Hormone Regulation of CCCH Zinc Finger Proteins in Plants
Abstract
:1. Introduction
2. Evolutionary Conservation of the CCCH Proteins in Plants
3. Responses of Plant CCCH Proteins to Several Major Hormones
3.1. ABA Signal Response
3.2. GA Signal Response
| Species | Corresponding Gene | ID | NO. of CCCH Motif | Subcellular Location | Responsive Hormone | Reference(s) |
|---|---|---|---|---|---|---|
| AtTZF1/AtC3H23 | AT2G25900 | 2 | Nucleus/cytoplasm | ABA/GA | [14] | |
| AtTZF2/OZF1/AtC3H20 | AT2G19810 | 2 | Cytoplasm | ABA/SA/JA | [16,17] | |
| AtTZF3/OZF2/AtC3H49 | AT4G29190 | 2 | Cytoplasm | ABA/JA | [16,18] | |
| AtTZF4/AtC3H2/SOMNUS | AT1G03790 | 2 | Nucleus | ABA/GA | [19,20] | |
| AtTZF5/AtC3H61 | AT5G44260 | 2 | Cytoplasm | ABA/GA | [19] | |
| AtTZF6/PEI1/AtC3H54 | AT5G07500 | 2 | Cytoplasm | ABA/GA | [19,21] | |
| AtKHZ1/AtC3H36 | AT3G12130 | 2 | Nucleus | ABA | [33] | |
| AtKHZ2/AtC3H52 | AT5G06770 | 2 | Nucleus | ABA | [33] | |
| AtC3H14 | AT1G66810 | 2 | Nucleus/cytoplasm | JA/ET/SA/BR | [24,25,26,27,28,29] | |
| AtC3H15 | AT1G68200 | 2 | Nucleus/cytoplasm | JA/ET/SA/BR | [24,25,26,27,28,29] | |
| AtCPSF30/AtC3H11 | AT1G30460 | 3 | Nucleus | Auxin/SA | [41,42] | |
| AtHIZ1 | AT1G32360 | 3 | Nucleus | Auxin | [43] | |
| rice | OsTZF1/OsC3H35 | LOC_Os05g10670 | 2 | Cytoplasm | ABA/JA/SA | [23] |
| OsTZF7 | LOC_Os10g37630 | 2 | Nucleus/cytoplasm | ABA | [34] | |
| OsC3H10 | LOC_Os01g53650 | 2 | Nucleus/cytoplasm | ABA | [35] | |
| OsC3H47 | LOC_Os07g04580 | 2 | - | ABA | [36] | |
| OsSAW1 | LOC_Os06g43120 | 5 | Nucleus | GA | [40] | |
| OsDOS/OsC3H2 | LOC_Os01g09620 | 2 | Nucleus | JA | [22] | |
| OsC3H12 | LOC_Os01g68860 | 5 | Nucleus/cytoplasm | JA | [44] | |
| OsLIC/OsC3H46 | LOC_Os06g49080 | 1 | Nucleus/cytoplasm | BR | [45,46] | |
| poplar | PaC3H17 | Potri.004G095100 | 2 | Nucleus/cytoplasm | Auxin/BR | [34,35] |
| PaC3H18 | Potri.017G119900 | 2 | Nucleus/cytoplasm | Auxin/BR | [34,35] | |
| bamboo | PeC3H74 | PH02Gene33725 | 2 | Cytoplasm | ABA | [7] |
| switchgrass | PvCCCH69 | Pavir.J04795.1 | 2 | Nucleus | ABA | [37] |
| sweet potato | IbC3H18 | MK396199 | 1 | Nucleus | ABA | [38] |
| cotton | GhZFP1 | EF403655 | 3 | - | JA | [47] |
| pepper | CaC3H14 | CA10g20930 | 3 | - | JA/ET/SA | [48] |
3.3. JA/ET Signal Response
3.4. BR Signal Response
3.5. SA Signal Response
3.6. Auxin Signal Response
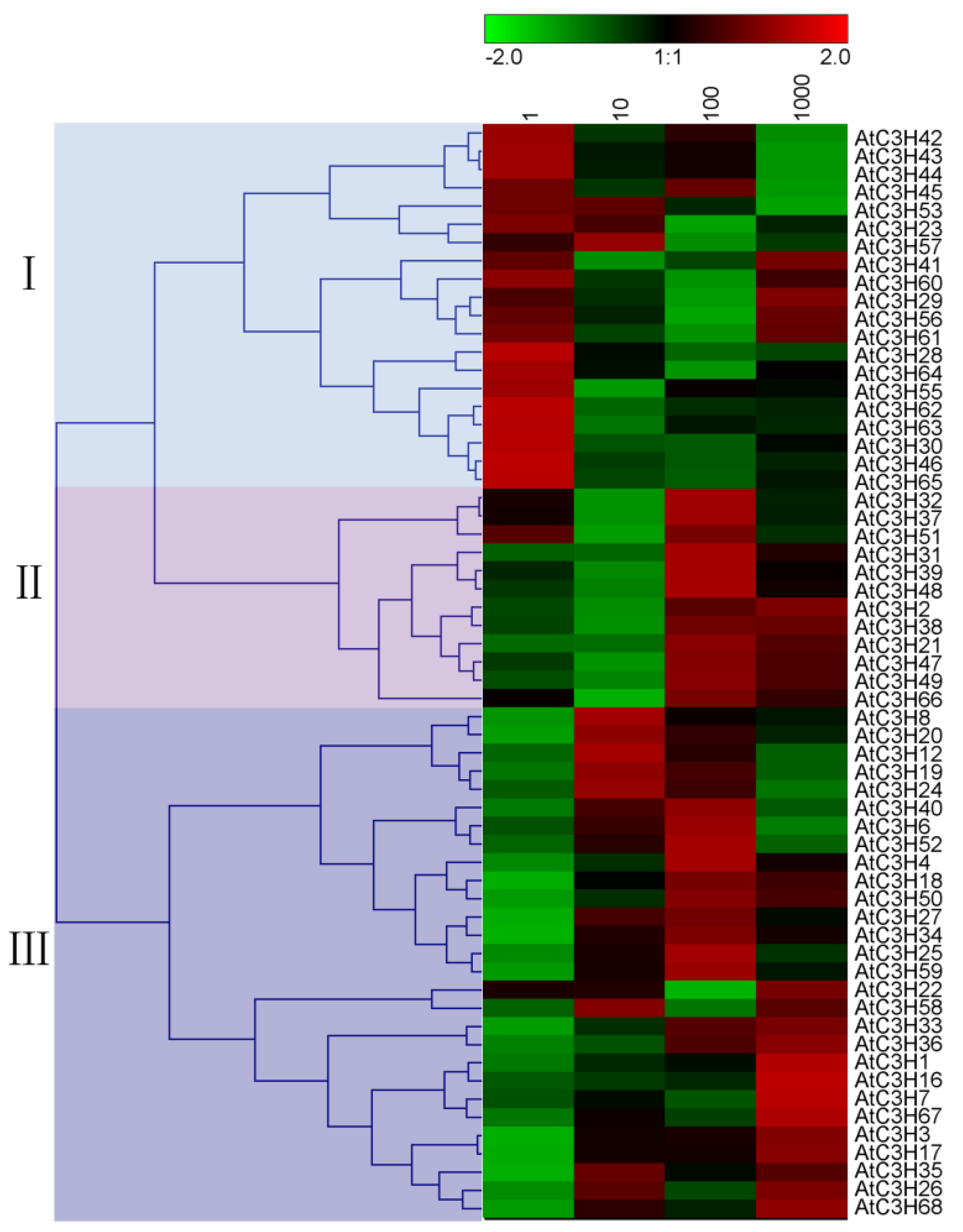
4. Prediction of the Responses of Arabidopsis CCCH Proteins to Several
Major Hormones
5. Regulatory Mechanisms of CCCH Proteins in Plants and Animals
