1. Introduction
SARS-CoV-2 is the human coronavirus (CoV) responsible for the CoV disease 19 (COVID-19) pandemic. Human CoVs are members of the Nidovirales order and belong to the Coronaviridae family. To date, seven species of human CoVs have been described: HCoV-NL63, HCoV-229E, HCoV-OC43, HCoVHKU1, SARS-CoV, MERS-CoV, and SARS-CoV-2. Like other CoVs, SARS-CoV-2 is an enveloped virus with a positive-sense, single-stranded RNA genome. SARS-CoV-2 belongs to the genus betacoronavirus, together with SARS-CoV and MERS-CoV (with 80% and 50% identity, respectively) [
1].
Coronaviruses, including SARS-CoV-2, have the largest genomes (26–32 kb) among all of the RNA virus families, which are flanked by 5′ and 3′ untranslated regions. SARS-CoV-2 RNA contains a common ‘leader’ sequence (of 70 nt) [
2]. Upon cell entry, the viral genomic RNA (gRNA) is translated to produce nonstructural proteins from two large open reading frames (ORFs), ORF1a, and ORF1b, via proteolytic cleavage. Among these, 15 nonstructural proteins make up the viral replication and transcription complex [
3]. Of importance, Nsp12 (which harbors RNA-dependent RNA polymerase; RdRp) leads the viral replication and transcription mechanisms by using viral RNA as the template.
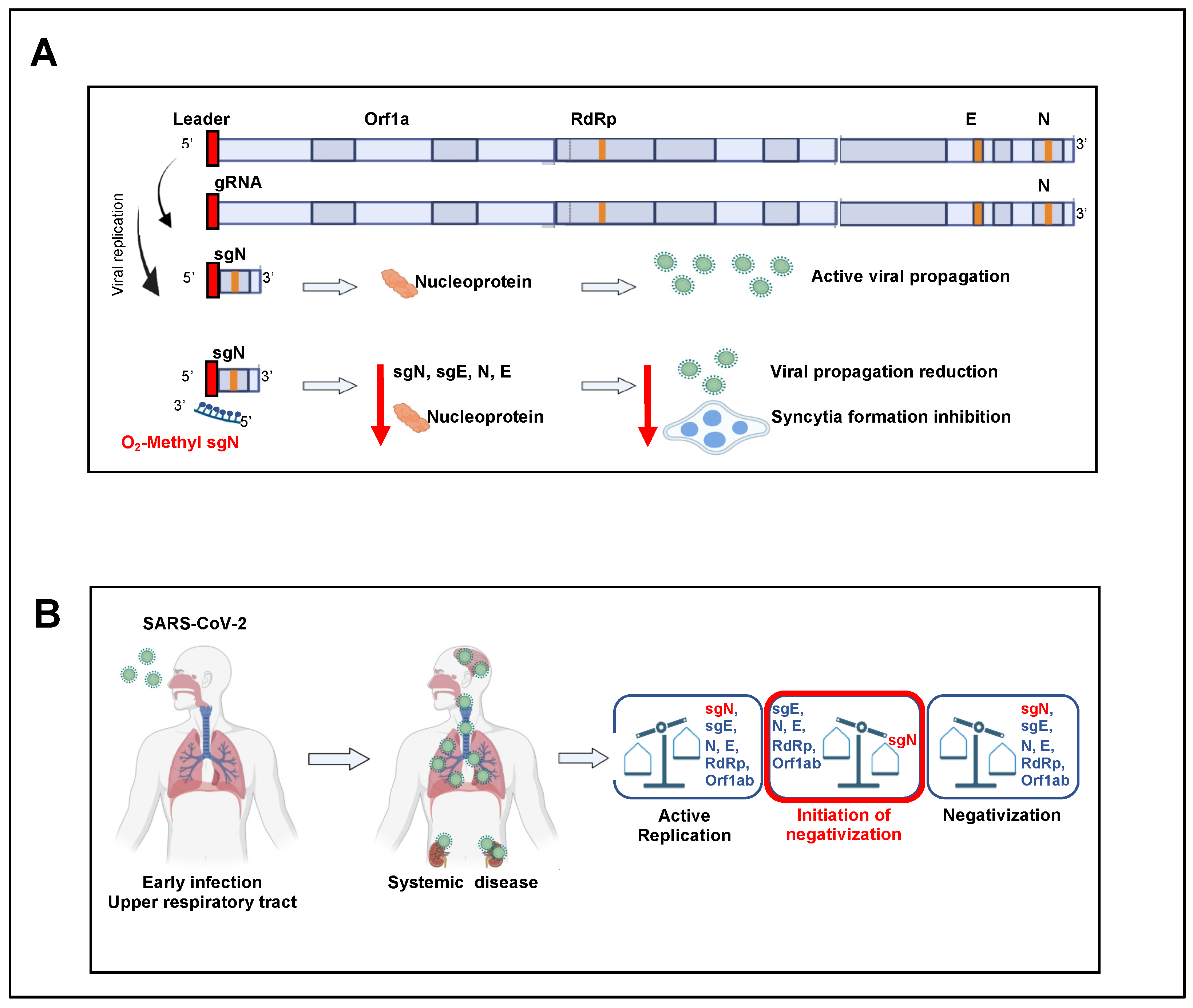
A hallmark of CoVs is a “discontinuous transcription” mechanism that produces a set of subgenomic RNAs (sgRNAs). Indeed, during their viral cycle, CoVs replicate their genomic RNA to produce full-length negative-sense RNA molecules that act as the templates for the synthesis of positive-sense gRNAs that are then packaged by the structural proteins into newly assembled virions. However, the ORFs are transcribed from the 3′ one-third of the genome to form sgRNAs that encode the SARS-CoV-2 structural proteins (i.e., spike [S], envelope [E], membrane [M], nucleocapsid [N]), and several accessory proteins (e.g., 3a, 6, 7a, 7b, 8, 10), according to the “leader-to-body fusion” model [
1]. Briefly, during the negative-strand synthesis, the viral replication and transcription complex interrupts transcription when it crosses a transcription regulatory sequence (TRS) upstream of most ORFs in the 3′ one-third of the viral genome (i.e., a TRS ‘body’ or TRS-B). The synthesis of the negative-strand RNA is then re-initiated at the TRS in the leader sequence (TRS-L) at 70 nucleotides from the 5′ end of the genome, due to the interaction between TRS-B of the negative-sense nascent RNA and TRS-L of the positive-sense gRNA. Upon re-initiation of RNA synthesis at the TRS-L region, a negative-strand copy of the leader sequence is added to the nascent RNA to complete the synthesis of negative-strand sgRNAs. These fused negative-strand intermediates are used as templates to synthesize positive-sense sgRNAs that are translated into both structural and accessory proteins [
3]. In addition, in a global landscape analysis of SARS-CoV-2 subgenome RNA expression, Wang et al., 2021 [
4] used computational analysis to identify novel modes of viral sgRNA biogenesis via a ‘TRS-independent’ mechanism.
To date, eight main sgRNAs have been reported to be produced in SARS-CoV-2-infected cells. In addition to these canonical sgRNAs, noncanonical RNA products of discontinuous transcription have also been reported for SARS-CoV-2, including fusions of the 5′ leader sequence to unexpected 3′ sites, TRS-L-independent long-distance fusions, and local fusions that result in small deletions mainly in structural and accessory genes [
