Effects of Cadmium Exposure on Gut Villi in Danio rerio
Abstract
:1. Introduction
2. Results
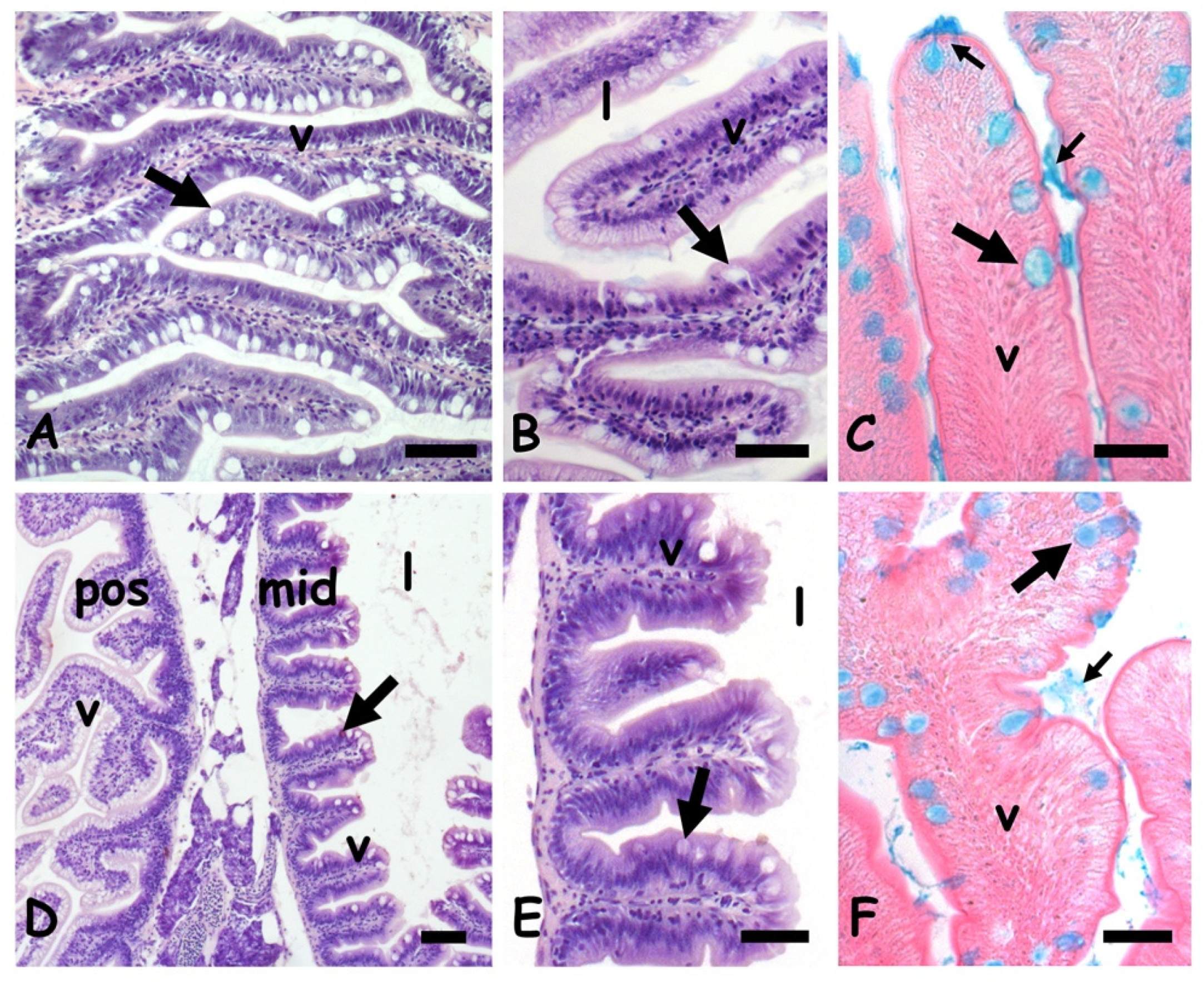
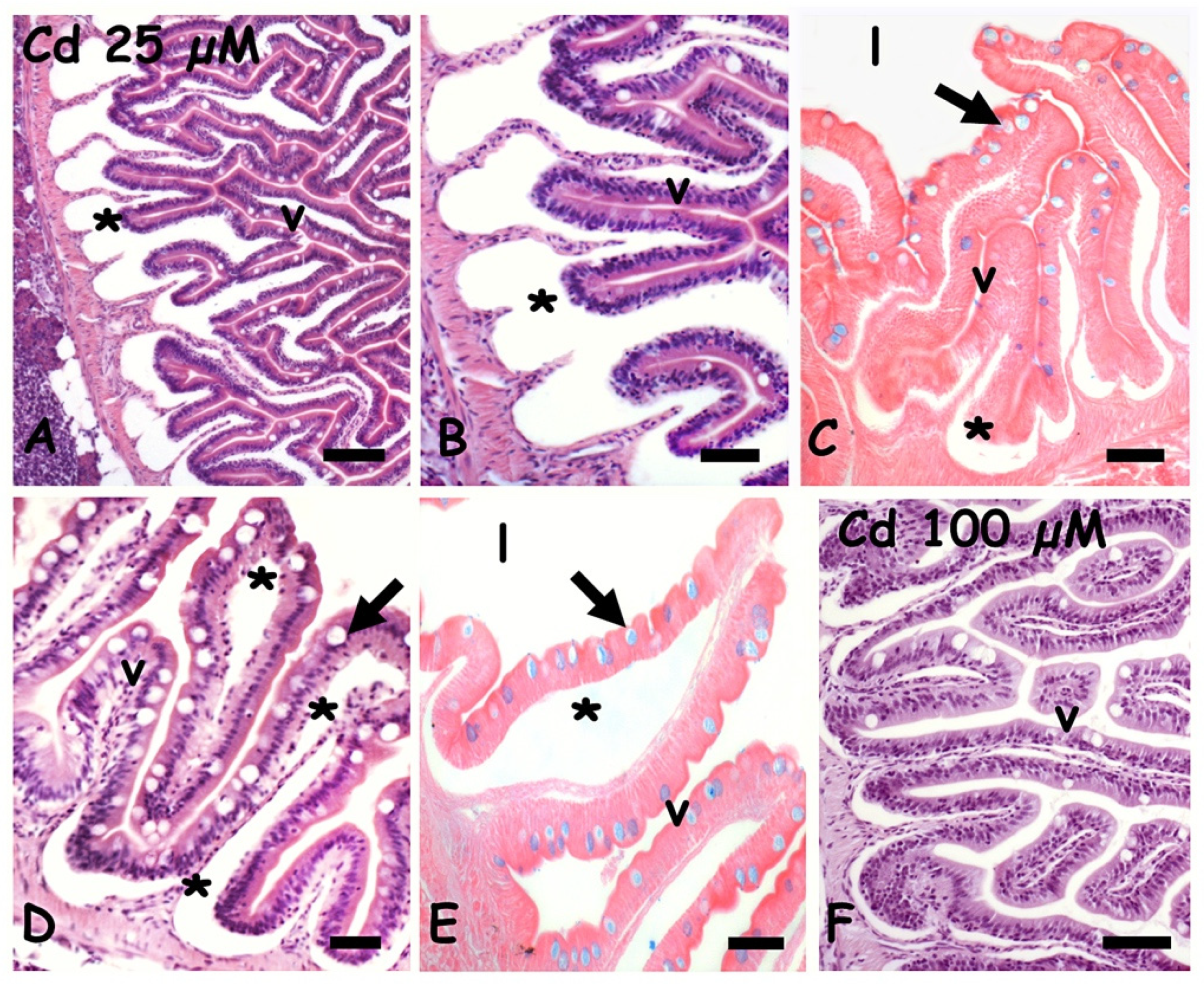
2.1. Effects of Cadmium on Gut Morphology
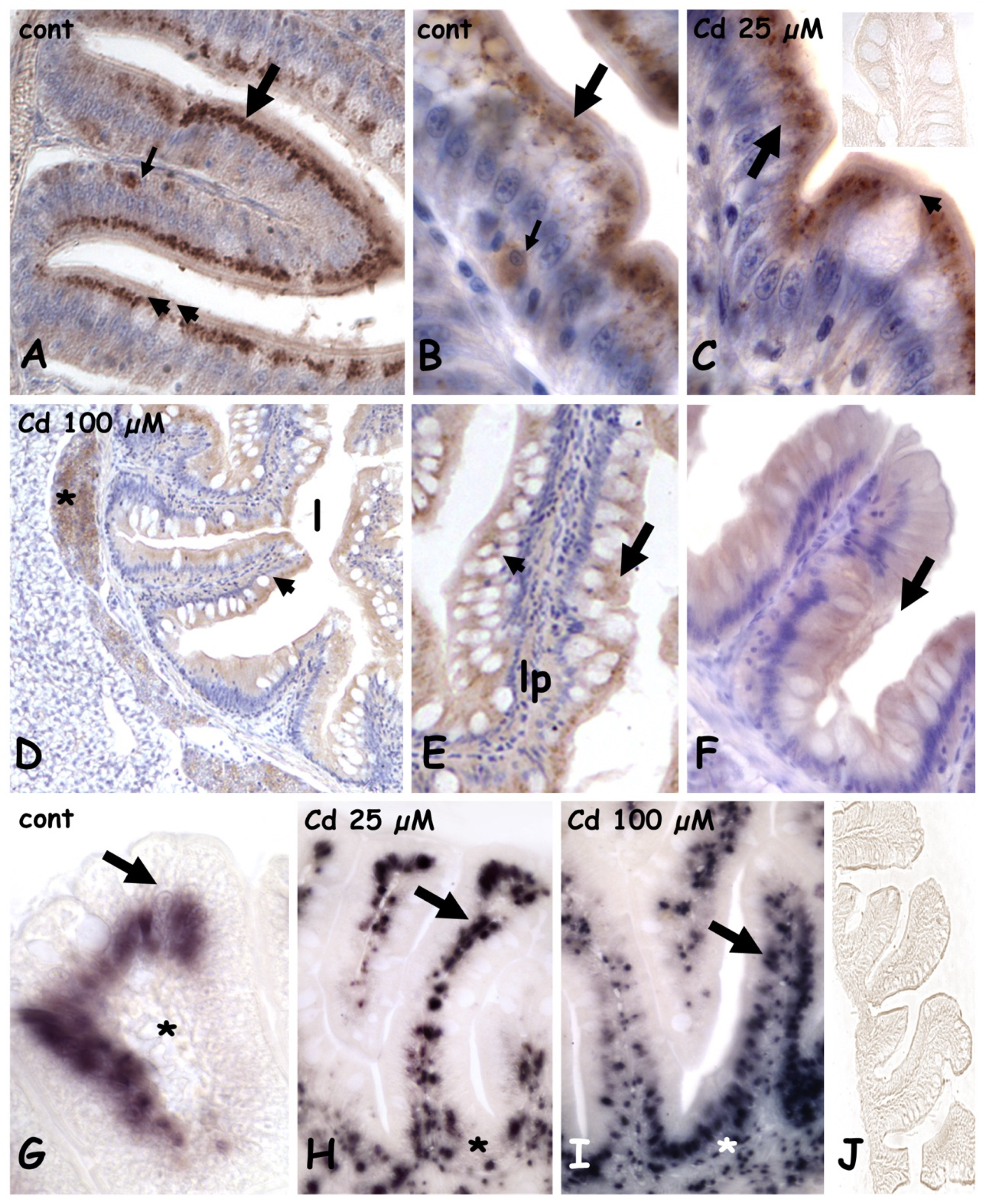
2.2. Effects of Cadmium on Metallothionein Expression
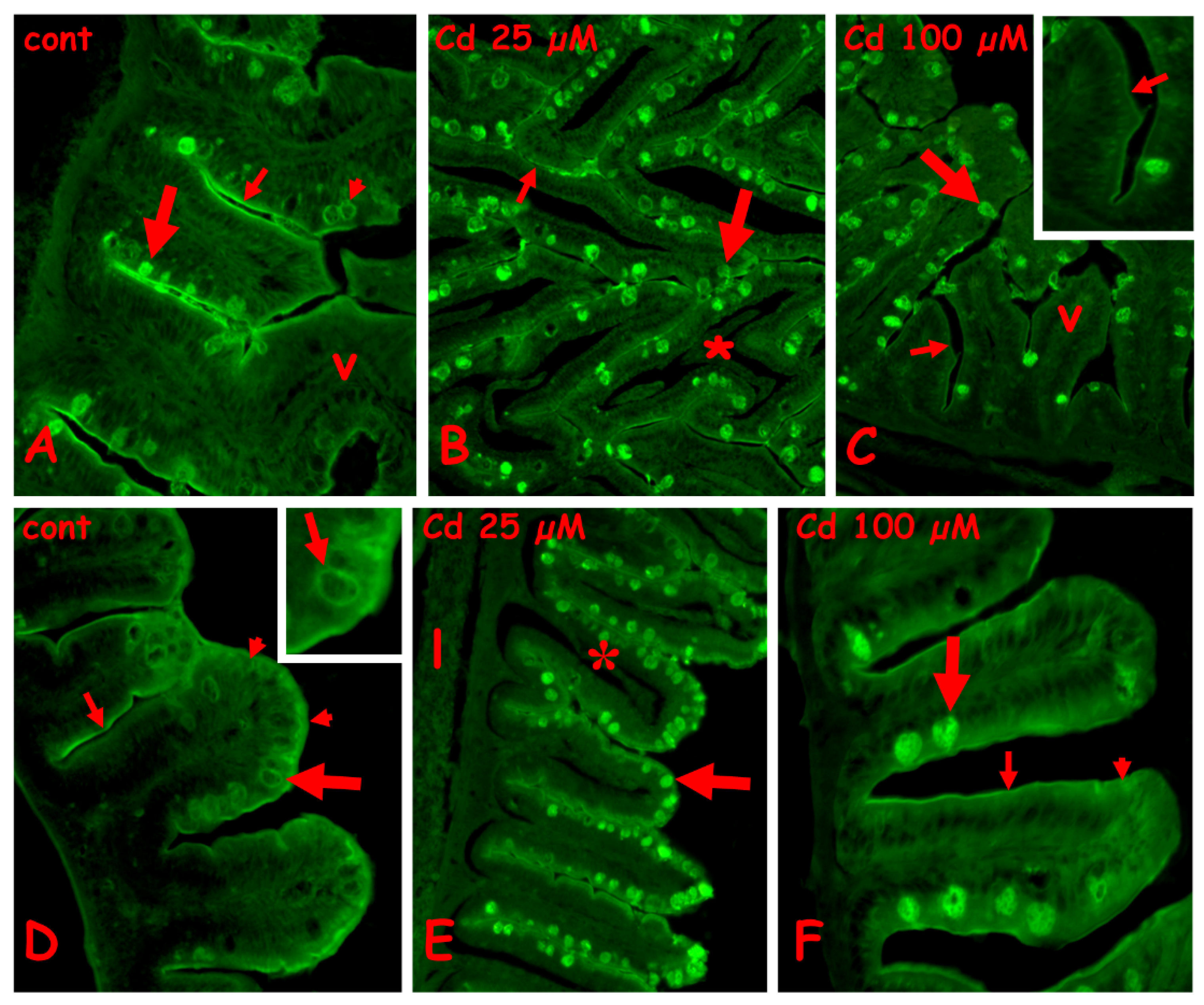
2.3. Effects of Cadmium on the Carbohydrate Composition of Gut Cells
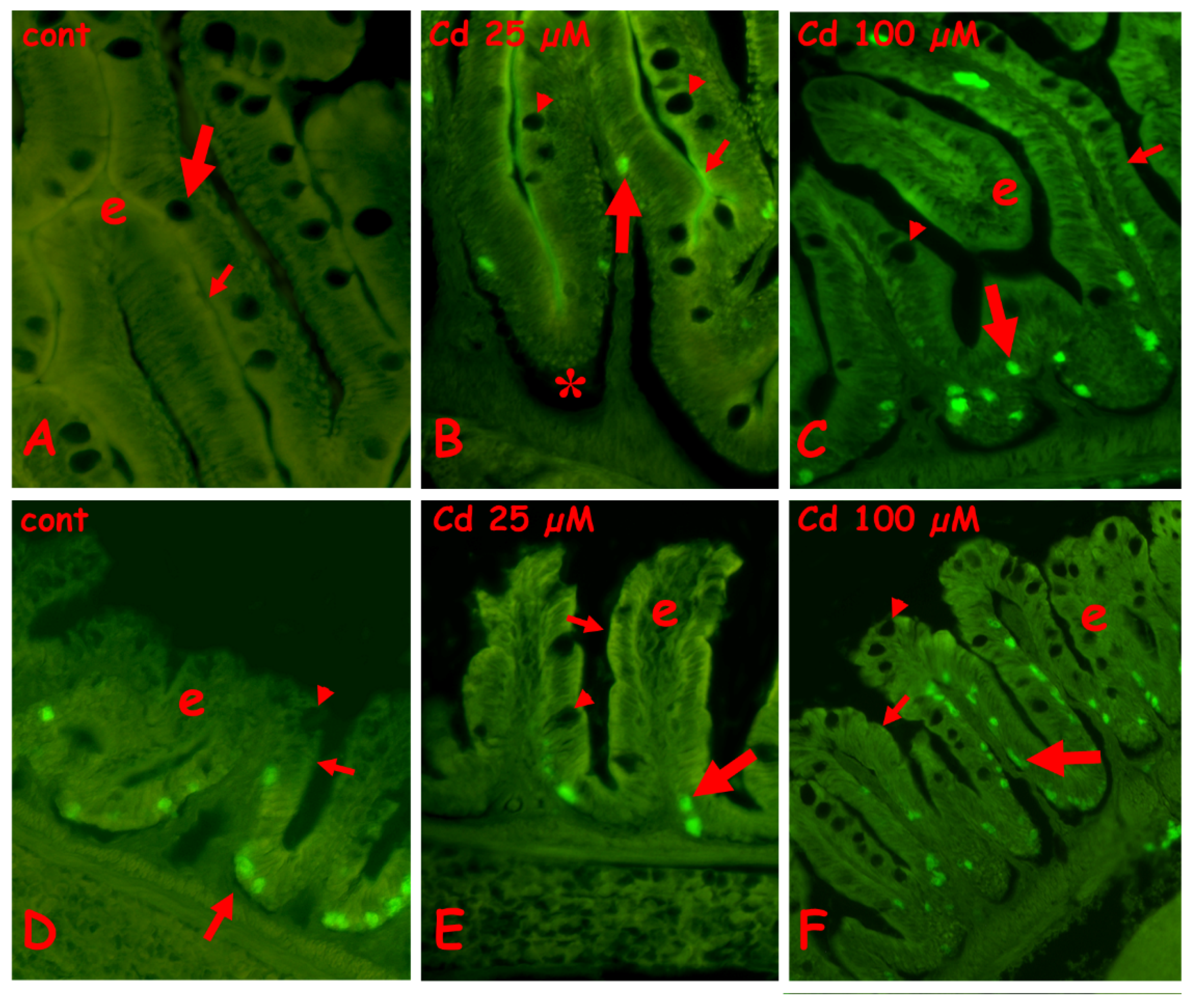
2.3.1. N-Acetyl-Glucosamine Staining with Fluorescent WGA Lectin
2.3.2. Galactose Staining with Fluorescent PNA Lectin
2.3.3. Staining for N-Acetyl-Galactosamine with Fluorescent RCA Lectin
3. Discussion
4. Material and Methods
4.1. Animals and Cadmium Treatments
4.2. Tissue Sampling, Processing, and Staining
4.3. Metallothionein Localization
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Thornton, I. Sources and pathways of cadmium in the environment. IARC Sci. Publ. 1992, 118, 149–162. [Google Scholar]
- Genchi, G.; Sinicropi, M.S.; Lauria, G.; Carocci, A.; Catalano, A. The effects of cadmium toxicity. Int. J. Environ. Res. Public Health 2020, 17, 3782. [Google Scholar] [CrossRef] [PubMed]
- Alonso, Á.; Valle-Torres, G. Feeding behavior of an aquatic snail as a simple endpoint to assess the exposure to cadmium. Bull. Environ. Contam. Toxicol. 2018, 100, 82–88. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, L.; Abel, G.M.; Storm, D.R.; Xia, Z. Cadmium exposure impairs cognition and olfactory memory in male C57BL/6 mice. Toxicol. Sci. 2018, 161, 87–102. [Google Scholar] [CrossRef] [Green Version]
- Capriello, T.; Grimaldi, M.C.; Cofone, R.; D’Aniello, S.; Ferrandino, I. Effects of aluminium and cadmium on hatching and swimming ability in developing zebrafish. Chemosphere 2019, 222, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Gumilar, F.; Bras, C.; Aggio, P.; Domínguez, S.; Bartos, M.; Gallegos, C.; Mónaco, N.; Minetti, A. Prenatal exposure to cadmium during organogenesis impairs memory in young rats. Int. J. Toxicol. 2019, 38, 312–318. [Google Scholar] [CrossRef]
