Lead Exposure Causes Spinal Curvature during Embryonic Development in Zebrafish
Abstract
:1. Introduction
2. Results
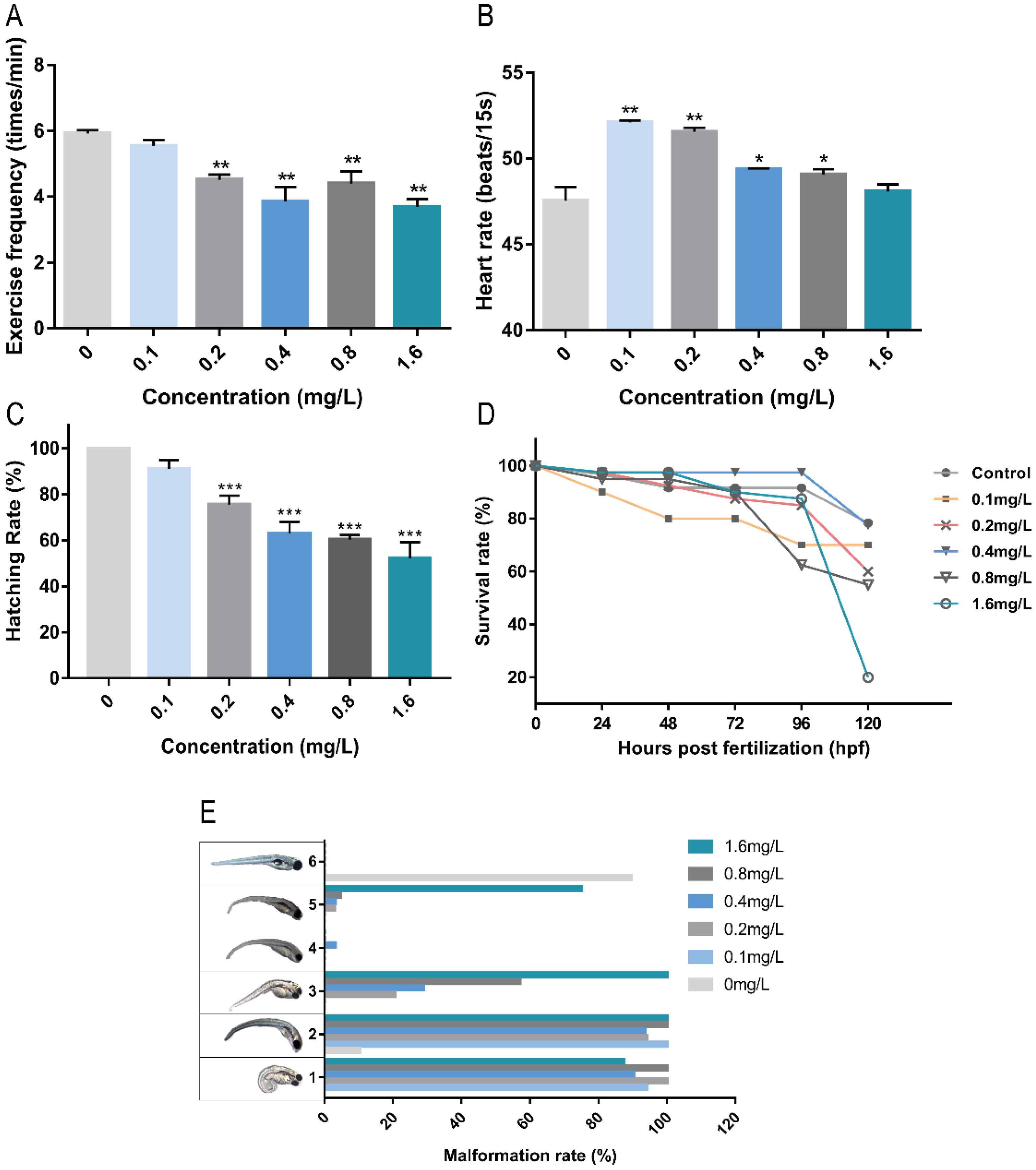
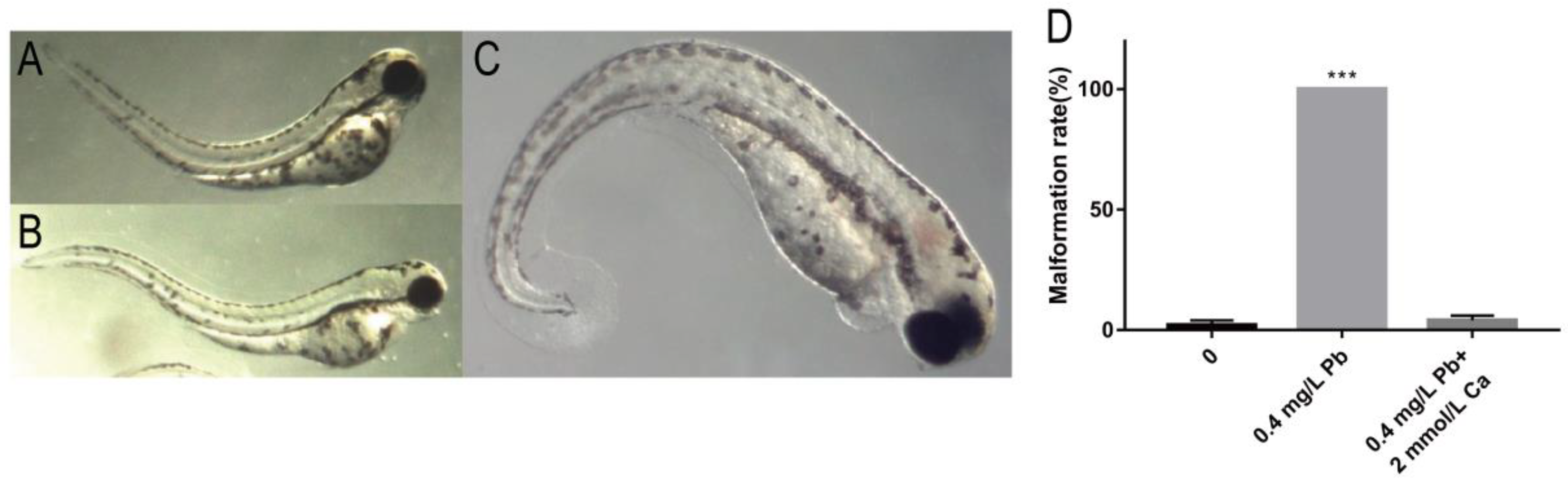
2.1. Toxicity of Pb2+ to Zebrafish Embryos
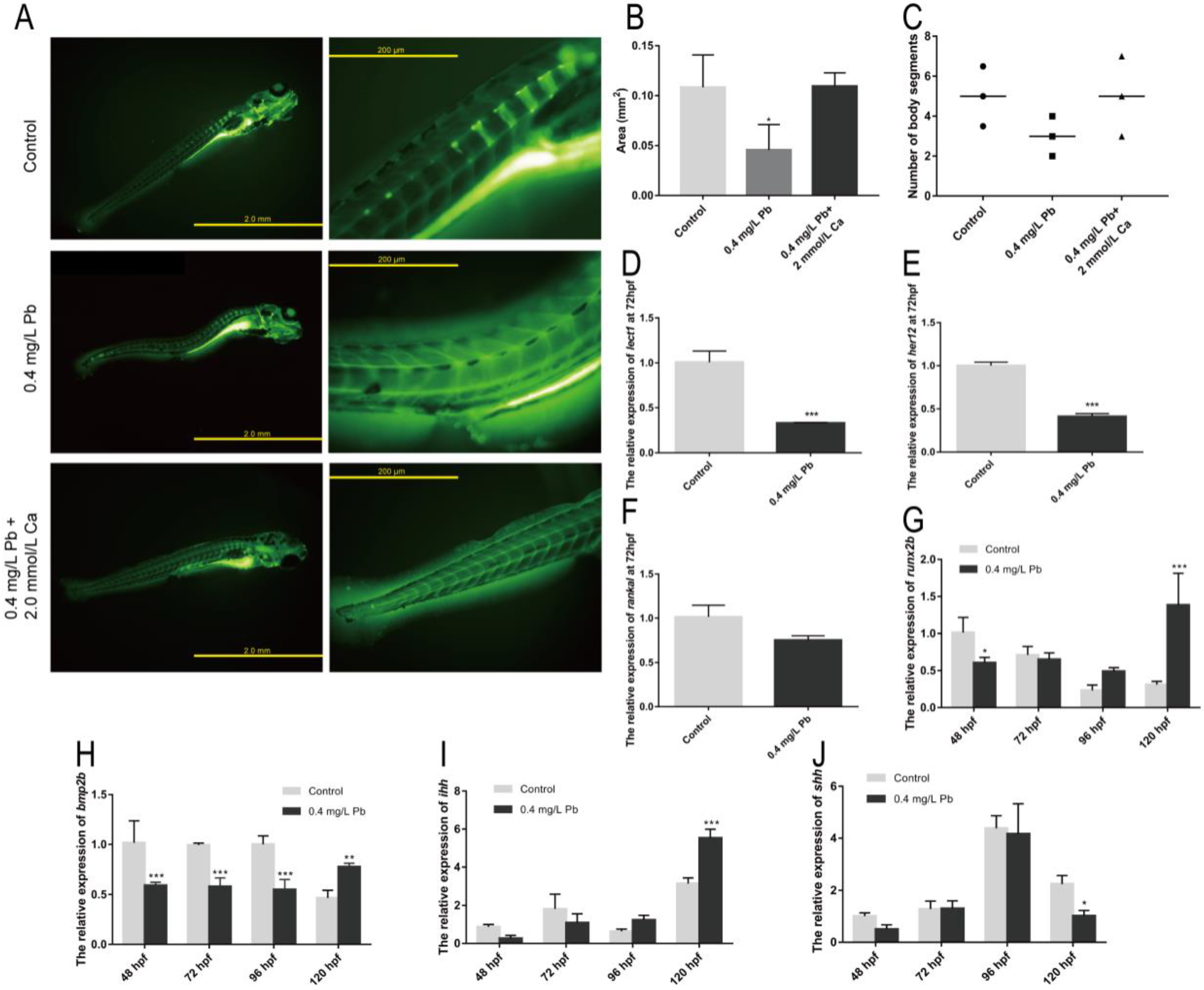
2.2. The Mechanism of Spinal Curvature
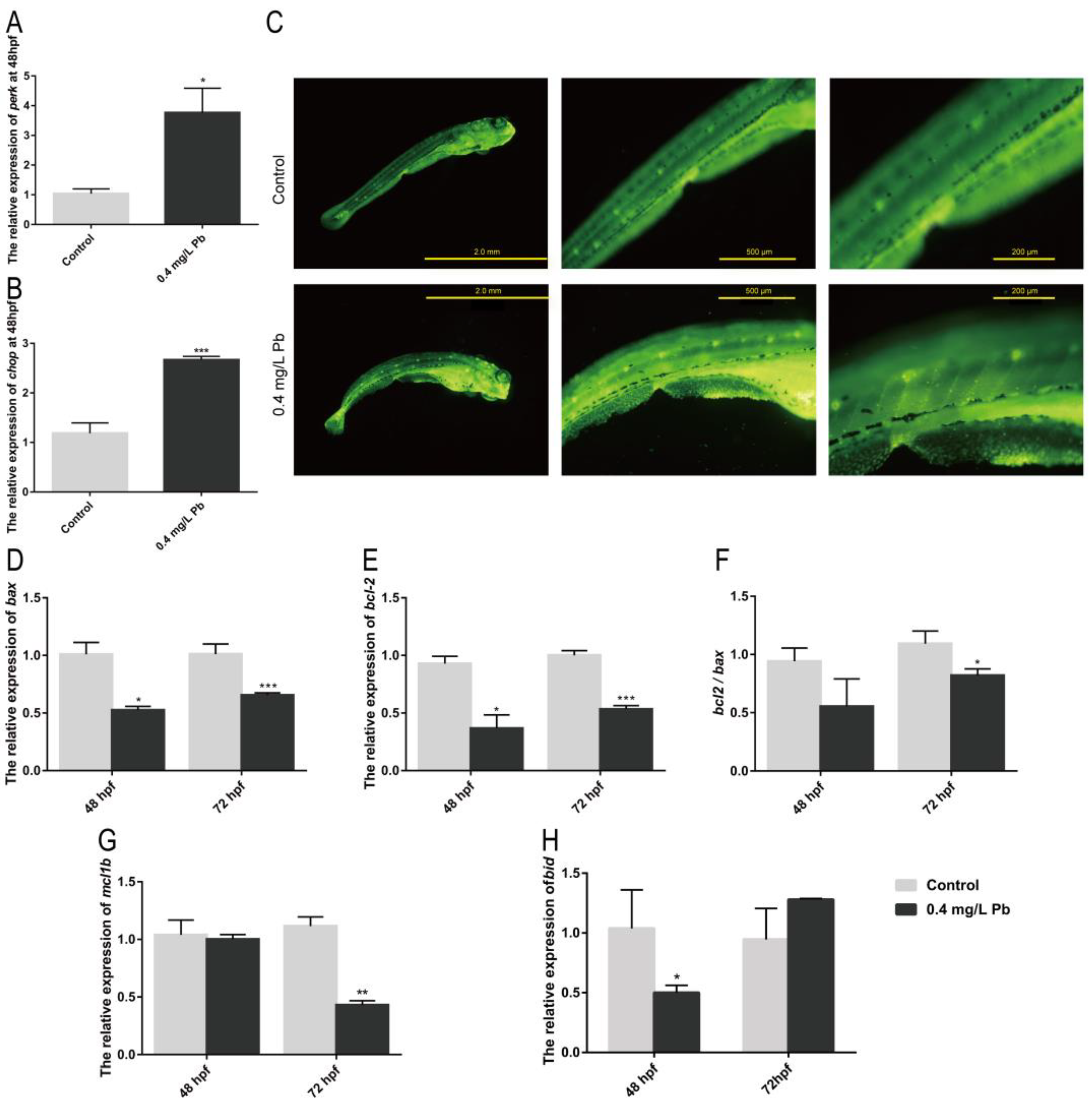
2.3. Pb2+ Induce Apoptosis by Activating Endoplasmic Reticulum (ER) Stress
3. Discussion
4. Materials and Methods
4.1. Materials and Reagents
4.2. Breeding and Spawning of Zebrafish
4.3. Zebrafish Pb2+ Exposure Experiment
4.4. Alizarin Red Staining
4.5. Calcein Staining
4.6. Acridine Orange Staining
4.7. Semi-Quantitative Polymerase Chain Reaction (PCR)
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Resongles, E.; Dietze, V.; Green, D.C.; Harrison, R.M.; Ochoa-Gonzalez, R.; Tremper, A.H.; Weiss, D.J. Strong evidence for the continued contribution of lead deposited during the 20th century to the atmospheric environment in London of today. Proc. Natl. Acad. Sci. USA 2021, 118, e2102791118. [Google Scholar] [CrossRef] [PubMed]
- Rossi, E. Low level environmental lead exposure–a continuing challenge. Clin. Biochem. Rev. 2008, 29, 63. [Google Scholar] [PubMed]
- Roy, N.M.; De Wolf, S.; Carneiro, B. Evaluation of the developmental toxicity of lead in the Danio rerio body. Aquat. Toxicol. 2015, 158, 138–148. [Google Scholar] [CrossRef]
