Oxidative Stress and DNA Damage Markers in Colorectal Cancer
Abstract
:1. Introduction
2. Results
2.1. Clinical and Anthropometric Characteristics
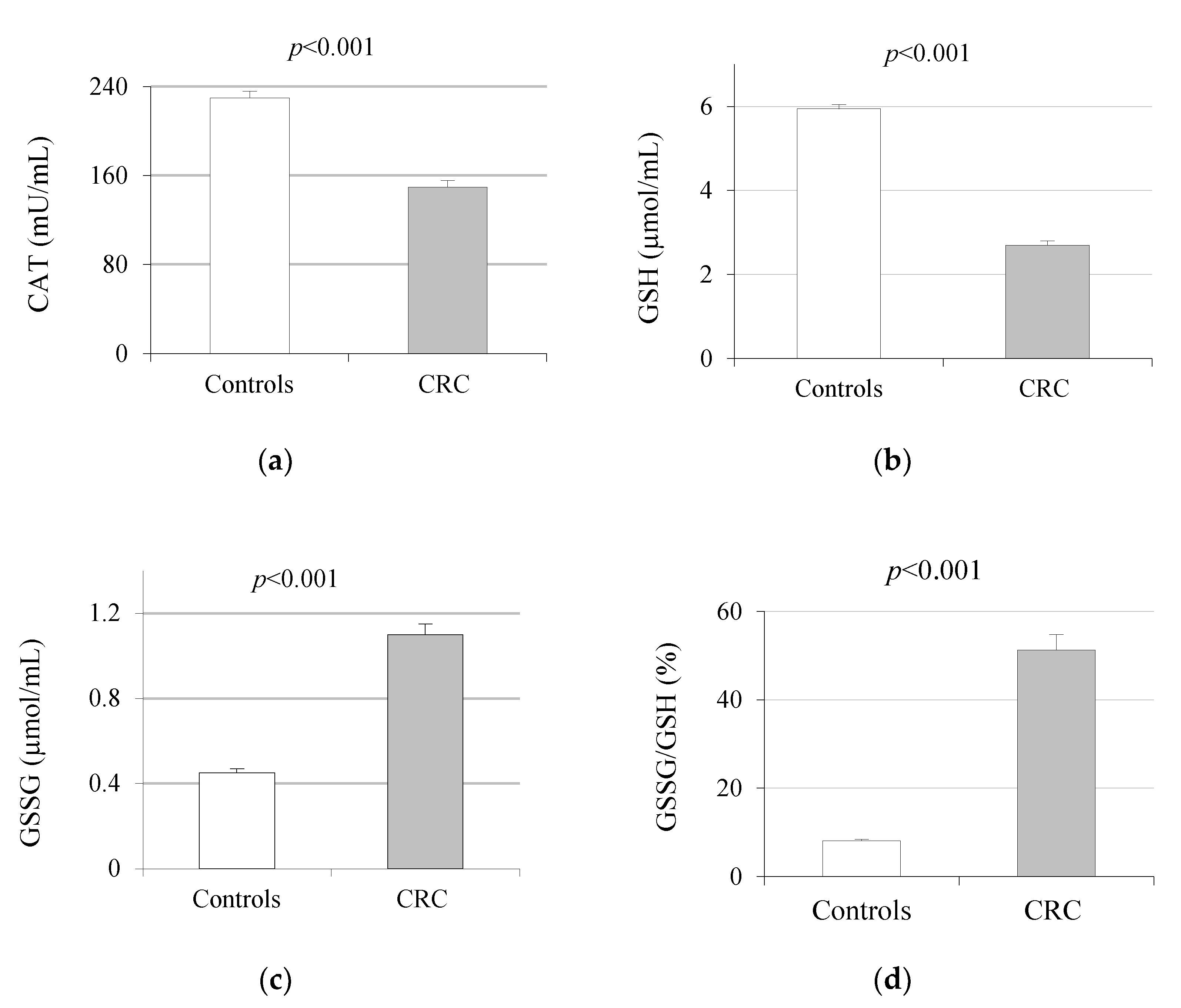
2.2. Oxidative Stress Markers
2.3. Association with Tumor Stages
2.3.1. Biochemical, Anemia, Tumor and Inflammation Markers
2.3.2. Oxidative Stress Markers
2.4. Correlation Analysis
3. Discussion
4. Materials and Methods
4.1. Study Design
4.2. Study Population
4.3. Analytical Assays
4.3.1. Biochemical and Hematological Studies in Serum and Blood Samples
4.3.2. Oxidative Stress and Inflammatory Profile
4.4. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- GLOBOCAN 2020: Colorectal Cancer. Available online: https://gco.iarc.fr/today/data/factsheets/cancers/10_8_9-Colorectum-fact-sheet.pdf (accessed on 13 September 2022).
