Effects of Combined Pentadecanoic Acid and Tamoxifen Treatment on Tamoxifen Resistance in MCF−7/SC Breast Cancer Cells
Abstract
:1. Introduction
2. Results
2.1. Characterization of Drug−Resistant Human Breast Cancer MCF−7/SC
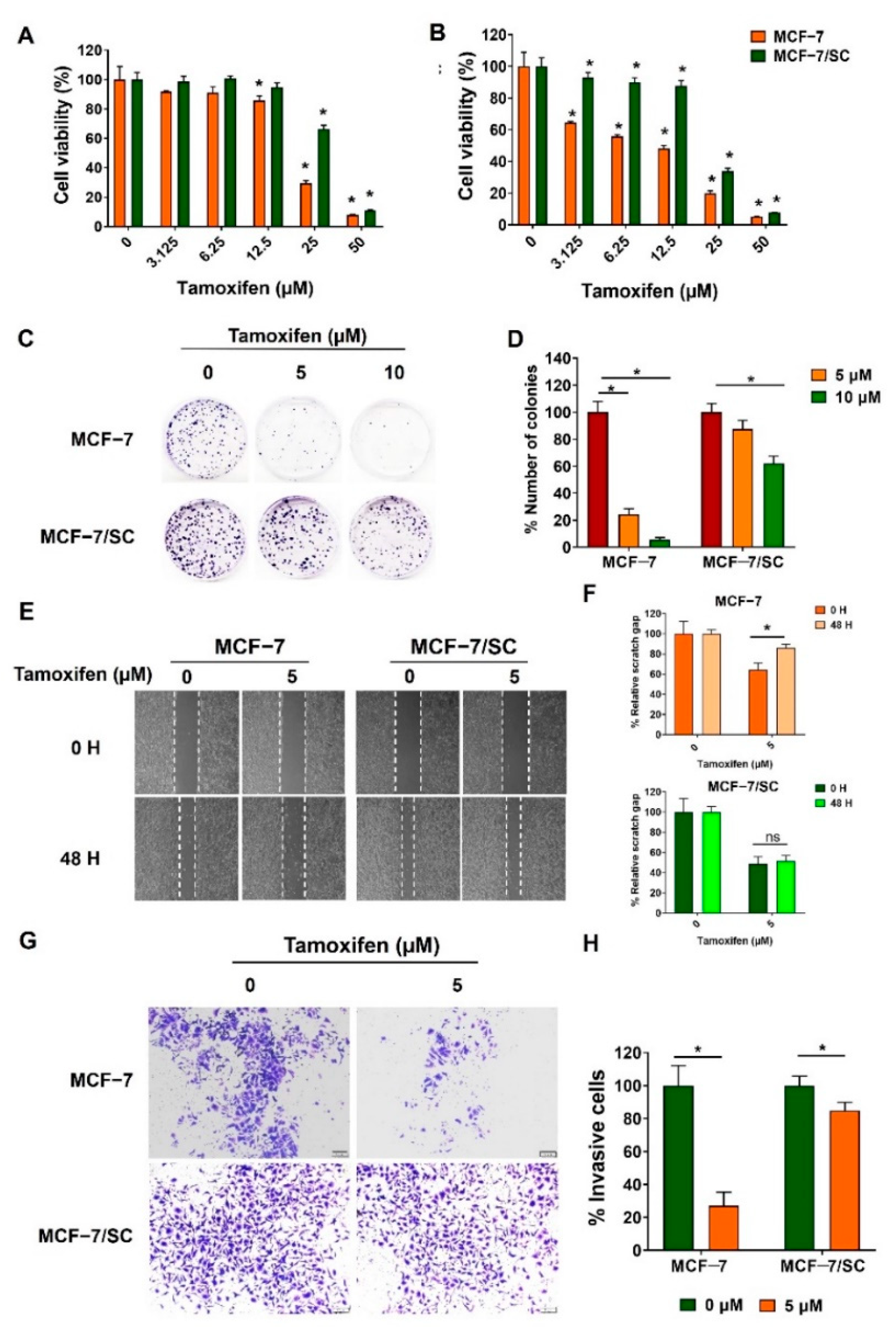
2.2. Tamoxifen Resistance of MCF−7/SC Cells Compared with the Parental MCF−7 Cells
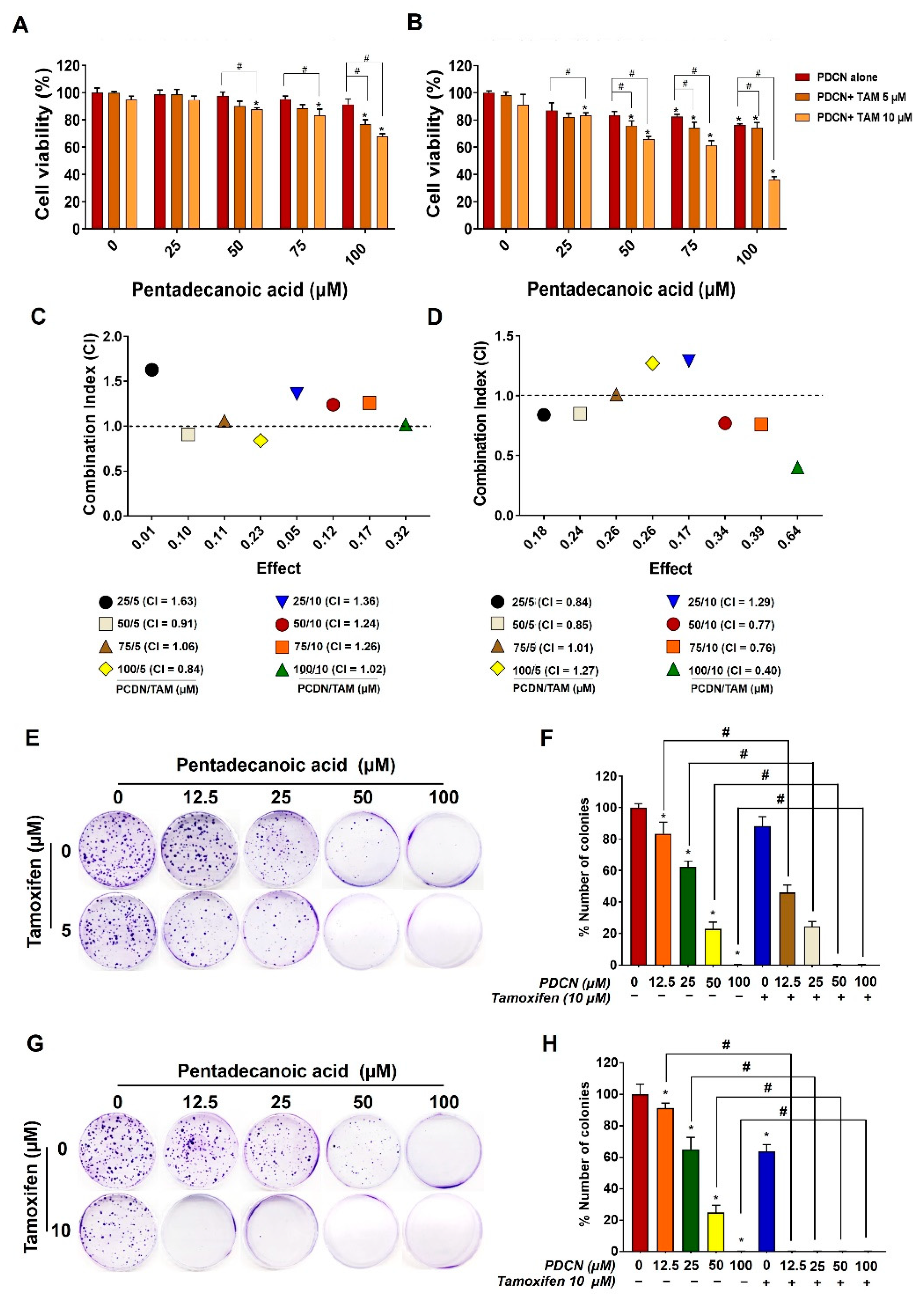
2.3. Effect of Combined Pentadecanoic acid and Tamoxifen Treatment on the Growth of MCF−7/SC Cells
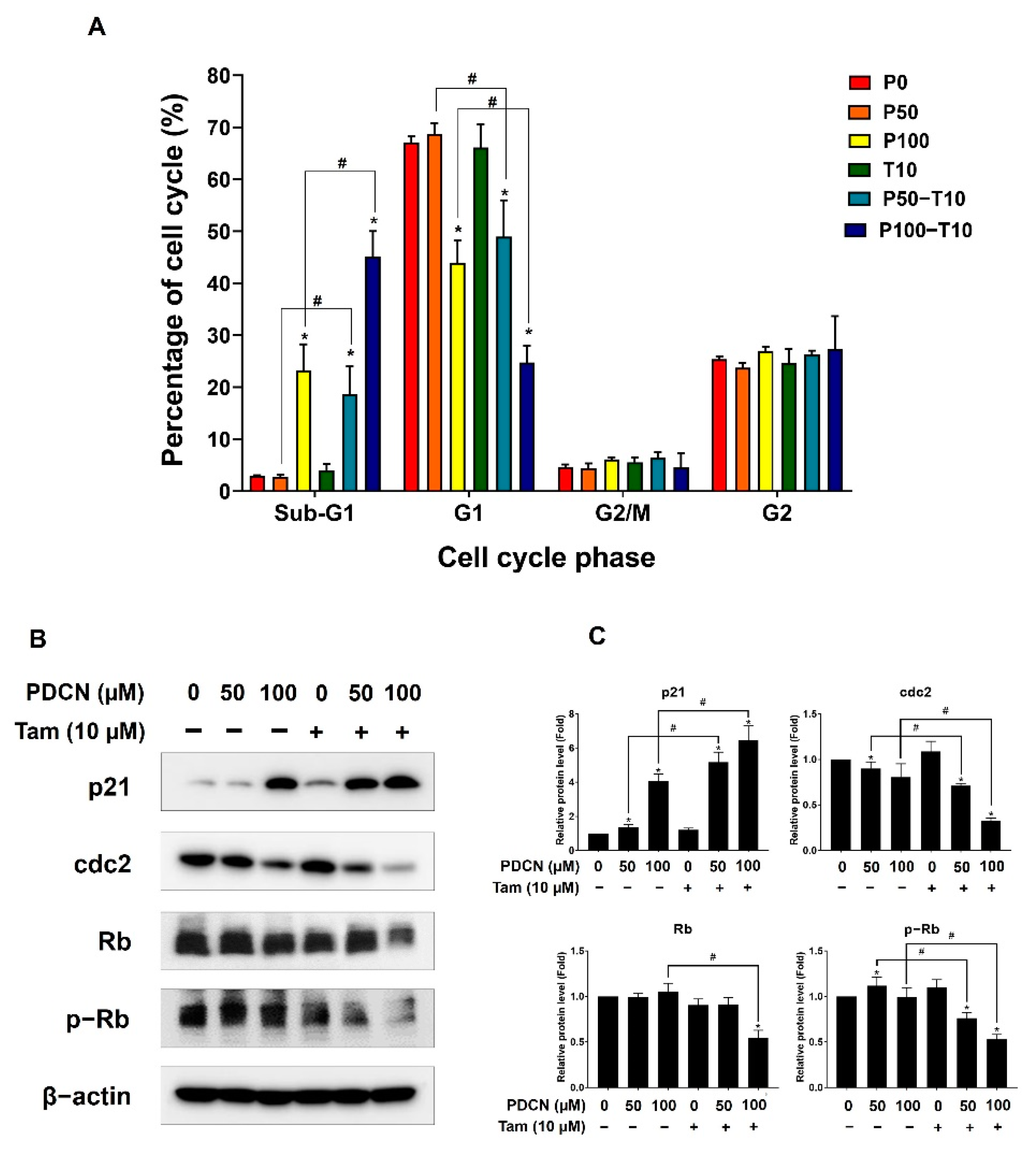
2.4. Effect of Combined Pentadecanoic Acid and Tamoxifen Treatment on Cell Cycle in MCF−7/SC
2.5. Effect of Combined Pentadecanoic Acid and Tamoxifen Treatment on Apoptosis in MCF−7/SC
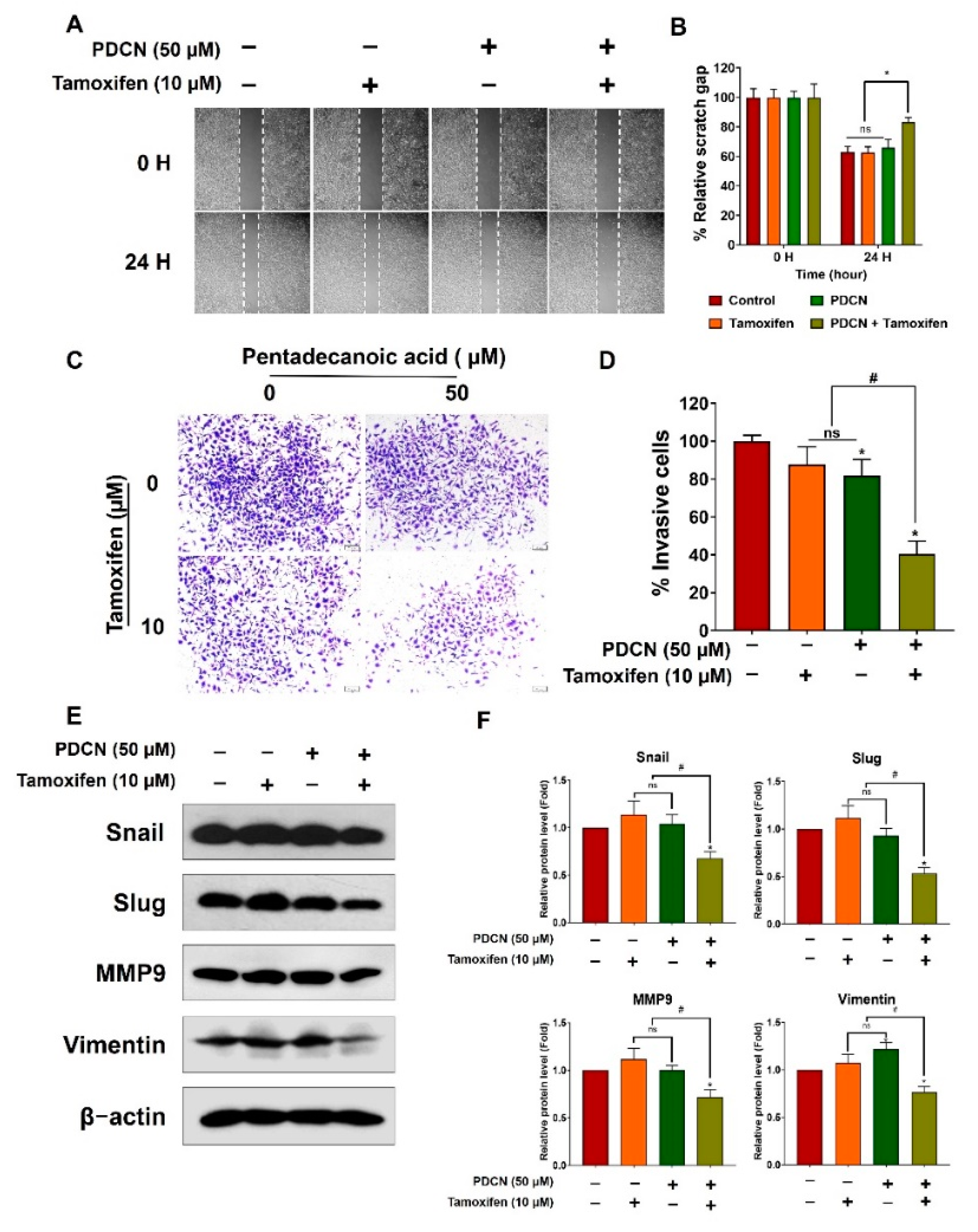
2.6. Effect of Combined Pentadecanoic Acid and Tamoxifen Treatment on Epithelial–Mesenchymal Transition
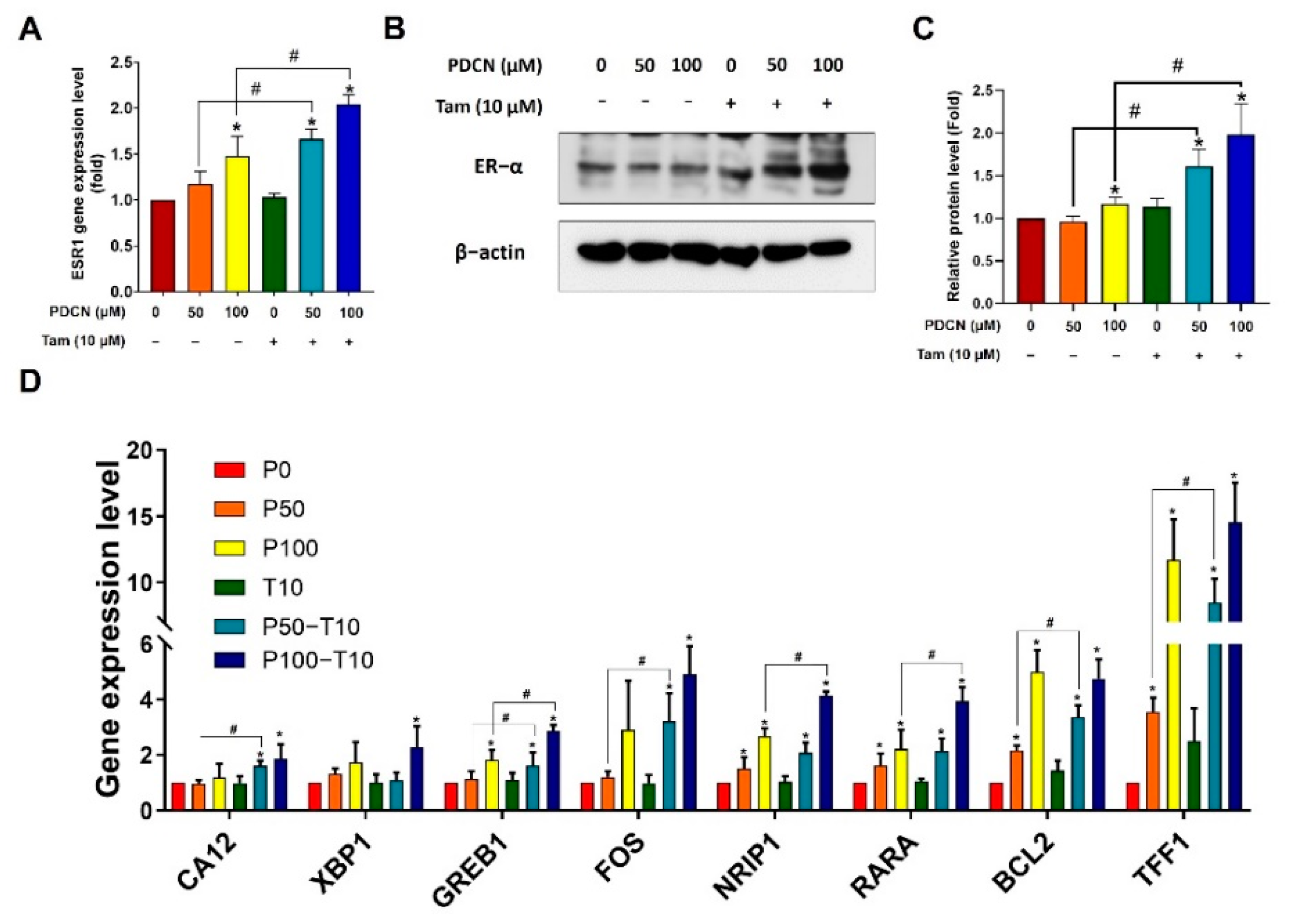
2.7. Effect of Combined Pentadecanoic Acid and Tamoxifen Treatment on ER−α Expression in MCF−7/SC Cells
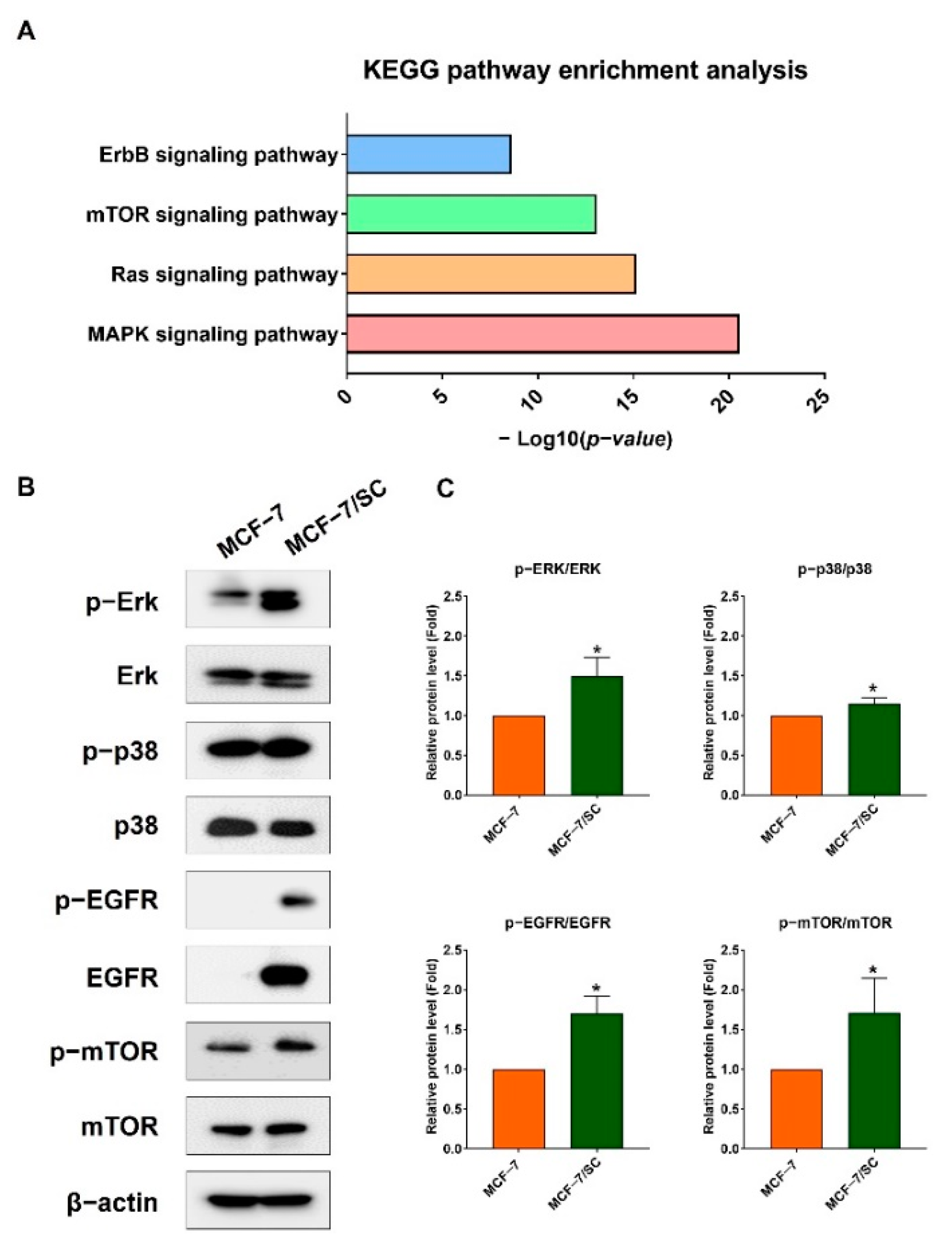
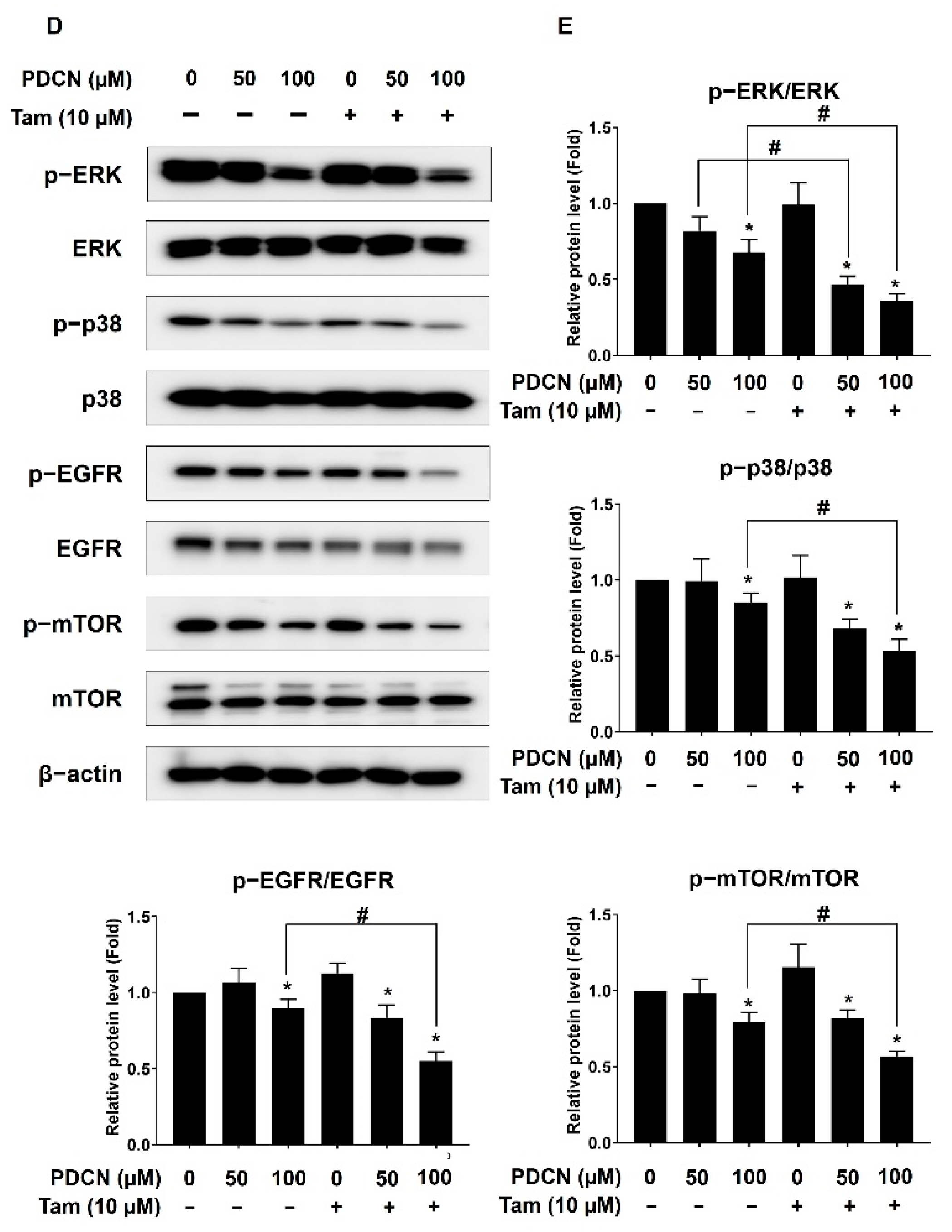
2.8. Effect of Combined Pentadecanoic Acid and Tamoxifen Treatment on Cell Survival Signaling Pathways
3. Discussion
4. Materials and Methods
4.1. Cell Lines and Cell Culture
4.2. Cell Viability Assay
4.3. Colony Formation Assay
4.4. Wound Healing Assay
4.5. Cell Invasion Assay
4.6. Flow Cytometry
4.7. Western Blotting
4.8. Quantitative Reverse Transcription PCR (qRT−PCR)
4.9. Transcriptomic Analysis
4.10. Correlation Analysis Using the Kaplan–Meier Plotter
4.11. Combination Index Analysis
4.12. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Jia, T.; Zhang, L.; Duan, Y.; Zhang, M.; Wang, G.; Zhang, J.; Zhao, Z. The differential susceptibilities of MCF−7 and MDA−MB−231 cells to the cytotoxic effects of curcumin are associated with the PI3K/Akt−SKP2−Cip/Kips pathway. Cancer Cell Int. 2014, 14, 126. [Google Scholar] [CrossRef] [PubMed]
- Holliday, D.L.; Speirs, V. Choosing the right cell line for breast cancer research. Breast Cancer Res. 2011, 13, 215. [Google Scholar] [CrossRef] [PubMed]
- Pusztai, L.; Mazouni, C.; Anderson, K.; Wu, Y.; Symmans, W.F. Molecular Classification of Breast Cancer: Limitations and Potential. Oncologist 2006, 11, 868–877. [Google Scholar] [CrossRef] [PubMed]
