Intact Type I Interferon Receptor Signaling Prevents Hepatocellular Necrosis but Not Encephalitis in a Dose-Dependent Manner in Rift Valley Fever Virus Infected Mice
Abstract
1. Introduction
2. Results
2.1. Survival and Clinical Signs
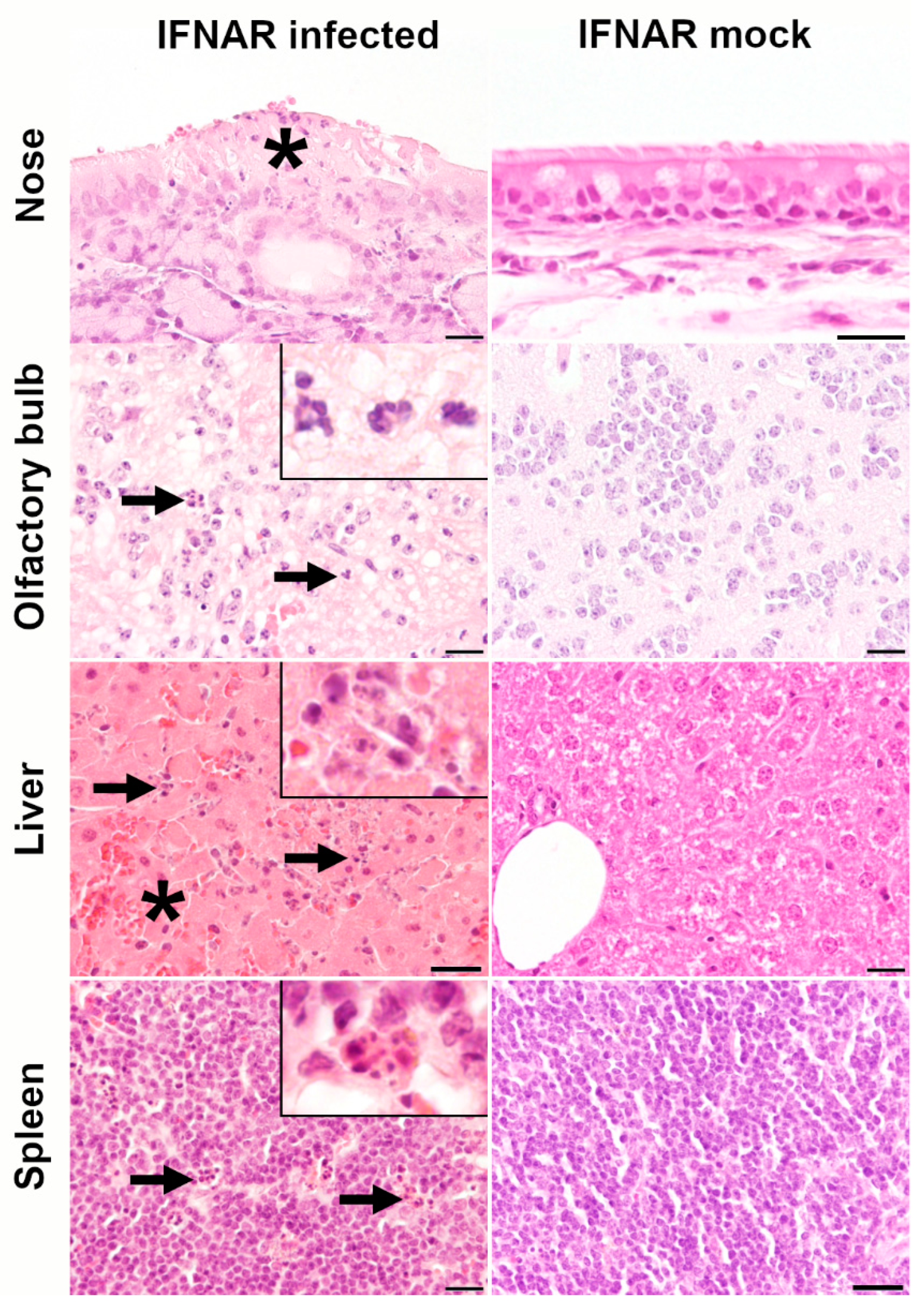
2.2. Histology
2.3. Immunohistochemistry and Immunofluorescence
2.4. Reverse Transcription-Quantitative Polymerase Chain Reaction (RT-qPCR)
3. Discussion
4. Materials and Methods
4.1. Animal Experiment
4.2. RNA Isolation and RT-qPCR
4.3. Histology, Immunohistochemistry and Immunofluorescence
4.4. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hartman, A. Rift Valley Fever. Clin. Lab. Med. 2017, 37, 285–301. [Google Scholar] [CrossRef] [PubMed]
- Ikegami, T.; Makino, S. The Pathogenesis of Rift Valley Fever. Viruses 2011, 3, 493–519. [Google Scholar] [CrossRef] [PubMed]
- Kwaśnik, M.; Rożek, W.; Rola, J. Rift Valley Fever-A Growing Threat to Humans and Animals. J. Vet. Res. 2021, 65, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Ulrich, R. Rift Valley Fever: An Ancient Plague on Its Way out of Africa? Vet. Pathol. 2019, 56, 178–179. [Google Scholar] [CrossRef]
- Wright, D.; Kortekaas, J.; Bowden, T.A.; Warimwe, G.M. Rift Valley Fever: Biology and Epidemiology. J. Gen. Virol. 2019, 100, 1187–1199. [Google Scholar] [CrossRef] [PubMed]
- Chevalier, V.; Pepin, M.; Plee, L.; Lancelot, R. Rift Valley Fever—A Threat for Europe? Euro. Surveill. 2010, 15, 19506. [Google Scholar] [CrossRef]
- Linthicum, K.J.; Britch, S.C.; Anyamba, A. Rift Valley Fever: An Emerging Mosquito-Borne Disease. Annu. Rev. Entomol. 2016, 61, 395–415. [Google Scholar] [CrossRef]
- McEntire, C.R.S.; Song, K.W.; McInnis, R.P.; Rhee, J.Y.; Young, M.; Williams, E.; Wibecan, L.L.; Nolan, N.; Nagy, A.M.; Gluckstein, J.; et al. Neurologic Manifestations of the World Health Organization’s List of Pandemic and Epidemic Diseases. Front Neurol. 2021, 12, 634827. [Google Scholar] [CrossRef]
- Bird, B.H.; McElroy, A.K. Rift Valley Fever Virus: Unanswered Questions. Antiviral. Res. 2016, 132, 274–280. [Google Scholar] [CrossRef]
- Meegan, J.M.; Watten, R.H.; Laughlin, L.W. Clinical Experience with Rift Valley Fever in Humans During the 1977 Egyptian Epizootic. In Rift Valley Fever: Proceedings of a Workshop on Rift Valley Fever, Herzlia, Israel, 18–21 March 1980; Karger: Basel, Switzerland, 1981. [Google Scholar]
- Léger, P.; Tetard, M.; Youness, B.; Cordes, N.; Rouxel, R.N.; Flamand, M.; Lozach, P.Y. Differential Use of the C-Type Lectins L-Sign and Dc-Sign for Phlebovirus Endocytosis. Traffic 2016, 17, 639–656. [Google Scholar] [CrossRef]
- Lozach, P.Y.; Kühbacher, A.; Meier, R.; Mancini, R.; Bitto, D.; Bouloy, M.; Helenius, A. Dc-Sign as a Receptor for Phleboviruses. Cell Host Microbe 2011, 10, 75–88. [Google Scholar] [CrossRef] [PubMed]
