Biological Functions and Applications of Virus-Related Bacterial Nanoparticles: A Review
Abstract
:1. Introduction
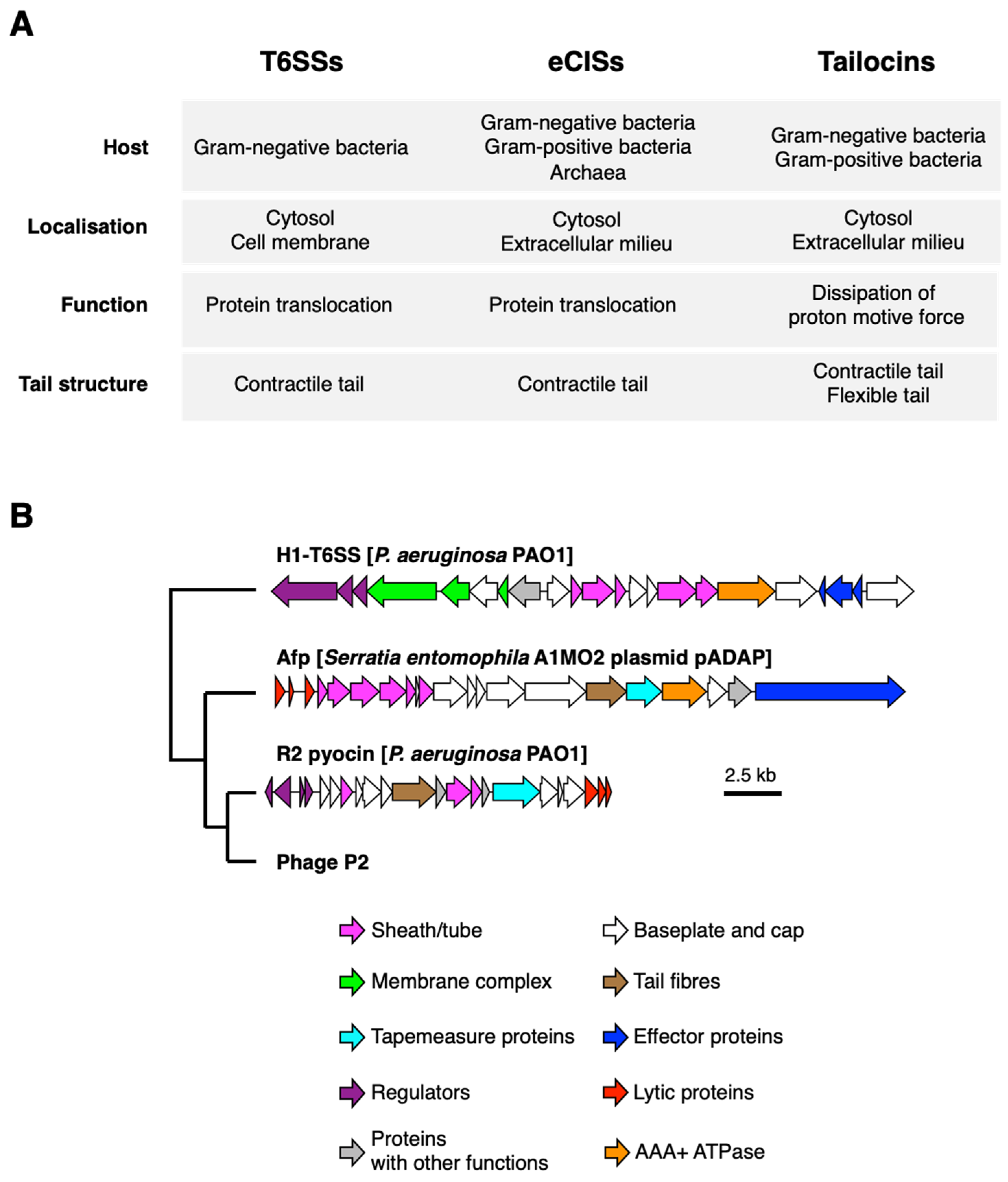
2. Intracellular and Extracellular Phage Tail-like Nanostructures
3. Tailocins
4. Extracellular Contractile Injections Systems (eCISs)
4.1. Distribution and Evolution of eCISs
4.2. Biological Functions of eCISs
4.2.1. Gram-Negative Bacterial eCISs
4.2.2. Gram-Positive Bacterial eCISs
5. Discussion
6. Conclusions and Future Directions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Toyofuku, M.; Nomura, N.; Eberl, L. Types and origins of bacterial membrane vesicles. Nat. Rev. Microbiol. 2019, 17, 13–24. [Google Scholar] [CrossRef]
- Nagakubo, T.; Nomura, N.; Toyofuku, M. Cracking Open Bacterial Membrane Vesicles. Front. Microbiol. 2019, 10, 3026. [Google Scholar] [CrossRef] [Green Version]
- Costa, T.; Felisberto-Rodrigues, C.; Meir, A.; Prevost, M.S.; Redzej, A.; Trokter, M.; Waksman, G. Secretion systems in Gram-negative bacteria: Structural and mechanistic insights. Nat. Rev. Genet. 2015, 13, 343–359. [Google Scholar] [CrossRef]
- Nóbrega, F.L.; Vlot, M.; de Jonge, P.A.; Dreesens, L.L.; Beaumont, H.J.E.; Lavigne, R.; Dutilh, B.E.; Brouns, S.J.J. Targeting mechanisms of tailed bacteriophages. Nat. Rev. Microbiol. 2018, 16, 760–773. [Google Scholar] [CrossRef]
- Mills, S.; Ross, R.; Hill, C. Bacteriocins and bacteriophage; a narrow-minded approach to food and gut microbiology. FEMS Microbiol. Rev. 2017, 41, S129–S153. [Google Scholar] [CrossRef]
- Sarris, P.F.; Ladoukakis, E.D.; Panopoulos, N.J.; Scoulica, E.V. A Phage Tail-Derived Element with Wide Distribution among Both Prokaryotic Domains: A Comparative Genomic and Phylogenetic Study. Genome Biol. Evol. 2014, 6, 1739–1747. [Google Scholar] [CrossRef] [PubMed]
- Ghequire, M.G.; De Mot, R. The Tailocin Tale: Peeling off Phage Tails. Trends Microbiol. 2015, 23, 587–590. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, F.; Li, N.; Wang, X.; Cheng, J.; Huang, Y.; Yang, Y.; Yang, J.; Cai, B.; Wang, Y.-P.; Jin, Q.; et al. Cryo-EM Structure and Assembly of an Extracellular Contractile Injection System. Cell 2019, 177, 370–383.e15. [Google Scholar] [CrossRef] [Green Version]
- Ericson, C.F.; Eisenstein, F.; Medeiros, J.M.; E Malter, K.; Cavalcanti, G.S.; Zeller, R.W.; Newman, D.K.; Pilhofer, M.; Shikuma, N.J. A contractile injection system stimulates tubeworm metamorphosis by translocating a proteinaceous effector. eLife 2019, 8, e46865. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Song, N.; Liu, B.; Zhang, N.; Alikhan, N.-F.; Zhou, Z.; Zhou, Y.; Zhou, S.; Zheng, D.; Chen, M.; et al. Genome-wide Identification and Characterization of a Superfamily of Bacterial Extracellular Contractile Injection Systems. Cell Rep. 2019, 29, 511–521.e2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ge, P.; Scholl, D.; Prokhorov, N.S.; Avaylon, J.; Shneider, M.M.; Browning, C.; Buth, S.A.; Plattner, M.; Chakraborty, U.; Ding, K.; et al. Action of a minimal contractile bactericidal nanomachine. Nature 2020, 580, 658–662. [Google Scholar] [CrossRef] [PubMed]
