Lipid Droplet-Associated Proteins Perilipin 1 and 2: Molecular Markers of Steatosis and Microvesicular Steatotic Foci in Chronic Hepatitis C
Abstract
1. Introduction
2. Results
2.1. LDs and Perilipin Expression in Liver
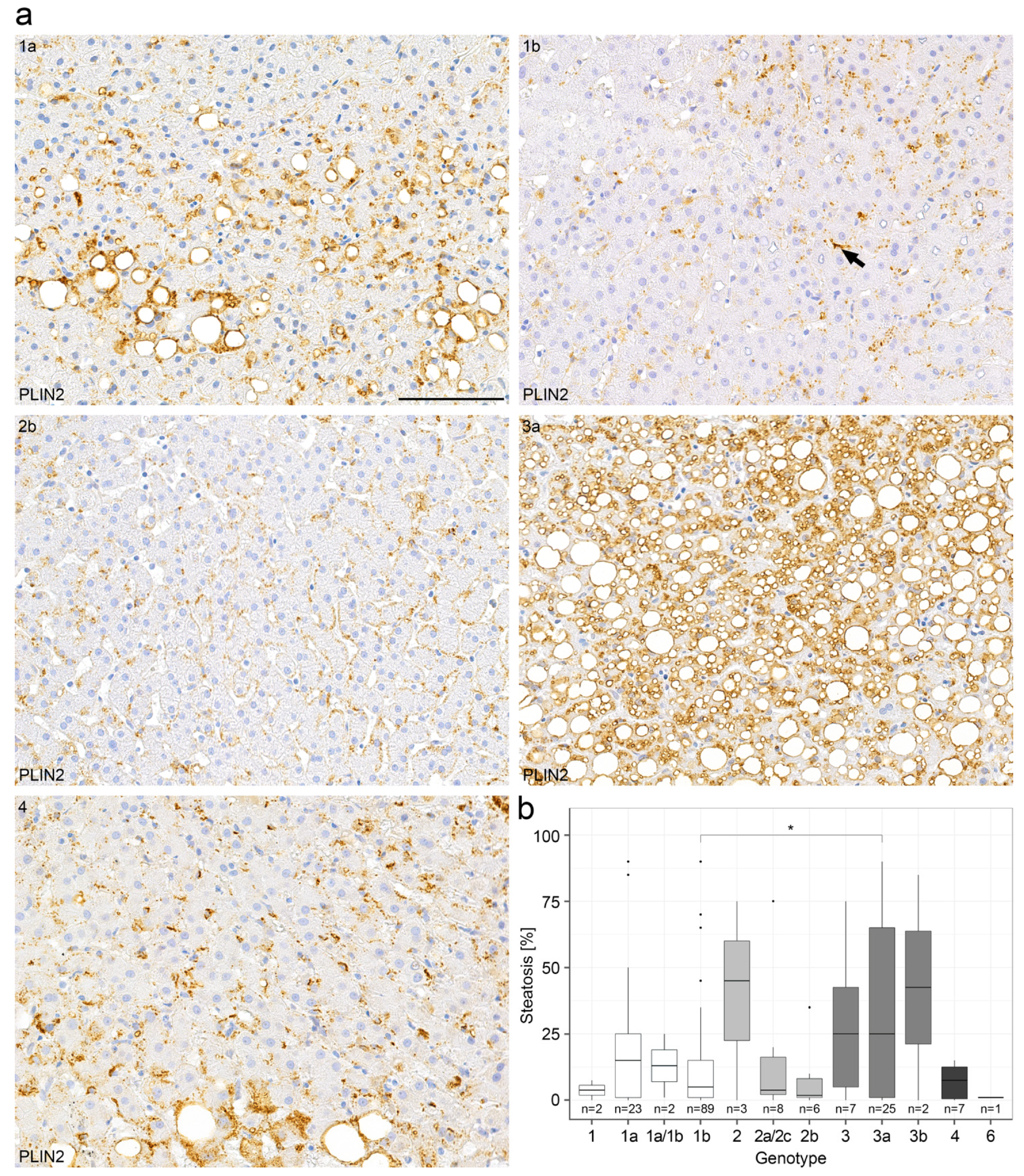
2.2. Steatosis in Chronic Hepatitis C
2.3. Steatotic Foci in Chronic Hepatitis C
2.4. Steatotic Foci Are Areas of Focal Metabolic Dysregulation
3. Discussion
4. Material & Methods
4.1. Tissue and Collectives
4.2. Immunohistochemistry and Histochemistry
4.3. Immunofluorescence Microscopy
4.4. RNA Chromogen In Situ Hybridization (CISH)
4.5. Electron Microscopy
4.6. Evaluation of Immunohistochemistry
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
Appendix A
| Parameter | HCV Genotypes | |||||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 6 | Not Tested/Unknown | |
| Age [a] | ||||||
| Median | 43 | 41 | 33 | 38 | 46 | 45.5 |
| mean | 43.43 | 41.82 | 33.09 | 39 | 46 | 45.68 |
| (SD) | (11.85) | (10.41) | (8.13) | (2.94) | - | (12.73) |
| N | 116 | 17 | 34 | 7 | 1 | 56 |
| Sex (♂) | ||||||
| [%] | 52.59 | 47.06 | 67.65 | 85.71 | 100 | 60.71 |
| AST [U/L] | ||||||
| mean | 28.1 | 26.8 | 29.9 | 35.7 | 24 | 29.7 |
| (SD) | (21.3) | (20.3) | (22.8) | (28) | - | (26.6) |
| N | 116 | 17 | 34 | 7 | 1 | 56 |
| ALT [U/L] | ||||||
| mean | 47.6 | 49.2 | 57.8 | 61.9 | 32 | 46.1 |
| SD | (42.3) | (48.1) | (52.5) | (44.9) | - | (39.1) |
| N | 116 | 17 | 34 | 7 | 1 | 56 |
| γ-GT [U/L] | ||||||
| mean | 40.6 | 31.6 | 31.9 | 53.9 | 21 | 66.6 |
| (SD) | (47.7) | (40) | (40.7) | (68.8) | - | (197.2) |
| N | 116 | 17 | 34 | 7 | 1 | 56 |
| AP [U/L] | ||||||
| mean | 120 | 124 | 126 | 140 | 49 | 123 |
| SD | (50) | (54) | (74) | (73) | - | (45) |
| N | 112 | 17 | 33 | 7 | 1 | 56 |
| Bilirubin (whole) [mg/dL] | ||||||
| mean | 0.64 | 0.88 | 0.67 | 0.59 | 0.6 | 0.82 |
| (SD) | (0.42) | (0.85) | (0.35) | (0.25) | - | (0.54) |
| N | 114 | 17 | 33 | 7 | 1 | 56 |
| Viral load [IU/mL] | ||||||
| mean | 798,569.34 | 622.200 | 693,757.5 | 363,500 | - | 800,366.67 |
| (SD) | (1009,947.01) | (466,505.16) | (657,027.44) | (181,726.44) | - | (667,698.51) |
| N | 61 | 10 | 18 | 2 | 0 | 15 |
| Parameter | HCV Genotypes | |||||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 6 | Not Tested/Unknown | |
| Albumin [g/L] | ||||||
| mean | 60.29 | 59.77 | 60.25 | 55.73 | 47.4 | 57.82 |
| (SD) | (4.80) | (6.54) | (6.51) | (11.02) | - | (7.06) |
| N | 106 | 17 | 33 | 7 | 1 | 43 |
| CHE [kU/L] | ||||||
| mean | 6.13 | 5.82 | 5.72 | 5.36 | 7 | 5.64 |
| SD | (1.57) | (1.76) | (1.82) | (2.24) | - | (1.81) |
| N | 112 | 17 | 33 | 7 | 1 | 43 |
| BMI [kg/m2] | ||||||
| mean | 24.61 | 25.71 | 23.09 | 23.11 | 22.40 | 25.79 |
| SD | (3.89) | (4.54) | (3.34) | (2.39) | - | (4.2) |
| N | 116 | 17 | 34 | 7 | 1 | 56 |
| Glucose [mg/dL] | ||||||
| mean | 95.93 | 100.71 | 89.12 | 90.86 | 73 | 98.23 |
| SD | (34.11) | (23.22) | (11.68) | (13.59) | - | (25.59) |
| N | 108 | 17 | 34 | 7 | 1 | 48 |
| TAG [mg/dL] | ||||||
| mean | 118.56 | 114.13 | 103.03 | 114.2 | 144 | 125.92 |
| SD | (78.65) | (77.99) | (60.87) | (35.56) | - | (77.55) |
| N | 108 | 16 | 33 | 5 | 1 | 48 |
| Cholesterol [mg/dL] | ||||||
| mean | 184.53 | 187.19 | 159.42 | 148.6 | 229 | 187.13 |
| SD | (37.80) | (45.00) | (48.34) | (30.40) | - | (42.73) |
| N | 108 | 16 | 33 | 5 | 1 | 48 |
| γ-Globulins [%] | ||||||
| mean | 17.13 | 18.18 | 17.56 | 21.73 | 26.8 | 19.59 |
| SD | (4.11) | (5.66) | (5.47) | (9.93) | - | (7.05) |
| N | 105 | 17 | 33 | 7 | 1 | 43 |
References
- MacSween, R.; Burt, A. Histologic Spectrum of Alcoholic Liver Disease. Semin. Liver Dis. 1986, 6, 221–232. [Google Scholar] [CrossRef] [PubMed]
- Lieber, C. Mechanism of ethanol induced hepatic injury. Pharmacol. Ther. 1990, 46, 1–41. [Google Scholar] [CrossRef] [PubMed]