- Simoniello, P.; Motta, C.M.; Scudiero, R.; Trinchella, F.; Filosa, S. Cadmium-induced teratogenicity in lizard embryos: Correlation with metallothionein gene expression. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2011, 153, 119–127. [Google Scholar] [CrossRef]
- Geng, H.X.; Wang, L. Cadmium: Toxic effects on placental and embryonic development. Environ. Toxicol. Pharmacol. 2019, 67, 102–107. [Google Scholar] [CrossRef]
- Carginale, V.; Capasso, C.; Scudiero, R.; Parisi, E. Identification of cadmium-sensitive genes in the Antarctic fish Chionodraco hamatus by messenger RNA differential display. Gene 2002, 299, 117–124. [Google Scholar] [CrossRef]
- Trinchella, F.; Cannetiello, M.; Simoniello, P.; Filosa, S.; Scudiero, R. Differential gene expression profiles in embryos of the lizard Podarcis sicula under in ovo exposure to cadmium. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2010, 151, 33–39. [Google Scholar] [CrossRef]
- Scudiero, R.; Motta, C.M.; Simoniello, P. Impact of environmental stressors on gene expression in the embryo of the Italian wall lizard. Appl. Sci. 2021, 11, 4723. [Google Scholar] [CrossRef]
- Simoniello, P.; Filosa, S.; Riggio, M.; Scudiero, R.; Tammaro, S.; Trinchella, F.; Motta, C.M. Responses to cadmium intoxication in the liver of the wall lizard Podarcis sicula. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2010, 151, 194–203. [Google Scholar] [CrossRef]
- Avallone, B.; Cerciello, R.; Cretì, P.; Pizzoleo, C.; Scudiero, R.; Tizzano, M.; Panzuto, R.; Simoniello, P.; Montinari, M.R.; Motta, C.M. Long term exposure to cadmium: Pathological effects on kidney tubules cells in Sparus aurata juveniles. Aquat. Toxicol. 2017, 193, 201–209. [Google Scholar] [CrossRef]
- Simoniello, P.; Trinchella, F.; Filosa, S.; Scudiero, R.; Magnani, D.; Theil, T.; Motta, C.M. Cadmium contaminated soil affects retinogenesis in lizard embryos. J. Exp. Zool. Part A Ecol. Genet. Physiol. 2014, 321, 207–219. [Google Scholar] [CrossRef]
