R-Ras1 and R-Ras2 Expression in Anatomical Regions and Cell Types of the Central Nervous System
Abstract
:1. Introduction
2. Results
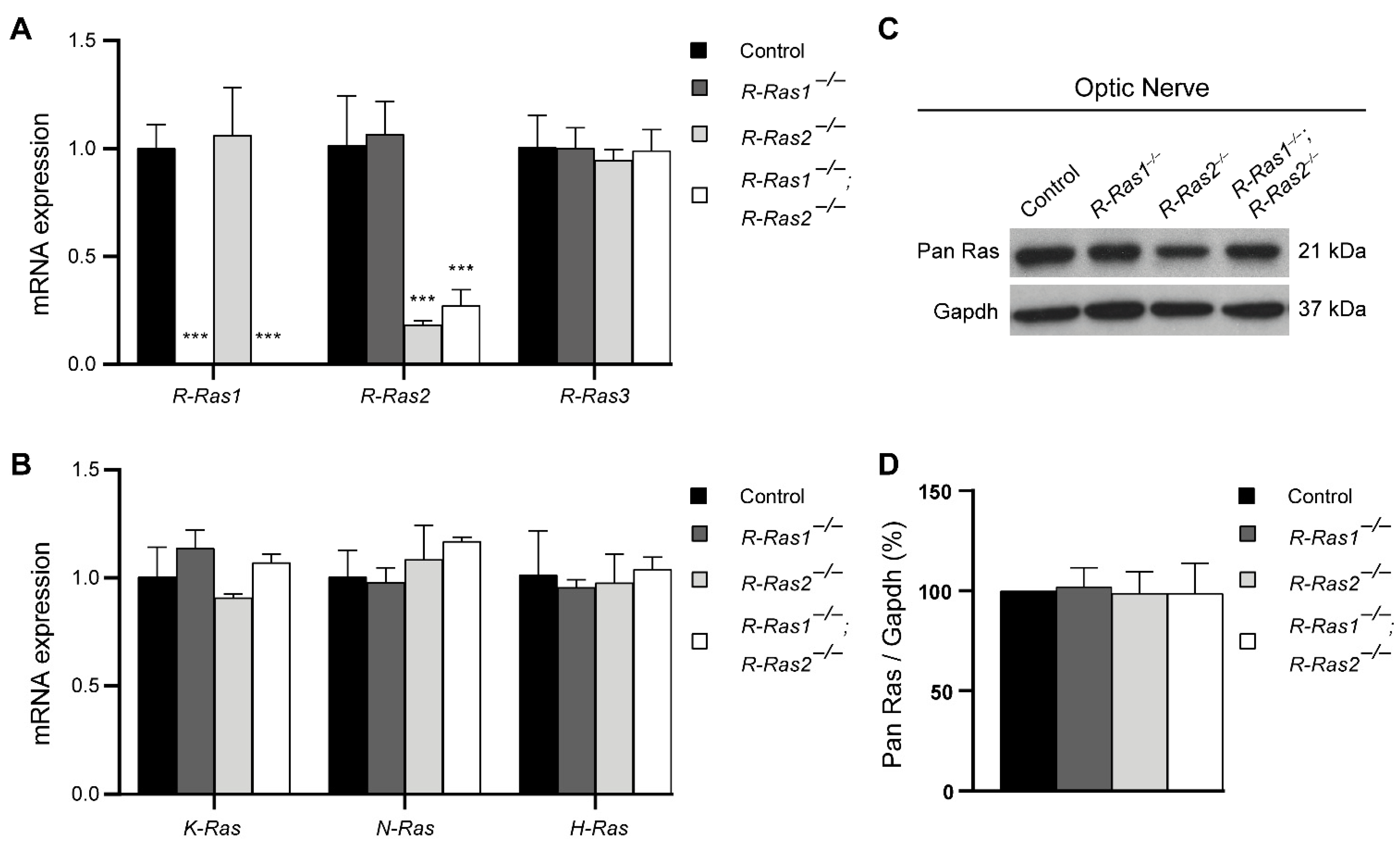
2.1. Loss of R-Ras1 and R-Ras2 Cannot Be Compensated by Either R-Ras3 or Classical Ras
2.2. R-Ras1 and R-Ras2 Are Expressed at Different Levels throughout the CNS
2.3. R-Ras1 and R-Ras2 Are Expressed by Neurons in the CNS, but Not in Astrocytes or Microglia
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Reverse Transcription Quantitative PCR (RT-qPCR)
4.3. Western Blotting
4.4. X-Gal Reaction
4.5. Immunohistochemistry
4.6. Confocal Microscopy
4.7. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Morena, J.; Gupta, A.; Hoyle, J.C. Charcot-Marie-Tooth: From Molecules to Therapy. Int. J. Mol. Sci. 2019, 20, 3419. [Google Scholar] [CrossRef] [Green Version]
- Kim, U.S.; Jurkute, N.; Yu-Wai-Man, P. Leber Hereditary Optic Neuropathy-Light at the End of the Tunnel? Asia-Pac. J. Ophthalmol. 2018, 7, 242–245. [Google Scholar] [CrossRef]
- Nave, K.-A.; Werner, H.B. Myelination of the Nervous System: Mechanisms and Functions. Annu. Rev. Cell Dev. Biol. 2014, 30, 503–533. [Google Scholar] [CrossRef]
- Jasiak-Zatonska, M.; Kalinowska-Lyszczarz, A.; Michalak, S.; Kozubski, W. The Immunology of Neuromyelitis Optica—Current Knowledge, Clinical Implications, Controversies and Future Perspectives. Int. J. Mol. Sci. 2016, 17, 273. [Google Scholar] [CrossRef] [Green Version]
- Gómez-Pinedo, U.; García-Ávila, Y.; Gallego-Villarejo, L.; Matías-Guiu, J.A.; Benito-Martín, M.S.; Esteban-García, N.; Sanclemente-Alamán, I.; Pytel, V.; Moreno-Jiménez, L.; Sancho-Bielsa, F.; et al. Sera from Patients with NMOSD Reduce the Differentiation Capacity of Precursor Cells in the Central Nervous System. Int. J. Mol. Sci. 2021, 22, 5192. [Google Scholar] [CrossRef] [PubMed]
- Benítez-Fernández, R.; Melero-Jerez, C.; Gil, C.; de la Rosa, E.J.; Martínez, A.; de Castro, F. Dynamics of Central Remyelination and Treatment Evolution in a Model of Multiple Sclerosis with Optic Coherence Tomography. Int. J. Mol. Sci. 2021, 22, 2440. [Google Scholar] [CrossRef]
- Charzewska, A.; Wierzba, J.; Iżycka-Świeszewska, E.; Bekiesińska-Figatowska, M.; Jurek, M.; Gintowt, A.; Kłosowska, A.; Bal, J.; Hoffman-Zacharska, D. Hypomyelinating Leukodystrophies-a Molecular Insight into the White Matter Pathology: Hypomyelinating Leukodystrophies. Clin. Genet. 2016, 90, 293–304. [Google Scholar] [CrossRef] [PubMed]
- Boespflug-Tanguy, O.; Labauge, P.; Fogli, A.; Vaurs-Barriere, C. Genes Involved in Leukodystrophies: A Glance at Glial Functions. Curr. Neurol. Neurosci. Rep. 2008, 8, 217–229. [Google Scholar] [CrossRef] [PubMed]