- Reinert, T.; Barrios, C.H. Optimal management of hormone receptor positive metastatic breast cancer in 2016. Ther. Adv. Med Oncol. 2015, 7, 304–320. [Google Scholar] [CrossRef] [PubMed]
- Rugo, H.S.; Rumble, R.B.; Macrae, E.; Barton, D.L.; Connolly, H.K.; Dickler, M.N.; Fallowfield, L.; Fowble, B.; Ingle, J.N.; Jahanzeb, M.; et al. Endocrine Therapy for Hormone Receptor–Positive Metastatic Breast Cancer: American Society of Clinical Oncology Guideline. J. Clin. Oncol. 2016, 34, 3069–3103. [Google Scholar] [CrossRef]
- Rodriguez, D.; Ramkairsingh, M.; Lin, X.; Kapoor, A.; Major, P.; Tang, D. The Central Contributions of Breast Cancer Stem Cells in Developing Resistance to Endocrine Therapy in Estrogen Receptor (ER)−Positive Breast Cancer. Cancers 2019, 11, 1028. [Google Scholar] [CrossRef]
- Ring, A.; Dowsett, M. Mechanisms of tamoxifen resistance. Endocr.−Relat. Cancer 2004, 11, 643–658. [Google Scholar] [CrossRef]
- Johnston, S.R. Acquired tamoxifen resistance in human breast cancer–potential mechanisms and clinical implications. Anti−Cancer Drugs 1997, 8, 911–930. [Google Scholar] [CrossRef]
- Roodi, N.; Bailey, L.R.; Kao, W.-Y.; Verrier, C.S.; Yee, C.J.; Dupont, W.D.; Parl, F.F. Estrogen Receptor Gene Analysis in Estrogen Receptor−Positive and Receptor−Negative Primary Breast Cancer. JNCI J. Natl. Cancer Inst. 1995, 87, 446–451. [Google Scholar] [CrossRef]
- Hervouet, E.; Cartron, P.-F.; Jouvenot, M.; Delage−Mourroux, R. Epigenetic regulation of estrogen signaling in breast cancer. Epigenetics 2013, 8, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Phillips, D.L.; Ferguson, A.T.; Nelson, W.G.; Herman, J.G.; Davidson, N.E. Synergistic activation of functional estrogen receptor (ER)−α by DNA methyltransferase and histone deacetylase inhibition in human ER−α−negative breast cancer cells. Cancer Res. 2001, 61, 7025–7029. [Google Scholar] [PubMed]
- Bovenzi, V.; Momparler, R.L. Antineoplastic action of 5−aza−2′−deoxycytidine and histone deacetylase inhibitor and their effect on the expression of retinoic acid receptor β and estrogen receptor α genes in breast carcinoma cells. Cancer Chemother. Pharmacol. 2001, 48, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Jang, E.R.; Lim, S.-J.; Lee, E.S.; Jeong, G.; Kim, T.-Y.; Bang, Y.-J.; Lee, J.-S. The histone deacetylase inhibitor trichostatin A sensitizes estrogen receptor α−negative breast cancer cells to tamoxifen. Oncogene 2004, 23, 1724–1736. [Google Scholar] [CrossRef] [PubMed]
