Selenium-Stimulated Exosomes Enhance Wound Healing by Modulating Inflammation and Angiogenesis
Abstract
:1. Introduction
2. Results
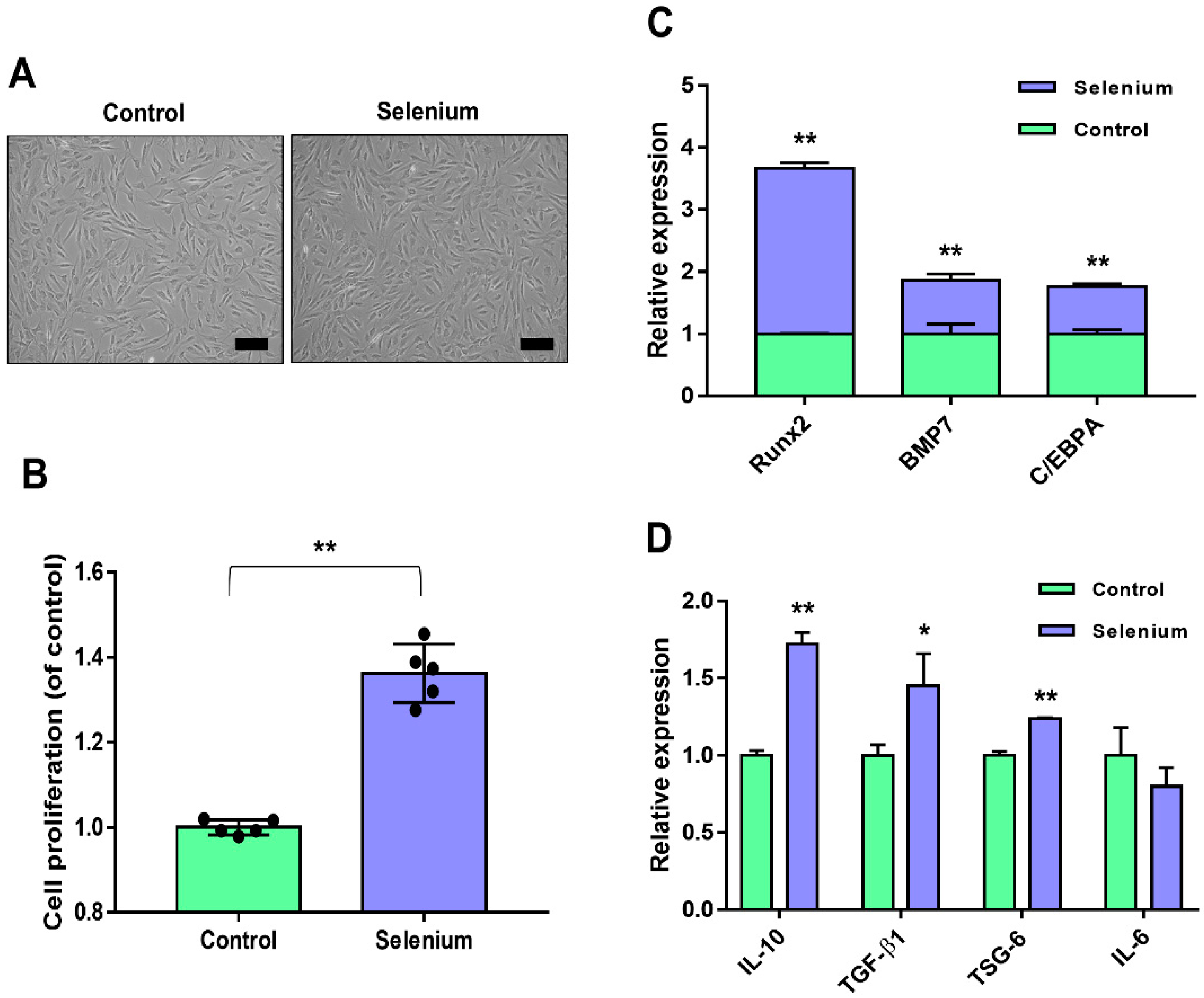
2.1. Selenium Treatment Improved AdMSC Functionality
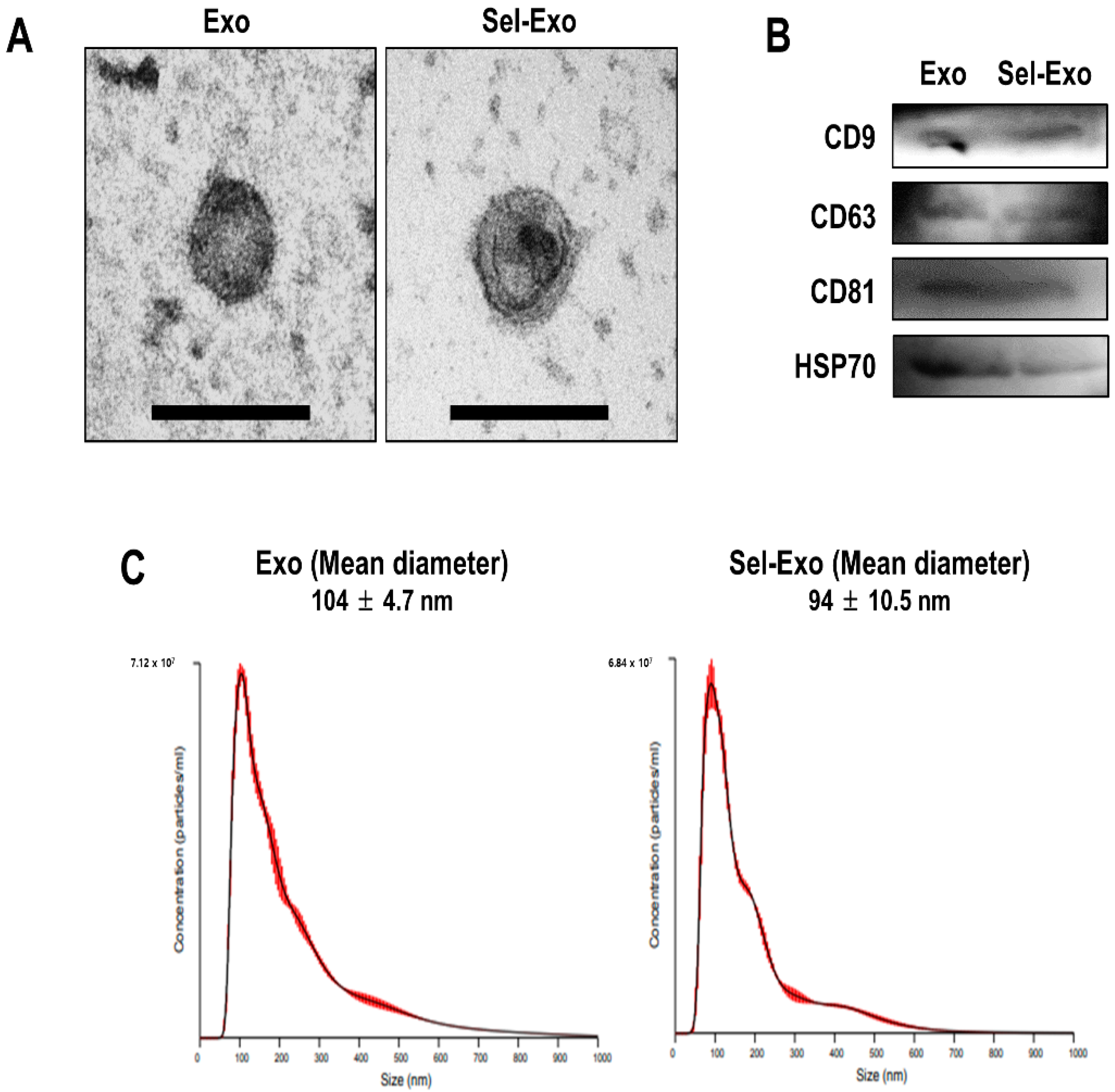
2.2. AdMSC-Derived Exosomes and Selenium-Stimulated Exosomes Showed Similar Characteristics
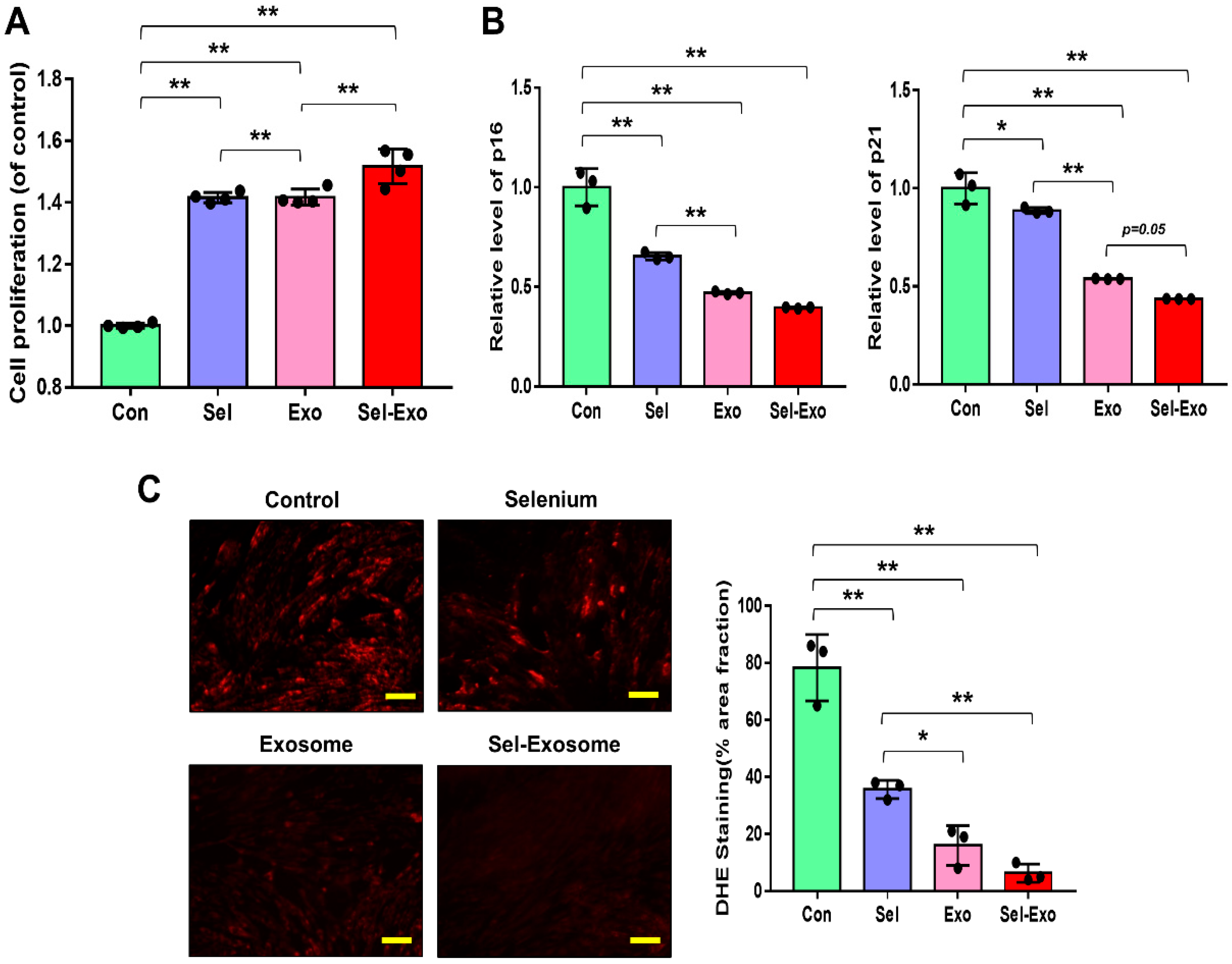
2.3. Selenium-Treated Exosomes Strongly Promoted Cell Proliferation by Reducing p16, p21, and Intracellular ROS Accumulation
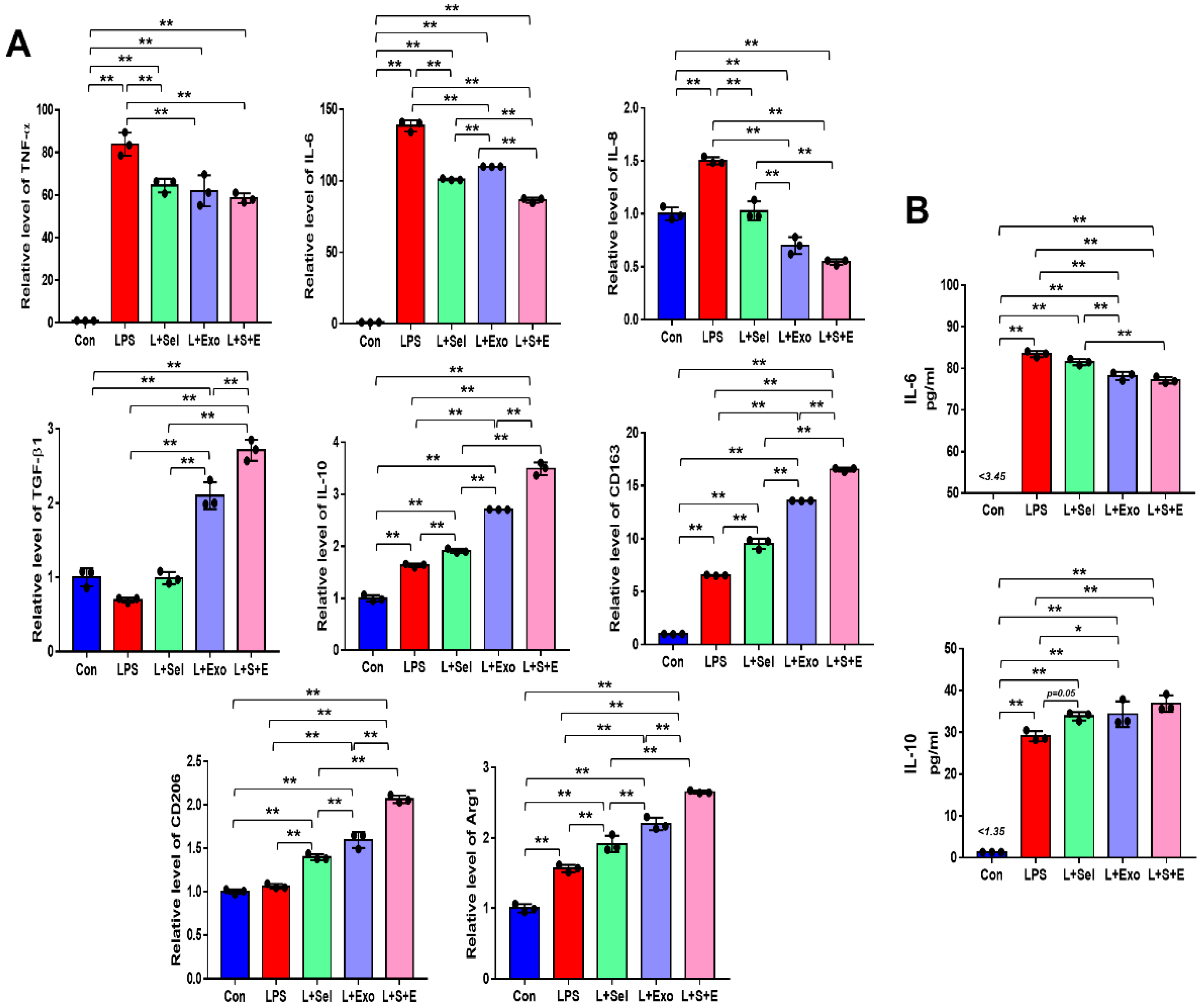
2.4. Selenium-Treated Exosomes Suppressed the Inflammatory Response by Regulating Pro-Inflammatory and Anti-Inflammatory Factors
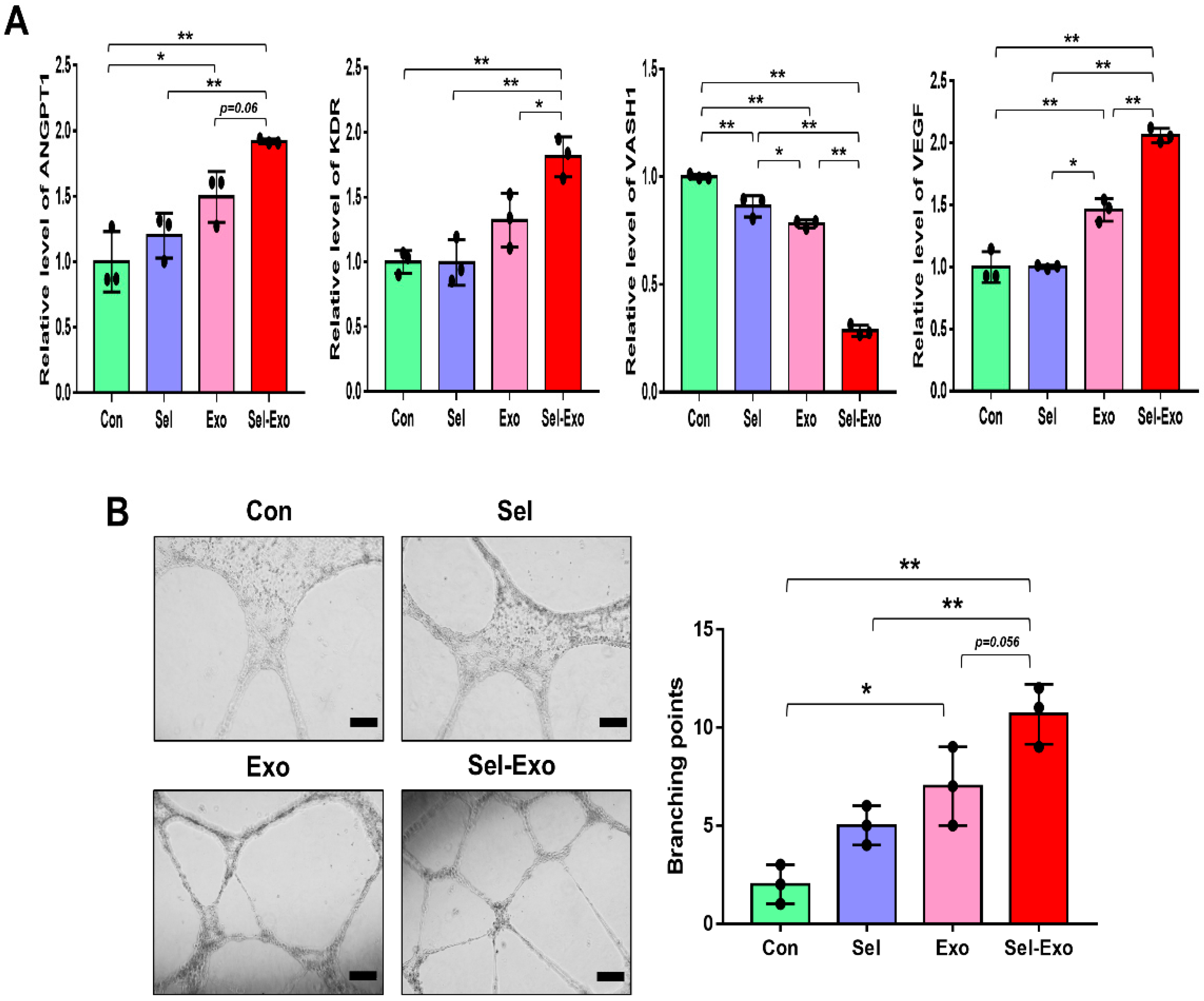
2.5. Selenium-Treated Exosomes Accelerated Endothelial Cell Angiogenesis In Vitro
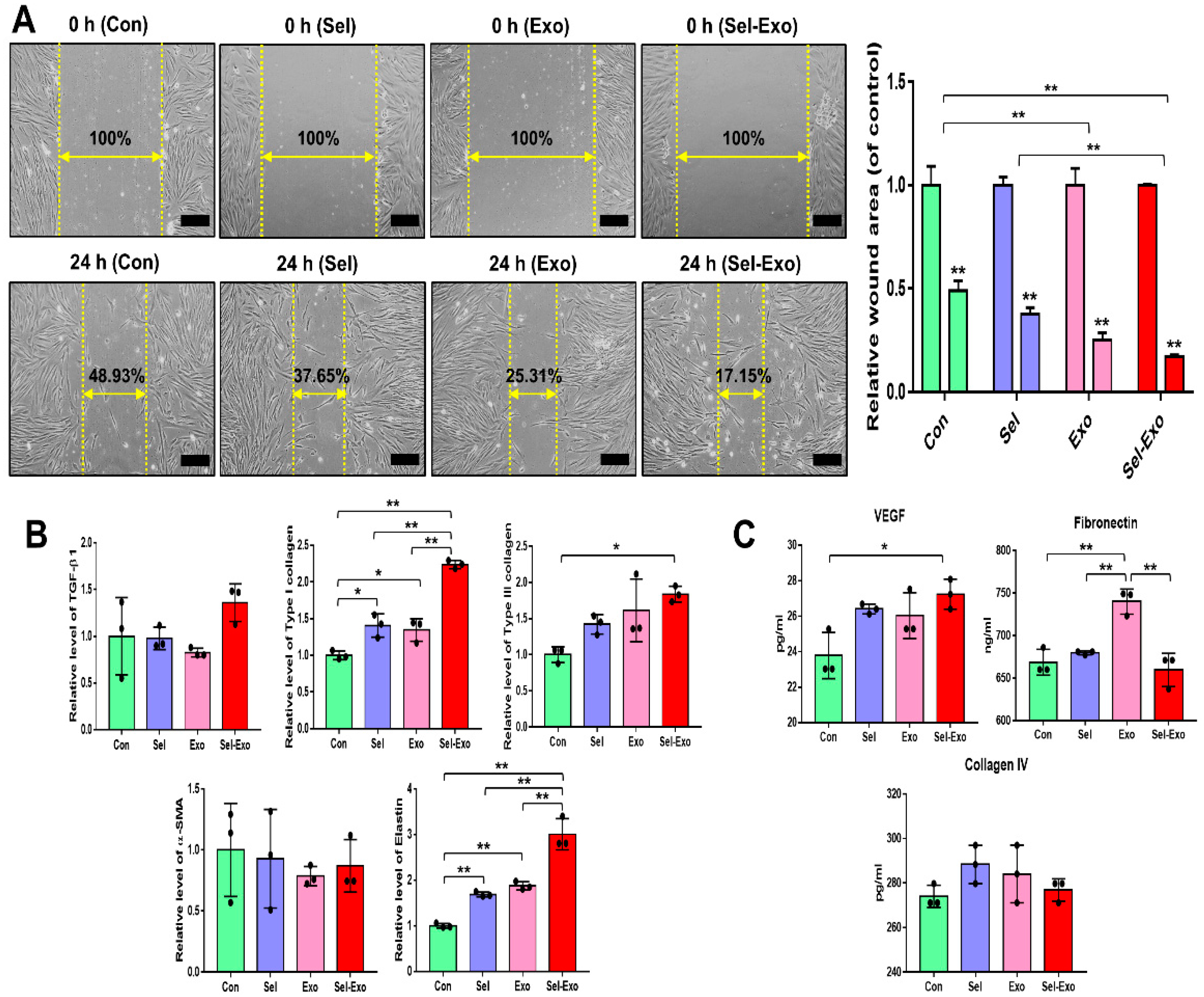
2.6. Selenium-Treated Exosomes Promoted Migration and Wound Closure by Upregulating the Factors Associated with Remodelling
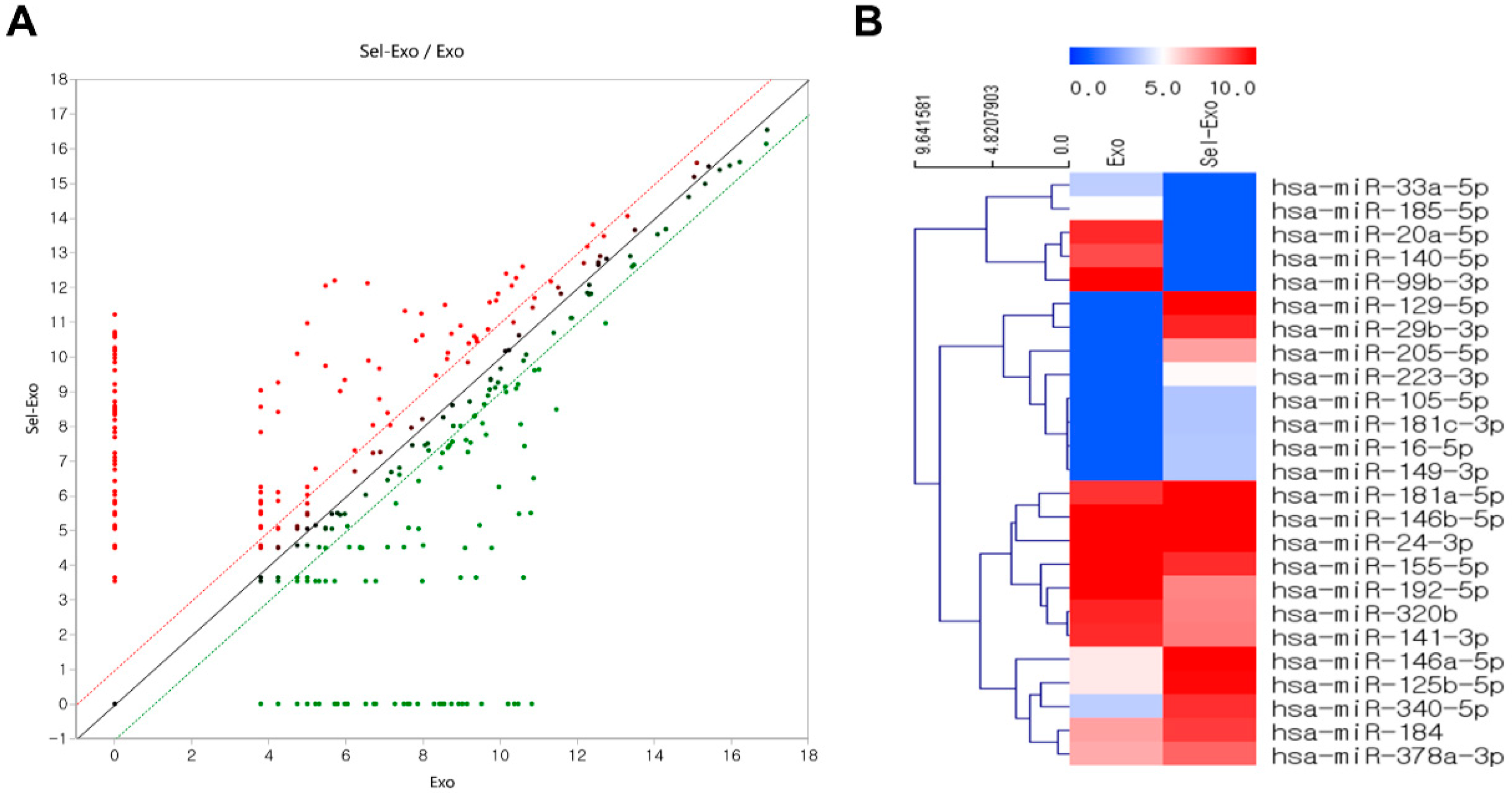
2.7. Selenium-Treated Exosomes Contained Many Beneficial miRNAs for Wound Healing
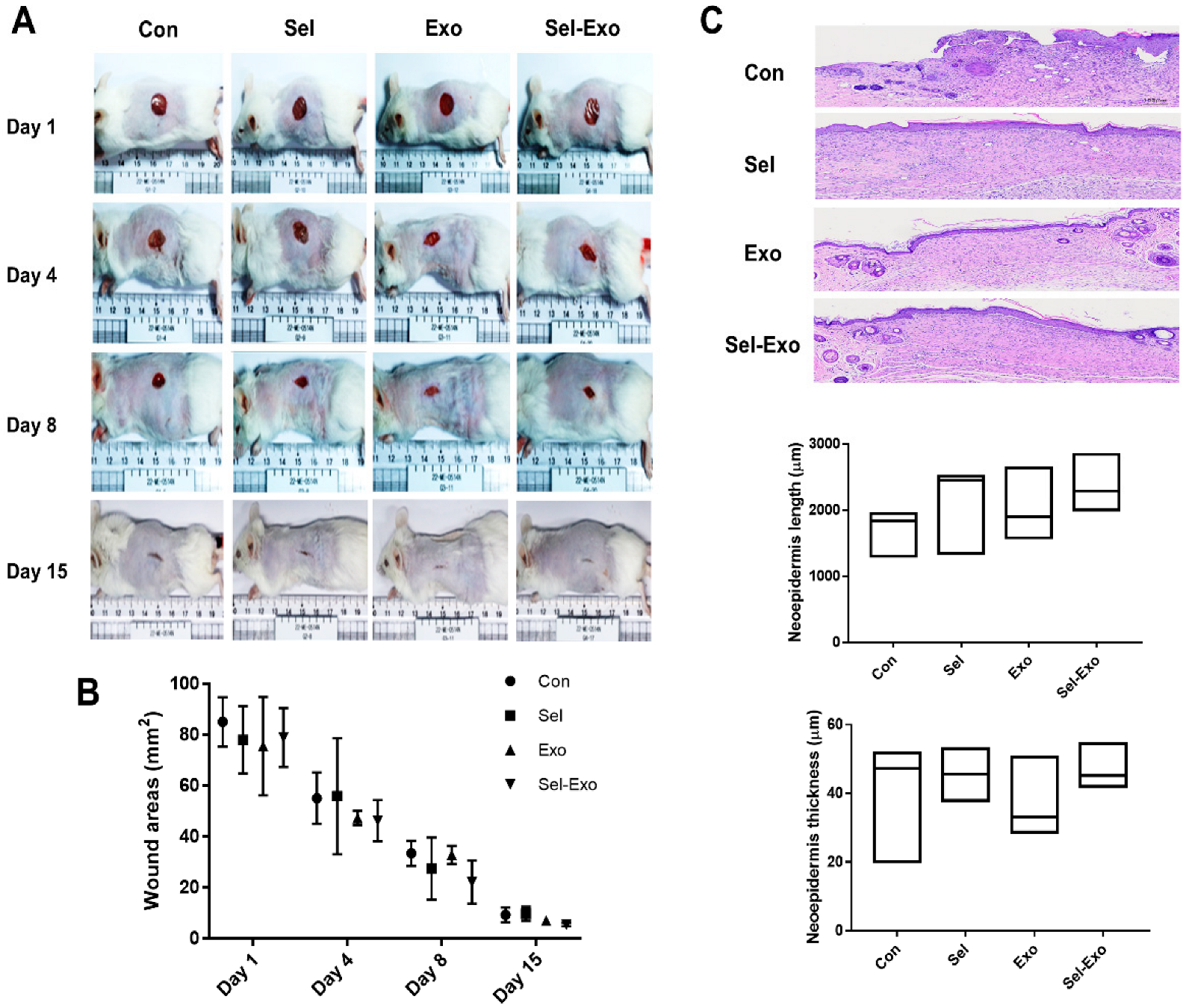
2.8. Selenium-Treated Exosomes Improved Wound Healing In Vivo
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Cell Proliferation Assay
4.3. Quantitative Reverse Transcription PCR (qRT-PCR)
4.4. Exosome Isolation and Characterization