1].
Of interest, the N protein sgRNA (sgN; which codes for the N protein) is the most abundant sgRNA during viral infection, mostly due to the low DG value in the duplex formation between TRS-L and TRS-B [
2]. On this basis, of all the sgRNAs, sgN has been shown to be the most abundant in SARS-CoV-2-infected cells [
5]. Furthermore, together with sgRNA for Orf7a, the same sgN also shows highest abundance in swab samples from COVID-19-affected patients [
6]. Another important finding related to the SARS-CoV-2 N protein encoded by the sgN transcript is its role in the regulation of the discontinuous transcription process through its C-terminal domain, which retains its interaction with TRS sequences, and the consequential regulation of transcription [
7]. To this end, early and continuous expression of the sgN transcript ensures the generation of the N protein in multiple copies in nascent viral genome particles, which is of great importance for SARS-CoV-2 replication and propagation.
As viral sgRNAs are transcribed in infected cells but are not packaged together with gRNAs into nascent virions, they might be useful indicators of the presence of active infection [
8]. However, to date, the literature data are contradictory on which sgRNAs might represent indicators of viral status. The detection of sgRNA for the E gene (i.e., sgE) has been shown in oro-pharyngeal throat swabs samples collected from days 4 to 9 after COVID-19 symptom onset. The authors would thus suggest that sgE can be used as an indicator of active SARS-CoV-2 infection [
9]. An additional study reported that the detection of sgRNAs was possible up to 11 and 17 days after first detection of SARS-CoV-2 infection through PCR and next-generation sequencing, respectively [
6]. Therefore, this study concluded that sgRNAs cannot be used as indicators of active CoV replication/infection, arguing that this might reflect the methodology used. Indeed, to date, the RNAs evaluated in diagnostic swab samples are likely to be a mixture of both gRNAs and sgRNAs. Alexandersen et al. [
6] concluded that these sgRNAs are protected from nuclease actions and hydrolysis by “double-membrane vesicles”, and they concluded that this is the main reason why sgRNAs survive longer in cells. In another study performed on in vivo infection of rhesus macaques, Dagotto et al. [
10] investigated whether an sgRNA assay can distinguish an input challenge virus from an actively replicating virus in vivo, through a comparison of the expression of both sgE and the standard N and E proteins (both at the level of gRNA and sgRNA detection). In summary, they suggested the use of sgRNAs to monitor the actively replicating virus in prophylactic and therapeutic SARS-CoV-2 studies during rhesus macaques’ infection. In our previous study, we showed that sgN detection might provide a better candidate biomarker for active and higher viral loads in SARS-CoV-2-infected patients than sgE. Of interest, sgN expression was not influenced by the expression of genomic transcription of the N gene [
11]. In a recent study, Oranger et al., 2021 [
12] used fine-tuned droplet digital PCR to show that both sgN and sgS directly correlate to gRNA copies, and then that the sgN and sgS expression levels are reduced in RNA samples with low viral RNA content. This thus indicated that the samples analyzed were mainly characterized by residual genomic SARS-CoV-2 material with little or no active viral transcription.