- Nobili, S.; Lippi, D.; Witort, E.; Donnini, M.; Bausi, L.; Mini, E.; Capaccioli, S. Natural compounds for cancer treatment and prevention. Pharmacol. Res. 2009, 59, 365–378. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, B.; West, J.A.; Koulman, A. A Review of Odd−Chain Fatty Acid Metabolism and the Role of Pentadecanoic Acid (C15:0) and Heptadecanoic Acid (C17:0) in Health and Disease. Molecules 2015, 20, 2425–2444. [Google Scholar] [CrossRef] [PubMed]
- To, N.B.; Nguyen, Y.T.-K.; Moon, J.Y.; Ediriweera, M.K.; Cho, S.K. Pentadecanoic Acid, an Odd−Chain Fatty Acid, Suppresses the Stemness of MCF−7/SC Human Breast Cancer Stem−Like Cells through JAK2/STAT3 Signaling. Nutrients 2020, 12, 1663. [Google Scholar] [CrossRef] [PubMed]
- Ediriweera, M.K.; To, N.B.; Lim, Y.; Cho, S.K. Odd−chain fatty acids as novel histone deacetylase 6 (HDAC6) inhibitors. Biochimie 2021, 186, 147–156. [Google Scholar] [CrossRef]
- Ma, H.; Li, L.; Dou, G.; Wang, C.; Li, J.; He, H.; Wu, M.; Qi, H. Z−ligustilide restores tamoxifen sensitivity of ERα negative breast cancer cells by reversing MTA1/IFI16/HDACs complex mediated epigenetic repression of ERα. Oncotarget 2017, 8, 29328. [Google Scholar] [PubMed]
- Sulaiman, A.; McGarry, S.; Lam, K.M.; El−Sahli, S.; Chambers, J.; Kaczmarek, S.; Li, L.; Addison, C.; Dimitroulakos, J.; Arnaout, A. Co−inhibition of mTORC1, HDAC and ESR1α retards the growth of triple−negative breast cancer and suppresses cancer stem cells. Cell Death Dis. 2018, 9, 815. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Meeran, S.M.; Tollefsbol, T.O. Combinatorial bioactive botanicals re−sensitize tamoxifen treatment in ER−negative breast cancer via epigenetic reactivation of ERα expression. Sci. Rep. 2017, 7, 9345. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, Y.T.-K.; To, N.B.; Truong, V.N.-P.; Kim, H.Y.; Ediriweera, M.K.; Lim, Y.; Cho, S.K. Impairment of Glucose Metabolism and Suppression of Stemness in MCF−7/SC Human Breast Cancer Stem Cells by Nootkatone. Pharmaceutics 2022, 14, 906. [Google Scholar] [CrossRef] [PubMed]
- Naugler, C. Estrogen receptor testing and 10−year mortality from breast cancer: A model for determining testing strategy. J. Pathol. Inform. 2012, 3, 19. [Google Scholar] [CrossRef] [PubMed]