- Odendaal, L.; Davis, A.S.; Venter, E.H. Insights into the Pathogenesis of Viral Haemorrhagic Fever Based on Virus Tropism and Tissue Lesions of Natural Rift Valley Fever. Viruses 2021, 13, 709. [Google Scholar] [CrossRef] [PubMed]
- Phoenix, I.; Nishiyama, S.; Lokugamage, N.; Hill, T.E.; Huante, M.B.; Slack, O.A.; Carpio, V.H.; Freiberg, A.N.; Ikegami, T. N-Glycans on the Rift Valley Fever Virus Envelope Glycoproteins Gn and Gc Redundantly Support Viral Infection Via Dc-Sign. Viruses 2016, 8, 149. [Google Scholar] [CrossRef] [PubMed]
- De Boer, S.M.; Kortekaas, J.; de Haan, C.A.; Rottier, P.J.; Moormann, R.J.; Bosch, B.J. Heparan Sulfate Facilitates Rift Valley Fever Virus Entry into the Cell. J. Virol. 2012, 86, 13767–13771. [Google Scholar] [CrossRef] [PubMed]
- Haisma, H.J.; Boesjes, M.; Beerens, A.M.; van der Strate, B.W.; Curiel, D.T.; Plüddemann, A.; Gordon, S.; Bellu, A.R. Scavenger Receptor A: A New Route for Adenovirus 5. Mol. Pharm. 2009, 6, 366–374. [Google Scholar] [CrossRef] [PubMed]
- MacLeod, D.T.; Nakatsuji, T.; Yamasaki, K.; Kobzik, L.; Gallo, R.L. Hsv-1 Exploits the Innate Immune Scavenger Receptor Marco to Enhance Epithelial Adsorption and Infection. Nat. Commun. 2013, 4, 1963. [Google Scholar] [CrossRef] [PubMed]
- Hise, A.G.; Traylor, Z.; Hall, N.B.; Sutherland, L.J.; Dahir, S.; Ermler, M.E.; Muiruri, S.; Muchiri, E.M.; Kazura, J.W.; LaBeaud, A.D.; et al. Association of Symptoms and Severity of Rift Valley Fever with Genetic Polymorphisms in Human Innate Immune Pathways. PLoS Negl. Trop. Dis. 2015, 9, e0003584. [Google Scholar] [CrossRef] [PubMed]
- Reed, C.; Lin, K.; Wilhelmsen, C.; Friedrich, B.; Nalca, A.; Keeney, A.; Donnelly, G.; Shamblin, J.; Hensley, L.E.; Olinger, G.; et al. Aerosol Exposure to Rift Valley Fever Virus Causes Earlier and More Severe Neuropathology in the Murine Model, Which Has Important Implications for Therapeutic Development. PLoS Negl. Trop. Dis. 2013, 7, e2156. [Google Scholar] [CrossRef]
- Boyles, D.A.; Schwarz, M.M.; Albe, J.R.; McMillen, C.M.; O’Malley, K.J.; Reed, D.S.; Hartman, A.L. Development of Rift Valley Fever Encephalitis in Rats Is Mediated by Early Infection of Olfactory Epithelium and Neuroinvasion across the Cribriform Plate. J. Gen. Virol. 2021, 102, 1522. [Google Scholar] [CrossRef]
- Dodd, K.A.; McElroy, A.K.; Jones, T.L.; Zaki, S.R.; Nichol, S.T.; Spiropoulou, C.F. Rift Valley Fever Virus Encephalitis Is Associated with an Ineffective Systemic Immune Response and Activated T Cell Infiltration into the Cns in an Immunocompetent Mouse Model. PLoS Negl. Trop. Dis. 2014, 8, e2874. [Google Scholar] [CrossRef]
- Wichgers Schreur, P.J.; Tacken, M.; Gutjahr, B.; Keller, M.; van Keulen, L.; Kant, J.; van de Water, S.; Lin, Y.; Eiden, M.; Rissmann, M.; et al. Vaccine Efficacy of Self-Assembled Multimeric Protein Scaffold Particles Displaying the Glycoprotein Gn Head Domain of Rift Valley Fever Virus. Vaccines 2021, 9, 301. [Google Scholar] [CrossRef] [PubMed]