6. Future Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Moore, M.; Ullman, C. Recent developments in the engineering of zinc finger proteins. Brief. Funct. Genom. Proteomic 2003, 1, 342–355. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brown, R.S. Zinc finger proteins: Getting a grip on RNA. Curr. Opin. Struct. Biol. 2005, 15, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Klug, A. The discovery of zinc fingers and their applications in gene regulation and genome manipulation. Annu. Rev. Biochem. 2010, 79, 213–231. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liang, J.; Wang, J.; Azfer, A.; Song, W.; Tromp, G.; Kolattukudy, P.; Fu, M. A novel CCCH-zinc finger protein family regulates proinflammatory activation of macrophages. J. Biol. Chem. 2008, 283, 6337–6346. [Google Scholar] [CrossRef] [Green Version]
- Wang, D.; Guo, Y.; Wu, C.; Yang, G.; Li, Y.; Zheng, C. Genome-wide analysis of CCCH zinc finger family in Arabidopsis and rice. BMC Genom. 2008, 9, 44. [Google Scholar] [CrossRef] [Green Version]
- Chai, G.; Hu, R.; Zhang, D.; Qi, G.; Zuo, R.; Cao, Y.; Chen, P.; Kong, Y.; Zhou, G. Comprehensive analysis of CCCH zinc finger family in poplar (Populus trichocarpa). BMC Genom. 2012, 13, 253. [Google Scholar] [CrossRef] [Green Version]
- Chen, F.; Liu, H.L.; Wang, K.; Gao, Y.M.; Wu, M.; Xiang, Y. Identification of CCCH zinc finger proteins family in Moso Bamboo (Phyllostachys edulis), and PeC3H74 confers drought tolerance to transgenic plants. Front. Plant Sci. 2020, 11, 579255. [Google Scholar] [CrossRef]
- Ngwa, C.J.; Farrukh, A.; Pradel, G. Zinc finger proteins of Plasmodium falciparum. Cell. Microbiol. 2021, 23, e13387. [Google Scholar] [CrossRef]
- Lai, W.; Carballo, E.; Thorn, J.; Kennington, E.; Blackshear, P. Interactions of CCCH zinc finger proteins with mRNA: Non-binding tristetraprolin mutants exert an inhibitory effect on degradation of AU-rich element-containing mRNAs. J. Biol. Chem. 2002, 277, 9606–9613. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Zhou, J.; Wei, Z.; Dong, H.; Yang, D.; Deng, Y.; Li, J.; Shi, S.; Sun, Y.; Lu, H.; et al. TTP-mediated regulation of mRNA stability in immune cells contributes to adaptive immunity, immune tolerance and clinical applications. RNA Biol. 2021, 18, 2150–2156. [Google Scholar] [CrossRef]
- Pomeranz, M.; Finer, J.; Jang, J. Putative molecular mechanisms underlying tandem CCCH zinc finger protein mediated plant growth, stress, and gene expression responses. Plant Signal. Behav. 2011, 6, 647–651. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jang, J. Arginine-rich motif-tandem CCCH zinc finger proteins in plant stress responses and post-transcriptional regulation of gene expression. Plant Sci. 2016, 252, 118–124. [Google Scholar] [CrossRef] [Green Version]
