Genome-Wide Identification of Laminin Family Related to Follicular Pseudoplacenta Development in Black Rockfish (Sebastes schlegelii)
Abstract
:1. Introduction
2. Results
2.1. Blackrock Fish Laminin Chains
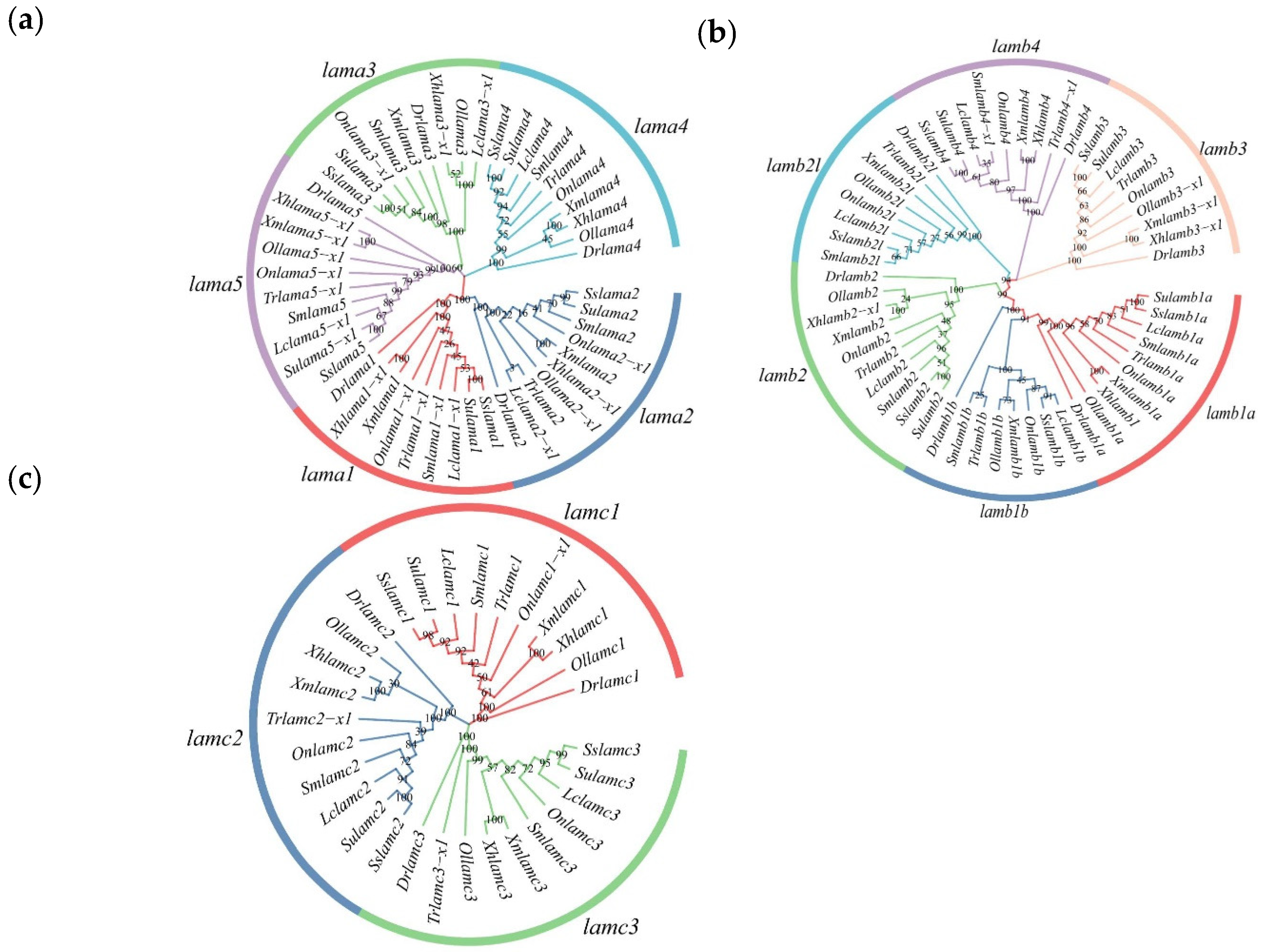
2.2. Phylogenic Analysis of Laminin Chains
2.3. Analysis of Gene Location in S. schlegelii and Other Teleosts
2.4. Gene Structure and Conserved Domain
2.5. Laminin Gene Expression Analysis at Different Ovarian Stages
2.6. The Location of Lama4 in Reproductive Cycle of S. schlegelii
3. Discussion
4. Materials and Methods
4.1. Identification of Laminin Genes
4.2. Gene Locations, Conserved Domains, Gene Structure, and Physicochemical Properties
4.3. Phylogenetic Analysis of Laminin Genes
4.4. RNA Extraction and qPCR Analysis of Gene Expression
4.5. In Situ Hybridization
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wourms, J.P. Viviparity: The Maternal-Fetal Relationship in Fishes. Am. Zool. 1981, 21, 473–515. [Google Scholar] [CrossRef]
- Blackburn, D.G. Evolution of vertebrate viviparity and specializations for fetal nutrition: A quantitative and qualitative analysis. J. Morphol. 2015, 276, 961–990. [Google Scholar] [CrossRef] [PubMed]
- Turner, C.L. Pseudoamnion, pseudochorion, and follicular pseudoplacenta in poeciliid fishes. J. Morphol. 1940, 67, 59–89. [Google Scholar] [CrossRef]
- Grove, B.D.; Wourms, J.P. Follicular Placenta of the Viviparous Fish, Heterandria formosa: II. Ultrastructure and Development of the Follicular Epithelium. J. Morphol. 1994, 220, 167–184. [Google Scholar] [CrossRef]
- Guernsey, M.W.; van Kruistum, H.; Reznick, D.N.; Pollux, B.J.A.; Baker, J.C. Molecular Signatures of Placentation and Secretion Uncovered in Poeciliopsis Maternal Follicles. Mol. Biol. Evol. 2020, 37, 2679–2690. [Google Scholar] [CrossRef]
- Uribe, M.C.; Grier, H.J.; Avila-Zuniga, S.A.; Garcia-Alarcon, A. Change of lecithotrophic to matrotrophic nutrition during gestation in the viviparous teleost Xenotoca eiseni (Goodeidae). J. Morphol. 2018, 279, 1336–1345. [Google Scholar] [CrossRef] [PubMed]
- Kawaguchi, M.; Okubo, R.; Harada, A.; Miyasaka, K.; Takada, K.; Hiroi, J.; Yasumasu, S. Morphology of brood pouch formation in the pot-bellied seahorse Hippocampus abdominalis. Zool. Lett. 2017, 3, 19. [Google Scholar] [CrossRef]
- Sasaki, M.; Kleinman, H.K.; Huber, H.; Deutzmann, R.; Yamada, Y. Laminin, a multidomain protein. The A chain has a unique globular domain and homology with the basement membrane proteoglycan and the laminin B chains. J. Biol. Chem. 1988, 263, 16536–16544. [Google Scholar] [CrossRef]
- Timpl, R.; Rohde, H.; Robey, P.G.; Rennard, S.I.; Foidart, J.M.; Martin, G.R. Laminin–a glycoprotein from basement membranes. J. Biol. Chem. 1979, 254, 9933–9937. [Google Scholar] [CrossRef]