On the basis of our previous results, to determine whether sgN detection can be used as a marker of active viral replication, we undertook a multicenter study here across five Diagnostic Coronet centers in Italy, which included 315 oro/nasopharyngeal swabs of COVID-19-positive patients. In this report, we define the limit of detection of sgN, sgE, and Orf1b, and we underline that this thus precedes patient negativization in home-isolated and hospitalized COVID-19-positive patients (by ~3/7 days from the first swab, respectively). We have also designed a new kit for detection of sgN, gene E, gene ORF1ab, and RNAse P gene that can detect their levels in oro/nasopharyngeal swabs and bronchial aspirates samples. SARS-CoV-2 life-cycle has been previously reported to trigger cell–cell fusion mechanisms, thus orchestrating the formation of multinucleated cells [
13,
14]. We then show that targeting the sgN sequence 2’-
O-methyl antisense RNA can impair viral replication and syncytia formation in human cells (i.e., HEK-293T cells overexpressing ACE2) upon infection with the VOC Alpha B.1.1.7-SARS-CoV-2 variant. Sequence analysis of the VOC variants shows that the sequences match to 100% identity to Beta and Gamma and 97.3% to Omicron, thus enhancing our hopes and findings. Altogether, these analyses indicate future therapeutic implications to target N protein synthesis to inhibit SARS-CoV-2 viral replication.
3. Discussion
SgRNAs are only produced during active infection to generate the N protein units to cover the nascent SARS-CoV-2 RNA genome that will be encapsulated in new virus particles, and that thus presents an accurate measure of replicating virus. The N protein, which is the only protein present in the coronavirus nucleocapsid, plays a critical role in ensuring coronavirus replication and successful intracellular lifecycle, thus it is considered a suitable target when designing new vaccines together with S-RBD protein [
22,
23]. At this time, a question can be raised on why do we observe a loss of sgN before the other markers during the time of clinical virus negativization? We thus think that this is due to the importance of sgN on supplying N protein for generating intact new genome RNA copies of the viral particles once assembled, hence underlying its functional importance on sustaining the viral stability and potency. In this case, missing N protein, as observed on measuring sgN RNA copies loss, supports the hypothesis that virus replication and infection capability is diminishing as a sign of negativization. At this time, we cannot exclude that sgN RNA, although present in a larger copy number during viral RNA genome replication (see data presented by Nanopore sequencing, BioProject PRJNA688696, [
5]) would be more sensible to RNA instability in comparison with other target viral mRNA genes. Future laboratory settings will address these hypotheses. The routine diagnostic tools based on RT-PCR assays typically target total or genomic SARS-CoV-2 RNA, and are thus not an optimal measure of newly replicating active virus. Here, we have developed a new Taqman-based diagnostic assay that can detect the expression of the SARS-CoV-2 sgN transcript together with viral gene E, gene ORF1ab, and human RNAse P gene. These data demonstrate the potential of measuring sgN transcripts rather than gRNA as a more specific measure of the replicating virus in samples with higher viral load. In this regard, sgN was not detectable in oro/nasopharyngeal swabs from COVID-19-affected patients showing Cq values >33.163 for viral gene E (
p < 0.0001;
Figure 1A), and >33.155 for gene ORF1ab (
p < 0.0001;
Figure 1B) genes. Of importance, this method has also been validated here in bronchial aspirate specimens (
Supplementary Figure S1A).
We further shown the ‘prognostic’ value of sgN detection in hospitalized (Intensive Care Unit) and home-isolated COVID-19-positive patients. Indeed, the SARS-CoV-2 Viral3 diagnostic kit was compared with the commercially available Allplex 2019-nCoV assay to monitor the time to reach negative results for the SARS-CoV-2 biomarkers, as a sign of benign disease and recovery from disease (follow-up: 3–7 days home isolated, 1–2 weeks hospitalized patients) through detection of viral sgN, gene E, gene ORF1ab, gene N, and human RNase P gene. The data presented here show that sgN is the first transcript that becomes undetectable during the recovery in both hospitalized and isolated COVID-19-affected patients (
Figure 2). These results suggest that sgN loss can be considered as a ‘predictive marker’ for lower SARS-CoV-2 replication activity, thus being of importance for both monitoring the therapeutic response and alerting clinicians that the SARS-CoV-2 negativization processes is underway.
