A Novel Antithrombocytopenia Agent, Rhizoma cibotii, Promotes Megakaryopoiesis and Thrombopoiesis through the PI3K/AKT, MEK/ERK, and JAK2/STAT3 Signaling Pathways
Abstract
1. Introduction
2. Results
2.1. Characterization of Chemical Constituents in the Aqueous Extracts of Cibotii rhizoma
2.2. Cytotoxicity of AECRs in K562 and Meg01 Cells
2.3. AECRs Induce Megakaryocyte Differentiation and Maturation
2.4. AECRs Accelerate PLT Recovery in Mice with Thrombocytopenia
2.5. AECRs Promote Megakaryocyte Differentiation and Maturation in Mice with Thrombocytopenia
2.6. Network Pharmacology Prediction of the Targets and Mechanisms of AECRs against Thrombocytopenia
2.7. AECRs Induce Megakaryocyte Differentiation through the Activation of the PI3K/Akt, MAPK, and JAK/STAT Signaling Pathways
3. Discussion
4. Materials and Methods
4.1. The Preparation of Aqueous Extracts of Cibotii rhizoma
4.2. The Main Ingredients of Aqueous Extracts of Cibotii rhizoma
4.3. Cell Culture
4.4. Cell Proliferation Assay
4.5. Lactate Dehydrogenase (LDH) Assay
4.6. Cell Apoptosis Assay
4.7. Morphologic Analysis
4.8. Giemsa Staining
4.9. Phalloidin Staining
4.10. Measurement of Megakaryocyte Differentiation by Flow Cytometry
4.11. Megakaryocyte Ploidy Analysis
4.12. Animals
4.13. Establishment of a Thrombocytopenia Mouse Model and Treatment with AECRs
4.14. Hemanalysis
4.15. Histopathological Analysis
4.16. Immunohistochemical Staining of CD41 and VWF
4.17. Flow Cytometry Analysis of Bone Marrow (BM), Spleen Cells, and Peripheral Blood
4.18. Network Pharmacology
4.19. Western Blot
4.20. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Praveen Kumar, M.K.; Shyama, S.K.; Sonaye, B.S.; Naik, U.R.; Kadam, S.B.; Bipin, P.D.; D’Costa, A.; Chaubey, R.C. Evaluation of γ-radiation-induced DNA damage in two species of bivalves and their relative sensitivity using comet assay. Aquat. Toxicol. 2014, 150, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Adhikari, J.S.; Rizvi, M.A.; Chaudhury, N.K. Melatonin attenuates 60Co γ-ray-induced hematopoietic, immunological and gastrointestinal injuries in C57BL/6 male mice. Environ. Toxicol. 2017, 32, 501–518. [Google Scholar] [CrossRef] [PubMed]
- Till, J.E.; McCulloch, E.A. Repair processes in irradiated mouse hematopoietic tissue. Ann. N. Y. Acad. Sci. 1964, 114, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Kiang, J.G.; Olabisi, A.O. Radiation: A poly-traumatic hit leading to multi-organ injury. Cell Biosci. 2019, 9, 25. [Google Scholar] [CrossRef] [PubMed]
- Satyamitra, M.M.; Cassatt, D.R.; Taliaferro, L.P. Meeting Commentary: A Poly-Pharmacy Approach to Mitigate Acute Radiation Syndrome (ARS). Radiat. Res. 2021, 196, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.K.; Newman, V.L.; Seed, T.M. Colony-stimulating factors for the treatment of the hematopoietic component of the acute radiation syndrome (H-ARS): A review. Cytokine 2015, 71, 22–37. [Google Scholar] [CrossRef]