- Desfosses, A.; Venugopal, H.; Joshi, T.; Felix, J.; Jessop, M.; Jeong, H.; Hyun, J.; Heymann, J.B.; Hurst, M.R.H.; Gutsche, I.; et al. Atomic structures of an entire contractile injection system in both the extended and contracted states. Nat. Microbiol. 2019, 4, 1885–1894. [Google Scholar] [CrossRef] [PubMed]
- Leiman, P.; Basler, M.; Ramagopal, U.A.; Bonanno, J.B.; Sauder, J.M.; Pukatzki, S.; Burley, S.; Almo, S.C.; Mekalanos, J.J. Type VI secretion apparatus and phage tail-associated protein complexes share a common evolutionary origin. Proc. Natl. Acad. Sci. USA 2009, 106, 4154–4159. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Basler, M.; Pilhofer, M.; Henderson, G.P.; Jensen, G.J.; Mekalanos, J.J. Type VI secretion requires a dynamic contractile phage tail-like structure. Nature 2012, 483, 182–186. [Google Scholar] [CrossRef]
- Pukatzki, S.; Ma, A.T.; Sturtevant, D.; Krastins, B.; Sarracino, D.; Nelson, W.C.; Heidelberg, J.F.; Mekalanos, J.J. Identification of a conserved bacterial protein secretion system in Vibrio cholerae using the Dictyostelium host model system. Proc. Natl. Acad. Sci. USA 2006, 103, 1528–1533. [Google Scholar] [CrossRef] [Green Version]
- Russell, A.; Hood, R.D.; Bui, N.K.; LeRoux, M.; Vollmer, W.; Mougous, J.D. Type VI secretion delivers bacteriolytic effectors to target cells. Nature 2011, 475, 343–347. [Google Scholar] [CrossRef] [Green Version]
- LaCourse, K.D.; Peterson, S.B.; Kulasekara, H.D.; Radey, M.C.; Kim, J.; Mougous, J.D. Conditional toxicity and synergy drive diversity among antibacterial effectors. Nat. Microbiol. 2018, 3, 440–446. [Google Scholar] [CrossRef]
- Smith, W.P.J.; Vettiger, A.; Winter, J.; Ryser, T.; Comstock, L.E.; Basler, M.; Foster, K.R. The evolution of the type VI secretion system as a disintegration weapon. PLoS Biol. 2020, 18, e3000720. [Google Scholar] [CrossRef]
- Molina-Santiago, C.; Pearson, J.R.; Navarro, Y.; Berlanga-Clavero, M.V.; Caraballo-Rodriguez, A.M.; Petras, D.; García-Martín, M.L.; Lamon, G.; Haberstein, B.; Cazorla, F.M.; et al. The extracellular matrix protects Bacillus subtilis colonies from Pseudomonas invasion and modulates plant co-colonization. Nat. Commun. 2019, 10, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Vettiger, A.; Basler, M. Type VI Secretion System Substrates Are Transferred and Reused among Sister Cells. Cell 2016, 167, 99–110.e12. [Google Scholar] [CrossRef] [Green Version]