- Cuzick, J.; Powles, T.; Veronesi, U.; Forbes, J.; Edwards, R.; Ashley, S.; Boyle, P. Overview of the main outcomes in breast−cancer prevention trials. Lancet 2003, 361, 296–300. [Google Scholar] [CrossRef]
- Li, W.; Shi, X.; Xu, Y.; Wan, J.; Wei, S.; Zhu, R. Tamoxifen promotes apoptosis and inhibits invasion in estrogen−positive breast cancer MCF−7 cells. Mol. Med. Rep. 2017, 16, 478–484. [Google Scholar] [CrossRef]
- Li, Y.; Meeran, S.M.; Patel, S.N.; Chen, H.; Hardy, T.M.; Tollefsbol, T.O. Epigenetic reactivation of estrogen receptor−α (ERα) by genistein enhances hormonal therapy sensitivity in ERα−negative breast cancer. Mol. Cancer 2013, 12, 9. [Google Scholar] [CrossRef]
- Vermeulen, K.; Van Bockstaele, D.R.; Berneman, Z.N. The cell cycle: A review of regulation, deregulation and therapeutic targets in cancer. Cell Prolif. 2003, 36, 131–149. [Google Scholar] [CrossRef]
- Darzynkiewicz, Z.; Bruno, S.; Del Bino, G.; Gorczyca, W.; Hotz, M.A.; Lassota, P.; Traganos, F. Features of apoptotic cells measured by flow cytometry. Cytometry 1992, 13, 795–808. [Google Scholar] [CrossRef]
- Du, B.; Shim, J.S. Targeting Epithelial–Mesenchymal Transition (EMT) to Overcome Drug Resistance in Cancer. Molecules 2016, 21, 965. [Google Scholar] [CrossRef]
- Mitra, A.; Mishra, L.; Li, S. EMT, CTCs and CSCs in tumor relapse and drug−resistance. Oncotarget 2015, 6, 10697–10711. [Google Scholar] [CrossRef] [Green Version]
- Chang, M. Tamoxifen resistance in breast cancer. Biomol. Ther. 2012, 20, 256–267. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.-J.; Lee, M.-H.; Kang, H.-L.; Kim, S.-H.; Ahn, J.-R.; Na, H.; Na, T.-Y.; Kim, Y.N.; Seong, J.K.; Lee, M.-O. Differential regulation of estrogen receptor α expression in breast cancer cells by metastasis−associated protein 1. Cancer Res. 2014, 74, 1484–1494. [Google Scholar] [CrossRef] [PubMed]
- Martinelli, E.; Morgillo, F.; Troiani, T.; Ciardiello, F. Cancer resistance to therapies against the EGFR−RAS−RAF pathway: The role of MEK. Cancer Treat. Rev. 2017, 53, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Bianco, R.; Garofalo, S.; Rosa, R.; Damiano, V.; Gelardi, T.; Daniele, G.; Marciano, R.; Ciardiello, F.; Tortora, G. Inhibition of mTOR pathway by everolimus cooperates with EGFR inhibitors in human tumours sensitive and resistant to anti−EGFR drugs. Br. J. Cancer 2008, 98, 923–930. [Google Scholar] [CrossRef] [PubMed]
