Diphenyl Diselenide Alleviates Tert-Butyl Hydrogen Peroxide-Induced Oxidative Stress and Lipopolysaccharide-Induced Inflammation in Rat Glomerular Mesangial Cells
Abstract
:1. Introduction
2. Results
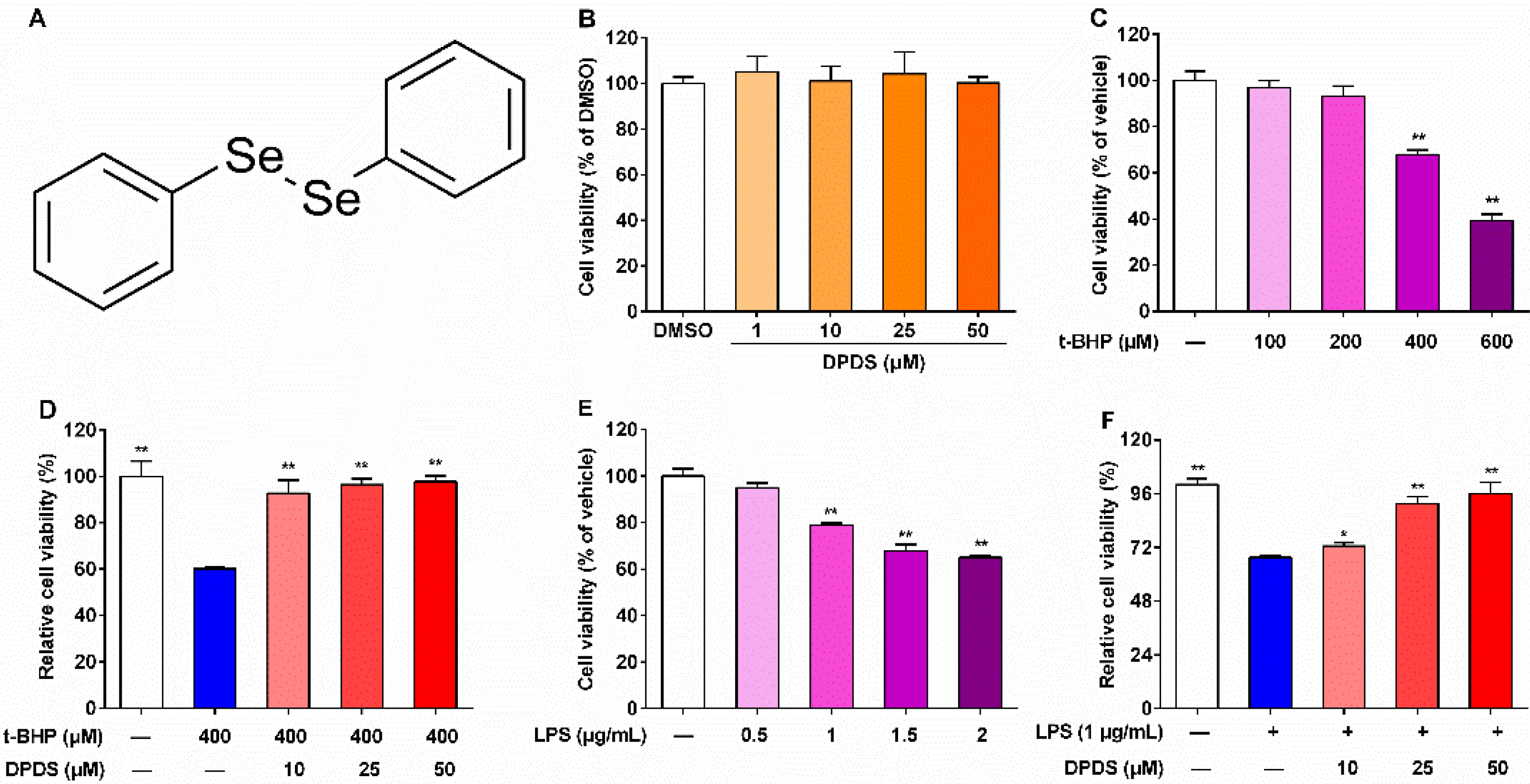
2.1. DPDS Protected HBZY-1 Cells against t-BHP and LPS-Induced Injury
2.2. DPDS Attenuated t-BHP-Induced Oxidative Stress in HBZY-1 Cells
2.3. DPDS Activated the Nrf2/Keap1 Signaling Pathway in t-BHP-Stimulated HBZY-1 Cells
2.4. DPDS Enhanced the Gene Expression of Nrf2 and Antioxidant Enzymes in t-BHP-Stimulated HBZY-1 Cells
2.5. DPDS Decreased Intracellular Contents and Gene Expression of Pro-Inflammatory Cytokines in LPS-Stimulated HBZY-1 Cells
2.6. DPDS Suppressed LPS-Induced NFκB and MAPK Signaling in HBZY-1 Cells
3. Discussion
4. Materials and Methods
4.1. Reagents and Antibodies
4.2. Cell Culture
4.3. Cell Viability Assay
4.4. Assessment of ROS Levels
4.5. Measurement of Intracellular Oxidative Stress Parameters
4.6. Enzyme-Linked Immunosorbent Assay (ELISA)
4.7. Western Blot
4.8. Gene Expression Analysis
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sagoo, M.K.; Gnudi, L. Diabetic nephropathy: An overview. Methods Mol. Biol. 2020, 2067, 3–7. [Google Scholar] [PubMed]
- Papadopoulou-Marketou, N.; Paschou, S.A.; Marketos, N.; Adamidi, S.; Adamidis, S.; Kanaka-Gantenbein, C. Diabetic nephropathy in type 1 diabetes. Minerva Med. 2018, 109, 218–228. [Google Scholar] [CrossRef]
- Samsu, N. Diabetic nephropathy: Challenges in pathogenesis, diagnosis, and treatment. Biomed. Res. Int. 2021, 2021, 1497449. [Google Scholar] [CrossRef] [PubMed]
- Sharma, D.; Gondaliya, P.; Tiwari, V.; Kalia, K. Kaempferol attenuates diabetic nephropathy by inhibiting RhoA/Rho-kinase mediated inflammatory signalling. Biomed. Pharmacother. 2019, 109, 1610–1619. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Chen, W.; Lu, P.; Ma, Y.; Liang, X.; Liu, Y. Ginsenoside Rg1 attenuates the inflammation and oxidative stress induced by diabetic nephropathy through regulating the PI3K/AKT/FOXO3 pathway. Ann. Transl. Med. 2021, 9, 1789. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xu, J.; Cheng, Z. YAP1 promotes high glucose-induced inflammation and extracellular matrix deposition in glomerular mesangial cells by modulating NF-κB/JMJD3 pathway. Exp. Ther. Med. 2021, 22, 1349. [Google Scholar] [CrossRef] [PubMed]