- Avallone, B.; Crispino, R.; Cerciello, R.; Simoniello, P.; Panzuto, R.; Motta, C.M. Cadmium effects on the retina of adult Danio rerio. CR Biol. 2015, 338, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Monaco, A.; Grimaldi, M.C.; Ferrandino, I. Neuroglial alterations in the zebrafish brain exposed to cadmium chloride. J. Appl. Toxicol. 2016, 36, 1629–1638. [Google Scholar] [CrossRef] [PubMed]
- Branca, J.J.V.; Morucci, G.; Pacini, A. Cadmium-induced neurotoxicity: Still much ado. Neural Regen Res. 2018, 13, 1879–1882. [Google Scholar] [PubMed]
- Lienesch, L.A.; Dumont, J.N.; Bantle, J.A. The effect of cadmium on oogenesis in Xenopus laevis. Chemosphere 2000, 41, 1651–1658. [Google Scholar] [CrossRef]
- Simoniello, P.; Filosa, S.; Scudiero, R.; Trinchella, F.; Motta, C.M. Cadmium impairment of reproduction in the female wall lizard Podarcis sicula. Environ. Toxicol. 2013, 28, 553–562. [Google Scholar] [CrossRef]
- Zhao, L.L.; Ru, Y.F.; Liu, M.; Tang, J.N.; Zheng, J.F.; Wu, B.; Gu, Y.H.; Shi, H.J. Reproductive effects of cadmium on sperm function and early embryonic development in vitro. PLoS ONE 2017, 12, e0186727. [Google Scholar] [CrossRef] [Green Version]
- Motta, C.M.; Simoniello, P.; Di Lorenzo, M.; Migliaccio, V.; Panzuto, R.; Califano, E.; Santovito, G. Endocrine disrupting effects of copper and cadmium in the oocytes of the Antarctic Emerald rockcod Trematomus bernacchii. Chemosphere 2021, 268, 129282. [Google Scholar] [CrossRef] [PubMed]
- Tao, S.; Liu, C.; Dawson, R.; Long, A.; Xu, F. Uptake of cadmium adsorbed on particulates by gills of goldfish (Carassius auratus). Ecotoxicol. Environ. Saf. 2000, 47, 306–313. [Google Scholar] [CrossRef] [PubMed]
- Capaldo, A.; Gay, F.; Scudiero, R.; Trinchella, F.; Caputo, I.; Lepretti, M.; Marabotti, A.; Esposito, C.; Laforgia, V. Histological changes, apoptosis and metallothionein levels in Triturus carnifex (Amphibia, Urodela) exposed to environmental cadmium concentrations. Aquat. Toxicol. 2016, 173, 63–73. [Google Scholar] [CrossRef]
- Chowdhury, M.J.; McDonald, D.G.; Wood, C.M. Gastrointestinal uptake and fate of cadmium in rainbow trout acclimated to sublethal dietary cadmium. Aquat. Toxicol. 2004, 69, 149–163. [Google Scholar] [CrossRef]