- Polyak, K. Heterogeneity in breast cancer. J. Clin. Investig. 2011, 121, 3786–3788. [Google Scholar] [CrossRef]
- Perou, C.M.; Sørlie, T.; Eisen, M.B.; Van De Rijn, M.; Jeffrey, S.S.; Rees, C.A.; Pollack, J.R.; Ross, D.T.; Johnsen, H.; Akslen, L.A.; et al. Molecular portraits of human breast tumours. Nature 2000, 406, 747–752. [Google Scholar] [CrossRef]
- Shao, W.; Brown, M. Advances in estrogen receptor biology: Prospects for improvements in targeted breast cancer therapy. Breast Cancer Res. 2003, 6, 39. [Google Scholar] [CrossRef]
- Clarke, R.; Liu, M.C.; Bouker, K.B.; Gu, Z.; Lee, R.Y.; Zhu, Y.; Skaar, T.C.; Gomez, B.; O’Brien, K.; Wang, Y.; et al. Antiestrogen resistance in breast cancer and the role of estrogen receptor signaling. Oncogene 2003, 22, 7316–7339. [Google Scholar] [CrossRef]
- Castrellon, A.B. Novel strategies to improve the endocrine therapy of breast cancer. Oncol. Rev. 2017, 11, 323. [Google Scholar] [CrossRef]
- Brinkman, J.A.; El−Ashry, D. ER Re−expression and Re−sensitization to Endocrine Therapies in ER−negative Breast Cancers. J. Mammary Gland Biol. Neoplasia 2009, 14, 67–78. [Google Scholar] [CrossRef]
- Sun, W.; Sanderson, P.E.; Zheng, W. Drug combination therapy increases successful drug repositioning. Drug Discov. Today 2016, 21, 1189–1195. [Google Scholar] [CrossRef] [PubMed]
- Pucci, B.; Kasten, M.; Giordano, A. Cell Cycle and Apoptosis. Neoplasia 2000, 2, 291–299. [Google Scholar] [CrossRef]
- Hiscox, S.; Jiang, W.G.; Obermeier, K.; Taylor, K.; Morgan, L.; Burmi, R.; Barrow, D.; Nicholson, R.I. Tamoxifen resistance in MCF7 cells promotes EMT−like behaviour and involves modulation of β−catenin phosphorylation. Int. J. Cancer 2007, 118, 290–301. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.-W.; Chen, M.-N.; Wei, X.-L.; Li, Y.-C.; Li, H.-Y.; Li, J.-W.; Du, C.-W.; Man, K.; Zhang, G.-J. The zinc−finger transcriptional factor Slug transcriptionally downregulates ERα by recruiting lysine−specific demethylase 1 in human breast cancer. Oncogenesis 2017, 6, e330. [Google Scholar] [CrossRef] [PubMed]
