Metagenomic Sequencing for Microbial DNA in Human Samples: Emerging Technological Advances
Abstract
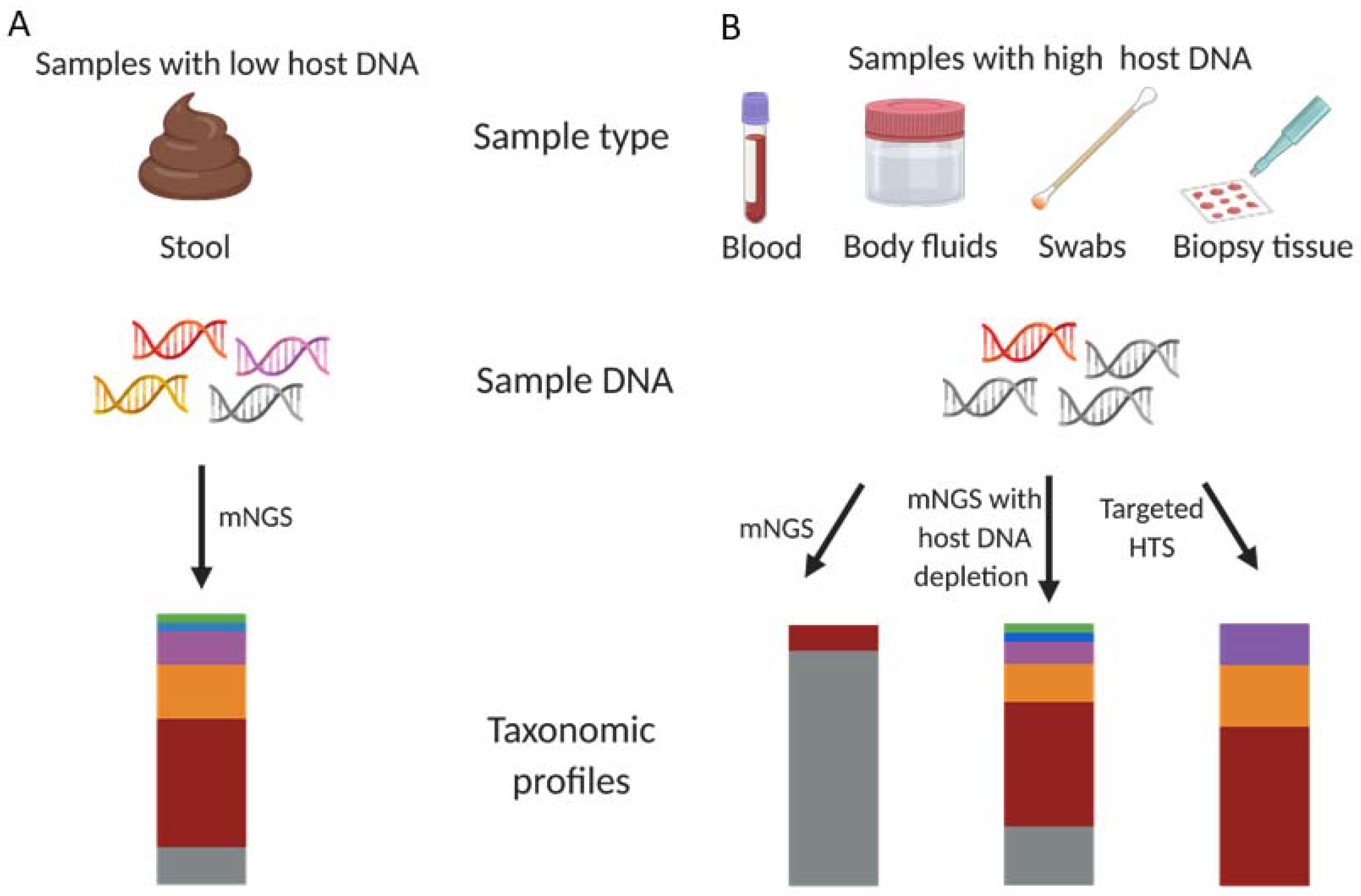
:1. Introduction
2. The Growing Need for Microbial DNA Enrichment Prior to Metagenomic Sequencing
3. Current Approaches to Host DNA Depletion
3.1. Removal of the Host Cells before DNA Extraction
3.2. Separating the Microbial DNA from the Host Background
3.3. Limitation and Controversy
4. Application in Microbiome Research and Clinical Metagenomics
5. New Strategies to Facilitate Metagenomic Sequencing in Samples with Overabundant Host DNA
5.1. The Removal of Unwanted High Abundance Species in Sequencing Libraries
5.2. Selective Sequencing
6. Conclusions and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Chiu, C.Y.; Mille, S.A. Clinical metagenomics. Nat. Rev. Genet. 2019, 20, 341–355. [Google Scholar] [CrossRef] [PubMed]
- Quick, J.; Grubaugh, N.D.; Pullan, S.T.; Claro, I.M.; Smith, A.D.; Gangavarapu, K.; Oliveira, G.; Robles-Sikisaka, R.; Rogers, T.F.; Beutler, N.A.; et al. Multiplex PCR method for MinION and Illumina sequencing of Zika and other virus genomes directly from clinical samples. Nat. Protoc. 2017, 12, 1261–1276. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Metsky, H.C.; Siddle, K.J.; Gladden-Young, A.; Qu, J.; Yang, D.K.; Brehio, P.; Goldfarb, A.; Piantadosi, A.; Wohl, S.; Carter, A.; et al. Capturing sequence diversity in metagenomes with comprehensive and scalable probe design. Nat. Biotechnol. 2019, 37, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Gao, X.; Meng, J.; Zhang, A.; Zhou, Y.; Long, M.; Li, B.; Deng, W.; Jin, L.; Zhao, S.; et al. Metagenomic analysis of bacteria, fungi, bacteriophages, and helminths in the gut of giant pandas. Front. Microbiol. 2018, 9, 1717. [Google Scholar] [CrossRef] [Green Version]
- Johnson, J.S.; Spakowicz, D.J.; Hong, B.Y.; Petersen, L.M.; Demkowicz, P.; Chen, L.; Leopold, S.R.; Hanson, B.M.; Agresta, H.O.; Gerstein, M.; et al. Evaluation of 16S rRNA gene sequencing for species and strain-level microbiome analysis. Nat. Commun. 2019, 10, 5029. [Google Scholar] [CrossRef] [Green Version]
- Di Cenzo, G.C.; Finan, T.M. The divided bacterial genome: Structure, function, and evolution. Microbiol. Mol. Biol. Rev. 2017, 81, e00019-17. [Google Scholar] [CrossRef] [Green Version]
- Wilson, M.R.; Sample, H.A.; Zorn, K.C.; Arevalo, S.; Yu, G.; Neuhaus, J.; Federman, S.; Stryke, D.; Briggs, B.; Langelier, C.; et al. Clinical metagenomic sequencing for diagnosis of meningitis and encephalitis. N. Engl. J. Med. 2019, 380, 2327–2340. [Google Scholar] [CrossRef]
- The Human Microbiome Project Consortium. A framework for human microbiome research. Nature 2012, 486, 215–221. [Google Scholar] [CrossRef] [Green Version]
- Simner, P.J.; Miller, S.; Carroll, K.C. Understanding the promises and hurdles of metagenomic next-generation sequencing as a diagnostic tool for infectious diseases. Clin. Infect. Dis. 2018, 66, 778–788. [Google Scholar] [CrossRef] [Green Version]
- Zhang, C.; Hu, T.; Xiu, L.; Li, Y.; Peng, J. Use of ultra-deep sequencing in a patient with tuberculous coxitis shows its limitations in extrapulmonary tuberculosis diagnostics: A case report. Infect. Drug Resist. 2019, 12, 3739–3743. [Google Scholar] [CrossRef] [Green Version]