- Hersch, S.J.; Watanabe, N.; Stietz, M.S.; Manera, K.; Kamal, F.; Burkinshaw, B.; Lam, L.; Pun, A.; Li, M.; Savchenko, A.; et al. Envelope stress responses defend against type six secretion system attacks independently of immunity proteins. Nat. Microbiol. 2020, 5, 706–714. [Google Scholar] [CrossRef] [PubMed]
- Wood, T.; Howard, S.A.; Förster, A.; Nolan, L.; Manoli, E.; Bullen, N.P.; Yau, H.; Hachani, A.; Hayward, R.D.; Whitney, J.C.; et al. The Pseudomonas aeruginosa T6SS Delivers a Periplasmic Toxin that Disrupts Bacterial Cell Morphology. Cell Rep. 2019, 29, 187–201.e7. [Google Scholar] [CrossRef] [Green Version]
- Russell, A.B.; Peterson, S.B.; Mougous, J.D. Type VI secretion system effectors: Poisons with a purpose. Nat. Rev. Microbiol. 2014, 12, 137–148. [Google Scholar] [CrossRef]
- Navarro-Garcia, F.; Ruiz-Perez, F.; Cataldi, Á.; Larzabal, M. Type VI Secretion System in Pathogenic Escherichia coli: Structure, Role in Virulence, and Acquisition. Front. Microbiol. 2019, 10, 1965. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brunet, Y.R.; Zoued, A.; Boyer, F.; Douzi, B.; Cascales, E. The Type VI Secretion TssEFGK-VgrG Phage-Like Baseplate Is Recruited to the TssJLM Membrane Complex via Multiple Contacts and Serves As Assembly Platform for Tail Tube/Sheath Polymerization. PLoS Genet. 2015, 11, e1005545. [Google Scholar] [CrossRef] [Green Version]
- Rybakova, D.; Schramm, P.; Mitra, A.; Hurst, M.R.H. Afp14 is involved in regulating the length of Anti-feeding prophage (Afp). Mol. Microbiol. 2015, 96, 815–826. [Google Scholar] [CrossRef] [Green Version]
- Vacheron, J.; Heiman, C.M.; Keel, C. Live cell dynamics of production, explosive release and killing activity of phage tail-like weapons for Pseudomonas kin exclusion. Commun. Biol. 2021, 4, 1–14. [Google Scholar] [CrossRef]
- Michel-Briand, Y.; Baysse, C. The pyocins of Pseudomonas aeruginosa. Biochimie 2002, 84, 499–510. [Google Scholar] [CrossRef]
- Canchaya, C.; Proux, C.; Fournous, G.; Bruttin, A.; Brüssow, H. Prophage Genomics. Microbiol. Mol. Biol. Rev. 2003, 67, 238–276. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fillol-Salom, A.; Alsaadi, A.; De Sousa, J.A.M.; Zhong, L.; Foster, K.R.; Rocha, E.P.C.; Penadés, J.R.; Ingmer, H.; Haaber, J. Bacteriophages benefit from generalized transduction. PLOS Pathog. 2019, 15, e1007888. [Google Scholar] [CrossRef]
- Fernández-Ruiz, I.; Coutinho, F.H.; Rodriguez-Valera, F. Thousands of Novel Endolysins Discovered in Uncultured Phage Genomes. Front. Microbiol. 2018, 9, 1033. [Google Scholar] [CrossRef] [Green Version]
- Wang, I.-N.; Smith, D.L.; Young, R. Holins: The Protein Clocks of Bacteriophage Infections. Annu. Rev. Microbiol. 2000, 54, 799–825. [Google Scholar] [CrossRef]