- Al Saleh, S.; Al−Mulla, F.; Luqmani, Y.A. Estrogen Receptor Silencing Induces Epithelial to Mesenchymal Transition in Human Breast Cancer Cells. PLoS ONE 2011, 6, e20610. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Zhao, X.; Xiao, Q.; Liu, Q.; Ding, K.; Yu, F.; Zhang, R.; Zhu, T.; Ge, G. Snail and Slug mediate tamoxifen resistance in breast cancer cells through activation of EGFR–ERK independent of epithelial–mesenchymal transition. J. Mol. Cell Biol. 2014, 6, 352–354. [Google Scholar] [CrossRef]
- Zhang, J.; Zhou, C.; Jiang, H.; Liang, L.; Shi, W.; Zhang, Q.; Sun, P.; Xiang, R.; Wang, Y.; Yang, S. ZEB1 induces ER−α promoter hypermethylation and confers antiestrogen resistance in breast cancer. Cell Death Dis. 2017, 8, e2732. [Google Scholar] [CrossRef] [PubMed]
- Dhasarathy, A.; Kajita, M.; Wade, P.A. The Transcription Factor Snail Mediates Epithelial to Mesenchymal Transitions by Repression of Estrogen Receptor−α. Mol. Endocrinol. 2007, 21, 2907–2918. [Google Scholar] [CrossRef]
- Geng, C.; Li, J.; Ding, F.; Wu, G.; Yang, Q.; Sun, Y.; Zhang, Z.; Dong, T.; Tian, X. Curcumin suppresses 4−hydroxytamoxifen resistance in breast cancer cells by targeting SLUG/Hexokinase 2 pathway. Biochem. Biophys. Res. Commun. 2016, 473, 147–153. [Google Scholar] [CrossRef]
- Chamard−Jovenin, C.; Jung, A.C.; Chesnel, A.; Abecassis, J.; Flament, S.; Ledrappier, S.; Macabre, C.; Boukhobza, T.; Dumond, H. From ERα66 to ERα36: A generic method for validating a prognosis marker of breast tumor progression. BMC Syst. Biol. 2015, 9, 28. [Google Scholar] [CrossRef] [Green Version]
- Creighton, C.J.; Hilger, A.M.; Murthy, S.; Rae, J.M.; Chinnaiyan, A.M.; El−Ashry, D. Activation of mitogen−activated protein kinase in estrogen receptor α–positive breast cancer cells in vitro induces an in vivo molecular phenotype of estrogen receptor α–negative human breast tumors. Cancer Res. 2006, 66, 3903–3911. [Google Scholar] [CrossRef] [PubMed]
- Bayliss, J.; Hilger, A.; Vishnu, P.; Diehl, K.; El−Ashry, D. Reversal of the Estrogen Receptor–Negative Phenotype in Breast Cancer and Restoration of Antiestrogen Response. Clin. Cancer Res. 2007, 13, 7029–7036. [Google Scholar] [CrossRef] [PubMed]
- Sainsbury, J.; Sherbet, G.; Farndon, J.; Harris, A. EPIDERMAL−GROWTH−FACTOR RECEPTORS AND OESTROGEN RECEPTORS IN HUMAN BREAST CANCER. Lancet 1985, 325, 364–366. [Google Scholar] [CrossRef]
