Tuning Immobilized Enzyme Features by Combining Solid-Phase Physicochemical Modification and Mineralization
Abstract
:1. Introduction
2. Results and Discussion
2.1. Preparation of the Immobilized and Chemically Modified TLL-Biocatalysts
2.2. Modification of Octyl-TLL with Different Phosphate Salts
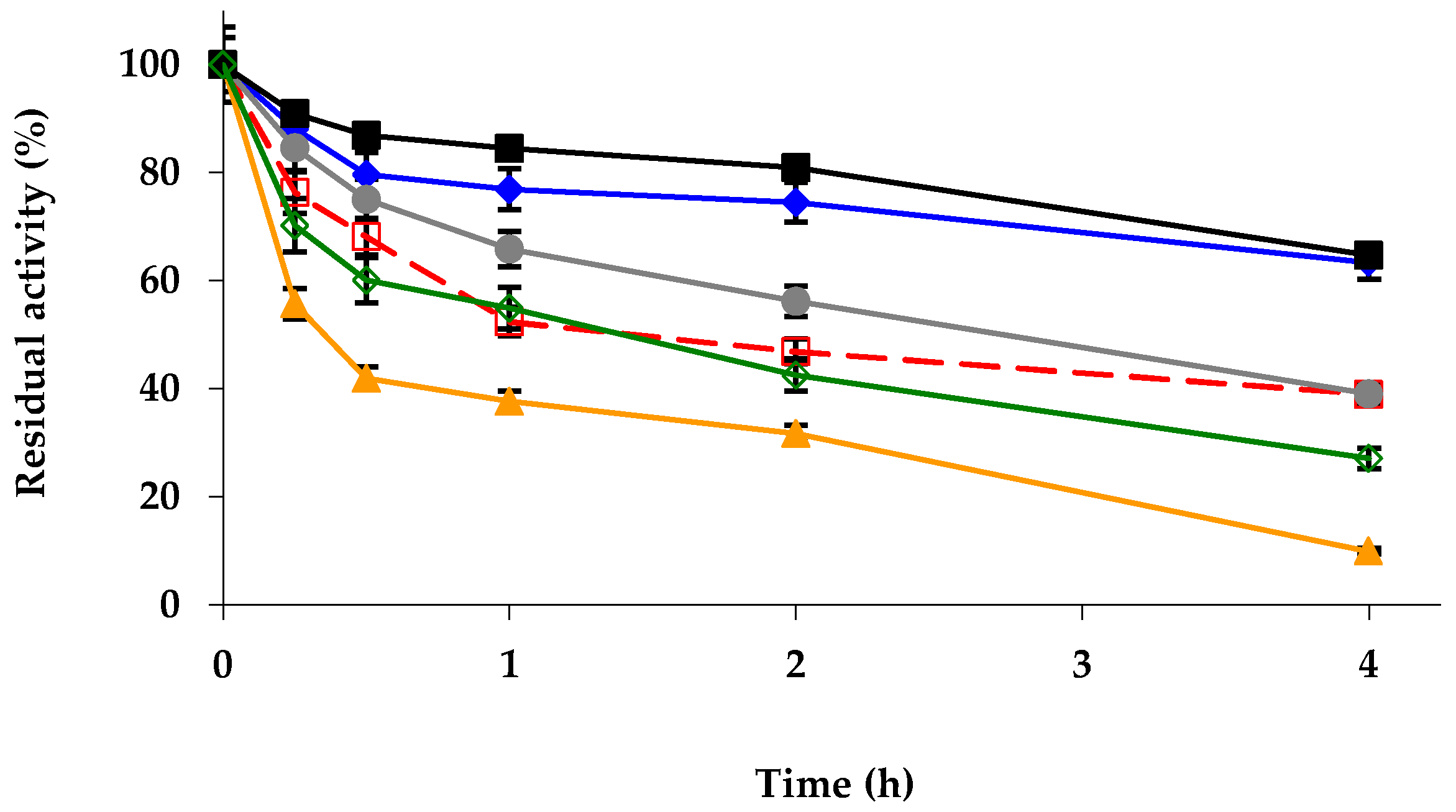
2.3. Effect of Mineralization on the Immobilized Enzyme Previously Modified
2.4. Preparation of the Immobilized and Chemically Modified CALB-Biocatalysts
2.5. Mineralization of Octyl-CALB
2.6. Effect of Mineralization on the Immobilized Enzyme Previously Modified
3. Materials and Methods
3.1. Materials
3.2. Methods
3.2.1. Immobilization of Lipases on Octyl-Agarose Beads
3.2.2. Immobilization of Lipases on Octyl-Agarose Beads
3.2.3. Modification of Immobilized Enzyme with Metallic Salt/Phosphate
3.2.4. Thermal Inactivation of Different Lipase Biocatalysts
3.2.5. Determination of the Biocatalysts Activities versus Different Substrates
Hydrolysis of p-NPB
Hydrolysis of Triacetin
Hydrolysis of R- or S-Methyl Mandelate
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sheldon, R.A.; Woodley, J.M. Role of biocatalysis in sustainable chemistry. Chem. Rev. 2018, 118, 801–838. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.-M.; Han, S.-S.; Kim, H.-S. Industrial applications of enzyme biocatalysis: Current status and future aspects. Biotechnol. Adv. 2015, 33, 1443–1454. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Snajdrova, R.; Moore, J.C.; Baldenius, K.; Bornscheuer, U.T. Biocatalysis: Enzymatic synthesis for industrial applications. Angew. Chemie Int. Ed. 2021, 60, 88–119. [Google Scholar] [CrossRef] [PubMed]
- Ferrer, M.; Martínez-Martínez, M.; Bargiela, R.; Streit, W.R.; Golyshina, O.V.; Golyshin, P.N. Estimating the success of enzyme bioprospecting through metagenomics: Current status and future trends. Microb. Biotechnol. 2016, 9, 22–34. [Google Scholar] [CrossRef]
- Fernández-Arrojo, L.; Guazzaroni, M.-E.; López-Cortés, N.; Beloqui, A.; Ferrer, M. Metagenomic era for biocatalyst identification. Curr. Opin. Biotechnol. 2010, 21, 725–733. [Google Scholar] [CrossRef] [PubMed]
- Guazzaroni, M.; Silva-Rocha, R.; Ward, R.J. Synthetic biology approaches to improve biocatalyst identification in metagenomic library screening. Microb. Biotechnol. 2015, 8, 52–64. [Google Scholar] [CrossRef] [PubMed]
- Araújo, R.; Silva, C.; O’Neill, A.; Micaelo, N.; Guebitz, G.; Soares, C.M.; Casal, M.; Cavaco-Paulo, A. Tailoring cutinase activity towards polyethylene terephthalate and polyamide 6,6 fibers. J. Biotechnol. 2007, 128, 849–857. [Google Scholar] [CrossRef] [Green Version]
- Woodyer, R.; van der Donk, W.A.; Zhao, H. Relaxing the nicotinamide cofactor specificity of phosphite dehydrogenase by rational design. Biochemistry 2003, 42, 11604–11614. [Google Scholar] [CrossRef]
- Arnold, F.H. Directed evolution: Bringing new chemistry to life. Angew. Chemie Int. Ed. 2018, 57, 4143–4148. [Google Scholar] [CrossRef] [Green Version]
- Kan, S.B.J.; Lewis, R.D.; Chen, K.; Arnold, F.H. Directed evolution of cytochrome c for carbon–silicon bond formation: Bringing silicon to life. Science 2016, 354, 1048–1051. [Google Scholar] [CrossRef]
- Renata, H.; Wang, Z.J.; Arnold, F.H. Expanding the enzyme universe: Accessing non-natural reactions by mechanism-guided directed evolution. Angew. Chemie Int. Ed. 2015, 54, 3351–3367. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Díaz-Rodríguez, A.; Davis, B.G. Chemical modification in the creation of novel biocatalysts. Curr. Opin. Chem. Biol. 2011, 15, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Pagar, A.D.; Patil, M.D.; Flood, D.T.; Yoo, T.H.; Dawson, P.E.; Yun, H. Recent advances in biocatalysis with chemical modification and expanded amino acid alphabet. Chem. Rev. 2021, 121, 6173–6245. [Google Scholar] [CrossRef] [PubMed]
- Carballares, D.; Morellon-Sterling, R.; Fernandez-Lafuente, R. Design of artificial enzymes bearing several active centers: New trends, opportunities and problems. Int. J. Mol. Sci. 2022, 23, 5304. [Google Scholar] [CrossRef]
- Alonso, S.; Santiago, G.; Cea-Rama, I.; Fernandez-Lopez, L.; Coscolín, C.; Modregger, J.; Ressmann, A.K.; Martínez-Martínez, M.; Marrero, H.; Bargiela, R.; et al. Genetically engineered proteins with two active sites for enhanced biocatalysis and synergistic chemo- and biocatalysis. Nat. Catal. 2020, 3, 319–328. [Google Scholar] [CrossRef]
- Santiago, G.; Martínez-Martínez, M.; Alonso, S.; Bargiela, R.; Coscolín, C.; Golyshin, P.N.; Guallar, V.; Ferrer, M. Rational engineering of multiple active sites in an ester hydrolase. Biochemistry 2018, 57, 2245–2255. [Google Scholar] [CrossRef] [Green Version]
- Roda, S.; Fernandez-Lopez, L.; Benedens, M.; Bollinger, A.; Thies, S.; Schumacher, J.; Coscolín, C.; Kazemi, M.; Santiago, G.; Gertzen, C.G.W.; et al. A plurizyme with transaminase and hydrolase activity catalyzes cascade reactions. Angew. Chemie Int. Ed. 2022, 134, e202207344. [Google Scholar] [CrossRef]
- Sheldon, R.A.; van Pelt, S. Enzyme immobilisation in biocatalysis: Why, what and how. Chem. Soc. Rev. 2013, 42, 6223–6235. [Google Scholar] [CrossRef] [Green Version]