- Chung, A.E.; Jaffe, R.; Freeman, I.L.; Vergnes, J.P.; Braginski, J.E.; Carlin, B. Properties of a basement membrane-related glycoprotein synthesized in culture by a mouse-embryonal carcinoma-derived cell-line. Cell 1979, 16, 277–287. [Google Scholar] [CrossRef]
- Malinda, K.M.; Kleinman, H.K. The laminins. Int. J. Biochem. Cell Biol. 1996, 28, 957–959. [Google Scholar] [CrossRef]
- Aumailley, M. The laminin family. Cell Adh. Migr. 2013, 7, 48–55. [Google Scholar] [CrossRef]
- Fahey, B.; Degnan, B.M. Origin and evolution of laminin gene family diversity. Mol. Biol. Evol. 2012, 29, 1823–1836. [Google Scholar] [CrossRef]
- Beck, K.; Hunter, I.; Engel, J. Structure and function of laminin-anatomy of a multidomain glycoprotein. FASEB J. 1990, 4, 148–160. [Google Scholar] [CrossRef] [PubMed]
- Hohenester, E.; Yurchenco, P.D. Laminins in basement membrane assembly. Cell Adh. Migr. 2013, 7, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Maltseva, D.V.; Rodin, S.A. Laminins in Metastatic Cancer. Mol. Biol. 2018, 52, 350–371. [Google Scholar] [CrossRef]
- Eble, J.A.; Niland, S. The Extracellular Matrix of Blood Vessels. Curr. Pharm. Des. 2009, 15, 1385–1400. [Google Scholar] [CrossRef]
- Spessotto, P.; Zucchetto, A.; Degan, M.; Wasserman, B.; Danussi, C.; Bomben, R.; Perris, R.; Canzonieri, V.; Radillo, O.; Colombatti, A.; et al. Laminin-332 (Laminin-5) is the major motility ligand for B cell chronic lymphocytic leukemia. Matrix Biol. 2007, 26, 473–484. [Google Scholar] [CrossRef]
- Wondimu, Z.; Geberhiwot, T.; Ingerpuu, S.; Juronen, E.; Xie, X.; Lindbom, L.; Doi, M.; Kortesmaa, J.; Thyboll, J.; Tryggvason, K.; et al. An endothelial laminin isoform, laminin 8 (alpha4beta1gamma1), is secreted by blood neutrophils, promotes neutrophil migration and extravasation, and protects neutrophils from apoptosis. Blood 2004, 104, 1859–1866. [Google Scholar] [CrossRef]
- Shibata, S.; Hayashi, R.; Okubo, T.; Kudo, Y.; Katayama, T.; Ishikawa, Y.; Toga, J.; Yagi, E.; Honma, Y.; Quantock, A.J.; et al. Selective Laminin-Directed Differentiation of Human Induced Pluripotent Stem Cells into Distinct Ocular Lineages. Cell Rep. 2018, 25, 1668–1679.e5. [Google Scholar] [CrossRef] [Green Version]
- Yap, L.; Tay, H.G.; Nguyen, M.T.X.; Tjin, M.S.; Tryggvason, K. Laminins in Cellular Differentiation. Trends Cell Biol. 2019, 29, 987–1000. [Google Scholar] [CrossRef] [PubMed]
- Rodin, S.; Antonsson, L.; Niaudet, C.; Simonson, O.E.; Salmela, E.; Hansson, E.M.; Domogatskaya, A.; Xiao, Z.; Damdimopoulou, P.; Sheikhi, M.; et al. Clonal culturing of human embryonic stem cells on laminin-521/E-cadherin matrix in defined and xeno-free environment. Nat. Commun. 2014, 5, 3195. [Google Scholar] [CrossRef] [PubMed]