- Stoica, A.; Saceda, M.; Doraiswamy, V.L.; Coleman, C.; Martin, M.B. Regulation of estrogen receptor−alpha gene expression by epidermal growth factor. J. Endocrinol. 2000, 165, 371–378. [Google Scholar] [CrossRef]
- Benz, C.C.; Scott, G.K.; Sarup, J.C.; Johnson, R.M.; Tripathy, D.; Coronado, E.; Shepard, H.M.; Osborne, C.K. Estrogen−dependent, tamoxifen−resistant tumorigenic growth of MCF−7 cells transfected with HER2/neu. Breast Cancer Res. Treat. 1992, 24, 85–95. [Google Scholar] [CrossRef]
- Xie, F.; Li, Z.; Wang, N.; Fang, J.; Huang, J.; Tian, F.; Li, C. Role of PKC−ERK signaling in tamoxifen−induced apoptosis and tamoxifen resistance in human breast cancer cells. Oncol. Rep. 2012, 27, 1879–1886. [Google Scholar] [CrossRef]
- Kelloff, G.J.; Lippman, S.M.; Dannenberg, A.J.; Sigman, C.C.; Pearce, H.L.; Reid, B.J.; Szabo, E.; Jordan, V.C.; Spitz, M.R.; Mills, G.B.; et al. Progress in Chemoprevention Drug Development: The Promise of Molecular Biomarkers for Prevention of Intraepithelial Neoplasia and Cancer—A Plan to Move Forward. Clin. Cancer Res. 2006, 12, 3661–3697. [Google Scholar] [CrossRef]
- Knowlden, J.M.; Hutcheson, I.R.; Jones, H.E.; Madden, T.; Gee, J.M.; Harper, M.E.; Barrow, D.; Wakeling, A.E.; Nicholson, R.I. Elevated levels of epidermal growth factor receptor/c−erbB2 heterodimers mediate an autocrine growth regulatory pathway in tamoxifen−resistant MCF−7 cells. Endocrinology 2003, 144, 1032–1044. [Google Scholar] [CrossRef]
- Degraffenried, L.A.; Friedrichs, W.E.; Russell, D.H.; Donzis, E.J.; Middleton, A.K.; Silva, J.M.; Roth, R.A.; Hidalgo, M. Inhibition of mTOR Activity Restores Tamoxifen Response in Breast Cancer Cells with Aberrant Akt Activity. Clin. Cancer Res. 2004, 10, 8059–8067. [Google Scholar] [CrossRef]
- Baylin, S.; Bestor, T.H. Altered methylation patterns in cancer cell genomes: Cause or consequence? Cancer Cell 2002, 1, 299–305. [Google Scholar] [CrossRef] [Green Version]
- Dworkin, A.M.; Huang, T.H.-M.; Toland, A.E. Epigenetic alterations in the breast: Implications for breast cancer detection, prognosis and treatment. Semin. Cancer Biol. 2009, 19, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yuan, Y.-Y.; Meeran, S.M.; O Tollefsbol, T. Synergistic epigenetic reactivation of estrogen receptor−α (ERα) by combined green tea polyphenol and histone deacetylase inhibitor in ERα−negative breast cancer cells. Mol. Cancer 2010, 9, 274. [Google Scholar] [CrossRef] [PubMed]