- Cretì, P.; Trinchella, F.; Scudiero, R. Heavy metals bioaccumulation and metallothionein content in tissues of the sea bream Sparus aurata from three different fish farming systems. Environ. Monit. Assess. 2010, 165, 321–329. [Google Scholar] [CrossRef]
- Tinkov, A.A.; Gritsenko, V.A.; Skalnaya, M.G.; Cherkasov, S.V.; Aaseth, J.; Skalny, A.V. Gut as a target for cadmium toxicity. Environ. Pollut. 2018, 235, 429–434. [Google Scholar] [CrossRef]
- Chang, X.; Li, H.; Feng, J.; Chen, Y.; Nie, G.; Zhang, J. Effects of cadmium exposure on the composition and diversity of the intestinal microbial community of common carp (Cyprinus carpio L.). Ecotoxicol. Environ. Saf. 2019, 171, 92–98. [Google Scholar] [CrossRef]
- Younis, E.; Abdel-Warith, A.W.; Al-Asgah, N.; Ebaid, H. Histopathological alterations in the liver and intestine of Nile tilapia Oreochromis niloticus exposed to long-term sublethal concentrations of cadmium chloride. Chin. J. Oceanol. Limnol. 2015, 33, 846–852. [Google Scholar] [CrossRef]
- Bais, U.E.; Lokhande, M.Y. Effect of cadmium chloride on histopathological changes in the freshwater fish Ophiocephalus striatus (Channa). Int. J. Zool. Res. 2012, 8, 23–32. [Google Scholar] [CrossRef]
- Ma, J.; Rubin, B.K.; Voynow, J.A. Mucins, Mucus, and Goblet Cells. Chest 2018, 154, 169–176. [Google Scholar] [CrossRef]
- Birchenough, G.M.; Johansson, M.E.; Gustafsson, J.K.; Bergström, J.H.; Hansson, G.C. New developments in goblet cell mucus secretion and function. Mucosal Immunol. 2015, 8, 712–719. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Knoop, K.A.; Newberry, R.D. Goblet cells: Multifaceted players in immunity at mucosal surfaces. Mucosal Immunol. 2018, 11, 1551–1557. [Google Scholar] [CrossRef] [PubMed]
- Heazlewood, C.K.; Cook, M.C.; Eri, R.; Price, G.R.; Tauro, S.B.; Taupin, D.; Thornton, D.J.; Png, C.W.; Crockford, T.L.; Cornall, R.J.; et al. Aberrant mucin assembly in mice causes endoplasmic reticulum stress and spontaneous inflammation resembling ulcerative colitis. PLoS Med. 2008, 5, e54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schroeder, B.O. Fight them or feed them: How the intestinal mucus layer manages the gut microbiota. Gastroenterol. Rep. 2019, 7, 3–12. [Google Scholar] [CrossRef] [Green Version]