- Marotz, C.A.; Sanders, J.G.; Zuniga, C.; Zaramela, L.S.; Knight, R.; Zengler, K. Improving saliva shotgun metagenomics by chemical host DNA depletion. Microbiome 2018, 6, 42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shi, X.; Shao, C.; Luo, C.; Chu, Y.; Wang, J.; Meng, Q.; Yu, J.; Gao, Z.; Kang, Y. Microfluidics-based enrichment and whole-genome amplification enable strain-level resolution for airway metagenomics. Msystems 2019, 4, e00198-19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liang, G.; Bushman, F.D. The human virome: Assembly, composition and host interactions. Nat. Rev. Microbiol. 2021, 19, 514–527. [Google Scholar] [CrossRef] [PubMed]
- Bal, A.; Pichon, M.; Picard, C.; Casalegno, J.S.; Valette, M.; Schuffenecker, I.; Billard, L.; Vallet, S.; Vilchez, G.; Cheynet, V.; et al. Quality control implementation for universal characterization of DNA and RNA viruses in clinical respiratory samples using single metagenomic next-generation sequencing workflow. BMC Infect. Dis. 2018, 18, 537. [Google Scholar] [CrossRef]
- Hasan, M.R.; Rawat, A.; Tang, P.; Jithesh, P.V.; Thomas, E.; Tan, R.; Tilley, P. Depletion of human DNA in spiked clinical specimens for improvement of sensitivity of pathogen detection by next-generation sequencing. J. Clin. Microbiol. 2016, 54, 919–927. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yeoh, Y.K. Removing host-derived DNA sequences from microbial metagenomes via mapping to reference genomes. Methods Mol. Biol. 2021, 2232, 147–153. [Google Scholar] [CrossRef]
- Fong, W.; Rockett, R.; Timms, V.; Sintchenko, V. Optimization of sample preparation for culture-independent sequencing of Bordetella pertussis. Microb. Genom. 2020, 6, e000332. [Google Scholar] [CrossRef]
- Zelenin, S.; Hansson, J.; Ardabili, S.; Ramachandraiah, H.; Brismar, H.; Russom, A. Microfluidic-based isolation of bacteria from whole blood for sepsis diagnostics. Biotechnol. Lett. 2015, 37, 825–830. [Google Scholar] [CrossRef]
- Anscombe, C.; Misra, R.V.; Gharbia, S. Whole genome amplification and sequencing of low cell numbers directly from a bacteria spiked blood model. bioRxiv 2018, 153965. [Google Scholar] [CrossRef] [Green Version]
- Charalampous, T.; Kay, G.L.; Richardson, H.; Aydin, A.; Baldan, R.; Jeanes, C.; Rae, D.; Grundy, S.; Turner, D.J.; Wain, J.; et al. Nanopore metagenomics enables rapid clinical diagnosis of bacterial lower respiratory infection. Nat. Biotechnol. 2019, 37, 783–792. [Google Scholar] [CrossRef]
- Bruggeling, C.E.; Garza, D.R.; Achouiti, S.; Mes, W.; Dutilh, B.E.; Boleij, A. Optimized bacterial DNA isolation method for microbiome analysis of human tissues. MicrobiologyOpen 2021, 10, e1191. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.; Xiao, F.; Wang, C.; Wang, Z. The impact of different methods of DNA extraction on microbial community measures of BALF samples based on metagenomic data. Am. J. Transl Res. 2016, 8, 1412–1425. [Google Scholar] [PubMed]
- Oechslin, C.P.; Lenz, N.; Liechti, N.; Ryter, S.; Agyeman, P.; Bruggmann, P.; Leib, S.L.; Beuret, C.M. Limited correlation of shotgun metagenomics following host depletion and routine diagnostics for viruses and bacteria in low concentrated surrogate and clinical samples. Front. Cell Infect. Microbiol. 2018, 8, 375. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nelson, M.T.; Pope, C.E.; Marsh, R.L.; Wolter, D.J.; Weiss, E.J.; Hager, K.R.; Vo, A.T.; Brittnacher, M.J.; Radey, M.C.; Hayden, H.S.; et al. Human and extracellular DNA depletion for metagenomic analysis of complex clinical infection samples yields optimized viable microbiome profiles. Cell Rep. 2019, 26, 2227–2240. [Google Scholar] [CrossRef] [Green Version]
- Ojo-Okunola, A.; Claassen-Weitz, S.; Mwaikono, K.S.; Gardner-Lubbe, S.; Zar, H.J.; Nicol, M.P.; du Toit, E. The influence of DNA extraction and lipid removal on human milk bacterial profiles. Methods Protoc. 2020, 3, 39. [Google Scholar] [CrossRef]
