Proliferation and Apoptosis Pathways and Factors in Oral Squamous Cell Carcinoma
Abstract
:1. Introduction
2. Molecular Hallmarks of Cancer
2.1. Enabling Replicative Immortality in Oral Cancer
2.2. Inducing Angiogenesis in Oral Cancer
2.3. Activating Invasion and Metastasis in Oral Cancer
3. Oral Cancer and Cellular Proliferation
3.1. Mitogenic Activation and Induction of Proliferation Signaling in OSCC
3.2. Ras-Raf-MEK-ERK/MAPK Pathway
3.3. PI3K-AKT-mTOR Pathway
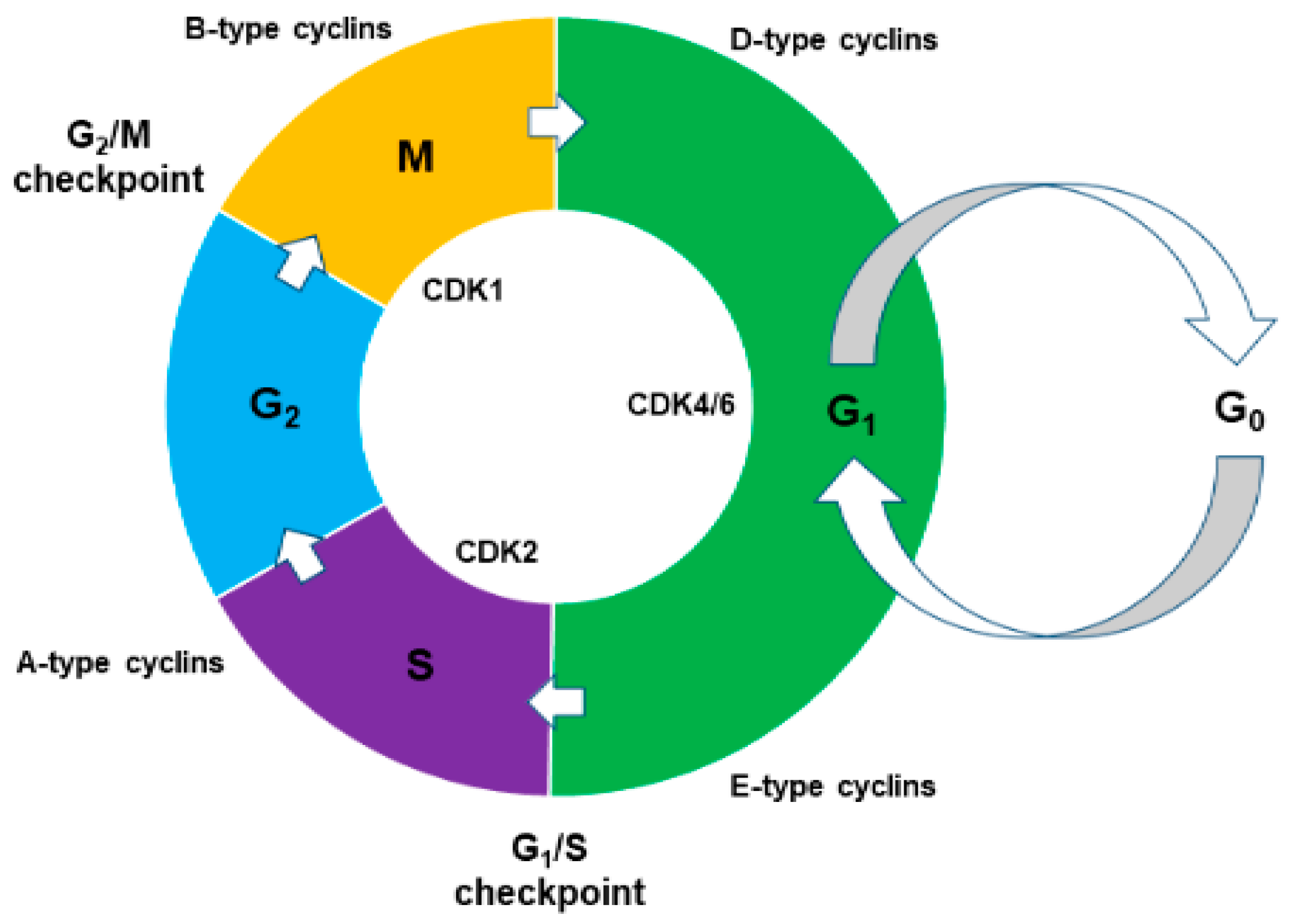
3.4. The Cell Cycle and Tumour Suppressor Genes
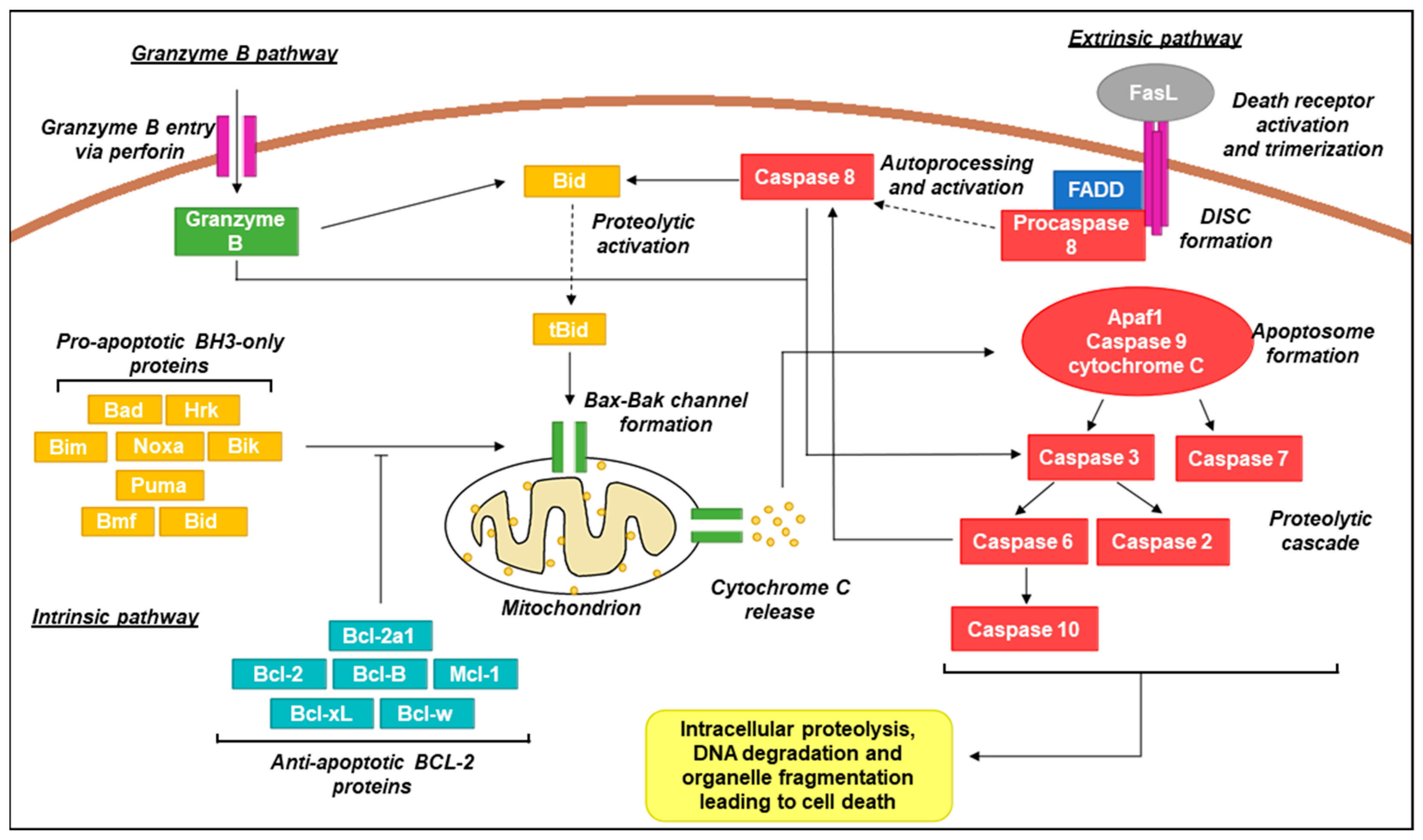
4. Oral Cancer and Apoptosis
| Protein | Role in Oral Cancer | Reference |
|---|---|---|
| Granzyme B | Cytotoxic T lymphocyte mediated tumour cell apoptosis | Zhu et al. [90] |
| Perforin | Takes part in NK cell mediated oral cancer cell destruction | Hadler-Olsen et al. [91] |
| FasL | Mediator of immune privilege in OSCC | Fang et al. [92] |
| Fas (CD95) | Considered a prognostic marker of OSCC | Peter et al. [93] |
| TNF-α | Promotes oral cancer growth and pain | Salvo et al. [94] |
| TNFR1 (CD120a) | Related to oral cancer pain and inflammation | Scheff et al. [95] |
| FADD | Prognostic implication in OSCC | Gonzales-Moles et al. [96] |
| Apaf1 | Helps in the formation of apoptosome | Dwivedi et al. [97] |
| Bcl-2 | Altered expression results in an increase in malignant transformation potential | Juneja et al. [98] |
| Bcl-2a1 (Bfl-1) and Cytochrome C | Promotes apoptosis in OSCC | Zheng et al. [99] |
| Mcl-1 | Overexpression of anti-apoptotic factor Mcl-1 leads to progression of OSCC | Sulkshane et al. [100] |
| Bcl-xL | Causes resistance to chemotherapeutic drugs | Alam et al. [101] |
| Bcl-w | Cooperates with oncogene activation in the development and progression of OSCC | Hartman et al. [102] |
| BH3 | Anti-anti-apoptotic functions in OSCC | Carter et al. [103] |
4.1. Extrinsic Apoptotic Signalling Receptors and Ligands
4.2. Bcl-2 Family Proteins
4.3. Apoptotic Caspase Proteins
5. Interplay between Apoptotic and Proliferative Signalling Networks
6. Method of Data Collection
7. Conclusions and Challenges for Future Research
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ren, Z.H.; Hu, C.Y.; He, H.R.; Li, Y.J.; Lyu, J. Global and regional burdens of oral cancer from 1990 to 2017: Results from the global burden of disease study. Cancer Commun. 2020, 40, 81–92. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Montero, P.H.; Patel, S.G. Cancer of the Oral Cavity. Surg. Oncol. Clin. N. Am. 2015, 24, 491–508. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Makarev, E.; Schubert, A.D.; Kanherkar, R.R.; London, N.; Teka, M.; Ozerov, I.; Lezhnina, K.; Bedi, A.; Ravi, R.; Mehra, R.; et al. In silico analysis of pathways activation landscape in oral squamous cell carcinoma and oral leukoplakia. Cell Death Discov. 2017, 3, 17022. [Google Scholar] [CrossRef] [PubMed]
- De Felice, F.; de Vincentiis, M.; Valentini, V.; Musio, D.; Mezi, S.; Lo Mele, L.; Terenzi, V.; D’Aguanno, V.; Cassoni, A.; Di Brino, M.; et al. Follow-up program in head and neck cancer. Crit. Rev. Oncol. Hematol. 2017, 113, 151–155. [Google Scholar] [CrossRef]
- Machiels, J.P.; René Leemans, C.; Golusinski, W.; Grau, C.; Licitra, L.; Gregoire, V. Squamous cell carcinoma of the oral cavity, larynx, oropharynx and hypopharynx: EHNS–ESMO–ESTRO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2020, 31, 1462–1475. [Google Scholar] [CrossRef]
- Cheraghlou, S.; Schettino, A.; Zogg, C.K.; Judson, B.L. Changing prognosis of oral cancer: An analysis of survival and treatment between 1973 and 2014. Laryngoscope 2018, 128, 2762–2769. [Google Scholar] [CrossRef]
- Akinkugbe, A.A.; Garcia, D.T.; Brickhouse, T.H.; Mosavel, M. Lifestyle risk factor related disparities in oral cancer examination in the U.S: A population-based cross-sectional study. BMC Public Health 2020, 20, 153. [Google Scholar] [CrossRef]
- Hung, L.C.; Kung, P.T.; Lung, C.H.; Tsai, M.H.; Liu, S.A.; Chiu, L.T.; Huang, K.H.; Tsai, W.C. Assessment of the risk of oral cancer incidence in a high-risk population and establishment of a predictive model for oral cancer incidence using a population-based cohort in Taiwan. Int. J. Environ. Res. Public Health 2020, 17, 665. [Google Scholar] [CrossRef] [Green Version]
- International Agency for Research on Cancer. IARC monographs on the evaluation of carcinogenic risks to humans, volume 100E. In Personal Habits and Indoor Combustions; IARC: Lyon, France, 2012; pp. 43–213. [Google Scholar]
- Roi, A.; Roi, C.I.; Andreescu, N.I.; Riviş, M.; Badea, I.D.; Meszaros, N.; Rusu, L.C.; Iurciuc, S. Oral cancer histopathological subtypes in association with risk factors: A 5-year retrospective study. Rom. J. Morphol. Embryol. 2021, 61, 1213–1220. [Google Scholar] [CrossRef] [PubMed]
- Athukorala, I.A.; Tilakaratne, W.M.; Jayasinghe, R.D. Areca Nut Chewing: Initiation, Addiction, and Harmful Effects Emphasizing the Barriers and Importance of Cessation. J. Addict. 2021, 2021, 9967097. [Google Scholar] [CrossRef] [PubMed]
