The Antivirulence Activity of Umbelliferone and Its Protective Effect against A. hydrophila-Infected Grass Carp
Abstract
:1. Introduction
2. Results
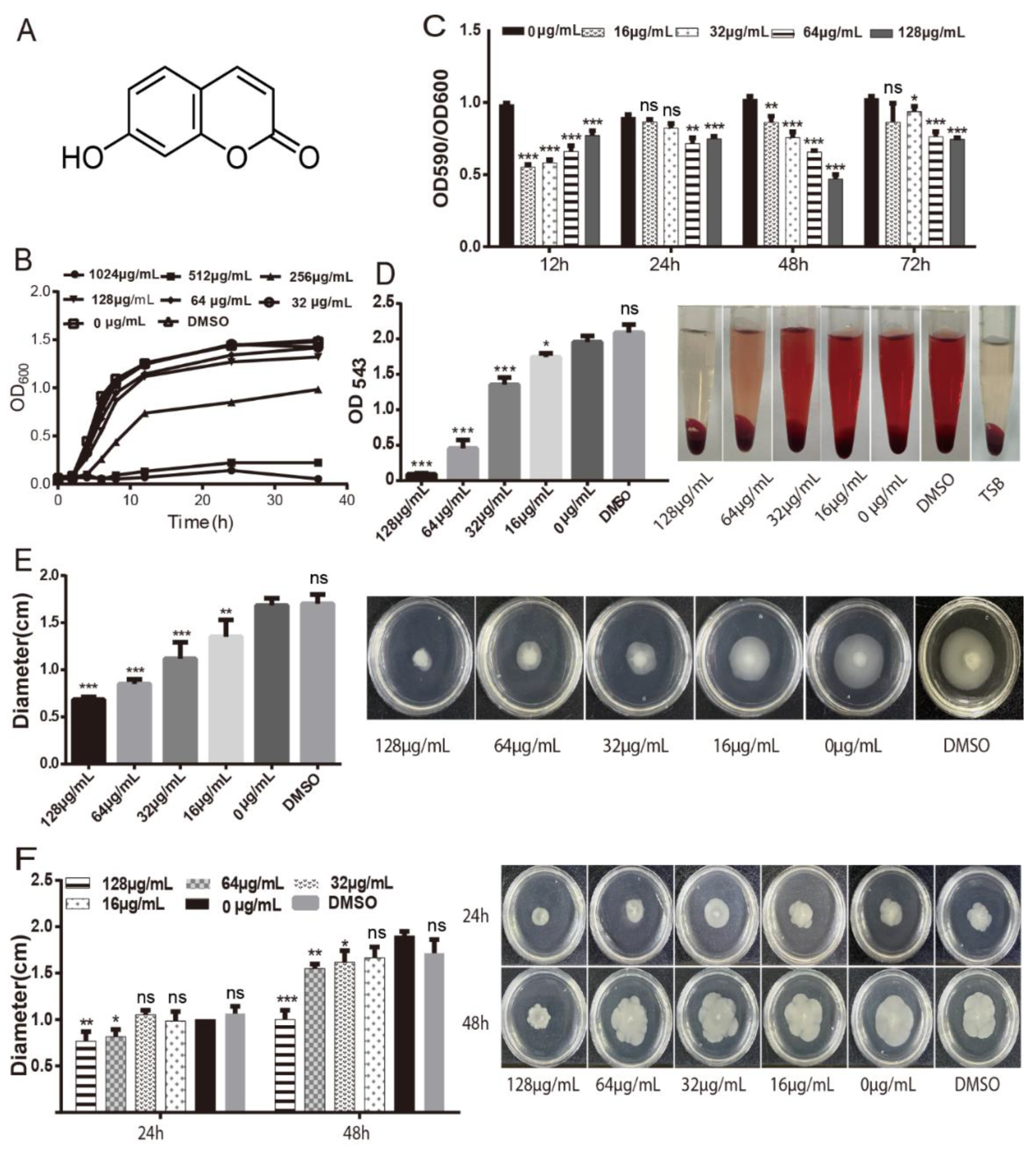
2.1. Inhibition of Virulence of A. hydrophila by Umbelliferone
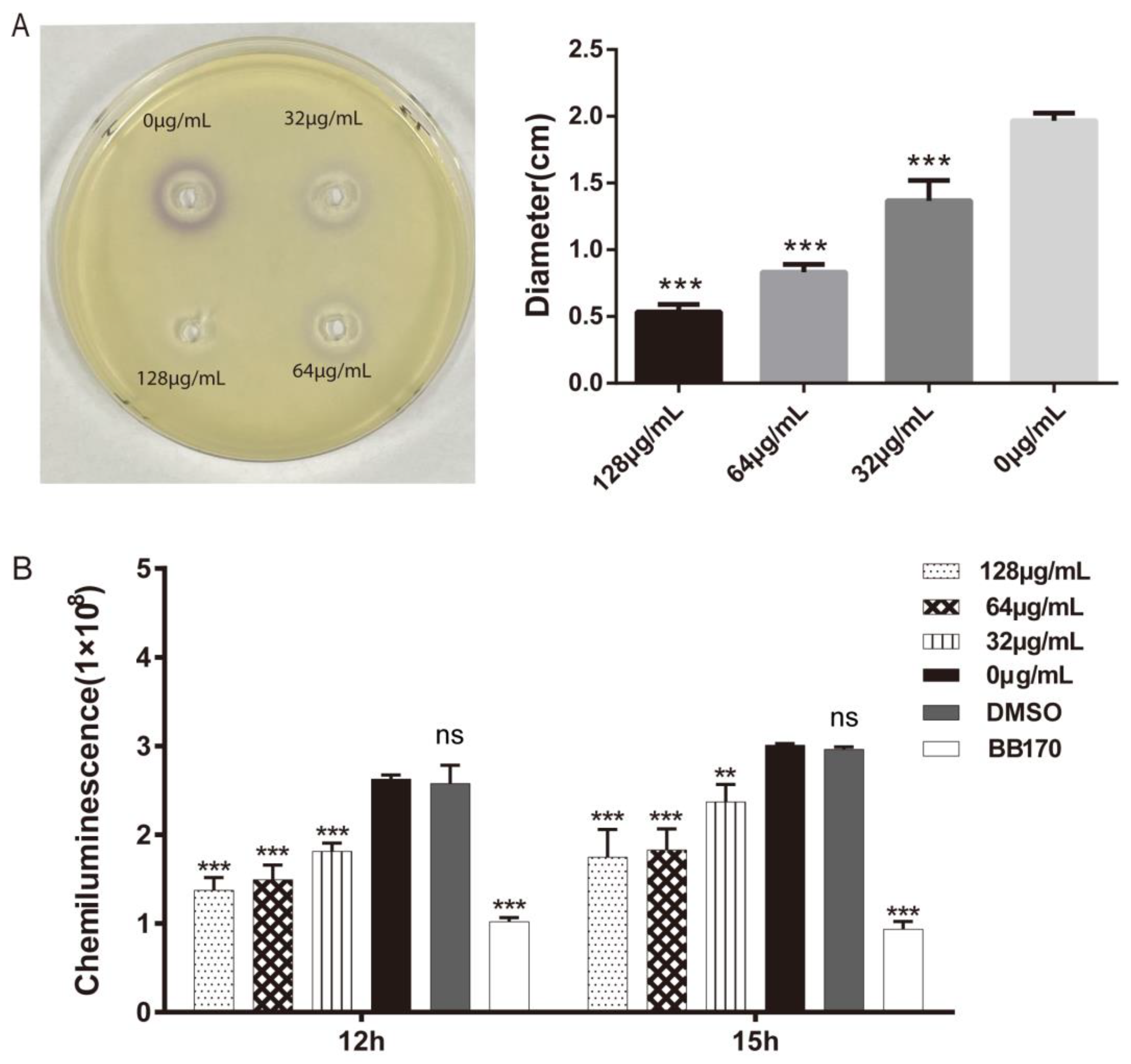
2.2. Effects of Umbelliferone on Quorum-Sensing Signals of A. hydrophila
2.3. Transcriptome Changes in A. hydrophila after Umbelliferone Treatment
2.4. qRT-PCR Validation of Transcriptome Data
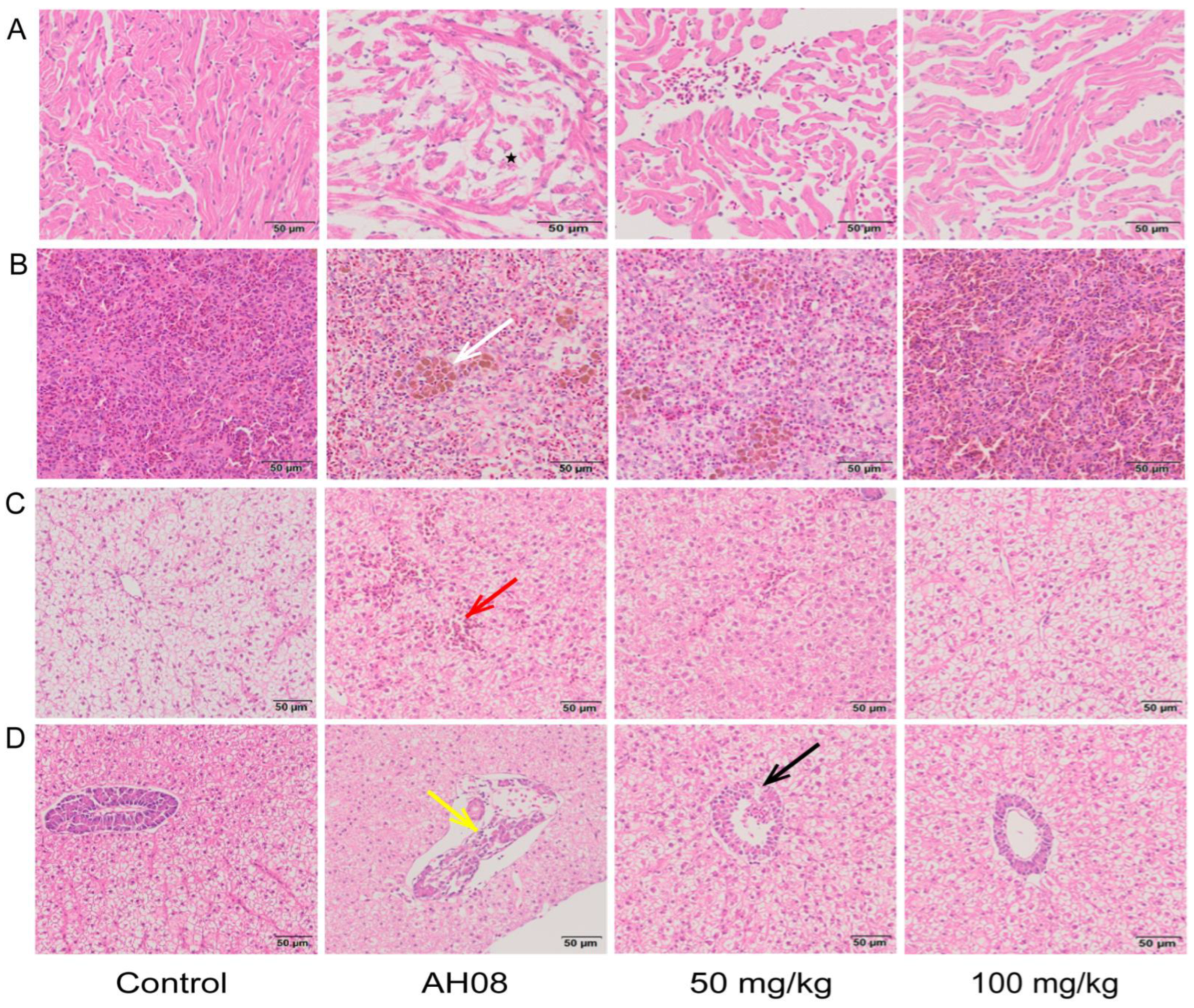
2.5. Protective Effect of Umbelliferone on Grass Carp Infected with A. hydrophila
3. Discussion
4. Materials and Methods
4.1. Bacterial Strains, Chemical Reagents, and Cultivation
4.2. Determinations of Minimum Inhibitory Concentration (MIC) and Growth Curve
4.3. Biofilm Formation
4.4. Motility Assays
4.5. Hemolysis Assay
4.6. Determination of Quorum-Sensing Signals
4.7. RNA Isolation and Sequencing
4.8. Quantification of Gene Expression Level
4.9. Differential Expression and Functional Enrichment Analyses
4.10. qRT-PCR
4.11. Animal Experiments
4.12. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chen, J.; Liu, N.; Zhang, H.; Zhao, Y.; Cao, X. The effects of Aeromonas hydrophila infection on oxidative stress, nonspecific immunity, autophagy, and apoptosis in the common carp. Dev. Comp. Immunol. 2020, 105, 103587. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Lin, Q.; Liu, L.; Liang, H.; Huang, Z.; Li, N. Display of ISKNV orf086 protein on the surface of Aeromonas hydrophila and its immunogenicity in Chinese perch (Siniperca chuatsi). Fish Shellfish Immunol. 2016, 56, 286–293. [Google Scholar] [CrossRef] [PubMed]
- Abdelhamed, H.; Banes, M.; Karsi, A.; Lawrence, M.L. Recombinant ATPase of Virulent Aeromonas hydrophila Protects Channel Catfish Against Motile Aeromonas Septicemia. Front. Immunol. 2019, 10, 1641. [Google Scholar] [CrossRef] [PubMed]
- Hazen, T.C.; Raker, M.L.; Esch, G.W.; Fliermans, C.B. Ultrastruct of red-sore lesions on largemouth bass (Micropterus salmoides): Association of the ciliate Epistylis sp. and the bacterium. Aeromonas hydrophila. J. Protozool. 1978, 25, 351–355. [Google Scholar] [CrossRef]