- Hussain Lodhi, A.; Ahmad, F.U.; Furwa, K.; Madni, A. Role of oxidative stress and reduced endogenous hydrogen sulfide in diabetic nephropathy. Drug Des. Dev. Ther. 2021, 15, 1031–1043. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.Q.; Ma, Y.L.; Chen, F.; Wang, S.Y.; Chen, B.P.; Shi, J. Artesunate ameliorates high glucose-induced rat glomerular mesangial cell injury by suppressing the TLR4/NF-κB/NLRP3 inflammasome pathway. Chem. Biol. Interact. 2018, 293, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Jian, P.; Erjiong, H.; Qihan, Z. Ginkgetin alleviates high glucose-evoked mesangial cell oxidative stress injury, inflammation, and extracellular matrix (ECM) deposition in an AMPK/mTOR-mediated autophagy axis. Chem. Biol. Drug Des. 2021, 98, 620–630. [Google Scholar] [CrossRef] [PubMed]
- Rayego-Mateos, S.; Morgado-Pascual, J.L.; Opazo-Ríos, L.; Guerrero-Hue, M.; García-Caballero, C.; Vázquez-Carballo, C.; Mas, S.; Sanz, A.B.; Herencia, C.; Mezzano, S.; et al. Pathogenic pathways and therapeutic approaches targeting inflammation in diabetic nephropathy. Int. J. Mol. Sci. 2020, 21, 3798. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Zhou, L. The signaling of cellular senescence in diabetic nephropathy. Oxid. Med. Cell Longev. 2019, 2019, 7495629. [Google Scholar] [CrossRef] [PubMed]
- Calle, P.; Hotter, G. Macrophage phenotype and fibrosis in diabetic nephropathy. Int. J. Mol. Sci. 2020, 21, 2806. [Google Scholar] [CrossRef]
- Xiang, E.; Han, B.; Zhang, Q.; Rao, W.; Wang, Z.; Chang, C.; Zhang, Y.; Tu, C.; Li, C.; Wu, D. Human umbilical cord-derived mesenchymal stem cells prevent the progression of early diabetic nephropathy through inhibiting inflammation and fibrosis. Stem Cell Res. Ther. 2020, 11, 336. [Google Scholar] [CrossRef] [PubMed]
- Adedara, I.A.; Owoeye, O.; Ajayi, B.O.; Awogbindin, I.O.; Rocha, J.B.T.; Farombi, E.O. Diphenyl diselenide abrogates chlorpyrifos-induced hypothalamic-pituitary-testicular axis impairment in rats. Biochem. Biophys. Res. Commun. 2018, 503, 171–176. [Google Scholar] [CrossRef]
- De Bem, A.F.; Farina, M.; Portella, R.D.L.; Nogueira, C.W.; Dinis, T.C.P.; Laranjinha, J.A.N.; Almeida, L.M.; Rocha, J.B.T. Diphenyl diselenide, a simple glutathione peroxidase mimetic, inhibits human LDL oxidation in vitro. Atherosclerosis 2008, 201, 92–100. [Google Scholar] [CrossRef]
- Luchese, C.; Prigol, M.; Duarte, M.M.; Nogueira, C.W. Diphenyl diselenide reduces inflammation in the mouse model of pleurisy induced by carrageenan: Reduction of pro-inflammatory markers and reactive species levels. Inflamm. Res. 2012, 61, 1117–1124. [Google Scholar] [CrossRef] [PubMed]
- Dobrachinski, F.; de Silva, M.H.; Tassi, C.L.C.; de Carvalho, N.R.; Dias, G.R.M.; Golombieski, R.M.; da Silva Loreto, E.L.; da Rocha, J.B.T.; Fighera, M.R.; Soares, F.A.A. Neuroprotective effect of diphenyl diselenide in an experimental stroke model: Maintenance of redox system in mitochondria of brain regions. Neurotox. Res. 2014, 26, 317–330. [Google Scholar] [CrossRef]
- Wilson, S.R.; Zucker, P.A.; Huang, R.R.C.; Spector, A. Development of synthetic compounds with glutathione peroxidase activity. J. Am. Chem. Soc. 1989, 111, 5936–5939. [Google Scholar] [CrossRef]