- Dunham, J. Encyclopedia of Environmental Health. Libr. J. 2011, 136, 104. [Google Scholar]
- Kolstad, K.; Thorland, I.; Refstie, T.; Gjerde, B. Genetic variation and genotype by location interaction in body weight, spinal deformity and sexual maturity in Atlantic cod (Gadus morhua) reared at different locations off Norway. Aquaculture 2006, 259, 66–73. [Google Scholar] [CrossRef]
- Witeska, M.; Jezierska, B. The effects of environmental factors on metal toxicity to fish (review). Fresen. Environ. Bull 2003, 12, 824–829. [Google Scholar]
- Hill, A.J.; Teraoka, H.; Heideman, W.; Peterson, R.E. Zebrafish as a model vertebrate for investigating chemical toxicity. Toxicol. Sci. 2005, 86, 6–19. [Google Scholar] [CrossRef] [Green Version]
- Dai, Y.J.; Jia, Y.F.; Chen, N.; Bian, W.P.; Li, Q.K.; Ma, Y.B.; Chen, Y.L.; Pei, D.S. Zebrafish as a Model System to Study Toxicology. Env. Toxicol. Chem. 2014, 33, 11–17. [Google Scholar] [CrossRef]
- Dou, C.M.; Zhang, J. Effects of lead on neurogenesis during zebrafish embryonic brain development. J. Hazard Mater. 2011, 194, 277–282. [Google Scholar] [CrossRef]
- Kimmel, C.B.; Ballard, W.W.; Kimmel, S.R.; Ullmann, B.; Schilling, T.F. Stages of Embryonic-Development of the Zebrafish. Dev. Dynam. 1995, 203, 253–310. [Google Scholar] [CrossRef]