- Wichgers Schreur, P.J.; van Keulen, L.; Kant, J.; Kortekaas, J. Four-Segmented Rift Valley Fever Virus-Based Vaccines Can Be Applied Safely in Ewes During Pregnancy. Vaccine 2017, 35, 3123–3128. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.R.; Steele, K.E.; Shamblin, J.; Honko, A.; Johnson, J.; Reed, C.; Kennedy, M.; Chapman, J.L.; Hensley, L.E. The Pathogenesis of Rift Valley Fever Virus in the Mouse Model. Virology 2010, 407, 256–267. [Google Scholar] [CrossRef] [PubMed]
- Walters, A.W.; Kujawa, M.R.; Albe, J.R.; Reed, D.S.; Klimstra, W.B.; Hartman, A.L. Vascular Permeability in the Brain Is a Late Pathogenic Event During Rift Valley Fever Virus Encephalitis in Rats. Virology 2019, 526, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Albe, J.R.; Boyles, D.A.; Walters, A.W.; Kujawa, M.R.; McMillen, C.M.; Reed, D.S.; Hartman, A.L. Neutrophil and Macrophage Influx into the Central Nervous System Are Inflammatory Components of Lethal Rift Valley Fever Encephalitis in Rats. PLoS Pathog. 2019, 15, e1007833. [Google Scholar] [CrossRef] [PubMed]
- Alrajhi, A.A.; Al-Semari, A.; Al-Watban, J. Rift Valley Fever Encephalitis. Emerg. Infect. Dis. 2004, 10, 554–555. [Google Scholar] [CrossRef]
- Van Velden, D.J.; Meyer, J.D.; Olivier, J.; Gear, J.H.S.; McIntosh, B. Rift Valley Fever Affecting Humans in South Africa-A Clinicopathological Study. S. Afr. Med. J. 1977, 51, 867–871. [Google Scholar] [CrossRef]
- Madani, T.A.; Al-Mazrou, Y.Y.; Al-Jeffri, M.H.; Mishkhas, A.A.; Al-Rabeah, A.M.; Turkistani, A.M.; Al-Sayed, M.O.; Abodahish, A.A.; Khan, A.S.; Ksiazek, T.G.; et al. Rift Valley Fever Epidemic in Saudi Arabia: Epidemiological, Clinical, and Laboratory Characteristics. Clin. Infect. Dis. 2003, 37, 1084–1092. [Google Scholar] [CrossRef] [PubMed]
- Dodd, K.A.; McElroy, A.K.; Jones, M.E.; Nichol, S.T.; Spiropoulou, C.F. Rift Valley Fever Virus Clearance and Protection from Neurologic Disease Are Dependent on Cd4+ T Cell and Virus-Specific Antibody Responses. J. Virol. 2013, 87, 6161–6171. [Google Scholar] [CrossRef]
- Lang, Y.; Henningson, J.; Jasperson, D.; Li, Y.; Lee, J.; Ma, J.; Li, Y.; Cao, N.; Liu, H.; Wilson, W.; et al. Mouse Model for the Rift Valley Fever Virus Mp12 Strain Infection. Vet. Microbiol. 2016, 195, 70–77. [Google Scholar] [CrossRef]
- Al-Hazmi, M.; Ayoola, E.A.; Abdurahman, M.; Banzal, S.; Ashraf, J.; El-Bushra, A.; Hazmi, A.; Abdullah, M.; Abbo, H.; Elamin, A.; et al. Epidemic Rift Valley Fever in Saudi Arabia: A Clinical Study of Severe Illness in Humans. Clin. Infect. Dis. 2003, 36, 245–252. [Google Scholar] [CrossRef]