Developing prophylactic actions for the SARS-CoV-2 virus is a public health priority. One of the most important actions in the diagnostic SARS-CoV-2 setting is to define when the diminishing viral load can be considered a benign phase (i.e., viral negativization). Our method of detection of sgN has a specificity of ≥99.9% and sensitivity for sgN, gene E, and gene ORF1ab of 300,000 to 30 viral copies, with hit rate of 95%. This positions the SARS-CoV-2 Viral3 kit as a valuable tool for early detection and negativization, and consequently to estimate the infectiousness of SARS-CoV-2 patients world-wide.
Viral structural proteins are essential for virus survival and propagation. These SARS-CoV-2 structural proteins (i.e., S, E, M, N proteins) are encoded by the SARS-CoV-2 sgRNAs that are also responsible for the synthesis of several other accessory proteins (i.e., 3a, 6, 7a, 7b, 8, 10). N protein encapsulates viral genomic RNAs during the viral life cycle to protect the genome and co-enter the host cell with the viral genomic RNAs, which indicates that N is essential for viral RNA replication, particularly at the initiation stage. SgRNAs are expressed in abundance [
4,
6], and among them, sgN has been shown to be the most abundant in SARS-CoV-2-infected cells [
1,
5]. To date, a siRNA-based approach that targets the leader sequence of SARS-CoV [
24] and a chemical inhibitor that targets the RNA-binding affinity of N protein [
25] have been tested in vitro only against SARS-CoV infection. The effectiveness of these therapeutic approaches against SARS-CoV-2 have not been tested to date. Further, there are no prophylactic treatments available. Here, we used sense and antisense 2′-O-methyl RNA oligonucleotides that specifically target the TRS sequence (following the leader sequence) at the 5′ end of the of N gene in B.1.1.7 SARS-CoV-2-infected HEK-293T cells overexpressing ACE2. Through qPCR assays, immunoblotting, and high-resolution immunofluorescence, we have shown that sense 2’-
O-methyl RNA oligonucleotide reduces sgN transcripts, and, as a consequence, N protein levels, and therefore inhibits SARS-CoV-2 infection and replication. Furthermore, N protein has been shown to have a critical role during the discontinuous transcription process as a positive regulator that influences the expression of the other sgRNAs (Yang et al. 2021). The results presented here indeed show that lowering the expression of sgN results in inhibition of the gRNA E and sgRNA E transcripts (
Figure 3E), which probably occurs via competition for the viral replication machinery.
In the future, it would appear reasonable to explore other sgRNAs in similar in vitro therapeutic assays. Of importance, we envision the use of technologies based on nanoparticles and stable nucleic acid lipid particles for delivery of 2′-
O-methyl antisense RNA in human cells infected with SARS-CoV-2 [
26], as proof-of-principle of the use of therapeutic nanoparticles. Together with an aerosol application formula, this would be of great value for the treatment of patients with respiratory failure [
5], and especially for individuals who cannot mount an immune response to the vaccines (e.g., immunosuppressed, under transplants) and thus requiring a prophylactic to prevent infection after being exposed to SARS-CoV-2. Additionally, by analyzing the 2′-
O-methyl antisense RNA sequence, we found 100% identity to VOC Beta and Gamma, 91.8% to VOC Delta, 97.3% to VOC Omicron, and 81.0% to VOC Alpha (
Figure 3B). This thus suggests further that 2′-
O-methyl antisense RNA can also act against the other VOC variants with a greater percentage identity as the VOC Alpha used here (
Figure 3C–E and
Figure 4A,B). Further studies should address its therapeutic usefulness for the other VOC variants.
Nevertheless, we should consider at this time that the canonical “leader to body fusion model” is not unique, and that others have been proposed (e.g., TRS-independent mechanism, multi-switch sgRNA synthesis), and thus other 2′-
O-methyl antisense RNAs with different switch junctions can be designed, as identified by [
4,
27].
One of the most common phenomena in SARS-CoV-2 infection is related to syncytia formation [
17,
28]. Syncytia can also be induced by certain types of infections by viruses, such as human immunodeficiency virus, respiratory syncytial virus, and herpes simplex virus [
20]. One of the most accredited models of syncytia formation is that this virus-induced cell fusion serves to facilitate the transfer of viral genomes to the neighboring cells, thus enhancing viral propagation [
21]. Here, we showed significant inhibition of syncytia formation (
p < 0.01) in those cells overexpressing the 2′-
O-methyl antisense RNA sgN sequence using SARS-CoV-2 VOC Alpha infected human cells (
Figure 4A). This is a further evidence that inhibition of N protein translation, and consequentially the transcription of the other proteins (e.g., S protein) [
7], negatively affects viral replication via the syncytia model of action.
4. Materials and Methods
4.1. Cell Culture
HEK-293T cells (CRL-3216, ATCC, Manassas, Virginia, USA), HEK-293T stable clones overexpressing human ACE2 (HEK293T-ACE2) and Vero E6 cells (C1008, ATCC) were grown in a humidified 37 °C incubator with 5% CO2. The cells were cultured in feeder-free conditions using Dulbecco’s modified Eagle’s medium (41966-029; Gibco, Thermo Fisher Scientific, Waltham, MA, USA) with 10% fetal bovine serum (10270-106; Gibco), 2 mM L-glutamine (25030-024; Gibco), and 1% penicillin/streptomycin (P0781; Sigma-Aldrich), with medium changed daily. Cells were dissociated with Trypsin-EDTA solution (T4049, Sigma-Aldrich, St. Louis, MO, USA) when the culture reached ~80% confluency.
