Grainyhead-like (Grhl) Target Genes in Development and Cancer
Abstract
1. Introduction
2. Neural Tube Closure and Morphogenesis
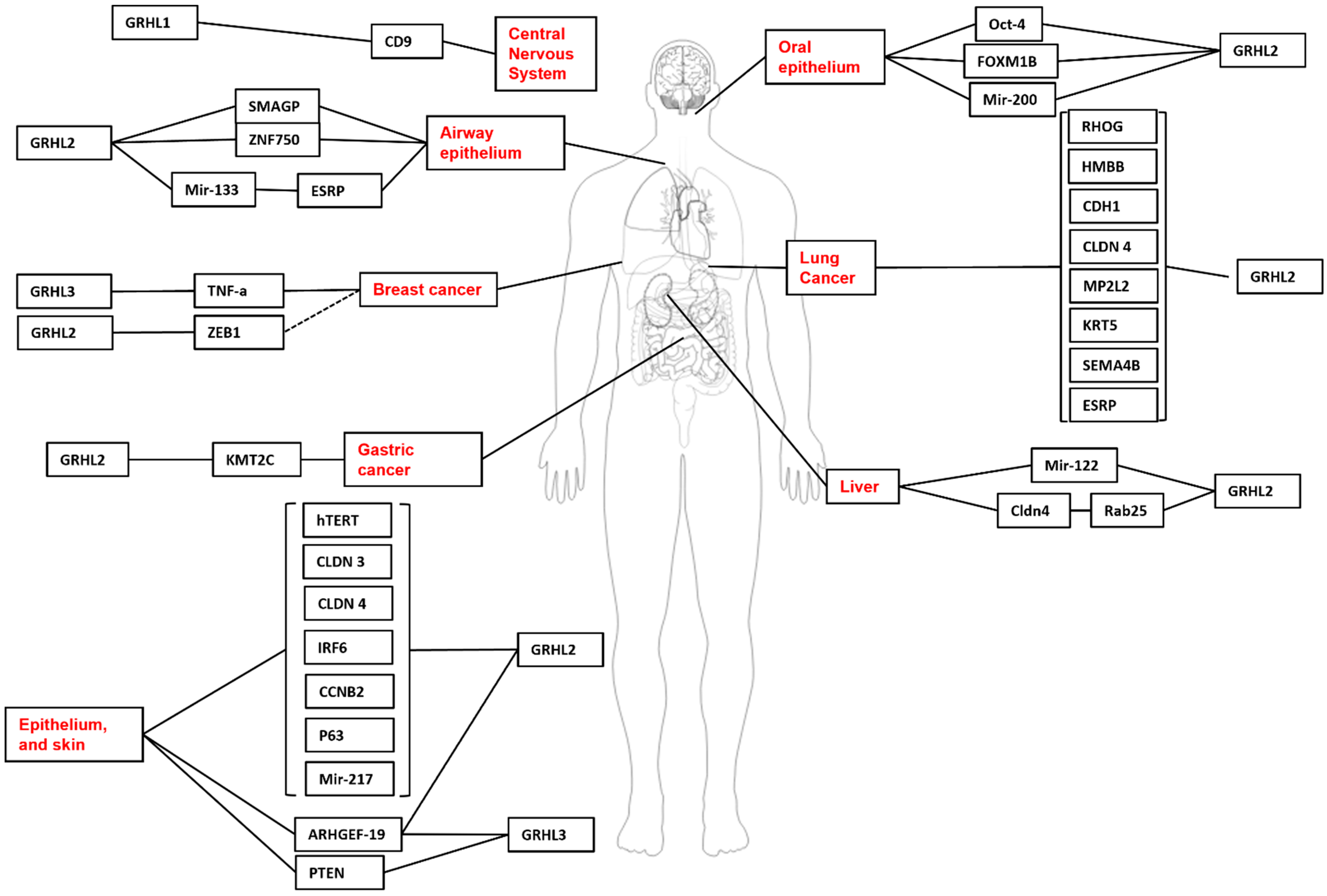
2.1. Brain—Neurogenesis, Morphogenesis and Cancer
2.2. Epidermal Development, Disease and Cancer
2.3. Craniofacial Development and Head and Neck Cancers
2.4. Kidney—Development and Cancer
2.5. Breast—Development and Cancer
3. Lung—Development and Cancer
4. Liver—Development and Cancer
4.1. Gastrointestinal Tract and Bladder—Development and Cancer
4.2. Development and Disease in Non-Epithelial Tissues
5. Endothelium
Placenta
6. Bone (Osteoblasts)
Funding
Acknowledgments
Conflicts of Interest
References
- Bray, S.J.; Kafatos, F.C. Developmental function of Elf-1: An essential transcription factor during embryogenesis in Drosophila. Genes Dev. 1991, 5, 1672–1683. [Google Scholar] [CrossRef] [PubMed]
- Wilanowski, T.; Tuckfield, A.; Cerruti, L.; O’Connell, S.; Saint, R.; Parekh, V.; Tao, J.; Cunningham, J.M.; Jane, S.M. A highly conserved novel family of mammalian developmental transcription factors related to Drosophila grainyhead. Mech. Dev. 2002, 114, 37–50. [Google Scholar] [CrossRef]
- Ting, S.B.; Wilanowski, T.; Auden, A.; Hall, M.; Voss, A.K.; Thomas, T.; Parekh, V.; Cunningham, J.M.; Jane, S.M. Inositol- and folate-resistant neural tube defects in mice lacking the epithelial-specific factor Grhl-3. Nat. Med. 2003, 9, 1513–1519. [Google Scholar] [CrossRef] [PubMed]
- Venkatesan, K.; McManus, H.R.; Mello, C.C.; Smith, T.F.; Hansen, U. Functional conservation between members of an ancient duplicated transcription factor family, LSF/Grainyhead. Nucleic Acids Res. 2003, 31, 4304–4316. [Google Scholar] [CrossRef]
- Jänicke, M.; Renisch, B.; Hammerschmidt, M. Zebrafish grainyhead-like1 is a common marker of different non-keratinocyte epidermal cell lineages, which segregate from each other in a Foxi3-dependent manner. Int. J. Dev. Biol. 2010, 54, 837–850. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Mu, Y.; Li, X.; Xu, P.; Tong, J.; Liu, Z.; Ma, T.; Zeng, G.; Yang, S.; Du, J.; et al. Grhl2 deficiency impairs otic development and hearing ability in a zebrafish model of the progressive dominant hearing loss DFNA28. Hum. Mol. Genet. 2011, 20, 3213–3226. [Google Scholar] [CrossRef]
- Dworkin, S.; Simkin, J.; Darido, C.; Partridge, D.D.; Georgy, S.R.; Caddy, J.; Wilanowski, T.; Lieschke, G.; Doggett, K.; Heath, J.; et al. Grainyhead-like 3 regulation of endothelin-1 in the pharyngeal endoderm is critical for growth and development of the craniofacial skeleton. Mech. Dev. 2014, 133, 77–90. [Google Scholar] [CrossRef]
- Paré, A.; Kim, M.; Juarez, M.T.; Brody, S.; McGinnis, W. The Functions of Grainy Head-Like Proteins in Animals and Fungi and the Evolution of Apical Extracellular Barriers. PLoS ONE 2012, 7, e36254. [Google Scholar] [CrossRef]
- Shelomi, M.; Tan, T.Z.; Huang, R.Y.-J. Evolution of CP2 transcription factors in Hexapoda. J. Genet. 2021, 100, 83. [Google Scholar] [CrossRef]
- Cieply, B.; Riley, P.; Pifer, P.M.; Widmeyer, J.; Addison, J.B.; Ivanov, A.; Denvir, J.; Frisch, S.M. Suppression of the Epithelial–Mesenchymal Transition by Grainyhead-like-2. Cancer Res. 2012, 72, 2440–2453. [Google Scholar] [CrossRef]
- Gao, X.; Vockley, C.M.; Pauli, F.; Newberry, K.M.; Xue, Y.; Randell, S.H.; Reddy, T.E.; Hogan, B.L.M. Evidence for multiple roles for grainyhead-like 2 in the establishment and maintenance of human mucociliary airway epithelium. Proc. Natl. Acad. Sci. USA 2013, 110, 9356–9361. [Google Scholar] [CrossRef] [PubMed]
- Peyrard-Janvid, M.; Leslie, E.J.; Kousa, M.Y.; Smith, T.L.; Dunnwald, M.; Magnusson, M.; Lentz, B.A.; Unneberg, P.; Fransson, I.; Koillinen, H.K.; et al. Dominant Mutations in GRHL3 Cause Van der Woude Syndrome and Disrupt Oral Periderm Development. Am. J. Hum. Genet. 2014, 94, 23–32. [Google Scholar] [CrossRef]
- Dworkin, S.; Darido, C.; Georgy, S.R.; Wilanowski, T.; Srivastava, S.; Ellett, F.; Pase, L.; Han, Y.; Meng, A.; Heath, J.; et al. Midbrain-hindbrain boundary patterning and morphogenesis are regulated by diverse grainy head-like 2-dependent pathways. Development 2012, 139, 525–536. [Google Scholar] [CrossRef] [PubMed]
- Carpinelli, M.; De Vries, M.; Jane, S.; Dworkin, S. Grainyhead-like Transcription Factors in Craniofacial Development. J. Dent. Res. 2017, 96, 1200–1209. [Google Scholar] [CrossRef] [PubMed]
- Werth, M.; Walentin, K.; Aue, A.; Schönheit, J.; Wuebken, A.; Pode-Shakked, N.; Vilianovitch, L.; Erdmann, B.; Dekel, B.; Bader, M.; et al. The transcription factor grainyhead-like 2 regulates the molecular composition of the epithelial apical junctional complex. Development 2010, 137, 3835–3845. [Google Scholar] [CrossRef] [PubMed]
- Menke, C.; Cionni, M.; Siggers, T.; Bulyk, M.L.; Beier, D.R.; Stottmann, R.W. Grhl2 is required in non-neural tissues for neural progenitor survival and forebrain development. Genesis 2015, 53, 573–582. [Google Scholar] [CrossRef]
- De Vries, M.; Carpinelli, M.; Rutland, E.; Hatzipantelis, A.; Partridge, D.; Auden, A.; Anderson, P.; De Groef, B.; Wu, H.; Osterwalder, M.; et al. Interrogating the Grainyhead-like 2 (Grhl2) genomic locus identifies an enhancer element that regulates palatogenesis in mouse. Dev. Biol. 2019, 459, 194–203. [Google Scholar] [CrossRef]
- De Vries, M.; Owens, H.G.; Carpinelli, M.R.; Partridge, D.; Kersbergen, A.; Sutherland, K.D.; Auden, A.; Anderson, P.J.; Jane, S.M.; Dworkin, S. Delineating the roles of Grhl2 in craniofacial development through tissue-specific conditional deletion and epistasis approaches in mouse. Dev. Dyn. 2021, 250, 1191–1209. [Google Scholar] [CrossRef]