- DiCosimo, R.; McAuliffe, J.; Poulose, A.J.; Bohlmann, G. Industrial use of immobilized enzymes. Chem. Soc. Rev. 2013, 42, 6437. [Google Scholar] [CrossRef]
- Liese, A.; Hilterhaus, L. Evaluation of immobilized enzymes for industrial applications. Chem. Soc. Rev. 2013, 42, 6236–6249. [Google Scholar] [CrossRef]
- Rodrigues, R.C.; Berenguer-Murcia, Á.; Carballares, D.; Morellon-Sterling, R.; Fernandez-Lafuente, R. Stabilization of enzymes via immobilization: Multipoint covalent attachment and other stabilization strategies. Biotechnol. Adv. 2021, 52, 107821. [Google Scholar] [CrossRef] [PubMed]
- Iyer, P.V.; Ananthanarayan, L. Enzyme stability and stabilization-Aqueous and non-aqueous environment. Process Biochem. 2008, 43, 1019–1032. [Google Scholar] [CrossRef]
- Mateo, C.; Palomo, J.M.; Fernandez-Lorente, G.; Guisan, J.M.; Fernandez-Lafuente, R. Improvement of enzyme activity, stability and selectivity via immobilization techniques. Enzyme Microb. Technol. 2007, 40, 1451–1463. [Google Scholar] [CrossRef]
- Rodrigues, R.C.; Ortiz, C.; Berenguer-Murcia, Á.; Torres, R.; Fernández-Lafuente, R. Modifying enzyme activity and selectivity by immobilization. Chem. Soc. Rev. 2013, 42, 6290–6307. [Google Scholar] [CrossRef]
- Garcia-Galan, C.; Berenguer-Murcia, Á.; Fernandez-Lafuente, R.; Rodrigues, R.C. Potential of different enzyme immobilization strategies to improve enzyme performance. Adv. Synth. Catal. 2011, 353, 2885–2904. [Google Scholar] [CrossRef]
- Barbosa, O.; Ortiz, C.; Berenguer-Murcia, Á.; Torres, R.; Rodrigues, R.C.; Fernandez-Lafuente, R. Strategies for the one-step immobilization-purification of enzymes as industrial biocatalysts. Biotechnol. Adv. 2015, 33, 435–456. [Google Scholar] [CrossRef] [Green Version]
- Rodrigues, R.C.; Virgen-Ortíz, J.J.; dos Santos, J.C.S.; Berenguer-Murcia, Á.; Alcantara, A.R.; Barbosa, O.; Ortiz, C.; Fernandez-Lafuente, R. Immobilization of lipases on hydrophobic supports: Immobilization mechanism, advantages, problems, and solutions. Biotechnol. Adv. 2019, 37, 746–770. [Google Scholar] [CrossRef] [Green Version]
- Fernandez-Lafuente, R. Stabilization of multimeric enzymes: Strategies to prevent subunit dissociation. Enzyme Microb. Technol. 2009, 45, 405–418. [Google Scholar] [CrossRef]
- Rodrigues, R.C.; Berenguer-Murcia, Á.; Fernandez-Lafuente, R. Coupling chemical modification and immobilization to improve the catalytic performance of enzymes. Adv. Synth. Catal. 2011, 353, 2216–2238. [Google Scholar] [CrossRef]
- Rueda, N.; dos Santos, J.C.S.; Ortiz, C.; Torres, R.; Barbosa, O.; Rodrigues, R.C.; Berenguer-Murcia, Á.; Fernandez-Lafuente, R. Chemical modification in the design of immobilized enzyme biocatalysts: Drawbacks and opportunities. Chem. Rec. 2016, 16, 1436–1455. [Google Scholar] [CrossRef]
- Rodrigues, R.C.; Barbosa, O.; Ortiz, C.; Berenguer-Murcia, Á.; Torres, R.; Fernandez-Lafuente, R. Amination of enzymes to improve biocatalyst performance: Coupling genetic modification and physicochemical tools. RSC Adv. 2014, 4, 38350–38374. [Google Scholar] [CrossRef] [Green Version]
- Guimarães, J.R.; Carballares, D.; Rocha-Martin, J.; Tardioli, P.W.; Fernandez-Lafuente, R. Stabilization of immobilized lipases by treatment with metallic phosphate salts. Int. J. Biol. Macromol. 2022, 213, 43–54. [Google Scholar] [CrossRef] [PubMed]
- Guimarães, J.R.; Carballares, D.; Tardioli, P.W.; Rocha-Martin, J.; Fernandez-Lafuente, R. Tuning immobilized commercial lipase preparations features by simple treatment with metallic phosphate salts. Molecules 2022, 27, 4486. [Google Scholar] [CrossRef] [PubMed]
- Ge, J.; Lei, J.; Zare, R.N. Protein–inorganic hybrid nanoflowers. Nat. Nanotechnol. 2012, 7, 428–432. [Google Scholar] [CrossRef]
- da Costa, F.P.; Cipolatti, E.P.; Furigo Junior, A.; Oliveira Henriques, R. Nanoflowers: A new approach of enzyme immobilization. Chem. Rec. 2022, 22, e202100293. [Google Scholar] [CrossRef]
- Wu, X.; Hou, M.; Ge, J. Metal-organic frameworks and inorganic nanoflowers: A type of emerging inorganic crystal nanocarrier for enzyme immobilization. Catal. Sci. Technol. 2015, 5, 5077–5085. [Google Scholar] [CrossRef]
- Altinkaynak, C.; Tavlasoglu, S.; Özdemir, N.; Ocsoy, I. A new generation approach in enzyme immobilization: Organic-inorganic hybrid nanoflowers with enhanced catalytic activity and stability. Enzyme Microb. Technol. 2016, 93, 105–112. [Google Scholar] [CrossRef]
- Cui, J.; Jia, S. Organic–inorganic hybrid nanoflowers: A novel host platform for immobilizing biomolecules. Coord. Chem. Rev. 2017, 352, 249–263. [Google Scholar] [CrossRef]
- Shende, P.; Kasture, P.; Gaud, R.S. Nanoflowers: The future trend of nanotechnology for multi-applications. Artif. Cells Nanomed. Biotechnol. 2018, 46, 413–422. [Google Scholar] [CrossRef] [Green Version]
- Cui, J.; Zhao, Y.; Liu, R.; Zhong, C.; Jia, S. Surfactant-activated lipase hybrid nanoflowers with enhanced enzymatic performance. Sci. Rep. 2016, 6, 27928. [Google Scholar] [CrossRef]