- Barbeau, D.J.; Albe, J.R.; Nambulli, S.; Tilston-Lunel, N.L.; Hartman, A.L.; Lakdawala, S.S.; Klein, E.; Duprex, W.P.; McElroy, A.K. Rift Valley Fever Virus Infection Causes Acute Encephalitis in the Ferret. mSphere 2020, 5, e00798-20. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, G.; Lopez-Gil, E.; Warimwe, G.M.; Brun, A. Understanding Rift Valley Fever: Contributions of Animal Models to Disease Characterization and Control. Mol. Immunol. 2015, 66, 78–88. [Google Scholar] [CrossRef] [PubMed]
- Ross, T.M.; Bhardwaj, N.; Bissel, S.J.; Hartman, A.L.; Smith, D.R. Animal Models of Rift Valley Fever Virus Infection. Virus. Res. 2012, 163, 417–423. [Google Scholar] [CrossRef]
- Wonderlich, E.R.; Caroline, A.L.; McMillen, C.M.; Walters, A.W.; Reed, D.S.; Barratt-Boyes, S.M.; Hartman, A.L. Peripheral Blood Biomarkers of Disease Outcome in a Monkey Model of Rift Valley Fever Encephalitis. J. Virol. 2018, 92, e01662-17. [Google Scholar] [CrossRef]
- Michaely, L.M.; Rissmann, M.; Keller, M.; König, R.; von Arnim, F.; Eiden, M.; Rohn, K.; Baumgärtner, W.; Groschup, M.; Ulrich, R. Nsg-Mice Reveal the Importance of a Functional Innate and Adaptive Immune Response to Overcome Rvfv Infection. Viruses 2022, 14, 350. [Google Scholar] [CrossRef]
- Detje, C.N.; Meyer, T.; Schmidt, H.; Kreuz, D.; Rose, J.K.; Bechmann, I.; Prinz, M.; Kalinke, U. Local Type I Ifn Receptor Signaling Protects against Virus Spread within the Central Nervous System. J. Immunol. 2009, 182, 2297–2304. [Google Scholar] [CrossRef] [PubMed]
- Bouloy, M.; Janzen, C.; Vialat, P.; Khun, H.; Pavlovic, J.; Huerre, M.; Haller, O. Genetic Evidence for an Interferon-Antagonistic Function of Rift Valley Fever Virus Nonstructural Protein Nss. J. Virol. 2001, 75, 1371–1377. [Google Scholar] [CrossRef] [PubMed]
- Ikegami, T.; Narayanan, K.; Won, S.; Kamitani, W.; Peters, C.J.; Makino, S. Dual Functions of Rift Valley Fever Virus Nss Protein: Inhibition of Host Mrna Transcription and Post-Transcriptional Downregulation of Protein Kinase Pkr. Ann. N. Y. Acad. Sci. 2009, 1171 (Suppl. 1), E75–E85. [Google Scholar] [CrossRef]
- Ikegami, T.; Narayanan, K.; Won, S.; Kamitani, W.; Peters, C.J.; Makino, S. Rift Valley Fever Virus Nss Protein Promotes Post-Transcriptional Downregulation of Protein Kinase Pkr and Inhibits Eif2alpha Phosphorylation. PLoS Pathog. 2009, 5, e1000287. [Google Scholar] [CrossRef]
- Kortekaas, J.; Oreshkova, N.; van Keulen, L.; Kant, J.; Bosch, B.J.; Bouloy, M.; Moulin, V.; Goovaerts, D.; Moormann, R.J. Comparative Efficacy of Two Next-Generation Rift Valley Fever Vaccines. Vaccine 2014, 32, 4901–4908. [Google Scholar] [CrossRef]
- Ivashkiv, L.B.; Donlin, L.T. Regulation of Type I Interferon Responses. Nat. Rev. Immunol. 2014, 14, 36–49. [Google Scholar] [CrossRef] [PubMed]