- Shikuma, N.J.; Pilhofer, M.; Weiss, G.L.; Hadfield, M.G.; Jensen, G.J.; Newman, D.K. Marine Tubeworm Metamorphosis Induced by Arrays of Bacterial Phage Tail–Like Structures. Science 2014, 343, 529–533. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Böck, D.; Medeiros, J.M.; Tsao, H.-F.; Penz, T.; Weiss, G.L.; Aistleitner, K.; Horn, M.; Pilhofer, M. In situ architecture, function, and evolution of a contractile injection system. Science 2017, 357, 713–717. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ito, S.; Kageyama, M.; Egami, F. Isolation and characterization of pyocins from several strains of pseudomonas aeruginosa. J. Gen. Appl. Microbiol. 1970, 16, 205–214. [Google Scholar] [CrossRef]
- Kuroda, K.; Kageyama, M. Comparative Study on F-Type Pyocins of Pseudomonas aeruginosa. J. Biochem. 1981, 89, 1721–1736. [Google Scholar] [CrossRef] [PubMed]
- Trought, T.E.T.; Jackson, T.A.; French, R.A. Incidence and transmission of a disease of grass grub(Costelytra zealandica)in Canterbury. N. Zealand J. Exp. Agric. 1982, 10, 79–82. [Google Scholar] [CrossRef] [Green Version]
- Glare, T.R.; Corbett, G.E.; Sadler, T.J. Association of a Large Plasmid with Amber Disease of the New Zealand Grass Grub, Costelytra zealandica, Caused by Serratia entomophila and Serratia proteamaculans. J. Invertebr. Pathol. 1993, 62, 165–170. [Google Scholar] [CrossRef]
- Hurst, M.R.H.; Glare, T.; Jackson, T.A.; Ronson, C. Plasmid-Located Pathogenicity Determinants of Serratia entomophila, the Causal Agent of Amber Disease of Grass Grub, Show Similarity to the Insecticidal Toxins of Photorhabdus luminescens. J. Bacteriol. 2000, 182, 5127–5138. [Google Scholar] [CrossRef] [Green Version]
- Hurst, M.R.H.; Glare, T.; Jackson, T.A. Cloning Serratia entomophila Antifeeding Genes—a Putative Defective Prophage Active against the Grass Grub Costelytra zealandica. J. Bacteriol. 2004, 186, 5116–5128. [Google Scholar] [CrossRef] [Green Version]
- Hurst, M.R.H.; Beard, S.S.; Jackson, T.A.; Jones, S.M. Isolation and characterization of theSerratia entomophilaantifeeding prophage. FEMS Microbiol. Lett. 2007, 270, 42–48. [Google Scholar] [CrossRef] [Green Version]
- Heymann, J.B.; Bartho, J.D.; Rybakova, D.; Venugopal, H.P.; Winkler, D.C.; Sen, A.; Hurst, M.R.H.; Mitra, A.K. Three-dimensional Structure of the Toxin-delivery Particle Antifeeding Prophage of Serratia entomophila. J. Biol. Chem. 2013, 288, 25276–25284. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, G.; Dowling, A.J.; Gerike, U.; Ffrench-Constant, R.; Waterfield, N. Photorhabdus Virulence Cassettes Confer Injectable Insecticidal Activity against the Wax Moth. J. Bacteriol. 2006, 188, 2254–2261. [Google Scholar] [CrossRef] [Green Version]
- Ffrench-Constant, R.; Waterfield, N.; Daborn, P.; Joyce, S.; Bennett, H.; Au, C.; Dowling, A.; Boundy, S.; Reynolds, S.; Clarke, D. Photorhabdus: Towards a functional genomic analysis of a symbiont and pathogen. FEMS Microbiol. Rev. 2003, 26, 433–456. [Google Scholar] [CrossRef] [PubMed]