- Ke, C.; Fan, Y.; Chen, Y.; Xu, L.; Yan, Y. A new lipase–inorganic hybrid nanoflower with enhanced enzyme activity. RSC Adv. 2016, 6, 19413–19416. [Google Scholar] [CrossRef]
- Lei, Z.; Gao, C.; Chen, L.; He, Y.; Ma, W.; Lin, Z. Recent advances in biomolecule immobilization based on self-assembly: Organic–inorganic hybrid nanoflowers and metal–organic frameworks as novel substrates. J. Mater. Chem. B 2018, 6, 1581–1594. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Wang, X.; Yang, J.; Han, H.; Li, Q.; Tang, J. Lipase-inorganic hybrid nanoflower constructed through biomimetic mineralization: A new support for biodiesel synthesis. J. Colloid Interface Sci. 2018, 514, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Mohammed Al-Antaki, A.H.; Igder, A.; Stubbs, K.A.; Su, P.; Zhang, W.; Weiss, G.A.; Raston, C.L. Vortex fluidic-mediated fabrication of fast gelated silica hydrogels with embedded laccase nanoflowers for real-time biosensing under flow. ACS Appl. Mater. Interfaces 2020, 12, 51999–52007. [Google Scholar] [CrossRef] [PubMed]
- Luo, M.; Li, M.; Jiang, S.; Shao, H.; Razal, J.; Wang, D.; Fang, J. Supported growth of inorganic-organic nanoflowers on 3D hierarchically porous nanofibrous membrane for enhanced enzymatic water treatment. J. Hazard. Mater. 2020, 381, 120947. [Google Scholar] [CrossRef]
- Sun, T.; Fu, M.; Xing, J.; Ge, Z. Magnetic nanoparticles encapsulated laccase nanoflowers: Evaluation of enzymatic activity and reusability for degradation of malachite green. Water Sci. Technol. 2020, 81, 29–39. [Google Scholar] [CrossRef]
- Fotiadou, R.; Patila, M.; Hammami, M.A.; Enotiadis, A.; Moschovas, D.; Tsirka, K.; Spyrou, K.; Giannelis, E.P.; Avgeropoulos, A.; Paipetis, A.; et al. Development of effective lipase-hybrid nanoflowers enriched with carbon and magnetic nanomaterials for biocatalytic transformations. Nanomaterials 2019, 9, 808. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Fei, X.; Tian, J.; Li, Y.; Zhi, H.; Wang, K.; Xu, L.; Wang, Y. Synthesis and continuous catalytic application of alkaline protease nanoflowers–PVA composite hydrogel. Catal. Commun. 2018, 116, 5–9. [Google Scholar] [CrossRef]
- Sun, B.; Wang, Z.; Wang, X.; Qiu, M.; Zhang, Z.; Wang, Z.; Cui, J.; Jia, S. Paper-based biosensor based on phenylalnine ammonia lyase hybrid nanoflowers for urinary phenylalanine measurement. Int. J. Biol. Macromol. 2021, 166, 601–610. [Google Scholar] [CrossRef]
- Lee, I.; Cheon, H.J.; Adhikari, M.D.; Tran, T.D.; Yeon, K.-M.; Kim, M.I.; Kim, J. Glucose oxidase-copper hybrid nanoflowers embedded with magnetic nanoparticles as an effective antibacterial agent. Int. J. Biol. Macromol. 2020, 155, 1520–1531. [Google Scholar] [CrossRef]
- Bilal, M.; Iqbal, H.M.N. Armoring bio-catalysis via structural and functional coordination between nanostructured materials and lipases for tailored applications. Int. J. Biol. Macromol. 2021, 166, 818–838. [Google Scholar] [CrossRef] [PubMed]
- Guimarães, J.R.; Carballares, D.; Rocha-martin, J.R.; Tardioli, P.W.; Fernandez-Lafuente, R. The immobilization protocol greatly alters the effects of metal phosphate modification on the activity/stability of immobilized lipases. Int. J. Biol. Macromol. 2022; in press. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Galan, C.; dos Santos, J.C.S.; Barbosa, O.; Torres, R.; Pereira, E.B.; Corberan, V.C.; Gonçalves, L.R.B.; Fernandez-Lafuente, R. Tuning of Lecitase features via solid-phase chemical modification: Effect of the immobilization protocol. Process Biochem. 2014, 49, 604–616. [Google Scholar] [CrossRef]
- Ruiz, M.; Galvis, M.; Barbosa, O.; Ortiz, C.; Torres, R.; Fernandez-Lafuente, R. Solid-phase modification with succinic polyethyleneglycol of aminated lipase B from Candida antarctica: Effect of the immobilization protocol on enzyme catalytic properties. J. Mol. Catal. B Enzym. 2013, 87, 75–82. [Google Scholar] [CrossRef]
- Barbosa, O.; Ruiz, M.; Ortiz, C.; Fernández, M.; Torres, R.; Fernandez-Lafuente, R. Modulation of the properties of immobilized CALB by chemical modification with 2,3,4-trinitrobenzenesulfonate or ethylendiamine. Advantages of using adsorbed lipases on hydrophobic supports. Process Biochem. 2012, 47, 867–876. [Google Scholar] [CrossRef]
- Galvis, M.; Barbosa, O.; Torres, R.; Ortiz, C.; Fernandez-Lafuente, R. Effect of solid-phase chemical modification on the features of the lipase from Thermomyces lanuginosus. Process Biochem. 2012, 47, 460–466. [Google Scholar] [CrossRef]
- Fernandez-Lopez, L.; Rueda, N.; Bartolome-Cabrero, R.; Rodriguez, M.D.; Albuquerque, T.L.; Dos Santos, J.C.S.; Barbosa, O.; Fernandez-Lafuente, R. Improved immobilization and stabilization of lipase from Rhizomucor miehei on octyl-glyoxyl agarose beads by using CaCl2. Process Biochem. 2016, 51, 48–52. [Google Scholar] [CrossRef]
- Fernandez-Lopez, L.; Bartolome-Cabrero, R.; Rodriguez, M.D.; Dos Santos, C.S.; Rueda, N.; Fernandez-Lafuente, R. Stabilizing effects of cations on lipases depend on the immobilization protocol. RSC Adv. 2015, 5, 83868–83875. [Google Scholar] [CrossRef]