- Xi, Y.; Xu, P. Global colorectal cancer burden in 2020 and projections to 2040. Transl. Oncol. 2021, 14, 101174. [Google Scholar] [CrossRef] [PubMed]
- American Society of Clinical Oncology (ASCO). Available online: https://www.cancer.net/cancer-types/colorectal-cancer/statistics (accessed on 15 September 2022).
- Assi, M. The differential role of reactive oxygen species in early and late stages of cancer. Am. J. Physiol. Integr. Comp. Physiol. 2017, 313, R646–R653. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jones, D.P. Radical-free biology of oxidative stress. Am. J. Physiol.-Cell Physiol. 2008, 295, C849–C868. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, N.; Baby, D.; Rajguru, J.P.; Patil, P.B.; Thakkannavar, S.S.; Pujari, V.B. Inflammation and cancer. Ann. Afr. Med. 2019, 3, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Todoric, J.; Antonucci, L.; Karin, M. Targeting Inflammation in Cancer Prevention and Therapy. Cancer Prev. Res. 2016, 9, 895–905. [Google Scholar] [CrossRef] [Green Version]
- Federico, A.; Morgillo, F.; Tuccillo, C.; Ciardiello, F.; Loguercio, C. Chronic inflammation and oxidative stress in human carcinogenesis. Int. J. Cancer 2007, 121, 2381–2386. [Google Scholar] [CrossRef] [PubMed]
- Perse, M. Oxidative stress in the pathogenesis of colorectal cancer: Cause or consequence? Biomed. Res. Int. 2013, 2013, 725710. [Google Scholar] [CrossRef] [Green Version]
- Basak, D.; Uddin, M.N.; Hancock, J. The Role of Oxidative Stress and Its Counteractive Utility in Colorectal Cancer (CRC). Cancers 2020, 12, 3336. [Google Scholar] [CrossRef]
- Brenner, D.R.; Scherer, D.; Muir, K.; Schildkraut, J.; Boffetta, P.; Spitz, M.R.; Le Marchand, L.; Chan, A.T.; Goode, E.L.; Ulrich, C.M.; et al. A Review of the Application of Inflammatory Biomarkers in Epidemiologic Cancer Research. Cancer Epidemiology Biomark. Prev. 2014, 23, 1729–1751. [Google Scholar] [CrossRef] [Green Version]
- Shibutani, S.; Takeshita, M.; Grollman, A.P. Insertion of specific bases during DNA synthesis past the oxidation-damaged base 8-oxodG. Nature 1991, 349, 431–434. [Google Scholar] [CrossRef] [PubMed]
- Borrego, S.; Vázquez, A.; Dasí, F.; Cerdá, C.; Iradi, A.; Tormos, C.; Sánchez, J.M.; Bagán, L.; Boix, J.; Zaragoza, C.; et al. Oxidative stress and DNA damage in human gastric carcinoma: 8-oxo-7’8-dihydro-2’-deoxyguanosine (8-oxo-dG) as a possible tumor marker. Int. J. Mol. Sci. 2013, 14, 3467–3486. [Google Scholar] [CrossRef] [PubMed]
- Czerska, M.; Zieliński, M.; Gromadzińska, J. Isoprostanes—A novel major group of oxidative stress markers. Int. J. Occup. Med. Environ. Health 2015, 29, 179–190. [Google Scholar] [CrossRef] [PubMed]