- Amar, Y.; Lagkouvardos, I.; Silva, R.L.; Ishola, O.A.; Foesel, B.U.; Kublik, S.; Schöler, A.; Niedermeier, S.; Bleuel, R.; Zink, A.; et al. Pre-digest of unprotected DNA by benzonase improves the representation of living skin bacteria and efficiently depletes host DNA. Microbiome 2021, 9, 123. [Google Scholar] [CrossRef]
- Israeli, O.; Makdasi, E.; Cohen-Gihon, I.; Zvi, A.; Lazar, S.; Shifman, O.; Levy, H.; Gur, D.; Laskar, O.; Beth-Din, A. A rapid high-throughput sequencing-based approach for the identification of unknown bacterial pathogens in whole blood. Future Sci. OA 2020, 6, Fso476. [Google Scholar] [CrossRef]
- Fittipaldi, M.; Nocker, A.; Codony, F. Progress in understanding preferential detection of live cells using viability dyes in combination with DNA amplification. J. Microbiol. Methods 2012, 91, 276–289. [Google Scholar] [CrossRef]
- Horz, H.P.; Scheer, S.; Vianna, M.E.; Conrads, G. New methods for selective isolation of bacterial DNA from human clinical specimens. Anaerobe 2010, 16, 47–53. [Google Scholar] [CrossRef]
- Thoendel, M.; Jeraldo, P.R.; Greenwood-Quaintance, K.E.; Yao, J.Z.; Chia, N.; Hanssen, A.D.; Abdel, M.P.; Patel, R. Comparison of microbial DNA enrichment tools for metagenomic whole genome sequencing. J. Microbiol. Methods 2016, 127, 141–145. [Google Scholar] [CrossRef] [Green Version]
- Bjerre, R.D.; Hugerth, L.W.; Boulund, F.; Seifert, M.; Johansen, J.D.; Engstrand, L. Effects of sampling strategy and DNA extraction on human skin microbiome investigations. Sci. Rep. 2019, 9, 17287. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heravi, F.S.; Zakrzewski, M.; Vickery, K.; Host, H.H. DNA depletion efficiency of microbiome DNA enrichment methods in infected tissue samples. J. Microbiol. Methods 2020, 170, 105856. [Google Scholar] [CrossRef] [PubMed]
- Rubiola, S.; Chiesa, F.; Dalmasso, A.; Di Ciccio, P.; Civera, T. Detection of Antimicrobial Resistance Genes in the Milk Production Environment: Impact of Host DNA and Sequencing Depth. Front. Microbiol. 2020, 11, 1983. [Google Scholar] [CrossRef] [PubMed]
- Yap, M.; Feehily, C.; Walsh, C.J.; Fenelon, M.; Murphy, E.F.; McAuliffe, F.M.; van Sinderen, D.; O’Toole, P.W.; O’Sullivan, O.; Cotter, P.D. Evaluation of methods for the reduction of contaminating host reads when performing shotgun metagenomic sequencing of the milk microbiome. Sci. Rep. 2020, 10, 21665. [Google Scholar] [CrossRef]
- Ganda, E.; Beck, K.L.; Haiminen, N.; Silverman, J.D.; Kawas, B.; Cronk, B.D.; Anderson, R.R.; Goodman, L.B.; Wiedmann, M. DNA extraction and host depletion methods significantly impact and potentially bias bacterial detection in a biological fluid. Msystems 2021, 6, e0061921. [Google Scholar] [CrossRef]
- Hansen, W.L.; Bruggeman, C.A.; Wolffs, P.F. Pre-analytical sample treatment and DNA extraction protocols for the detection of bacterial pathogens from whole blood. Methods Mol. Biol. 2013, 943, 81–90. [Google Scholar] [CrossRef] [Green Version]
- Avanzi, C.; Del-Pozo, J.; Benjak, A.; Stevenson, K.; Simpson, V.R.; Busso, P.; McLuckie, J.; Loiseau, C.; Lawton, C.; Schoening, J.; et al. Red squirrels in the British Isles are infected with leprosy bacilli. Science 2016, 354, 744–747. [Google Scholar] [CrossRef] [Green Version]
- Thoendel, M.J.; Jeraldo, P.R.; Greenwood-Quaintance, K.E.; Yao, J.Z.; Chia, N.; Hanssen, A.D.; Abdel, M.P.; Patel, R. Identification of prosthetic joint infection pathogens using a shotgun metagenomics approach. Clin. Infect. Dis. 2018, 67, 1333–1338. [Google Scholar] [CrossRef] [Green Version]