- Bercury, K.K.; Macklin, W.B. Dynamics and Mechanisms of CNS Myelination. Dev. Cell 2015, 32, 447–458. [Google Scholar] [CrossRef] [Green Version]
- Snaidero, N.; Simons, M. Myelination at a Glance. J. Cell Sci. 2014, 127, 2999–3004. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Laursen, L.S.; Chan, C.W.; ffrench-Constant, C. An Integrin-Contactin Complex Regulates CNS Myelination by Differential Fyn Phosphorylation. J. Neurosci. 2009, 29, 9174–9185. [Google Scholar] [CrossRef] [Green Version]
- Baron, W.; Colognato, H.; Ffrench-Constant, C. Integrin-Growth Factor Interactions as Regulators of Oligodendroglial Development and Function. Glia 2005, 49, 467–479. [Google Scholar] [CrossRef] [PubMed]
- Snaidero, N.; Simons, M. The Logistics of Myelin Biogenesis in the Central Nervous System. Glia 2017, 65, 1021–1031. [Google Scholar] [CrossRef]
- Foster, A.Y.; Bujalka, H.; Emery, B. Axoglial Interactions in Myelin Plasticity: Evaluating the Relationship between Neuronal Activity and Oligodendrocyte Dynamics. Glia 2019, 67, 2038–2049. [Google Scholar] [CrossRef] [PubMed]
- Hill, R.A.; Grutzendler, J. Uncovering the Biology of Myelin with Optical Imaging of the Live Brain. Glia 2019, 67, 2008–2019. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Morrison, B.M.; Li, Y.; Lengacher, S.; Farah, M.H.; Hoffman, P.N.; Liu, Y.; Tsingalia, A.; Jin, L.; Zhang, P.-W.; et al. Oligodendroglia Metabolically Support Axons and Contribute to Neurodegeneration. Nature 2012, 487, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Saab, A.S.; Tzvetanova, I.D.; Nave, K.-A. The Role of Myelin and Oligodendrocytes in Axonal Energy Metabolism. Curr. Opin. Neurobiol. 2013, 23, 1065–1072. [Google Scholar] [CrossRef]
- Simons, M.; Nave, K.-A. Oligodendrocytes: Myelination and Axonal Support. Cold Spring Harb. Perspect. Biol. 2016, 8, a020479. [Google Scholar] [CrossRef]
- Gaesser, J.M.; Fyffe-Maricich, S.L. Intracellular Signaling Pathway Regulation of Myelination and Remyelination in the CNS. Exp. Neurol. 2016, 283, 501–511. [Google Scholar] [CrossRef] [Green Version]
- Alcover-Sanchez, B.; Garcia-Martin, G.; Wandosell, F.; Cubelos, B. R-Ras GTPases Signaling Role in Myelin Neurodegenerative Diseases. Int. J. Mol. Sci. 2020, 21, 5911. [Google Scholar] [CrossRef]
- Colicelli, J. Human RAS Superfamily Proteins and Related GTPases. Sci. Signal. 2004, 2004, re13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, W.N.; Yan, M.; Chan, A.M. A Thirty-Year Quest for a Role of R-Ras in Cancer: From an Oncogene to a Multitasking GTPase. Cancer Lett. 2017, 403, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Simanshu, D.K.; Nissley, D.V.; McCormick, F. RAS Proteins and Their Regulators in Human Disease. Cell 2017, 170, 17–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goitre, L.; Trapani, E.; Trabalzini, L.; Retta, S.F. The Ras Superfamily of Small GTPases: The Unlocked Secrets. In Ras Signaling; Trabalzini, L., Retta, S.F., Eds.; Methods in Molecular Biology; Humana Press: Totowa, NJ, USA, 2014; Volume 1120, pp. 1–18. ISBN 978-1-62703-790-7. [Google Scholar]