- Kersbergen, A.; Best, S.; Dworkin, S.; Ah-Cann, C.; de Vries, M.E.; Asselin-Labat, M.-L.; Ritchie, M.E.; Jane, S.M.; Sutherland, K.D. Lung morphogenesis is orchestrated through Grainyhead-like 2 (Grhl2) transcriptional programs. Dev. Biol. 2018, 443, 1–9. [Google Scholar] [CrossRef]
- Ting, S.B.; Caddy, J.; Hislop, N.; Wilanowski, T.; Auden, A.; Zhao, L.-L.; Ellis, S.; Kaur, P.; Uchida, Y.; Holleran, W.M.; et al. A Homolog of Drosophila grainy head Is Essential for Epidermal Integrity in Mice. Science 2005, 308, 411–413. [Google Scholar] [CrossRef]
- Cangkrama, M.; Darido, C.; Georgy, S.R.; Partridge, D.; Auden, A.; Srivastava, S.; Wilanowski, T.; Jane, S.M. Two Ancient Gene Families Are Critical for Maintenance of the Mammalian Skin Barrier in Postnatal Life. J. Investig. Dermatol. 2016, 136, 1438–1448. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, J.; Atkins, M.; Davie, K.; Imrichova, H.; Romanelli, L.; Christiaens, V.; Hulselmans, G.; Potier, D.; Wouters, J.; Taskiran, I.I.; et al. The transcription factor Grainy head primes epithelial enhancers for spatiotemporal activation by displacing nucleosomes. Nat. Genet. 2018, 50, 1011–1020. [Google Scholar] [CrossRef] [PubMed]
- Nevil, M.; Bondra, E.R.; Schulz, K.N.; Kaplan, T.; Harrison, M.M. Stable Binding of the Conserved Transcription Factor Grainy Head to its Target Genes Throughout Drosophila melanogaster Development. Genetics 2017, 205, 605–620. [Google Scholar] [CrossRef] [PubMed]
- Sundararajan, V.; Pang, Q.Y.; Choolani, M.; Huang, R.Y.-J. Spotlight on the Granules (Grainyhead-Like Proteins)—From an Evolutionary Conserved Controller of Epithelial Trait to Pioneering the Chromatin Landscape. Front. Mol. Biosci. 2020, 7, 213. [Google Scholar] [CrossRef] [PubMed]
- Kotarba, G.; Taracha-Wisniewska, A.; Wilanowski, T. Grainyhead-like transcription factors in cancer—Focus on recent developments. Exp. Biol. Med. 2020, 245, 402–410. [Google Scholar] [CrossRef] [PubMed]
- Darido, C.; Georgy, S.R.; Wilanowski, T.; Dworkin, S.; Auden, A.; Zhao, Q.; Rank, G.; Srivastava, S.; Finlay, M.J.; Papenfuss, A.T.; et al. Targeting of the Tumor Suppressor GRHL3 by a miR-21-Dependent Proto-Oncogenic Network Results in PTEN Loss and Tumorigenesis. Cancer Cell 2011, 20, 635–648. [Google Scholar] [CrossRef]
- Georgy, S.R.; Cangkrama, M.; Srivastava, S.; Partridge, D.; Auden, A.; Dworkin, S.; McLean, C.A.; Jane, S.M.; Darido, C. Identification of a Novel Proto-oncogenic Network in Head and Neck Squamous Cell Carcinoma. J. Natl. Cancer Inst. 2015, 107, djv152. [Google Scholar] [CrossRef]
- Xu, H.; Liu, C.; Zhao, Z.; Gao, N.; Chen, G.; Wang, Y.; Cui, J. Clinical implications of GRHL3 protein expression in breast cancer. Tumor Biol. 2014, 35, 1827–1831. [Google Scholar] [CrossRef]
- Bhandari, A.; Gordon, W.; Dizon, D.; Hopkin, A.S.; Gordon, E.; Yu, Z.; Andersen, B. The Grainyhead transcription factor Grhl3/Get1 suppresses miR-21 expression and tumorigenesis in skin: Modulation of the miR-21 target MSH2 by RNA-binding protein DND1. Oncogene 2012, 32, 1497–1507. [Google Scholar] [CrossRef]
- Kikulska, A.; Rausch, T.; Krzywińska, E.; Pawlak, M.; Wilczyński, B.; Benes, V.; Rutkowski, P.; Wilanowski, T. Coordinated expression and genetic polymorphisms in Grainyhead-like genes in human non-melanoma skin cancers. BMC Cancer 2018, 18, 23. [Google Scholar] [CrossRef]
- Tanaka, Y.; Kanai, F.; Tada, M.; Tateishi, R.; Sanada, M.; Nannya, Y.; Ohta, M.; Asaoka, Y.; Seto, M.; Shiina, S.; et al. Gain of GRHL2 is associated with early recurrence of hepatocellular carcinoma. J. Hepatol. 2008, 49, 746–757. [Google Scholar] [CrossRef] [PubMed]
- Werner, S.; Frey, S.; Riethdorf, S.; Schulze, C.; Alawi, M.; Kling, L.; Vafaizadeh, V.; Sauter, G.; Terracciano, L.; Schumacher, U.; et al. Dual Roles of the Transcription Factor Grainyhead-like 2 (GRHL2) in Breast Cancer. J. Biol. Chem. 2013, 288, 22993–23008. [Google Scholar] [CrossRef]
- Quan, Y.; Xu, M.; Cui, P.; Ye, M.; Zhuang, B.; Min, Z. Grainyhead-like 2 Promotes Tumor Growth and is Associated with Poor Prognosis in Colorectal Cancer. J. Cancer 2015, 6, 342–350. [Google Scholar] [CrossRef] [PubMed]
- Quan, Y.; Jin, R.; Huang, A.; Zhao, H.; Feng, B.; Zang, L.; Zheng, M. Downregulation of GRHL2 inhibits the proliferation of colorectal cancer cells by targeting ZEB1. Cancer Biol. Ther. 2014, 15, 878–887. [Google Scholar] [CrossRef]
- Nishino, H.; Takano, S.; Yoshitomi, H.; Suzuki, K.; Kagawa, S.; Shimazaki, R.; Shimizu, H.; Furukawa, K.; Miyazaki, M.; Ohtsuka, M. Grainyhead-like 2 (GRHL2) regulates epithelial plasticity in pancreatic cancer progression. Cancer Med. 2017, 6, 2686–2696. [Google Scholar] [CrossRef]
- Pifer, P.M.; Farris, J.C.; Thomas, A.L.; Stoilov, P.; Denvir, J.; Smith, D.M.; Frisch, S.M. Grainyhead-like 2 inhibits the coactivator p300, suppressing tubulogenesis and the epithelial-mesenchymal transition. Mol. Biol. Cell. 2016, 27, 2479–2492. [Google Scholar] [CrossRef] [PubMed]
- Farris, J.C.; Pifer, P.M.; Zheng, L.; Gottlieb, E.; Denvir, J.; Frisch, S.M. Grainyhead-like 2 Reverses the Metabolic Changes Induced by the Oncogenic Epithelial–Mesenchymal Transition: Effects on Anoikis. Mol. Cancer Res. 2016, 14, 528–538. [Google Scholar] [CrossRef] [PubMed]