- Carpenter, B.P.; Talosig, A.R.; Mulvey, J.T.; Merham, J.G.; Esquivel, J.; Rose, B.; Ogata, A.F.; Fishman, D.A.; Patterson, J.P. Role of molecular modification and protein folding in the nucleation and growth of protein–metal–organic frameworks. Chem. Mater. 2022, 34, 8336–8344. [Google Scholar] [CrossRef]
- Sarmah, N.; Revathi, D.; Sheelu, G.; Yamuna Rani, K.; Sridhar, S.; Mehtab, V.; Sumana, C. Recent advances on sources and industrial applications of lipases. Biotechnol. Prog. 2018, 34, 5–28. [Google Scholar] [CrossRef]
- Zhong, L.; Feng, Y.; Wang, G.; Wang, Z.; Bilal, M.; Lv, H.; Jia, S.; Cui, J. Production and use of immobilized lipases in/on nanomaterials: A review from the waste to biodiesel production. Int. J. Biol. Macromol. 2020, 152, 207–222. [Google Scholar] [CrossRef] [PubMed]
- Facin, B.R.; Melchiors, M.S.; Valério, A.; Oliveira, J.V.; de Oliveira, D. Driving immobilized lipases as biocatalysts: 10 years state of the art and future prospects. Ind. Eng. Chem. Res. 2019, 58, 5358–5378. [Google Scholar] [CrossRef]
- Dwivedee, B.P.; Soni, S.; Sharma, M.; Bhaumik, J.; Laha, J.K.; Banerjee, U.C. Promiscuity of lipase-catalyzed reactions for organic synthesis: A recent update. Chem. Sel. 2018, 3, 2441–2466. [Google Scholar] [CrossRef]
- Chen, G.; Khan, I.M.; He, W.; Li, Y.; Jin, P.; Campanella, O.H.; Zhang, H.; Huo, Y.; Chen, Y.; Yang, H.; et al. Rebuilding the lid region from conformational and dynamic features to engineering applications of lipase in foods: Current status and future prospects. Compr. Rev. Food Sci. Food Saf. 2022, 21, 2688–2714. [Google Scholar] [CrossRef]
- Wang, S.; Xu, Y.; Yu, X.-W. A phenylalanine dynamic switch controls the interfacial activation of Rhizopus chinensis lipase. Int. J. Biol. Macromol. 2021, 173, 1–12. [Google Scholar] [CrossRef]
- Verger, R. ‘Interfacial activation’ of lipases: Facts and artifacts. Trends Biotechnol. 1997, 15, 32–38. [Google Scholar] [CrossRef]
- Brzozowski, A.M.; Derewenda, U.; Derewenda, Z.S.; Dodson, G.G.; Lawson, D.M.; Turkenburg, J.P.; Bjorkling, F.; Huge-Jensen, B.; Patkar, S.A.; Thim, L. A model for interfacial activation in lipases from the structure of a fungal lipase-inhibitor complex. Nature 1991, 351, 491–494. [Google Scholar] [CrossRef]
- Manoel, E.A.; dos Santos, J.C.S.; Freire, D.M.G.; Rueda, N.; Fernandez-Lafuente, R. Immobilization of lipases on hydrophobic supports involves the open form of the enzyme. Enzyme Microb. Technol. 2015, 71, 53–57. [Google Scholar] [CrossRef]
- Migneault, I.; Dartiguenave, C.; Bertrand, M.J.; Waldron, K.C. Glutaraldehyde: Behavior in aqueous solution, reaction with proteins, and application to enzyme crosslinking. Biotechniques 2004, 37, 790–802. [Google Scholar] [CrossRef]
- Barbosa, O.; Ortiz, C.; Berenguer-Murcia, Á.; Torres, R.; Rodrigues, R.C.; Fernandez-Lafuente, R. Glutaraldehyde in bio-catalysts design: A useful crosslinker and a versatile tool in enzyme immobilization. RSC Adv. 2014, 4, 1583–1600. [Google Scholar] [CrossRef]
- Monsan, P. Optimization of glutaraldehyde activation of a support for enzyme immobilization. J. Mol. Catal. 1978, 3, 371–384. [Google Scholar] [CrossRef]
- Barbosa, O.; Torres, R.; Ortiz, C.; Fernandez-Lafuente, R. The slow-down of the CALB immobilization rate permits to control the inter and intra molecular modification produced by glutaraldehyde. Process Biochem. 2012, 47, 766–774. [Google Scholar] [CrossRef]
- Fernandez-Lafuente, R.; Rosell, C.M.; Rodriguez, V.; Guisan, J.M. Strategies for enzyme stabilization by intramolecular crosslinking with bifunctional reagents. Enzyme Microb. Technol. 1995, 17, 517–523. [Google Scholar] [CrossRef]
- Hoare, D.G.; Koshland, D.E. A method for the quantitative modification and estimation of carboxylic acid groups in proteins. J. Biol. Chem. 1967, 242, 2447–2453. [Google Scholar] [CrossRef]
- Carraway, K.L.; Spoerl, P.; Koshland, D.E. Carboxyl group modification in chymotrypsin and chymotrypsinogen. J. Mol. Biol. 1969, 42, 133–137. [Google Scholar] [CrossRef]
- Snyder, S.L.; Sobocinski, P.Z. An improved 2,4,6-trinitrobenzenesulfonic acid method for the determination of amines. Anal. Biochem. 1975, 64, 284–288. [Google Scholar] [CrossRef]
- Virgen-Ortíz, J.J.; dos Santos, J.C.S.; Berenguer-Murcia, Á.; Barbosa, O.; Rodrigues, R.C.; Fernandez-Lafuente, R. Polyethylenimine: A very useful ionic polymer in the design of immobilized enzyme biocatalysts. J. Mater. Chem. B 2017, 5, 7461–7490. [Google Scholar] [CrossRef] [Green Version]
- Fernandez-Lopez, L.; Virgen-OrtÍz, J.J.; Pedrero, S.G.; Lopez-Carrobles, N.; Gorines, B.C.; Otero, C.; Fernandez-Lafuente, R. Optimization of the coating of octyl-CALB with ionic polymers to improve stability and decrease enzyme leakage. Biocatal. Biotransf. 2018, 36, 47–56. [Google Scholar] [CrossRef]
- Carballares, D.; Rocha-Martin, J.; Fernandez-Lafuente, R. Preparation of a six-enzyme multilayer combi-biocatalyst: Reuse of the most stable enzymes after inactivation of the least stable one. ACS Sustain. Chem. Eng. 2022, 10, 3920–3934. [Google Scholar] [CrossRef]
- Fernandez-Lopez, L.; Pedrero, S.G.; Lopez-Carrobles, N.; Virgen-Ortíz, J.J.; Gorines, B.C.; Otero, C.; Fernandez-Lafuente, R. Physical crosslinking of lipase from Rhizomucor miehei immobilized on octyl agarose via coating with ionic polymers: Avoiding enzyme release from the support. Process Biochem. 2017, 54, 81–88. [Google Scholar] [CrossRef]
