Functional Interplay between Arginyl-tRNA Synthetases and Arginyltransferase
Abstract
:1. Introduction
2. Results
2.1. Intracellular Measurements of Arginylation Using an Arginylation Sensor Plasmid
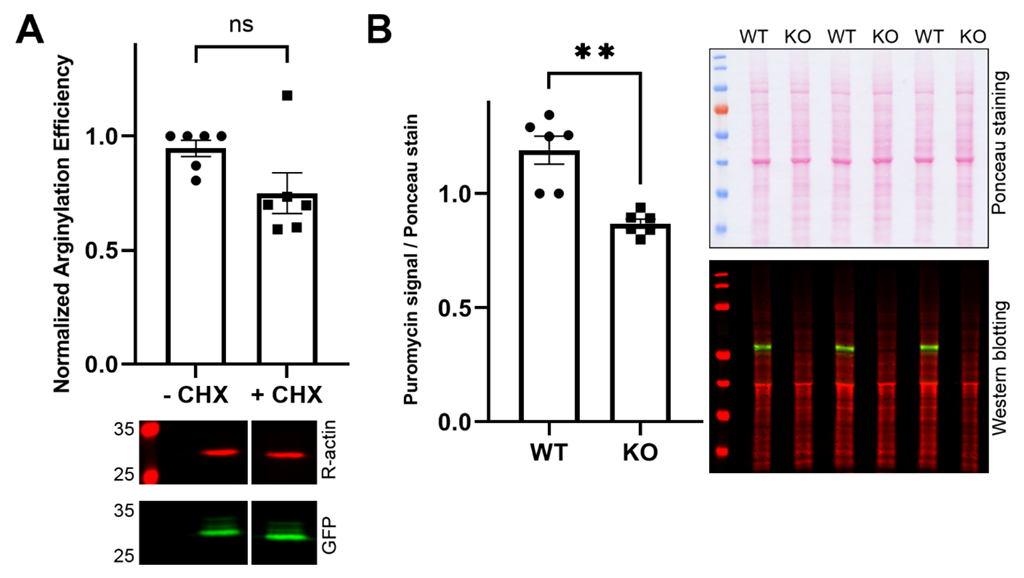
2.2. Intracellular Arginylation Depends on the Physiological State of the Cell but Does Not Compete with Active Translation
2.3. ATE1 Can Interact with the Long and Short RARS, but Overexpression of RARS Does Not Facilitate Arginylation
2.4. The ATE1–RARS Interaction Is tRNA-Independent
2.5. Displacement of the Long RARS from the MSC Increases Intracellular Arginylation
2.6. Displacement of RARS from the MSC Increases the Cytosolic Fraction of ATE1
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Total Protein Analysis and Cell Fractionation
4.3. Cell Treatments for Arginylation Sensor Measurements, Translation Inhibition and the Puromycylation Assay
4.4. Immunoprecipitation
4.5. Western Blotting
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Saha, S.; Kashina, A. Posttranslational arginylation as a global biological regulator. Dev. Biol. 2011, 358, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Han, X.; Leu, N.A.; Sterling, S.; Kurosaka, S.; Fina, M.; Lee, V.M.; Dong, D.W.; Yates, J.R.; Kashina, A. Protein arginylation targets alpha synuclein, facilitates normal brain health, and prevents neurodegeneration. Sci. Rep. 2017, 7, 11323. [Google Scholar] [CrossRef] [PubMed]
- Cornachione, A.S.; Leite, F.S.; Wang, J.; Leu, N.A.; Kalganov, A.; Volgin, D.; Han, X.; Xu, T.; Cheng, Y.S.; Yates, J.R., III; et al. Arginylation of myosin heavy chain regulates skeletal muscle strength. Cell Rep. 2014, 8, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Han, X.; Wong, C.C.; Cheng, H.; Aslanian, A.; Xu, T.; Leavis, P.; Roder, H.; Hedstrom, L.; Yates, J.R., III; et al. Arginyltransferase ATE1 catalyzes midchain arginylation of proteins at side chain carboxylates in vivo. Chem Biol. 2014, 21, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Saha, S.; Kashina, A. Arginylation-dependent regulation of a proteolytic product of talin is essential for cell-cell adhesion. J. Cell Biol. 2012, 197, 819–836. [Google Scholar] [CrossRef]
- Saha, S.; Wong, C.C.; Xu, T.; Namgoong, S.; Zebroski, H.; Yates, J.R., III; Kashina, A. Arginylation and methylation double up to regulate nuclear proteins and nuclear architecture in vivo. Chem. Biol. 2011, 18, 1369–1378. [Google Scholar] [CrossRef]
- Wong, C.C.L.; Xu, T.; Rai, R.; OBailey, A.; Yates, J.R.; Wolf, Y.; Zebroski, H.; Kashina, A. Global analysis of posttranslational protein arginylation. PLoS Biol. 2007, 5, e258. [Google Scholar] [CrossRef]
- Karakozova, M.; Kozak, M.; Wong, C.C.; Bailey, A.O.; Yates, J.R., III; Mogilner, A.; Zebroski, H.; Kashina, A. Arginylation of beta-actin regulates actin cytoskeleton and cell motility. Science 2006, 313, 192–196. [Google Scholar] [CrossRef]
- Zhang, F.; Patel, D.M.; Colavita, K.; Rodionova, I.; Buckley, B.; Scott, D.A.; Kumar, A.; Shabalina, S.A.; Saha, S.; Chernov, M.; et al. Arginylation regulates purine nucleotide biosynthesis by enhancing the activity of phosphoribosyl pyrophosphate synthase. Nat. Commun. 2015, 6, 7517. [Google Scholar] [CrossRef]