- Chung, V.Y.; Tan, T.Z.; Tan, M.; Wong, M.K.; Kuay, K.T.; Yang, Z.; Ye, J.; Muller, J.; Koh, C.M.; Guccione, E.; et al. GRHL2-miR-200-ZEB1 maintains the epithelial status of ovarian cancer through transcriptional regulation and histone modification. Sci. Rep. 2016, 6, 19943. [Google Scholar] [CrossRef]
- Fabian, J.; Lodrini, M.; Oehme, I.; Schier, M.C.; Thole, T.M.; Hielscher, T.; Kopp-Schneider, A.; Opitz, L.; Capper, D.; von Deimling, A.; et al. GRHL1 Acts as Tumor Suppressor in Neuroblastoma and Is Negatively Regulated by MYCN and HDAC3. Cancer Res. 2014, 74, 2604–2616. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Li, Z.; Guan, X.; Qin, Y. Suppressor gene GRHL1 is associated with prognosis in patients with oesophageal squamous cell carcinoma. Oncol. Lett. 2019, 17, 4313–4320. [Google Scholar] [CrossRef]
- Mlacki, M.; Darido, C.; Jane, S.M.; Wilanowski, T. Loss of Grainy Head-Like 1 Is Associated with Disruption of the Epidermal Barrier and Squamous Cell Carcinoma of the Skin. PLoS ONE 2014, 9, e89247. [Google Scholar] [CrossRef] [PubMed]
- Mathiyalagan, N.; Miles, L.B.; Anderson, P.J.; Wilanowski, T.; Grills, B.L.; McDonald, S.; Keightley, M.C.; Charzynska, A.; Dabrowski, M.; Dworkin, S. Meta-Analysis of Grainyhead-Like Dependent Transcriptional Networks: A Roadmap for Identifying Novel Conserved Genetic Pathways. Genes 2019, 10, 876. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Feng, C.; Zhu, H.; Wu, S.; Jin, P.; Xu, T. Grainyhead-like 2 as a double-edged sword in development and cancer. Am. J. Transl. Res. 2020, 12, 310–331. [Google Scholar] [PubMed]
- Dynlacht, B.D.; Attardi, L.D.; Admon, A.; Freeman, M.; Tjian, R. Functional analysis of NTF-1, a developmentally regulated Drosophila transcription factor that binds neuronal cis elements. Genes Dev. 1989, 3, 1677–1688. [Google Scholar] [CrossRef] [PubMed]
- Dynlacht, B.D.; Hoey, T.; Tjian, R. Isolation of coactivators associated with the TATA-binding protein that mediate transcriptional activation. Cell 1991, 66, 563–576. [Google Scholar] [CrossRef]
- Mace, K.A.; Pearson, J.C.; McGinnis, W. An Epidermal Barrier Wound Repair Pathway in Drosophila Is Mediated by grainy head. Science 2005, 308, 381–385. [Google Scholar] [CrossRef]
- Uv, A.E.; Thompson, C.R.; Bray, S.J. The Drosophila tissue-specific factor Grainyhead contains novel DNA-binding and dimerization domains which are conserved in the human protein CP2. Mol. Cell. Biol. 1994, 14, 4020–4031. [Google Scholar] [PubMed]
- Liaw, G.J.; Rudolph, K.M.; Huang, J.D.; Dubnicoff, T.; Courey, A.J.; Lengyel, J.A. The torso response element binds GAGA and NTF-1/Elf-1, and regulates tailless by relief of repression. Genes Dev. 1995, 9, 3163–3176. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.D.; Dubnicoff, T.; Liaw, G.J.; Bai, Y.; Valentine, S.A.; Shirokawa, J.M.; Lengyel, J.A.; Courey, A.J. Binding sites for transcription factor NTF-1/Elf-1 contribute to the ventral repression of decapentaplegic. Genes Dev. 1995, 9, 3177–3189. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, Y.; Yamagishi, M.; Nishimoto, Y.; Taguchi, O.; Matsukage, A.; Yamaguchi, M. A Binding Site for the Transcription Factor Grainyhead/Nuclear Transcription Factor-1 Contributes to Regulation of theDrosophila Proliferating Cell Nuclear Antigen Gene Promoter. J. Biol. Chem. 1999, 274, 35080–35088. [Google Scholar] [CrossRef]
- Brody, T.; Odenwald, W.F. Programmed Transformations in Neuroblast Gene Expression during Drosophila CNS Lineage Development. Dev. Biol. 2000, 226, 34–44. [Google Scholar] [CrossRef] [PubMed]
- Fuss, B.; Hoch, M. Notch signaling controls cell fate specification along the dorsoventral axis of the Drosophila gut. Curr. Biol. 2002, 12, 171–179. [Google Scholar] [CrossRef][Green Version]
- Ostrowski, S.; Dierick, H.A.; Bejsovec, A. Genetic control of cuticle formation during embryonic development of Drosophila melanogaster. Genetics 2002, 161, 171–182. [Google Scholar] [CrossRef] [PubMed]
- Hemphäla, J.; Uv, A.; Cantera, R.; Bray, S.; Samakovlis, C. Grainy head controls apical membrane growth and tube elongation in response to Branchless/FGF signalling. Development 2003, 130, 249–258. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Adler, P.N. The grainy head transcription factor is essential for the function of the frizzled pathway in the Drosophila wing. Mech. Dev. 2003, 121, 37–49. [Google Scholar] [CrossRef] [PubMed]
- Cenci, C.; Gould, A.P. Drosophila Grainyhead specifies late programmes of neural proliferation by regulating the mitotic activity and Hox-dependent apoptosis of neuroblasts. Development 2005, 132, 3835–3845. [Google Scholar] [CrossRef] [PubMed]
- Almeida, M.; Bray, S. Regulation of post-embryonic neuroblasts by Drosophila Grainyhead. Mech. Dev. 2005, 122, 1282–1293. [Google Scholar] [CrossRef] [PubMed]
- Blastyák, A.; Mishra, R.K.; Karch, F.; Gyurkovics, H. Efficient and Specific Targeting of Polycomb Group Proteins Requires Cooperative Interaction between Grainyhead and Pleiohomeotic. Mol. Cell. Biol. 2006, 26, 1434–1444. [Google Scholar] [CrossRef] [PubMed]
- Lattorff, H.M.; Moritz, R.F.; Crewe, R.M.; Solignac, M. Control of reproductive dominance by the thelytoky gene in honeybees. Biol. Lett. 2007, 3, 292–295. [Google Scholar] [CrossRef] [PubMed]
- Narasimha, M.; Uv, A.; Krejci, A.; Brown, N.H.; Bray, S. Grainy head promotes expression of septate junction proteins and influences epithelial morphogenesis. J. Cell Sci. 2008, 121, 747–752. [Google Scholar] [CrossRef] [PubMed]
- Florence, B.L.; Faller, D.V. Drosophila female sterile (1) homeotic is a multifunctional transcriptional regulator that is modulated by Ras signaling. Dev. Dyn. 2008, 237, 554–564. [Google Scholar] [CrossRef] [PubMed]