- Thompson, I.O.C.; Van der Bijl, P.; Van Wyk, C.W.; Van Eyk, A.D. A comparative light-microscopic, electron-microscopic and chemical study of human vaginal and buccal epithelium. Arch. Oral Biol. 2001, 46, 1091–1098. [Google Scholar] [CrossRef]
- Campisi, G.; Panzarella, V.; Giuliani, M.; Lajolo, C.; Di Fede, O.; Falaschini, S.; Di Liberto, C.; Scully, C.; Lo Muzio, L. Human papillomavirus: Its identikit and controversial role in oral oncogenesis, premalignant and malignant lesions (Review). Int. J. Oncol. 2007, 30, 813–823. [Google Scholar] [CrossRef] [Green Version]
- Park, N.-H.; Min, B.-M.; Li, S.; Huang, M.Z.; Doniger, J. Immortalization of normal human oral keratinocytes with type 16 human papillomavirus. Carcinogenesis 1991, 12, 1627–1631. [Google Scholar] [CrossRef]
- Pytynia, K.B.; Dahlstrom, K.R.; Sturgis, E.M. Epidemiology of HPV-associated oropharyngeal cancer. Oral Oncol. 2014, 50, 380–386. [Google Scholar] [CrossRef] [Green Version]
- Farah, C.S. Molecular landscape of head and neck cancer and implications for therapy. Ann. Transl. Med. 2021, 9, 915. [Google Scholar] [CrossRef]
- Krüger, M.; Pabst, A.M.; Walter, C.; Sagheb, K.; Günther, C.; Blatt, S.; Weise, K.; Al-Nawas, B.; Ziebart, T. The prevalence of human papilloma virus (HPV) infections in oral squamous cell carcinomas: A retrospective analysis of 88 patients and literature overview. J. Cranio-Maxillofac. Surg. 2014, 42, 1506–1514. [Google Scholar] [CrossRef]
- Wu, W.; Wang, Z.; Zhou, Z. Role of the human papillomavirus in malignant transformation of oral leukoplakia distinct from oropharyngeal squamous cell carcinoma: A study of 76 patients with internal-control specimens. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2019, 128, 273–279. [Google Scholar] [CrossRef]
- Pindborg, J.J.; Daftary, D.K.; Mehta, F.S. A follow-up study of sixty-one oral dysplastic precancerous lesions in Indian villagers. Oral Surg. Oral Med. Oral Pathol. 1977, 43, 383–390. [Google Scholar] [CrossRef]
- Reibel, J. Prognosis of oral pre-malignant lesions: Significance of clinical, histopathological, and molecular biological characteristics. Crit. Rev. Oral Biol. Med. 2001, 6, 47–62. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, A.K.; Udaltsova, N.; Engels, E.A.; Katzel, J.A.; Yanik, E.L.; Katki, H.A.; Lingen, M.W.; Silverberg, M.J. Oral Leukoplakia and Risk of Progression to Oral Cancer: A Population-Based Cohort Study. JNCI J. Natl. Cancer Inst. 2020, 112, 1047–1054. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van der Waal, I. Oral leukoplakia, the ongoing discussion on definition and terminology. Med. Oral Patol. Oral Cir. Bucal 2015, 20, e685–e692. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raghunandan, B.; Sanjai, K.; Kumaraswamy, J.; Papaiah, L.; Pandey, B.; Jyothi, B. Expression of human telomerase reverse transcriptase protein in oral epithelial dysplasia and oral squamous cell carcinoma: An immunohistochemical study. J. Oral Maxillofac. Pathol. 2016, 20, 96–101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Essa, A.A.M.; Deraz, E.M. Expression of CD44 (NKI-P1) in oral squamous cell carcinoma associated vascular endothelial cells: A relationship to tumor angiogenesis. Saudi Dent. J. 2021, 34, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Berx, G.; van Roy, F. Involvement of members of the cadherin superfamily in cancer. Cold Spring Harb. Perspect. Biol. 2009, 1, a003129. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.L.; Xie, Y.G.; Li, Z.; Ma, J.H.; Xu, X. E-cadherin expression and prognosis of oral cancer: A meta-analysis. Tumor Biol. 2014, 35, 5533–5537. [Google Scholar] [CrossRef]
- Micalizzi, D.S.; Farabaugh, S.M.; Ford, H.L. Epithelial-mesenchymal transition in cancer: Parallels between normal development and tumor progression. J. Mammary Gland Biol. Neoplasia 2010, 15, 117–134. [Google Scholar] [CrossRef] [Green Version]
- Potten, C.S.; Loeffler, M. Stem cells: Attributes, cycles, spirals, pitfalls and uncertainties. Lessons for and from the crypt. Development 1990, 110, 1001–1020. [Google Scholar] [CrossRef]
- Wong, R.W.C.; Guillaud, L. The role of epidermal growth factor and its receptors in mammalian CNS. Cytokine Growth Factor Rev. 2004, 15, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Pierre, S.; Bats, A.S.; Coumoul, X. Understanding SOS (Son of Sevenless). Biochem. Pharmacol. 2011, 82, 1049–1056. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Terrell, E.M.; Morrison, D.K. Ras-mediated activation of the Raf family kinases. Cold Spring Harb. Perspect. Med. 2019, 9, a033746. [Google Scholar] [CrossRef] [PubMed]