- Fernandez-Lafuente, R. Lipase from Thermomyces lanuginosus: Uses and prospects as an industrial biocatalyst. J. Mol. Catal. B Enzym. 2010, 62, 197–212. [Google Scholar] [CrossRef]
- Anderson, E.M.; Larsson, K.M.; Kirk, O. One biocatalyst-many applications: The use of Candida antarctica B-lipase in organic synthesis. Biocatal. Biotransf. 1998, 16, 181–204. [Google Scholar] [CrossRef]
- Gotor-Fernández, V.; Busto, E.; Gotor, V. Candida antarctica lipase B: An ideal biocatalyst for the preparation of nitrogenated organic compounds. Adv. Synth. Catal. 2006, 348, 797–812. [Google Scholar] [CrossRef]
- Tsai, S.-W. Enantiopreference of Candida antarctica lipase B toward carboxylic acids: Substrate models and enantioselectivity thereof. J. Mol. Catal. B Enzym. 2016, 127, 98–116. [Google Scholar] [CrossRef]
- Palomo, J.M.; Fernández-Lorente, G.; Guisán, J.M.; Fernández-Lafuente, R. Modulation of immobilized lipase enantioselectivity via chemical amination. Adv. Synth. Catal. 2007, 349, 1119–1127. [Google Scholar] [CrossRef]
- Cabrera, Z.; Fernandez-Lorente, G.; Fernandez-Lafuente, R.; Palomo, J.M.; Guisan, J.M. Enhancement of Novozym-435 catalytic properties by physical or chemical modification. Process Biochem. 2009, 44, 226–231. [Google Scholar] [CrossRef]
- Gonçalves, M.C.P.; Amaral, J.C.; Lopes, L.A.; Fernandez-Lafuente, R.; Tardioli, P.W. Stabilization and operational selectivity alteration of Lipozyme 435 by its coating with polyethyleneimine: Comparison of the biocatalyst performance in the synthesis of xylose fatty esters. Int. J. Biol. Macromol. 2021, 192, 665–674. [Google Scholar] [CrossRef]
- Rodrigues, R.C.; Godoy, C.A.; Volpato, G.; Ayub, M.A.Z.; Fernandez-Lafuente, R.; Guisan, J.M. Immobilization–stabilization of the lipase from Thermomyces lanuginosus: Critical role of chemical amination. Process Biochem. 2009, 44, 963–968. [Google Scholar] [CrossRef]
- Zaak, H.; Fernandez-Lopez, L.; Otero, C.; Sassi, M.; Fernandez-Lafuente, R. Improved stability of immobilized lipases via modification with polyethylenimine and glutaraldehyde. Enzyme Microb. Technol. 2017, 106, 67–74. [Google Scholar] [CrossRef]
- Fernandez-Lopez, L.; Pedrero, S.G.; Lopez-Carrobles, N.; Gorines, B.C.; Virgen-Ortíz, J.J.; Fernandez-Lafuente, R. Effect of protein load on stability of immobilized enzymes. Enzyme Microb. Technol. 2017, 98, 18–25. [Google Scholar] [CrossRef]
- Zaak, H.; Siar, E.-H.; Kornecki, J.F.; Fernandez-Lopez, L.; Pedrero, S.G.; Virgen-Ortíz, J.J.; Fernandez-Lafuente, R. Effect of immobilization rate and enzyme crowding on enzyme stability under different conditions. The case of lipase from Thermomyces lanuginosus immobilized on octyl agarose beads. Process Biochem. 2017, 56, 117–123. [Google Scholar] [CrossRef]
- Arana-Peña, S.; Rios, N.S.; Mendez-Sanchez, C.; Lokha, Y.; Carballares, D.; Gonçalves, L.R.B.; Fernandez-Lafuente, R. Coimmobilization of different lipases: Simple layer by layer enzyme spatial ordering. Int. J. Biol. Macromol. 2020, 145, 856–864. [Google Scholar] [CrossRef] [PubMed]
- Rios, N.S.; Arana-Peña, S.; Mendez-Sanchez, C.; Lokha, Y.; Cortes-Corberan, V.; Gonçalves, L.R.B.; Fernandez-Lafuente, R. Increasing the enzyme loading capacity of porous supports by a layer-by-layer immobilization strategy using PEI as glue. Catalysts 2019, 9, 576. [Google Scholar] [CrossRef] [Green Version]
- Arana-Peña, S.; Rios, N.S.; Mendez-Sanchez, C.; Lokha, Y.; Gonçalves, L.R.B.; Fernández-Lafuente, R. Use of polyethylenimine to produce immobilized lipase multilayers biocatalysts with very high volumetric activity using octyl-agarose beads: Avoiding enzyme release during multilayer production. Enzyme Microb. Technol. 2020, 137, 109535. [Google Scholar] [CrossRef] [PubMed]
- Arana-Peña, S.; Carballares, D.; Cortés Corberan, V.; Fernandez-Lafuente, R. Multi-combilipases: Co-immobilizing lipases with very different stabilities combining immobilization via interfacial activation and ion exchange. The reuse of the most stable co-immobilized enzymes after inactivation of the least stable ones. Catalysts 2020, 10, 1207. [Google Scholar] [CrossRef]
- Arana-Peña, S.; Rios, N.S.; Carballares, D.; Mendez-Sanchez, C.; Lokha, Y.; Gonçalves, L.R.B.; Fernandez-Lafuente, R. Effects of enzyme loading and immobilization conditions on the catalytic features of lipase from Pseudomonas fluorescens immobilized on octyl-agarose beads. Front. Bioeng. Biotechnol. 2020, 8, 36. [Google Scholar] [CrossRef] [Green Version]
- Fernández-Lorente, G.; Palomo, J.M.; Mateo, C.; Munilla, R.; Ortiz, C.; Cabrera, Z.; Guisán, J.M.; Fernández-Lafuente, R. Glutaraldehyde cross-linking of lipases adsorbed on aminated supports in the presence of detergents leads to improved performance. Biomacromolecules 2006, 7, 2610–2615. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Arana-Peña, S.; Lokha, Y.; Fernández-Lafuente, R. Immobilization on octyl-agarose beads and some catalytic features of commercial preparations of lipase a from Candida antarctica (Novocor ADL): Comparison with immobilized lipase B from Candida antarctica. Biotechnol. Prog. 2019, 35, e2735. [Google Scholar] [CrossRef] [Green Version]