- Tomas, J.M. The main Aeromonas pathogenic factors. ISRN Microbiol. 2012, 2012, 256261. [Google Scholar] [CrossRef]
- Barger, P.C.; Liles, M.R.; Beck, B.H.; Newton, J.C. Differential production and secretion of potentially toxigenic extracellular proteins from hypervirulent Aeromonas hydrophila under biofilm and planktonic culture. BMC Microbiol. 2021, 21, 8. [Google Scholar] [CrossRef]
- Hossain, S.; Dahanayake, P.S.; De Silva, B.; Wickramanayake, M.; Wimalasena, S.; Heo, G.J. Multidrug resistant Aeromonas spp. isolated from zebrafish (Danio rerio): Antibiogram, antimicrobial resistance genes and class 1 integron gene cassettes. Lett. Appl. Microbiol. 2019, 68, 370–377. [Google Scholar] [CrossRef]
- Preena, P.G.; Arathi, D.; Raj, N.S.; Arun, K.T.; Arun, R.S.; Reshma, R.N.; Raja, S.T. Diversity of antimicrobial-resistant pathogens from a freshwater ornamental fish farm. Lett. Appl. Microbiol. 2020, 71, 108–116. [Google Scholar] [CrossRef]
- Tan, N.; Yazici-Tutunis, S.; Bilgin, M.; Tan, E.; Miski, M. Antibacterial Activities of Pyrenylated Coumarins from the Roots of Prangos hulusii. Molecules 2017, 22, 1098. [Google Scholar] [CrossRef]
- Wang, G.; Pang, J.; Hu, X.; Nie, T.; Lu, X.; Li, X.; Wang, X.; Lu, Y.; Yang, X.; Jiang, J.; et al. Daphnetin: A Novel Anti-Helicobacter pylori Agent. Int. J. Mol. Sci. 2019, 20, 850. [Google Scholar] [CrossRef] [Green Version]
- Maracic, S.; Kraljevic, T.G.; Paljetak, H.C.; Peric, M.; Matijasic, M.; Verbanac, D.; Cetina, M.; Raic-Malic, S. 1,2,3-Triazole pharmacophore-based benzofused nitrogen/sulfur heterocycles with potential anti-Moraxella catarrhalis activity. Bioorg. Med. Chem. 2015, 23, 7448–7463. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez-Barranquero, J.A.; Reen, F.J.; McCarthy, R.R.; O’Gara, F. Deciphering the role of coumarin as a novel quorum sensing inhibitor suppressing virulence phenotypes in bacterial pathogens. Appl. Microbiol. Biotechnol. 2015, 99, 3303–3316. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Kim, Y.G.; Cho, H.S.; Ryu, S.Y.; Cho, M.H.; Lee, J. Coumarins reduce biofilm formation and the virulence of Escherichia coli O157:H7. Phytomedicine 2014, 21, 1037–1042. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Ding, W.; Xu, Y.; Wu, D.; Li, S.; Chen, J.; Guo, B. New insights into the antibacterial activity of hydroxycoumarins against ralstonia solanacearum. Molecules 2016, 21, 468. [Google Scholar] [CrossRef]
- Singh, S.; Singh, S.K.; Chowdhury, I.; Singh, R. Understanding the Mechanism of Bacterial Biofilms Resistance to Antimicrobial Agents. Open Microbiol. J. 2017, 11, 53–62. [Google Scholar] [CrossRef]
- Yang, L.; Li, S.; Qin, X.; Jiang, G.; Chen, J.; Li, B.; Yao, X.; Liang, P.; Zhang, Y.; Ding, W. Exposure to Umbelliferone Reduces Ralstonia solanacearum Biofilm Formation, Transcription of Type III Secretion System Regulators and Effectors and Virulence on Tobacco. Front. Microbiol. 2017, 8, 1234. [Google Scholar] [CrossRef]
- Patriquin, G.M.; Banin, E.; Gilmour, C.; Tuchman, R.; Greenberg, E.P.; Poole, K. Influence of quorum sensing and iron on twitching motility and biofilm formation in Pseudomonas aeruginosa. J. Bacteriol. 2008, 190, 662–671. [Google Scholar] [CrossRef]
- Yeung, A.T.; Torfs, E.C.; Jamshidi, F.; Bains, M.; Wiegand, I.; Hancock, R.E.; Overhage, J. Swarming of Pseudomonas aeruginosa is controlled by a broad spectrum of transcriptional regulators, including MetR. J. Bacteriol. 2009, 191, 5592–5602. [Google Scholar] [CrossRef]
- Monte, J.; Abreu, A.C.; Borges, A.; Simoes, L.C.; Simoes, M. Antimicrobial Activity of Selected Phytochemicals against Escherichia coli and Staphylococcus aureus and Their Biofilms. Pathogens 2014, 3, 473–498. [Google Scholar] [CrossRef]
- Chopra, A.K.; Houston, C.W.; Peterson, J.W.; Jin, G.F. Cloning, expression, and sequence analysis of a cytolytic enterotoxin gene from Aeromonas hydrophila. Can. J. Microbiol. 1993, 39, 513–523. [Google Scholar] [CrossRef]