- Leicht, D.T.; Balan, V.; Kaplun, A.; Singh-Gupta, V.; Kaplun, L.; Dobson, M.; Tzivion, G. Raf kinases: Function, regulation and role in human cancer. Biochim. Biophys. Acta Mol. Cell Res. 2007, 1773, 1196–1212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wee, P.; Wang, Z. Epidermal growth factor receptor cell proliferation signaling pathways. Cancers 2017, 9, 52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, S.; Jee, K.; Kim, D.; Koh, H.; Chung, J. Cyclic AMP Inhibits Akt Activity by Blocking the Membrane Localization of PDK1. J. Biol. Chem. 2001, 276, 12864–12870. [Google Scholar] [CrossRef] [Green Version]
- Manning, B.D.; Cantley, L.C. AKT/PKB Signaling: Navigating Downstream. Cell 2007, 129, 1261–1274. [Google Scholar] [CrossRef] [Green Version]
- Morita, M.; Gravel, S.P.; Chénard, V.; Sikström, K.; Zheng, L.; Alain, T.; Gandin, V.; Avizonis, D.; Arguello, M.; Zakaria, C.; et al. MTORC1 controls mitochondrial activity and biogenesis through 4E-BP-dependent translational regulation. Cell Metab. 2013, 18, 698–711. [Google Scholar] [CrossRef] [Green Version]
- Bartek, J.; Lukas, J. Pathways governing G1/S transition and their response to DNA damage. FEBS Lett. 2001, 490, 117–122. [Google Scholar] [CrossRef] [Green Version]
- Burkhart, D.L.; Sage, J. Cellular mechanisms of tumour suppression by the retinoblastoma gene. Nat. Rev. Cancer 2008, 8, 671–682. [Google Scholar] [CrossRef]
- Malumbres, M.; Barbacid, M. Cell cycle, CDKs and cancer: A changing paradigm. Nat. Rev. Cancer 2009, 9, 153–166. [Google Scholar] [CrossRef] [PubMed]
- Bernardes, V.F.; Gleber-Netto, F.O.; Sousa, S.F.; Silva, T.A.; Aguiar, M.C.F. Clinical significance of EGFR, Her-2 and EGF in oral squamous cell carcinoma: A case control study. J. Exp. Clin. Cancer Res. 2010, 29, 40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, L.Y.; Li, E.M.; Wu, Z.Y.; Cao, H.H.; Shen, J.H.; Xu, X.E.; Chen, B.; Wu, J.Y.; Xu, L.Y. Overexpression of GRB2 is correlated with lymph node metastasis and poor prognosis in esophageal squamous cell carcinoma. Int. J. Clin. Exp. Pathol. 2014, 7, 3132–3140. [Google Scholar] [PubMed]
- Li, X.; Yang, Y.; Hu, Y.; Dang, D.; Regezi, J.; Schmidt, B.L.; Atakilit, A.; Chen, B.; Ellis, D.; Ramos, D.M. αvβ6-Fyn Signaling Promotes Oral Cancer Progression. J. Biol. Chem. 2003, 278, 41646–41653. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baltanás, F.C.; Zarich, N.; Rojas-Cabañeros, J.M.; Santos, E. SOS GEFs in health and disease. Biochim. Biophys. Acta Rev. Cancer 2020, 1874, 188445. [Google Scholar] [CrossRef] [PubMed]
- Batta, N.; Pandey, M. Mutational spectrum of tobacco associated oral squamous carcinoma and its therapeutic significance. World J. Surg. Oncol. 2019, 17, 198. [Google Scholar] [CrossRef]
- Hallums, D.P.; Gomez, R.; Doyle, A.P.; Viet, C.T.; Schmidt, B.L.; Jeske, N.A. RAF Kinase Inhibitory Protein Expression and Phosphorylation Profiles in Oral Cancers. Clin. Surg. 2016, 1, 1100. [Google Scholar]
- Kashyap, T.; Pramanik, K.K.; Nath, N.; Mishra, P.; Singh, A.K.; Nagini, S.; Rana, A.; Mishra, R. Crosstalk between Raf-MEK-ERK and PI3K-Akt-GSK3β signaling networks promotes chemoresistance, invasion/migration and stemness via expression of CD44 variants (v4 and v6) in oral cancer. Oral Oncol. 2018, 86, 234–243. [Google Scholar] [CrossRef]
- Peng, Q.; Deng, Z.; Pan, H.; Gu, L.; Liu, O.; Tang, Z. Mitogen-activated protein kinase signaling pathway in oral cancer (Review). Oncol. Lett. 2018, 15, 1379–1388. [Google Scholar] [CrossRef] [Green Version]
- Harsha, C.; Banik, K.; Ang, H.L.; Girisa, S.; Vikkurthi, R.; Parama, D.; Rana, V.; Shabnam, B.; Khatoon, E.; Kumar, A.P.; et al. Targeting AKT/mTOR in oral cancer: Mechanisms and advances in clinical trials. Int. J. Mol. Sci. 2020, 21, 3285. [Google Scholar] [CrossRef]
- Sushma, P.S.; Jamil, K.; Kumar, P.U.; Satyanarayana, U.; Ramakrishna, M.; Triveni, B. PTEN and p16 genes as epigenetic biomarkers in oral squamous cell carcinoma (OSCC): A study on south Indian population. Tumor Biol. 2016, 37, 7625–7632. [Google Scholar] [CrossRef] [PubMed]
- Sambandam, V.; Frederick, M.J.; Shen, L.; Tong, P.; Rao, X.; Peng, S.; Singh, R.; Mazumdar, T.; Huang, C.; Li, Q.; et al. PDK1 Mediates NOTCH1-Mutated Head and Neck Squamous Carcinoma Vulnerability to Therapeutic PI3K/mTOR Inhibition. Clin. Cancer Res. 2019, 25, 3329. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.; Feng, X.; Molinolo, A.A.; Martin, D.; Vitale-Cross, L.; Nohata, N.; Ando, M.; Wahba, A.; Amornphimoltham, P.; Wu, X.; et al. 4E-BP1 Is a Tumor Suppressor Protein Reactivated by mTOR Inhibition in Head and Neck Cancer. Cancer Res. 2019, 79, 1438–1450. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramakrishna, A.; Shreedhar, B.; Narayan, T.; Mohanty, L.; Shenoy, S.; Jamadar, S. Cyclin D1 an early biomarker in oral carcinogenesis. J. Oral Maxillofac. Pathol. 2013, 17, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Kujan, O.; Huang, G.; Ravindran, A.; Vijayan, M.; Farah, C.S. CDK4, CDK6, cyclin D1 and Notch1 immunocytochemical expression of oral brush liquid-based cytology for the diagnosis of oral leukoplakia and oral cancer. J. Oral Pathol. Med. 2019, 48, 566–573. [Google Scholar] [CrossRef] [PubMed]