- Machlus, K.R.; Italiano, J.E., Jr. The incredible journey: From megakaryocyte development to platelet formation. J. Cell Biol. 2013, 201, 785–796. [Google Scholar] [CrossRef]
- Roweth, H.G.; Parvin, S.; Machlus, K.R. Megakaryocyte modification of platelets in thrombocytopenia. Curr. Opin. Hematol. 2018, 25, 410–415. [Google Scholar] [CrossRef]
- Noetzli, L.J.; French, S.L.; Machlus, K.R. New Insights Into the Differentiation of Megakaryocytes From Hematopoietic Progenitors. Arterioscler. Thromb. Vasc. Biol. 2019, 39, 1288–1300. [Google Scholar] [CrossRef]
- Bartley, T.D.; Bogenberger, J.; Hunt, P.; Li, Y.S.; Lu, H.S.; Martin, F.; Chang, M.S.; Samal, B.; Nichol, J.L.; Swift, S.; et al. Identification and cloning of a megakaryocyte growth and development factor that is a ligand for the cytokine receptor Mpl. Cell 1994, 77, 1117–1124. [Google Scholar] [CrossRef]
- De Sauvage, F.J.; Hass, P.E.; Spencer, S.D.; Malloy, B.E.; Gurney, A.L.; Spencer, S.A.; Darbonne, W.C.; Henzel, W.J.; Wong, S.C.; Kuang, W.J.; et al. Stimulation of megakaryocytopoiesis and thrombopoiesis by the c-Mpl ligand. Nature 1994, 369, 533–538. [Google Scholar] [CrossRef]
- Kaushansky, K.; Lok, S.; Holly, R.D.; Broudy, V.C.; Lin, N.; Bailey, M.C.; Forstrom, J.W.; Buddle, M.M.; Oort, P.J.; Hagen, F.S.; et al. Promotion of megakaryocyte progenitor expansion and differentiation by the c-Mpl ligand thrombopoietin. Nature 1994, 369, 568–571. [Google Scholar] [CrossRef]
- Lok, S.; Kaushansky, K.; Holly, R.D.; Kuijper, J.L.; Lofton-Day, C.E.; Oort, P.J.; Grant, F.J.; Heipel, M.D.; Burkhead, S.K.; Kramer, J.M.; et al. Cloning and expression of murine thrombopoietin cDNA and stimulation of platelet production in vivo. Nature 1994, 369, 565–568. [Google Scholar] [CrossRef]
- Kaushansky, K. The mpl ligand: Molecular and cellular biology of the critical regulator of megakaryocyte development. Stem. cells 1994, 12 (Suppl. 1), 91–97. [Google Scholar]
- Kuter, D.J.; Beeler, D.L.; Rosenberg, R.D. The purification of megapoietin: A physiological regulator of megakaryocyte growth and platelet production. Proc. Natl. Acad. Sci. USA 1994, 91, 11104–11108. [Google Scholar] [CrossRef]
- Sohma, Y.; Akahori, H.; Seki, N.; Hori, T.; Ogami, K.; Kato, T.; Shimada, Y.; Kawamura, K.; Miyazaki, H. Molecular cloning and chromosomal localization of the human thrombopoietin gene. FEBS Lett. 1994, 353, 57–61. [Google Scholar] [CrossRef]
- Wendling, F.; Maraskovsky, E.; Debili, N.; Florindo, C.; Teepe, M.; Titeux, M.; Methia, N.; Breton-Gorius, J.; Cosman, D.; Vainchenker, W. cMpl ligand is a humoral regulator of megakaryocytopoiesis. Nature 1994, 369, 571–574. [Google Scholar] [CrossRef]
