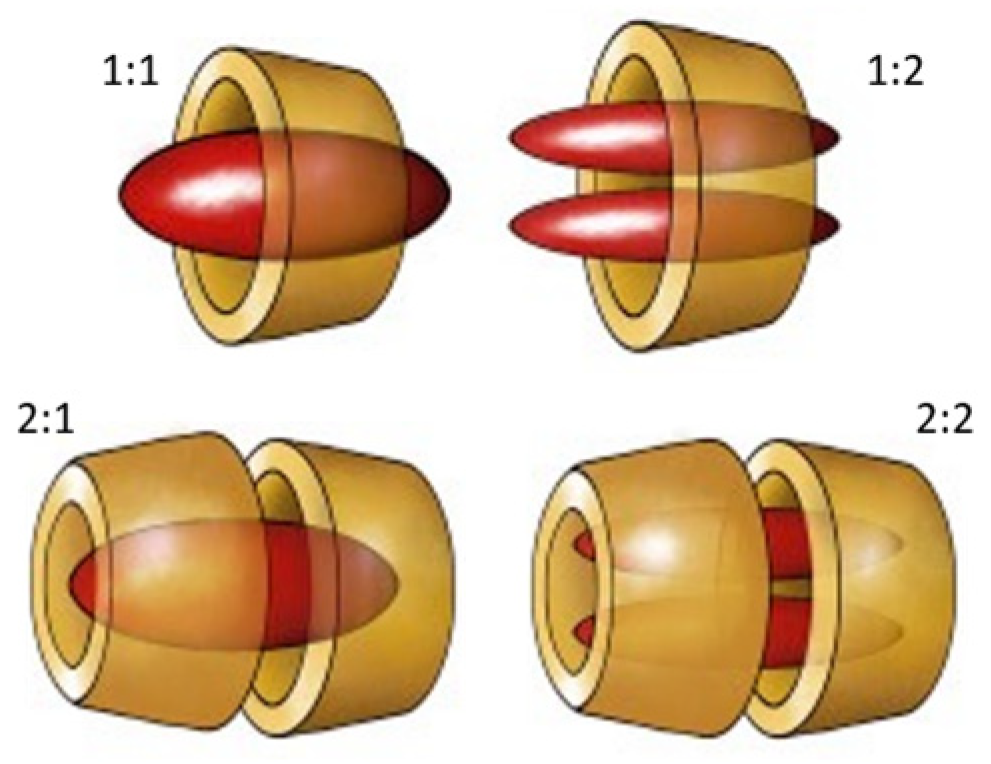
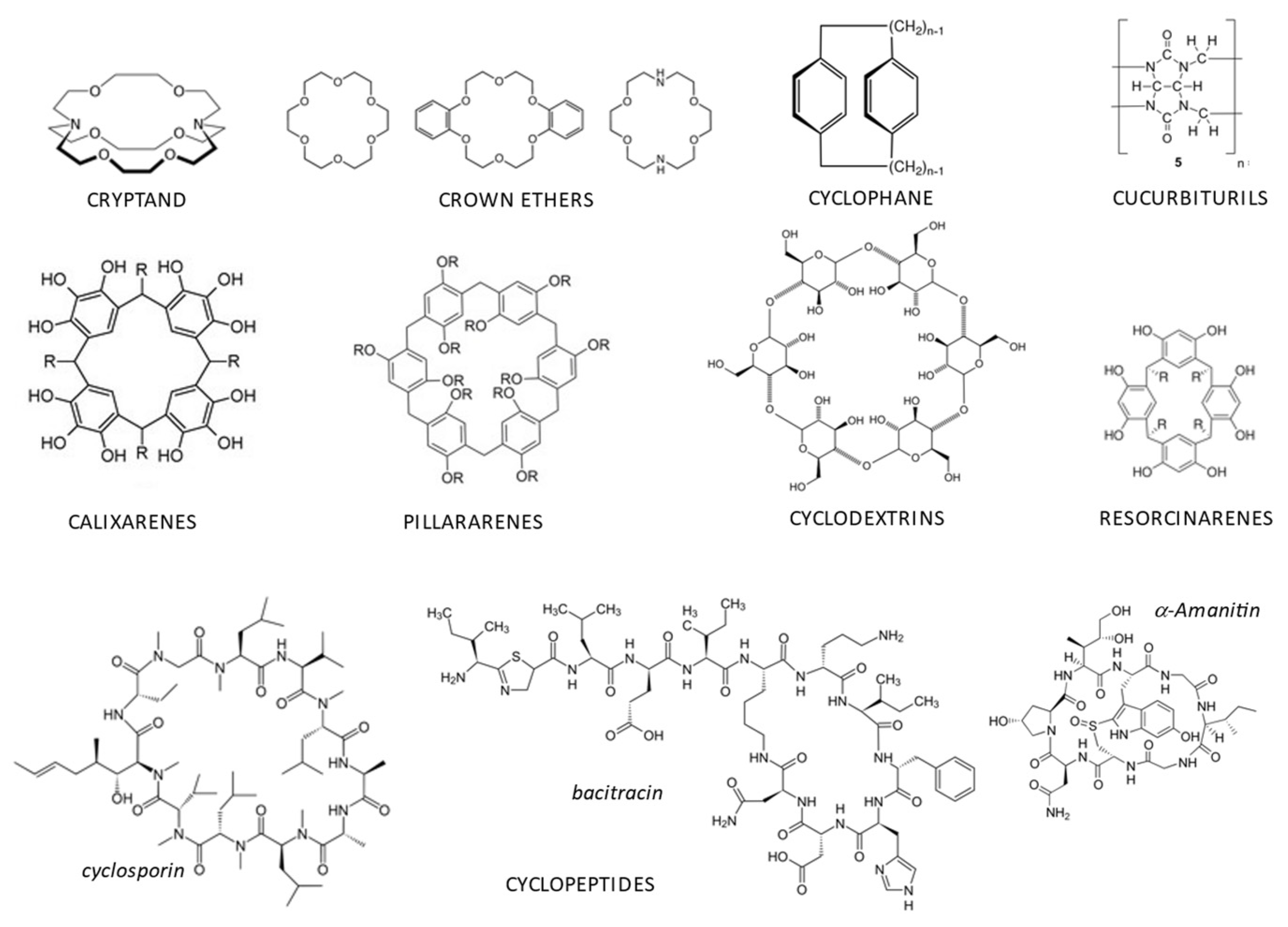
Host–Guest Complexes
Conflicts of Interest
References
- Steed, J.W.; Atwood, J.L. Supramolecular Chemistry, 2nd ed.; Wiley: Hoboken, NJ, USA, 2013. [Google Scholar]
- Lehn, J.M. Supramolecular Shemistry. Science 1993, 260, 1762–1763. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Yan, X.; Huang, F.; Gibson, H.W. Stimuli-Responsive Host–Guest Systems Based on the Recognition of Cryptands by Organic Guests. Acc. Chem. Res. 2014, 47, 1995–2005. [Google Scholar] [CrossRef] [PubMed]
- Kralij, M.; Tusek-Bozic, L.; Frkanec, L. Biomedical potentials of crown ethers: Prospective antitumor agents. ChemMedChem 2008, 3, 1478–1492. [Google Scholar] [CrossRef]
- Goek, G.W.; Leevy, W.M.; Weber, M.E. Crown Ethers: Sensors for Ions and Molecular Scaffolds for Materials and Biological Models. Chem. Rev. 2004, 104, 2723–2750. [Google Scholar]
- Hof, F. Host–guest chemistry that directly targets lysine methylation: Synthetic host molecules as alternatives to bio-reagents. Chem. Commun. 2016, 52, 10093–10108. [Google Scholar] [CrossRef] [PubMed]
- Ramaiah, D.; Neelakandan, P.P.; Naira, A.K.; Aviraha, R.R. Functional cyclophanes: Promising hosts for optical biomolecular recognition. Chem. Soc. Rev. 2010, 39, 4158–4168. [Google Scholar] [CrossRef]
- Tan, N.-H.; Zhou, J. Plant Cyclopeptides. Chem. Rev. 2006, 106, 840–895. [Google Scholar] [CrossRef]
- Galan, M.C.; Dumy, P.; Renaudet, O. Multivalent glyco(cyclo)peptides. Chem. Soc. Rev. 2013, 42, 4599–4612. [Google Scholar] [CrossRef]
- Crini, G. Review: A History of Cyclodextrins. Chem. Rev. 2014, 114, 10940–10975. [Google Scholar] [CrossRef]
- Mellet, C.O.; Fernandez, J.M.G.; Benito, J.M. Cyclodextrin-based gene delivery systems. Chem. Soc. Rev. 2011, 40, 1586–1608. [Google Scholar] [CrossRef]