- Goodman, Z.D.; Ishak, K.G. Histopathology of Hepatitis C Virus Infection. Semin. Liver Dis. 1995, 15, 70–81. [Google Scholar] [CrossRef] [PubMed]
- Gluchowski, N.L.; Becuwe, M.; Walther, T.C.; Farese, R.V., Jr. Lipid droplets and liver disease: From basic biology to clinical implica-tions. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 343–355. [Google Scholar] [CrossRef] [PubMed]
- Younossi, Z.M.; Stepanova, M.; Younossi, Y.; Golabi, P.; Mishra, A.; Rafiq, N.; Henry, L. Epidemiology of chronic liver diseases in the USA in the past three decades. Gut 2019, 69, 564–568. [Google Scholar] [CrossRef]
- Bedogni, G.; Miglioli, L.; Masutti, F.; Tiribelli, C.; Marchesini, G.; Bellentani, S. Prevalence of and risk factors for nonalcoholic fatty liver disease: The Dionysos nutrition and liver study. Hepatology 2005, 42, 44–52. [Google Scholar] [CrossRef]
- Stein, E.; Cruz-Lemini, M.; Altamirano, J.; Ndugga, N.; Couper, D.; Abraldes, J.G.; Bataller, R. Heavy daily alcohol intake at the population level predicts the weight of alcohol in cirrhosis burden worldwide. J. Hepatol. 2016, 65, 998–1005. [Google Scholar] [CrossRef]
- Bataller, R.; Arteel, G.E.; Moreno, C.; Shah, V. Alcohol-related liver disease: Time for action. J. Hepatol. 2019, 70, 221–222. [Google Scholar] [CrossRef]
- World Health Organization. Global Hepatitis Report 2017; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Fartoux, L.; Poujol-Robert, A.; Guéchot, J.; Wendum, D.; Poupon, R.; Serfaty, L. Insulin resistance is a cause of steatosis and fibrosis progression in chronic hepatitis C. Gut 2005, 54, 1003–1008. [Google Scholar] [CrossRef]
- Torbenson, M.; Zen, Y.; Yeh, M. Tumors of the Liver, AFIP Atlas of Tumor Pathology Series 4; American Registry of Pathology: Washington, DC, USA, 2018; pp. 39–112. [Google Scholar]
- Younossi, Z.; Tacke, F.; Arrese, M.; Sharma, B.C.; Mostafa, I.; Bugianesi, E.; Wong, V.W.-S.; Yilmaz, Y.; George, J.; Fan, J.; et al. Global Perspectives on Nonalcoholic Fatty Liver Disease and Nonalcoholic Steatohepatitis. Hepatology 2019, 69, 2672–2682. [Google Scholar] [CrossRef]
- Tannapfel, A.; Denk, H.; Dienes, H.-P.; Langner, C.; Schirmacher, P.; Trauner, M.; Flott-Rahmel, B. Histopathological diagnosis of non-alcoholic and alcoholic fatty liver disease. Virchows Arch. 2011, 458, 511–523. [Google Scholar] [CrossRef] [PubMed]
- Celli, R.; Zhang, X. Pathology of Alcoholic Liver Disease. J. Clin. Transl. Hepatol. 2014, 2, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Bedossa, P. Pathology of non-alcoholic fatty liver disease. Liver Int. 2017, 37, 85–89. [Google Scholar] [CrossRef]
- Caldwell, S.; Ikura, Y.; Dias, D.; Isomoto, K.; Yabu, A.; Moskaluk, C.; Pramoonjago, P.; Simmons, W.; Scruggs, H.; Rosenbaum, N.; et al. Hepatocellular ballooning in NASH. J. Hepatol. 2010, 53, 719–723. [Google Scholar] [CrossRef]
- Kakisaka, K.; Cazanave, S.C.; Werneburg, N.W.; Razumilava, N.; Mertens, J.C.; Bronk, S.F.; Gores, G.J. A hedgehog survival pathway in ’undead’ lipotoxic hepatocytes. J. Hepatol. 2012, 57, 844–851. [Google Scholar] [CrossRef] [PubMed]
- Lackner, C.; Gogg-Kamerer, M.; Zatloukal, K.; Stumptner, C.; Brunt, E.M.; Denk, H. Ballooned hepatocytes in steatohepatitis: The value of keratin immunohistochemistry for diagnosis. J. Hepatol. 2008, 48, 821–828. [Google Scholar] [CrossRef]
- Blaner, W.S.; O’Byrne, S.M.; Wongsiriroj, N.; Kluwe, J.; D’Ambrosio, D.M.; Jiang, H.; Schwabe, R.F.; Hillman, E.M.; Piantedosi, R.; Libien, J. Hepatic stellate cell lipid droplets: A specialized lipid droplet for retinoid storage. Biochim. et Biophys. Acta (BBA) Mol. Cell Biol. Lipids 2009, 1791, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Scorletti, E.; Carr, R.M. A new perspective on NAFLD: Focusing on lipid droplets. J. Hepatol. 2021, 76, 934–945. [Google Scholar] [CrossRef]