- Garcia, M.; Stathopoulos, A. Lateral Gene Expression in Drosophila Early Embryos Is Supported by Grainyhead-Mediated Activation and Tiers of Dorsally-Localized Repression. PLoS ONE 2011, 6, e29172. [Google Scholar] [CrossRef]
- Juarez, M.T.; Patterson, R.A.; Sandoval-Guillen, E.; McGinnis, W. Duox, Flotillin-2, and Src42A Are Required to Activate or Delimit the Spread of the Transcriptional Response to Epidermal Wounds in Drosophila. PLoS Genet. 2011, 7, e1002424. [Google Scholar] [CrossRef] [PubMed]
- Tao, J.; Kuliyev, E.; Wang, X.; Li, X.; Wilanowski, T.; Jane, S.M.; Mead, P.E.; Cunningham, J.M. BMP4-dependent expression of Xenopus Grainyhead-like 1 is essential for epidermal differentiation. Development 2005, 132, 1021–1034. [Google Scholar] [CrossRef] [PubMed]
- De la Garza, G.; Schleiffarth, J.R.; Dunnwald, M.; Mankad, A.; Weirather, J.L.; Bonde, G.; Butcher, S.; Mansour, T.A.; Kousa, Y.A.; Fukazawa, C.F.; et al. Interferon Regulatory Factor 6 Promotes Differentiation of the Periderm by Activating Expression of Grainyhead-Like 3. J. Investig. Dermatol. 2012, 133, 68–77. [Google Scholar]
- Miles, L.B.; Darido, C.; Kaslin, J.; Heath, J.K.; Jane, S.M.; Dworkin, S. Mis-expression of grainyhead-like transcription factors in zebrafish leads to defects in enveloping layer (EVL) integrity, cellular morphogenesis and axial extension. Sci. Rep. 2017, 7, 17607. [Google Scholar] [CrossRef] [PubMed]
- Kudryavtseva, E.I.; Sugihara, T.M.; Wang, N.; Lasso, R.J.; Gudnason, J.F.; Lipkin, S.M.; Andersen, B. Identification and characterization of Grainyhead-like epithelial transactivator (GET-1), a novel mammalian Grainyhead-like factor. Dev. Dyn. 2003, 226, 604–617. [Google Scholar] [CrossRef] [PubMed]
- Cogram, P.; Hynes, A.; Dunlevy, L.P.; Greene, N.; Copp, A. Specific isoforms of protein kinase C are essential for prevention of folate-resistant neural tube defects by inositol. Hum. Mol. Genet. 2003, 13, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Brynczka, C.; Labhart, P.; Merrick, B.A. NGF-mediated transcriptional targets of p53 in PC12 neuronal differentiation. BMC Genom. 2007, 8, 139. [Google Scholar] [CrossRef] [PubMed]
- Wilanowski, T.; Caddy, J.; Ting, S.B.; Hislop, N.R.; Cerruti, L.; Auden, A.; Zhao, L.-L.; Asquith, S.; Ellis, S.; Sinclair, R.; et al. Perturbed desmosomal cadherin expression in grainy head-like 1-null mice. EMBO J. 2008, 27, 886–897. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Bhandari, A.; Mannik, J.; Pham, T.; Xu, X.; Andersen, B. Grainyhead-like factor Get1/Grhl3 regulates formation of the epidermal leading edge during eyelid closure. Dev. Biol. 2008, 319, 56–67. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Mannik, J.; Soto, A.; Lin, K.K.; Andersen, B. The epidermal differentiation-associated Grainyhead gene Get1/Grhl3 also regulates urothelial differentiation. EMBO J. 2009, 28, 1890–1903. [Google Scholar] [CrossRef] [PubMed]
- Aue, A.; Hinze, C.; Walentin, K.; Ruffert, J.; Yurtdas, Y.; Werth, M.; Chen, W.; Rabien, A.; Kilic, E.; Schulzke, J.D.; et al. A Grainyhead-Like 2/Ovo-Like 2 Pathway Regulates Renal Epithelial Barrier Function and Lumen Expansion. J. Am. Soc. Nephrol. 2015, 26, 2704–2715. [Google Scholar] [CrossRef] [PubMed]
- Hinze, C.; Ruffert, J.; Walentin, K.; Himmerkus, N.; Nikpey, E.; Tenstad, O.; Wiig, H.; Mutig, K.; Yurtdas, Z.Y.; Klein, J.D.; et al. GRHL2 Is Required for Collecting Duct Epithelial Barrier Function and Renal Osmoregulation. J. Am. Soc. Nephrol. 2017, 29, 857–868. [Google Scholar] [CrossRef] [PubMed]
- Caddy, J.; Wilanowski, T.; Darido, C.; Dworkin, S.; Ting, S.B.; Zhao, Q.; Rank, G.; Auden, A.; Srivastava, S.; Papenfuss, T.A.; et al. Epidermal wound repair is regulated by the planar cell polarity signaling pathway. Dev. Cell 2010, 19, 138–147. [Google Scholar] [CrossRef][Green Version]
- De Castro, S.C.P.; Malhas, A.; Leung, K.-Y.; Gustavsson, P.; Vaux, D.J.; Copp, A.J.; Greene, N.D.E. Lamin B1 Polymorphism Influences Morphology of the Nuclear Envelope, Cell Cycle Progression, and Risk of Neural Tube Defects in Mice. PLoS Genet. 2012, 8, e1003059. [Google Scholar] [CrossRef] [PubMed]
- Varma, S.; Cao, Y.; Tagne, J.-B.; Lakshminarayanan, M.; Li, J.; Friedman, T.B.; Morell, R.J.; Warburton, D.; Kotton, D.N.; Ramirez, M.I. The Transcription Factors Grainyhead-like 2 and NK2-Homeobox 1 Form a Regulatory Loop That Coordinates Lung Epithelial Cell Morphogenesis and Differentiation. J. Biol. Chem. 2012, 287, 37282–37295. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Liu, Z.X.; Oh, J.-E.; Shin, K.-H.; Kim, R.H.; Jiang, M.; Park, N.-H.; Kang, M.K. Grainyhead-like 2 (GRHL2) inhibits keratinocyte differentiation through epigenetic mechanism. Cell Death Dis. 2012, 3, e450. [Google Scholar] [CrossRef]
- Tanimizu, N.; Mitaka, T. Role of grainyhead-like 2 in the formation of functional tight junctions. Tissue Barriers 2013, 1, e23495. [Google Scholar] [CrossRef] [PubMed]
- Pawlak, M.; Walkowska, A.; Mlącki, M.; Pistolic, J.; Wrzesiński, T.; Benes, V.; Jane, S.M.; Wesoły, J.; Kompanowska-Jezierska, E.; Wilanowski, T. Consequences of the loss of the Grainyhead-like 1 gene for renal gene expression, regulation of blood pressure and heart rate in a mouse model. Acta Biochim. Pol. 2015, 62, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Walentin, K.; Hinze, C.; Werth, M.; Haase, N.; Varma, S.; Morell, R.; Aue, A.; Pötschke, E.; Warburton, D.; Qiu, A.; et al. A Grhl2-dependent gene network controls trophoblast branching morphogenesis. Development 2015, 142, 1125–1136. [Google Scholar] [CrossRef] [PubMed]