- Moharil, R.B.; Khandekar, S.; Dive, A.; Bodhade, A. Cyclin D1 in oral premalignant lesions and oral squamous cell carcinoma: An immunohistochemical study. J. Oral Maxillofac. Pathol. 2020, 24, 397. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, L.S.; Diniz-Freitas, M.; Warnakulasuriya, S.; Garcia-Caballero, T.; Forteza-Vila, J.; Fraga, M. Prognostic Significance of Cyclins A2, B1, D1, and E1 and CCND1 Numerical Aberrations in Oral Squamous Cell Carcinomas. Anal. Cell. Pathol. 2018, 2018, 7253510. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.; Zhang, F.-H.; Chen, Q.-E.; Wang, Y.-Y.; Wang, Y.-L.; He, J.-C.; Zhou, J. The clinical significance of CDK1 expression in oral squamous cell carcinoma. Med. Oral Patol. Oral Cir. Bucal 2015, 20, e7–e12. [Google Scholar] [CrossRef]
- Mihara, M.; Shintani, S.; Nakahara, Y.; Kiyota, A.; Ueyama, Y.; Matsumura, T.; Wong, D.T. Overexpression of CDK2 is a prognostic indicator of oral cancer progression. Jpn. J. Cancer Res. 2001, 92, 352–360. [Google Scholar] [CrossRef]
- Shin, M.-K.; Pitot, H.C.; Lambert, P.F. Pocket proteins suppress head and neck cancer. Cancer Res. 2012, 72, 1280–1289. [Google Scholar] [CrossRef] [Green Version]
- Parikh, R.A.; Appleman, L.J.; Bauman, J.E.; Sankunny, M.; Lewis, D.W.; Vlad, A.; Gollin, S.M. Upregulation of the ATR-CHEK1 pathway in oral squamous cell carcinomas. Genes Chromosomes Cancer 2014, 53, 25–37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ragos, V.; Mastronikolis, N.S.; Tsiambas, E.; Baliou, E.; Mastronikolis, S.N.; Tsoukalas, N.; Patsouri, E.E.; Fotiades, P.P. p53 mutations in oral cavity carcinoma. J. BUON 2018, 23, 1569–1572. [Google Scholar] [PubMed]
- Pérez-Sayáns, M.; Suárez-Peñaranda, J.-M.; Gayoso-Diz, P.; Barros-Angueira, F.; Gándara-Rey, J.-M.; García-García, A. The role of p21Waf1/CIP1 as a Cip/Kip type cell-cycle regulator in oral squamous cell carcinoma (Review). Med. Oral Patol. Oral Cir. Bucal 2013, 18, e219–e225. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Kamboj, M.; Shreedhar, B. Expression of p16 in oral leukoplakia and oral squamous cell carcinoma and correlation of its expression with individual atypical features. J. Oral Biol. Craniofacial Res. 2019, 9, 156–160. [Google Scholar] [CrossRef]
- Cheng, N.; Chytil, A.; Shyr, Y.; Joly, A.; Moses, H.L. TGF-β signaling deficient fibroblasts enhance Hepatocyte Growth Factor signaling in mammary carcinoma cells to promotes cattering and invasion. Mol. Cancer 2009, 6, 1521–1533. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lawrence, M.S.; Sougnez, C.; Lichtenstein, L.; Cibulskis, K.; Lander, E.; Gabriel, S.B.; Getz, G.; Ally, A.; Balasundaram, M.; Birol, I.; et al. Comprehensive genomic characterization of head and neck squamous cell carcinomas. Nature 2015, 517, 576–582. [Google Scholar]
- Roshan, V.D.; Sinto, M.; Vargees, B.T.; Kannan, S. Loss of CDKN2A and CDKN2B expression is associated with disease recurrence in oral cancer. J. Oral Maxillofac. Pathol. 2019, 23, 82–89. [Google Scholar]
- Bader, A.G.; Kang, S.; Zhao, L.; Vogt, P.K. Oncogenic PI3K deregulates transcription and translation. Nat. Rev. Cancer 2005, 5, 921–929. [Google Scholar] [CrossRef]
- Murugan, A.K.; Munirajan, A.K.; Tsuchida, N. Ras oncogenes in oral cancer: The past 20 years. Oral Oncol. 2012, 48, 383–392. [Google Scholar] [CrossRef]
- Bruckman, K.C.; Schnleben, F.; Qiu, W.; Woo, V.L.; Su, G.H. Mutational analyses of the BRAF, KRAS, and PIK3CA genes in oral squamous cell carcinoma. Oral Surgery Oral Med. Oral Pathol. Oral Radiol. Endodontology 2010, 110, 632–637. [Google Scholar] [CrossRef] [Green Version]
- Yoon, S.; Seger, R. The extracellular signal-regulated kinase: Multiple substrates regulate diverse cellular functions. Growth Factors 2006, 24, 21–44. [Google Scholar] [CrossRef] [PubMed]
- Mishima, K.; Inoue, K.; Hayashi, Y. Overexpression of extracellular-signal regulated kinases on oral squamous cell carcinoma. Oral Oncol. 2002, 38, 468–474. [Google Scholar] [CrossRef]
- Valiathan, G.M.; Thenumgal, S.J.; Jayaraman, B.; Palaniyandi, A.; Ramkumar, H.; Jayakumar, K.; Bhaskaran, S.; Ramanathan, A. Common docking domain mutation E322k of the ERK2 gene is infrequent in oral squamous cell carcinomas. Asian Pac. J. Cancer Prev. 2012, 13, 6155–6157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caulin, C.; Nguyen, T.; Longley, M.A.; Zhou, Z.; Wang, X.J.; Roop, D.R. Inducible activation of oncogenic K-ras results in tumor formation in the oral cavity. Cancer Res. 2004, 64, 5054–5058. [Google Scholar] [CrossRef] [Green Version]
- Vitale-Cross, L.; Amornphimoltham, P.; Fisher, G.; Molinolo, A.A.; Gutkind, J.S. Conditional expression of K-ras in an epithelial compartment that includes the stem cells is sufficient to promote squamous cell carcinogenesis. Cancer Res. 2004, 64, 8804–8807. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kozaki, K.I.; Imoto, I.; Pimkhaokham, A.; Hasegawa, S.; Tsuda, H.; Omura, K.; Inazawa, J. PIK3CA mutation is an oncogenic aberration at advanced stages of oral squamous cell carcinoma. Cancer Sci. 2006, 97, 1351–1358. [Google Scholar] [CrossRef] [PubMed]