- Iivanainen, A.; Sainio, K.; Sariola, H.; Tryggvason, K. Primary structure and expression of a novel human laminin a4 chain. FEBS Lett. 1995, 365, 183–188. [Google Scholar] [CrossRef]
- Richards, A.; Al-lmara, L.; Pope, F.M. The complete cDNA sequence of laminin a4 and its relationship to the other human laminin a chains. Eur. J. Biochem. 1996, 238, 813–821. [Google Scholar] [CrossRef]
- Liu, M. Expression level of LAMA4 protein in placental tissue of preeclampsia patients and its correlation with invasion of trophoblast cells and placental hypoxia. Matern. Child Health Care China 2017, 32, 2597–2599. [Google Scholar]
- Wang, P.; Nan, S.; Wang, Y.; Qi, H. Expression and function of LAMA4 in the placentas and trophoblast. J. Chongqing Med. Univ. 2016, 41, 694–698. [Google Scholar]
- Miner, J.H.; Cunningham, J.; Sanes, J.R. Roles for laminin in embryogenesis: Exencephaly, syndactyly, and placentopathy in mice lacking the laminin alpha 5 chain. J. Cell Biol. 1998, 143, 1713–1723. [Google Scholar] [CrossRef]
- Miner, J.H. Laminins and their roles in mammals. Microsc. Res. Tech. 2008, 71, 349–356. [Google Scholar] [CrossRef]
- Nakagawa, M.; Okouchi, H.; Adachi, J.; Hattori, K.; Yamashita, Y. Effectiveness of stock enhancement of hatchery-released black rockfish Sebastes schlegeli in Yamada Bay—Evaluation by a Fish Market survey. Aquaculture 2007, 263, 295–302. [Google Scholar] [CrossRef]
- Boehlert, G.W.; Kusakari, M.; Shimizu, M.; Yamada, J. Energetics during embryonic-development in Kurosoi, Sebastes schlegeli hilgendorf. J. Exp. Mar. Biol. Ecol. 1986, 101, 239–256. [Google Scholar] [CrossRef]
- Yamada, J.; Kusakari, M. Staging and the time course of embryonic-development in Kurosoi, Sebastes schlegeli. Environ. Biol. Fishes 1991, 30, 103–110. [Google Scholar] [CrossRef]
- Xu, X.; Wang, X.; Liu, Q.; Qi, X.; Zhou, L.; Liu, H.; Li, J. New insights on folliculogenesis and follicular placentation in marine viviparous fish black rockfish (Sebastes schlegelii). Gene 2022, 827, 146444. [Google Scholar] [CrossRef]
- Tengfei, D.; Yongshuang, X.; Haixia, Z.; Li, Z.; Qinghua, L.; Xueying, W.; Jun, L.; Shihong, X.; Yanfeng, W.; Jiachen, Y.; et al. Multiple Fetal Nutritional Patterns Before Parturition in Viviparous Fish Sebastes schlegelii (Hilgendorf, 1880). Front. Mar. Sci. 2021, 7, 571946. [Google Scholar] [CrossRef]
- Zinkevich, N.S.; Bosenko, D.V.; Link, B.A.; Semina, E.V. laminin alpha 1 gene is essential for normal lens development in zebrafish. BMC Dev. Biol. 2006, 6, 13. [Google Scholar] [CrossRef] [PubMed]
- Sittaramane, V.; Sawant, A.; Wolman, M.A.; Maves, L.; Halloran, M.C.; Chandrasekhar, A. The cell adhesion molecule Tag1, transmembrane protein Stbm/Vangl2, and Lamininalpha1 exhibit genetic interactions during migration of facial branchiomotor neurons in zebrafish. Dev. Biol. 2009, 325, 363–373. [Google Scholar] [CrossRef]