- Matsushita, T.; Sakai, M.; Yoshida, H.; Morita, S.; Hieda, Y.; Sakai, T. Grhl2 regulation of SPINT1 expression controls salivary gland development. Biochem. Biophys. Res. Commun. 2018, 504, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Kashgari, G.; Venkatesh, S.; Refuerzo, S.; Pham, B.; Bayat, A.; Klein, R.H.; Ramos, R.; Ta, A.P.; Plikus, M.V.; Wang, P.H.; et al. GRHL3 activates FSCN1 to relax cell-cell adhesions between migrating keratinocytes during wound reepithelialization. JCI Insight 2021, 6, e142577. [Google Scholar] [CrossRef]
- Guardiola-Serrano, F.; Haendeler, J.; Lukosz, M.; Sturm, K.; von Melchner, H.; Altschmied, J. Gene trapping identifies a putative tumor suppressor and a new inducer of cell migration. Biochem. Biophys. Res. Commun. 2008, 376, 748–752. [Google Scholar] [CrossRef]
- Kang, X.; Chen, W.; Kim, R.H.; Kang, M.K.; Park, N.-H. Regulation of the hTERT promoter activity by MSH2, the hnRNPs K and D, and GRHL2 in human oral squamous cell carcinoma cells. Oncogene 2008, 28, 565–574. [Google Scholar] [CrossRef]
- Lukosz, M.; Mlynek, A.; Czypiorski, P.; Altschmied, J.; Haendeler, J. The transcription factor Grainyhead like 3 (GRHL3) affects endothelial cell apoptosis and migration in a NO-dependent manner. Biochem. Biophys. Res. Commun. 2011, 412, 648–653. [Google Scholar] [CrossRef]
- Senga, K.; Mostov, K.E.; Mitaka, T.; Miyajima, A.; Tanimizu, N. Grainyhead-like 2 regulates epithelial morphogenesis by establishing functional tight junctions through the organization of a molecular network among claudin3, claudin4, and Rab25. Mol. Biol. Cell 2012, 23, 2845–2855. [Google Scholar] [CrossRef]
- Xiang, X.; Deng, Z.; Zhuang, X.; Ju, S.; Mu, J.; Jiang, H.; Zhang, L.; Yan, J.; Miller, D.; Zhang, H.G. Grhl2 determines the epithelial phenotype of breast cancers and promotes tumor progression. PLoS ONE 2012, 7, e50781. [Google Scholar] [CrossRef]
- Gao, X.; Bali, A.S.; Randell, S.H.; Hogan, B.L. GRHL2 coordinates regeneration of a polarized mucociliary epithelium from basal stem cells. J. Cell. Biol. 2015, 211, 669–682. [Google Scholar] [CrossRef]
- Tanimizu, N.; Kobayashi, S.; Ichinohe, N.; Mitaka, T. Downregulation of miR122 by grainyhead-like 2 restricts the hepatocytic differentiation potential of adult liver progenitor cells. Development 2014, 141, 4448–4456. [Google Scholar] [CrossRef]
- Mehrazarin, S.; Chen, W.; Oh, J.-E.; Liu, Z.X.; Kang, K.L.; Yi, J.K.; Kim, R.H.; Shin, K.-H.; Park, N.-H.; Kang, M.K. The p63 Gene Is Regulated by Grainyhead-like 2 (GRHL2) through Reciprocal Feedback and Determines the Epithelial Phenotype in Human Keratinocytes. J. Biol. Chem. 2015, 290, 19999–20008. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Hou, L.; Liu, J.; Li, Z. MiR-217 is down-regulated in psoriasis and promotes keratinocyte differentiation via targeting GRHL2. Biochem. Biophys. Res. Commun. 2016, 471, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Yi, J.K.; Shimane, T.; Mehrazarin, S.; Lin, Y.-L.; Shin, K.-H.; Kim, R.H.; Park, N.-H.; Kang, M.K. Grainyhead-like 2 regulates epithelial plasticity and stemness in oral cancer cells. Carcinogenesis 2016, 37, 500–510. [Google Scholar] [CrossRef] [PubMed]
- Xiang, J.; Fu, X.; Ran, W.; Wang, Z. Grhl2 reduces invasion and migration through inhibition of TGFbeta-induced EMT in gastric cancer. Oncogenesis 2017, 6, e284. [Google Scholar] [CrossRef]
- Yu, X.; An, J.; Hua, Y.; Li, Z.; Yan, N.; Fan, W.; Su, C. MicroRNA-194 regulates keratinocyte proliferation and differentiation by targeting Grainyhead-like 2 in psoriasis. Pathol. Res. Pract. 2017, 213, 89–97. [Google Scholar] [CrossRef]
- Pan, X.; Zhang, R.; Xie, C.; Gan, M.; Yao, S.; Yao, Y.; Jin, J.; Han, T.; Huang, Y.; Gong, Y.; et al. GRHL2 suppresses tumor metastasis via regulation of transcriptional activity of RhoG in non-small cell lung cancer. Am. J. Transl. Res. 2017, 9, 4217–4226. [Google Scholar]
- Chen, L.; He, X.; Xie, Y.; Huang, Y.; Wolff, D.W.; Abel, P.W.; Tu, Y. Up-regulated miR-133a orchestrates epithelial-mesenchymal transition of airway epithelial cells. Sci. Rep. 2018, 8, 15543. [Google Scholar] [CrossRef]
- MacFawn, I.; Wilson, H.; Selth, L.A.; Leighton, I.; Serebriiskii, I.; Bleackley, R.C.; Elzamzamy, O.; Farris, J.; Pifer, P.M.; Richer, J.; et al. Grainyhead-like-2 confers NK-sensitivity through interactions with epigenetic modifiers. Mol. Immunol. 2018, 105, 137–149. [Google Scholar] [CrossRef]
- Jander, K.; Greulich, J.; Gonnissen, S.; Ale-Agha, N.; Goy, C.; Jakobs, P.; Farrokh, S.; Marziano, C.; Sonkusare, S.; Haendeler, J.; et al. Extra-Nuclear Functions of the Transcription Factor Grainyhead-Like 3 in the Endothelium—Interaction with Endothelial Nitric Oxide Synthase. Antioxidants 2021, 10, 428. [Google Scholar] [CrossRef]
- He, Y.; Gan, M.; Wang, Y.; Huang, T.; Wang, J.; Han, T.; Yu, B. EGFR-ERK induced activation of GRHL1 promotes cell cycle progression by up-regulating cell cycle related genes in lung cancer. Cell Death Dis. 2021, 12, 430. [Google Scholar] [CrossRef]
- Głowacka, A.; Bieganowski, P.; Jurewicz, E.; Leśniak, W.; Wilanowski, T.; Filipek, A. Regulation of S100A10 Gene Expression. Biomolecules 2021, 11, 974. [Google Scholar] [CrossRef] [PubMed]
- Greene, N.D.E.; Copp, A.J. Development of the vertebrate central nervous system: Formation of the neural tube. Prenat. Diagn. 2009, 29, 303–311. [Google Scholar] [CrossRef]