- Vlisidou, I.; Hapeshi, A.; Healey, J.R.; Smart, K.; Yang, G.; Waterfield, N.R. The Photorhabdus asymbiotica virulence cassettes deliver protein effectors directly into target eukaryotic cells. eLife 2019, 8, e46259. [Google Scholar] [CrossRef]
- Waterfield, N.R.; Daborn, P.J.; Ffrench-Constant, R.H. Insect pathogenicity islands in the insect pathogenic bacterium Photorhabdus. Physiol. Èntomol. 2004, 29, 240–250. [Google Scholar] [CrossRef]
- Duchaud, E.; Rusniok, C.; Frangeul, L.; Buchrieser, C.; Givaudan, A.; Taourit, S.; Bocs, S.; Boursaux-Eude, C.; Chandler, M.; Charles, J.-F.; et al. The genome sequence of the entomopathogenic bacterium Photorhabdus luminescens. Nat. Biotechnol. 2003, 21, 1307–1313. [Google Scholar] [CrossRef] [PubMed]
- Hadfield, M.G. Biofilms and Marine Invertebrate Larvae: What Bacteria Produce That Larvae Use to Choose Settlement Sites. Annu. Rev. Mar. Sci. 2011, 3, 453–470. [Google Scholar] [CrossRef]
- Shikuma, N.J.; Antoshechkin, I.; Medeiros, J.M.; Pilhofer, M.; Newman, D.K. Stepwise metamorphosis of the tubewormHydroides elegansis mediated by a bacterial inducer and MAPK signaling. Proc. Natl. Acad. Sci. USA 2016, 113, 10097–10102. [Google Scholar] [CrossRef] [Green Version]
- Rocchi, I.; Ericson, C.F.; Malter, K.E.; Zargar, S.; Eisenstein, F.; Pilhofer, M.; Beyhan, S.; Shikuma, N.J. A Bacterial Phage Tail-like Structure Kills Eukaryotic Cells by Injecting a Nuclease Effector. Cell Rep. 2019, 28, 295–301.e4. [Google Scholar] [CrossRef]
- Horn, M.; Harzenetter, M.D.; Linner, T.; Schmid, E.N.; Muller, K.-D.; Michel, R.; Wagner, M. Members of the Cytophaga-Flavobacterium-Bacteroides phylum as intracellular bacteria of acanthamoebae: Proposal of ‘Candidatus Amoebophilus asiaticus’. Environ. Microbiol. 2001, 3, 440–449. [Google Scholar] [CrossRef] [PubMed]
- Furusawa, G.; Yoshikawa, T.; Takano, Y.; Mise, K.; Furusawa, I.; Okuno, T.; Sakata, T. Characterization of cytoplasmic fibril structures found in gliding cells of Saprospira sp. Can. J. Microbiol. 2005, 51, 875–880. [Google Scholar] [CrossRef]
- Ueda, M.; Takagi, A. Rhapidosomes in clostridium botulinum. J. Gen. Appl. Microbiol. 1972, 18, 81–98. [Google Scholar] [CrossRef] [Green Version]
- Pazirandeh, M.; Campbell, J.R. Protein composition of rhapidosomes isolated from Aquaspirillum itersonii. J. Gen. Microbiol. 1993, 139, 859–864. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bush, M.; Tschowri, N.; Schlimpert, S.; Flärdh, K.; Buttner, M. c-di-GMP signalling and the regulation of developmental transitions in streptomycetes. Nat. Rev. Genet. 2015, 13, 749–760. [Google Scholar] [CrossRef]
- Chater, K.F.; Chandra, G. The use of the rare UUA codon to define “Expression Space” for genes involved in secondary metabolism, development and environmental adaptation in Streptomyces. J. Microbiol. 2008, 46, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Higo, A.; Horinouchi, S.; Ohnishi, Y. Strict regulation of morphological differentiation and secondary metabolism by a positive feedback loop between two global regulators AdpA and BldA in Streptomyces griseus. Mol. Microbiol. 2011, 81, 1607–1622. [Google Scholar] [CrossRef] [PubMed]