- Probst, A.J.; Weinmaier, T.; DeSantis, T.Z.; Domingo, J.W.S.; Ashbolt, N. New perspectives on microbial community distortion after whole-genome amplification. PLoS ONE 2015, 10, e0124158. [Google Scholar] [CrossRef] [Green Version]
- Cheng, M.; Cao, L.; Ning, K. Microbiome big-data mining and applications using single-cell technologies and metagenomics approaches toward precision medicine. Front. Genet. 2019, 10, 972. [Google Scholar] [CrossRef]
- Sidore, A.M.; Lan, F.; Lim, S.W.; Abate, A.R. Enhanced sequencing coverage with digital droplet multiple displacement amplification. Nucleic Acids Res. 2016, 44, e66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruan, Q.; Ruan, W.; Lin, X.; Wang, Y.; Zou, F.; Zhou, L.; Zhu, Z.; Yang, C. Digital-WGS: Automated, highly efficient whole-genome sequencing of single cells by digital microfluidics. Sci. Adv. 2020, 6, eabd6454. [Google Scholar] [CrossRef] [PubMed]
- Kubicek, J.; Singer, T.; Sander, A.-K. Method, Lysis Solution and Kit for Selectively Depleting Animal Nucleic Acids in a Sample. 2018, QIAGEN GmbH: U.S. Patent No. 10,655,122, 19 May 2020. [Google Scholar]
- Lorenz, M.G. Use of Nucleases for Degrading Nucleic Acids in the Presence of Chaotropic Agents and/or Surfactants. U.S. Patent Application No. 11/885,256, 3 July 2008. [Google Scholar]
- Radzieta, M.; Sadeghpour-Heravi, F.; Peters, T.J.; Hu, H.; Vickery, K.; Jeffries, T.; Dickson, H.G.; Schwarzer, S.; Jensen, S.O.; Malone, M. A multiomics approach to identify host-microbe alterations associated with infection severity in diabetic foot infections: A pilot study. NPJ Biofilms Microbiomes 2021, 7, 29. [Google Scholar] [CrossRef]
- Feehery, G.R.; Yigit, E.; Oyola, S.O.; Langhorst, B.W.; Schmidt, V.T.; Stewart, F.J.; Dimalanta, E.T.; Amaral-Zettler, L.A.; Davis, T.; Quail, M.A.; et al. A method for selectively enriching microbial DNA from contaminating vertebrate host DNA. PLoS ONE 2013, 8, e76096. [Google Scholar] [CrossRef] [PubMed]
- Osterloh, D.; Felsmann, K. Methods for Separating, Detecting or Enriching Different DNA Species. U.S. Patent Application No.14/056,756, 17 October 2013. [Google Scholar]
- Smith, M.; Campino, S.; Gu, Y.; Clark, T.G.; Otto, T.D.; Maslen, G.; Manske, M.; Imwong, M.; Dondorp, A.M.; Kwiatkowski, D.P.; et al. An in-solution hybridisation method for the isolation of pathogen DNA from human DNA-rich clinical samples for analysis by NGS. Open Genom. J. 2012, 5, 10–2174. [Google Scholar] [CrossRef] [PubMed]
- Willbanks, A.; Leary, M.; Greenshields, M.; Tyminski, C.; Heerboth, S.; Lapinska, K.; Haskins, K.; Sarkar, S. The evolution of epigenetics: From prokaryotes to humans and its biological consequences. Genet. Epigenet. 2016, 8, 825–836. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bird, A.P. CpG-rich islands and the function of DNA methylation. Nature 1986, 321, 209–213. [Google Scholar] [CrossRef]
- Xu, C.; Bian, C.; Lam, R.; Dong, A.; Min, J. The structural basis for selective binding of non-methylated CpG islands by the CFP1 CXXC domain. Nat. Commun. 2011, 2, 227. [Google Scholar] [CrossRef] [Green Version]
- Glassing, A.; Dowd, S.E.; Galandiuk, S.; Davis, B.; Jorden, J.R.; Chiodini, R.J. Changes in 16s RNA gene microbial community profiling by concentration of prokaryotic DNA. J. Microbiol. Methods 2015, 119, 239–242. [Google Scholar] [CrossRef]
- Oyola, S.O.; Gu, Y.; Manske, M.; Otto, T.D.; O’Brien, J.; Alcock, D.; Macinnis, B.; Berriman, M.; Newbold, C.I.; Kwiatkowski, D.P.; et al. Efficient depletion of host DNA contamination in malaria clinical sequencing. J. Clin. Microbiol. 2013, 51, 745–751. [Google Scholar] [CrossRef] [Green Version]