- Paulus, J.D.; Halloran, M.C. Zebrafish bashful/laminin-alpha 1 mutants exhibit multiple axon guidance defects. Dev. Dyn. 2006, 235, 213–224. [Google Scholar] [CrossRef]
- Parsons, M.J.; Pollard, S.M.; Saude, L.; Feldman, B.; Coutinho, P.; Hirst, E.M.A.; Stemple, D.L. Zebrafish mutants identify an essential role for laminins in notochord formation. Development 2002, 129, 3137–3146. [Google Scholar] [CrossRef]
- Domogatskaya, A.; Rodin, S.; Tryggvason, K. Functional diversity of laminins. Annu. Rev. Cell Dev. Biol. 2012, 28, 523–553. [Google Scholar] [CrossRef]
- Meyer, A.; Van de Peer, Y. From 2R to 3R: Evidence for a fish-specific genome duplication (FSGD). Bioessays 2005, 27, 937–945. [Google Scholar] [CrossRef]
- Nielsen, P.K.; Yamada, Y. Identification of cell-binding sites on the Laminin alpha 5 N-terminal domain by site-directed mutagenesis. J. Biol. Chem. 2001, 276, 10906–10912. [Google Scholar] [CrossRef]
- Sasaki, T.; Timpl, R. Domain IVa of laminin alpha 5 chain is cell-adhesive and binds beta 1 and alpha V beta 3 integrins through Arg-Gly-Asp. FEBS Lett. 2001, 509, 181–185. [Google Scholar] [CrossRef]
- Akkoyunlu, G.; Demir, R.; Ustunel, I. Distribution patterns of TGF-alpha, laminin and fibronectin and their relationship with folliculogenesis in rat ovary. Acta Histochem. 2003, 105, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Shan, N.; Zhang, X.; Xiao, X.; Zhang, H.; Tong, C.; Luo, X.; Chen, Y.; Liu, X.; Yin, N.; Deng, Q.; et al. Laminin alpha4 (LAMA4) expression promotes trophoblast cell invasion, migration, and angiogenesis, and is lowered in preeclamptic placentas. Placenta 2015, 36, 809–820. [Google Scholar] [CrossRef]
- Virolle, T.; Coraux, C.; Ferrigno, O.; Cailleteau, L.; Ortonne, J.P.; Pognonec, P.; Aberdam, D. Binding of USF to a non-canonical E-box following stress results in a cell-specific derepression of the lama3 gene. Nucleic Acids Res. 2002, 30, 1789–1798. [Google Scholar] [CrossRef] [PubMed]
- Miner, J.H.; Patton, B.L.; Lentz, S.I.; Gilbert, D.J.; Jenkins, N.A.; Copeland, N.G.; Sanes, J.R. The laminin alpha chains: Expression, developmental transitions, and chromosomal locations of alpha 1-5, identification of heterotrimeric laminins 8-11, and cloning of a novel alpha 3 isoform. J. Cell Biol. 1997, 137, 685–701. [Google Scholar] [CrossRef]
- Chen, C.; Chen, H.; Zhang, Y.; Thomas, H.R.; Frank, M.H.; He, Y.; Xia, R. TBtools: An Integrative Toolkit Developed for Interactive Analyses of Big Biological Data. Mol. Plant. 2020, 13, 1194–1202. [Google Scholar] [CrossRef]