- Ybot-Gonzalez, P.; Savery, D.; Gerrelli, D.; Signore, M.; Mitchell, C.E.; Faux, C.H.; Greene, N.; Copp, A.J. Convergent extension, planar-cell-polarity signalling and initiation of mouse neural tube closure. Development 2007, 134, 789–799. [Google Scholar] [CrossRef] [PubMed]
- Gustavsson, P.; Greene, N.; Lad, D.; Pauws, E.; De Castro, S.C.; Stanier, P.; Copp, A. Increased expression of Grainyhead-like-3 rescues spina bifida in a folate-resistant mouse model. Hum. Mol. Genet. 2007, 16, 2640–2646. [Google Scholar] [CrossRef] [PubMed]
- Rifat, Y.; Parekh, V.; Wilanowski, T.; Hislop, N.R.; Auden, A.; Ting, S.B.; Cunningham, J.M.; Jane, S.M. Regional neural tube closure defined by the Grainy head-like transcription factors. Dev. Biol. 2010, 345, 237–245. [Google Scholar] [CrossRef]
- Brouns, M.R.; De Castro, S.C.; Terwindt-Rouwenhorst, E.A.; Massa, V.; Hekking, J.W.; Hirst, C.S.; Savery, D.; Munts, C.; Partridge, D.; Lamers, W.; et al. Over-expression of Grhl2 causes spina bifida in the Axial defects mutant mouse. Hum. Mol. Genet. 2011, 20, 1536–1546. [Google Scholar] [CrossRef]
- Pyrgaki, C.; Liu, A.; Niswander, L. Grainyhead-like 2 regulates neural tube closure and adhesion molecule expression during neural fold fusion. Dev. Biol. 2011, 353, 38–49. [Google Scholar] [CrossRef]
- Ray, H.J.; Niswander, L. Grainyhead-like 2 downstream targets act to suppress epithelial-to-mesenchymal transition during neural tube closure. J. Cell Sci. 2016, 129, e1.2. [Google Scholar] [CrossRef]
- Nikolopoulou, E.; Hirst, C.S.; Galea, G.; Venturini, C.; Moulding, D.; Marshall, A.R.; Rolo, A.; De Castro, S.C.P.; Copp, A.J.; Greene, N.D.E. Spinal neural tube closure depends on regulation of surface ectoderm identity and biomechanics by Grhl2. Nat. Commun. 2019, 10, 2487. [Google Scholar] [CrossRef]
- Hashimoto, H.; Munro, E. Differential Expression of a Classic Cadherin Directs Tissue-Level Contractile Asymmetry during Neural Tube Closure. Dev. Cell 2019, 51, 158–172.e4. [Google Scholar] [CrossRef]
- Yu, Z.; Lin, K.K.; Bhandari, A.; Spencer, J.A.; Xu, X.; Wang, N.; Lu, Z.; Gill, G.N.; Roop, D.R.; Wertz, P.; et al. The Grainyhead-like epithelial transactivator Get-1/Grhl3 regulates epidermal terminal differentiation and interacts functionally with LMO4. Dev. Biol. 2006, 299, 122–136. [Google Scholar] [CrossRef] [PubMed]
- Van Straaten, H.W.; Copp, A.J. Curly tail: A 50-year history of the mouse spina bifida model. Anat. Embryo. 2001, 203, 225–237. [Google Scholar] [CrossRef] [PubMed]
- Lemay, P.; De Marco, P.; Emond, A.; Spiegelman, D.; Dionne-Laporte, A.; Laurent, S.; Merello, E.; Accogli, A.; Rouleau, G.; Capra, V.; et al. Rare deleterious variants in GRHL3 are associated with human spina bifida. Hum. Mutat. 2017, 38, 716–724. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Xiao, Y.; Tian, T.; Jin, L.; Wang, L.; Ren, A. Genetic variants in GRHL3 and risk for neural tube defects: A case–control and case–parent triad/control study. Birth Defects Res. 2018, 111, 1468–1478. [Google Scholar] [CrossRef] [PubMed]
- Tian, T.; Wang, L.; Shen, Y.; Zhang, B.; Finnell, R.H.; Ren, A. Hypomethylation of GRHL3 gene is associated with the occurrence of neural tube defects. Epigenomics 2018, 10, 891–901. [Google Scholar] [CrossRef] [PubMed]
- De Castro, S.C.P.; Gustavsson, P.; Marshall, A.R.; Gordon, W.M.; Galea, G.; Nikolopoulou, E.; Savery, D.; Rolo, A.; Stanier, P.; Andersen, B.; et al. Overexpression of Grainyhead-like 3 causes spina bifida and interacts genetically with mutant alleles of Grhl2 and Vangl2 in mice. Hum. Mol. Genet. 2018, 27, 4218–4230. [Google Scholar] [CrossRef]
- Kimura-Yoshida, C.; Mochida, K.; Ellwanger, K.; Niehrs, C.; Matsuo, I. Fate Specification of Neural Plate Border by Canonical Wnt Signaling and Grhl3 is Crucial for Neural Tube Closure. EBioMedicine 2015, 2, 513–527. [Google Scholar] [CrossRef]
- Zhou, C.J.; Ji, Y.; Reynolds, K.; McMahon, M.; Garland, M.A.; Zhang, S.; Sun, B.; Gu, R.; Islam, M.; Liu, Y.; et al. Non-neural surface ectodermal rosette formation and F-actin dynamics drive mammalian neural tube closure. Biochem. Biophys. Res. Commun. 2020, 526, 647–653. [Google Scholar] [CrossRef]
- Jaffe, E.; Niswander, L. Loss of Grhl3 is correlated with altered cellular protrusions in the non-neural ectoderm during neural tube closure. Dev. Dyn. 2020, 250, 732–744. [Google Scholar] [CrossRef]
- Dworkin, S.; Auden, A.; Partridge, D.D.; Daglas, M.; Medcalf, R.L.; Mantamadiotis, T.; Georgy, S.R.; Darido, C.; Jane, S.M.; Ting, S.B. Grainyhead-like 3 (Grhl3 ) deficiency in brain leads to altered locomotor activity and decreased anxiety-like behaviors in aged mice. Dev. Neurobiol. 2017, 77, 775–788. [Google Scholar] [CrossRef]
- Dworkin, S.; Jane, S.M. Novel mechanisms that pattern and shape the midbrain-hindbrain boundary. Cell. Mol. Life Sci. 2013, 70, 3365–3374. [Google Scholar] [CrossRef] [PubMed]
- Auden, A.; Caddy, J.; Wilanowski, T.; Ting, S.B.; Cunningham, J.M.; Jane, S.M. Spatial and temporal expression of the Grainyhead-like transcription factor family during murine development. Gene Expr. Patterns 2006, 6, 964–970. [Google Scholar] [CrossRef] [PubMed]
- Dworkin, S.; Jane, S.M.; Darido, C. The planar cell polarity pathway in vertebrate epidermal development, homeostasis and repair. Organogenesis 2011, 7, 202–208. [Google Scholar] [CrossRef]
- Cangkrama, M.; Ting, S.B.; Darido, C. Stem Cells behind the Barrier. Int. J. Mol. Sci. 2013, 14, 13670–13686. [Google Scholar] [CrossRef] [PubMed]