- Miyakawa, Y.; Drachman, J.G.; Gallis, B.; Kaushansky, A.; Kaushansky, K. A structure-function analysis of serine/threonine phosphorylation of the thrombopoietin receptor, c-Mpl. J. Biol. Chem. 2000, 275, 32214–32219. [Google Scholar] [CrossRef]
- Miyakawa, Y.; Oda, A.; Druker, B.J.; Kato, T.; Miyazaki, H.; Handa, M.; Ikeda, Y. Recombinant thrombopoietin induces rapid protein tyrosine phosphorylation of Janus kinase 2 and Shc in human blood platelets. Blood 1995, 86, 23–27. [Google Scholar] [CrossRef]
- Miyakawa, Y.; Oda, A.; Druker, B.J.; Miyazaki, H.; Handa, M.; Ohashi, H.; Ikeda, Y. Thrombopoietin induces tyrosine phosphorylation of Stat3 and Stat5 in human blood platelets. Blood 1996, 87, 439–446. [Google Scholar] [CrossRef]
- Yamada, M.; Komatsu, N.; Okada, K.; Kato, T.; Miyazaki, H.; Miura, Y. Thrombopoietin induces tyrosine phosphorylation and activation of mitogen-activated protein kinases in a human thrombopoietin-dependent cell line. Biochem. Biophys. Res. Commun. 1995, 217, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Sharma, P.; Tyler, L.N. Transfusion of blood and blood products: Indications and complications. Am. Fam. Physician 2011, 83, 719–724. [Google Scholar] [PubMed]
- Gilreath, J.; Lo, M.; Bubalo, J. Thrombopoietin Receptor Agonists (TPO-RAs): Drug Class Considerations for Pharmacists. Drugs 2021, 81, 1285–1305. [Google Scholar] [CrossRef]
- Al-Samkari, H.; Kuter, D.J. Optimal use of thrombopoietin receptor agonists in immune thrombocytopenia. Ther. Adv. Hematol. 2019, 10, 2040620719841735. [Google Scholar] [CrossRef] [PubMed]
- Ghanima, W.; Cooper, N.; Rodeghiero, F.; Godeau, B.; Bussel, J.B. Thrombopoietin receptor agonists: Ten years later. Haematologica 2019, 104, 1112–1123. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.K.; Seed, T.M. Radiation countermeasures for hematopoietic acute radiation syndrome: Growth factors, cytokines and beyond. Int. J. Radiat. Biol. 2021, 97, 1526–1547. [Google Scholar] [CrossRef]
- Vadhan-Raj, S.; Verschraegen, C.F.; Bueso-Ramos, C.; Broxmeyer, H.E.; Kudelkà, A.P.; Freedman, R.S.; Edwards, C.L.; Gershenson, D.; Jones, D.; Ashby, M.; et al. Recombinant human thrombopoietin attenuates carboplatin-induced severe thrombocytopenia and the need for platelet transfusions in patients with gynecologic cancer. Ann. Intern. Med. 2000, 132, 364–368. [Google Scholar] [CrossRef]
- Moskowitz, C.H.; Hamlin, P.A.; Gabrilove, J.; Bertino, J.R.; Portlock, C.S.; Straus, D.J.; Gencarelli, A.N.; Nimer, S.D.; Zelenetz, A.D. Maintaining the dose intensity of ICE chemotherapy with a thrombopoietic agent, PEG-rHuMGDF, may confer a survival advantage in relapsed and refractory aggressive non-Hodgkin lymphoma. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2007, 18, 1842–1850. [Google Scholar] [CrossRef]