- Denk, H.; Franke, W.W.; Eckerstorfer, R.; Schmid, E.; Kerjaschki, D. Formation and involution of Mallory bodies (“alcoholic hyalin”) in murine and human liver revealed by immunofluorescence microscopy with antibodies to prekeratin. Proc. Natl. Acad. Sci. USA 1979, 76, 4112–4116. [Google Scholar] [CrossRef]
- Strnad, P.; Zatloukal, K.; Stumptner, C.; Kulaksiz, H.; Denk, H. Mallory-Denk-bodies: Lessons from keratin-containing hepatic in-clusion bodies. Biochim. Et Biophys. Acta 2008, 1782, 764–774. [Google Scholar] [CrossRef]
- Xu, Z.; Zhai, L.; Yi, T.; Gao, H.; Fan, F.; Li, Y.; Wang, Y.; Li, N.; Xing, X.; Su, N.; et al. Hepatitis B virus X induces inflammation and cancer in mice liver through dysregulation of cytoskeletal remodeling and lipid metabolism. Oncotarget 2016, 7, 70559–70574. [Google Scholar] [CrossRef] [PubMed]
- Miyanari, Y.; Atsuzawa, K.; Usuda, N.; Watashi, K.; Hishiki, T.; Zayas, M.; Bartenschlager, R.; Wakita, T.; Hijikata, M.; Shimotohno, K. The lipid droplet is an important organelle for hepatitis C virus production. Nature 2007, 9, 1089–1097. [Google Scholar] [CrossRef] [PubMed]
- Barba, G.; Harper, F.; Harada, T.; Kohara, M.; Goulinet, S.; Matsuura, Y.; Eder, G.; Schaff, Z.; Chapman, M.J.; Miyamura, T.; et al. Hepatitis C virus core protein shows a cytoplasmic localization and associates to cellular lipid storage droplets. Proc. Natl. Acad. Sci. USA 1997, 94, 1200–1205. [Google Scholar] [CrossRef] [PubMed]
- Bulankina, A.V.; Richter, R.M.; Welsch, C. Regulatory Role of Phospholipids in Hepatitis C Virus Replication and Protein Function. Pathogens 2022, 11, 102. [Google Scholar] [CrossRef] [PubMed]
- Finkelmeier, F.; Dultz, G.; Peiffer, K.-H.; Kronenberger, B.; Krauss, F.; Zeuzem, S.; Sarrazin, C.; Vermehren, J.; Waidmann, O. Risk of de novo Hepatocellular Carcinoma after HCV Treatment with Direct-Acting Antivirals. Liver Cancer 2018, 7, 190–204. [Google Scholar] [CrossRef] [PubMed]
- Antala, S.; Diamond, T.; Kociolek, L.K.; Shah, A.A.; Chapin, C.A. Severe Hepatitis in Pediatric Coronavirus Disease 2019. J. Craniofacial Surg. 2022, 74, 631–635. [Google Scholar] [CrossRef] [PubMed]
- Cooper, S.; Tobar, A.; Konen, O.; Orenstein, N.; Gilad, N.; Landau, Y.E.; Mozer-Glassberg, Y.; Rozenfeld Bar-Lev, M.; Shaoul, R.; Shamir, R.; et al. Long COVID-19 Liver Manifestation in Children. J. Pediatr. Gastroenterol. Nutr. 2022, 75, 244–251. [Google Scholar] [CrossRef]
- Gutierrez Sanchez, L.H.; Shiau, H.; Baker, J.M.; Saaybi, S.; Buchfellner, M.; Britt, W.; Sanchez, V.; Potter, J.L.; Ingram, L.A.; Kelly, D.; et al. A Case Series of Children with Acute Hepatitis and Human Adenovirus Infection. N. Engl. J. Med. 2022, 387, 620–630. [Google Scholar] [CrossRef]
- Morfopoulou, S.; Buddle, S.; Torres Montaguth, O.E.; Atkinson, L.; Guerra-Assunção, J.A.; Storey, N.; Roy, S.; Lennon, A.; Lee, J.C.D.; Williams, R.; et al. Genomic Investigations of Acute Hepatitis of Unknown Aetiology in Children. medRxiv 2022, 27, 7963. [Google Scholar]
- Itabe, H.; Yamaguchi, T.; Nimura, S.; Sasabe, N. Perilipins: A diversity of intracellular lipid droplet proteins. Lipids Health Dis. 2017, 16, 83. [Google Scholar] [CrossRef]
- Franke, W.W.; Hergt, M.; Grund, C. Rearrangement of the vimentin cytoskeleton during adipose conversion: Formation of an intermediate filament cage around lipid globules. Cell 1987, 49, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Heid, H.; Rickelt, S.; Zimbelmann, R.; Winter, S.; Schumacher, H.; Dorflinger, Y. Lipid droplets, perilipins and cy-tokeratins--unravelled liaisons in epithelium-derived cells. PLoS ONE 2013, 8, e63061. [Google Scholar] [CrossRef] [PubMed]
- Heid, H.; Rickelt, S.; Zimbelmann, R.; Winter, S.; Schumacher, H.; Dörflinger, Y.; Kuhn, C.; Franke, W.W. On the Formation of Lipid Droplets in Human Adipocytes: The Organization of the Perilipin–Vimentin Cortex. PLoS ONE 2014, 9, e90386. [Google Scholar] [CrossRef] [PubMed]