- Boglev, Y.; Wilanowski, T.; Caddy, J.; Parekh, V.; Auden, A.; Darido, C.; Hislop, N.R.; Cangkrama, M.; Ting, S.B.; Jane, S.M. The unique and cooperative roles of the Grainy head-like transcription factors in epidermal development reflect unexpected target gene specificity. Dev. Biol. 2011, 349, 512–522. [Google Scholar] [CrossRef] [PubMed]
- Petrof, G.; Nanda, A.; Howden, J.; Takeichi, T.; McMillan, J.R.; Aristodemou, S.; Ozoemena, L.; Liu, L.; South, A.P.; Pourreyron, C.; et al. Mutations in GRHL2 Result in an Autosomal-Recessive Ectodermal Dysplasia Syndrome. Am. J. Hum. Genet. 2014, 95, 308–314. [Google Scholar] [CrossRef]
- Goldie, S.J.; Cottle, D.L.; Tan, F.H.; Roslan, S.; Srivastava, S.; Brady, R.; Partridge, D.D.; Auden, A.; Smyth, I.; Jane, S.M.; et al. Loss of GRHL3 leads to TARC/CCL17-mediated keratinocyte proliferation in the epidermis. Cell Death Dis. 2018, 9, 1072. [Google Scholar] [CrossRef] [PubMed]
- Darido, C.; Georgy, S.R.; Cullinane, C.; Partridge, D.D.; Walker, R.; Srivastava, S.; Roslan, S.; Carpinelli, M.R.; Dworkin, S.; Pearson, R.B.; et al. Stage-dependent therapeutic efficacy in PI3K/mTOR-driven squamous cell carcinoma of the skin. Cell Death Differ. 2018, 25, 1146–1159. [Google Scholar] [CrossRef]
- Youssef, M.; Cuddihy, A.; Darido, C. Long-Lived Epidermal Cancer-Initiating Cells. Int. J. Mol. Sci. 2017, 18, 1369. [Google Scholar] [CrossRef]
- Van Laer, L.; Van Eyken, E.; Fransen, E.; Huyghe, J.R.; Topsakal, V.; Hendrickx, J.-J.; Hannula, S.; Mäki-Torkko, E.; Jensen, M.; Demeester, K.; et al. The grainyhead like 2 gene (GRHL2), alias TFCP2L3, is associated with age-related hearing impairment. Hum. Mol. Genet. 2007, 17, 159–169. [Google Scholar] [CrossRef]
- Eshete, M.; Liu, H.; Li, M.; Adeyemo, W.; Gowans, L.J.J.; Mossey, P.; Busch, T.; Deressa, W.; Donkor, P.; Olaitan, P.; et al. Loss-of-Function GRHL3 Variants Detected in African Patients with Isolated Cleft Palate. J. Dent. Res. 2017, 97, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Leslie, E.J.; Liu, H.; Carlson, J.; Shaffer, J.R.; Feingold, E.; Wehby, G.; Laurie, C.A.; Jain, D.; Laurie, C.C.; Doheny, K.F.; et al. A Genome-wide Association Study of Nonsyndromic Cleft Palate Identifies an Etiologic Missense Variant in GRHL3. Am. J. Hum. Genet. 2016, 98, 744–754. [Google Scholar] [CrossRef] [PubMed]
- Carpinelli, M.R.; de Vries, M.E.; Auden, A.; Butt, T.; Deng, Z.; Partridge, D.D.; Miles, L.B.; Georgy, S.R.; Haigh, J.J.; Darido, C.; et al. Inactivation of Zeb1 in GRHL2-deficient mouse embryos rescues mid-gestation viability and secondary palate closure. Dis. Model. Mech. 2020, 13, dmm042218. [Google Scholar] [CrossRef] [PubMed]
- Goldie, S.J.; Arhatari, B.D.; Anderson, P.; Auden, A.; Partridge, D.D.; Jane, S.M.; Dworkin, S. Mice lacking the conserved transcription factor Grainyhead-like 3 (Grhl3) display increased apposition of the frontal and parietal bones during embryonic development. BMC Dev. Biol. 2016, 16, 37. [Google Scholar] [CrossRef] [PubMed]
- Kurihara, Y.; Kurihara, H.; Suzuki, H.; Kodama, T.; Maemura, K.; Nagai, R.; Oda, H.; Kuwaki, T.; Cao, W.H.; Kamada, N.; et al. Elevated blood pressure and craniofacial abnormalities in mice deficient in endothelin-1. Nature 1994, 368, 703–710. [Google Scholar] [CrossRef] [PubMed]
- Adhikari, N.; Neupane, S.; Gwon, G.J.; Kim, J.Y.; An, C.H.; Lee, S.; Sohn, W.J.; Lee, Y.; Kim, J.Y. Grhl3 modulates epithelial structure formation of the circumvallate papilla during mouse development. Histochem. Cell Biol. 2017, 147, 5–16. [Google Scholar] [CrossRef]
- Bai, Y.; Zhao, Z.; Boath, J.; van Denderen, B.J.; Darido, C. The functional GRHL3-filaggrin axis maintains a tumor differentiation potential and influences drug sensitivity. Mol. Ther. 2021, 29, 2571–2582. [Google Scholar] [CrossRef]
- Chen, W.; Shimane, T.; Kawano, S.; AlShaikh, A.; Kim, S.; Chung, S.; Kim, R.; Shin, K.; Walentin, K.; Park, N.; et al. Human Papillomavirus 16 E6 Induces FoxM1B in Oral Keratinocytes through GRHL2. J. Dent. Res. 2018, 97, 795–802. [Google Scholar] [CrossRef]
- Boivin, F.J.; Schmidt-Ott, K.M. Functional roles of Grainyhead-like transcription factors in renal development and disease. Pediatr. Nephrol. 2018, 35, 181–190. [Google Scholar] [CrossRef]
- Walkowska, A.; Pawlak, M.; Jane, S.M.; Kompanowska-Jezierska, E.; Wilanowski, T. Effects of High and Low Sodium Diet on Blood Pressure and Heart Rate in Mice Lacking the Functional Grainyhead-Like 1 Gene. Physiol. Res. 2017, 66, 163–165. [Google Scholar] [CrossRef]
- Butz, H.; Szabó, P.M.; Nofech-Mozes, R.; Rotondo, F.; Kovacs, K.; Mirham, L.; Girgis, H.; Boles, D.; Patocs, A.; Yousef, G.M. Integrative Bioinformatics Analysis Reveals New Prognostic Biomarkers of Clear Cell Renal Cell Carcinoma. Clin. Chem. 2014, 60, 1314–1326. [Google Scholar] [CrossRef] [PubMed]
- Pawlak, M.; Kikulska, A.; Wrzesinski, T.; Rausch, T.; Kwias, Z.; Wilczynski, B.; Benes, V.; Wesoly, J.; Wilanowski, T. Potential protective role of Grainyhead-like genes in the development of clear cell renal cell carcinoma. Mol. Carcinog. 2017, 56, 2414–2423. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, J.J.; Purdue, M.P.; Signoretti, S.; Swanton, C.; Albiges, L.; Schmidinger, M.; Heng, D.Y.; Larkin, J.; Ficarra, V. Renal cell carcinoma. Nat. Rev. Dis. Primers 2017, 3, 17009. [Google Scholar] [CrossRef] [PubMed]
- Riethdorf, S.; Frey, S.; Santjer, S.; Stoupiec, M.; Otto, B.; Riethdorf, L.; Koop, C.; Wilczak, W.; Simon, R.; Sauter, G.; et al. Diverse expression patterns of the EMT suppressor grainyhead-like 2 (GRHL2) in normal and tumour tissues. Int. J. Cancer 2015, 138, 949–963. [Google Scholar] [CrossRef] [PubMed]