- Murugan, A.K.; Thi Hong, N.; Fukui, Y.; Munirajan, A.K.; Tsuchida, N. Oncogenic mutations of the PIK3CA gene in head and neck squamous cell carcinomas. Int. J. Oncol. 2008, 32, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Qiu, W.; Schönleben, F.; Li, X.; Ho, D.J.; Close, L.G.; Manolidis, S.; Bennett, B.P.; Su, G.H. PIK3CA mutations in head and neck squamous cell carcinoma. Clin. Cancer Res. 2006, 12, 1441–1446. [Google Scholar] [CrossRef] [Green Version]
- Bian, Y.; Hall, B.; Sun, Z.J.; Molinolo, A.; Chen, W.; Gutkind, J.S.; Waes, C.V.; Kulkarni, A.B. Loss of TGF-β signaling and PTEN promotes head and neck squamous cell carcinoma through cellular senescence evasion and cancer-related inflammation. Oncogene 2012, 31, 3322–3332. [Google Scholar] [CrossRef] [Green Version]
- Tokunaga, C.; Yoshino, K.I.; Yonezawa, K. mTOR integrates amino acid- and energy-sensing pathways. Biochem. Biophys. Res. Commun. 2004, 313, 443–446. [Google Scholar] [CrossRef]
- Williams, G.H.; Stoeber, K. The Cell Cycle and Cancer. J. Pathol. 2012, 226, 352–364. [Google Scholar] [CrossRef] [PubMed]
- Vousden, K.H.; Lane, D.P. P53 in Health and Disease. Nat. Rev. Mol. Cell Biol. 2007, 8, 275–283. [Google Scholar] [CrossRef] [PubMed]
- William, W.N.; Lee, W.-C.; Lee, J.J.; Lin, H.Y.; Eterovic, A.K.; El-Naggar, A.K.; Gillenwater, A.M.; Pisegna, M.; Tong, P.; Li, L.; et al. Genomic and transcriptomic landscape of oral pre-cancers (OPCs) and risk of oral cancer (OC). J. Clin. Oncol. 2019, 37, 6009. [Google Scholar] [CrossRef]
- Opitz, O.G.; Harada, H.; Suliman, Y.; Rhoades, B.; Sharpless, N.E.; Kent, R.; Kopelovich, L.; Nakagawa, H.; Rustgi, A.K. A mouse model of human oral-esophageal cancer. J. Clin. Investig. 2002, 110, 761–769. [Google Scholar] [CrossRef]
- Li, Z.; Gonzalez, C.L.; Wang, B.; Zhang, Y.; Mejia, O.; Katsonis, P.; Lichtarge, O.; Myers, J.N.; El-Naggar, A.K.; Caulin, C. Cdkn2a suppresses metastasis in squamous cell carcinomas induced by the gain-of-function mutant p53R172H. J. Pathol. 2016, 240, 224–234. [Google Scholar] [CrossRef]
- Brown, V.L.; Harwood, C.A.; Crook, T.; Cronin, J.G.; Kelsell, D.R.; Proby, C.M. p16INK4a and p14ARF tumor suppressor genes are commonly inactivated in cutaneous squamous cell carcinoma. J. Investig. Dermatol. 2004, 122, 1284–1292. [Google Scholar] [CrossRef] [Green Version]
- Takeshima, M.; Saitoh, M.; Kusano, K.; Nagayasu, H.; Kurashige, Y.; Malsantha, M.; Arakawa, T.; Takuma, T.; Chiba, I.; Kaku, T.; et al. High frequency of hypermethylation of p14, p15 and p16 in oral pre-cancerous lesions associated with betel-quid chewing in Sri Lanka. J. Oral Pathol. Med. 2008, 37, 475–479. [Google Scholar] [CrossRef]
- Elmore, S. Apoptosis: A Review of Programmed Cell Death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef]
- Zhu, Z.; Ying, Z.; Zeng, M.; Zhang, Q.; Liao, G.; Liang, Y.; Li, C.; Zhang, C.; Wang, X.; Jiang, W.; et al. Trichosanthin cooperates with Granzyme B to restrain tumor formation in tongue squamous cell carcinoma. BMC Complement. Med. Ther. 2021, 21, 88. [Google Scholar] [CrossRef]
- Hadler-Olsen, E.; Wirsing, A.M. Tissue-infiltrating immune cells as prognostic markers in oral squamous cell carcinoma: A systematic review and meta-analysis. Br. J. Cancer 2019, 120, 714–727. [Google Scholar] [CrossRef] [Green Version]
- Fang, L.; Sun, L.; Hu, F.-F.; Chen, Q.-E. Effects of FasL Expression in Oral Squamous Cell Cancer. Asian Pac. J. Cancer Prev. 2013, 14, 281–285. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peter, M.E.; Hadji, A.; Murmann, A.E.; Brockway, S.; Putzbach, W.; Pattanayak, A.; Ceppi, P. The role of CD95 and CD95 ligand in cancer. Cell Death Differ. 2015, 22, 549–559. [Google Scholar] [CrossRef] [PubMed]
- Salvo, E.; Tu, N.H.; Scheff, N.N.; Dubeykovskaya, Z.A.; Chavan, S.A.; Aouizerat, B.E.; Ye, Y. TNFα promotes oral cancer growth, pain, and Schwann cell activation. Sci. Rep. 2021, 11, 1840. [Google Scholar] [CrossRef] [PubMed]
- Scheff, N.N.; Ye, Y.; Bhattacharya, A.; MacRae, J.; Hickman, D.N.; Sharma, A.K.; Dolan, J.C.; Schmidt, B.L. Tumor necrosis factor alpha secreted from oral squamous cell carcinoma contributes to cancer pain and associated inflammation. Pain 2017, 158, 2396–2409. [Google Scholar] [CrossRef] [PubMed]
- González-Moles, M.Á.; Ayén, Á.; González-Ruiz, I.; de Porras-Carrique, T.; González-Ruiz, L.; Ruiz-Ávila, I.; Ramos-García, P. Prognostic and Clinicopathological Significance of FADD Upregulation in Head and Neck Squamous Cell Carcinoma: A Systematic Review and Meta-Analysis. Cancers 2020, 12, 2393. [Google Scholar] [CrossRef]
- Dwivedi, R.; Pandey, R.; Chandra, S.; Mehrotra, D. Apoptosis and genes involved in oral cancer—A comprehensive review. Oncol. Rev. 2020, 14, 472. [Google Scholar] [CrossRef]
- Juneja, S.; Chaitanya, N.; Agarwal, M. Immunohistochemical expression of Bcl-2 in oral epithelial dysplasia and oral squamous cell carcinoma. Indian J. Cancer 2015, 52, 505–510. [Google Scholar] [CrossRef]