- Heid, H.; Zimbelmann, R.; Dörflinger, Y.; Rickelt, S. Formation and degradation of lipid droplets in human adipocytes and the expression of aldehyde oxidase (AOX). Cell Tissue Res. 2019, 379, 45–62. [Google Scholar] [CrossRef] [PubMed]
- Robenek, H.; Severs, N.J. Lipid droplet growth by fusion: Insights from freeze-fracture imaging. J. Cell. Mol. Med. 2009, 13, 4657–4661. [Google Scholar] [CrossRef]
- Jüngst, C.; Winterhalder, M.J.; Zumbusch, A. Fast and long term lipid droplet tracking with CARS microscopy. J. Biophotonics 2011, 4, 435–441. [Google Scholar] [CrossRef]
- Paar, M.; Jüngst, C.; Steiner, N.A.; Magnes, C.; Sinner, F.; Kolb, D.; Lass, A.; Zimmermann, R.; Zumbusch, A.; Kohlwein, S.D.; et al. Remodeling of Lipid Droplets during Lipolysis and Growth in Adipocytes. J. Biol. Chem. 2012, 287, 11164–11173. [Google Scholar] [CrossRef]
- Beller, M.; Bulankina, A.V.; Hsiao, H.-H.; Urlaub, H.; Jäckle, H.; Kühnlein, R.P. PERILIPIN-Dependent Control of Lipid Droplet Structure and Fat Storage in Drosophila. Cell Metab. 2010, 12, 521–532. [Google Scholar] [CrossRef]
- Greenberg, A.S.; Egan, J.J.; Wek, S.A.; Garty, N.B.; Blanchette-Mackie, E.J.; Londos, C. Perilipin, a major hormonally regulated adipo-cyte-specific phosphoprotein associated with the periphery of lipid storage droplets. J. Biol. Chem. 1991, 266, 11341–11346. [Google Scholar] [CrossRef]
- Brasaemle, D.L.; Barber, T.; Wolins, N.E.; Serrero, G.; Blanchette-Mackie, E.J.; Londos, C. Adipose differentiation-related protein is an ubiquitously expressed lipid storage droplet-associated protein. J. Lipid Res. 1997, 38, 2249–2263. [Google Scholar] [CrossRef]
- Heid, H.W.; Moll, R.; Schwetlick, I.; Rackwitz, H.-R.; Keenan, T.W. Adipophilin is a specific marker of lipid accumulation in diverse cell types and diseases. Cell Tissue Res. 1998, 294, 309–321. [Google Scholar] [CrossRef] [PubMed]
- Bulankina, A.V.; Deggerich, A.; Wenzel, D.; Mutenda, K.; Wittmann, J.G.; Rudolph, M.G.; Burger, K.N.; Höning, S. TIP47 functions in the biogenesis of lipid droplets. J. Cell Biol. 2009, 185, 641–655. [Google Scholar] [CrossRef] [PubMed]
- Wolins, N.E.; Skinner, J.R.; Schoenfish, M.J.; Tzekov, A.; Bensch, K.G.; Bickel, P.E. Adipocyte protein S3-12 coats nascent lipid droplets. J. Biol. Chem. 2003, 278, 37713–37721. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Matsushita, S.; Motojima, K.; Hirose, F.; Osumi, T. MLDP, a Novel PAT Family Protein Localized to Lipid Droplets and Enriched in the Heart, Is Regulated by Peroxisome Proliferator-activated Receptor α. J. Biol. Chem. 2006, 281, 14232–14240. [Google Scholar] [CrossRef] [PubMed]
- Wolins, N.E.; Quaynor, B.K.; Skinner, J.R.; Tzekov, A.; Croce, M.A.; Gropler, M.C.; Varma, V.; Yao-Borengasser, A.; Rasouli, N.; Kern, P.A.; et al. OXPAT/PAT-1 is a PPAR-induced lipid droplet protein that promotes fatty acid utilization. Diabetes 2006, 55, 3418–3428. [Google Scholar] [CrossRef] [PubMed]
- Sztalryd, C.; Brasaemle, D.L. The perilipin family of lipid droplet proteins: Gatekeepers of intracellular lipolysis. Biochim. Biophys. Acta (BBA) Mol. Cell Biol. Lipids 2017, 1862, 1221–1232. [Google Scholar] [CrossRef]
- Straub, B.K.; Gyoengyoesi, B.; Koenig, M.; Hashani, M.; Pawella, L.M.; Herpel, E.; Mueller, W.; Macher-Goeppinger, S.; Heid, H.; Schirmacher, P. Adipophilin/perilipin-2 as a lipid droplet-specific marker for metabolically active cells and diseases associated with metabolic dysregulation. Histopathology 2012, 62, 617–631. [Google Scholar] [CrossRef]
- Straub, B.K.; Stöffel, P.D.; Heid, H.; Zimbelmann, R.; Schirmacher, P. Differential pattern of lipid droplet-associated proteins and de novo perilipin expression in hepatocyte steatogenesis. Hepatology 2008, 46, 1936–1946. [Google Scholar] [CrossRef]