- Liu, G.; Weston, C.Q.; Pham, L.K.; Waltz, S.; Barnes, H.; King, P.; Sphar, D.; Yamamoto, R.T.; Forsyth, R.A. Epigenetic segregation of microbial genomes from complex samples using restriction endonucleases HpaII and McrB. PLoS ONE 2016, 11, e0146064. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blow, M.J.; Clark, T.A.; Daum, C.G.; Deutschbauer, A.M.; Fomenkov, A.; Fries, R.; Froula, J.; Kang, D.D.; Malmstrom, R.R.; Morgan, R.D.; et al. The epigenomic landscape of prokaryotes. PLoS Genet. 2016, 12, e1005854. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Douvlataniotis, K.; Bensberg, M.; Lentini, A.; Gylemo, B.; Nestor, C.E. No evidence for DNA N6 methyladenine in mammals. Sci. Adv. 2020, 6, eaay3335. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ratel, D.; Ravanat, J.L.; Berger, F.; Wion, D. N6-methyladenine: The other methylated base of DNA. Bioessays 2006, 28, 309–315. [Google Scholar] [CrossRef] [Green Version]
- Barnes, H.E.; Liu, G.; Weston, C.Q.; King, P.; Pham, L.K.; Waltz, S.; Helzer, K.T.; Day, L.; Sphar, D.; Yamamoto, R.T.; et al. Selective microbial genomic DNA isolation using restriction endonucleases. PLoS ONE 2014, 9, e109061. [Google Scholar] [CrossRef] [Green Version]
- Ferretti, P.; Farina, S.; Cristofolini, M.; Girolomoni, G.; Tett, A.; Segata, N. Exerimental metagenomics and ribosomal profiling of the human skin microbiome. Exp. Dermatol. 2017, 26, 211–219. [Google Scholar] [CrossRef] [Green Version]
- Krebes, J.; Morgan, R.D.; Bunk, B.; Spröer, C.; Luong, K.; Parusel, R.; Anton, B.P.; König, C.; Josenhans, C.; Overmann, J.; et al. The complex methylome of the human gastric pathogen Helicobacter pylori. Nucleic Acids Res. 2014, 42, 2415–2432. [Google Scholar] [CrossRef]
- Bewick, A.J.; Hofmeister, B.T.; Powers, R.A.; Mondo, S.J.; Grigoriev, I.V.; James, T.Y.; Stajich, J.E.; Schmitz, R.J. Diversity of cytosine methylation across the fungal tree of life. Nat. Ecol. Evol. 2019, 3, 479–490. [Google Scholar] [CrossRef]
- Hoelzer, K.; Shackelton, L.A.; Parrish, C.R. Presence and role of cytosine methylation in DNA viruses of animals. Nucleic Acids Res. 2008, 36, 2825–2837. [Google Scholar] [CrossRef]
- Snitkin, E.S.; Zelazny, A.M.; Thomas, P.J.; Stock, F.; Henderson, D.K.; Palmore, T.N.; Segre, J.A. Tracking a hospital outbreak of carbapenem-resistant Klebsiella pneumoniae with whole-genome sequencing. Sci. Transl. Med. 2012, 4, 148ra116. [Google Scholar] [CrossRef] [Green Version]
- Gire, S.K.; Goba, A.; Andersen, K.G.; Sealfon, R.S.; Park, D.J.; Kanneh, L.; Jalloh, S.; Momoh, M.; Fullah, M.; Dudas, G.; et al. Genomic surveillance elucidates Ebola virus origin and transmission during the 2014 outbreak. Science 2014, 345, 1369–1372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gu, W.; Miller, S.; Chiu, C.Y. Clinical metagenomic next-generation sequencing for pathogen detection. Annu. Rev. Pathol. 2019, 14, 319–338. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, K.; Mwaigwisya, S.; Crossman, L.C.; Doumith, M.; Munroe, D.; Pires, C.; Khan, A.M.; Woodford, N.; Saunders, N.J.; Wain, J.; et al. Identification of bacterial pathogens and antimicrobial resistance directly from clinical urines by nanopore-based metagenomic sequencing. J. Antimicrob. Chemother. 2017, 72, 104–114. [Google Scholar] [CrossRef] [Green Version]
- Graham, R.M.; Doyle, C.J.; Jennison, A.V. Epidemiological typing of Neisseria gonorrhoeae and detection of markers associated with antimicrobial resistance directly from urine samples using next generation sequencing. Sex Transm. Infect. 2017, 93, 65–67. [Google Scholar] [CrossRef] [Green Version]