- Cao, X.D.; Ma, L.Q.; Chen, M.; Singh, S.P.; Harris, W.G. Impacts of phosphate amendments on lead biogeochemistry at a contaminated site. Environ. Sci. Technol. 2002, 36, 5296–5304. [Google Scholar] [CrossRef] [PubMed]
- Mundlos, S.; Olsen, B.R. Heritable diseases of the skeleton. Part I: Molecular insights into skeletal development-transcription factors and signaling pathways. FASEB J. 1997, 11, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Gan, S.; Huang, Z.; Liu, N.; Su, R.; Xie, G.; Zhong, B.; Zhang, K.; Wang, S.; Hu, X.; Zhang, J.; et al. MicroRNA-140-5p impairs zebrafish embryonic bone development via targeting BMP-2. FEBS Lett. 2016, 590, 1438–1446. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van der Meulen, T.; Kranenbarg, S.; Schipper, H.; Samallo, J.; van Leeuwen, J.L.; Franssen, H. Identification and characterisation of two runx2 homologues in zebrafish with different expression patterns. Biochim. Biophys. Acta 2005, 1729, 105–117. [Google Scholar] [CrossRef] [PubMed]
- Winata, C.L.; Korzh, S.; Kondrychyn, I.; Zheng, W.L.; Korzh, V.; Gong, Z.Y. Development of zebrafish swimbladder: The requirement of Hedgehog signaling in specification and organization of the three tissue layers. Dev. Biol. 2009, 331, 222–236. [Google Scholar] [CrossRef] [PubMed]
- Braunstein, J.A.; Robbins, A.E.; Stewart, S.; Stankunas, K. Basal epidermis collective migration and local Sonic hedgehog signaling promote skeletal branching morphogenesis in zebrafish fins. Dev. Biol. 2021, 477, 177–190. [Google Scholar] [CrossRef] [PubMed]