- Lokha, Y.; Arana-Peña, S.; Rios, N.S.; Mendez-Sanchez, C.; Gonçalves, L.R.B.; Lopez-Gallego, F.; Fernandez-Lafuente, R. Modulating the properties of the lipase from Thermomyces lanuginosus immobilized on octyl agarose beads by altering the immobilization conditions. Enzyme Microb. Technol. 2020, 133, 109461. [Google Scholar] [CrossRef]
- Wang, M.; Shi, H.; Wu, D.; Han, H.; Zhang, J.; Xing, Z.; Wang, S.; Li, Q. Glutaraldehyde cross-linking of immobilized thermophilic esterase on hydrophobic macroporous resin for application in poly(ε-caprolactone) synthesis. Molecules 2014, 19, 9838–9849. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Virgen-Ortíz, J.; Pedrero, S.; Fernandez-Lopez, L.; Lopez-Carrobles, N.; Gorines, B.; Otero, C.; Fernandez-Lafuente, R. Desorption of lipases immobilized on octyl-agarose beads and coated with ionic polymers after thermal inactivation. Stronger adsorption of polymers/unfolded protein composites. Molecules 2017, 22, 91. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Palomo, J.M.; Fuentes, M.; Fernández-Lorente, G.; Mateo, C.; Guisan, J.M.; Fernández-Lafuente, R. General trend of lipase to self-assemble giving bimolecular aggregates greatly modifies the enzyme functionality. Biomacromolecules 2003, 4, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Palomo, J.M.; Ortiz, C.; Fernández-Lorente, G.; Fuentes, M.; Guisán, J.M.; Fernández-Lafuente, R. Lipase-lipase interactions as a new tool to immobilize and modulate the lipase properties. Enzyme Microb. Technol. 2005, 36, 447–454. [Google Scholar] [CrossRef]
- de Lima, L.N.; Aragon, C.C.; Mateo, C.; Palomo, J.M.; Giordano, R.L.C.; Tardioli, P.W.; Guisan, J.M.; Fernandez-Lorente, G. Immobilization and stabilization of a bimolecular aggregate of the lipase from Pseudomonas fluorescens by multipoint covalent attachment. Process Biochem. 2013, 48, 118–123. [Google Scholar] [CrossRef]
- Volpato, G.; Filice, M.; Ayub, M.A.Z.; Guisan, J.M.; Palomo, J.M. Single-step purification of different lipases from Staphylococcus warneri. J. Chromatogr. A 2010, 1217, 473–478. [Google Scholar] [CrossRef]
- Fernández-Lorente, G.; Palomo, J.M.; Fuentes, M.; Mateo, C.; Guisán, J.M.; Fernández-Lafuente, R. Self-assembly of Pseudomonas fluorescens lipase into bimolecular aggregates dramatically affects functional properties. Biotechnol. Bioeng. 2003, 82, 232–237. [Google Scholar] [CrossRef]
- Palomo, J.M.; Ortiz, C.; Fuentes, M.; Fernandez-Lorente, G.; Guisan, J.M.; Fernandez-Lafuente, R. Use of immobilized lipases for lipase purification via specific lipase-lipase interactions. J. Chromatogr. A 2004, 1038, 267–273. [Google Scholar] [CrossRef]
- Wilson, L.; Palomo, J.M.; Fernández-Lorente, G.; Illanes, A.; Guisán, J.M.; Fernández-Lafuente, R. Effect of lipase–lipase interactions in the activity, stability and specificity of a lipase from Alcaligenes sp. Enzyme Microb. Technol. 2006, 39, 259–264. [Google Scholar] [CrossRef]
- Lombardo, D.; Guy, O. Effect of alcohols on the hydrolysis catalyzed by human pancreatic carboxylic-ester hydrolase. Biochim. Biophys. Acta-Enzymol. 1981, 657, 425–437. [Google Scholar] [CrossRef]
- Hernandez, K.; Garcia-Verdugo, E.; Porcar, R.; Fernandez-Lafuente, R. Hydrolysis of triacetin catalyzed by immobilized lipases: Effect of the immobilization protocol and experimental conditions on diacetin yield. Enzyme Microb. Technol. 2011, 48, 510–517. [Google Scholar] [CrossRef] [PubMed]
- Souza, P.M.P.; Carballares, D.; Lopez-Carrobles, N.; Gonçalves, L.R.B.; Lopez-Gallego, F.; Rodrigues, S.; Fernandez-Lafuente, R. Enzyme-support interactions and inactivation conditions determine Thermomyces lanuginosus lipase inactivation pathways: Functional and florescence studies. Int. J. Biol. Macromol. 2021, 191, 79–91. [Google Scholar] [CrossRef]
- dos Santos, J.C.S.; Rueda, N.; Gonçalves, L.R.B.; Fernandez-Lafuente, R. Tuning the catalytic properties of lipases immobilized on divinylsulfone activated agarose by altering its nanoenvironment. Enzyme Microb. Technol. 2015, 77, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Uppenberg, J.; Hansen, M.T.; Patkar, S.; Jones, T.A. The sequence, crystal structure determination and refinement of two crystal forms of lipase B from Candida antarctica. Structure 1994, 2, 293–308. [Google Scholar] [CrossRef] [Green Version]
- Arana-Peña, S.; Rios, N.S.; Carballares, D.; Gonçalves, L.R.B.; Fernandez-Lafuente, R. Immobilization of lipases via interfacial activation on hydrophobic supports: Production of biocatalysts libraries by altering the immobilization conditions. Catal. Today 2021, 362, 130–140. [Google Scholar] [CrossRef]
- Bolivar, J.M.; Nidetzky, B. On the relationship between structure and catalytic effectiveness in solid surface-immobilized enzymes: Advances in methodology and the quest for a single-molecule perspective. Biochim. Biophys. Acta-Proteins Proteom. 2020, 1868, 140333. [Google Scholar] [CrossRef]