4.2. Generation of HEK293T-ACE2 Stable Clones
HEK-293T cells were plated in 6-well plates in 2 mL of Dulbecco’s modified Eagle’s medium (41966-029; Gibco) with 10% fetal bovine serum (10270-106; Gibco). When the culture reached ~70% confluency, they were transfected with pCEP4-myc-ACE2 plasmid (#141185, Addgene) with X-tremeGENE 9 DNA Transfection Reagent (06365779001; Sigma-Aldrich). Briefly, X-tremeGENE 9 DNA Transfection Reagent was equilibrated at room temperature (+15 to +25 °C) and diluted with serum-free Dulbecco’s modified Eagle’s medium (41966-029; Gibco) to a concentration of 3 μL reagent/100 μL medium. Then, 1 μg of DNA plasmid was added to 100 μL of diluted X-tremeGENE 9 DNA Transfection Reagent (3:1 ratio [μL]). The transfection reagent:DNA complex was then incubated for 15 min at room temperature. Finally, the transfection complex was added to the cells in a dropwise manner. Following forty-eight hours from transfection, the cell culture medium was changed, and the cells’ clones were selected using 800 μg/mL hygromycin.
4.3. Transient Transfection with 2′-O-Methyl RNA Oligos Targeting sgN
4.3.1. 2′-O-Methyl RNA Oligos Design
The following 2’-O-methyl RNA oligonucleotides were designed against the junction site between the leader sequence, the transcription-regulating sequences (TRSs) and the 5′ end of gene N (leader-“junction”-TRS-“junction”-N) of SARS-CoV-2:
Sense: TCTCTAAACGAACAAACTAAAATGTCTGATAATGGAC
Antisense: GTCCATTATCAGACATTTTAGTTTGTTCGTTTAGAGA
To assess the initiation on the different viral transcripts, and enable leader–junction site unique alignment, we used RNA Nanopore sequencing data obtained from previously SARS-CoV-2 infected (20A clade) Vero E6 monkey kidney cells [
5]. This sequence was further aligned with all of the SARS-CoV-2 variants identified to date, and was confirmed by Sanger sequence analysis from a swab sample from a COVID-19-affected patient. These analyses allowed the determination of the best match sequence junction between the leader, TRS-B and the subgenomic N transcript (sgN). The 2’-
O-methyl RNA oligonucleotides were synthetized at the DNA lab Facility at CEINGE, Biotecnologie Avanzate (Naples).
4.3.2. 2’-O-Methyl RNA Oligos Targeting sgN Transfection in HEK293T-ACE2 Cells before Infection with SARS-CoV-2
HEK293T-ACE2 cells were transfected with 1 μg of sense or antisense 2’-O-methyl RNA oligonucleotides against SgN. Transient transfections were performed with X-tremeGENE 9 DNA Transfection Reagent (06365779001; Sigma-Aldrich). To this end, X-tremeGENE 9 DNA Transfection Reagent was equilibrated at room temperature and diluted with serum-free Dulbecco’s modified Eagle’s medium (41966-029; Gibco) (3 μL reagent/100 μL medium). Then, 1 μg of sense or antisense 2′-O-methyl RNA oligonucleotides were added to 100 μL of diluted X-tremeGENE 9 DNA Transfection Reagent (3:1 ratio [μL]). The transfection reagent:RNA complex was incubated for 15 min at room temperature. The transfection complex was then added to the cells in a dropwise manner. Twelve hours after transfection, the cell culture medium was changed, and the cells were infected with SARS-CoV-2 particles (GISAID accession number: EPI_ISL_736997). Forty-eight hours after transfection (i.e., after 36 h of infection), the HEK293T-ACE2 cells were lysed.
4.4. SARS-CoV-2 Isolation and Infection
SARS-CoV-2 was isolated from a nasopharyngeal swab obtained from an Italian patient sample as previously described [
5]. Briefly, Vero E6 cells (8 × 105) were trypsinized and resuspended in Dulbecco’s modified Eagle’s medium (41966-029; Gibco) with 2% FBS in T25 flasks, to which the clinical specimen (100 μL) was added. The inoculated cultures were grown in a humidified 37 °C incubator with 5% CO
2. Seven days after infection, when cytopathic effects were observed, the cell monolayers were scrapped with the back of a pipette tip, while the cell culture supernatant containing the viral particles was aliquoted and frozen at −80 °C. Viral lysates were used for total nucleic acid extraction for confirmatory testing and sequencing (GISAID accession number: EPI_ISL_736997).