- Kim, Y.S.; Ho, S.B. Intestinal goblet cells and mucins in health and disease: Recent insights and progress. Curr. Gastroenterol. Rep. 2010, 12, 319–330. [Google Scholar] [CrossRef] [Green Version]
- Pothuraju, R.; Krishn, S.R.; Gautam, S.K.; Priya Pai, P.; Ganguly, K.; Chaudhary, S.; Rachagani, S.; Kaur, S.; Batra, S.K. Mechanistic and functional shades of mucins and associated glycans in colon cancer. Cancers 2020, 12, 649. [Google Scholar] [CrossRef] [Green Version]
- Xie, D.; Li, Y.; Liu, Z.; Chen, Q. Inhibitory effect of cadmium exposure on digestive activity, antioxidant capacity and immune defense in the intestine of yellow catfish (Pelteobagrus fulvidraco). Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2019, 222, 65–73. [Google Scholar] [CrossRef]
- Liu, J.; Pang, J.J.; Tu, Z.C.; Wang, H.; Sha, X.M.; Shao, Y.H.; Liu, G.X. The accumulation, histopathology, and intestinal microorganism effects of waterborne cadmium on Carassius auratus gibelio. Fish Physiol. Biochem. 2019, 45, 231–243. [Google Scholar] [CrossRef]
- Jiang, Z.; Mu, W.; Yang, Y.; Sun, M.; Liu, Y.; Gao, Z.; Li, J.; Gu, P.; Wang, H.; Lu, Y.; et al. Cadmium exacerbates dextran sulfate sodium-induced chronic colitis and impairs intestinal barrier. Sci. Total Environ. 2020, 744, 140844. [Google Scholar] [CrossRef]
- Xie, S.; Jiang, L.; Wang, M.; Sun, W.; Yu, S.; Turner, J.-R.; Yu, Q. Cadmium ingestion exacerbates Salmonella infection, with a loss of goblet cells through activation of Notch signaling pathways by ROS in the intestine. J. Hazard Mater. 2020, 391, 122262. [Google Scholar] [CrossRef]
- Gallego Ríos, S.E.; Ramírez Botero, C.M.; López Marín, B.E.; Velásquez Rodríguez, C.M. Evaluation of mercury, lead, and cadmium in the waste material of crevalle jack fish from the Gulf of Urabá, Colombian Caribbean, as a possible raw material in the production of sub-products. Environ. Monit. Assess. 2018, 190, 115. [Google Scholar] [CrossRef]