- Zhang, S.; Liu, N.; Liang, W.; Han, Q.; Zhang, W.; Li, C. Quorum sensing-disrupting coumarin suppressing virulence phenotypes in Vibrio splendidus. Appl. Microbiol. Biotechnol. 2017, 101, 3371–3378. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Sass, A.; Van Acker, H.; Wille, J.; Verhasselt, B.; Van Nieuwerburgh, F.; Kaever, V.; Crabbe, A.; Coenye, T. Coumarin Reduces Virulence and Biofilm Formation in Pseudomonas aeruginosa by Affecting Quorum Sensing, Type III Secretion and C-di-GMP Levels. Front. Microbiol. 2018, 9, 1952. [Google Scholar] [CrossRef] [PubMed]
- Lo, H.R.; Lin, J.H.; Chen, Y.H.; Chen, C.L.; Shao, C.P.; Lai, Y.C.; Hor, L.I. RTX toxin enhances the survival of Vibrio vulnificus during infection by protecting the organism from phagocytosis. J. Infect. Dis. 2011, 203, 1866–1874. [Google Scholar] [CrossRef]
- Fabret, C.; Feher, V.A.; Hoch, J.A. Two-component signal transduction in Bacillus subtilis: How one organism sees its world. J. Bacteriol. 1999, 181, 1975–1983. [Google Scholar] [CrossRef] [PubMed]
- Mahan, M.J.; Slauch, J.M.; Mekalanos, J.J. Selection of bacterial virulence genes that are specifically induced in host tissues. Science 1993, 259, 686–688. [Google Scholar] [CrossRef]
- Jackson-Litteken, C.D.; Di Venanzio, G.; Le, N.H.; Scott, N.E.; Djahanschiri, B.; Distel, J.S.; Pardue, E.J.; Ebersberger, I.; Felman, M.F. InvL, an invasin-like adhesin, is a type II secretion system substrate required for Acinetobacter baumannii uropathogenesis. mBio 2022, 3, e00258-22. [Google Scholar] [CrossRef]
- Green, E.R.; Meccas, J. Bacterial Secretion Systems: An Overview. Microbiol. Spectr. 2016, 4, 215–239. [Google Scholar] [CrossRef]
- Zoued, A.; Brunet, Y.R.; Durand, E.; Aschtgen, M.S.; Logger, L.; Douzi, B.; Journet, L.; Cambillau, C.; Cascales, E. Architecture and assembly of the Type VI secretion system. Biochim. Biophys. Acta 2014, 1843, 1664–1673. [Google Scholar] [CrossRef]
- Cianfanelli, F.R.; Monlezun, L.; Coulthurst, S.J. Aim, Load, Fire: The Type VI Secretion System, a Bacterial Nanoweapon. Trends Microbiol. 2016, 24, 51–62. [Google Scholar] [CrossRef]