4.5. Western Blotting
4.6. ROS Analysis
4.7. Multiplex Supernatant Cytokine Assay
4.8. Tube Formation Assay
4.9. In Vitro Wound Healing Assay
4.10. In Vivo Wound Healing Assay
4.11. Library Preparation and Sequencing
4.12. Statistical Analysis
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rodriguez-Menocal, L.; Salgado, M.; Ford, D.; Van Badiavas, E. Stimulation of skin and wound fibroblast migration by mesenchymal stem cells derived from normal donors and chronic wound patients. Stem Cells Transl. Med. 2012, 1, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Falanga, V. Wound healing and its impairment in the diabetic foot. Lancet 2005, 366, 1736–1743. [Google Scholar] [CrossRef]
- Zhao, P.; Sui, B.D.; Liu, N.; Lv, Y.J.; Zheng, C.X.; Lu, Y.B.; Huang, W.T.; Zhou, C.H.; Chen, J.; Pang, D.L.; et al. Anti-aging pharmacology in cutaneous wound healing: Effects of metformin, resveratrol, and rapamycin by local application. Aging Cell 2017, 16, 1083–1093. [Google Scholar] [CrossRef] [PubMed]
- Hassanshahi, A.; Hassanshahi, M.; Khabbazi, S.; Hosseini-Khah, Z.; Peymanfar, Y.; Ghalamkari, S.; Su, Y.W.; Xian, C.J. Adipose-derived stem cells for wound healing. J. Cell. Physiol. 2019, 234, 7903–7914. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.O.; Han, J.W.; Kim, J.M.; Cho, H.J.; Park, C.; Lee, N.; Kim, D.W.; Yoon, Y.S. Malignant tumor formation after transplantation of short-term cultured bone marrow mesenchymal stem cells in experimental myocardial infarction and diabetic neuropathy. Circ. Res. 2011, 108, 1340–1347. [Google Scholar] [CrossRef]