- Petronilho, F.; Michels, M.; Danielski, L.G.; Goldim, M.P.; Florentino, D.; Vieira, A.; Mendonça, M.G.; Tournier, M.; Piacentini, B.; Giustina, A.D.; et al. Diphenyl diselenide attenuates oxidative stress and inflammatory parameters in ulcerative colitis: A comparison with ebselen. Pathol. Res. Pract. 2016, 212, 755–760. [Google Scholar] [CrossRef]
- de Bem, A.F.; Fiuza, B.; Calcerrada, P.C.; Brito, P.M.; Peluffo, G.; Dinis, T.C.P.; Trujillo, M.; Rocha, J.B.T.; Radi, R.; Almeida, L.M. Protective effect of diphenyl diselenide against peroxynitrite-mediated endothelial cell death: A comparison with ebselen. Nitric Oxide 2013, 31, 20–30. [Google Scholar] [CrossRef] [Green Version]
- Nogueira, C.W.; Meotti, F.C.; Curte, E.; Pilissão, C.; Zeni, G.; Rocha, J.B.T. Investigations into the potential neurotoxicity induced by diselenides in mice and rats. Toxicology 2003, 183, 29–37. [Google Scholar] [CrossRef]
- Meotti, F.C.; Borges, V.C.; Zeni, G.; Rocha, J.B.T.; Nogueira, C.W. Potential renal and hepatic toxicity of diphenyl diselenide, diphenyl ditelluride and Ebselen for rats and mice. Toxicol. Lett. 2003, 143, 9–16. [Google Scholar] [CrossRef]
- Barbosa, N.B.V.; Rocha, J.B.T.; Wondracek, D.C.; Perottoni, J.; Zeni, G.; Nogueira, C.W. Diphenyl diselenide reduces temporarily hyperglycemia: Possible relationship with oxidative stress. Chem. Biol. Interact. 2006, 163, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, C.N.; Huan, Y.; Cao, H.; Sun, S.J.; Lei, L.; Liu, Q.; Liu, S.N.; Ji, W.M.; Huang, K.X.; et al. Diphenyl diselenide ameliorates diabetic nephropathy in streptozotocin-induced diabetic rats via suppressing oxidative stress and inflammation. Chem. Biol. Interact. 2021, 338, 109427. [Google Scholar] [CrossRef] [PubMed]
- Bellezza, I.; Giambanco, I.; Minelli, A.; Donato, R. Nrf2-Keap1 signaling in oxidative and reductive stress. Biochim. Biophys. Acta Mol. Cell Res. 2018, 1865, 721–733. [Google Scholar] [CrossRef]
- Qiao, S.M.; Liu, R.; Lv, C.J.; Miao, Y.M.; Yue, M.F.; Tao, Y.; Wei, Z.F.; Xia, Y.F.; Dai, Y. Bergenin impedes the generation of extracellular matrix in glomerular mesangial cells and ameliorates diabetic nephropathy in mice by inhibiting oxidative stress via the mTOR/β-TrcP/Nrf2 pathway. Free Radic. Biol. Med. 2019, 145, 118–135. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, L.; Wen, X.; Hao, D.; Zhang, N.; He, G.; Jiang, X. NF-κB signaling in skin aging. Mech. Ageing Dev. 2019, 184, 111160. [Google Scholar] [CrossRef]
- Ren, J.; Su, D.; Li, L.; Cai, H.; Zhang, M.; Zhai, J.; Li, M.; Wu, X.; Hu, K. Anti-inflammatory effects of Aureusidin in LPS-stimulated RAW264.7 macrophages via suppressing NF-κB and activating ROS- and MAPKs-dependent Nrf2/HO-1 signaling pathways. Toxicol. Appl. Pharmacol. 2020, 387, 114846. [Google Scholar] [CrossRef]