- Ohba, Y.; Mochizuki, N.; Yamashita, S.; Chan, A.M.; Schrader, J.W.; Hattori, S.; Nagashima, K.; Matsuda, M. Regulatory Proteins of R-Ras, TC21/R-Ras2, and M-Ras/R-Ras3. J. Biol. Chem. 2000, 275, 20020–20026. [Google Scholar] [CrossRef] [Green Version]
- Gutierrez-Erlandsson, S.; Herrero-Vidal, P.; Fernandez-Alfara, M.; Hernandez-Garcia, S.; Gonzalo-Flores, S.; Mudarra-Rubio, A.; Fresno, M.; Cubelos, B. R-RAS2 Overexpression in Tumors of the Human Central Nervous System. Mol. Cancer 2013, 12, 127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kimmelman, A.C.; Rodriguez, N.N.; Chan, A.M.-L. R-Ras3/M-Ras Induces Neuronal Differentiation of PC12 Cells through Cell-Type-Specific Activation of the Mitogen-Activated Protein Kinase Cascade. Mol. Cell. Biol. 2002, 22, 5946–5961. [Google Scholar] [CrossRef] [Green Version]
- Tasaka, G.-I.; Negishi, M.; Oinuma, I. Semaphorin 4D/Plexin-B1-Mediated M-Ras GAP Activity Regulates Actin-Based Dendrite Remodeling through Lamellipodin. J. Neurosci. 2012, 32, 8293–8305. [Google Scholar] [CrossRef] [Green Version]
- Kimmelman, A.; Tolkacheva, T.; Lorenzi, M.V.; Osada, M.; Chan, A.M.-L. Identi®cation and Characterization of R-Ras3: A Novel Member of the RAS Gene Family with a Non-Ubiquitous Pattern of Tissue Distribution. Oncogene 1997, 15, 2675–2685. [Google Scholar] [CrossRef] [Green Version]
- Sanz-Rodriguez, M.; Gruart, A.; Escudero-Ramirez, J.; de Castro, F.; Delgado-García, J.M.; Wandosell, F.; Cubelos, B. R-Ras1 and R-Ras2 Are Essential for Oligodendrocyte Differentiation and Survival for Correct Myelination in the Central Nervous System. J. Neurosci. 2018, 38, 5096–5110. [Google Scholar] [CrossRef] [Green Version]
- Alcover-Sanchez, B.; Garcia-Martin, G.; Escudero-Ramirez, J.; Gonzalez-Riano, C.; Lorenzo, P.; Gimenez-Cassina, A.; Formentini, L.; Villa-Polo, P.; Pereira, M.P.; Wandosell, F.; et al. Absence of R-Ras1 and R-Ras2 Causes Mitochondrial Alterations That Trigger Axonal Degeneration in a Hypomyelinating Disease Model. Glia 2021, 69, 619–637. [Google Scholar] [CrossRef]
- Ivins, J.K.; Yurchenco, P.D.; Lander, A.D. Regulation of Neurite Outgrowth by Integrin Activation. J. Neurosci. 2000, 20, 6551–6560. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Negishi, M.; Oinuma, I.; Katoh, H. R-Ras as a Key Player for Signaling Pathway of Plexins. Mol. Neurobiol. 2005, 32, 217–222. [Google Scholar] [CrossRef]