- Toma, C.; Wagner, W.R.; Bowry, S.; Schwartz, A.; Villanueva, F. Fate of culture-expanded mesenchymal stem cells in the microvasculature: In vivo observations of cell kinetics. Circ. Res. 2009, 104, 398–402. [Google Scholar] [CrossRef]
- Ratajczak, M.Z.; Kucia, M.; Jadczyk, T.; Greco, N.J.; Wojakowski, W.; Tendera, M.; Ratajczak, J. Pivotal role of paracrine effects in stem cell therapies in regenerative medicine: Can we translate stem cell-secreted paracrine factors and microvesicles into better therapeutic strategies? Leukemia 2012, 26, 1166–1173. [Google Scholar] [CrossRef]
- Nikfarjam, S.; Rezaie, J.; Zolbanin, N.M.; Jafari, R. Mesenchymal stem cell derived-exosomes: A modern approach in translational medicine. J. Transl. Med. 2020, 18, 449. [Google Scholar] [CrossRef]
- Phinney, D.G.; Pittenger, M.F. Concise review: Msc-derived exosomes for cell-free therapy. Stem Cells 2017, 35, 851–858. [Google Scholar] [CrossRef]
- Lai, R.C.; Chen, T.S.; Lim, S.K. Mesenchymal stem cell exosome: A novel stem cell-based therapy for cardiovascular disease. Regen. Med. 2011, 6, 481–492. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Bi, J.; Huang, J.; Tang, Y.; Du, S.; Li, P. Exosome: A review of its classification, isolation techniques, storage, diagnostic and targeted therapy applications. Int. J. Nanomed. 