- García-Río, L.; Hevés, P.; Leis, J.R.; Mejuto, J.C.; Perez-Juste, J.; Rodríguez-Dafonte, P. Evidence for complexes of different stoichiometries between organic solvents and cyclodextrins. Org. Biomol. Chem. 2006, 4, 1038–1048. [Google Scholar] [CrossRef] [PubMed]
- Timmerman, P.; Verboom, W.; Reinhoudt, D.N. Resorcinarenes. Tetrahedron 1996, 52, 2663–2704. [Google Scholar] [CrossRef]
- Lagona, J.; Mukhopadhyay, P.; Chakrabarti, S.; Isaacs, L. The Cucurbit[n]uril Family. Angew. Chem. Int. Ed. 2005, 44, 4844–4870. [Google Scholar] [CrossRef] [PubMed]
- Barrow, S.J.; Kasera, S.; Rowland, M.J.; Barrio, J.; Scherman, O.A. Cucurbituril-Based Molecular Recognition. Chem. Rev. 2015, 115, 12320–12406. [Google Scholar] [CrossRef]
- Diamond, D.; Mckervey, M.A. Calixarene-based sensing agents. Chem. Soc. Rev. 1996, 25, 15–24. [Google Scholar] [CrossRef]
- Ikeda, A.; Shinkai, S. Novel Cavity Design Using Calix[n]arene Skeletons: Toward Molecular Recognition and Metal Binding. Chem. Rev. 1997, 97, 1713–1734. [Google Scholar] [CrossRef]
- Böhmer, V. Calixarenes, Macrocycles with (Almost) Unlimited Possibilities. Angew. Chem. Int. Ed. Engl. 1995, 34, 713–745. [Google Scholar] [CrossRef]
- Nimse, S.B.; Kim, T. Biological applications of functionalized calixarenes. Chem. Soc. Rev. 2013, 42, 366–386. [Google Scholar] [CrossRef]
- Sathiyajith, C.W.; Shaikh, R.R.; Han, Q.; Zhang, Y.; Meguellati, K.; Yang, Y.-W. Biological and related applications of pillar[n]arenes. Chem. Commun. 2017, 53, 677–696. [Google Scholar] [CrossRef]
- Gómez-González, B.; García-Río, L.; Basílio, N.; Mejuto, J.C.; Simal-Gándara, J. Molecular Molecular Recognition by Pillar [5]arenes: Evidence for Simultaneous Electrostatic and Hydrophobic Interactions. Pharmaceutics 2022, 14, 60. [Google Scholar] [CrossRef]