- Li, J.; Yang, C.; Xia, Y.; Bertino, A.; Glaspy, J.; Roberts, M.; Kuter, D.J. Thrombocytopenia caused by the development of antibodies to thrombopoietin. Blood 2001, 98, 3241–3248. [Google Scholar] [CrossRef]
- Basser, R.L.; O’Flaherty, E.; Green, M.; Edmonds, M.; Nichol, J.; Menchaca, D.M.; Cohen, B.; Begley, C.G. Development of pancytopenia with neutralizing antibodies to thrombopoietin after multicycle chemotherapy supported by megakaryocyte growth and development factor. Blood 2002, 99, 2599–2602. [Google Scholar] [CrossRef]
- Tang, B.; Huang, L.; Liu, H.; Cheng, S.; Song, K.; Zhang, X.; Yao, W.; Ning, L.; Wan, X.; Sun, G.; et al. Recombinant human thrombopoietin promotes platelet engraftment after umbilical cord blood transplantation. Blood Adv. 2020, 4, 3829–3839. [Google Scholar] [CrossRef]
- Al-Samkari, H.; Soff, G.A. Clinical challenges and promising therapies for chemotherapy-induced thrombocytopenia. Exp. Rev. Hematol. 2021, 14, 437–448. [Google Scholar] [CrossRef]
- Mazan, S.; Michot, B.; Bachellerie, J.P. Mouse U3-RNA-processed pseudogenes are nonrandomly integrated into genomic DNA. Implications for the process of retrogene formation. Eur. J. Biochem. 1989, 181, 599–605. [Google Scholar] [CrossRef]
- Mai, W.; Chen, D.; Li, X. Antioxidant Activity of Rhizoma Cibotii in vitro. Adv. Pharm. Bull. 2012, 2, 107–114. [Google Scholar] [CrossRef]
- Wu, Q.; Yang, X.W. The constituents of Cibotium barometz and their permeability in the human Caco-2 monolayer cell model. J. Ethnopharmacol. 2009, 125, 417–422. [Google Scholar] [CrossRef]
- Poulter, N.S.; Thomas, S.G. Cytoskeletal regulation of platelet formation: Coordination of F-actin and microtubules. Int. J. Biochem. Cell Biol. 2015, 66, 69–74. [Google Scholar] [CrossRef]
- Bunin, D.I.; Bakke, J.; Green, C.E.; Javitz, H.S.; Fielden, M.; Chang, P.Y. Romiplostim (Nplate®) as an effective radiation countermeasure to improve survival and platelet recovery in mice. Int. J. Radiat. Biol. 2020, 96, 145–154. [Google Scholar] [CrossRef]
- Ogawa, M.; Shirasago, Y.; Ando, S.; Shimojima, M.; Saijo, M.; Fukasawa, M. Caffeic acid, a coffee-related organic acid, inhibits infection by severe fever with thrombocytopenia syndrome virus in vitro. J. Infect. Chemother. Off. J. Jpn. Soc. Chemother. 2018, 24, 597–601. [Google Scholar] [CrossRef]
- Qin, P.; Wei, Y.; Hou, M.; Zhao, C.; Shen, Z. A multicenter clinical trial of caffeic acid tablet in treatment of 103 primary immune thrombocytopenia patients. Zhonghua Xue Ye Xue Za Zhi. 2015, 36, 103–106. [Google Scholar] [CrossRef]