- Miller, S.; Naccache, S.N.; Samayoa, E.; Messacar, K.; Arevalo, S.; Federman, S.; Stryke, D.; Pham, E.; Fung, B.; Bolosky, W.J.; et al. Laboratory validation of a clinical metagenomic sequencing assay for pathogen detection in cerebrospinal fluid. Genome Res. 2019, 29, 831–842. [Google Scholar] [CrossRef] [Green Version]
- Yang, L.; Haidar, G.; Zia, H.; Nettles, R.; Qin, S.; Wang, X.; Shah, F.; Rapport, S.F.; Charalampous, T.; Methé, B.; et al. Metagenomic identification of severe pneumonia pathogens in mechanically-ventilated patients: A feasibility and clinical validity study. Respir. Res. 2019, 20, 265. [Google Scholar] [CrossRef]
- Ivy, M.I.; Thoendel, M.J.; Jeraldo, P.R.; Greenwood-Quaintance, K.E.; Hanssen, A.D.; Abdel, M.P.; Chia, N.; Yao, J.Z.; Tande, A.J.; Mandrekar, J.N.; et al. Direct detection and identification of prosthetic joint infection pathogens in synovial fluid by metagenomic shotgun sequencing. J. Clin. Microbiol. 2018, 56, e00402-18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruppé, E.; Lazarevic, V.; Girard, M.; Mouton, W.; Ferry, T.; Laurent, F.; Schrenzel, J. Clinical metagenomics of bone and joint infections: A proof of concept study. Sci. Rep. 2017, 7, 7718. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kolb, M.; Lazarevic, V.; Emonet, S.; Calmy, A.; Girard, M.; Gaïa, N.; Charretier, Y.; Cherkaoui, A.; Keller, P.; Huber, C.; et al. Next-generation sequencing for the diagnosis of challenging culture-negative endocarditis. Front. Med. 2019, 6, 203. [Google Scholar] [CrossRef] [PubMed]
- Lazarevic, V.; Gaïa, N.; Girard, M.; Leo, S.; Cherkaoui, A.; Renzi, G.; Emonet, S.; Jamme, S.; Ruppé, E.; Vijgen, S.; et al. When bacterial culture fails, metagenomics can help: A case of chronic hepatic brucelloma assessed by next-generation sequencing. Front Microbiol. 2018, 9, 1566. [Google Scholar] [CrossRef]
- Sanabria, A.; Hjerde, E.; Johannessen, M.; Sollid, J.E.; Simonsen, G.S.; Hanssen, A.M. Shotgun-metagenomics on positive blood culture bottles inoculated with prosthetic joint tissue: A proof of concept study. Front. Microbiol. 2020, 11, 1687. [Google Scholar] [CrossRef] [PubMed]
- Vijayvargiya, P.; Jeraldo, P.R.; Thoendel, M.J.; Greenwood-Quaintance, K.E.; Garrigos, Z.E.; Rizwan Sohail, M.; Chia, N.; Pritt, B.S.; Patel, R. Application of metagenomic shotgun sequencing to detect vector-borne pathogens in clinical blood samples. PLoS ONE 2019, 14, e0222915. [Google Scholar] [CrossRef] [PubMed]
- Parize, P.; Muth, E.; Richaud, C.; Gratigny, M.; Pilmis, B.; Lamamy, A.; Mainardi, J.L.; Cheval, J.; de Visser, L.; Jagorel, F.; et al. Untargeted next-generation sequencing-based first-line diagnosis of infection in immunocompromised adults: A multicentre, blinded, prospective study. Clin. Microbiol. Infect. 2017, 23, e571–e574. [Google Scholar] [CrossRef] [PubMed]
- Votintseva, A.A.; Bradley, P.; Pankhurst, L.; Elias, C.d.; Loose, M.; Nilgiriwala, K.; Chatterjee, A.; Smith, E.G.; Sanderson, N.; Walker, T.M.; et al. Same-day diagnostic and surveillance data for tuberculosis via whole-genome sequencing of direct respiratory samples. J. Clin. Microbiol. 2017, 55, 1285–1298. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Street, T.L.; Sanderson, N.D.; Atkins, B.L.; Brent, A.J.; Cole, K.; Foster, D.; McNally, M.A.; Oakley, S.; Peto, L.; Taylor, A.; et al. Molecular diagnosis of orthopedic-device-related infection directly from sonication fluid by metagenomic sequencing. J. Clin. Microbiol. 2017, 55, 2334–2347. [Google Scholar] [CrossRef] [Green Version]