- Han, Y.; Xu, X.; Tang, C.; Gao, P.; Chen, X.; Xiong, X.; Yang, M.; Yang, S.; Zhu, X.; Yuan, S.; et al. Reactive oxygen species promote tubular injury in diabetic nephropathy: The role of the mitochondrial ros-txnip-nlrp3 biological axis. Redox Biol. 2018, 16, 32–46. [Google Scholar] [CrossRef]
- Victor, P.; Umapathy, D.; George, L.; Juttada, U.; Ganesh, G.V.; Amin, K.N.; Viswanathan, V.; Ramkumar, K.M. Crosstalk between endoplasmic reticulum stress and oxidative stress in the progression of diabetic nephropathy. Cell Stress Chaperones 2021, 26, 311–321. [Google Scholar] [CrossRef]
- Du, L.; Wang, L.; Wang, B.; Wang, J.; Hao, M.; Chen, Y.B.; Li, X.Z.; Li, Y.; Jiang, Y.F.; Li, C.C.; et al. A novel compound AB38b attenuates oxidative stress and ECM protein accumulation in kidneys of diabetic mice through modulation of Keap1/Nrf2 signaling. Acta. Pharmacol. Sin. 2020, 41, 358–372. [Google Scholar] [CrossRef]
- Xu, Y.; Bai, L.W.; Chen, X.H.; Li, Y.; Qin, Y.; Meng, X.Y.; Zhang, Q.G. 6-Shogaol ameliorates diabetic nephropathy through anti-inflammatory, hyperlipidemic, anti-oxidative activity in db/db mice. Biomed. Pharmacother. 2018, 97, 633–641. [Google Scholar] [CrossRef]
- Suryavanshi, S.V.; Garud, M.S.; Barve, K.; Addepalli, V.; Utpat, S.V.; Kulkarni, Y.A. Triphala ameliorates nephropathy via inhibition of TGF-β1 and oxidative stress in siabetic rats. Pharmacology 2020, 105, 681–691. [Google Scholar] [CrossRef]
- Shen, H.; Wang, W. Effect of glutathione liposomes on diabetic nephropathy based on oxidative stress and polyol pathway mechanism. J. Liposome Res. 2021, 31, 317–325. [Google Scholar] [CrossRef]
- Matsumoto, N.; Omagari, D.; Ushikoshi-Nakayama, R.; Yamazaki, T.; Inoue, H.; Saito, I. Hyperglycemia induces generation of reactive oxygen species and accelerates apoptotic cell death in salivary gland cells. Pathobiology 2021, 88, 234–241. [Google Scholar] [CrossRef]
- Zhang, Q.; Hu, Y.; Hu, J.E.; Ding, Y.; Shen, Y.; Xu, H.; Chen, H.; Wu, N. Sp1-mediated upregulation of Prdx6 expression prevents podocyte injury in diabetic nephropathy via mitigation of oxidative stress and ferroptosis. Life Sci. 2021, 278, 119529. [Google Scholar] [CrossRef]
- Huang, S.; Tan, M.; Guo, F.; Dong, L.; Liu, Z.; Yuan, R.; Dongzhi, Z.; Lee, D.S.; Wang, Y.; Li, B. Nepeta angustifolia C. Y. Wu improves renal injury in HFD/STZ-induced diabetic nephropathy and inhibits oxidative stress-induced apoptosis of mesangial cells. J. Ethnopharmacol. 2020, 255, 112771. [Google Scholar] [CrossRef]
- Bravo-Sánchez, E.; Peña-Montes, D.; Sánchez-Duarte, S.; Saavedra-Molina, A.; Sánchez-Duarte, E.; Montoya-Pérez, R. Effects of apocynin on heart muscle oxidative stress of rats with experimental diabetes: Implications for mitochondria. Antioxidants 2021, 10, 335. [Google Scholar] [CrossRef]