- Han, G.; Qiao, Z.; Li, Y.; Wang, C.; Wang, B. The roles of CCCH zinc-finger proteins in plant abiotic stress tolerance. Int. J. Mol. Sci. 2021, 22, 8327. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.; Pomeranz, M.; Jikumaru, Y.; Kang, S.; Hah, C.; Fujioka, S.; Kamiya, Y.; Jang, J. The Arabidopsis tandem zinc finger protein AtTZF1 affects ABA- and GA-mediated growth, stress and gene expression responses. Plant J. 2011, 65, 253–268. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Dudley, J.; Nei, M.; Kumar, S. MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol. Biol. Evol. 2007, 24, 1596–1599. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Jung, H.; Kang, H.; Kim, S. Arabidopsis zinc finger proteins AtC3H49/AtTZF3 and AtC3H20/AtTZF2 are involved in ABA and JA responses. Plant Cell Physiol. 2012, 53, 673–686. [Google Scholar] [CrossRef]
- Singh, N.; Swain, S.; Singh, A.; Nandi, A.K. AtOZF1 positively regulates defense against bacterial pathogens and NPR1-independent salicylic acid signaling. Mol. Plant Microbe Interact. 2018, 31, 323–333. [Google Scholar] [CrossRef] [Green Version]
- Huang, P.; Ju, H.W.; Min, J.H.; Zhang, X.; Chung, J.S.; Cheong, H.S.; Kim, C.S. Molecular and physiological characterization of the Arabidopsis thaliana. Oxidation-related Zinc Finger 2, a plasma membrane protein involved in ABA and salt stress response through the ABI2-mediated signaling pathway. Plant Cell Physiol. 2012, 53, 193–203. [Google Scholar] [CrossRef] [Green Version]
- Bogamuwa, S.; Jang, J. The Arabidopsis tandem CCCH zinc finger proteins AtTZF4, 5 and 6 are involved in light-, abscisic acid- and gibberellic acid-mediated regulation of seed germination. Plant Cell Environ. 2013, 36, 1507–1519. [Google Scholar] [CrossRef]
- Kim, D.; Yamaguchi, S.; Lim, S.; Oh, E.; Park, J.; Hanada, A.; Kamiya, Y.; Choi, G. SOMNUS, a CCCH-type zinc finger protein in Arabidopsis, negatively regulates light-dependent seed germination downstream of PIL5. Plant Cell 2008, 20, 1260–1277. [Google Scholar] [CrossRef]
- Li, Z.; Thomas, T. PEI1, an embryo-specific zinc finger protein gene required for heart-stage embryo formation in Arabidopsis. Plant Cell 1998, 10, 383–398. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kong, Z.; Li, M.; Yang, W.; Xu, W.; Xue, Y. A novel nuclear-localized CCCH-type zinc finger protein, OsDOS, is involved in delaying leaf senescence in rice. Plant Physiol. 2006, 141, 1376–1388. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jan, A.; Maruyama, K.; Todaka, D.; Kidokoro, S.; Abo, M.; Yoshimura, E.; Shinozaki, K.; Nakashima, K.; Yamaguchi-Shinozaki, K. OsTZF1, a CCCH-tandem zinc finger protein, confers delayed senescence and stress tolerance in rice by regulating stress-related genes. Plant Physiol. 2013, 161, 1202–1216. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, W.; Kim, J.; Ko, J.; Kang, H.; Kim, J.; Han, K. AtC3H14, a plant-specific tandem CCCH zinc-finger protein, binds to its target mRNAs in a sequence-specific manner and affects cell elongation in Arabidopsis thaliana. Plant J. 2014, 80, 772–784. [Google Scholar] [CrossRef] [PubMed]