- Zheng, Q.; Gan, G.; Gao, X.; Luo, Q.; Chen, F. Targeting the IDO-BCL2A1-Cytochrome c Pathway Promotes Apoptosis in Oral Squamous Cell Carcinoma. Onco. Targets. Ther. 2021, 14, 1673–1687. [Google Scholar] [CrossRef]
- Sulkshane, P.; Pawar, S.N.; Waghole, R.; Pawar, S.S.; Rajput, P.; Uthale, A.; Oak, S.; Kalkar, P.; Wani, H.; Patil, R.; et al. Elevated USP9X drives early-to-late-stage oral tumorigenesis via stabilisation of anti-apoptotic MCL-1 protein and impacts outcome in oral cancers. Br. J. Cancer 2021, 125, 547–560. [Google Scholar] [CrossRef]
- Alam, M.; Mishra, R. Bcl-xL expression and regulation in the progression, recurrence, and cisplatin resistance of oral cancer. Life Sci. 2021, 280, 119705. [Google Scholar] [CrossRef]
- Hartman, M.L.; Czyz, M. BCL-w: Apoptotic and non-apoptotic role in health and disease. Cell Death Dis. 2020, 11, 260. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carter, R.J.; Milani, M.; Butterworth, M.; Alotibi, A.; Harper, N.; Yedida, G.; Greaves, G.; Al-Zebeeby, A.; Jorgensen, A.L.; Schache, A.G.; et al. Exploring the potential of BH3 mimetic therapy in squamous cell carcinoma of the head and neck. Cell Death Dis. 2019, 10, 912. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giam, M.; Huang, D.C.S.; Bouillet, P. BH3-only proteins and their roles in programmed cell death. Oncogene 2008, 27, S128–S136. [Google Scholar] [CrossRef] [PubMed]
- Lüthi, A.U.; Martin, S.J. The CASBAH: A searchable database of caspase substrates. Cell Death Differ. 2007, 14, 641–650. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taylor, R.C.; Cullen, S.P.; Martin, S.J. Apoptosis: Controlled demolition at the cellular level. Nat. Rev. Mol. Cell Biol. 2008, 9, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Kruidering, M.; Evan, G.I. Caspase-8 in apoptosis: The beginning of “the end”? IUBMB Life 2000, 50, 85–90. [Google Scholar] [CrossRef]
- Korsmeyer, S.J.; Wei, M.C.; Saito, M.; Weiler, S.; Oh, K.J.; Schlesinger, P.H. Pro-apoptotic cascade activates BID, which oligomerizes BAK or BAX into pores that result in the release of cytochrome c. Cell Death Differ. 2000, 7, 1166–1173. [Google Scholar] [CrossRef]
- Waterhouse, N.J.; Sedelies, K.A.; Browne, K.A.; Wowk, M.E.; Newbold, A.; Sutton, V.R.; Clarke, C.J.P.; Oliaro, J.; Lindemann, R.K.; Bird, P.I.; et al. A central role for Bid in granzyme B-induced apoptosis. J. Biol. Chem. 2005, 280, 4476–4482. [Google Scholar] [CrossRef] [Green Version]
- Das, S.N.; Khare, P.; Singh, M.K.; Sharma, S.C. Fas receptor (CD95) & fas ligand (CD178) expression in patients with tobacco-related intraoral squamous cell carcinoma. Indian J. Med. Res. 2011, 134, 54–60. [Google Scholar]
- Loro, L.L.; Vintermyr, O.K.; Johannessen, A.C.; Liavaag, P.G.; Jonsson, R. Suppression of Fas receptor and negative correlation of Fas ligand with differentiation and apoptosis in oral squamous cell carcinoma. J. Oral Pathol. Med. 1999, 28, 82–87. [Google Scholar] [CrossRef]
- Jeong, W.K.; Wieckowski, E.; Taylor, D.D.; Reichert, T.E.; Watkins, S.; Whiteside, T.L. Fas ligand-positive membranous vesicles isolated from sera of patients with oral cancer induce apoptosis of activated T lymphocytes. Clin. Cancer Res. 2005, 11, 1010–1020. [Google Scholar]
- Wang, L.H.; Ting, S.C.; Chen, C.H.; Tsai, C.C.; Lung, O.; Liu, T.C.; Lee, C.W.; Wang, Y.Y.; Tsai, C.L.; Lin, Y.C. Polymorphisms in the apoptosis-associated genes FAS and FASL and risk of oral cancer and malignant potential of oral premalignant lesions in a Taiwanese population. J. Oral Pathol. Med. 2010, 39, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Yapijakis, C.; Serefoglou, Z.; Vylliotis, A.; Nkenke, E.; Derka, S.; Vassiliou, S.; Avgoustidis, D.; Neukam, F.W.; Patsouris, E.; Vairaktaris, E. Association of polymorphisms in tumor necrosis factor alpha and beta genes with increased risk for oral cancer. Anticancer Res. 2009, 29, 2379–2386. [Google Scholar] [PubMed]
- Marín-Rubio, J.L.; Vela-Martín, L.; Fernández-Piqueras, J.; Villa-Morales, M. FADD in cancer: Mechanisms of altered expression and function, and clinical implications. Cancers 2019, 11, 1462. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chien, H.T.; Cheng, S.D.; Chuang, W.Y.; Liao, C.T.; Wang, H.M.; Huang, S.F. Clinical implications of FADD gene amplification and protein overexpression in taiwanese oral cavity squamous cell carcinomas. PLoS ONE 2016, 11, e0164870. [Google Scholar] [CrossRef]
- Lo Muzio, L.; Sartini, D.; Santarelli, A.; Rocchetti, R.; Morganti, S.; Pozzi, V.; Rubini, C.; Bambini, F.; Emanuelli, M. Expression and prognostic significance of apoptotic genes in oral squamous cell carcinoma. Mol. Carcinog. 2014, 53, 264–271. [Google Scholar] [CrossRef] [PubMed]
- Loro, L.L.; Vintermyr, O.K.; Liavaag, P.G.; Jonsson, R.; Johannessen, A.C. Oral squamous cell carcinoma is associated with decreased bcl-2/bax expression ratio and increased apoptosis. Hum. Pathol. 1999, 30, 1097–1105. [Google Scholar] [CrossRef]