- Krahmer, N.; Farese, R.V., Jr.; Walther, T.C. Balancing the fat: Lipid droplets and human disease. EMBO Mol. Med. 2013, 5, 973–983. [Google Scholar] [CrossRef]
- Carr, R.M.; Ahima, R.S. Pathophysiology of lipid droplet proteins in liver diseases. Exp. Cell Res. 2016, 340, 187–192. [Google Scholar] [CrossRef]
- Najt, C.P.; Senthivinayagam, S.; Aljazi, M.B.; Fader, K.A.; Olenic, S.D.; Brock, J.R.; Lydic, T.A.; Jones, A.D.; Atshaves, B.P. Liver-specific loss of Perilipin 2 alleviates di-et-induced hepatic steatosis, inflammation, and fibrosis. Am. J. Physiol.-Gastrointest. Liver Physiol. 2016, 310, G726–G738. [Google Scholar] [CrossRef] [PubMed]
- Warburg, O. On the Origin of Cancer Cells. Science 1956, 123, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Straub, B.K.; Schirmacher, P. Pathology and Biopsy Assessment of Non-Alcoholic Fatty Liver Disease. Dig. Dis. 2010, 28, 197–202. [Google Scholar] [CrossRef]
- Straub, B.K.; Witzel, H.R.; Pawella, L.M.; Renner, M.; Eiteneuer, E.; Hashani, M.; Schirmacher, P.; Roth, W.; Mechtersheimer, G. Perilipin 1 Expression Differentiates Liposarcoma from Other Types of Soft Tissue Sarcoma. Am. J. Pathol. 2019, 189, 1547–1558. [Google Scholar] [CrossRef]
- Fujii, H.; Ikura, Y.; Arimoto, J.; Sugioka, K.; Iezzoni, J.C.; Park, S.H.; Naruko, T.; Itabe, H.; Kawada, N.; Caldwell, S.H.; et al. Expression of Perilipin and Adipophilin in Nonalcoholic Fatty Liver Disease; Relevance to Oxidative Injury and Hepatocyte Ballooning. J. Atheroscler. Thromb. 2009, 16, 893–901. [Google Scholar] [CrossRef] [PubMed]
- Pawella, L.M.; Hashani, M.; Eiteneuer, E.; Renner, M.; Bartenschlager, R.; Schirmacher, P.; Straub, B.K. Perilipin discerns chronic from acute hepatocellular steatosis. J. Hepatol. 2013, 60, 633–642. [Google Scholar] [CrossRef] [PubMed]
- Boulant, S.; Douglas, M.W.; Moody, L.; Budkowska, A.; Targett-Adams, P.; McLauchlan, J. Hepatitis C Virus Core Protein Induces Lipid Droplet Redistribution in a Microtubule- and Dynein-Dependent Manner. Traffic 2008, 9, 1268–1282. [Google Scholar] [CrossRef]
- Piodi, A.; Chouteau, P.; Lerat, H.; Hezode, C.; Pawlotsky, J. Morphological changes in intracellular lipid droplets induced by different hepatitis C virus genotype core sequences and relationship with steatosis. Hepatology 2008, 48, 16–27. [Google Scholar] [CrossRef]
- Jamal, S.; Mubarik, A.; Zubair, A. Morphometric analysis of hepatic steatosis in chronic hepatitis C infection. Saudi J. Gastroenterol. 2009, 15, 11–14. [Google Scholar] [CrossRef]
- Campana, B.; Calabrese, D.; Matter, M.S.; Terracciano, L.M.; Wieland, S.F.; Heim, M.H. In vivo analysis at the cellular level reveals similar steatosis induction in both hepatitis C virus genotype 1 and 3 infections. J. Viral Hepat. 2017, 25, 262–271. [Google Scholar] [CrossRef]
- Ribback, S.; Calvisi, D.F.; Cigliano, A.; Sailer, V.; Peters, M.; Rausch, J.; Heidecke, C.-D.; Birth, M.; Dombrowski, F. Molecular and metabolic changes in human liver clear cell foci resemble the alterations occurring in rat hepatocarcinogenesis. J. Hepatol. 2013, 58, 1147–1156. [Google Scholar] [CrossRef] [PubMed]
- Asselah, T.; Rubbia-Brandt, L.; Marcellin, P.; Negro, F. Steatosis in chronic hepatitis C: Why does it really matter? Gut 2006, 55, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Hourioux, C.; Patient, R.; Morin, A.; Blanchard, E.; Moreau, A.; Trassard, S.; Giraudeau, B.; Roingeard, P. The genotype 3-specific hepatitis C virus core protein residue phenylalanine 164 increases steatosis in an in vitro cellular model. Gut 2007, 56, 1302–1308. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.F.; Dai, C.Y.; Yeh, M.L.; Huang, C.I.; Tai, C.M.; Hsieh, M.H.; Yang, H.-L.; Huang, J.-F.; Lin, Z.-Y.; Chen, S.-C.; et al. Association of diabetes and PNPLA3 genetic variants with disease severity of patients with chronic hepatitis C virus infection. J. Hepatol. 2015, 62, 512–518. [Google Scholar] [CrossRef] [PubMed]