- Muller, R.; Saluzzo, J.F.; Lopez, N.; Dreier, T.; Turell, M.; Smith, J.; Bouloy, M. Characterization of Clone 13, a Naturally Attenuated Avirulent Isolate of Rift Valley Fever Virus, Which Is Altered in the Small Segment. Am. J. Trop. Med. Hyg. 1995, 53, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Vialat, P.; Billecocq, A.; Kohl, A.; Bouloy, M. The S Segment of Rift Valley Fever Phlebovirus (Bunyaviridae) Carries Determinants for Attenuation and Virulence in Mice. J. Virol. 2000, 74, 1538–1543. [Google Scholar] [CrossRef] [PubMed]
- Habjan, M.; Pichlmair, A.; Elliott, R.M.; Overby, A.K.; Glatter, T.; Gstaiger, M.; Superti-Furga, G.; Unger, H.; Weber, F. Nss Protein of Rift Valley Fever Virus Induces the Specific Degradation of the Double-Stranded Rna-Dependent Protein Kinase. J. Virol. 2009, 83, 4365–4375. [Google Scholar] [CrossRef] [PubMed]
- Le May, N.; Dubaele, S.; Proietti De Santis, L.; Billecocq, A.; Bouloy, M.; Egly, J.M. Tfiih Transcription Factor, a Target for the Rift Valley Hemorrhagic Fever Virus. Cell 2004, 116, 541–550. [Google Scholar] [CrossRef]
- Mansuroglu, Z.; Josse, T.; Gilleron, J.; Billecocq, A.; Leger, P.; Bouloy, M.; Bonnefoy, E. Nonstructural Nss Protein of Rift Valley Fever Virus Interacts with Pericentromeric DNA Sequences of the Host Cell, Inducing Chromosome Cohesion and Segregation Defects. J. Virol. 2010, 84, 928–939. [Google Scholar] [CrossRef]
- Hartman, A.L.; Powell, D.S.; Bethel, L.M.; Caroline, A.L.; Schmid, R.J.; Oury, T.; Reed, D.S. Aerosolized Rift Valley Fever Virus Causes Fatal Encephalitis in African Green Monkeys and Common Marmosets. J. Virol. 2014, 88, 2235–2245. [Google Scholar] [CrossRef]
- Nicholas, D.E.; Jacobsen, K.H.; Waters, N.M. Risk Factors Associated with Human Rift Valley Fever Infection: Systematic Review and Meta-Analysis. Trop. Med. Int. Health 2014, 19, 1420–1429. [Google Scholar] [CrossRef]
- Van Riel, D.; Verdijk, R.; Kuiken, T. The Olfactory Nerve: A Shortcut for Influenza and Other Viral Diseases into the Central Nervous System. J. Pathol. 2015, 235, 277–287. [Google Scholar] [CrossRef]
- Reed, C.; Steele, K.E.; Honko, A.; Shamblin, J.; Hensley, L.E.; Smith, D.R. Ultrastructural Study of Rift Valley Fever Virus in the Mouse Model. Virology 2012, 431, 58–70. [Google Scholar] [CrossRef]
- Chhatbar, C.; Detje, C.N.; Grabski, E.; Borst, K.; Spanier, J.; Ghita, L.; Elliott, D.A.; Jordão, M.J.C.; Mueller, N.; Sutton, J.; et al. Type I Interferon Receptor Signaling of Neurons and Astrocytes Regulates Microglia Activation During Viral Encephalitis. Cell Rep. 2018, 25, 118–129.e4. [Google Scholar] [CrossRef]
- Detje, C.N.; Lienenklaus, S.; Chhatbar, C.; Spanier, J.; Prajeeth, C.K.; Soldner, C.; Tovey, M.G.; Schlüter, D.; Weiss, S.; Stangel, M.; et al. Upon Intranasal Vesicular Stomatitis Virus Infection, Astrocytes in the Olfactory Bulb Are Important Interferon Beta Producers That Protect from Lethal Encephalitis. J. Virol. 2015, 89, 2731–2738. [Google Scholar] [CrossRef] [PubMed]