- Chen, K.-J.; Garcia, M.A.; Wang, H.; Tseng, H.-R. Supramolecular Nanoparticles for Molecular Diagnostics and Therapeutics; Supramolecular Chemistry; Wiley: Hoboken, NJ, USA, 2012. [Google Scholar]
- Ma, X.; Zhao, Y. Biomedical applications of supramolecular systems based on host-guest interactions. Chem. Rev. 2015, 115, 7794–7839. [Google Scholar] [CrossRef] [PubMed]
- Cid-Samamed, A.; Rakmai, J.; Mejuto, J.C.; Simal-Gandara, J.; Astray, G. Cyclodextrins inclusion complex: Preparation methos, analytical techniques and food industry applications. Food Chem. 2022, 384, 132467. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Pereira, A.; Carpena, M.; García-Oliveira, P.; Mejuto, J.C.; Prieto, M.A.; Simal-Gandara, J. Main Applications of Cyclodextrins in the Food Industry as the Compounds of Choice to Form Host–Guest Complexes. Int. J. Mol. Sci. 2021, 22, 1339. [Google Scholar] [CrossRef] [PubMed]
- Astray, G.; Mejuto, J.C.; Simal-Gandara, J. Latest developments in the application of cyclodextrins host-guest complexes in beverage technology processes. Food Hydrocoll. 2020, 106, 105882. [Google Scholar] [CrossRef]
- Galmés, B.; Franconetti, A.; Frontera, A. Nitropyridine-1-Oxides as Excellent π-Hole Donors: Interplay between σ-Hole (Halogen, Hydrogen, Triel, and Coordination Bonds) and π-Hole Interactions. Int. J. Mol. Sci. 2019, 20, 3440. [Google Scholar] [CrossRef]
- Bai, H.; Wang, J.; Li, Z.; Tang, G. Macrocyclic Compounds for Drug and Gene Delivery in Immune-Modulating Therapy. Int. J. Mol. Sci. 2019, 20, 2097. [Google Scholar] [CrossRef] [PubMed]
- Raffaini, G.; Ganazzoli, F. A Molecular Dynamics Study of a Photodynamic Sensitizer for Cancer Cells: Inclusion Complexes of γ-Cyclodextrins with C70. Int. J. Mol. Sci. 2019, 20, 4831. [Google Scholar] [CrossRef]