- Oinuma, I.; Katoh, H.; Negishi, M. R-Ras Controls Axon Specification Upstream of Glycogen Synthase Kinase-3β through Integrin-Linked Kinase. J. Biol. Chem. 2007, 282, 303–318. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iwasawa, N.; Negishi, M.; Oinuma, I. R-Ras Controls Axon Branching through Afadin in Cortical Neurons. Mol. Biol. Cell 2012, 23, 2793–2804. [Google Scholar] [CrossRef] [PubMed]
- Delgado, P.; Cubelos, B.; Calleja, E.; Martínez-Martín, N.; Ciprés, A.; Mérida, I.; Bellas, C.; Bustelo, X.R.; Alarcón, B. Essential Function for the GTPase TC21 in Homeostatic Antigen Receptor Signaling. Nat. Immunol. 2009, 10, 880–888. [Google Scholar] [CrossRef] [Green Version]
- Robaglia-Schlupp, A.; Pizant, J.; Norreel, J.-C.; Passage, E.; Sabéran-Djoneidi, D.; Ansaldi, J.-L.; Vinay, L.; Figarella-Branger, D.; Lévy, N.; Clarac, F.; et al. PMP22 Overexpression Causes Dysmyelination in Mice. Brain 2002, 125, 2213–2221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Michailov, G.V.; Sereda, M.W.; Brinkmann, B.G.; Fischer, T.M.; Haug, B.; Birchmeier, C.; Role, L.; Lai, C.; Schwab, M.H.; Nave, K.-A. Axonal Neuregulin-1 Regulates Myelin Sheath Thickness. Science 2004, 304, 700–703. [Google Scholar] [CrossRef] [Green Version]
- Lee, X.; Hu, Y.; Zhang, Y.; Yang, Z.; Shao, Z.; Qiu, M.; Pepinsky, B.; Miller, R.H.; Mi, S. Oligodendrocyte Differentiation and Myelination Defects in OMgp Null Mice. Mol. Cell. Neurosci. 2011, 46, 752–761. [Google Scholar] [CrossRef]
- Olsen, I.M.; ffrench-Constant, C. Dynamic Regulation of Integrin Activation by Intracellular and Extracellular Signals Controls Oligodendrocyte Morphology. BMC Biol. 2005, 3, 25. [Google Scholar] [CrossRef] [Green Version]
- Laurell, H.; Iacovoni, J.S.; Abot, A.; Svec, D.; Maoret, J.-J.; Arnal, J.-F.; Kubista, M. Correction of RT–QPCR Data for Genomic DNA-Derived Signals with ValidPrime. Nucleic Acids Res. 2012, 40, e51. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods San Diego Calif. 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Burn, S.F. Detection of β-Galactosidase Activity: X-Gal Staining. In Kidney Development: Methods and Protocols; Michos, O., Ed.; Methods in Molecular BiologyTM; Humana Press: Totowa, NJ, USA, 2012; pp. 241–250. ISBN 978-1-61779-851-1. [Google Scholar]


| Target | Forward Primer | Reverse Primer | Size 1 |
|---|---|---|---|
| R-Ras1+/+ | CGCTCTAGCTGAGCCTCTGT | TACAGGGTCTTGTGGGGAAA | 138 |
| R-Ras2+/+ | TGAAACAGGATCATGTTGTGGAG | CAGGAGGAGTCCAAGAAGAC | 266 |
| R-Ras1−/− | GGAGCAAGAGGAGGGAAGGAATG | CTTCGAGAGGACTCAGTTCAATCC | 1000 |
| R-Ras2−/− | TGAAACAGGATCATGTTGTGGAG | ATAAACCCTCTTGCAGTTGCATC | 128 |
| Target | Forward Primer | Reverse Primer | Size 1 |
|---|---|---|---|
| R-Ras1 | TCACAAGCTGGTGGTCGTAG | TGGGATCATAGTCAGACACAAAG | 95 |