- Degasperi, E.; Galmozzi, E.; Pelusi, S.; D’Ambrosio, R.; Soffredini, R.; Borghi, M.; Perbellini, R.; Facchetti, F.; Iavarone, M.; SanGiovanni, A.; et al. Hepatic Fat—Genetic Risk Score Predicts Hepatocellular Carcinoma in Patients With Cirrhotic HCV Treated With DAAs. Hepatology 2020, 72, 1912–1923. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-Y.; Cortese, M.; Haselmann, U.; Tabata, K.; Romero-Brey, I.; Funaya, C.; Schieber, N.L.; Qiang, Y.; Bartenschlager, M.; Kallis, S.; et al. Spatiotemporal Coupling of the Hepatitis C Virus Replication Cycle by Creating a Lipid Droplet- Proximal Membranous Replication Compartment. Cell Rep. 2019, 27, 3602–3617.e5. [Google Scholar] [CrossRef]
- Camus, G.; Schweiger, M.; Herker, E.; Harris, C.; Kondratowicz, A.S.; Tsou, C.L.; Herath, K.; Previs, S.F.; Roddy, T.P.; Pinto, S.; et al. The hepatitis C virus core protein inhibits adipose triglyceride lipase (ATGL)-mediated lipid mobilization and enhances the ATGL interaction with comparative gene identifica-tion 58 (CGI-58) and lipid droplets. J. Biol. Chem. 2014, 289, 35770–35780. [Google Scholar] [CrossRef]
- Zhang, J.; Gao, X.; Yuan, Y.; Sun, C.; Zhao, Y.; Xiao, L.; Yang, Y.; Gu, Y.; Yang, R.; Hu, P.; et al. Perilipin 5 alleviates HCV NS5A-induced lipotoxic injuries in liver. Lipids Health Dis. 2019, 18, 1–12. [Google Scholar] [CrossRef]
- Geidl-Flueck, B.; Hochuli, M.; Németh, Á.; Eberl, A.; Derron, N.; Köfeler, H.C.; Tappy, L.; Berneis, K.; Spinas, G.A.; Gerber, P.A. Fructose- and sucrose- but not glucose-sweetened beverages promote hepatic de novo lipogenesis: A randomized controlled trial. J. Hepatol. 2021, 75, 46–54. [Google Scholar] [CrossRef]
- Kim, S.-A.; Shin, S. Fruit and vegetable consumption and non-alcoholic fatty liver disease among Korean adults: A prospective cohort study. J. Epidemiol. Community Health 2020, 74, 1035–1042. [Google Scholar] [CrossRef]
- Aminian, A.; Al-Kurd, A.; Wilson, R.; Bena, J.; Fayazzadeh, H.; Singh, T.; Albaugh, V.L.; Shariff, F.U.; Rodriguez, N.A.; Jin, J.; et al. Association of Bariatric Surgery With Major Adverse Liver and Cardiovascular Outcomes in Patients With Biopsy-Proven Nonalcoholic Steatohepatitis. JAMA 2021, 326, 2031. [Google Scholar] [CrossRef] [PubMed]
- Brawer, M.K.; Austin, G.E.; Lewin, K.J. Focal fatty change of the liver, a hitherto poorly recognized entity. Gastroenterology 1980, 78, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Grove, A.; Vyberg, B.; Vyberg, M. Focal fatty change of the liver. A review and a case associated with continuous ambulatory peritoneal dialysis. Virchows Archiv. A Pathol. Anat. Histopathol. 1991, 419, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Sterling, R.K.; Herbener, T.E.; Jacobs, G.H.; Post, A.B.; Carey, J.T.; Haaga, J.R. Multifocal hepatic lesions in AIDS: An unusual presentation of steatosis. Am. J. Gastroenterol. 1997, 92, 1934–1936. [Google Scholar]
- Patton, H.M.; Lavine, J.E. Focal Fatty liver: More than just a radiographic curiosity? Eur. J. Gastroenterol. Hepatol. 2007, 3, 199–200. [Google Scholar]
- Bannasch, P.; Ribback, S.; Su, Q.; Mayer, D. Clear cell hepatocellular carcinoma: Origin, metabolic traits and fate of glycogenotic clear and ground glass cells. Hepatobiliary Pancreat. Dis. Int. 2017, 16, 570–594. [Google Scholar] [CrossRef]
- Metzendorf, C.; Wineberger, K.; Rausch, J.; Cigliano, A.; Peters, K.; Sun, B.; Mennerich, D.; Kietzmann, T.; Calvisi, D.; Dombrowski, F.; et al. Transcriptomic and Proteomic Analysis of Clear Cell Foci (CCF) in the Human Non-Cirrhotic Liver Identifies Several Differentially Expressed Genes and Proteins with Functions in Cancer Cell Biology and Glycogen Metabolism. Molecules 2020, 25, 4141. [Google Scholar] [CrossRef]