- Rodrigo, M.A.M.; Jimemez, A.M.J.; Haddad, Y.; Bodoor, K.; Adam, P.; Krizkova, S.; Heger, Z.; Adam, V. Metallothionein isoforms as double agents—Their roles in carcinogenesis, cancer progression and chemoresistance. Drug Resist. Updat. 2020, 52, 100691. [Google Scholar] [CrossRef] [PubMed]
- Takano, H.; Satoh, M.; Shimada, A.; Sagai, M.; Yoshikawa, T.; Tohyama, C. Cytoprotection by metallothionein against gastroduodenal mucosal injury caused by ethanol in mice. Lab. Investig. 2000, 80, 371–377. [Google Scholar] [CrossRef] [Green Version]
- Hanna, M.I.; Shaheed, I.B.; Elias, N.S. A contribution on chromium and lead toxicity in cultured Oreochromis niloticus. Egypt. J. Aquat. Biol. Fish. 2005, 9, 177–209. [Google Scholar]
- Thévenod, F. Catch me if you can! Novel aspects of cadmium transport in mammalian cells. Biometals 2010, 23, 857–875. [Google Scholar] [CrossRef] [PubMed]
- Choong, G.; Liu, Y.; Templeton, D.M. Interplay of calcium and cadmium in mediating cadmium toxicity. Chem. Biol. Interact. 2014, 211, 54–65. [Google Scholar] [CrossRef]
- Earley, B.J.; Cubillas, C.; Warnhoff, K.; Ahmad, R.; Alcantar, A.; Lyon, M.D.; Schneider, D.L.; Kornfeld, K. Cadmium hijacks the high zinc response by binding and activating the HIZR-1 nuclear receptor. Proc. Natl. Acad. Sci. USA 2021, 118, e2022649118. [Google Scholar] [CrossRef] [PubMed]
- Dostie, K.E.; Thees, A.V.; Lynes, M.A. Metallothionein: A novel therapeutic target for treatment of inflammatory bowel disease. Curr. Pharm. Des. 2018, 24, 3155–3161. [Google Scholar] [CrossRef]
- Hart, S.; Wrathmell, A.B.; Harris, J.E.; Grayson, T.H. Gut immunology in fish: A review. Dev. Comp. Immunol. 1988, 12, 453–480. [Google Scholar] [CrossRef]
- Soufy, H.; Soliman, M.K.; El-Manakhly, E.M.; Gaafar, A.Y. Some biochemical and pathological investigations on monosex Tilapia following chronic exposure to carbofuran pesticides. Global Vet. 2007, 1, 45–52. [Google Scholar]
- Irato, P.; Santovito, G.; Piccinni, E.; Albergoni, V. Oxidative burst and metallothionein as a scavenger in macrophages. Immunol. Cell. Biol. 2001, 79, 251–254. [Google Scholar] [CrossRef] [PubMed]