- Nguyen, N.T.T.; Vincens, P.; Roest Crollius, H.; Louis, A. Genomicus 2018: Karyotype evolutionary trees and on-the-fly synteny computing. Nucleic Acids Res. 2018, 46, D816–D822. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, J.; Jiao, W.; Li, J.; Xun, X.; Sun, Y.; Guo, X.; Huan, P.; Dong, B.; Zhang, L.; et al. Scallop genome provides insights into evolution of bilaterian karyotype and development. Nat. Ecol. Evol. 2017, 1, 120. [Google Scholar] [CrossRef] [Green Version]






| Gene | Number of Amino Acids | Molecular Weight (kDa) | Theoretical pI | Subcellular Location |
|---|---|---|---|---|
| lama1 | 3087 | 333.5 | 5.73 | Membrane bound Extracellular (Secreted) |
| lama2 | 2306 | 251.46 | 6.38 | Membrane bound Extracellular (Secreted) |
| lama3 | 1544 | 171.9 | 7.15 | Plasma membrane |
| lama4 | 1804 | 200.16 | 5.6 | Plasma membrane |
| lama5 | 3666 | 401.73 | 5.64 | Membrane bound Extracellular (Secreted) |
| lamb1a | 1799 | 197.73 | 4.99 | Membrane bound Extracellular (Secreted) |
| lamb1b | 1800 | 197.81 | 4.94 | Membrane bound Extracellular (Secreted) |
| lamb2l | 1789 | 199.48 | 6.27 | Plasma membrane |
| lamb2 | 1685 | 184.97 | 6.31 | Membrane bound Extracellular (Secreted) |
| lamb3 | 1169 | 128.05 | 5.37 | Extracellular (Secreted) |
| lamb4 | 1803 | 199.87 | 5.34 | Membrane bound Extracellular (Secreted) |
| lamc1 | 1605 | 176.31 | 5.16 | Membrane bound Extracellular (Secreted) |
| lamc2 | 1125 | 121.23 | 5.95 | Membrane bound Extracellular (Secreted) |
| lamc3 | 1421 | 156.92 | 5.72 | Membrane bound Extracellular (Secreted) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, N.; Wang, X.; Wang, T.; Xu, X.; Liu, Q.; Li, J. Genome-Wide Identification of Laminin Family Related to Follicular Pseudoplacenta Development in Black Rockfish (Sebastes schlegelii). Int. J. Mol. Sci. 2022, 23, 10523. https://doi.org/10.3390/ijms231810523
Zhao N, Wang X, Wang T, Xu X, Liu Q, Li J. Genome-Wide Identification of Laminin Family Related to Follicular Pseudoplacenta Development in Black Rockfish (Sebastes schlegelii). International Journal of Molecular Sciences. 2022; 23(18):10523. https://doi.org/10.3390/ijms231810523
Chicago/Turabian StyleZhao, Ning, Xueying Wang, Tao Wang, Xiaojie Xu, Qinghua Liu, and Jun Li. 2022. "Genome-Wide Identification of Laminin Family Related to Follicular Pseudoplacenta Development in Black Rockfish (Sebastes schlegelii)" International Journal of Molecular Sciences 23, no. 18: 10523. https://doi.org/10.3390/ijms231810523
APA StyleZhao, N., Wang, X., Wang, T., Xu, X., Liu, Q., & Li, J. (2022). Genome-Wide Identification of Laminin Family Related to Follicular Pseudoplacenta Development in Black Rockfish (Sebastes schlegelii). International Journal of Molecular Sciences, 23(18), 10523. https://doi.org/10.3390/ijms231810523