2020, 15, 6917–6934. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Jia, L.; Zheng, Y.; Li, W. Salivary exosomes: Emerging roles in systemic disease. Int. J. Biol. Sci. 2018, 14, 633–643. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Wang, M.; Gong, A.; Zhang, X.; Wu, X.; Zhu, Y.; Shi, H.; Wu, L.; Zhu, W.; Qian, H.; et al. Hucmsc-exosome mediated-wnt4 signaling is required for cutaneous wound healing. Stem Cells 2015, 33, 2158–2168. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chen, C.; Hu, B.; Niu, X.; Liu, X.; Zhang, G.; Zhang, C.; Li, Q.; Wang, Y. Exosomes derived from human endothelial progenitor cells accelerate cutaneous wound healing by promoting angiogenesis through erk1/2 signaling. Int. J. Biol. Sci. 2016, 12, 1472–1487. [Google Scholar] [CrossRef]
- Heo, J.S.; Kim, S. Human adipose mesenchymal stem cells modulate inflammation and angiogenesis through exosomes. Sci. Rep. 2022, 12, 2776. [Google Scholar] [CrossRef]
- Heo, J.S.; Kim, H.O.; Song, S.Y.; Lew, D.H.; Choi, Y.; Kim, S. Poly-l-lysine prevents senescence and augments growth in culturing mesenchymal stem cells ex vivo. BioMed Res. Int. 2016, 2016, 8196078. [Google Scholar] [CrossRef]
- Hu, C.; Li, L. Preconditioning influences mesenchymal stem cell properties in vitro and in vivo. J. Cell Mol. Med. 2018, 22, 1428–1442. [Google Scholar] [CrossRef] [PubMed]
