. [Google Scholar] [CrossRef]
- Alcaino, C.; Knutson, K.R.; Treichel, A.J.; Yildiz, G.; Strege, P.R.; Linden, D.R.; Li, J.H.; Leiter, A.B.; Szurszewski, J.H.; Farrugia, G.; et al. A population of gut epithelial enterochromaffin cells is mechanosensitive and requires Piezo2 to convert force into serotonin release. Proc. Natl. Acad. Sci. USA 2018, 115, E7632–E7641. [Google Scholar] [CrossRef]
- Nozawa, K.; Kawabata-Shoda, E.; Doihara, H.; Kojima, R.; Okada, H.; Mochizuki, S.; Sano, Y.; Inamura, K.; Matsushime, H.; Koizumi, T.; et al. TRPA1 regulates gastrointestinal motility through serotonin release from enterochromaffin cells. Proc. Natl. Acad. Sci. USA 2009, 106, 3408–3413. [Google Scholar] [CrossRef]
- Purhonen, A.; Louhivuori, L.; Kiehne, K.; Åkerman, K.; Herzig, K. TRPA1 channel activation induces cholecystokinin release via extracellular calcium. FEBS Lett. 2008, 582, 229–232. [Google Scholar] [CrossRef]
- Moran, M.M. TRP Channels as Potential Drug Targets. Annu. Rev. Pharmacol. Toxicol. 2018, 58, 309–330. [Google Scholar] [CrossRef]
- Coste, B.; Mathur, J.; Schmidt, M.; Earley, T.J.; Ranade, S.; Petrus, M.J.; Dubin, A.E.; Patapoutian, A. Piezo1 and Piezo2 Are Essential Components of Distinct Mechanically Activated Cation Channels. Science 2010, 330, 55–60. [Google Scholar] [CrossRef]
- Wang, F.; Knutson, K.; Alcaino, C.; Linden, D.R.; Gibbons, S.J.; Kashyap, P.; Grover, M.; Oeckler, R.; Gottlieb, P.A.; Li, H.J.; et al. Mechanosensitive ion channel Piezo2 is important for enterochromaffin cell response to mechanical forces. J. Physiol. 2017, 595, 79–91. [Google Scholar] [CrossRef]
- Liñán-Rico, A.; Wunderlich, J.E.; Grants, I.S.; Frankel, W.L.; Xue, J.; Williams, K.C.; Harzman, A.; Enneking, J.T.; Cooke, H.J.; Christofi, F.L. Purinergic autocrine regulation of mechanosensitivity and serotonin release in a human EC model: ATP-gated P2X3 channels in EC are downregulated in ulcerative colitis. Inflamm. Bowel Dis. 2013, 19, 2366–2379. [Google Scholar] [CrossRef]
- Liñán-Rico, A.; Ochoa-Cortes, F.; Zuleta-Alarcon, A.; Alhaj, M.; Tili, E.; Enneking, J.; Harzman, A.; Grants, I.; Bergese, S.; Christofi, F.L. UTP—Gated Signaling Pathways of 5-HT Release from BON Cells as a Model of Human Enterochromaffin Cells. Front. Pharmacol. 2017, 8, 429. [Google Scholar] [CrossRef]
- Zhou, H.-R.; Pestka, J.J. Deoxynivalenol (Vomitoxin)-Induced Cholecystokinin and Glucagon-Like Peptide-1 Release in the STC-1 Enteroendocrine Cell Model Is Mediated by Calcium-Sensing Receptor and Transient Receptor Potential Ankyrin-1 Channel. Toxicol. Sci. 2015, 145, 407–417. [Google Scholar] [CrossRef]
- Kumar, V.; Khare, P.; Devi, K.; Kaur, J.; Kumar, V.; Kondepudi, K.K.; Chopra, K.; Bishnoi, M. Short-chain fatty acids increase intracellular calcium levels and enhance gut hormone release from STC-1 cells via transient receptor potential Ankyrin1. Fundam. Clin. Pharmacol. 2021, 35, 1004–1017. [Google Scholar] [CrossRef]
- Shah, B.P.; Liu, P.; Yu, T.; Hansen, D.R.; Gilbertson, T.A. TRPM5 is critical for linoleic acid-induced CCK secretion from the enteroendocrine cell line, STC-1. Am. J. Physiol. Physiol. 2012, 302, C210–C219. [Google Scholar] [CrossRef]