- Kumazoe, M.; Fujimura, Y.; Hidaka, S.; Kim, Y.; Murayama, K.; Takai, M.; Huang, Y.; Yamashita, S.; Murata, M.; Miura, D.; et al. Metabolic profiling-based data-mining for an effective chemical combination to induce apoptosis of cancer cells. Sci. Rep. 2015, 5, 9474. [Google Scholar] [CrossRef]
- Ash, D.; Subramanian, M.; Surolia, A.; Shaha, C. Nitric oxide is the key mediator of death induced by fisetin in human acute monocytic leukemia cells. Am. J. Cancer Res. 2015, 5, 481–497. [Google Scholar] [PubMed]
- Adan, A.; Baran, Y. Fisetin and hesperetin induced apoptosis and cell cycle arrest in chronic myeloid leukemia cells accompanied by modulation of cellular signaling. Tumour Biol. J. Int. Soc. Oncodev. Biol. Med. 2016, 37, 5781–5795. [Google Scholar] [CrossRef] [PubMed]
- Jang, K.Y.; Jeong, S.J.; Kim, S.H.; Jung, J.H.; Kim, J.H.; Koh, W.; Chen, C.Y.; Kim, S.H. Activation of reactive oxygen species/AMP activated protein kinase signaling mediates fisetin-induced apoptosis in multiple myeloma U266 cells. Cancer Lett. 2012, 319, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.F.; Lai, G.F.; Efferth, T.; Cao, J.X.; Luo, S.D. New glycosides from Tetracentron sinense and their cytotoxic activity. Chem. Biodivers. 2006, 3, 1023–1030. [Google Scholar] [CrossRef] [PubMed]
- Roux, P.P.; Blenis, J. ERK and p38 MAPK-activated protein kinases: A family of protein kinases with diverse biological functions. Microbiol. Mol. Biol. Rev. MMBR 2004, 68, 320–344. [Google Scholar] [CrossRef]
- Garcia, J.; de Gunzburg, J.; Eychène, A.; Gisselbrecht, S.; Porteu, F. Thrombopoietin-mediated sustained activation of extracellular signal-regulated kinase in UT7-Mpl cells requires both Ras-Raf-1- and Rap1-B-Raf-dependent pathways. Mol. Cell. Biol. 2001, 21, 2659–2670. [Google Scholar] [CrossRef][Green Version]
- Rouyez, M.C.; Boucheron, C.; Gisselbrecht, S.; Dusanter-Fourt, I.; Porteu, F. Control of thrombopoietin-induced megakaryocytic differentiation by the mitogen-activated protein kinase pathway. Mol. Cell. Biol. 1997, 17, 4991–5000. [Google Scholar] [CrossRef]
- Rojnuckarin, P.; Drachman, J.G.; Kaushansky, K. Thrombopoietin-induced activation of the mitogen-activated protein kinase (MAPK) pathway in normal megakaryocytes: Role in endomitosis. Blood 1999, 94, 1273–1282. [Google Scholar] [CrossRef]
- Sattler, M.; Durstin, M.A.; Frank, D.A.; Okuda, K.; Kaushansky, K.; Salgia, R.; Griffin, J.D. The thrombopoietin receptor c-MPL activates JAK2 and TYK2 tyrosine kinases. Exp. Hematol. 1995, 23, 1040–1048. [Google Scholar]
- Fichelson, S.; Freyssinier, J.M.; Picard, F.; Fontenay-Roupie, M.; Guesnu, M.; Cherai, M.; Gisselbrecht, S.; Porteu, F. Megakaryocyte growth and development factor-induced proliferation and differentiation are regulated by the mitogen-activated protein kinase pathway in primitive cord blood hematopoietic progenitors. Blood 1999, 94, 1601–1613. [Google Scholar] [CrossRef]