- Pfefferkorn, C.; Kallfass, C.; Lienenklaus, S.; Spanier, J.; Kalinke, U.; Rieder, M.; Conzelmann, K.K.; Michiels, T.; Staeheli, P. Abortively Infected Astrocytes Appear to Represent the Main Source of Interferon Beta in the Virus-Infected Brain. J. Virol. 2016, 90, 2031–2038. [Google Scholar] [CrossRef]
- Hum, N.R.; Bourguet, F.A.; Sebastian, A.; Lam, D.; Phillips, A.M.; Sanchez, K.R.; Rasley, A.; Loots, G.G.; Weilhammer, D.R. MAVS Mediates a Protective Immune Response in the Brain to Rift Valley Fever Virus. PLoS Pathog. 2022, 18, e1010231. [Google Scholar] [CrossRef] [PubMed]
- Guan, J.; Miah, S.M.; Wilson, Z.S.; Erick, T.K.; Banh, C.; Brossay, L. Role of Type I Interferon Receptor Signaling on NK Cell Development and Functions. PLoS ONE 2014, 9, e111302. [Google Scholar] [CrossRef] [PubMed]
- Kavrochorianou, N.; Evangelidou, M.; Markogiannaki, M.; Tovey, M.; Thyphronitis, G.; Haralambous, S. IFNAR Signaling Directly Modulates T Lymphocyte Activity, Resulting in Milder Experimental Autoimmune Encephalomyelitis Development. J. Leukoc. Biol. 2016, 99, 175–188. [Google Scholar] [CrossRef]
- Metidji, A.; Rieder, S.A.; Glass, D.D.; Cremer, I.; Punkosdy, G.A.; Shevach, E.M. IFN-Alpha/beta Receptor Signaling Promotes Regulatory T Cell Development and Function Under Stress Conditions. J. Immunol. 2015, 194, 4265–4276. [Google Scholar] [CrossRef]
- Taylor, M.P.; Enquist, L.W. Axonal Spread of Neuroinvasive Viral Infections. Trends Microbiol. 2015, 23, 283–288. [Google Scholar] [CrossRef] [PubMed]
- Miller, K.D.; Schnell, M.J.; Rall, G.F. Keeping It in Check: Chronic Viral Infection and Antiviral Immunity in the Brain. Nat. Rev. Neurosci. 2016, 17, 766–776. [Google Scholar] [CrossRef] [PubMed]
- Borrego, B.; Brun, A. A Hyper-Attenuated Variant of Rift Valley Fever Virus Generated by a Mutagenic Drug (Favipiravir) Unveils Potential Virulence Markers. Front Microbiol. 2020, 11, 621463. [Google Scholar] [CrossRef] [PubMed]
- Ritter, M.; Bouloy, M.; Vialat, P.; Janzen, C.; Haller, O.; Frese, M. Resistance to Rift Valley Fever Virus in Rattus Norvegicus: Genetic Variability within Certain ‘Inbred’ Strains. J. Gen. Virol. 2000, 81, 2683–2688. [Google Scholar] [CrossRef] [PubMed]
- Müller, U.; Steinhoff, U.; Reis, L.F.; Hemmi, S.; Pavlovic, J.; Zinkernagel, R.M.; Aguet, M. Functional Role of Type I and Type II Interferons in Antiviral Defense. Science 1994, 264, 1918–1921. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, I.; Jansen, S.; Fall, G.; Lorenzen, S.; Rudolf, M.; Huber, K.; Heitmann, A.; Schicht, S.; Ndiaye, E.H.; Watson, M.; et al. Rna Interference Restricts Rift Valley Fever Virus in Multiple Insect Systems. mSphere 2017, 2, e00090-17. [Google Scholar] [CrossRef]