- Veldhuis, N.A.; Poole, D.P.; Grace, M.; McIntyre, P.; Bunnett, N.W. The G Protein–Coupled Receptor–Transient Receptor Potential Channel Axis: Molecular Insights for Targeting Disorders of Sensation and Inflammation. Pharmacol. Rev. 2015, 67, 36–73. [Google Scholar] [CrossRef]
- Billing, L.J.; Larraufie, P.; Lewis, J.; Leiter, A.; Li, J.; Lam, B.; Yeo, G.S.; Goldspink, D.A.; Kay, R.G.; Gribble, F.M.; et al. Single cell transcriptomic profiling of large intestinal enteroendocrine cells in mice—Identification of selective stimuli for insulin-like peptide-5 and glucagon-like peptide-1 co-expressing cells. Mol. Metab. 2019, 29, 158–169. [Google Scholar] [CrossRef]
- Gehart, H.; van Es, J.H.; Hamer, K.; Beumer, J.; Kretzschmar, K.; Dekkers, J.F.; Rios, A.; Clevers, H. Identification of Enteroendocrine Regulators by Real-Time Single-Cell Differentiation Mapping. Cell 2019, 176, 1158–1173.e16. [Google Scholar] [CrossRef]
- Ye, L.; Bae, M.; Cassilly, C.D.; Jabba, S.V.; Thorpe, D.W.; Martin, A.M.; Lu, H.-Y.; Wang, J.; Thompson, J.D.; Lickwar, C.R.; et al. Enteroendocrine cells sense bacterial tryptophan catabolites to activate enteric and vagal neuronal pathways. Cell Host Microbe 2021, 29, 179–196.e9. [Google Scholar] [CrossRef] [PubMed]
- Prescott, S.L.; Liberles, S.D. Internal senses of the vagus nerve. Neuron 2022, 110, 579–599. [Google Scholar] [CrossRef] [PubMed]
- Steinert, R.E.; Feinle-Bisset, C.; Asarian, L.; Horowitz, M.; Beglinger, C.; Geary, N. Ghrelin, CCK, GLP-1, and PYY(3-36): Secretory Controls and Physiological Roles in Eating and Glycemia in Health, Obesity, and After RYGB. Physiol. Rev. 2017, 97, 411–463. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Simasko, S.M. Role of Transient Receptor Potential Channels in Cholecystokinin-Induced Activation of Cultured Vagal Afferent Neurons. Endocrinology 2010, 151, 5237–5246. [Google Scholar] [CrossRef] [PubMed]
- Bohórquez, D.V.; Chandra, R.; Samsa, L.; Vigna, S.R.; Liddle, R.A. Characterization of basal pseudopod-like processes in ileal and colonic PYY cells. J. Mol. Histol. 2011, 42, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Bohórquez, D.V.; Shahid, R.A.; Erdmann, A.; Kreger, A.M.; Wang, Y.; Calakos, N.; Wang, F.; Liddle, R.A. Neuroepithelial circuit formed by innervation of sensory enteroendocrine cells. J. Clin. Investig. 2015, 125, 782–786. [Google Scholar] [CrossRef] [PubMed]
- Kaelberer, M.M.; Buchanan, K.L.; Klein, M.E.; Barth, B.B.; Montoya, M.M.; Shen, X.; Bohórquez, D.V. A gut-brain neural circuit for nutrient sensory transduction. Science 2018, 361, eaat5236. [Google Scholar] [CrossRef]
- Field, M. Intestinal ion transport and the pathophysiology of diarrhea. J. Clin. Investig. 2003, 111, 931–943. [Google Scholar] [CrossRef]
- Jensen, R.T. Overview of chronic diarrhea caused by functional neuroendocrine neoplasms. Semin. Gastrointest. Dis. 1999, 10, 156–172. [Google Scholar]
- Gupta, K.; Ng, I.C.; Balachander, G.M.; Nguyen, B.P.; Tucker-Kellogg, L.; Low, B.C.; Yu, H. Bile canaliculi contract autonomously by releasing calcium into hepatocytes via mechanosensitive calcium channel. Biomaterials 2020, 259, 120283. [Google Scholar] [CrossRef]
- Thiagarajah, J.R.; Donowitz, M.; Verkman, A.S. Secretory diarrhoea: Mechanisms and emerging therapies. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 446–457. [Google Scholar] [CrossRef]