- Leskiw, B.K.; Lawlor, E.J.; Fernandez-Abalos, J.M.; Chater, K.F. TTA codons in some genes prevent their expression in a class of developmental, antibiotic-negative, Streptomyces mutants. Proc. Natl. Acad. Sci. USA 1991, 88, 2461–2465. [Google Scholar] [CrossRef] [Green Version]
- Hesketh, A.; Bucca, G.; Laing, E.; Flett, F.; Hotchkiss, G.; Smith, C.P.; Chater, K.F. New pleiotropic effects of eliminating a rare tRNA from Streptomyces coelicolor, revealed by combined proteomic and transcriptomic analysis of liquid cultures. BMC Genom. 2007, 8, 261. [Google Scholar] [CrossRef] [Green Version]
- Kim, D.-W.; Chater, K.F.; Lee, K.-J.; Hesketh, A. Effects of growth phase and the developmentally significant bldA-specified tRNA on the membrane-associated proteome of Streptomyces coelicolor. Microbiology 2005, 151, 2707–2720. [Google Scholar] [CrossRef] [Green Version]
- Nagakubo, T.; Yamamoto, T.; Asamizu, S.; Toyofuku, M.; Nomura, N.; Onaka, H. Phage tail-like nanostructures affect microbial interactions between Streptomyces and fungi. Sci. Rep. 2021, 11, 1–11. [Google Scholar] [CrossRef]
- Seipke, R.; Kaltenpoth, M.; Hutchings, M.I. Streptomycesas symbionts: An emerging and widespread theme? FEMS Microbiol. Rev. 2012, 36, 862–876. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sunderland, K.S.; Yang, M.; Mao, C. Phage-Enabled Nanomedicine: From Probes to Therapeutics in Precision Medicine. Angew. Chem. Int. Ed. 2016, 56, 1964–1992. [Google Scholar] [CrossRef] [Green Version]
- Alfaleh, M.A.; Alsaab, H.O.; Mahmoud, A.B.; Alkayyal, A.A.; Jones, M.L.; Mahler, S.M.; Hashem, A.M. Phage Display Derived Monoclonal Antibodies: From Bench to Bedside. Front. Immunol. 2020, 11. [Google Scholar] [CrossRef]
- Wu, C.-H.; Liu, I.-J.; Lu, R.-M.; Wu, H.-C. Advancement and applications of peptide phage display technology in biomedical science. J. Biomed. Sci. 2016, 23, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, H.; Cao, B.; George, A.; Mao, C. Self-Assembly and Mineralization of Genetically Modifiable Biological Nanofibers Driven by β-Structure Formation. Biomacromolecules 2011, 12, 2193–2199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Geller, A.M.; Pollin, I.; Zlotkin, D.; Danov, A.; Nachmias, N.; Andreopoulos, W.B.; Shemesh, K.; Levy, A. The extracellular contractile injection system is enriched in environmental microbes and associates with numerous toxins. Nat. Commun. 2021, 12, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Brown, L.; Wolf, J.M.; Prados-Rosales, R.; Casadevall, A. Through the wall: Extracellular vesicles in Gram-positive bacteria, mycobacteria and fungi. Nat. Rev. Microbiol. 2015, 13, 620–630. [Google Scholar] [CrossRef] [Green Version]