- Gào, X.; Brenner, H.; Holleczek, B.; Cuk, K.; Zhang, Y.; Anusruti, A.; Xuan, Y.; Xu, Y.; Schöttker, B. Urinary 8-isoprostane levels and occurrence of lung, colorectal, prostate, breast and overall cancer: Results from a large, population-based cohort study with 14 years of follow-up. Free Radic. Biol. Med. 2018, 123, 20–26. [Google Scholar] [CrossRef]
- Dusak, A.; Atasoy, N.; Demir, H.; Doğan, E.; Gürsoy, T.; Sarıkaya, E. Investigation of levels of oxidative stress and antioxidant enzymes in colon cancers. J. Clin. Anal. Med. 2017, 8, 469–473. [Google Scholar]
- Chang, D.; Wang, F.; Zhao, Y.-S.; Pan, H.-Z. Evaluation of Oxidative Stress in Colorectal Cancer Patients. Biomed. Environ. Sci. 2008, 21, 286–289. [Google Scholar] [CrossRef]
- Rasool, M.; Malik, A.; Ghuman, A.A.; Ashraf, M.A.B.; Arooj, M.; Waquar, S.; Zahid, S.; Shaheen, S.; Qazi, A.; Naseer, M.I.; et al. Implications of Isoprostanes and Matrix Metalloproteinase-7 Having Potential Role in the Development of Colorectal Cancer in Males. Front. Oncol. 2018, 8, 205. [Google Scholar] [CrossRef] [PubMed]
- Aleksandrova, K.; Nimptsch, K.; Pischon, T. Obesity and colorectal cancer. Front. Biosci. (Elite Ed.) 2013, 5, 61–77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murdolo, G.; Piroddi, M.; Luchetti, F.; Tortoioli, C.; Canonico, B.; Zerbinati, C.; Galli, F.; Iuliano, L. Oxidative stress and lipid peroxidation by-products at the crossroad between adipose organ dysregulation and obesity-linked insulin resistance. Biochimie 2012, 95, 585–594. [Google Scholar] [CrossRef]
- Horn, R.C.; Gelatti, G.T.; Mori, N.C.; Tissiani, A.C.; Mayer, M.S.; Pereira, E.A.; Ross, M.; Moreira, P.R.; Bortolotto, J.W.; Felippin, T. Obesity, bariatric surgery and oxidative stress. Rev. Assoc. Med. Bras. (1992) 2017, 63, 229–235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cecerska-Heryć, E.; Surowska, O.; Heryć, R.; Serwin, N.; Napiontek-Balińska, S.; Dołęgowska, B. Are antioxidant enzymes essential markers in the diagnosis and monitoring of cancer patients—A review. Clin. Biochem. 2021, 93, 1–8. [Google Scholar] [CrossRef]
- Piecuch, A.; Kurek, J.; Kucharzewski, M.; Wyrobiec, G.; Jasiński, D.; Brzozowa-Zasada, M. Catalase immunoexpression in colorectal lesions. Gastroenterol. Rev. 2020, 15, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Chung-man Ho, J.; Zheng, S.; Comhair, S.A.; Farver, C.; Erzurum, S.C. Differential expression of manganese superoxide dismutase and catalase in lung cancer. Cancer Res. 2001, 61, 8578–8585. [Google Scholar]
- Didžiapetrienė, J.; Bublevič, J.; Smailytė, G.; Kazbarienė, B.; Stukas, R. Significance of blood serum catalase activity and malondialdehyde level for survival prognosis of ovarian cancer patients. Medicina 2014, 50, 204–208. [Google Scholar] [CrossRef] [PubMed]