- Sauer, M.; Tausch, S.; Zieger, M.; Zintl, F.; Nowak, G.; Kaufmann, R. Evidence for a novel thrombopoietin signalling event: Activation of protein kinase A in human megakaryoblastic CMK cells. Cytokine 2001, 15, 75–79. [Google Scholar] [CrossRef]
- Geddis, A.E.; Fox, N.E.; Kaushansky, K. Phosphatidylinositol 3-kinase is necessary but not sufficient for thrombopoietin-induced proliferation in engineered Mpl-bearing cell lines as well as in primary megakaryocytic progenitors. J. Biol. Chem. 2001, 276, 34473–34479. [Google Scholar] [CrossRef]
- Zhang, Y.; Sun, S.; Wang, Z.; Thompson, A.; Kaluzhny, Y.; Zimmet, J.; Ravid, K. Signaling by the Mpl receptor involves IKK and NF-kappaB. J. Cell. Biochem. 2002, 85, 523–535. [Google Scholar] [CrossRef]
- Zheng, C.; Yang, R.; Han, Z.; Zhou, B.; Liang, L.; Lu, M. TPO-independent megakaryocytopoiesis. Crit. Rev. Oncol./Hematol. 2008, 65, 212–222. [Google Scholar] [CrossRef]
- Malara, A.; Balduini, A. Blood platelet production and morphology. Thromb. Res. 2012, 129, 241–244. [Google Scholar] [CrossRef]
- Di Martino, J.F.; Selleri, L.; Traver, D.; Firpo, M.T.; Rhee, J.; Warnke, R.; O’Gorman, S.; Weissman, I.L.; Cleary, M.L. The Hox cofactor and proto-oncogene Pbx1 is required for maintenance of definitive hematopoiesis in the fetal liver. Blood 2001, 98, 618–626. [Google Scholar] [CrossRef]
- Huang, H.; Cantor, A.B. Common features of megakaryocytes and hematopoietic stem cells: What’s the connection? J. Cell. Biochem. 2009, 107, 857–864. [Google Scholar] [CrossRef]
- Cai, M.; Langer, E.M.; Gill, J.G.; Satpathy, A.T.; Albring, J.C.; Kc, W.; Murphy, T.L.; Murphy, K.M. Dual actions of Meis1 inhibit erythroid progenitor development and sustain general hematopoietic cell proliferation. Blood 2012, 120, 335–346. [Google Scholar] [CrossRef]
- Hock, H.; Hamblen, M.J.; Rooke, H.M.; Schindler, J.W.; Saleque, S.; Fujiwara, Y.; Orkin, S.H. Gfi-1 restricts proliferation and preserves functional integrity of haematopoietic stem cells. Nature 2004, 431, 1002–1007. [Google Scholar] [CrossRef]
- Zeng, H.; Yücel, R.; Kosan, C.; Klein-Hitpass, L.; Möröy, T. Transcription factor Gfi1 regulates self-renewal and engraftment of hematopoietic stem cells. EMBO J. 2004, 23, 4116–4125. [Google Scholar] [CrossRef]
- Schlaeger, T.M.; Mikkola, H.K.; Gekas, C.; Helgadottir, H.B.; Orkin, S.H. Tie2Cre-mediated gene ablation defines the stem-cell leukemia gene (SCL/tal1)-dependent window during hematopoietic stem-cell development. Blood 2005, 105, 3871–3874. [Google Scholar] [CrossRef] [PubMed]