HEK-293T cell clones stably overexpressing ACE2 (8 × 105 cells) were plated in T25 flasks for transfection with sense or antisense 2’-O-methyl RNA oligos targeting sgN. After 12 h, the cell culture medium was changed, and the 2’-O-methyl RNA transfected cells were then infected with viral particles of the 20I/501Y.V1 (B.1.1.7) clades (0.03 MOI), (GISAID accession number: EPI_ISL_736997). Uninfected cells were used as the negative control. After 36 h of infection, the cells were lysed or fixed. These experiments were performed in a BLS3-authorized laboratory.
4.5. RNA Extraction and qPCR from Oro/Nasopharyngeal Swabs from Patients
Oro/nasopharyngeal swab samples (200 μL) were taken for RNA extraction using nucleic acid extraction kits (T-1728; ref: 1000021043; MGI tech) with automated procedures on a high-throughput automated sample preparation system (MGISP-960; MGI Tech), following the manufacturer’s instructions. RNA samples (5 μL) were used to perform qPCR with the SARS-CoV-2 Viral3 kit (BioMol laboratories) and the IVD-approved Allplex 2019-nCoV assay (Seegene).
Allplex 2019-nCoV assay for viral E, RdRP, and N gene detection. This diagnostic kit provides a specific quantitative detection of the viral E, RdRP, and N gene (from the SARS-CoV-2) by using differentially labeled target probes into two mixes (E, FAM; RdRp, Cal Red 610; N: Quasar 670) N2, VIC; Second mix: N3, VIC; RNase P, CY5). These runs were performed by using 5 μL RNA on a PCR machine (CFX96; BioRad; in vitro diagnostics IVD approved) under the following conditions:
- ○
Reverse transcription: 50 °C for 20 min;
- ○
Denaturation: 95 °C for 15 min;
- ○
Denaturation and annealing (×44 cycles): [95 °C for 10 s; 60 °C for 15 s, and 72 °C for 10 s].
SARS-CoV-2 Viral3 kit for viral sgN, gene E, gene Orf1ab and human RNAse P detection. These runs were performed using 5 μL RNA on a PCR machine (CFX96; BioRad; in vitro diagnostics IVD approved) under the following conditions:
- ○
UNG incubation: 25 °C for 2 min;
- ○
Reverse transcription: 50 °C for 15 min;
- ○
Inactivation/denaturation: 95 °C for 3 min;
- ○
Denaturation and annealing (for 44 cycles): [95 °C for 3 s and 60 °C for 45 s].
SARS-CoV-2 Viral3 kit was produced by following CDC 2019-Novel Coronavirus Real-Time RT-PCR Diagnostic Panel and WHO-technical-guidance for oligo sequences (Sequences MT810943.1. and NM_001104546.2). The details of the primers used in these assays (SARS-CoV-2 Viral3 kit) are provided below:
sgN Forward: CAACCAACTTTCGATCTCTTGTA
sgN Reverse: TCTGCTCCCTTCTGCGTAGA
sgN Probe: 5′-FAM-ACTTCCTCAAGGAACAACATTGCCA-BBQ1-3′
Orf1ab Forward: CCCTGTGGGTTTTACACTTAA
Orf1ab Reverse: ACGATTGTGCATCAGCTGA
Orf1ab Probe:5’-ROX- CCGTCTGCGGTATGTGGAAAGGTTATGG-BBQ2-3’
E Forward: ACAGGTACGTTAATAGTTAATAGCGT
E Reverse: ATATTGCAGCAGTACGCACACA
E Probe: 5’ CY5-ACACTAGCCATCCTTACTGCGCTTCG BBQ2-3’
RNAse P Forward: ATGGCGGTGTTTGCAGATTT
RNAse P Reverse: AGCAACAACTGAATAGCCAAGG
RNAse P Probe: 5′-HEX-TTCTGACCTGAAGGCTCTGCGCG-BHQ1-3′
Taqman assays for viral sgE and human β-Actin (ACTB) detection. Reverse transcription was performed with SuperScript IV VILO Master Mix (11756500, Invitrogen, Carlsbad, CA, USA), following the manufacturer’s instructions. The reverse transcription products (cDNA) were amplified by qRT-PCR using an RT-PCR system (7900; Applied Biosystems, Foster City, CA, USA). The cDNA preparation was through the cycling method by incubating the complete reaction mix as follows:
- ○
cDNA reactions: [25 °C for 5 min and 42 °C for 30 min]
- ○
Heat-inactivation: 85 °C for 5 min
- ○
Hold stage: 4 °C
The target sgE and ACTB were detected with Taqman approach [
9,
11]. These runs were performed on a PCR machine (Quantstudio 12K Flex, Applied Biosystems, Waltham, MA, USA) with the following thermal protocol:
- ○
Denaturation Step: 95 °C for 20 s;
- ○
Denaturation and annealing (×50 cycles): [95 °C for 1 s and 60 °C for 20 s].