- English, G.; Byron, O.; Cianfanelli, F.R.; Prescott, A.R.; Coulthurst, S.J. Biochemical analysis of TssK, a core component of the bacterial Type VI secretion system, reveals distinct oligomeric states of TssK and identifies a TssK-TssFG subcomplex. Biochem. J. 2014, 461, 291–304. [Google Scholar] [CrossRef] [Green Version]
- Felisberto-Rodrigues, C.; Durand, E.; Aschtgen, M.S.; Blangy, S.; Ortiz-Lombardia, M.; Douzi, B.; Cambillau, C.; Cascales, E. Towards a structural comprehension of bacterial type VI secretion systems: Characterization of the TssJ-TssM complex of an Escherichia coli pathovar. PLoS Pathog. 2011, 7, e1002386. [Google Scholar] [CrossRef] [PubMed]
- Juarez, G.E.; Galvan, E.M. Role of nutrient limitation in the competition between uropathogenic strains of Klebsiella pneumoniae and Escherichia coli in mixed biofilms. Biofouling 2018, 34, 287–298. [Google Scholar] [CrossRef] [PubMed]
- Hsu, J.L.; Manouvakhova, O.V.; Clemons, K.V.; Inayathullah, M.; Tu, A.B.; Sobel, R.A.; Tian, A.; Nazik, H.; Pothineni, V.R.; Pasupneti, S.; et al. Microhemorrhage-associated tissue iron enhances the risk for Aspergillus fumigatus invasion in a mouse model of airway transplantation. Sci. Transl. Med. 2018, 10, eaag2616. [Google Scholar] [CrossRef] [PubMed]
- Noinaj, N.; Guillier, M.; Barnard, T.J.; Buchanan, S.K. TonB-dependent transporters: Regulation, structure, and function. Annu. Rev. Microbiol. 2010, 64, 43–60. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Zhang, L.; Liu, Y.; Xu, N.; Zhou, S.; Yang, Q.; Yang, Y.; Ai, X. Thymol Protects Channel Catfish from Aeromonas hydrophila Infection by Inhibiting Aerolysin Expression and Biofilm Formation. Microorganisms 2020, 8, 636. [Google Scholar] [CrossRef]
- Langmead, B.; Salzberg, S.L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 2012, 9, 357–359. [Google Scholar] [CrossRef]
- Anders, S.; Pyl, P.T.; Huber, W. HTSeq—A Python framework to work with high-throughput sequencing data. Bioinformatics 2015, 31, 166–169. [Google Scholar] [CrossRef]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef]
- Yu, G.; Wang, L.G.; Han, Y.; He, Q.Y. clusterProfiler: An R package for comparing biological themes among gene clusters. OMICS 2012, 16, 284–287. [Google Scholar] [CrossRef]