- Manneck, D.; Manz, G.; Braun, H.S.; Rosendahl, J.; Stumpff, F. The TRPA1 Agonist Cinnamaldehyde Induces the Secretion of HCO3- by the Porcine Colon. Int. J. Mol. Sci. 2021, 22, 5198. [Google Scholar] [CrossRef]
- Brown, P.M.; Drossman, D.A.; Wood, A.J.; Cline, G.A.; Frazier, K.S.; Jackson, J.I.; Bronner, J.; Freiman, J.; Zambrowicz, B.; Sands, A.; et al. The Tryptophan Hydroxylase Inhibitor LX1031 Shows Clinical Benefit in Patients with Nonconstipating Irritable Bowel Syndrome. Gastroenterology 2011, 141, 507–516. [Google Scholar] [CrossRef]
- Zhang, T.; Perkins, M.H.; Chang, H.; Han, W.; de Araujo, I.E. An inter-organ neural circuit for appetite suppression. Cell 2022, 185, 2478–2494.e28. [Google Scholar] [CrossRef]
- De Araujo, I.E.; Schatzker, M.; Small, D.M. Rethinking Food Reward. Annu. Rev. Psychol. 2020, 71, 139–164. [Google Scholar] [CrossRef]
- Powley, T.L.; Phillips, R.J. Gastric satiation is volumetric, intestinal satiation is nutritive. Physiol. Behav. 2004, 82, 69–74. [Google Scholar] [CrossRef]
- Augustine, V.; Lee, S.; Oka, Y. Neural Control and Modulation of Thirst, Sodium Appetite, and Hunger. Cell 2020, 180, 25–32. [Google Scholar] [CrossRef]
- Zhao, Q.; Yu, C.D.; Wang, R.; Xu, Q.J.; Pra, R.D.; Le Zhang, L.; Chang, R.B. A multidimensional coding architecture of the vagal interoceptive system. Nature 2022, 603, 878–884. [Google Scholar] [CrossRef]
- Wang, Y.; Sims, C.E.; Allbritton, N.L. Enterochromaffin Cell-Enriched Monolayer Platform for Assaying Serotonin Release from Human Primary Intestinal Cells. Anal. Chem. 2020, 92, 12330–12337. [Google Scholar] [CrossRef]
- Min, S.; Oh, Y.; Verma, P.; Whitehead, S.C.; Yapici, N.; Van Vactor, D.; Suh, G.S.; Liberles, S. Control of feeding by Piezo-mediated gut mechanosensation in Drosophila. eLife 2021, 10, e63049. [Google Scholar] [CrossRef]
- Hughes, K.; Shah, A.; Bai, X.; Adams, J.; Bauer, R.; Jackson, J.; Harris, E.; Ficca, A.; Freebairn, P.; Mohammed, S.; et al. Distinct mechanoreceptor pezo-1 isoforms modulate food intake in the nematode Caenorhabditis elegans. G3 GenesGenomGenet. 2022, 12, jkab429. [Google Scholar] [CrossRef]
- Oh, Y.; Lai, J.S.-Y.; Min, S.; Huang, H.-W.; Liberles, S.D.; Ryoo, H.D.; Suh, G.S. Periphery signals generated by Piezo-mediated stomach stretch and Neuromedin-mediated glucose load regulate the Drosophila brain nutrient sensor. Neuron 2021, 109, 1979–1995.e6. [Google Scholar] [CrossRef]
- Williams, E.K.; Chang, R.B.; Strochlic, D.E.; Umans, B.D.; Lowell, B.B.; Liberles, S.D. Sensory Neurons that Detect Stretch and Nutrients in the Digestive System. Cell 2016, 166, 209–221. [Google Scholar] [CrossRef]
- Bai, L.; Mesgarzadeh, S.; Ramesh, K.; Huey, E.L.; Liu, Y.; Gray, L.A.; Aitken, T.J.; Chen, Y.; Beutler, L.; Ahn, J.S.; et al. Genetic Identification of Vagal Sensory Neurons That Control Feeding. Cell 2019, 179, 1129–1143.e23. [Google Scholar] [CrossRef]
- Page, A.J.; Brierley, S.M.; Martin, C.M.; Price, M.P.; Symonds, E.; Butler, R.; Wemmie, J.A.; Blackshaw, L.A. Different contributions of ASIC channels 1a, 2, and 3 in gastrointestinal mechanosensory function. Gut 2005, 54, 1408–1415. [Google Scholar] [CrossRef]