- Schwechheimer, C.; Kuehn, M.J. Outer-membrane vesicles from Gram-negative bacteria: Biogenesis and functions. Nat. Rev. Genet. 2015, 13, 605–619. [Google Scholar] [CrossRef] [Green Version]
- Turnbull, L.; Toyofuku, M.; Hynen, A.; Kurosawa, M.; Pessi, G.; Petty, N.K.; Osvath, S.R.; Cárcamo-Oyarce, G.; Gloag, E.S.; Shimoni, R.; et al. Explosive cell lysis as a mechanism for the biogenesis of bacterial membrane vesicles and biofilms. Nat. Commun. 2016, 7, 11220. [Google Scholar] [CrossRef] [Green Version]
- Toyofuku, M.; Cárcamo-Oyarce, G.; Yamamoto, T.; Eisenstein, F.; Hsiao, C.-C.; Kurosawa, M.; Gademann, K.; Pilhofer, M.; Nomura, N.; Eberl, L. Prophage-triggered membrane vesicle formation through peptidoglycan damage in Bacillus subtilis. Nat. Commun. 2017, 8, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nagakubo, T.; Tahara, Y.O.; Miyata, M.; Nomura, N.; Toyofuku, M. Mycolic acid-containing bacteria trigger distinct types of membrane vesicles through different routes. iScience 2021, 24, 102015. [Google Scholar] [CrossRef]
- Forterre, P.; Soler, N.; Krupovic, M.; Marguet, E.; Ackermann, H.-W. Fake virus particles generated by fluorescence microscopy. Trends Microbiol. 2013, 21, 1–5. [Google Scholar] [CrossRef]
- Forterre, P. The virocell concept and environmental microbiology. ISME J. 2012, 7, 233–236. [Google Scholar] [CrossRef] [PubMed]
- Soler, N.; Krupovic, M.; Marguet, E.; Forterre, P. Membrane vesicles in natural environments: A major challenge in viral ecology. ISME J. 2014, 9, 793–796. [Google Scholar] [CrossRef] [Green Version]
- Biller, S.J.; Schubotz, F.; Roggensack, S.E.; Thompson, A.W.; Summons, R.E.; Chisholm, S.W. Bacterial Vesicles in Marine Ecosystems. Science 2014, 343, 183–186. [Google Scholar] [CrossRef]
- Patz, S.; Becker, Y.; Richert-Pöggeler, K.R.; Berger, B.; Ruppel, S.; Huson, D.H.; Becker, M. Phage tail-like particles are versatile bacterial nanomachines – A mini-review. J. Adv. Res. 2019, 19, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Scholl, D. Phage Tail–Like Bacteriocins. Annu. Rev. Virol. 2017, 4, 453–467. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nagakubo, T. Biological Functions and Applications of Virus-Related Bacterial Nanoparticles: A Review. Int. J. Mol. Sci. 2022, 23, 2595. https://doi.org/10.3390/ijms23052595
Nagakubo T. Biological Functions and Applications of Virus-Related Bacterial Nanoparticles: A Review. International Journal of Molecular Sciences. 2022; 23(5):2595. https://doi.org/10.3390/ijms23052595
Chicago/Turabian StyleNagakubo, Toshiki. 2022. "Biological Functions and Applications of Virus-Related Bacterial Nanoparticles: A Review" International Journal of Molecular Sciences 23, no. 5: 2595. https://doi.org/10.3390/ijms23052595