- Rai, R.; Zhang, F.; Colavita, K.; Leu, N.A.; Kurosaka, S.; Kumar, A.; Birnbaum, M.D.; Győrffy, B.; Dong, D.W.; Shtutman, M.; et al. Arginyltransferase suppresses cell tumorigenic potential and inversely correlates with metastases in human cancers. Oncogene 2016, 35, 4058–4068. [Google Scholar] [CrossRef] [Green Version]
- Rai, R.; Kashina, A. Identification of mammalian arginyltransferases that modify a specific subset of protein substrates. Proc. Natl. Acad. Sci. USA 2005, 102, 10123–10238. [Google Scholar] [CrossRef] [PubMed]
- Avcilar-Kucukgoze, I.; Gamper, H.; Polte, C.; Ignatova, Z.; Kraetzner, R.; Shtutman, M.; Hou, Y.M.; Dong, D.W.; Kashina, A. tRNA(Arg)-Derived Fragments Can Serve as Arginine Donors for Protein Arginylation. Cell Chem. Biol. 2020, 27, 839–849 e4. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.G.; Wei, H.; Ling, C.; Xu, M.G.; Wang, E.D. Two forms of human cytoplasmic arginyl-tRNA synthetase produced from two translation initiations by a single mRNA. Biochemistry 2006, 45, 1338–1344. [Google Scholar] [CrossRef] [PubMed]
- Sivaram, P.; Deutscher, M.P. Existence of two forms of rat liver arginyl-tRNA synthetase suggests channeling of aminoacyl-tRNA for protein synthesis. Proc. Natl. Acad. Sci. USA 1990, 87, 3665–3669. [Google Scholar] [CrossRef] [PubMed]
- Mirande, M. The Aminoacyl-tRNA Synthetase Complex. Subcell Biochem. 2017, 83, 505–522. [Google Scholar]
- Fu, Y.; Kim, Y.; Jin, K.S.; Kim, H.S.; Kim, J.H.; Wang, D.; Park, M.; Jo, C.H.; Kwon, N.H.; Kim, D.; et al. Structure of the ArgRS-GlnRS-AIMP1 complex and its implications for mammalian translation. Proc. Natl. Acad. Sci. USA 2014, 111, 15084–15089. [Google Scholar] [CrossRef]
- Cui, H.; Kapur, M.; Diedrich, J.K.; Yates, J.R., III; Ackerman, S.L.; Schimmel, P. Regulation of ex-translational activities is the primary function of the multi-tRNA synthetase complex. Nucleic Acids Res. 2021, 49, 3603–3616. [Google Scholar] [CrossRef]
- Nathanson, L.; Deutscher, M.P. Active aminoacyl-tRNA synthetases are present in nuclei as a high molecular weight multienzyme complex. J. Biol. Chem. 2000, 275, 31559–31562. [Google Scholar] [CrossRef]
- Bachmair, A.; Finley, D.; Varshavsky, A. In vivo half-life of a protein is a function of its amino-terminal residue. Science 1986, 234, 179–186. [Google Scholar] [CrossRef]
- Varshavsky, A. Ubiquitin fusion technique and its descendants. Methods Enzymol. 2000, 327, 578–593. [Google Scholar]
- Zhang, F.; Saha, S.; Shabalina, S.A.; Kashina, A. Differential arginylation of actin isoforms is regulated by coding sequence-dependent degradation. Science 2010, 329, 1534–1537. [Google Scholar] [CrossRef]
- Kumar, A.; Birnbaum, M.D.; Patel, D.M.; Morgan, W.M.; Singh, J.; Barrientos, A.; Zhang, F. Posttranslational arginylation enzyme Ate1 affects DNA mutagenesis by regulating stress response. Cell Death Dis. 2016, 7, e2378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, L.; Kashina, A. Quantification of intracellular N-terminal beta-actin arginylation. Sci. Rep. 2019, 9, 16669. [Google Scholar] [CrossRef] [PubMed]
- Bereiter-Hahn, J.; Munnich, A.; Woiteneck, P. Dependence of energy metabolism on the density of cells in culture. Cell Struct. Funct. 1998, 23, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Aviner, R. The science of puromycin: From studies of ribosome function to applications in biotechnology. Comput. Struct Biotechnol. J. 2020, 18, 1074–1083. [Google Scholar] [CrossRef]