- Haist, V.; Ulrich, R.; Kalkuhl, A.; Deschl, U.; Baumgärtner, W. Distinct Spatio-Temporal Extracellular Matrix Accumulation within Demyelinated Spinal Cord Lesions in Theiler’s Murine Encephalomyelitis. Brain Pathol. 2012, 22, 188–204. [Google Scholar] [CrossRef] [PubMed]
- Riedelsheimer, B.U.W. Romeis Mikroskopische Technik; Springer: Berlin/Heidelberg, Germany, 2010; Volume 18. [Google Scholar]
- Allnoch, L.; Beythien, G.; Leitzen, E.; Becker, K.; Kaup, F.J.; Stanelle-Bertram, S.; Schaumburg, B.; Mounogou Kouassi, N.; Beck, S.; Zickler, M.; et al. Vascular Inflammation Is Associated with Loss of Aquaporin 1 Expression on Endothelial Cells and Increased Fluid Leakage in SARS-CoV-2 Infected Golden Syrian Hamsters. Viruses 2021, 13, 639. [Google Scholar] [CrossRef]
- Allnoch, L.; Baumgärtner, W.; Hansmann, F. Impact of Astrocyte Depletion Upon Inflammation and Demyelination in a Murine Animal Model of Multiple Sclerosis. Int. J. Mol. Sci. 2019, 20, 3922. [Google Scholar] [CrossRef] [PubMed]
- Gregor, K.M.; Michaely, L.M.; Gutjahr, B.; Rissmann, M.; Keller, M.; Dornbusch, S.; Naccache, F.; Schön, K.; Jansen, S.; Heitmann, A.; et al. Rift Valley Fever Virus Detection in Susceptible Hosts with Special Emphasis in Insects. Sci. Rep. 2021, 11, 9822. [Google Scholar] [CrossRef]
- Huang, B.; Zdora, I.; de Buhr, N.; Lehmbecker, A.; Baumgärtner, W.; Leitzen, E. Phenotypical Peculiarities and Species-Specific Differences of Canine and Murine Satellite Glial Cells of Spinal Ganglia. J. Cell Mol. Med. 2021, 25, 6909–6924. [Google Scholar] [CrossRef]





| WT | IFNAR−/− | |||||
|---|---|---|---|---|---|---|
| Infection Dose | 104 PFU (n = 5, 7–21 dpi) * | 103 PFU (n = 10, 21 dpi) | 103 PFU (n = 10, 2–21 dpi) ** | |||
| Staining | HE | RVF-Ag | HE | RVF-Ag | HE | RVF-Ag |
| Brain | multifocal neuronal necrosis of varying severity (n = 4) multifocal mild (n = 2) to moderate (n = 2) leptomeningitis | many neurons (n = 4) | focal mild meningitis (n = 2) | n.d. | focal mild neuronal necrosis (n = 1) multifocal mild meningitis (n = 2) | few neurons (n = 1) |
| Spinal cord | multifocal mild neuronal necrosis (n = 2) | many neurons (n = 2) | n.s.m.l. | n.d. | n.s.m.l. | n.d. |
| Liver | n.s.m.l. | n.d. | n.s.m.l. | n.d. | diffuse hepatocellular necrosis (n = 5) | many hepatocytes (n = 5) |
| Nose | n.s.m.l. | n.d. | n.s.m.l. | n.d. | multifocal mild necro-suppurative rhinitis | single cells in olfactory and respiratory mucosa (n = 1) |
| Spleen | n.s.m.l. | n.d. | n.s.m.l. | n.d. | multifocal lymphocyte necrosis (n = 5) | many lymphocytes (n = 5) |
| Blood vessels | n.s.m.l. | n.d. | n.s.m.l. | n.d. | n.s.m.l. | many monocytes (n = 5) |
| Bone marrow | n.s.m.l. | n.d. | n.s.m.l. | n.d. | n.s.m.l. | many hematopoietic precursor cells (n = 5) |
| Antigen | Host Species | Pretreatment | Dilution | Supplier |