- Barraud, O.; Ravry, C.; François, B.; Daix, T.; Ploy, M.C.; Vignon, P. Shotgun metagenomics for microbiome and resistome detection in septic patients with urinary tract infection. Int. J. Antimicrob. Agents 2019, 54, 803–808. [Google Scholar] [CrossRef]
- Ji, P.; Zhang, Y.; Wang, J.; Zhao, F. MetaSort untangles metagenome assembly by reducing microbial community complexity. Nat. Commun. 2017, 8, 14306. [Google Scholar] [CrossRef]
- Gu, W.; Deng, X.; Lee, M.; Sucu, Y.D.; Arevalo, S.; Stryke, D.; Federman, S.; Gopez, A.; Reyes, K.; Zorn, K.; et al. Rapid pathogen detection by metagenomic next-generation sequencing of infected body fluids. Nat. Med. 2021, 27, 115–124. [Google Scholar] [CrossRef]
- Strong, M.J.; Xu, G.; Morici, L.; Bon-Durant, S.S.; Baddoo, M.; Lin, Z.; Fewell, C.; Taylor, C.M.; Flemington, E.K. Microbial contamination in next generation sequencing: Implications for sequence-based analysis of clinical samples. PLoS Pathog. 2014, 10, e1004437. [Google Scholar] [CrossRef] [Green Version]
- Carpenter, M.L.; Bustamante, C.D.; Gourguechon, S.B. Compositions and Methods for Targeted Depletion, Enrichment, and Partitioning of Nucleic Acids Using Crispr/Cas System Proteins. U.S. Patent Application No.16/231,338, 21 December 2018. [Google Scholar]
- Gu, W.; Crawford, E.D.; O’Donovan, B.D.; Wilson, M.R.; Chow, E.D.; Retallack, H.; DeRisi, J.L. Depletion of abundant sequences by hybridization (DASH): Using Cas9 to remove unwanted high-abundance species in sequencing libraries and molecular counting applications. Genome Biol. 2016, 17, 41. [Google Scholar] [CrossRef] [Green Version]
- Deininger, P. Alu elements: Know the SINEs. Genome Biol. 2011, 12, 236. [Google Scholar] [CrossRef] [Green Version]
- Loose, M.; Malla, S.; Stout, M. Real-time selective sequencing using nanopore technology. Nat. Methods 2016, 13, 751–754. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kovaka, S.; Fan, Y.; Ni, B.; Timp, W.; Schatz, M.C. Targeted nanopore sequencing by real-time mapping of raw electrical signal with uncalled. Nat. Biotechnol. 2021, 39, 431–441. [Google Scholar] [CrossRef] [PubMed]
- Payne, A.; Holmes, N.; Clarke, T.; Munro, R.; Debebe, B.J.; Loose, M. Readfish enables targeted nanopore sequencing of gigabase-sized genomes. Nat. Biotechnol. 2021, 39, 442–450. [Google Scholar] [CrossRef] [PubMed]


| Kit | Principle | Pros | Cons | Hands-On Time Per Sample | Cost Per Sample (USD) | Ref. |
|---|---|---|---|---|---|---|
| QIAamp DNA Microbiome (Qiagen, Hilden, Germany) | Lysis of host cell by saponin, degrade extracellular DNA with Benzonase nuclease | Ultra-clean columns to minimise contamination risk | Requires fresh sample | 160 min | 13 | [43] |
| MolYsis™ Complete/Ultra-Deep Microbiome Prep (Molzym, Bremen, Germany) | Chaotropic lysis of host cell, degrade extracellular DNA with MolDNase | Applicable for body fluids, tissue and swab samples. Enrichment of bacterial and fungal DNA | Fresh sample is recommended | 120 min | 11 | [44] |
| HostZERO Microbial DNA Kit (Zymo Research, Irvine, CA, USA) | Lysis of host cell, degrade extracellular DNA with microbial selection enzyme | Protocols for both tissue and liquid samples are provided | Requires intact (living) bacteria cells | 30 min | 10 | [45] |
| NEBNext Microbiome DNA Enrichment (New England BioLabs, Ipswich, MA, USA) | Capture methylated host DNA | Can retain cell-free DNA from dead organisms to avoid DNA loss | Requires high molecular weight intact DNA. Bias to high CpG-methylated microbes | 30 min * | 39 * | [46] |
| LOOXSTER Enrichment Kit (Analytik Jena GmbH, Jena, Germany) | Capture non-methylated CpG dinucleotides | Can retain cell-free DNA from dead organisms to avoid DNA loss | Requires high molecular weight intact DNA. Bias to high CpG-methylated microbes | 75 min * | 34 * | [47] |