- Chagraoui, H.; Kassouf, M.; Banerjee, S.; Goardon, N.; Clark, K.; Atzberger, A.; Pearce, A.C.; Skoda, R.C.; Ferguson, D.J.; Watson, S.P.; et al. SCL-mediated regulation of the cell-cycle regulator p21 is critical for murine megakaryopoiesis. Blood 2011, 118, 723–735. [Google Scholar] [CrossRef] [PubMed]
- Ramsay, R.G.; Gonda, T.J. MYB function in normal and cancer cells. Nat. Rev. Cancer 2008, 8, 523–534. [Google Scholar] [CrossRef] [PubMed]
- Mucenski, M.L.; McLain, K.; Kier, A.B.; Swerdlow, S.H.; Schreiner, C.M.; Miller, T.A.; Pietryga, D.W.; Scott, W.J., Jr.; Potter, S.S. A functional c-myb gene is required for normal murine fetal hepatic hematopoiesis. Cell 1991, 65, 677–689. [Google Scholar] [CrossRef]
- Gewirtz, A.M.; Calabretta, B. A c-myb antisense oligodeoxynucleotide inhibits normal human hematopoiesis in vitro. Science 1988, 242, 1303–1306. [Google Scholar] [CrossRef] [PubMed]
- Pattabiraman, D.R.; Gonda, T.J. Role and potential for therapeutic targeting of MYB in leukemia. Leukemia 2013, 27, 269–277. [Google Scholar] [CrossRef]
- Perdomo, J.; Yan, F.; Leung, H.H.L.; Chong, B.H. Megakaryocyte Differentiation and Platelet Formation from Human Cord Blood-derived CD34+ Cells. J. Vis. Exp. JoVE 2017, 130, e56420. [Google Scholar] [CrossRef]
- Gandhi, M.J.; Drachman, J.G.; Reems, J.A.; Thorning, D.; Lannutti, B.J. A novel strategy for generating platelet-like fragments from megakaryocytic cell lines and human progenitor cells. Blood Cells Mol. Dis. 2005, 35, 70–73. [Google Scholar] [CrossRef]
- Choi, E.S.; Nichol, J.L.; Hokom, M.M.; Hornkohl, A.C.; Hunt, P. Platelets generated in vitro from proplatelet-displaying human megakaryocytes are functional. Blood 1995, 85, 402–413. [Google Scholar] [CrossRef]
- Bruno, S.; Gunetti, M.; Gammaitoni, L.; Danè, A.; Cavalloni, G.; Sanavio, F.; Fagioli, F.; Aglietta, M.; Piacibello, W. In vitro and in vivo megakaryocyte differentiation of fresh and ex-vivo expanded cord blood cells: Rapid and transient megakaryocyte reconstitution. Haematologica 2003, 88, 379–387. [Google Scholar]
- Iraqi, M.; Perdomo, J.; Yan, F.; Choi, P.Y.; Chong, B.H. Immune thrombocytopenia: Antiplatelet autoantibodies inhibit proplatelet formation by megakaryocytes and impair platelet production in vitro. Haematologica 2015, 100, 623–632. [Google Scholar] [CrossRef]
- Lev, P.R.; Grodzielski, M.; Goette, N.P.; Glembotsky, A.C.; Espasandin, Y.R.; Pierdominici, M.S.; Contrufo, G.; Montero, V.S.; Ferrari, L.; Molinas, F.C.; et al. Impaired proplatelet formation in immune thrombocytopenia: A novel mechanism contributing to decreased platelet count. Br. J. Haematol. 2014, 165, 854–864. [Google Scholar] [CrossRef]
- Salzmann, M.; Hoesel, B.; Haase, M.; Mussbacher, M.; Schrottmaier, W.C.; Kral-Pointner, J.B.; Finsterbusch, M.; Mazharian, A.; Assinger, A.; Schmid, J.A. A novel method for automated assessment of megakaryocyte differentiation and proplatelet formation. Platelets 2018, 29, 357–364. [Google Scholar] [CrossRef]