- Asgher, M.; Ahmed, S.; Sehar, Z.; Gautam, H.; Gandhi, S.G.; Khan, N.A. Hydrogen peroxide modulates activity and expression of antioxidant enzymes and protects photosynthetic activity from arsenic damage in rice (Oryza sativa L.). J. Hazard. Mater. 2021, 401, 123365. [Google Scholar] [CrossRef]
- Wang, Y.; Branicky, R.; Noë, A.; Hekimi, S. Superoxide dismutases: Dual roles in controlling ROS damage and regulating ROS signaling. J. Cell Biol. 2018, 217, 1915–1928. [Google Scholar] [CrossRef] [Green Version]
- Hatori, Y.; Kubo, T.; Sato, Y.; Inouye, S.; Akagi, R.; Seyama, T. Visualization of the redox status of cytosolic glutathione using the organelle- and cytoskeleton-targeted redox sensors. Antioxidants 2020, 9, 129. [Google Scholar] [CrossRef]
- Mei, Y.; Wang, Z.H.; Zhang, Y.F.; Wan, T.; Xue, J.C.; He, W.; Luo, Y.; Xu, Y.J.; Bai, X.; Wang, Q.; et al. FA-97, a new synthetic caffeic acid phenethyl ester derivative, ameliorates DSS-induced colitis against oxidative stress by activating Nrf2/HO-1 pathway. Front. Immunol. 2020, 10, 2969. [Google Scholar] [CrossRef]
- Li, S.; Zheng, L.; Zhang, J.; Liu, X.; Wu, Z. Inhibition of ferroptosis by up-regulating Nrf2 delayed the progression of diabetic nephropathy. Free Radic. Biol. Med. 2021, 162, 435–449. [Google Scholar] [CrossRef]
- Akino, N.; Wada-Hiraike, O.; Isono, W.; Terao, H.; Honjo, H.; Miyamoto, Y.; Tanikawa, M.; Sone, K.; Hirano, M.; Harada, M.; et al. Activation of Nrf2/Keap1 pathway by oral Dimethylfumarate administration alleviates oxidative stress and age-associated infertility might be delayed in the mouse ovary. Reprod. Biol. Endocrinol. 2019, 17, 23. [Google Scholar] [CrossRef]
- Mancini, G.; Straliotto, M.R.; da Rocha, J.B.T.; de Bem, A.F. Diphenyl diselenide improves the antioxidant response via activation of the Nrf-2 pathway in macrophage cells. Free Radic. Biol. Med. 2014, 75 (Suppl. S1), S40. [Google Scholar] [CrossRef]
- Muller, S.G.; Jardim, N.S.; Quines, C.B.; Nogueira, C.W. Diphenyl diselenide regulates Nrf2/Keap-1 signaling pathway and counteracts hepatic oxidative stress induced by bisphenol A in male mice. Environ. Res. 2018, 164, 280–287. [Google Scholar] [CrossRef]
- Yang, X.D.; Yang, Y.Y. Ferroptosis as a novel therapeutic target for diabetes and its complications. Front. Endocrinol. 2022, 13, 853822. [Google Scholar] [CrossRef]
- Tuo, Q.Z.; Masaldan, S.; Southon, A.; Mawal, C.; Ayton, S.; Bush, A.I.; Lei, P.; Belaidi, A.A. Characterization of selenium compounds for anti-ferroptotic activity in neuronal cells and after cerebral ischemia-reperfusion injury. Neurotherapeutics 2021, 18, 2682–2691. [Google Scholar] [CrossRef]