- Rhoades, J.; Nelson, J.C.; Nwabudike, I.; Yu, S.K.; McLachlan, I.G.; Madan, G.K.; Abebe, E.; Powers, J.R.; Colón-Ramos, D.A.; Flavell, S.W. ASICs Mediate Food Responses in an Enteric Serotonergic Neuron that Controls Foraging Behaviors. Cell 2019, 176, 85–97.e14. [Google Scholar] [CrossRef]
- Choi, M.-J.; Jin, Z.; Park, Y.S.; Rhee, Y.K.; Jin, Y.-H. Transient receptor potential (TRP) A1 activated currents in TRPV1 and cholecystokinin-sensitive cranial visceral afferent neurons. Brain Res. 2011, 1383, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Navarrete, J.; Liang-Guallpa, J.; Lu, C.; Funderburk, S.C.; Chang, R.B.; Liberles, S.D.; Olson, D.P.; Krashes, M.J. Defined Paraventricular Hypothalamic Populations Exhibit Differential Responses to Food Contingent on Caloric State. Cell Metab. 2019, 29, 681–694.e5. [Google Scholar] [CrossRef] [PubMed]
- Dockray, G.J. Enteroendocrine cell signalling via the vagus nerve. Curr. Opin. Pharmacol. 2013, 13, 954–958. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Zhou, H.-R.; Pestka, J.J. Potential roles for calcium-sensing receptor (CaSR) and transient receptor potential ankyrin-1 (TRPA1) in murine anorectic response to deoxynivalenol (vomitoxin). Arch. Toxicol. 2017, 91, 495–507. [Google Scholar] [CrossRef] [PubMed]
- Kentish, S.J.; Frisby, C.L.; Kritas, S.; Li, H.; Hatzinikolas, G.; O’Donnell, T.A.; Wittert, G.; Page, A.J. TRPV1 Channels and Gastric Vagal Afferent Signalling in Lean and High Fat Diet Induced Obese Mice. PLoS ONE 2015, 10, e0135892. [Google Scholar] [CrossRef]
- Hibberd, T.; Feng, J.; Luo, J.; Yang, P.; Samineni, V.; Gereau, R.W.; Kelley, N.; Hu, H.; Spencer, N.J. Optogenetic Induction of Colonic Motility in Mice. Gastroenterology 2018, 155, 514–528.e6. [Google Scholar] [CrossRef]
- Spencer, N.J.; Travis, L.; Hibberd, T.; Kelly, N.; Feng, J.; Hu, H. Effects of optogenetic activation of the enteric nervous system on gastrointestinal motility in mouse small intestine. Auton. Neurosci. 2020, 229, 102733. [Google Scholar] [CrossRef]
- Spencer, N.J.; Smith, T.K. Mechanosensory S-neurons rather than AH-neurons appear to generate a rhythmic motor pattern in guinea-pig distal colon. J. Physiol. 2004, 558, 577–596. [Google Scholar] [CrossRef]
- Johnson, J.A.G.; Liu, H.; Höger, U.; Rogers, S.M.; Sivapalan, K.; French, A.S.; Torkkeli, P.H. Mechanotransduction channel Piezo is widely expressed in the spider, Cupiennius salei, mechanosensory neurons and central nervous system. Sci. Rep. 2021, 11, 7994. [Google Scholar] [CrossRef]
- Morarach, K.; Mikhailova, A.; Knoflach, V.; Memic, F.; Kumar, R.; Li, W.; Ernfors, P.; Marklund, U. Diversification of molecularly defined myenteric neuron classes revealed by single-cell RNA sequencing. Nat. Neurosci. 2021, 24, 34–46. [Google Scholar] [CrossRef]
- Treichel, A.J.; Finholm, I.; Knutson, K.R.; Alcaino, C.; Whiteman, S.T.; Brown, M.R.; Matveyenko, A.; Wegner, A.; Kacmaz, H.; Mercado-Perez, A.; et al. Specialized Mechanosensory Epithelial Cells in Mouse Gut Intrinsic Tactile Sensitivity. Gastroenterology 2022, 162, 535–547.e13. [Google Scholar] [CrossRef]
- Jones, L.A.; Jin, B.; Martin, A.M.; Wei, L.; Ro, S.; Keating, D.J. Diminished Piezo2-Dependent Tactile Sensitivity Occurs in Aging Human Gut and Slows Gastrointestinal Transit in Mice. Gastroenterology 2022, 162, 1755–1757.e2. [Google Scholar] [CrossRef]