| R-Ras2 | CGTGATGAGTTTCCCATGATT | TAACTGCTGCCCTTCTTCCT | 87 |
| R-Ras3 | TGGGCCATCTTGGATGTT | CTGTGCGCATGTATTGTTCC | 76 |
| K-Ras | TGTGGATGAGTATGACCCTACG | CCCTCATTGCACTGTACTCCT | 122 |
| N-Ras | GAACTGGCCAAGAGTTACGG | TGTAAAAGGCATCCTCCACA | 79 |
| H-Ras | CGCCAGCAAGCGGTG | GGTAGGAGTCCTCTATAGTGGGATCA | 174 |
| ActB | CTAAGGCCAACCGTGAAAAG | ACCAGAGGCATACAGGGACA | 104 |
| Arbp | GATGCCCAGGGAAGACAG | TCCAAAAGTTGGATGATCTTGA | 66 |
| Gapdh | CACCACCAACTGCTTAGCCC | TGTGGTCATGAGCCCTTCC | 76 |
| GusB | AGCCGCTACGGGAGTCG | GCTGCTTCTTGGGTGATGTCA | 76 |
| Hprt1 | TCCTCCTCAGACCGCTTTT | CCTGGTTCATCATCGCTAATC | 90 |
| Tbp | CCACAGGGCGCCATGA | GCTGTGGAGTAAGTCCTGTGCC | 76 |
| Target | Host | Dilution | Manufacturer | Catalog | RRID |
|---|---|---|---|---|---|
| GAPDH | Mouse | 1:1000 | Santa Cruz Biotechnology | Sc-365062 | AB_10847862 |
| Pan-Ras | Mouse | 1:300 | Millipore | OP40 | AB_213400 |
| R-Ras1 | Rabbit | 1:200 | Abcam | Ab154962 | AB_2894924 |
| R-Ras2 | Rabbit | 1:200 | Professor B. Alarcón (CBMSO) [36] | AB_2895064 | |
| Anti-Mouse | Goat-HRP | 1:5000 | Santa Cruz Biotechnology | Sc-2005 | AB_631736 |
| Anti-Rabbit | Goat-HRP | 1:5000 | Southern Biotech | 4030-05 | AB_2687483 |
| Target | Host | Dilution | Manufacturer | Catalog | RRID |
|---|---|---|---|---|---|
| GFAP | Mouse | 1:1000 | Sigma-Aldrich | G3893 | AB_477010 |
| Iba-1 | Mouse | 1:1000 | Millipore | MABN92 | AB_10917271 |
| NeuN | Mouse | 1:500 | Millipore | MAB377 | AB_2298772 |
| R-Ras1 | Rabbit | 1:200 | Abcam | Ab154962 | AB_2894924 |
| R-Ras2 | Rabbit | 1:200 | Professor B. Alarcón (CBMSO) | ||
| Anti-Mouse | Donkey-Alexa488 | 1:500 | Thermo Fisher | A-21202 | AB_141607 |
| Anti-Rabbit | Donkey-Alexa555 | 1:500 | Thermo Fisher | A-31572 | AB_162543 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garcia-Martin, G.; Sanz-Rodriguez, M.; Alcover-Sanchez, B.; Pereira, M.P.; Wandosell, F.; Cubelos, B. R-Ras1 and R-Ras2 Expression in Anatomical Regions and Cell Types of the Central Nervous System. Int. J. Mol. Sci. 2022, 23, 978. https://doi.org/10.3390/ijms23020978
Garcia-Martin G, Sanz-Rodriguez M, Alcover-Sanchez B, Pereira MP, Wandosell F, Cubelos B. R-Ras1 and R-Ras2 Expression in Anatomical Regions and Cell Types of the Central Nervous System. International Journal of Molecular Sciences. 2022; 23(2):978. https://doi.org/10.3390/ijms23020978
Chicago/Turabian StyleGarcia-Martin, Gonzalo, Miriam Sanz-Rodriguez, Berta Alcover-Sanchez, Marta P. Pereira, Francisco Wandosell, and Beatriz Cubelos. 2022. "R-Ras1 and R-Ras2 Expression in Anatomical Regions and Cell Types of the Central Nervous System" International Journal of Molecular Sciences 23, no. 2: 978. https://doi.org/10.3390/ijms23020978