- Cano, L.; Cerapio, J.P.; Ruiz, E.; Marchio, A.; Turlin, B.; Casavilca, S.; Taxa, L.; Marti, G.; Deharo, E.; Pineau, P.; et al. Liver clear cell foci and viral infection are associated with non-cirrhotic, non-fibrolamellar hepatocellular carcinoma in young patients from South America. Sci. Rep. 2018, 8, 9945. [Google Scholar] [CrossRef]
- Kutami, R.; Nakashima, Y.; Nakashima, O.; Shiota, K.; Kojiro, M. Pathomorphologic study on the mechanism of fatty change in small hepatocellular carcinoma of humans. J. Hepatol. 2000, 33, 282–289. [Google Scholar] [CrossRef]
- International Consensus Group for Hepatocellular NeoplasiaThe International Consensus Group for Hepatocellular N. Path-ologic diagnosis of early hepatocellular carcinoma: A report of the international consensus group for hepatocellular neoplasia. Hepatology 2009, 49, 658–664. [Google Scholar] [CrossRef]
- Straub, B.K.; Herpel, E.; Singer, S.; Zimbelmann, R.; Breuhahn, K.; Macher-Goeppinger, S.; Warth, A.; Lehmann-Koch, J.; Longerich, T.; Heid, H.; et al. Lipid droplet-associated PAT-proteins show frequent and differential expression in neoplastic steatogenesis. Mod. Pathol. 2010, 23, 480–492. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Mao, N.Y.; Zhang, Y.; Xu, W.W. Consideration on the emerging acute severe hepatitis cases with unknown etiology in children of some countries. Zhonghua Yu Fang Yi Xue Za Zhi 2022, 56, 679–681. [Google Scholar] [PubMed]
- Kelgeri, C.; Couper, M.; Gupte, G.L.; Brant, A.; Patel, M.; Johansen, L.; Valamparampil, J.; Ong, E.; Hartog, H.; Perera, M.; et al. Clinical Spectrum of Children with Acute Hepatitis of Unknown Cause. N. Engl. J. Med. 2022, 387, 611–619. [Google Scholar] [CrossRef] [PubMed]
- Ho, A.; Orton, R.; Tayler, R.; Asamaphan, P.; Tong, L.; Smollett, K.; Davis, C.; Manali, M.; McDonald, S.E.; Pollock, L.; et al. Adeno-associated virus 2 infection in children with non-A-E hepatitis. medRxiv 2022, 27, 7425. [Google Scholar]
- Agnello, V.; Ábel, G.; Knight, G.B.; Muchmore, E. Detection of widespread hepatocyte infection in chronic hepatitis C. Hepatology 1998, 28, 573–584. [Google Scholar] [CrossRef]
- Remmele, W.; Stegner, H.E. Recommendation for uniform definition of an immunoreactive score (IRS) for immunohistochemical estrogen receptor detection (ER-ICA) in breast cancer tissue. Pathologe 1987, 8, 138–140. [Google Scholar]
- Kleiner, D.E.; Brunt, E.M.; Van Natta, M.; Behling, C.; Contos, M.J.; Cummings, O.W.; Ferrell, L.D.; Liu, Y.-C.; Torbenson, M.S.; Unalp-Arida, A.; et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 2005, 41, 1313–1321. [Google Scholar] [CrossRef]
- Desmet, V.J.; Gerber, M.; Hoofnagle, J.H.; Manns, M.; Scheuer, P.J. Classification of chronic hepatitis: Diagnosis, grading and staging. Hepatology 1994, 19, 1513–1520. [Google Scholar] [CrossRef]



| Parameter | HCV Genotypes | |||||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 6 | Not Tested/Unknown | |
| PLIN2 score [0–3] | - | |||||
| mean (SD) | 1.85 (0.79) | 1.75 (0.73) | 2.27 (0.79) | 1.79 (0.49) | 1.92 (0.74) | |
| N | 114 | 16 | 34 | 7 | 56 | |
| PLIN2 foci share [%] | 35.09 | 50 | 23.53 | 57.14 | - | 39.29 |
| N | 114 | 16 | 34 | 7 | 56 | |
| PLIN1 score [0–3] | - | |||||
| mean (SD) | 1.15 (0.5) | 1.3 (0.56) | 1.66 (0.71) | 0.71(0.27) | 1.26 (0.53) | |
| N | 114 | 16 | 34 | 7 | 56 | |
| PLIN1 foci share [%] | 29.82 | 18.75 | 11.76 | 0 | - | 16.07 |
| N | 114 | 16 | 34 | 7 | 56 | |
| Foci share of all hepatocytes [%] | - | |||||
| mean (SD) | 13 (15) | 8 (4) | 45 (38) | 10 (6) | 23 (26) | |
| N | 51 | 10 | 16 | 4 | 33 | |
| Steatosis in PLIN2 [% of parenchyme area] | ||||||
| mean | 15.72 | 16.15 | 35.71 | 14.36 | 1 | 23.24 |
| (SD) | (20.94) | (24.07) | (32.63) | (15.59) | - | (24.47) |