- Nylander, K.; Schildt, E.-B.; Eriksson, M.; Roos, G. Squamous cell carcinoma of the head and neck. Anal. Cell. Pathol. 1997, 14, 101–110. [Google Scholar] [CrossRef] [Green Version]
- Jordan, R.C.; Catzavelos, G.C.; Barrett, A.W.; Speight, P.M. Differential expression of bcl-2 and bax in squamous cell carcinomas of the oral cavity. Eur. J. Cancer Part B Oral Oncol. 1996, 32B, 394–400. [Google Scholar] [CrossRef]
- Ravi, D.; Nalinakumari, K.R.; Rajaram, R.S.; Nair, M.K.; Pillai, M.R. Expression of programmed cell death regulatory p53 and bcl-2 proteins in oral lesions. Cancer Lett. 1996, 105, 139–146. [Google Scholar] [CrossRef]
- Teni, T.; Pawar, S.; Sanghvi, V.; Saranath, D. Expression of Bcl-2 and bax in chewing tobacco-induced oral cancers and oral lesions from India. Pathol. Oncol. Res. 2002, 8, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Popović, B.; Jekić, B.; Novaković, I.; Luković, L.J.; Tepavčević, Z.; Jurišić, V.; Vukadinović, M.; Milašin, J. Bcl-2 expression in oral squamous cell carcinoma. Ann. N. Y. Acad. Sci. 2007, 1095, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Kayano, T.; Takagi, M. Dysregulated expression of bcl-2 and bax in oral carcinomas: Evidence of post-transcriptional control. J. Oral Pathol. Med. 2000, 29, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Brayer, S.; Joannes, A.; Jaillet, M.; Gregianin, E.; Mahmoudi, S.; Marchal Sommé, J.; Fabre, A.; Mordant, P.; Cazes, A.; Crestani, B.; et al. The pro-apoptotic BAX protein influences cell growth and differentiation from the nucleus in healthy interphasic cells. Cell Cycle 2017, 16, 2108–2118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, J.; Fu, Y.; Tu, Y.Y.; Liu, Y.; Tan, Y.R.; Ju, W.T.; Pickering, C.R.; Myers, J.N.; Zhang, Z.Y.; Zhong, L.P. Mutation allele frequency threshold does not affect prognostic analysis using next-generation sequencing in oral squamous cell carcinoma. BMC Cancer 2018, 18, 758. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, R.; Das, S.; Datta, S.; Mazumdar, A.; Biswas, N.K.; Maitra, A.; Majumder, P.P.; Ghose, S.; Roy, B. Study of Caspase 8 mutation in oral cancer and adjacent precancer tissues and implication in progression. PLoS ONE 2020, 15, e0233058. [Google Scholar] [CrossRef] [PubMed]
- Coutinho-Camillo, C.M.; Lourenço, S.V.; Nishimoto, I.N.; Kowalski, L.P.; Soares, F.A. Caspase expression in oral squamous cell carcinoma. Head Neck 2011, 33, 1191–1198. [Google Scholar] [CrossRef]
- Liu, P.F.; Hu, Y.C.; Kang, B.H.; Tseng, Y.K.; Wu, P.C.; Liang, C.C.; Hou, Y.Y.; Fu, T.Y.; Liou, H.H.; Hsieh, I.C.; et al. Expression levels of cleaved caspase-3 and caspase-3 in tumorigenesis and prognosis of oral tongue squamous cell carcinoma. PLoS ONE 2017, 12, e0180620. [Google Scholar] [CrossRef] [Green Version]
- Huang, J.S.; Yang, C.M.; Wang, J.S.; Liou, H.H.; Hsieh, I.C.; Li, G.C.; Huang, S.J.; Shu, C.W.; Fu, T.Y.; Lin, Y.C.; et al. Caspase-3 expression in tumorigenesis and prognosis of buccal mucosa squamous cell carcinoma. Oncotarget 2017, 8, 84237–84247. [Google Scholar] [CrossRef] [Green Version]
- Coutinho-Camillo, C.M.; Lourenço, S.V.; Puga, R.D.; Damascena, A.S.; Teshima, T.H.N.; Kowalski, L.P.; Soares, F.A. Profile of apoptotic proteins in oral squamous cell carcinoma: A cluster analysis of 171 cases. Appl. Cancer Res. 2017, 37, 2. [Google Scholar] [CrossRef] [Green Version]
- Su, T.T. Non-apoptotic roles of apoptotic proteases: New tricks for an old dog. Open Biol. 2020, 10, 200130. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Dong, H.; Yang, G.; Song, Y.; Mou, Y.; Ni, Y. Mouse Tumor-Bearing Models as Preclinical Study Platforms for Oral Squamous Cell Carcinoma. Front. Oncol. 2020, 10, 212. [Google Scholar] [CrossRef] [PubMed]
- Amôr, N.G.; Buzo, R.F.; Ortiz, R.C.; Lopes, N.M.; Saito, L.M.; Mackenzie, I.C.; Rodini, C.O. In vitro and in vivo characterization of cancer stem cell subpopulations in oral squamous cell carcinoma. J. Oral Pathol. Med. 2021, 50, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Cox, A.D.; Der, C.J. The dark side of Ras: Regulation of apoptosis. Oncogene 2003, 22, 8999–9006. [Google Scholar] [CrossRef] [PubMed]
- Tomita, Y.; Marchenko, N.; Erster, S.; Nemajerova, A.; Dehner, A.; Klein, C.; Pan, H.; Kessler, H.; Pancoska, P.; Moll, U.M. WT p53, but not tumor-derived mutants, bind to Bcl2 via the DNA binding domain and induce mitochondrial permeabilization. J. Biol. Chem. 2006, 281, 8600–8606. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kanojia, D.; Vaidya, M.M. 4-Nitroquinoline-1-oxide induced experimental oral carcinogenesis. Oral Oncol. 2006, 42, 655–667. [Google Scholar] [CrossRef]
- Okazaki, Y.; Tanaka, Y.; Tonogi, M.; Yamane, G. Investigation of environmental factors for diagnosing malignant potential in oral epithelial dysplasia. Oral Oncol. 2002, 38, 562–573. [Google Scholar] [CrossRef]