- Joshi, V.; Strege, P.R.; Farrugia, G.; Beyder, A. Mechanotransduction in gastrointestinal smooth muscle cells: Role of mechanosensitive ion channels. Am. J. Physiol. Liver Physiol. 2021, 320, G897–G906. [Google Scholar] [CrossRef]
- Vogt, M.; Schulz, B.; Wagdi, A.; Lebert, J.; van Belle, G.J.; Christoph, J.; Bruegmann, T.; Patejdl, R. Direct optogenetic stimulation of smooth muscle cells to control gastric contractility. Theranostics 2021, 11, 5569–5584. [Google Scholar] [CrossRef]
- O’Grady, G.; Angeli, T.; Du, P.; Cheng, L.K. Concerning the validity of gastrointestinal extracellular recordings. Physiol. Rev. 2015, 95, 691–692. [Google Scholar] [CrossRef]
- Sanders, K.M.; Ward, S.M.; Koh, S.D. Reply to O’Grady et al. Physiol. Rev. 2015, 95, 693–694. [Google Scholar] [CrossRef]
- Bulbring, E.; Lin, R. The action of 5-hydroxytryptamine (5-HT) on peristalsis. J. Physiol. 1957, 138, 12. [Google Scholar]
- Wei, L.; Singh, R.; Ha, S.E.; Martin, A.M.; Jones, L.A.; Jin, B.; Jorgensen, B.G.; Zogg, H.; Chervo, T.; Gottfried-Blackmore, A.; et al. Serotonin Deficiency Is Associated with Delayed Gastric Emptying. Gastroenterology 2021, 160, 2451–2466.e19. [Google Scholar] [CrossRef]
- Okaty, B.W.; Commons, K.G.; Dymecki, S.M. Embracing diversity in the 5-HT neuronal system. Nat. Rev. Neurosci. 2019, 20, 397–424. [Google Scholar] [CrossRef]
- Sugisawa, E.; Takayama, Y.; Takemura, N.; Kondo, T.; Hatakeyama, S.; Kumagai, Y.; Sunagawa, M.; Tominaga, M.; Maruyama, K. RNA Sensing by Gut Piezo1 Is Essential for Systemic Serotonin Synthesis. Cell 2020, 182, 609–624.e21. [Google Scholar] [CrossRef]
- Michel, K.; Zeller, F.; Langer, R.; Nekarda, H.; Kruger, D.; Dover, T.J.; Brady, C.A.; Barnes, N.M.; Schemann, M. Serotonin Excites Neurons in the Human Submucous Plexus via 5-HT3 Receptors. Gastroenterology 2005, 128, 1317–1326. [Google Scholar] [CrossRef]
- Hagbom, M.; Hellysaz, A.; Istrate, C.; Nordgren, J.; Sharma, S.; de-Faria, F.M.; Magnusson, K.E.; Svensson, L. The 5-HT(3) Receptor Affects Rotavirus-Induced Motility. J. Virol. 2021, 95, e0075121. [Google Scholar] [CrossRef]
- Hao, M.M.; Fung, C.; Boesmans, W.; Lowette, K.; Tack, J.; Berghe, P.V. Development of the intrinsic innervation of the small bowel mucosa and villi. Am. J. Physiol. Liver Physiol. 2020, 318, G53–G65. [Google Scholar] [CrossRef]
- Drokhlyansky, E.; Smillie, C.S.; Van Wittenberghe, N.; Ericsson, M.; Griffin, G.K.; Eraslan, G.; Dionne, D.; Cuoco, M.S.; Goder-Reiser, M.N.; Sharova, T.; et al. The Human and Mouse Enteric Nervous System at Single-Cell Resolution. Cell 2020, 182, 1606–1622.e23. [Google Scholar] [CrossRef] [PubMed]
- Poole, D.P.; Pelayo, J.C.; Cattaruzza, F.; Kuo, Y.; Gai, G.; Chiu, J.V.; Bron, R.; Furness, J.B.; Grady, E.F.; Bunnett, N.W. Transient Receptor Potential Ankyrin 1 Is Expressed by Inhibitory Motoneurons of the Mouse Intestine. Gastroenterology 2011, 141, 565–575.e4. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Wang, S.; Kobayashi, K.; Hao, Y.; Kanda, H.; Kondo, T.; Kogure, Y.; Yamanaka, H.; Yamamoto, S.; Li, J.; et al. TRPA1-expressing lamina propria mesenchymal cells regulate colonic motility. JCI Insight 2019, 4, e122402. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Yan, Z.; Li, B.; Jan, L.Y.; Jan, Y.N. Identification of motor neurons and a mechanosensitive sensory neuron in the defecation circuitry of Drosophila larvae. eLife 2014, 3, e03293. [Google Scholar] [CrossRef] [PubMed]