| N | 115 | 17 | 34 | 7 | 1 | 55 |
| Steatosis in H&E [% of parenchyme area] | ||||||
| mean | 12.54 | 17.09 | 34.03 | 6.93 | 1 | 18.15 |
| (SD) | (18.46) | (25.38) | (32.69) | (6.72) | - | (22.95) |
| N | 116 | 17 | 34 | 7 | 1 | 55 |
| Grade of Steatosis in % (as Evaluated in H&E) | Number of Cases | Percentage of PLIN2-Positive Foci [%] | Percentage of PLIN1-Positive Foci [%] |
|---|---|---|---|
| <5 | 89 | 41.57 | 25.84 |
| 5–33 | 94 | 42.55 | 27.66 |
| 33–66 | 27 | 14.81 | 3.70 |
| >66 | 16 | 0 | 0 |
| Antibody (Clone) | Immunoglobulin Class | Provider |
|---|---|---|
| Akt (C67E7) | Monoclonal rabbit | Cell Signaling Technology, Danvers, MA, USA |
| Beta-Catenin (β-Catenin-1) | Monoclonal mouse | Agilent, Santa Clara, CA, USA |
| CK8/18 (GP11) | Polyclonal guinea pig | PROGEN, Heidelberg, Germany |
| E-cadherin (36/E-Cadherin) | Monoclonal mouse | BD Biosciences, San Jose, CA, USA |
| Desmin (D9) | Monoclonal mouse | PROGEN |
| Desmin | Polyclonal rabbit | PROGEN |
| L-FABP1 | Polyclonal rabbit | Sigma-Aldrich, St Louis, MO, USA |
| Glutaminsynthetase (GS-6) | Monoclonal mouse | Cell Marque/Sigma Aldrich |
| Glypican 3 (GC33) | Monoclonal mouse | Agilent |
| HCV Core antigen (C7-50, sc-57800) | Monoclonal mouse | Santa Cruz Biotechnology, Dallas, TX, USA |
| Heat shock protein 70 (W27) | Monoclonal mouse | Santa Cruz Biotechnology |
| Hepatitis B surface antigen (AI0FI) | Monoclonal mouse | DCS Innovative Diagnostics, Hamburg, Germany |
| Ki-67 (MIB-1) | Monoclonal mouse | Agilent |
| N-cadherin (36/N-Cadherin) | Monoclonal mouse | BD Biosciences |
| p53 (DO-7) | Monoclonal mouse | Agilent |
| Perilipin 1 (PERI 112.17) | Monoclonal mouse | PROGEN |
| Perilipin 1 (GP29) | Polyclonal guinea pig | PROGEN |
| Perilipin 1A | Polyclonal rabbit | Sigma Aldrich |
| Perilipin 2 (AP 125) | Monoclonal mouse | PROGEN |
| Perilipin 2 (GP40, GP41) | Polyclonal guinea pig | PROGEN |
| Perilipin 3 (GP30, GP32) | Polyclonal guinea pig | PROGEN |
| Perilipin 4 (GP38) | Polyclonal guinea pig | PROGEN |
| Perilipin 5 (GP31) | Polyclonal guinea pig | PROGEN |
| Phospho-mTor (49F9) | Monoclonal rabbit | Cell Signaling Technology |
| Vimentin (3B4, V9) | Monoclonal mouse | PROGEN |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schelbert, S.; Schindeldecker, M.; Drebber, U.; Witzel, H.R.; Weinmann, A.; Dries, V.; Schirmacher, P.; Roth, W.; Straub, B.K. Lipid Droplet-Associated Proteins Perilipin 1 and 2: Molecular Markers of Steatosis and Microvesicular Steatotic Foci in Chronic Hepatitis C. Int. J. Mol. Sci. 2022, 23, 15456. https://doi.org/10.3390/ijms232415456
Schelbert S, Schindeldecker M, Drebber U, Witzel HR, Weinmann A, Dries V, Schirmacher P, Roth W, Straub BK. Lipid Droplet-Associated Proteins Perilipin 1 and 2: Molecular Markers of Steatosis and Microvesicular Steatotic Foci in Chronic Hepatitis C. International Journal of Molecular Sciences. 2022; 23(24):15456. https://doi.org/10.3390/ijms232415456
Chicago/Turabian StyleSchelbert, Selina, Mario Schindeldecker, Uta Drebber, Hagen Roland Witzel, Arndt Weinmann, Volker Dries, Peter Schirmacher, Wilfried Roth, and Beate Katharina Straub. 2022. "Lipid Droplet-Associated Proteins Perilipin 1 and 2: Molecular Markers of Steatosis and Microvesicular Steatotic Foci in Chronic Hepatitis C" International Journal of Molecular Sciences 23, no. 24: 15456. https://doi.org/10.3390/ijms232415456
APA StyleSchelbert, S., Schindeldecker, M., Drebber, U., Witzel, H. R., Weinmann, A., Dries, V., Schirmacher, P., Roth, W., & Straub, B. K. (2022). Lipid Droplet-Associated Proteins Perilipin 1 and 2: Molecular Markers of Steatosis and Microvesicular Steatotic Foci in Chronic Hepatitis C. International Journal of Molecular Sciences, 23(24), 15456. https://doi.org/10.3390/ijms232415456