- Jun, E.K.; Zhang, Q.; Yoon, B.S.; Moon, J.H.; Lee, G.; Park, G.; Kang, P.J.; Lee, J.H.; Kim, A.; You, S. Hypoxic conditioned medium from human amniotic fluid-derived mesenchymal stem cells accelerates skin wound healing through tgf-β/smad2 and pi3k/akt pathways. Int. J. Mol. Sci. 2014, 15, 605–628. [Google Scholar] [CrossRef]
- Qazi, T.H.; Mooney, D.J.; Duda, G.N.; Geissler, S. Biomaterials that promote cell-cell interactions enhance the paracrine function of mscs. Biomaterials 2017, 140, 103–114. [Google Scholar] [CrossRef]
- Qiu, Y.; Guo, J.; Mao, R.; Chao, K.; Chen, B.L.; He, Y.; Zeng, Z.R.; Zhang, S.H.; Chen, M.H. Tlr3 preconditioning enhances the therapeutic efficacy of umbilical cord mesenchymal stem cells in tnbs-induced colitis via the tlr3-jagged-1-notch-1 pathway. Mucosal Immunol. 2017, 10, 727–742. [Google Scholar] [CrossRef] [Green Version]
- Ding, J.; Wang, X.; Chen, B.; Zhang, J.; Xu, J. Exosomes derived from human bone marrow mesenchymal stem cells stimulated by deferoxamine accelerate cutaneous wound healing by promoting angiogenesis. BioMed Res. Int. 2019, 2019, 9742765. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Yu, M.; Xie, D.; Wang, L.; Ye, C.; Zhu, Q.; Liu, F.; Yang, L. Melatonin-stimulated msc-derived exosomes improve diabetic wound healing through regulating macrophage m1 and m2 polarization by targeting the pten/akt pathway. Stem Cell Res. Ther. 2020, 11, 259. [Google Scholar] [CrossRef]