- Chai, G.; Kong, Y.; Zhu, M.; Yu, L.; Qi, G.; Tang, X.; Wang, Z.; Cao, Y.; Yu, C.; Zhou, G. Arabidopsis C3H14 and C3H15 have overlapping roles in the regulation of secondary wall thickening and anther development. J. Exp. Bot. 2015, 66, 2595–2609. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, D.; Xu, H.; Huang, J.; Kong, Y.; AbuQamar, S.; Yu, D.; Liu, S.; Zhou, G.; Chai, G. The Arabidopsis CCCH protein C3H14 contributes to basal defense against Botrytis cinerea mainly through the WRKY33-dependent pathway. Plant Cell Environ. 2020, 43, 1792–1806. [Google Scholar] [CrossRef]
- Wang, D.; Chai, G.; Xu, L.; Yang, K.; Zhuang, Y.; Yang, A.; Liu, S.; Kong, Y.; Zhou, G. Phosphorylation-mediated inactivation of C3H14 by MPK4 enhances bacterial-triggered immunity in Arabidopsis. Plant Physiol. 2022, 190, 1941–1959. [Google Scholar] [CrossRef]
- Lu, P.; Chai, M.; Yang, J.; Ning, G.; Wang, G.; Ma, H. The Arabidopsis CALLOSE DEFECTIVE MICROSPORE1 gene is required for male fertility through regulating callose metabolism during microsporogenesis. Plant Physiol. 2014, 164, 1893–1904. [Google Scholar] [CrossRef] [Green Version]
- Chai, G.; Qi, G.; Wang, D.; Zhuang, Y.; Xu, H.; Bai, Z.; Bai, M.; Hu, R.; Wang, Z.; Zhou, G.; et al. The CCCH zinc finger protein C3H15 negatively regulates cell elongation by inhibiting brassinosteroid signaling. Plant Physiol. 2022, 189, 285–300. [Google Scholar] [CrossRef]
- Chai, G.; Qi, G.; Cao, Y.; Wang, Z.; Yu, L.; Tang, X.; Yu, Y.; Wang, D.; Kong, Y.; Zhou, G. Poplar PdC3H17 and PdC3H18 are direct targets of PdMYB3 and PdMYB21, and positively regulate secondary wall formation in Arabidopsis and poplar. New Phytol. 2014, 203, 520–534. [Google Scholar] [CrossRef]
- Tang, X.; Wang, D.; Liu, Y.; Lu, M.; Zhuang, Y.; Xie, Z.; Wang, C.; Wang, S.; Kong, Y.; Chai, G.; et al. Dual regulation of xylem formation by an auxin-mediated PaC3H17-PaMYB199 module in Populus. New Phytol. 2020, 225, 1545–1561. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Li, G.J.; Bressan, R.A.; Song, C.P.; Zhu, J.K.; Zhao, Y. Abscisic acid dynamics, signaling, and functions in plants. J. Integr. Plant Biol. 2020, 62, 25–54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yan, Z.; Shi, H.; Liu, Y.; Jing, M.; Han, Y. KHZ1 and KHZ2, novel members of the autonomous pathway, repress the splicing efficiency of FLC pre-mRNA in Arabidopsis. J. Exp. Bot. 2020, 71, 1375–1386. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, C.; Chen, L.; Cui, Y.; Tang, M.; Guo, Y.; Yi, Y.; Li, Y.; Liu, L.; Chen, L. RNA binding protein OsTZF7 traffics between the nucleus and processing bodies/stress granules and positively regulates drought stress in rice. Front. Plant Sci. 2022, 13, 802337. [Google Scholar] [CrossRef]