- Milas, O.; Gadalean, F.; Vlad, A.; Dumitrascu, V.; Velciov, S.; Gluhovschi, C.; Bob, F.; Popescu, R.; Ursoniu, S.; Jianu, D.C.; et al. Pro-inflammatory cytokines are associated with podocyte damage and proximal tubular dysfunction in the early stage of diabetic kidney disease in type 2 diabetes mellitus patients. J. Diabetes Complicat. 2020, 34, 107479. [Google Scholar] [CrossRef]
- Chen, B.; Wu, M.; Zang, C.; Li, Y.; Xu, Z. Association between IL-6 polymorphisms and diabetic nephropathy risk: A meta-analysis. Am. J. Med. Sci. 2019, 358, 363–373. [Google Scholar] [CrossRef]
- Wu, R.; Liu, X.; Yin, J.; Wu, H.; Cai, X.; Wang, N.; Qian, Y.; Wang, F. IL-6 receptor blockade ameliorates diabetic nephropathy via inhibiting inflammasome in mice. Metabolism 2018, 83, 18–24. [Google Scholar] [CrossRef]
- Lampropoulou, I.T.; Stangou, Μ.; Sarafidis, P.; Gouliovaki, A.; Giamalis, P.; Tsouchnikas, I.; Didangelos, T.; Papagianni, A. TNF-α pathway and T-cell immunity are activated early during the development of diabetic nephropathy in type II diabetes mellitus. Clin. Immunol. 2020, 215, 108423. [Google Scholar] [CrossRef]
- Barnabei, L.; Laplantine, E.; Mbongo, W.; Rieux-Laucat, F.; Weil, R. NF-κB: At the borders of autoimmunity and inflammation. Front. Immunol. 2021, 12, 716469. [Google Scholar] [CrossRef]
- Kunnumakkara, A.B.; Shabnam, B.; Girisa, S.; Harsha, C.; Banik, K.; Devi, T.B.; Choudhury, R.; Sahu, H.; Parama, D.; Sailo, B.L.; et al. Inflammation, NF-κB, and chronic diseases: How are they linked? Crit. Rev. Immunol. 2020, 40, 1–39. [Google Scholar] [CrossRef]
- Pérez-Morales, R.E.; Del Pino, M.D.; Valdivielso, J.M.; Ortiz, A.; Mora-Fernández, C.; Navarro-González, J.F. Inflammation in diabetic kidney disease. Nephron 2019, 143, 12–16. [Google Scholar] [CrossRef]
- Zhang, S.; Xu, L.; Liang, R.; Yang, C.; Wang, P. Baicalin suppresses renal fibrosis through microRNA-124/TLR4/NF-κB axis in streptozotocin-induced diabetic nephropathy mice and high glucose-treated human proximal tubule epithelial cells. J. Physiol. Biochem. 2020, 76, 407–416. [Google Scholar] [CrossRef]
- Xu, X.; Zhang, L.; Hua, F.; Zhang, C.; Zhang, C.; Mi, X.; Qin, N.; Wang, J.; Zhu, A.; Qin, Z.; et al. FOXM1-activated SIRT4 inhibits NF-κB signaling and NLRP3 inflammasome to alleviate kidney injury and podocyte pyroptosis in diabetic nephropathy. Exp. Cell Res. 2021, 408, 112863. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.Y.; Ni, X.; Hu, K.Q.; Meng, F.L.; Li, M.; Ma, X.L.; Meng, T.T.; Wu, H.H.; Ge, D.; Zhao, J.; et al. Cilostazol alleviate nicotine induced cardiomyocytes hypertrophy through modulation of autophagy by CTSB/ROS/p38MAPK/JNK feedback loop. Int. J. Biol. Sci. 2020, 16, 2001–2013. [Google Scholar] [CrossRef]