The details of the primers used in these Taqman assays are provided below:
sgE Forward (Taqman) [
9,
11]: CGATCTCTTGTAGATCTGTTCTC
sgE Reverse (Taqman) [
9,
11]: ATATTGCAGCAGTACGCACACA
sgE probe (Taqman) [
9,
11]: 5′-FAM-ACACTAGCCATCCTTACTGCGCTTCG-BBQ-3′
ACTB Forward (Taqman): Hs01060665_g1 (Thermo Fisher Scientific, Waltham, Massachusetts, USA)
The quantification cycle (Cq) values of sgN and sgE are reported as means ± SD normalized to the control (human ACTB) of three replicates.
4.6. RNA Extraction and qPCR from HEK-293T Cells Overexpressing ACE2
RNA samples were extracted with TRIzol RNA isolation reagent according to the manufacturer’s instructions. Reverse transcription was performed with 5× All-In-One RT MasterMix (catalog no. g486; ABM), according to the manufacturer’s instructions. The reverse transcription products (cDNA) were amplified by qRT-PCR using an RT-PCR system (7900; Applied Biosystems, Foster City, CA, USA). The cDNA preparation was through the cycling method by incubating the complete reaction mix as follows:
- ○
cDNA reactions: [25 °C for 5 min and 42 °C for 30 min];
- ○
Heat-inactivation: 85 °C for 5 min;
- ○
Hold stage: 4 °C.
Viral N and human RNAse P detection. The detection of viral N gene and human RNase P was performed using the in vitro diagnostics IVD-approved “Quanty COVID-19” kit (RT-25; Clonit). This diagnostic kit provides a specific quantitative detection of the viral N1, N2, and N3 fragments (from the SARS-CoV-2 N gene) and human RNaseP gene by using differentially labeled target probes into two mixes (First mix: N1, FAM; N2, VIC; Second mix: N3, VIC; RNase P, CY5). These runs were performed on a PCR machine (CFX96; Bio-Rad; IVD approved) according to the manufacturer’s instructions. Briefly, 5 μL RNA was used for the following thermal protocol below:
- ○
UNG incubation: 25 °C for 2 min;
- ○
Reverse transcription: 50 °C for 15 min;
- ○
Inactivation/denaturation: 95 °C for 2 min;
- ○
Denaturation and annealing (×45 cycles): [95 °C for 3 s and 55 °C for 30 s].
The quantification cycle (Cq) values of N1, N2, and N3 are reported as means ±SD normalized to the internal control (human RNase P) of three replicates.
Viral E, human ACE2, and β-Actin (ACTB) detection (SYBR green). The targets E, ACE2, and ACTB were detected with SYBR green approach by using BrightGreen 2X qRT-PCR MasterMix Low-ROX (MasterMix-LR; ABM). Human ACTB was used as the housekeeping gene used to normalize the quantification cycle (Cq) values of the other genes. These runs were performed on a PCR machine (Quantstudio5, Lifetechnologies) with the following thermal protocol:
- ○
Hold stage: 50 °C for 2 min;
- ○
Denaturation step: 95 °C for 10 min;
- ○
Denaturation and annealing (×45 cycles): [95 °C for 15 s and 60 °C for 60 s];
- ○
Melt curve stage: [95 °C for 15 s, 60 °C for 1 min, and 95 °C for 15 s].
The details of the primers used in these SYBR green assays are provided below:
E Forward (SYBR green) [
5]: ACAGGTACGTTAATAGTTAATAGCGT
E Reverse (SYBR green) [
5]: ATATTGCAGCAGTACGCACACA
ACE2 Forward (SYBR green) [
5]: GAAATTCCCAAAGACCAGTGGA
ACE2 Reverse (SYBR green) [
5]: CCCCAACTATCTCTCGCTTCAT
ACTB Forward (SYBR green) [
5]: GACCCAGATCATGTTTGAGACCTT
ACTB Reverse (SYBR green) [
5]: CCAGAGGCGTACAGGGATAGC
The relative expression of the target genes was determined using the 2−ΔΔCq method, as the fold increase compared with the controls. The data are presented as means ± SD of the 2−ΔΔCq values (normalized to human ACTB) of three replicates.
Viral sgN, sgE and human β-Actin (ACTB) detection (Taqman). The target sgN, sgE, and ACTB were detected with the Taqman approach [
5]. These runs were performed on a PCR machine (Quantstudio 12K Flex, Appliedbiosystems) with the following thermal protocol:
- ○
Denaturation Step: 95 °C for 20 s;
- ○
Denaturation and Annealing (×50 cycles): [95 °C for 1 s and 60 °C for 20 s].
The details of the primers used in these Taqman assays are provided below:
sgN Forward (Taqman) [
11]: CAACCAACTTTCGATCTCTTGTA
sgN Reverse (Taqman) [
11]: TCTGCTCCCTTCTGCGTAGA
sgN Probe (Taqman) [
11]: 5′FAM-ACTTCCTCAAGGAACAACATTGCCA-BBQ-3′
sgE Forward (Taqman) [
9,
11]: CGATCTCTTGTAGATCTGTTCTC
sgE Reverse (Taqman) [
9,
11]: ATATTGCAGCAGTACGCACACA
sgE probe (Taqman) [
9,
11]: 5′-FAM-ACACTAGCCATCCTTACTGCGCTTCG-BBQ-3′
ACTB Forward (Taqman): Hs01060665_g1 (Thermo Fisher Scientific)
The quantification cycle (Cq) values of sgN and sgE are reported as means ± SD normalized to the control (human ACTB) of three replicates.