- Seong, S.Y.; Shim, J.S.; Bang, S.W.; Kim, J.K. Overexpression of OsC3H10, a CCCH-Zinc finger, improves drought tolerance in rice by regulating stress-related genes. Plants 2020, 9, 1298. [Google Scholar] [CrossRef]
- Wang, W.; Liu, B.; Xu, M.; Jamil, M.; Wang, G. ABA-induced CCCH tandem zinc finger protein decreases ABA sensitivity and promotes drought tolerance in Oryza sativa. Biochem. Biophys. Res. Commun. 2015, 464, 33–37. [Google Scholar] [CrossRef]
- Xie, Z.; Yu, G.; Lei, S.; Zhang, C.; Xu, B.; Huang, B. CCCH protein-PvCCCH69 acted as a repressor for leaf senescence through suppressing ABA-signaling pathway. Hortic. Res. 2021, 8, 165. [Google Scholar] [CrossRef]
- Zhang, H.; Gao, X.; Zhi, Y.; Li, X.; Zhang, Q.; Niu, J.; Wang, J.; Zhai, H.; Zhao, N.; Li, J.; et al. A non-tandem CCCH-type zinc-finger protein, IbC3H18, functions as a nuclear transcriptional activator and enhances abiotic stress tolerance in sweet potato. New Phytol. 2019, 223, 1918–1936. [Google Scholar] [CrossRef]
- Shtin, M.; Ioio, R.D.; Bianco, M.D. It’s Time for a Change: The role of gibberellin in root meristem development. Front. Plant Sci. 2022, 13, 882517. [Google Scholar] [CrossRef]
- Wang, B.; Fang, R.; Chen, F.; Han, J.; Liu, Y.; Chen, L.; Zhu, Q. A novel CCCH-type zinc finger protein SAW1 activates OsGA20ox3 to regulate gibberellin homeostasis and anther development in rice. J. Integr. Plant Biol. 2020, 62, 1594–1606. [Google Scholar] [CrossRef]
- Bruggeman, Q.; Garmier, M.; Bont, L.; Soubigou-Taconnat, L.; Mazubert, C.; Benhamed, M.; Raynaud, C.; Bergounioux, C.; Delarue, M. The polyadenylation factor subunit CLEAVAGE AND POLYADENYLATION SPECIFICITY FACTOR30: A key factor of programmed cell death and a regulator of immunity in Arabidopsis. Plant Physiol. 2014, 165, 732–746. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hong, L.; Ye, C.; Lin, J.; Fu, H.; Wu, X.; Li, Q. Alternative polyadenylation is involved in auxin-based plant growth and development. Plant J. 2018, 93, 246–258. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, M.; Bodi, Z.; Mackinnon, K.; Zhong, S.; Archer, N.; Mongan, N.; Simpson, G.; Fray, R. Two zinc finger proteins with functions in mA writing interact with HAKAI. Nat. Commun. 2022, 13, 1127. [Google Scholar] [CrossRef] [PubMed]
- Deng, H.; Liu, H.; Li, X.; Xiao, J.; Wang, S. A CCCH-type zinc finger nucleic acid-binding protein quantitatively confers resistance against rice bacterial blight disease. Plant Physiol. 2012, 158, 876–889. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, L.; Xu, Y.; Zhang, C.; Ma, Q.; Joo, S.H.; Kim, S.K.; Xu, Z.; Chong, K. OsLIC, a novel CCCH-type zinc finger protein with transcription activation, mediates rice architecture via brassinosteroids signaling. PLoS ONE 2008, 3, e3521. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, C.; Xu, Y.; Guo, S.; Zhu, J.; Huan, Q.; Liu, H.; Wang, L.; Luo, G.; Wang, X.; Chong, K. Dynamics of brassinosteroid response modulated by negative regulator LIC in rice. PLoS Genet. 2012, 8, e1002686. [Google Scholar] [CrossRef] [Green Version]