- Zińczuk, J.; Maciejczyk, M.; Zaręba, K.; Romaniuk, W.; Markowski, A.; Kędra, B.; Zalewska, A.; Pryczynicz, A.; Matowicka-Karna, J.; Guzińska-Ustymowicz, K. Antioxidant barrier, redox status, and oxidative damage to biomolecules in patients with colorectal cancer. Can malondialdehyde and catalase be markers of colorectal cancer advancement? Biomolecules 2019, 9, 637. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baltruskeviciene, E.; Kazbariene, B.; Badaras, R.; Bagdonaitė, L.; Krikštaponienė, A.; Zdanavičius, L.; Aleknavicius, E.; Didziapetrienė, J. Glutathione and glutathione S-transferase levels in patients with liver metastases of colorectal cancer and other hepatic disorders. Turk. J. Gastroenterol. 2016, 27, 336–341. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Acevedo-León, D.; Monzó-Beltrán, L.; Gómez-Abril, S.; Estañ-Capell, N.; Camarasa-Lillo, N.; Pérez-Ebri, M.; Escandón-Álvarez, J.; Alonso-Iglesias, E.; Santaolaria-Ayora, M.; Carbonell-Moncho, A.; et al. The Effectiveness of Glutathione Redox Status as a Possible Tumor Marker in Colorectal Cancer. Int. J. Mol. Sci. 2021, 22, 6183. [Google Scholar] [CrossRef]
- Skrzydlewska, E.; Sulkowski, S.; Koda, M.; Zalewski, B.; Kanczuga-Koda, L.; Sulkowska, M. Lipid peroxidation and antioxidant status in colorectal cancer. World J. Gastroenterol. 2005, 11, 403–406. [Google Scholar] [CrossRef]
- Kruk, J.; Aboul-Enein, H.Y. Reactive oxygen and nitrogen species in carcinogenesis: Implications of oxidative stress on the progression and development of several cancer types. Mini-Reviews Med. Chem. 2017, 17, 904–919. [Google Scholar] [CrossRef]
- Kubo, N.; Morita, M.; Nakashima, Y.; Kitao, H.; Egashira, A.; Saeki, H.; Oki, E.; Kakeji, Y.; Oda, Y.; Maehara, Y. Oxidative DNA damage in human esophageal cancer: Clinicopathological analysis of 8-hydroxydeoxyguanosine and its repair enzyme. Dis. Esophagus 2013, 27, 285–293. [Google Scholar] [CrossRef]
- Roszkowski, K.; Jozwicki, W.; Blaszczyk, P.; Mucha-Malecka, A.; Siomek, A. Oxidative damage DNA: 8-oxoGua and 8-oxodG as molecular markers of cancer. Med. Sci. Monit. 2011, 17, CR329–CR333. [Google Scholar] [CrossRef] [Green Version]
- Jelic, M.; Mandic, A.; Maricic, S.; Srdjenovic, B. Oxidative stress and its role in cancer. J. Cancer Res. Ther. 2021, 17, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Acevedo-León, D.; Gómez-Abril, S.; Monzó-Beltrán, L.; Estañ-Capell, N.; Arroyo-Montañés, R.; Bañuls, C.; Salas-Salvadó, J.; Sáez, G. Adherence to the Mediterranean Diet Has a Protective Role against Metabolic and DNA Damage Markers in Colorectal Cancer Patients. Antioxidants 2022, 11, 499. [Google Scholar] [CrossRef] [PubMed]
- Amitay, E.L.; Carr, P.R.; Gies, A.; Laetsch, D.C.; Brenner, H. Probiotic/Synbiotic Treatment and Postoperative Complications in Colorectal Cancer Patients: Systematic Review and Meta-analysis of Randomized Controlled Trials. Clin. Transl. Gastroenterol. 2020, 11, e00268. [Google Scholar] [CrossRef] [PubMed]