- Czapik, A.; Jelecki, M.; Kwit, M. Chiral Cocrystal Solid Solutions, Molecular Complexes, and Salts of N-Triphenylacetyl-l-Tyrosine and Diamines. Int. J. Mol. Sci. 2019, 20, 5004. [Google Scholar] [CrossRef]
- Couto, V.M.; de Oliveira-Nascimento, L.; Cabeça, L.F.; Geraldes, D.C.; Costa, J.S.R.; Riske, K.A.; Franz-Montan, M.; Yokaychiya, F.; Franco, M.K.K.D.; de Paula, E. Capsaicin-Cyclodextrin Complex Enhances Mepivacaine Targeting and Improves Local Anesthesia in Inflamed Tissues. Int. J. Mol. Sci. 2020, 21, 5741. [Google Scholar] [CrossRef]
- de Almeida Magalhães, T.S.S.; de Oliveira Macedo, P.C.; Kawashima Pacheco, S.Y.; Silva, S.S.D.; Barbosa, E.G.; Pereira, R.R.; Costa, R.M.R.; Silva Junior, J.O.C.; da Silva Ferreira, M.A.; de Almeida, J.C.; et al. Development and Evaluation of Antimicrobial and Modulatory Activity of Inclusion Complex of Euterpe oleracea Mart Oil and β-Cyclodextrin or HP-β-Cyclodextrin. Int. J. Mol. Sci. 2020, 21, 942. [Google Scholar] [CrossRef]
- García-Pérez, P.; Losada-Barreiro, S.; Gallego, P.P.; Bravo-Díaz, C. Cyclodextrin-Elicited Bryophyllum Suspension Cultured Cells: Enhancement of the Production of Bioactive Compounds. Int. J. Mol. Sci. 2019, 20, 5180. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mejuto, J.C.; Simal-Gandara, J. Host–Guest Complexes. Int. J. Mol. Sci. 2022, 23, 15730. https://doi.org/10.3390/ijms232415730
Mejuto JC, Simal-Gandara J. Host–Guest Complexes. International Journal of Molecular Sciences. 2022; 23(24):15730. https://doi.org/10.3390/ijms232415730
Chicago/Turabian StyleMejuto, Juan C., and Jesus Simal-Gandara. 2022. "Host–Guest Complexes" International Journal of Molecular Sciences 23, no. 24: 15730. https://doi.org/10.3390/ijms232415730
APA StyleMejuto, J. C., & Simal-Gandara, J. (2022). Host–Guest Complexes. International Journal of Molecular Sciences, 23(24), 15730. https://doi.org/10.3390/ijms232415730