- Guo, Y.; Yu, Y.; Wang, D.; Wu, C.; Yang, G.; Huang, J.; Zheng, C. GhZFP1, a novel CCCH-type zinc finger protein from cotton, enhances salt stress tolerance and fungal disease resistance in transgenic tobacco by interacting with GZIRD21A and GZIPR5. New Phytol. 2009, 183, 62–75. [Google Scholar] [CrossRef]
- Qiu, A.; Lei, Y.; Yang, S.; Wu, J.; Li, J.; Bao, B.; Cai, Y.; Wang, S.; Lin, J.; Wang, Y. CaC3H14 encoding a tandem CCCH zinc finger protein is directly targeted by CaWRKY40 and positively regulates the response of pepper to inoculation by Ralstonia solanacearum. Mol. Plant Pathol. 2018, 19, 2221. [Google Scholar] [CrossRef] [Green Version]
- Wasternack, C. Jasmonates: An update on biosynthesis, signal transduction and action in plant stress response, growth and development. Ann. Bot. 2007, 100, 681–697. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, T.H.; Goossens, A.; Lacchini, E. Jasmonate: A hormone of primary importance for plant metabolism. Curr. Opin. Plant Biol. 2022, 67, 102197. [Google Scholar] [CrossRef]
- Sharma, A.; Ramakrishnan, M.; Khanna, K.; Landi, M.; Prasad, R.; Bhardwaj, R.; Zheng, B. Brassinosteroids and metalloids: Regulation of plant biology. J. Hazard. Mater. 2022, 424, 127518. [Google Scholar] [CrossRef] [PubMed]
- Nolan, T.M.; Vukašinović, N.; Liu, D.; Russinova, E.; Yin, Y. Brassinosteroids: Multidimensional regulators of plant growth, development, and stress responses. Plant Cell 2020, 32, 295–318. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ren, H.; Willige, B.C.; Jaillais, Y.; Geng, S.; Park, M.Y.; Gray, W.M.; Chory, J. BRASSINOSTEROID-SIGNALING KINASE 3, a plasma membrane-associated scaffold protein involved in early brassinosteroid signaling. PLoS Genet. 2019, 15, e1007904. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Li, X. Salicylic acid: Biosynthesis, perception, and contributions to plant immunity. Curr. Opin. Plant Biol. 2019, 50, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Cancé, C.; Martin-Arevalillo, R.; Boubekeur, K.; Dumas, R. Auxin response factors are keys to the many auxin doors. New Phytol. 2022, 235, 402–419. [Google Scholar] [CrossRef]
- Fu, M.; Blackshear, P.J. RNA-binding proteins in immune regulation: A focus on CCCH zinc finger proteins. Nat. Rev. Immunol. 2017, 17, 130–143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fabian, M.R.; Frank, F.; Rouya, C.; Siddiqui, N.; Lai, W.S.; Karetnikov, A.; Blackshear, P.J.; Nagar, B.; Sonenberg, N. Structural basis for the recruitment of the human CCR4-NOT deadenylase complex by tristetraprolin. Nat. Struct. Mol. Biol. 2013, 20, 735–739. [Google Scholar] [CrossRef] [Green Version]