- Pourrajab, B.; Fatahi, S.; Sohouli, M.H.; Găman, M.-A.; Shidfar, F. The effects of probiotic/synbiotic supplementation compared to placebo on biomarkers of oxidative stress in adults: A systematic review and meta-analysis of randomized controlled trials. Crit. Rev. Food Sci. Nutr. 2020, 62, 490–507. [Google Scholar] [CrossRef]
- Milne, G.L.; Dai, Q.; Roberts, L.J. The isoprostanes-25 years later. Biochim. Biophys. Acta 2015, 1851, 433–445. [Google Scholar] [CrossRef] [Green Version]
- Kadiiska, M.B.; Gladen, B.C.; Baird, D.; Germolec, D.; Graham, L.B.; Parker, C.E.; Nyska, A.; Wachsman, J.T.; Ames, B.N.; Basu, S.; et al. Biomarkers of Oxidative Stress Study II: Are oxidation products of lipids, proteins, and DNA markers of CCl4 poisoning? Free Radic. Biol. Med. 2005, 38, 698–710. [Google Scholar] [CrossRef]
- Forman, H.J.; Augusto, O.; Brigelius-Flohe, R.; Dennery, P.A.; Kalyanaraman, B.; Ischiropoulos, H.; Mann, G.E.; Radi, R.; Roberts, L.J.; Vina, J.; et al. Even free radicals should follow some rules: A Guide to free radical research terminology and methodology. Free Radic. Biol. Med. 2014, 78, 233–235. [Google Scholar] [CrossRef]
- Il’ Yasova, D.; Scarbrough, P.; Spasojevic, I. Urinary biomarkers of oxidative status. Clin. Chim. Acta 2012, 413, 1446–1453. [Google Scholar] [CrossRef] [Green Version]
- Khadem-Ansari, M.-H.; Shahsavari, Z.; Rasmi, Y.; Mahmoodlo, R. Elevated levels of urinary 8-hydroxy-2Ͳ-deoxyguanosine and 8-isoprostane in esophageal squamous cell carcinoma. J. Carcinog. 2011, 10, 14. [Google Scholar] [CrossRef]
- Asare, G.A.; Akuffo, G.; Doku, D.; Asiedu, B.; Santa, S. Dynamics of urinary oxidative stress biomarkers: 8-hydroxy-2′-deoxyguanosine and 8-isoprostane in uterine leiomyomas. J. Mid-Life Health 2016, 7, 8–14. [Google Scholar]
- Basbug, M.; Arikanoglu, Z.; Bulbuller, N.; Cetinkaya, Z.; Aygen, E.; Akbulut, S.; Satici, O. Prognostic value of preoperative CEA and CA 19-9 levels in patients with colorectal cancer. Hepatogastroenterology 2011, 58, 400–405. [Google Scholar] [PubMed]
- Kasprzak, A. The Role of Tumor Microenvironment Cells in Colorectal Cancer (CRC) Cachexia. Int. J. Mol. Sci. 2021, 22, 1565. [Google Scholar] [CrossRef]
- Tuomisto, A.E.; Mäkinen, M.J.; Väyrynen, J.P. Systemic inflammation in colorectal cancer: Underlying factors, effects, and prognostic significance. World J. Gastroenterol. 2019, 25, 4383–4404. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Fang, J.; Wu, J.; Zhou, X.; Wei, H. Prognostic value of combining preoperative serum tumor markers and peripheral blood routine indexes in patients with colorectal cancer. Zhonghua Wei Chang Wai Ke Za Zhi = Chin. J. Gastrointest. Surg. 2018, 21, 1421–1426. [Google Scholar]