- Capasso, R.; Aviello, G.; Romano, B.; Borrelli, F.; De Petrocellis, L.; Di Marzo, V.; Izzo, A.A. Modulation of mouse gastrointestinal motility by allyl isothiocyanate, a constituent of cruciferous vegetables (Brassicaceae): Evidence for TRPA1-independent effects. Br. J. Pharmacol. 2012, 165, 1966–1977. [Google Scholar] [CrossRef]
- Mihara, H.; Suzuki, N.; Yamawaki, H.; Tominaga, M.; Sugiyama, T. TRPV2 ion channels expressed in inhibitory motor neurons of gastric myenteric plexus contribute to gastric adaptive relaxation and gastric emptying in mice. Am. J. Physiol. Liver Physiol. 2013, 304, G235–G240. [Google Scholar] [CrossRef]
- Mihara, H.; Boudaka, A.; Shibasaki, K.; Yamanaka, A.; Sugiyama, T.; Tominaga, M. Involvement of TRPV2 Activation in Intestinal Movement through Nitric Oxide Production in Mice. J. Neurosci. 2010, 30, 16536–16544. [Google Scholar] [CrossRef]
- Najjar, S.A.; Edwards, B.S.; Albers, K.M.; Davis, B.M.; Smith-Edwards, K.M. Optogenetic activation of the distal colon epithelium engages enteric nervous system circuits to initiate motility patterns. Am. J. Physiol. Liver Physiol. 2021, 321, G426–G435. [Google Scholar] [CrossRef]
- Kim, M.J.; Son, H.J.; Song, S.H.; Jung, M.; Kim, Y.; Rhyu, M.-R. The TRPA1 Agonist, Methyl Syringate Suppresses Food Intake and Gastric Emptying. PLoS ONE 2013, 8, e71603. [Google Scholar] [CrossRef]
- Someya, S.; Nagao, M.; Shibata, C.; Tanaka, N.; Sasaki, H.; Kikuchi, D.; Miyachi, T.; Naitoh, T.; Unno, M. Intracolonic Administration of the TRPA1 Agonist Allyl Isothiocyanate Stimulates Colonic Motility and Defecation in Conscious Dogs. J. Gastrointest. Surg. 2015, 19, 1342–1349. [Google Scholar] [CrossRef]
- Tsuchiya, K.; Kubota, K.; Ohbuchi, K.; Kaneko, A.; Ohno, N.; Mase, A.; Matsushima, H.; Yamamoto, M.; Miyano, K.; Uezono, Y.; et al. Transient receptor potential ankyrin 1 agonists improve intestinal transit in a murine model of postoperative ileus. Neurogastroenterol. Motil. 2016, 28, 1792–1805. [Google Scholar] [CrossRef]
- Koseki, J.; Oshima, T.; Kondo, T.; Tomita, T.; Fukui, H.; Watari, J.; Hattori, T.; Kase, Y.; Miwa, H. Role of Transient Receptor Potential Ankyrin 1 in Gastric Accommodation in Conscious Guinea Pigs. J. Pharmacol. Exp. Ther. 2012, 341, 205–212. [Google Scholar] [CrossRef]
- Yoshimura, M.; Mikami, T.; Kuroda, K.; Nishida, M.; Ito, K.; Mondal, A.; Koyama, K.; Jogahara, T.; Sakata, I.; Sakai, T. Involvement of Transient Receptor Potential Vanilloid Receptor 1, (TRPV1)-Expressing Vagal Nerve in the Inhibitory Effect of Gastric Acidification on Exogenous Motilin-Induced Gastric Phase III Contractions in Suncus murinus. Dig. Dis. Sci. 2016, 61, 1501–1511. [Google Scholar] [CrossRef]
- Rahmati, R. The transient receptor potential vanilloid receptor 1, TRPV1 (VR1) inhibits peristalsis in the mouse jejunum. Arch. Iran. Med. 2012, 15, 433–438. [Google Scholar]