- Foy, J.P.; Tortereau, A.; Caulin, C.; Le Texier, V.; Lavergne, E.; Thomas, E.; Chabaud, S.; Perol, D.; Lachuer, J.; Lang, W.; et al. The dynamics of gene expression changes in a mouse model of oral tumorigenesis may help refine prevention and treatment strategies in patients with oral cancer. Oncotarget 2016, 7, 35932–35945. [Google Scholar] [CrossRef]
- Tang, X.H.; Knudsen, B.; Bemis, D.; Tickoo, S.; Gudas, L.J. Oral Cavity and Esophageal Carcinogenesis Modeled in Carcinogen-Treated Mice. Clin. Cancer Res. 2004, 10, 301–313. [Google Scholar] [CrossRef] [Green Version]
- Khare, R.; Leaman, R.; Lu, Z. Accessing Biomedical Literature in the Current Information Landscape. Biomed. Lit. Min. 2014, 1159, 11–31. [Google Scholar]
- Greenspan, N.; Si, Y.; Roberts, K. Extracting Concepts for Precision Oncology from the Biomedical Literature. In AMIA Annual Symposium Proceedings; American Medical Informatics Association: Bethesda, MD, USA, 2021; Volume 2021, pp. 276–285. [Google Scholar]
- Aksnes, D.W.; Langfeldt, L.; Wouters, P. Citations, Citation Indicators, and Research Quality: An Overview of Basic Concepts and Theories. SAGE Open 2019, 9, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Irimie, A.I.; Ciocan, C.; Gulei, D.; Mehterov, N.; Atanasov, A.G.; Dudea, D.; Berindan-Neagoe, I. Current insights into oral cancer epigenetics. Int. J. Mol. Sci. 2018, 19, 670. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chakraborty, R.; Vickery, K.; Darido, C.; Ranganathan, S.; Hu, H. Bacterial Antigens Reduced the Inhibition Effect of Capsaicin on Cal 27 Oral Cancer Cell Proliferation. Int. J. Mol. Sci. 2021, 22, 8686. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, R.; Hu, H.; Darido, C.; Vickery, K.; Ranganathan, S. ML218 HCl Is More Efficient Than Capsaicin in Inhibiting Bacterial Antigen-Induced Cal 27 Oral Cancer Cell Proliferation. Int. J. Mol. Sci. 2021, 22, 12559. [Google Scholar] [CrossRef]
- Chakraborty, R.; Hu, H.; Mangani, A.S.; Vickery, K.; Ranganathan, S. Combined bacterial antigen lipopolysaccharide and lipoteichoic acid increase Cal 27 oral cancer cell proliferation. Dent. Oral Maxillofac. Res. 2021, 4, 1–6. [Google Scholar]

| Protein | Role in Oral Cancer | Reference |
|---|---|---|
| EGF | Modulates growth and differentiation of oral cancer cells | Bernades et al. [43] |
| ErbB proteins (EGFR, ErbB2, ErbB3, ErbB4) | Progression and pathogenesis of OSCC | Bernades et al. [43] |
| GRB2 | Overexpression is correlated with lymph node metastasis | Li et al. [44] |
| Shc | Activation of Shc results in couple β6 signaling to the Raf-ERK/MAPK pathway | Li et al. [45] |
| SOS1 | Useful biomarker in OSCC | Baltanas et al. [46] |
| Ras | Ras mutations confer therapeutic resistance | Batta et al. [47] |
| Raf | Oral cancer behaves similarly like other cancer in terms of perturbations in Raf kinase inhibitor protein | Hallums et al. [48] |
| MEK/ERK | Related to chemoresistance in oral cancer | Kashyap et al. [49] |
| MAPK | Promotes tumour cell proliferation and anti-apoptosis | Peng et al. [50] |
| PI3K/Akt/mTOR | Overexpression related to poor prognosis of oral cancer | Harsha et al. [51] |
| PTEN | Epigenetic biomarker in OSCC | Sushma et al. [52] |
| PDK1 | Targeting PDK1 sensitizes NOTCH1 to PI3K/mTOR pathway | Sambandam et al. [53] |
| 4E-BP | Reactivated by mTOR inhibition in OSCC | Wang et al. [54] |
| Cyclin D | Early biomarker in oral cancer | Ramakrishna et al. [55] |
| CDK4, CDK6 | The index scores of CDK4, CDK6, and cyclin D1 are associated with the transformation from pre-cancer to oral cancer stage | Kujan et al. [56] |
| Cyclin E | Related to progression of cancer | Moharil et al. [57] |
| Cyclin A, Cyclin B | Prognostic significance in OSCC | Monteiro et al. [58] |
| CDK1, CDK2 | CDK1 serve as a prognostic marker and malignant degree for the survival of OSCC patients. CDK2 overexpression serve as a prognostic marker of OSCC | Chen et al. [59]; Mihara et al. [60] |
| Pocket proteins (RB, RBL1/p107, RBL2/p130) | Suppress OSCC | Shin et al. [61] |
| ATM and ATR | Related to radiosensitivity in OSCC | Parikh et al. [62] |
| p53 | Inactivated in OSCC resulting in prolonged cell cycle | Ragos et al. [63] |
| CIP/KIP family (p21, p27, p57) | Cell cycle regulators | Perez-Sayans et al. [64] |
| INK4 family (p15, p16, p18, p19) | Related to transformation from precancer to oral cancer stage | Agarwal et al. [65] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, S.; Chakraborty, R.; Ranganathan, S. Proliferation and Apoptosis Pathways and Factors in Oral Squamous Cell Carcinoma. Int. J. Mol. Sci. 2022, 23, 1562. https://doi.org/10.3390/ijms23031562
He S, Chakraborty R, Ranganathan S. Proliferation and Apoptosis Pathways and Factors in Oral Squamous Cell Carcinoma. International Journal of Molecular Sciences. 2022; 23(3):1562. https://doi.org/10.3390/ijms23031562
Chicago/Turabian StyleHe, Steven, Rajdeep Chakraborty, and Shoba Ranganathan. 2022. "Proliferation and Apoptosis Pathways and Factors in Oral Squamous Cell Carcinoma" International Journal of Molecular Sciences 23, no. 3: 1562. https://doi.org/10.3390/ijms23031562