- Aydemir, T.B.; Blanchard, R.K.; Cousins, R.J. Zinc supplementation of young men alters metallothionein, zinc transporter, and cytokine gene expression in leukocyte populations. Proc. Natl. Acad. Sci. USA 2006, 103, 1699–1704. [Google Scholar] [CrossRef] [Green Version]
- Mon, K.K.Z.; Kern, C.; Chanthavixay, G.; Wang, Y.; Zhou, H. Tolerogenic immunoregulation towards Salmonella enteritidis contributes to colonization persistence in young chicks. Infect. Immun. 2021, 89, e0073620. [Google Scholar] [CrossRef] [PubMed]
- Biniaminov, M.; Ramot, B.; Rosenthal, E.; Novogrodsky, A. Galactose oxidase-induced blastogenesis of human lymphocytes and the effect of macrophages on the reaction. Clin. Exp. Immunol. 1975, 19, 93–98. [Google Scholar] [PubMed]
- Kurata, O.; Hatai, K. Activation of carp leukocytes by a galactose-binding protein from Aphanomyces piscicida. Dev. Comp. Immunol. 2002, 26, 461–469. [Google Scholar] [CrossRef]
- Scott, J.E. Alcian blue. Now you see it, now you don’t. Eur. J. Oral Sci. 1996, 104, 2–9. [Google Scholar] [CrossRef]
- Avallone, B.; Agnisola, C.; Cerciello, R.; Panzuto, R.; Simoniello, P.; Cretì, P.; Motta, C.M. Structural and functional changes in the zebrafish (Danio rerio) skeletal muscle after cadmium exposure. Cell. Biol. Toxicol. 2015, 31, 273–283. [Google Scholar] [CrossRef]
- Arellano, J.; Dinis, M.T.; Sarasquete, C. Histomorphological and histochemical characteristics of the intestine of the Senegal sole, Solea senegalensis. Eur. J. Histochem. 1999, 43, 121–133. [Google Scholar]
- Marchetti, L.; Capacchietti, M.; Sabbieti, M.G.; Accili, D.; Materazzi, G.; Menghi, G. Histology and carbohydrate histochemistry of the alimentary canal in the rainbow trout Oncorhynchus mykiss. J. Fish Biol. 2006, 68, 1808–1821. [Google Scholar] [CrossRef]
- Fiertak, A.; Kilarski, W.M. Glycoconjugates of the intestinal goblet cells of four cyprinids. Cell. Mol. Life Sci. 2002, 59, 1724–1733. [Google Scholar] [CrossRef]