- Bolivar, J.M.; Eisl, I.; Nidetzky, B. Advanced characterization of immobilized enzymes as heterogeneous biocatalysts. Catal. Today 2016, 259, 66–80. [Google Scholar] [CrossRef]
- Guo, W.; Zou, X.; Jiang, H.; Koebke, K.J.; Hoarau, M.; Crisci, R.; Lu, T.; Wei, T.; Marsh, E.N.G.; Chen, Z. Molecular structure of the surface-immobilized super uranyl binding protein. J. Phys. Chem. B 2021, 125, 7706–7716. [Google Scholar] [CrossRef]
- Zou, X.; Wei, S.; Badieyan, S.; Schroeder, M.; Jasensky, J.; Brooks, C.L.; Marsh, E.N.G.; Chen, Z. Investigating the effect of two-point surface attachment on enzyme stability and activity. J. Am. Chem. Soc. 2018, 140, 16560–16569. [Google Scholar] [CrossRef]
- Jasensky, J.; Ferguson, K.; Baria, M.; Zou, X.; McGinnis, R.; Kaneshiro, A.; Badieyan, S.; Wei, S.; Marsh, E.N.G.; Chen, Z. Simultaneous observation of the orientation and activity of surface-immobilized enzymes. Langmuir 2018, 34, 9133–9140. [Google Scholar] [CrossRef]
- Li, Y.; Ogorzalek, T.L.; Wei, S.; Zhang, X.; Yang, P.; Jasensky, J.; Brooks, C.L.; Marsh, E.N.G.; Chen, Z. Effect of immobilization site on the orientation and activity of surface-tethered enzymes. Phys. Chem. Chem. Phys. 2018, 20, 1021–1029. [Google Scholar] [CrossRef] [PubMed]
- Badieyan, S.; Wang, Q.; Zou, X.; Li, Y.; Herron, M.; Abbott, N.L.; Chen, Z.; Marsh, E.N.G. Engineered surface-immobilized enzyme that retains high levels of catalytic activity in air. J. Am. Chem. Soc. 2017, 139, 2872–2875. [Google Scholar] [CrossRef] [PubMed]
- Ogorzalek, T.L.; Wei, S.; Liu, Y.; Wang, Q.; Brooks, C.L.; Chen, Z.; Marsh, E.N.G. Molecular-level insights into orientation-dependent changes in the thermal stability of enzymes covalently immobilized on surfaces. Langmuir 2015, 31, 6145–6153. [Google Scholar] [CrossRef] [PubMed]
- Mohamad, N.R.; Marzuki, N.H.C.; Buang, N.A.; Huyop, F.; Wahab, R.A. An overview of technologies for immobilization of enzymes and surface analysis techniques for immobilized enzymes. Biotechnol. Biotechnol. Equip. 2015, 29, 205–220. [Google Scholar] [CrossRef]
- Carlsson, N.; Gustafsson, H.; Thörn, C.; Olsson, L.; Holmberg, K.; Åkerman, B. Enzymes immobilized in mesoporous silica: A physical–chemical perspective. Adv. Colloid Interface Sci. 2014, 205, 339–360. [Google Scholar] [CrossRef]





| Biocatalysts | Activity (U/g) | |||
|---|---|---|---|---|
| p-NPB | Triacetin | R-Methyl Mandelate | S-Methyl Mandelate | |
| Octyl-TLL | 1416 ± 51 | 601 ± 6 | 1.2 ± 0.1 | 3.2 ± 0.2 |
| Octyl-TLL-GA | 1350 ± 91 | 48 ± 2 | 0.8 ± 0.1 | 2.4 ± 0.1 |
| Octyl-TLL-Amin | 1675 ± 49 | 88 ± 4 | 0.6 ± 0.1 | 3.2 ± 0.2 |
| Octyl-TLL-TNBS | 1764 ± 82 | 60 ± 3 | 0.9 ± 0.1 | 3.1 ± 0.2 |
| Octyl-TLL-PEI | 1363 ± 61 | 90 ± 4 | 9.8 ± 0.5 | 7.7 ± 0.4 |
| Octyl-TLL-DS | 985 ± 52 | 28 ± 1 | 7.5 ± 0.4 | 6.5 ± 0.2 |
| Biocatalysts | Activity (U/g) | |||
|---|---|---|---|---|
| p-NPB | Triacetin | R-Methyl Mandelate | S-Methyl Mandelate | |
| Octyl-TLL | 1416 ± 51 | 61 ± 6 | 1.2 ± 0.1 | 3.2 ± 0.2 |
| Octyl-TLL-ZnP * | 1040 ± 64 | 88 ± 6 | 0.9 ± 0.1 | 3.0 ± 0.1 |
| Octyl-TLL-CoP | 1207 ± 72 | 65 ± 5 | 1.1 ± 0.1 | 2.9 ± 0.2 |
| Octyl-TLL-CuP | 1144 ± 90 | 67 ± 6 | 0.8 ± 0.1 | 2.7 ± 0.1 |
| Octyl-TLL-NiP | 1371 ± 87 | 53 ± 3 | 1.5 ± 0.1 | 3.0 ± 0.2 |
| Octyl-TLL-MgP | 1665 ± 41 | 66 ± 3 | 1.5 ± 0.1 | 2.4 ± 0.1 |
| Line | Biocatalysts | Activity (U/g) | |||
|---|---|---|---|---|---|
| 1 | p-NPB | Triacetin | R-Methyl Mandelate | S-Methyl Mandelate | |
| 2 | Octyl-TLL-GA | 1350 ± 91 | 48 ± 2 | 0.8 ± 0.1 | 2.4 ± 0.1 |
| 3 | Octyl-TLL-GA-ZnP | 1408 ± 84 | 50 ± 2 | 0.7 ± 0.1 | 3.0 ± 0.1 |
| 4 | Octyl-TLL-GA-CoP | 1133 ± 61 | 35 ± 2 | 0.2 ± 0.1 | 2.0 ± 0.1 |
| 5 | Octyl-TLL-GA-CuP | 1142 ± 42 | 38 ± 2 | 0.6 ± 0.1 | 2.1 ± 0.1 |
| 6 | Octyl-TLL-GA-NiP | 1308 ± 54 | 48 ± 2 | 0.6 ± 0.1 | 2.2 ± 0.1 |
| 7 | Octyl-TLL-GA-MgP | 1404 ± 120 | 46 ± 2 | 0.7 ± 0.1 | 2.4 ± 0.1 |
| 8 | Octyl-TLL-Amin | 1675 ± 49 | 88 ± 4 | 0.6 ± 0.1 | 3.2 ± 0.2 |
| 9 | Octyl-TLL-Amin-ZnP | 1422 ± 208 | 78 ± 4 | 0.6 ± 0.1 | 3.1 ± 0.2 |
| 10 | Octyl-TLL-Amin-CoP | 1647 ± 87 | 53 ± 3 | 0.1 ± 0.1 | 2.2 ± 0.1 |
| 11 | Octyl-TLL-Amin-CuP | 1618 ± 36 | 79 ± 4 | 0.9 ± 0.4 | 3.0 ± 0.2 |
| 12 | Octyl-TLL-Amin-NiP | 1863 ± 107 | 63 ± 3 | 0.6 ± 0.1 | 2.8 ± 0.1 |
| 13 | Octyl-TLL-Amin-MgP | 1979 ± 90 | 79 ± 4 | 0.8 ± 0.1 | 2.9 ± 0.1 |
| 14 | Octyl-TLL-TNBS | 1763 ± 82 | 60 ± 3 | 0.9 ± 0.1 | 3.1 ± 0.2 |
| 15 | Octyl-TLL-TNBS-ZnP | 1836 ± 97 | 47 ± 2. | 0.9 ± 0.1 | 2.7 ± 0.1 |
| 16 | Octyl-TLL-TNBS-CoP | 1591 ± 109 | 46 ± 2 | 0.4 ± 0.1 | 2.4 ± 0.1 |
| 17 | Octyl-TLL-TNBS-CuP | 1501 ± 76 | 45 ± 2 | 1.0 ± 0. 14 | 3.0 ± 0.2 |
| 18 | Octyl-TLL-TNBS-NiP | 1592 ± 128 | 44 ± 2 | 1.4 ± 0.1 | 3.1 ± 0.2 |
| 19 | Octyl-TLL-TNBS-MgP | 1544 ± 60 | 46 ± 2 | 1.1 ± 0.1 | 3.0 ± 0.2 |
| Line | Biocatalysts | Activity (U/g) | |||
|---|---|---|---|---|---|
| 1 | p-NPB | Triacetin | R-Methyl Mandelate | S-Methyl Mandelate | |
| 2 | Octyl-TLL-PEI | 1363 ± 61 | 90 ± 4 | 9.8 ± 0.5 | 7.7 ± 0.4 |
| 3 | Octyl-TLL-PEI-ZnP | 1479 ± 20 | 106 ± 5 | 8.2 ± 0.4 | 8.2 ± 0.5 |
| 4 | Octyl-TLL-PEI-CoP | 1442 ± 31 | 108 ± 6 | 7.7 ± 0.4 | 8.1 ± 0.3 |
| 5 | Octyl-TLL-PEI-CuP | 1351 ± 80 | 120 ± 6 | 8.7 ± 0.4 | 8.0 ± 0.2 |