- García-Heredia, J.M.; Carnero, A. The cargo protein MAP17 (PDZK1IP1) regulates the immune microenvironment. Oncotarget 2017, 8, 98580–98597. [Google Scholar] [CrossRef] [Green Version]
- Waldner, M.J.; Foersch, S.; Neurath, M.F. Interleukin-6—A Key Regulator of Colorectal Cancer Development. Int. J. Biol. Sci. 2012, 8, 1248–1253. [Google Scholar] [CrossRef] [PubMed]
- Nikiteas, N.I.; Tzanakis, N.; Gazouli, M.; Rallis, G.; Daniilidis, K.; Theodoropoulos, G.; Kostakis, A.; Peros, G. Serum IL-6, TNF alpha and CRP levels in Greek colorectal cancer patients: Prognostic implications. World J. Gastroenterol. 2005, 11, 1639–1643. [Google Scholar] [CrossRef] [PubMed]
- Ghuman, S.; Van Hemelrijck, M.; Garmo, H.; Holmberg, L.; Malmström, H.; Lambe, M.; Hammar, N.; Walldius, G.; Jungner, I.; Wulaningsih, W. Serum inflammatory markers and colorectal cancer risk and survival. Br. J. Cancer 2017, 116, 1358–1365. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, S.M.; Buring, J.E.; Lee, I.-M.; Cook, N.R.; Ridker, P.M. C-Reactive Protein Levels Are Not Associated with Increased Risk for Colorectal Cancer in Women. Ann. Intern. Med. 2005, 142, 425–432. [Google Scholar] [CrossRef]
- Chung, Y.C.; Chang, Y.F. Serum C-reactive protein correlates with survival in colorectal cancer patients but is not an independent prognostic indicator. Eur. J. Gastroenterol. Hepatol. 2003, 15, 369–373. [Google Scholar] [CrossRef]
- Gethi.org. Grupo Español de Tumores Huérfanos e Infrecuentes (GETHI). Síndromes paraneoplásicos. Capítulo 3. In Síndromes Paraneoplásicos Hematológicos; Grupo Español de Tumores Huérfanos e Infrecuentes (GETHI): Barcelona, Spain, 2016; pp. 28–45. [Google Scholar]
- Wilson, M.; Dekker, J.W.; Harlaar, J.J.; Jeekel, H.; Schipperus, M.; Zwaginga, J.J. The role of preoperative iron deficiency in colorectal cancer patients: Prevalence and treatment. Int. J. Color. Dis. 2017, 32, 1617–1624. [Google Scholar] [CrossRef] [Green Version]
- Sugimoto, K.; Sakamoto, K.; Kawai, M.; Kawano, S.; Munakata, S.; Ishiyama, S.; Takahashi, M.; Kojima, Y.; Tomiki, Y. Serum oxidative stress is an independent prognostic marker in colorectal cancer. Transl. Cancer Res. 2019, 8, 1699–1708. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Deng, S.; Yan, L.; Gu, J.; Mao, F.; Xue, Y.; Zheng, C.; Yang, M.; Liu, H.; Liu, L.; et al. An Oxidative Stress Index-Based Score for Prognostic Prediction in Colorectal Cancer Patients Undergoing Surgery. Oxidative Med. Cell. Longev. 2021, 2021, 6693707. [Google Scholar] [CrossRef] [PubMed]
- Boakye, D.; Jansen, L.; Schöttker, B.; Jansen, E.H.J.M.; Schneider, M.; Halama, N.; Gào, X.; Chang-Claude, J.; Hoffmeister, M.; Brenner, H. Blood markers of oxidative stress are strongly associated with poorer prognosis in colorectal cancer patients. Int. J. Cancer 2020, 147, 2373–2386. [Google Scholar] [CrossRef] [PubMed]