- Nathans, D. Puromycin Inhibition of Protein Synthesis: Incorporation of Puromycin into Peptide Chains. Proc. Natl. Acad. Sci. USA 1964, 51, 585–592. [Google Scholar] [CrossRef]
- Yarmolinsky, M.B.; Haba, G.L. Inhibition by Puromycin of Amino Acid Incorporation into Protein. Proc. Natl. Acad. Sci. USA 1959, 45, 1721–1729. [Google Scholar] [CrossRef]
- Schmidt, E.K.; Clavarino, G.; Ceppi, M.; Pierre, P. SUnSET, a nonradioactive method to monitor protein synthesis. Nat. Methods. 2009, 6, 275–277. [Google Scholar] [CrossRef]
- Ciechanover, A.; Ferber, S.; Ganoth, D.; Elias, S.; Hershko, A.; Arfin, S. Purification and characterization of arginyl-tRNA-protein transferase from rabbit reticulocytes. Its involvement in post-translational modification and degradation of acidic NH2 termini substrates of the ubiquitin pathway. J. Biol. Chem. 1988, 263, 11155–11167. [Google Scholar] [CrossRef]
- Wang, J.; Han, X.; Saha, S.; Xu, T.; Rai, R.; Zhang, F.; Wolf, Y.I.; Wolfson, A.; Yates, J.R.; Kashina, A. Arginyltransferase is an ATP-independent self-regulating enzyme that forms distinct functional complexes in vivo. Chem. Biol. 2011, 18, 121–130. [Google Scholar] [CrossRef]
- Kwon, Y.T.; Kashina, A.S.; Varshavsky, A. Alternative splicing results in differential expression, activity, and localization of the two forms of arginyl-tRNA-protein transferase, a component of the N-end rule pathway. Mol. Cell Biol. 1999, 19, 182–193. [Google Scholar] [CrossRef] [PubMed]
- Fissolo, S.; Bongiovanni, G.; Decca, M.B.; Hallak, M.E. Post-translational arginylation of proteins in cultured cells. Neurochem. Res. 2000, 25, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Evans, M.; Clark, W.C.; Zheng, G.; Pan, T. Determination of tRNA aminoacylation levels by high-throughput sequencing. Nucleic Acids Res. 2017, 45, e133. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Moorthy, B.T.; Patel, D.M.; Kumar, A.; Morgan, W.M.; Alfonso, B.; Huang, J.; Lampidis, T.J.; Isom, D.G.; Barrientos, A.; et al. Regulation of Mitochondrial Respiratory Chain Complex Levels, Organization, and Function by Arginyltransferase 1. Front. Cell Dev. Biol. 2020, 8, 603688. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Kashina, A.S. Bacterial Expression and Purification of Recombinant Arginyltransferase (ATE1) and Arg-tRNA Synthetase (RRS) for Arginylation Assays. Methods Mol. Biol. 2015, 1337, 67–71. [Google Scholar]
- Avcilar-Kucukgoze, I.; Gamper, H.; Hou, Y.-M.; Kashina, A. Purification and Use of tRNA for Enzymatic Post-translational Addition of Amino Acids to Proteins. STAR Protoc. 2020, 1, 100207. [Google Scholar] [CrossRef]






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Avcilar-Kucukgoze, I.; MacTaggart, B.; Kashina, A. Functional Interplay between Arginyl-tRNA Synthetases and Arginyltransferase. Int. J. Mol. Sci. 2022, 23, 10160. https://doi.org/10.3390/ijms231710160
Avcilar-Kucukgoze I, MacTaggart B, Kashina A. Functional Interplay between Arginyl-tRNA Synthetases and Arginyltransferase. International Journal of Molecular Sciences. 2022; 23(17):10160. https://doi.org/10.3390/ijms231710160
Chicago/Turabian StyleAvcilar-Kucukgoze, Irem, Brittany MacTaggart, and Anna Kashina. 2022. "Functional Interplay between Arginyl-tRNA Synthetases and Arginyltransferase" International Journal of Molecular Sciences 23, no. 17: 10160. https://doi.org/10.3390/ijms231710160