| 6 | Octyl-TLL-PEI-NiP | 1309 ± 72 | 94 ± 4 | 8.5 ± 0.5 | 7.9 ± 0.3 |
| 7 | Octyl-TLL-PEI-MgP | 1323 ± 50 | 80 ± 5 | 8.2 ± 0.4 | 7.7 ± 0.3 |
| 8 | Octyl-TLL-DS | 985 ± 52 | 28 ± 1 | 7.5 ± 0.4 | 6.5 ± 0.2 |
| 9 | Octyl-TLL-DS-ZnP | 1041 ± 31 | 37 ± 2 | 9.9 ± 0.4 | 7.3 ± 0.4 |
| 10 | Octyl-TLL-DS-CoP | 1084 ± 46 | 27 ± 1 | 8.1 ± 0.3 | 6.1 ± 0.2 |
| 11 | Octyl-TLL-DS-CuP | 898 ± 54 | 30 ± 2 | 8.9 ± 0.4 | 7.2 ± 0.4 |
| 12 | Octyl-TLL-DS-NiP | 1041 ± 40 | 28 ± 1 | 8.5 ± 0.5 | 7.2 ± 0.3 |
| 13 | Octyl-TLL-DS-MgP | 951 ± 23 | 31 ± 1 | 8.9 ± 0.4 | 7.4 ± 0.4 |
| Biocatalysts | Activity (U/g) | |||
|---|---|---|---|---|
| p-NPB | Triacetin | R-Methyl Mandelate | S-Methyl Mandelate | |
| Octyl-CALB | 1151 ± 62 | 132 ± 6 | 19.5 ± 0.9 | 39.0 ± 1.9 |
| Octyl-CALB-GA | 1325 ± 23 | 94 ± 5 | 16.8 ± 0.9 | 28.0 ± 1.4 |
| Octyl-CALB-Amin | 1312 ± 36 | 77 ± 3 | 26.5 ± 1.3 | 36.3 ± 1.9 |
| Octyl-CALB-TNBS | 1336 ± 36 | 89 ± 7 | 18.3 ± 0.8 | 41.1 ± 2.0 |
| Octyl-CALB-PEI | 1518 ± 91 | 143 ± 9 | 26.1 ± 1.5 | 33.3 ± 1.7 |
| Octyl-CALB-DS | 1294 ± 85 | 121 ± 4 | 20.0 ± 0.8 | 43.2 ± 2.2 |
| Biocatalysts | Activity (U/g) | |||
|---|---|---|---|---|
| p-NPB | Triacetin | R-Methyl Mandelate | S-Methyl Mandelate | |
| Octyl-CALB | 1151 ± 62 | 132 ± 6 | 19.5 ± 0.9 | 39.0 ± 1.9 |
| Octyl-CALB-ZnP | 1172 ± 43 | 194 ± 7 | 25.6 ± 1.3 | 49.5 ± 2.9 |
| Octyl-CALB-CoP | 967 ± 52 | 220 ± 13 | 27.7 ± 1.4 | 50.2 ± 2.2 |
| Octyl-CALB-CuP | 998 ± 73 | 270 ± 14 | 26.8 ± 1.3 | 50.1 ± 2.9 |
| Octyl-CALB-NiP | 1218 ± 78 | 222 ± 13 | 20.4 ± 1.0 | 43.5 ± 2.1 |
| Octyl-CALB-MgP | 1141 ± 49 | 82 ± 5 | 17.5 ± 0.9 | 29.5 ± 1.6 |
| Line | Biocatalysts | Activity (U/g) | |||
|---|---|---|---|---|---|
| 1 | p-NPB | Triacetin | R-Methyl Mandelate | S-Methyl Mandelate | |
| 2 | Octyl-CALB-GA | 1324 ± 23 | 94 ± 5 | 16.8 ± 0.9 | 28.0 ± 1.4 |
| 3 | Octyl-CALB-GA-ZnP | 1195 ± 23 | 92 ± 5 | 9.7 ± 0.5 | 25.0 ± 1.9 |
| 4 | Octyl-CALB-GA-CoP | 1324 ± 39 | 85 ± 4 | 15.5 ± 0.8 | 26.6 ± 1.3 |
| 5 | Octyl-CALB-GA-CuP | 1195 ± 29 | 82 ± 4 | 14.1 ± 0.7 | 31.4 ± 1.4 |
| 6 | Octyl-CALB-GA-NiP | 1263 ± 44 | 78 ± 2 | 18. 9 ± 0.8 | 27.8 ± 1.2 |
| 7 | Octyl-CALB-GA-MgP | 1264 ± 56 | 121 ± 6 | 23.8 ± 1.0 | 29.2 ± 1.6 |
| 8 | Octyl-CALB-Amin | 1312 ± 36 | 77 ± 3 | 26.5 ± 1.3 | 36.3 ± 1.9 |
| 9 | Octyl-CALB-Amin-ZnP | 1266 ± 21 | 112 ± 7 | 23.6 ± 1.4 | 27.9 ± 1.3 |
| 10 | Octyl-CALB-Amin-CoP | 1225 ± 69 | 122 ± 6 | 25.3 ± 1.1 | 27.0 ± 1.0 |
| 11 | Octyl-CALB-Amin-CuP | 1341 ± 63 | 97 ± 6 | 24.3 ± 0.9 | 27.6 ± 1.2 |
| 12 | Octyl-CALB-Amin-NiP | 1369 ± 102 | 116 ± 6 | 24.7 ± 1.4 | 37.0 ± 1.9 |
| 13 | Octyl-CALB-Amin-MgP | 1264 ± 56 | 121 ± 6 | 23.8 ± 1.0 | 29.2 ± 1.6 |
| 14 | Octyl-CALB-TNBS | 1336 ± 36 | 89 ± 7 | 18.3 ± 0.8 | 41.1 ± 2.0 |
| 15 | Octyl-CALB-TNBS-ZnP | 1266 ± 70 | 139 ± 7 | 16.7 ± 0.8 | 39.1 ± 2.0 |
| 16 | Octyl-CALB-TNBS-CoP | 1263 ± 81 | 140 ± 8 | 17.4 ± 0.7 | 37.5 ± 1.9 |
| 17 | Octyl-CALB-TNBS-CuP | 1128 ± 58 | 169 ± 8 | 19.3 ± 0.9 | 43.3 ± 2.1 |
| 18 | Octyl-CALB-TNBS-NiP | 1214 ± 119 | 153 ± 7 | 20.0 ± 0.8 | 38.2 ± 1.9 |
| 19 | Octyl-CALB-TNBS-MgP | 1170 ± 34.9 | 145 ± 9 | 18.6 ± 1.2 | 39.8 ± 2.0 |
| Line | Biocatalysts | Activity (U/g) | |||
|---|---|---|---|---|---|
| 1 | p-NPB | Triacetin | R-Methyl Mandelate | S-Methyl Mandelate | |
| 2 | Octyl-CALB-PEI | 1518 ± 91 | 143 ± 9 | 26.1 ± 1.5 | 33.3 ± 1.7 |
| 3 | Octyl-CALB-PEI-ZnP | 1769 ± 30 | 181 ± 7 | 26.9 ± 1.4 | 38.2 ± 2.0 |
| 4 | Octyl-CALB-PEI-CoP | 1638 ± 33 | 201 ± 11 | 26.3 ± 1.4 | 40.8 ± 2.1 |
| 5 | Octyl-CALB-PEI-CuP | 1656 ± 48 | 219 ± 14 | 17.2 ± 0.9 | 45.9 ± 2.9 |
| 6 | Octyl-CALB-PEI-NiP | 1489 ± 93 | 200 ± 11 | 12.5 ± 0.7 | 47.8 ± 2.3 |
| 7 | Octyl-CALB-PEI-MgP | 1468 ± 89 | 207 ± 13 | 19.1 ± 1.0 | 48.5 ± 2.6 |
| 8 | Octyl-CALB-DS | 1294 ± 85 | 121 ± 4 | 20.0 ± 0.8 | 43.2 ± 2.2 |
| 9 | Octyl-CALB-DS-ZnP | 1251 ± 51 | 132 ± 8 | 23.6 ± 1.1 | 43.2 ± 1.9 |
| 10 | Octyl-CALB-DS-CoP | 1248 ± 57 | 144 ± 8 | 21.4 ± 1.0 | 44.8 ± 2.6 |
| 11 | Octyl-CALB-DS-CuP | 1188 ± 101 | 182 ± 10 | 20.9 ± 0.8 | 46.6 ± 2.5 |
| 12 | Octyl-CALB-DS-NiP | 1232 ± 31 | 159 ± 9 | 19.3 ± 0.6 | 41.0 ± 1.9 |
| 13 | Octyl-CALB-DS-MgP | 1308 ± 27 | 179 ± 10 | 21.5 ± 0.9 | 44.3 ± 1.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guimarães, J.R.; Carballares, D.; Rocha-Martin, J.; Tardioli, P.W.; Fernandez-Lafuente, R. Tuning Immobilized Enzyme Features by Combining Solid-Phase Physicochemical Modification and Mineralization. Int. J. Mol. Sci. 2022, 23, 12808. https://doi.org/10.3390/ijms232112808
Guimarães JR, Carballares D, Rocha-Martin J, Tardioli PW, Fernandez-Lafuente R. Tuning Immobilized Enzyme Features by Combining Solid-Phase Physicochemical Modification and Mineralization. International Journal of Molecular Sciences. 2022; 23(21):12808. https://doi.org/10.3390/ijms232112808
Chicago/Turabian StyleGuimarães, José R., Diego Carballares, Javier Rocha-Martin, Paulo W. Tardioli, and Roberto Fernandez-Lafuente. 2022. "Tuning Immobilized Enzyme Features by Combining Solid-Phase Physicochemical Modification and Mineralization" International Journal of Molecular Sciences 23, no. 21: 12808. https://doi.org/10.3390/ijms232112808