- Yucel, A.F.; Kemik, O.; Kemik, A.S.; Purisa, S.; Tuzun, I.S. Relationship Between the Levels of Oxidative Stress in Mesenteric and Peripheral Serum and Clinicopathological Variables in Colorectal Cancer. Balk. Med. J. 2012, 29, 144–147. [Google Scholar] [CrossRef] [PubMed]
- Bomfim Gomes Campos, S.; Sanuelly da Paz Martins, A.; Karolyne Dos Santos, A.; Goulart, M.O.F.; Andréa Moura, F. Interleukin-6 and triceps skinfold are associated with severity/cancer stage in newly-diagnosed colorectal cancer patients. Nutr. Hosp. 2021, 38, 1034–1039. [Google Scholar]

| Variable | Control (n = 60) | CRC (n = 80) | p-Value | * Adjusted p-Value |
|---|---|---|---|---|
| Age (years) | 64.0 ± 9.0 | 67.5 ± 11.8 | 0.052 | - |
| Male/Female (n; %) | 36/24; 60/40 | 52/28; 65/35 | 0.548 | - |
| Weight (kg) | 74.4 ± 16.3 | 77.3 ± 15 | <0.001 | - |
| Height (cm) | 168 ± 11 | 165.5 ± 9.8 | <0.001 | - |
| BMI (kg/m2) | 26.1 ± 3.0 | 28.1 ± 3.9 | 0.001 | - |
| Glucose (mg/dL) | 96.2 ± 14.4 | 116.6 ± 52.3 | <0.001 | 0.001 |
| Creatinine (mg/dL) | 0.9 ± 0.2 | 2.0 ± 8.6 | 0.319 | 0.269 |
| Urea (mg/dL) | 40.9 ± 7.2 | 38.8 ± 15.9 | 0.878 | 0.296 |
| EGF (mL/min) | 81.1 ± 8.7 | 78.9 ± 20.9 | 0.399 | 0.720 |
| Total cholesterol (mg/dL) | 195.7 ± 34.3 | 180.4 ± 39.1 | 0.018 | 0.026 |
| HDL cholesterol (mg/dL) | 50.7 ± 12.8 | 43.2 ± 10.8 | <0.001 | <0.001 |
| LDL cholesterol (mg/dL) | 144.9 ± 30.0 | 114.5 ± 34.7 | <0.001 | <0.001 |
| Triglycerides (mg/dL) | 112.0 (98; 142.8) | 108.5 (83.3; 141) | 0.954 | 0.777 |
| Uric acid (mg/dL) | 4.5 ± 1.6 | 5.3 ± 1.7 | 0.003 | 0.058 |
| Albumin (g/dL) | 3.9 ± 0.4 | 4.2 ± 0.5 | 0.001 | <0.001 |
| Total proteins (g/dL) | 7.0 ± 0.5 | 6.9 ± 0.4 | 0.472 | 0.577 |
| Ferritin (µg/L) | 133.5 ± 75.4 | 67.5 ± 144.9 | 0.002 | 0.008 |
| Iron (µg/dL) | 79.7 ± 19.1 | 57.6 ± 41.0 | <0.001 | <0.001 |
| Transferrin (mg/dL) | 269.3 ± 46.5 | 291.1 ± 51.4 | 0.011 | 0.016 |
| TSI (%) | 30.5 ± 8.9 | 16.2 ± 11.1 | <0.001 | <0.001 |
| CRP (mg/L) | 6.2 ± 1.3 | 11.0 ± 23.6 | 0.072 | 0.076 |
| IL-6 (pg/mL) | 2.7 ± 1.4 | 19.7 ± 26.5 | <0.001 | <0.001 |
| Leukocytes (×103/mm3) | 7.0 ± 1.7 | 7.7 ± 1.8 | 0.016 | 0.018 |
| Neutrophils (%) | 56.0 ± 6.0 | 62.9 ± 8.4 | <0.001 | <0.001 |
| Lymphocytes (%) | 29.8 ± 10.2 | 27.7 ± 12.0 | 0.221 | 0.531 |
| N/L (-) | 2.1 ± 1.4 | 2.7 ± 1.6 | 0.005 | 0.013 |
| Platelets (x105/mm3) | 206.0 ± 60.0 | 253.5 ± 74.0 | <0.001 | <0.001 |
| Fibrinogen (mg/dL) | 352.3 ± 70.8 | 483.8 ± 101.7 | <0.001 | <0.001 |
| Hemoglobin (g/dL) | 14.2 ± 1.5 | 12.9 ± 1.9 | <0.001 | <0.001 |
| Hematocrit (%) | 42.5 ± 4.6 | 37.7 ± 5.1 | <0.001 | <0.001 |
| MCV (fL) | 85.7 ± 4.8 | 83.4 ± 8.4 | 0.046 | 0.135 |
| CEA (ng/mL) | 2.35 ± 1.15 | 7.51 ± 11.0 | <0.001 | 0.004 |