|---|---|---|---|---|
| CD3 (T lymphocyte) | rabbit | microwave, citrate buffer, 20 min | 1:2000 | DakoCytomation, Hamburg, Germany |
| CD45R (B lymphocyte) | rat | microwave, citrate buffer, 20 min | 1:1000 | Becton Dickinson, Franklin Lakes, USA |
| Cleaved caspase 3 (apoptotic cells) | rabbit | microwave, citrate buffer, 20 min | 1:200 | Cell Signaling Technology, Cambridge, UK |
| Entactin/Nidogen-1 | rabbit | microwave, citrate buffer, 20 min | 1:3000 | Abcam, Cambridge, UK |
| GFAP (astrocyte) | rabbit | None | 1:1000 | DakoCytomation, Hamburg, Germany |
| Iba-1 (macrophage/microglia) | rabbit | microwave, citrate buffer, 20 min | 1:1000 | Wako Chemicals, Richmond, USA |
| Laminin | rabbit | None | 1:50 | Sigma-Aldrich Chemie GmbH, Taufkirchen, Germany |
| Non-phosphorylated neurofilament | mouse | microwave, citrate buffer, 20 min | 1:2000 | BioLegend, San Diego, USA |
| RVFV nucleoprotein | sheep | None | 1:50,000 | Friedrich-Loeffler-Institute, Greifswald, Germany [70] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Michaely, L.M.; Schuwerk, L.; Allnoch, L.; Schön, K.; Waltl, I.; Larsen, P.-K.; Pavlou, A.; Prajeeth, C.K.; Rimmelzwaan, G.F.; Becker, S.C.; et al. Intact Type I Interferon Receptor Signaling Prevents Hepatocellular Necrosis but Not Encephalitis in a Dose-Dependent Manner in Rift Valley Fever Virus Infected Mice. Int. J. Mol. Sci. 2022, 23, 12492. https://doi.org/10.3390/ijms232012492
Michaely LM, Schuwerk L, Allnoch L, Schön K, Waltl I, Larsen P-K, Pavlou A, Prajeeth CK, Rimmelzwaan GF, Becker SC, et al. Intact Type I Interferon Receptor Signaling Prevents Hepatocellular Necrosis but Not Encephalitis in a Dose-Dependent Manner in Rift Valley Fever Virus Infected Mice. International Journal of Molecular Sciences. 2022; 23(20):12492. https://doi.org/10.3390/ijms232012492
Chicago/Turabian StyleMichaely, Lukas Mathias, Lukas Schuwerk, Lisa Allnoch, Kathleen Schön, Inken Waltl, Pia-Katharina Larsen, Andreas Pavlou, Chittappen Kandiyil Prajeeth, Guus F. Rimmelzwaan, Stefanie C. Becker, and et al. 2022. "Intact Type I Interferon Receptor Signaling Prevents Hepatocellular Necrosis but Not Encephalitis in a Dose-Dependent Manner in Rift Valley Fever Virus Infected Mice" International Journal of Molecular Sciences 23, no. 20: 12492. https://doi.org/10.3390/ijms232012492
APA StyleMichaely, L. M., Schuwerk, L., Allnoch, L., Schön, K., Waltl, I., Larsen, P.-K., Pavlou, A., Prajeeth, C. K., Rimmelzwaan, G. F., Becker, S. C., Kalinke, U., Baumgärtner, W., & Gerhauser, I. (2022). Intact Type I Interferon Receptor Signaling Prevents Hepatocellular Necrosis but Not Encephalitis in a Dose-Dependent Manner in Rift Valley Fever Virus Infected Mice. International Journal of Molecular Sciences, 23(20), 12492. https://doi.org/10.3390/ijms232012492