- Shanu, A.; Groebler, L.; Kim, H.B.; Wood, S.; Weekley, C.M.; Aitken, J.B.; Harris, H.H.; Witting, P.K. Selenium inhibits renal oxidation and inflammation but not acute kidney injury in an animal model of rhabdomyolysis. Antioxid. Redox Signal. 2013, 18, 756–769. [Google Scholar] [CrossRef]
- Park, S.H.; Kim, J.H.; Nam, S.W.; Kim, B.W.; Kim, G.Y.; Kim, W.J.; Choi, Y.H. Selenium improves stem cell potency by stimulating the proliferation and active migration of 3t3-l1 preadipocytes. Int. J. Oncol. 2014, 44, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Lee, J.H.; Yoon, B.S.; Jun, E.K.; Lee, G.; Kim, I.Y.; You, S. Additive effect of bfgf and selenium on expansion and paracrine action of human amniotic fluid-derived mesenchymal stem cells. Stem Cell Res. Ther. 2018, 9, 293. [Google Scholar] [CrossRef]
- Heo, J.S.; Kim, S.; Yang, C.E.; Choi, Y.; Song, S.Y.; Kim, H.O. Human adipose mesenchymal stem cell-derived exosomes: A key player in wound healing. Tissue Eng. Regen. Med. 2021, 18, 537–548. [Google Scholar] [CrossRef] [PubMed]
- Ding, M.H.; Lozoya, E.G.; Rico, R.N.; Chew, S.A. The role of angiogenesis-inducing micrornas in vascular tissue engineering. Tissue Eng. Part A 2020, 26, 1283–1302. [Google Scholar] [CrossRef]