- Caeser, R.; Hulton, C.; Costa, E.; Durani, V.; Little, M.; Chen, X.; Tischfield, S.E.; Asher, M.; Kombak, F.E.; Chavan, S.S.; et al. MAPK pathway activation selectively inhibits ASCL1-driven small cell lung cancer. iScience 2021, 24, 103224. [Google Scholar] [CrossRef]
- Cheng, S.; Li, L.; Song, C.; Jin, H.; Ma, S.; Kang, P. Sitagliptin relieves diabetic nephropathy fibrosis via the MAPK/ERK signaling pathway. Minerva Endocrinol. 2020, 45, 273–275. [Google Scholar] [CrossRef]
- Li, Y.; Hou, J.G.; Liu, Z.; Gong, X.J.; Hu, J.N.; Wang, Y.P.; Liu, W.C.; Lin, X.H.; Wang, Z.; Li, W. Alleviative effects of 20(R)-Rg3 on HFD/STZ-induced diabetic nephropathy via MAPK/NF-κB signaling pathways in C57BL/6 mice. J. Ethnopharmacol. 2021, 267, 113500. [Google Scholar] [CrossRef]
- Wang, Y.Q.; Fan, C.C.; Chen, B.P.; Shi, J. Retracted: Resistin-like molecule Beta (RELM-β) regulates proliferation of human diabetic nephropathy mesangial cells via mitogen-activated protein kinases (MAPK) signaling pathway. Med. Sci. Monit. 2022, 28, e936542. [Google Scholar] [CrossRef]
- Wang, X.; Huan, Y.; Li, C.N.; Cao, H.; Sun, S.J.; Lei, L.; Liu, Q.; Liu, S.N.; Ji, W.M.; Liu, H.M.; et al. Diphenyl diselenide alleviates diabetic peripheral neuropathy in rats with streptozotocin-induced diabetes by modulating oxidative stress. Biochem. Pharmacol. 2020, 182, 114221. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Huan, Y.; Jiang, Q.; Sun, S.J.; Jia, C.M.; Shen, Z.F. Effects and potential mechanisms of pioglitazone on lipid metabolism in obese diabetic KKAy mice. PPAR Res. 2014, 2014, 538183. [Google Scholar] [CrossRef] [PubMed] [Green Version]






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Huan, Y.; Liu, S.; Li, C.; Cao, H.; Lei, L.; Liu, Q.; Ji, W.; Sun, S.; Huang, K.; et al. Diphenyl Diselenide Alleviates Tert-Butyl Hydrogen Peroxide-Induced Oxidative Stress and Lipopolysaccharide-Induced Inflammation in Rat Glomerular Mesangial Cells. Int. J. Mol. Sci. 2022, 23, 11215. https://doi.org/10.3390/ijms231911215
Wang X, Huan Y, Liu S, Li C, Cao H, Lei L, Liu Q, Ji W, Sun S, Huang K, et al. Diphenyl Diselenide Alleviates Tert-Butyl Hydrogen Peroxide-Induced Oxidative Stress and Lipopolysaccharide-Induced Inflammation in Rat Glomerular Mesangial Cells. International Journal of Molecular Sciences. 2022; 23(19):11215. https://doi.org/10.3390/ijms231911215
Chicago/Turabian StyleWang, Xing, Yi Huan, Shuainan Liu, Caina Li, Hui Cao, Lei Lei, Quan Liu, Wenming Ji, Sujuan Sun, Kaixun Huang, and et al. 2022. "Diphenyl Diselenide Alleviates Tert-Butyl Hydrogen Peroxide-Induced Oxidative Stress and Lipopolysaccharide-Induced Inflammation in Rat Glomerular Mesangial Cells" International Journal of Molecular Sciences 23, no. 19: 11215. https://doi.org/10.3390/ijms231911215