| Sample Type | Potential Clinical Indication | Sample Size | Depletion Method | Sequencing Platform | Reads Number | Ref. |
|---|---|---|---|---|---|---|
| Cerebrospinal fluid | Infectious aetiology identification | 13 | Selective lysis by a bead-beater tissue homogeniser followed by a Benzonase nuclease treatment | Ion Torrent PGM | N/A | [23] |
| Prosthetic joint sonicate fluid | Pathogen identification | 408 | MolYsis basic kit | Illumina HiSeq | 2.8 million, mean | [38] |
| Urine | Pathogen identification | 10 | Differential centrifugation and MolYsis kit | MinION | 0.026 million, median | [66] |
| Urine | Antimicrobial resistance marker identification | 13 | NEBNext microbiome kit | Ion Torrent PGM | N/A | [67] |
| Sputum | Pathogen detection | 6 | Microfluidic separation followed by DNase digestion | Illumina HiSeq | 36.3 million, mean | [12] |
| Sputum, bronchoalveolar lavage and endotracheal aspirates | Diagnosis of known and unknown infections | 40 | Saponin-based differential lysis followed by HL-SAN DNase digestion | MinION | 0.041 million, mean | [20] |
| Cerebrospinal fluid | Diagnosis of known and unknown infections | 95 | NEB Microbiome Enrichment Kit | Illumina HiSeq | 5~10 million | [68] |
| Endotracheal aspirates | Pathogen identification | 22 | Saponin-based differential lysis followed by HL-SAN DNase digestion | MinION | 6628, median | [69] |
| Synovial fluid | Pathogen detection | 168 | MolYsis basic kit | Illumina HiSeq | 30 million, mean | [70] |
| Bone and joint infectious tissue | Pathogen detection and antibiotic susceptibility prediction | 24 | Ultra-Deep Microbiome Prep kit | Illumina HiSeq | 20 million, mean | [71] |
| Valve tissue | Pathogen identification | 1 | Ultra-Deep Microbiome Prep kit | Illumina MiSeq | 1.4 million, mean | [72] |
| Hepatic tissue | Diagnosis of unknown infections | 1 | Ultra-Deep Microbiome Prep kit | Illumina MiSeq | 1.1 million, mean | [73] |
| Blood culture bottles inoculated with prosthetic joint tissue | Pathogen identification | 9 | MolYsis basic kit | Illumina MiSeq | 10.3 million, mean | [74] |
| Blood | Pathogen detection | 8 | MolYsis complete kit and WGA | Illumina HiSeq | 27.5 million, mean | [75] |
| Whole blood | Diagnosis of infection | 101 | MolYsis complete kit | Ion Torrent | N/A | [76] |
| Sputum | M. tuberculosis detection and antibiotic susceptibility prediction | 40 | MolYsis basic kit | Illumina MiSeq and MinION | 3.6 million, mean | [77] |
| Prosthetic joint sonication fluid | Diagnosis of prosthetic joint infections | 97 | A 5 μm pore size filter | Illumina MiSeq | N/A | [78] |
| Urine | Pathogen detection and antimicrobial susceptibility prediction | 40 | NEB Microbiome Enrichment Kit | Ion Proton | N/A | [79] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, Y.; Wang, G.; Lau, H.C.-H.; Yu, J. Metagenomic Sequencing for Microbial DNA in Human Samples: Emerging Technological Advances. Int. J. Mol. Sci. 2022, 23, 2181. https://doi.org/10.3390/ijms23042181
Shi Y, Wang G, Lau HC-H, Yu J. Metagenomic Sequencing for Microbial DNA in Human Samples: Emerging Technological Advances. International Journal of Molecular Sciences. 2022; 23(4):2181. https://doi.org/10.3390/ijms23042181
Chicago/Turabian StyleShi, Yu, Guoping Wang, Harry Cheuk-Hay Lau, and Jun Yu. 2022. "Metagenomic Sequencing for Microbial DNA in Human Samples: Emerging Technological Advances" International Journal of Molecular Sciences 23, no. 4: 2181. https://doi.org/10.3390/ijms23042181