- Matsumoto, K.; Kurosawa, E.; Terui, H.; Hosoya, T.; Tashima, K.; Murayama, T.; Priestley, J.V.; Horie, S. Localization of TRPV1 and contractile effect of capsaicin in mouse large intestine: High abundance and sensitivity in rectum and distal colon. Am. J. Physiol. Liver Physiol. 2009, 297, G348–G360. [Google Scholar] [CrossRef]
- De Winter, B.Y.; Bredenoord, A.J.; Van Nassauw, L.; De Man, J.G.; De Schepper, H.; Timmermans, J.-P.; Pelckmans, P.A. Involvement of afferent neurons in the pathogenesis of endotoxin-induced ileus in mice: Role of CGRP and TRPV1 receptors. Eur. J. Pharmacol. 2009, 615, 177–184. [Google Scholar] [CrossRef]
- Szymaszkiewicz, A.; Włodarczyk, J.; Wasilewski, A.; Di Marzo, V.; Storr, M.; Fichna, J.; Zielińska, M. Desensitization of transient receptor potential vanilloid type-1 (TRPV1) channel as promising therapy of irritable bowel syndrome: Characterization of the action of palvanil in the mouse gastrointestinal tract. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2020, 393, 1357–1364. [Google Scholar] [CrossRef]
- Luo, J.; Qian, A.; Oetjen, L.K.; Yu, W.; Yang, P.; Feng, J.; Xie, Z.; Liu, S.; Yin, S.; Dryn, D.; et al. TRPV4 Channel Signaling in Macrophages Promotes Gastrointestinal Motility via Direct Effects on Smooth Muscle Cells. Immunity 2018, 49, 107–119.e4. [Google Scholar] [CrossRef]
- Fichna, J.; Poole, D.P.; Veldhuis, N.; MacEachern, S.J.; Saur, D.; Zakrzewski, P.K.; Cygankiewicz, A.I.; Mokrowiecka, A.; Małecka-Panas, E.; Krajewska, W.M.; et al. Transient receptor potential vanilloid 4 inhibits mouse colonic motility by activating NO-dependent enteric neurotransmission. J. Mol. Med. 2015, 93, 1297–1309. [Google Scholar] [CrossRef]
- Mihara, H.; Suzuki, N.; Boudaka, A.; Muhammad, J.S.; Tominaga, M.; Tabuchi, Y.; Sugiyama, T. Transient receptor potential vanilloid 4-dependent calcium influx and ATP release in mouse and rat gastric epithelia. World J. Gastroenterol. 2016, 22, 5512–5519. [Google Scholar] [CrossRef]
- Rao, M. An increasingly complex view of intestinal motility. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 72–73. [Google Scholar] [CrossRef]
- Costa, M.; Spencer, N.J.; Brookes, S.J. The role of enteric inhibitory neurons in intestinal motility. Auton. Neurosci. 2021, 235, 102854. [Google Scholar] [CrossRef]
- Heitmann, P.T.; Vollebregt, P.F.; Knowles, C.H.; Lunniss, P.J.; Dinning, P.G.; Scott, S.M. Understanding the physiology of human defaecation and disorders of continence and evacuation. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 751–769. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, Z.; Wu, Y.; Liu, Z.; Li, Y.; Jiang, M. Role of Ion Channels in the Chemotransduction and Mechanotransduction in Digestive Function and Feeding Behavior. Int. J. Mol. Sci. 2022, 23, 9358. https://doi.org/10.3390/ijms23169358
Zhu Z, Wu Y, Liu Z, Li Y, Jiang M. Role of Ion Channels in the Chemotransduction and Mechanotransduction in Digestive Function and Feeding Behavior. International Journal of Molecular Sciences. 2022; 23(16):9358. https://doi.org/10.3390/ijms23169358
Chicago/Turabian StyleZhu, Zhenya, Yuhao Wu, Ziyu Liu, Yuezhou Li, and Mizu Jiang. 2022. "Role of Ion Channels in the Chemotransduction and Mechanotransduction in Digestive Function and Feeding Behavior" International Journal of Molecular Sciences 23, no. 16: 9358. https://doi.org/10.3390/ijms23169358