- Landskroner-Eiger, S.; Moneke, I.; Sessa, W.C. Mirnas as modulators of angiogenesis. Cold Spring Harb Perspect. Med. 2013, 3, a006643. [Google Scholar] [CrossRef]
- Mulholland, E.J.; Dunne, N.; McCarthy, H.O. Microrna as therapeutic targets for chronic wound healing. Mol. Ther. Nucleic Acids 2017, 8, 46–55. [Google Scholar] [CrossRef]
- Nejad, C.; Stunden, H.J.; Gantier, M.P. A guide to mirnas in inflammation and innate immune responses. FEBS J. 2018, 285, 3695–3716. [Google Scholar] [CrossRef]
- Soliman, A.M.; Das, S.; Abd Ghafar, N.; Teoh, S.L. Role of microrna in proliferation phase of wound healing. Front. Genet. 2018, 9, 38. [Google Scholar] [CrossRef] [PubMed]
- Suárez, Y.; Sessa, W.C. Micrornas as novel regulators of angiogenesis. Circ. Res. 2009, 104, 442–454. [Google Scholar] [CrossRef]
- Lee, J.Y.; Kim, H.S. Extracellular vesicles in regenerative medicine: Potentials and challenges. Tissue Eng. Regen. Med. 2021, 18, 479–484. [Google Scholar] [CrossRef]
- Bodnar, R.J. Chemokine regulation of angiogenesis during wound healing. Adv. Wound Care 2015, 4, 641–650. [Google Scholar] [CrossRef] [PubMed]
- Koh, T.J.; DiPietro, L.A. Inflammation and wound healing: The role of the macrophage. Expert Rev. Mol. Med. 2011, 13, e23. [Google Scholar] [CrossRef] [PubMed]