- Godwin, O.C.; Clifford, A.N.; Agatha, A. Evaluation of the morphological adaptations of the small intestine of the African pied crow (Corvus albus). J. Basic Appl. Zool. 2016, 75, 54–60. [Google Scholar] [CrossRef]
- Wallace, K.N.; Akhter, S.; Smith, E.M.; Lorent, K.; Pack, M. Intestinal growth and differentiation in zebrafish. Mech. Dev. 2005, 122, 157–173. [Google Scholar] [CrossRef] [PubMed]
- Motta, C.M.; Tammaro, S.; Simoniello, P.; Prisco, M.; Ricchiari, L.; Andreuccetti, P.; Filosa, S. Characterization of cortical alveoli content in several species of Antarctic notothenioids. J. Fish Biol. 2005, 66, 442–453. [Google Scholar] [CrossRef]
- Motta, C.M.; Frezza, V.; Simoniello, P. Caspase 3 in molluscan tissues: Localization and possible function. J. Exp. Zool. Part A Ecol. Genet. Physiol. 2013, 319, 548–559. [Google Scholar] [CrossRef] [PubMed]
- Scudiero, R.; Carginale, V.; Riggio, M.; Capasso, C.; Capasso, A.; Kille, P.; di Prisco, G.; Parisi, E. Difference in hepatic metallothionein content in Antarctic red-blooded and haemoglobinless fish: Undetectable metallothionein levels in haemoglobinless fish is accompanied by accumulation of untranslated metallothionein mRNA. Biochem. J. 1997, 322, 207–211. [Google Scholar] [CrossRef] [Green Version]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Motta, C.M.; Califano, E.; Scudiero, R.; Avallone, B.; Fogliano, C.; De Bonis, S.; Raggio, A.; Simoniello, P. Effects of Cadmium Exposure on Gut Villi in Danio rerio. Int. J. Mol. Sci. 2022, 23, 1927. https://doi.org/10.3390/ijms23041927
Motta CM, Califano E, Scudiero R, Avallone B, Fogliano C, De Bonis S, Raggio A, Simoniello P. Effects of Cadmium Exposure on Gut Villi in Danio rerio. International Journal of Molecular Sciences. 2022; 23(4):1927. https://doi.org/10.3390/ijms23041927
Chicago/Turabian StyleMotta, Chiara Maria, Emanuela Califano, Rosaria Scudiero, Bice Avallone, Chiara Fogliano, Salvatore De Bonis, Anja Raggio, and Palma Simoniello. 2022. "Effects of Cadmium Exposure on Gut Villi in Danio rerio" International Journal of Molecular Sciences 23, no. 4: 1927. https://doi.org/10.3390/ijms23041927