- Musatov, A.; Robinson, N.C. Susceptibility of mitochondrial electron-transport complexes to oxidative damage. Focus on cytochrome c oxidase. Free Radic. Res. 2012, 46, 1313–1326. [Google Scholar] [CrossRef]
- Huang, R.; Hui, Z.; Wei, S.; Li, D.; Li, W.; Daping, W.; Alahdal, M. IRE1 signaling regulates chondrocyte apoptosis and death fate in the osteoarthritis. J. Cell Physiol. 2022, 237, 118–127. [Google Scholar] [CrossRef]
- Schonthal, A.H. Endoplasmic reticulum stress: Its role in disease and novel prospects for therapy. Science 2012, 2012, 857516. [Google Scholar] [CrossRef] [Green Version]
- Minina, E.; Wenzel, H.; Kreschel, C.; Karp, S.; Gaffield, W.; McMahon, A.; Vortkamp, A. BMP and Ihh/PTHrP signaling interact to coordinate chondrocyte proliferation and differentiation. Development 2001, 128, 4523–4534. [Google Scholar] [CrossRef]
- Reichert, J.; Schmalzl, J.; Prager, P.; Gilbert, F.; Quent, V.; Steinert, A.; Rudert, M.; Nöth, U. Synergistic effect of Indian hedgehog and bone morphogenetic protein-2 gene transfer to increase the osteogenic potential of human mesenchymal stem cells. Stem Cell Res. Ther. 2013, 4, 105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Polak-Juszczak, L. Impact of strontium on skeletal deformities in Baltic cod (Gadus morhua callaris L.). Chemosphere 2011, 83, 486–491. [Google Scholar] [CrossRef] [PubMed]