- Makita, S.; Takatori, H.; Nakajima, H. Post-transcriptional regulation of immune responses and inflammatory diseases by RNA-binding ZFP36 family proteins. Front. Immunol. 2021, 12, 711633. [Google Scholar] [CrossRef]
- Essig, K.; Kronbeck, N.; Guimaraes, J.; Lohs, C.; Schlundt, A.; Hoffmann, A.; Behrens, G.; Brenner, S.; Kowalska, J.; Lopez-Rodriguez, C.; et al. Roquin targets mRNAs in a 3’-UTR-specific manner by different modes of regulation. Nat. Commun. 2018, 9, 3810. [Google Scholar] [CrossRef] [Green Version]
- Hoefig, K.; Reim, A.; Gallus, C.; Wong, E.; Behrens, G.; Conrad, C.; Xu, M.; Kifinger, L.; Ito-Kureha, T.; Defourny, K.; et al. Defining the RBPome of primary T helper cells to elucidate higher-order Roquin-mediated mRNA regulation. Nat. Commun. 2021, 12, 5208. [Google Scholar] [CrossRef]
- Lai, W.; Stumpo, D.; Blackshear, P. Rapid insulin-stimulated accumulation of an mRNA encoding a proline-rich protein. J. Biol. Chem. 1990, 265, 16556–16563. [Google Scholar] [CrossRef]
- Qu, J.; Kang, S.; Wang, W.; Musier-Forsyth, K.; Jang, J. The Arabidopsis thaliana tandem zinc finger 1 (AtTZF1) protein in RNA binding and decay. Plant J. 2014, 78, 452–467. [Google Scholar] [CrossRef] [Green Version]
- Taylor, G.A.; Thompson, M.J.; Lai, W.S.; Blackshear, P.J. Mitogens stimulate the rapid nuclear to cytosolic translocation of tristetraprolin, a potential zinc-finger transcription factor. Mol. Endocrinol. 1996, 10, 140–146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seok, H.; Woo, D.; Park, H.; Lee, S.; Tran, H.; Lee, E.; Nguyen, L.V.; Moon, Y. AtC3H17, a non-tandem CCCH zinc finger protein, functions as a nuclear transcriptional activator and has pleiotropic effects on vegetative development, flowering and seed development in Arabidopsis. Plant Cell Physiol. 2016, 57, 603–615. [Google Scholar] [CrossRef] [PubMed]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Q.; Song, S.; Lu, X.; Wang, Y.; Chen, Y.; Wu, X.; Tan, L.; Chai, G. Hormone Regulation of CCCH Zinc Finger Proteins in Plants. Int. J. Mol. Sci. 2022, 23, 14288. https://doi.org/10.3390/ijms232214288
Wang Q, Song S, Lu X, Wang Y, Chen Y, Wu X, Tan L, Chai G. Hormone Regulation of CCCH Zinc Finger Proteins in Plants. International Journal of Molecular Sciences. 2022; 23(22):14288. https://doi.org/10.3390/ijms232214288
Chicago/Turabian StyleWang, Qiao, Shangfa Song, Xintong Lu, Yiqing Wang, Yan Chen, Xiuwen Wu, Li Tan, and Guohua Chai. 2022. "Hormone Regulation of CCCH Zinc Finger Proteins in Plants" International Journal of Molecular Sciences 23, no. 22: 14288. https://doi.org/10.3390/ijms232214288