| Gene Name | Log2 Fold Change | Description |
|---|---|---|
| Genes associated with exotoxin | ||
| VU14_RS08845 | −1.6900 | HBL/NHE enterotoxin family protein |
| VU14_RS08840 | −2.304 | Bacillus hemolytic enterotoxin (HBL) |
| rtxA | −1.5406 | MARTX multifunctional-autoprocessing repeats-in-toxin holotoxin RtxA |
| VU14_RS05570 | −1.5204 | Thermostable hemolysin |
| Genes associated with two-component system | ||
| VU14_RS20395 | −1.8170 | Two-component system response regulator |
| VU14_RS05225 | −1.5966 | Response regulator receiver domain |
| yehT | −1.6300 | Two-component system response regulator BtsR |
| uhpB | −1.5975 | Signal transduction histidine-protein kinase/phosphatase UhpB |
| Genes associated with secretory systems | ||
| exeI | −1.6425 | GspI family T2SS minor pseudopilin variant ExeI |
| exeN | −1.5519 | GspN family type II secretion system protein ExeN |
| VU14_RS12995 | −2.4463 | Type VI secretion system ImpA family |
| VU14_RS12990 | −2.0408 | Type VI secretion system PAAR protein |
| vgrG | −1.7650 | Type VI secretion system effector VgrG |
| tssK | −1.7623 | Type VI secretion system baseplate subunit TssK |
| tssM | −1.6745 | Type VI secretion system membrane subunit TssM |
| vasH | −1.1018 | σ-54-dependent transcriptional regulator VasH |
| VU14_RS13025 | −1.5086 | Type VI secretion system protein DotU |
| Genes associated with iron transport | ||
| VU14_RS01835 | −2.1770 | Ligand-gated channel protein |
| VU14_RS04515 | −1.8007 | Energy transducer TonB |
| VU14_RS20175 | −1.7896 | TonB-dependent siderophore receptor |
| VU14_RS04520 | −1.8444 | Biopolymer transporter ExbD |
| VU14_RS14250 | −1.7692 | TonB-dependent hemoglobin/transferrin/lactoferrin family receptor |
| VU14_RS17650 | −1.7498 | TonB-dependent hemoglobin/transferrin/lactoferrin family receptor |
| VU14_RS20010 | −1.5912 | TonB-dependent copper receptor |
| VU14_RS12465 | −1.5400 | TonB-dependent siderophore receptor |
| amoA | −1.5178 | Isochorismate synthases AmoA |
| amoG | −1.5635 | Amonabactin biosynthesis nonribosomal peptide synthetase AmoG |