| Malformation | 72 hpf | 96 hpf | 120 hpf |
|---|---|---|---|
| upward curvature | 52.0 ± 1.5% | 0% | 0% |
| S-shaped curvature | 0% | 85.0 ± 2.5% | 0% |
| downward curvature | 0% | 0% | 100% |
| Total | 52% | 85% | 100% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, X.; Chen, C.; He, M.; Yu, L.; Liu, R.; Ma, C.; Zhang, Y.; Jia, J.; Li, B.; Li, L. Lead Exposure Causes Spinal Curvature during Embryonic Development in Zebrafish. Int. J. Mol. Sci. 2022, 23, 9571. https://doi.org/10.3390/ijms23179571
Li X, Chen C, He M, Yu L, Liu R, Ma C, Zhang Y, Jia J, Li B, Li L. Lead Exposure Causes Spinal Curvature during Embryonic Development in Zebrafish. International Journal of Molecular Sciences. 2022; 23(17):9571. https://doi.org/10.3390/ijms23179571
Chicago/Turabian StyleLi, Xueting, Ce Chen, Mingyue He, Lidong Yu, Renhao Liu, Chunmeng Ma, Yu Zhang, Jianbo Jia, Bingsheng Li, and Li Li. 2022. "Lead Exposure Causes Spinal Curvature during Embryonic Development in Zebrafish" International Journal of Molecular Sciences 23, no. 17: 9571. https://doi.org/10.3390/ijms23179571
APA StyleLi, X., Chen, C., He, M., Yu, L., Liu, R., Ma, C., Zhang, Y., Jia, J., Li, B., & Li, L. (2022). Lead Exposure Causes Spinal Curvature during Embryonic Development in Zebrafish. International Journal of Molecular Sciences, 23(17), 9571. https://doi.org/10.3390/ijms23179571