- Asthana, J.; Kapoor, S.; Mohan, R.; Panda, D. Inhibition of HDAC6 Deacetylase Activity Increases Its Binding with Microtubules and Suppresses Microtubule Dynamic Instability in MCF−7 Cells. J. Biol. Chem. 2013, 288, 22516–22526. [Google Scholar] [CrossRef] [PubMed]
- Oba, T.; Ono, M.; Matoba, H.; Uehara, T.; Hasegawa, Y.; Ito, K.-I. −I. HDAC6 inhibition enhances the anti−tumor effect of eribulin through tubulin acetylation in triple−negative breast cancer cells. Breast Cancer Res. Treat. 2021, 186, 37–51. [Google Scholar] [CrossRef] [PubMed]
- Azuma, K.; Urano, T.; Horie−Inoue, K.; Hayashi, S.-I.; Sakai, R.; Ouchi, Y.; Inoue, S. Association of Estrogen Receptor α and Histone Deacetylase 6 Causes Rapid Deacetylation of Tubulin in Breast Cancer Cells. Cancer Res. 2009, 69, 2935–2940. [Google Scholar] [CrossRef]
- Van Phuc, P.; Keng, S.C.; Nguyet, N.T.M.; Thuy, D.T.; Ngoc, P.K. Isolation and comparison of tumorigenicity of different cell populations from the MCF−7 breast cancer cell line based on CD44 and CD24 markers. Vietnam. J. Sci. Technol. 2011, 9, 13–19. [Google Scholar]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real−time quantitative PCR and the 2(−Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Chou, T.-C. Drug combination studies and their synergy quantification using the Chou−Talalay method. Cancer Res. 2010, 70, 440–446. [Google Scholar] [CrossRef] [Green Version]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
To, N.B.; Truong, V.N.-P.; Ediriweera, M.K.; Cho, S.K. Effects of Combined Pentadecanoic Acid and Tamoxifen Treatment on Tamoxifen Resistance in MCF−7/SC Breast Cancer Cells. Int. J. Mol. Sci. 2022, 23, 11340. https://doi.org/10.3390/ijms231911340
To NB, Truong VN-P, Ediriweera MK, Cho SK. Effects of Combined Pentadecanoic Acid and Tamoxifen Treatment on Tamoxifen Resistance in MCF−7/SC Breast Cancer Cells. International Journal of Molecular Sciences. 2022; 23(19):11340. https://doi.org/10.3390/ijms231911340
Chicago/Turabian StyleTo, Ngoc Bao, Vi Nguyen-Phuong Truong, Meran Keshawa Ediriweera, and Somi Kim Cho. 2022. "Effects of Combined Pentadecanoic Acid and Tamoxifen Treatment on Tamoxifen Resistance in MCF−7/SC Breast Cancer Cells" International Journal of Molecular Sciences 23, no. 19: 11340. https://doi.org/10.3390/ijms231911340