4.7. Sanger Sequencing
The cDNA was obtained by random primer RT-PCR using SensiFASTcDNA synthesis kits (Bioline, provided by Life Technologies Italia, Monza, MB, Italy), using 5 μL RNA extracted from nasopharyngeal swabs. We used the primer setting (sgN-For AAAC- CAACCAACTTTCGATCTCTTGTA and sgN-Rev TCTGGTTACTGCCAGTTGAATC) to amplify the sgN region, and to perform the Sanger sequencing.
4.8. Immunoblotting
Cells were lysed in 20 mM sodium phosphate, pH 7.4, 150 mM NaCl, 10% (v/v) glycerol, 1% (w/v) sodium deoxycholate, 1% (v/v) Triton X-100, supplemented with protease inhibitors (Roche). The cell lysates were cleared by centrifugation at 16,200× g for 30 min at room temperature, and the supernatants were removed and assayed for protein concentrations with protein assay dye reagent (Bio-Rad Laboratories, Berkeley, CA, USA). The cell lysates (20 μg) were resolved on 10% SDS-PAGE gels. The proteins were transferred to PVDF membranes (Millipore). After 1 h in blocking solution with 5% (w/v) dry milk fat in Tris-buffered saline containing 0.02% [v/v] Tween-20, the PVDF membranes were incubated with the primary antibody overnight at 4 °C: anti-ACE2 (1:1000; ab15348), anti-SARS-CoV-2 N protein (1:250; 35-579; ProSci Inc., Poway, San Diego, CA, USA), or anti-β-actin (1:10,000; A5441; Sigma-Aldrich, St. Louis, MO, USA). The membranes were then incubated with the required secondary antibodies for 1 h at room temperature: secondary mouse or rabbit horseradish-peroxidase-conjugated antibodies (NC 15 27606; ImmunoReagents, Inc.), diluted in 5% (w/v) milk in TBS-Tween. The protein bands were visualized by chemiluminescence detection (Pierce-Thermo Fisher Scientific Inc., Rockford, IL, USA). Densitometry analysis was performed with the ImageJ software. The peak areas of the bands were measured on the densitometry plots, and the relative proportions (%) were calculated. Then, the density areas of the peaks were normalized with those of the loading controls, and the ratios for the corresponding controls are presented as fold-changes. Immunoblotting was performed in triplicate. The densitometry analyses shown were derived from three independent experiments.
4.9. Immunofluorescence
SARS-CoV-2-infected HEK293T-ACE2 cells were fixed in 4% paraformaldehyde in phosphate-buffered saline (PBS) for 30 min, washed three times with PBS, and permeabilized for 15 min with 0.1% Triton X-100 (215680010; Acros Organics, Thermo Fisher Scientific, Waltham, MA, USA) diluted in PBS. The cells were then blocked with 3% bovine serum albumin (A9418; Sigma-Aldrich, St. Louis, MO, USA) in PBS for 1 h at room temperature. The samples were incubated with the appropriate primary antibodies overnight at 4 °C: anti-ACE2 (1:1000; ab15348; Abcam. Cambridge, UK) or anti-SARS S protein (1:100; ab272420; Abcam). After washing with PBS, the samples were incubated with the secondary antibody at room temperature for 1 h: anti-mouse Alexa Fluor 488 (1:200; ab150113; Abcam) or anti-rabbit Alexa Fluor 647 (1:200; ab150075; Abcam). DNA was stained with DAPI (1:1000; #62254; Thermo Fisher). The slides were washed and mounted with cover slips with 50% glycerol (G5150; Sigma-Aldrich). Microscopy images were obtained using the Elyra 7 platform (Zeiss) with the optical Lattice SIM2 technology (with the ZEN software, Zeiss, blue edition), using the 63× oil immersion objective.
4.10. Statistical Analysis
Statistical significance was defined as p < 0.05 by unpaired two-tailed student’s t-tests. All of the data are given as means ± SD. In the Figures, statistical significance is represented as follows: * p < 0.05, ** p < 0.01, and *** p < 0.001.
For the determination of the limit of detection (cut-off) for sgN detection, one-way analysis of variance (ANOVA) was used through IBM SPSS Statistics. Briefly, the samples were stratified into three groups according to the Cq values of sgN. The first group consisted of those samples where the Cq t value for sgN was below the median value (i.e., 30.51; 99 samples). The second group of samples were characterized by Cq values of sgN ranging from the Cq median value (30.51) to 40 (96 samples). The third group comprised the samples in which sgN was not detectable (i.e., Cq > 40; 120 samples). All the experiments were performed in triplicate.