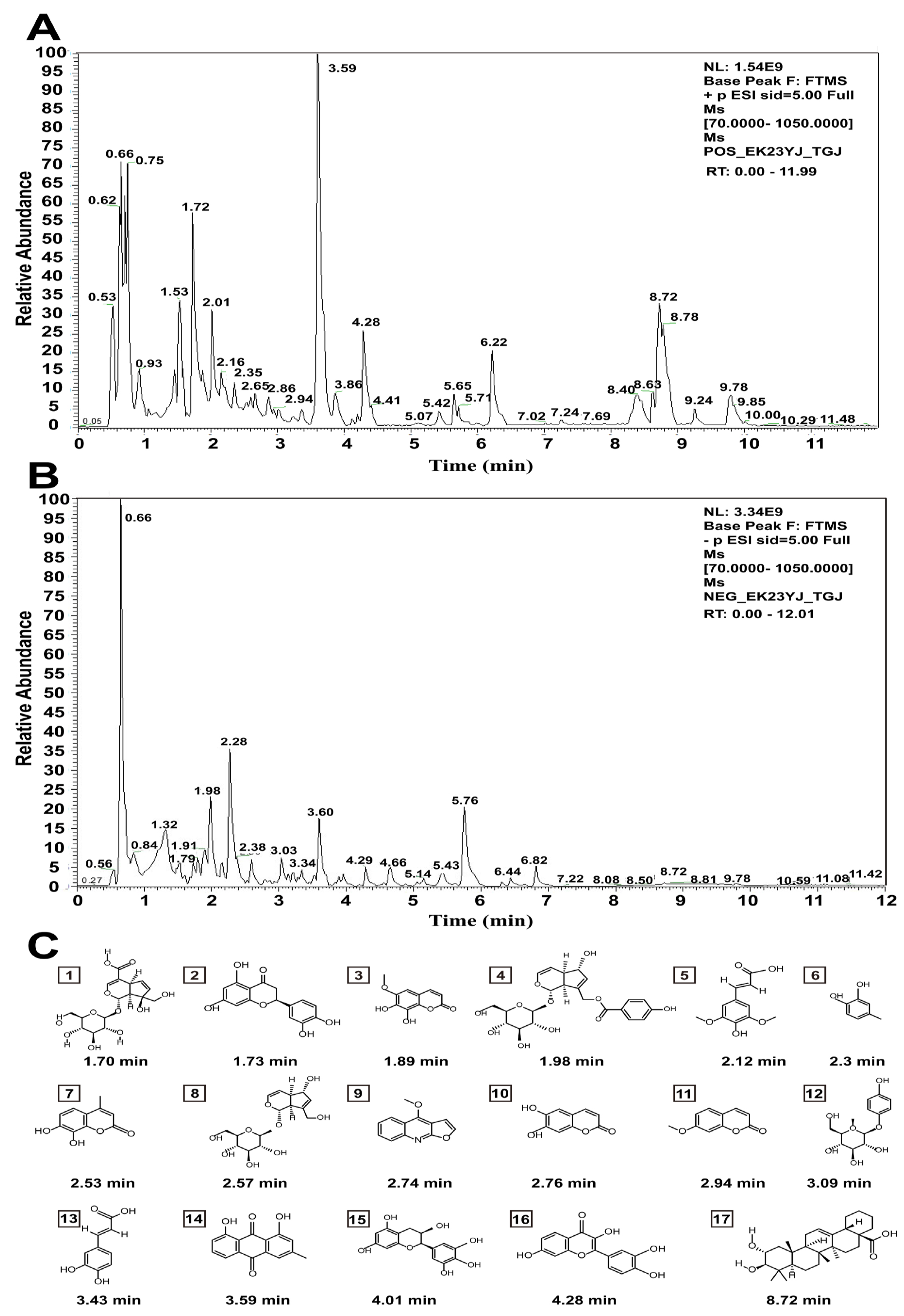








| No. | Identity | Mode | Molecular Formula | RT(min) | [M+H]+ (m/z) |
|---|---|---|---|---|---|
| 1 | Monotropein | Neg | C16H22O11 | 1.70 | 391.12401 |
| 2 | Eriodictyol | Neg | C15H12O6 | 1.73 | 289.07119 |
| 3 | Fraxetin | Neg | C10H8O5 | 1.89 | 209.04497 |
| 4 | Agnuside | Neg | C22H26O11 | 1.98 | 467.15531 |
| 5 | Sinapic acid | Neg | C11H12O5 | 2.12 | 225.07627 |
| 6 | 4-Methylcatechol | Neg | C7H8O2 | 2.30 | 125.06023 |
| 7 | 4-Methyldaphnetin | Neg | C10H8O4 | 2.53 | 193.05006 |
| 8 | Aucubin | Neg | C15H22O9 | 2.57 | 347.13418 |
| 9 | Dictamnine | Pos | C12H9NO2 | 2.74 | 200.07113 |
| 10 | Esculetin | Neg | C9H6O4 | 2.76 | 179.03441 |
| 11 | Herniarin | Pos | C10H8O3 | 2.94 | 177.05514 |
| 12 | Arbutin | Neg | C12H16O7 | 3.09 | 273.0974 |
| 13 | Caffeic Acid | Neg | C9H8O4 | 3.43 | 181.05006 |
| 14 | Chrysophanol | Pos | C15H10O4 | 3.59 | 255.06571 |
| 15 | Epigallocatechin | Neg | C15H14O7 | 4.02 | 307.08175 |
| 16 | Fisetin | Pos | C15H10O6 | 4.28 | 287.05554 |
| 17 | Maslinic Acid | Pos | C30H48O4 | 8.72 | 473.36306 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, W.; Zhu, L.; Wang, L.; Zeng, J.; Wen, M.; Xu, X.; Zou, L.; Huang, F.; Huang, Q.; Qin, D.; et al. A Novel Antithrombocytopenia Agent, Rhizoma cibotii, Promotes Megakaryopoiesis and Thrombopoiesis through the PI3K/AKT, MEK/ERK, and JAK2/STAT3 Signaling Pathways. Int. J. Mol. Sci. 2022, 23, 14060. https://doi.org/10.3390/ijms232214060
Chen W, Zhu L, Wang L, Zeng J, Wen M, Xu X, Zou L, Huang F, Huang Q, Qin D, et al. A Novel Antithrombocytopenia Agent, Rhizoma cibotii, Promotes Megakaryopoiesis and Thrombopoiesis through the PI3K/AKT, MEK/ERK, and JAK2/STAT3 Signaling Pathways. International Journal of Molecular Sciences. 2022; 23(22):14060. https://doi.org/10.3390/ijms232214060
Chicago/Turabian StyleChen, Wang, Linjie Zhu, Long Wang, Jing Zeng, Min Wen, Xiyan Xu, LiLe Zou, Feihong Huang, Qianqian Huang, Dalian Qin, and et al. 2022. "A Novel Antithrombocytopenia Agent, Rhizoma cibotii, Promotes Megakaryopoiesis and Thrombopoiesis through the PI3K/AKT, MEK/ERK, and JAK2/STAT3 Signaling Pathways" International Journal of Molecular Sciences 23, no. 22: 14060. https://doi.org/10.3390/ijms232214060
APA StyleChen, W., Zhu, L., Wang, L., Zeng, J., Wen, M., Xu, X., Zou, L., Huang, F., Huang, Q., Qin, D., Mei, Q., Yang, J., Wang, Q., & Wu, J. (2022). A Novel Antithrombocytopenia Agent, Rhizoma cibotii, Promotes Megakaryopoiesis and Thrombopoiesis through the PI3K/AKT, MEK/ERK, and JAK2/STAT3 Signaling Pathways. International Journal of Molecular Sciences, 23(22), 14060. https://doi.org/10.3390/ijms232214060