| amoH | −1.5880 | Amonabactin biosynthesis glycine adenylation protein AmoH |
| Gene Name | Log2 Fold Change | Description |
|---|---|---|
| Genes associated with ribosomes | ||
| rpsG | 2.2465 | 30S ribosomal protein S7 |
| rpsF | 2.2355 | 30S ribosomal protein S6 |
| rpsJ | 2.1849 | 30S ribosomal protein S10 |
| rpsR | 2.1632 | 30S ribosomal protein S18 |
| Genes associated with citrate cycle (TCA cycle) | ||
| mdh | 2.3949 | Malate dehydrogenase |
| VU14_RS12215 | 2.6071 | Fumarate hydratase |
| frdD | 2.3141 | Fumarate reductase subunit FrdD |
| sucD | 1.9545 | Succinate-CoA ligase subunit α |
| Genes associated with carbon metabolism | ||
| VU14_RS04850 | 2.2821 | Glyceraldehyde-3-phosphate dehydrogenase |
| fadB | 2.4898 | Fatty acid oxidation complex subunit α FadB |
| VU14_RS06340 | 1.6773 | Formate dehydrogenase subunit α |
| Genes associated with oxidative phosphorylation | ||
| frdD | 2.3141 | Fumarate reductase subunit FrdD |
| sdhD | 1.7136 | Succinate dehydrogenase hydrophobic membrane anchor protein |
| sdhC | 1.5912 | Succinate dehydrogenase cytochrome b556 subunit |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, L.; Jin, X.; Xiong, Z.; Tang, H.; Guo, H.; Ye, G.; Chen, D.; Yang, S.; Yin, Z.; Fu, H.; et al. The Antivirulence Activity of Umbelliferone and Its Protective Effect against A. hydrophila-Infected Grass Carp. Int. J. Mol. Sci. 2022, 23, 11119. https://doi.org/10.3390/ijms231911119
Zhao L, Jin X, Xiong Z, Tang H, Guo H, Ye G, Chen D, Yang S, Yin Z, Fu H, et al. The Antivirulence Activity of Umbelliferone and Its Protective Effect against A. hydrophila-Infected Grass Carp. International Journal of Molecular Sciences. 2022; 23(19):11119. https://doi.org/10.3390/ijms231911119
Chicago/Turabian StyleZhao, Ling, Xiaoyu Jin, Ziqian Xiong, Huaqiao Tang, Hongrui Guo, Gang Ye, Defang Chen, Shiyong Yang, Zhongqiong Yin, Hualin Fu, and et al. 2022. "The Antivirulence Activity of Umbelliferone and Its Protective Effect against A. hydrophila-Infected Grass Carp" International Journal of Molecular Sciences 23, no. 19: 11119. https://doi.org/10.3390/ijms231911119