- Vono, R.; Jover Garcia, E.; Spinetti, G.; Madeddu, P. Oxidative stress in mesenchymal stem cell senescence: Regulation by coding and noncoding rnas. Antioxid. Redox Signal. 2018, 29, 864–879. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Wang, S.Y.; Kwak, G.; Yang, Y.; Kwon, I.C.; Kim, S.H. Exosome-guided phenotypic switch of m1 to m2 macrophages for cutaneous wound healing. Adv. Sci. 2019, 6, 1900513. [Google Scholar] [CrossRef]
- Brem, H.; Tomic-Canic, M. Cellular and molecular basis of wound healing in diabetes. J. Clin. Investig. 2007, 117, 1219–1222. [Google Scholar] [CrossRef]
- Mohd Hilmi, A.B.; Halim, A.S.; Jaafar, H.; Asiah, A.B.; Hassan, A. Chitosan dermal substitute and chitosan skin substitute contribute to accelerated full-thickness wound healing in irradiated rats. BioMed Res. Int. 2013, 2013, 795458. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Heo, J.S. Selenium-Stimulated Exosomes Enhance Wound Healing by Modulating Inflammation and Angiogenesis. Int. J. Mol. Sci. 2022, 23, 11543. https://doi.org/10.3390/ijms231911543
Heo JS. Selenium-Stimulated Exosomes Enhance Wound Healing by Modulating Inflammation and Angiogenesis. International Journal of Molecular Sciences. 2022; 23(19):11543. https://doi.org/10.3390/ijms231911543
Chicago/Turabian StyleHeo, June Seok. 2022. "Selenium-Stimulated Exosomes Enhance Wound Healing by Modulating Inflammation and Angiogenesis" International Journal of Molecular Sciences 23, no. 19: 11543. https://doi.org/10.3390/ijms231911543