| CA 19.9 (UI/mL) | 9.03 ± 6.53 | 23.0 ± 30.4 | 0.002 | 0.011 |
| Variable | Stage 0 | Stage 1 | Stage 2 | p-Value |
|---|---|---|---|---|
| CAT (mU/mL) | 160.1 ± 5.43 a | 140.0 ± 3.92 b | 88.9 ± 4.32 b | 0.002 |
| GSH (µmol/mL) | 2.52 ± 0.72 a | 2.32 ± 0.65 a,b | 1.86 ± 0.09 b | 0.045 |
| GSSG (µmol/mL) | 1.06 ± 0.44 a | 1.03 ± 0.39 a | 1.68 ± 1.68 b | 0.002 |
| GSSG/GSH (%) | 47.6 ± 31.3 a | 48.3 ± 25.7 a | 90.7 ± 33.9 b | 0.002 |
| 8-oxodG (nmol/mmol crea) | 12.2 ± 4.35 a | 12.0 ± 3.97 a | 19.3 ± 6.36 b | 0.001 |
| F2-IsoPs (pg/mg crea) | 106.6 ± 3.8 a | 107.2 ± 3.5 a | 128.2 ± 6.3 b | 0.004 |
| CAT (mU/mL) | GSH (μmol/mL) | GSSG (μmol/mL) | GSSG/GSH (%) | 8-oxodG (nmol/mmol crea) | F2-IsoPs (pg/mg crea) | |
|---|---|---|---|---|---|---|
| Tumor markers | ||||||
| CEA (ng/mL) | −0.379 *** | −0.270 ** | 0.292 ** | 0.276 ** | 0.300 *** | 0.183 ** |
| CA 19.9 (IU/mL) | −0.412 *** | −0.292 ** | 0.345 *** | 0.322 *** | 0.257 ** | 0.343 *** |
| Anemia markers | ||||||
| Hemoglobin (g/dL) | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. |
| Hematocrit (%) | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. |
| MCV (fL) | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. |
| Inflammatory markers | ||||||
| CRP (mg/L) | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. |
| IL-6 (pg/mL) | −0.237 *** | −0.328 *** | 0.419 *** | 0.385 *** | 0.366*** | 0.367 *** |
| Leukocytes (×103/mm3) | −0.237 ** | n.s. | 0.186 * | 0.173 * | 0.193 * | 0.260 ** |
| Neutrophils (%) | −0.236 ** | −0.318 *** | 0.362 *** | 0.330 *** | 0.332 *** | 0.356 *** |
| N/L (-) | −0.187 * | −0.175 * | 0.232 ** | 0.181 * | 0.246 ** | 0.268 *** |
| Platelets (×105/mm3) | −0.256 ** | −0.300 *** | 0.240 ** | 0.239 ** | 0.302 *** | 0.294 *** |
| Fibrinogen (mg/dL) | −0.471 *** | −0.462 *** | 0.521 *** | 0.521 *** | 0.522 *** | 0.535 *** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Acevedo-León, D.; Monzó-Beltrán, L.; Pérez-Sánchez, L.; Naranjo-Morillo, E.; Gómez-Abril, S.Á.; Estañ-Capell, N.; Bañuls, C.; Sáez, G. Oxidative Stress and DNA Damage Markers in Colorectal Cancer. Int. J. Mol. Sci. 2022, 23, 11664. https://doi.org/10.3390/ijms231911664
Acevedo-León D, Monzó-Beltrán L, Pérez-Sánchez L, Naranjo-Morillo E, Gómez-Abril SÁ, Estañ-Capell N, Bañuls C, Sáez G. Oxidative Stress and DNA Damage Markers in Colorectal Cancer. International Journal of Molecular Sciences. 2022; 23(19):11664. https://doi.org/10.3390/ijms231911664
Chicago/Turabian StyleAcevedo-León, Delia, Lidia Monzó-Beltrán, Laura Pérez-Sánchez, Eva Naranjo-Morillo, Segundo Ángel Gómez-Abril, Nuria Estañ-Capell, Celia Bañuls, and Guillermo Sáez. 2022. "Oxidative Stress and DNA Damage Markers in Colorectal Cancer" International Journal of Molecular Sciences 23, no. 19: 11664. https://doi.org/10.3390/ijms231911664