- Matsuoka, S.; Suzuki, H.; Kato, C.; Kamikawa-Tokai, M.; Kamikawa, A.; Okamatu-Ogura, Y.; Kimura, K. Expression of Grainyhead-like 2 in the Process of Ductal Development of Mouse Mammary Gland. J. Histochem. Cytochem. 2021, 69, 373–388. [Google Scholar] [CrossRef]
- Cieply, B.; Farris, J.; Denvir, J.; Ford, H.L.; Frisch, S.M. Epithelial–Mesenchymal Transition and Tumor Suppression Are Controlled by a Reciprocal Feedback Loop between ZEB1 and Grainyhead-like-2. Cancer Res. 2013, 73, 6299–6309. [Google Scholar] [CrossRef]
- Holding, A.N.; Giorgi, F.M.; Donnelly, A.; Cullen, A.E.; Nagarajan, S.; Selth, L.A.; Markowetz, F. VULCAN integrates ChIP-seq with patient-derived co-expression networks to identify GRHL2 as a key co-regulator of ERa at enhancers in breast cancer. Genome Biol. 2019, 20, 91. [Google Scholar] [CrossRef]
- Cocce, K.J.; Jasper, J.S.; Desautels, T.K.; Everett, L.; Wardell, S.; Westerling, T.; Baldi, R.; Wright, T.M.; Tavares, K.; Yllanes, A.; et al. The Lineage Determining Factor GRHL2 Collaborates with FOXA1 to Establish a Targetable Pathway in Endocrine Therapy-Resistant Breast Cancer. Cell Rep. 2019, 29, 889–903.e10. [Google Scholar] [CrossRef]
- Nagarajan, S.; Bedi, U.; Budida, A.; Hamdan, F.; Mishra, V.K.; Najafova, Z.; Xie, W.; Alawi, M.; Indenbirken, D.; Knapp, S.; et al. BRD4 promotes p63 and GRHL3 expression downstream of FOXO in mammary epithelial cells. Nucleic Acids Res. 2016, 45, 3130–3145. [Google Scholar] [CrossRef]
- Metzger, D.E.; Stahlman, M.T.; Shannon, J.M. Misexpression of ELF5 disrupts lung branching and inhibits epithelial differentiation. Dev. Biol. 2008, 320, 149–160. [Google Scholar] [CrossRef][Green Version]
- Darido, C.; Georgy, S.R.; Jane, S.M. The role of barrier genes in epidermal malignancy. Oncogene 2016, 35, 5705–5712. [Google Scholar] [CrossRef] [PubMed]
- Raynaud, P.; Carpentier, R.; Antoniou, A.; Lemaigre, F.P. Biliary differentiation and bile duct morphogenesis in development and disease. Int. J. Biochem. Cell Biol. 2011, 43, 245–256. [Google Scholar] [CrossRef] [PubMed]
- Tanimizu, N.; Miyajima, A. Notch signaling controls hepatoblast differentiation by altering the expression of liver-enriched transcription factors. J. Cell Sci. 2004, 117, 3165–3174. [Google Scholar] [CrossRef]
- Satishchandran, A.; Ambade, A.; Rao, S.; Hsueh, Y.-C.; Iracheta-Vellve, A.; Tornai, D.; Lowe, P.; Gyongyosi, B.; Li, J.; Catalano, D.; et al. MicroRNA 122, Regulated by GRLH2, Protects Livers of Mice and Patients From Ethanol-Induced Liver Disease. Gastroenterology 2018, 154, 238–252.e7. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.; the HBVstudy consortium; Liu, H.; Xu, H.; Lu, H.; Yu, Y.; Xu, X.; Yu, M.; Zhang, T.; Tian, X.; et al. Genome-wide association study identifies new loci associated with risk of HBV infection and disease progression. BMC Med. Genom. 2021, 14, 1–12. [Google Scholar] [CrossRef]
- Bell, S.M.; Zhang, L.; Mendell, A.; Xu, Y.; Haitchi, H.M.; Lessard, J.L.; Whitsett, J.A. Kruppel-like factor 5 is required for formation and differentiation of the bladder urothelium. Dev. Biol. 2011, 358, 79–90. [Google Scholar] [CrossRef]
- Xiang, J.; Fu, X.; Ran, W.; Chen, X.; Hang, Z.; Mao, H.; Wang, Z. Expression and role of grainyhead-like 2 in gastric cancer. Med. Oncol. 2013, 30, 714. [Google Scholar] [CrossRef]
- Yuan, M.; Wang, J.; Fang, F. Grainyhead-Like Genes Family May Act as Novel Biomarkers in Colon Cancer. OncoTargets Ther. 2020, 13, 3237–3245. [Google Scholar] [CrossRef]
- Shen, J.; Lv, X.; Zhang, L. GRHL2 Acts as an Anti-Oncogene in Bladder Cancer by Regulating ZEB1 in Epithelial-Mesenchymal Transition (EMT) Process. OncoTargets Ther. 2020, 13, 2511–2522. [Google Scholar] [CrossRef]
- Wezel, F.; Lustig, J.; Azoitei, A.; Liu, J.; Meessen, S.; Najjar, G.; Zehe, V.; Faustmann, P.; Zengerling, F.; John, A.; et al. Grainyhead-Like 3 Influences Migration and Invasion of Urothelial Carcinoma Cells. Int. J. Mol. Sci. 2021, 22, 2959. [Google Scholar] [CrossRef]
- Salazar, V.S.; Ohte, S.; Capelo, L.P.; Gamer, L.; Rosen, V. Specification of osteoblast cell fate by canonical Wnt signaling requires Bmp2. Development 2016, 143, 4352–4367. [Google Scholar] [CrossRef] [PubMed]
- Marx, V. Method of the Year: Spatially resolved transcriptomics. Nat. Methods 2021, 18, 9–14. [Google Scholar] [CrossRef] [PubMed]



| GENE(S) | SPECIES | MAMMALIAN | BIOLOGICAL ROLE | REFERENCE(S) |
|---|---|---|---|---|
| ORTHOLOGUE | ||||
| Ultrabithorax (Ubx) | Drosophila | HoxB6 | Grh can bind to and/or directly activate transcription from the promoters of these genes during the development of neuronal cells. | [44,45] |
| Dopa Decarboxylase (Ddc) | DDC | |||
| Fushi-tarazu(Ftz) | Drosophila | NR5A2 | Grh regulates Ftz during epidermal wound repair. | [44,46] |
| Suppressor of Hairless (Su (H)) | Drosophila | CBF1 | Modulation of Notch signalling in regulation of cell fate. | [47] |
| Torso (tor) | Drosophila | TEK receptor tyrosine kinase | The torsos receptor tyrosine kinase (RTK) can remove Grh-mediated repression of the tailless gene during development. | [48] |
| Tailless (tll) | NR2E1 | |||
| Zerknullt (zen) | Drosophila | Hox A3/B3/D3 | Grh binds to a zen repression element in a ventral repression region | [49] |
| Decapentaplegic (dpp) | Drosophila | BMP2/BMP4 | Grh binds to a dpp repression element in a ventral repression region | [49] |
| Proliferating cell nuclear antigen (Pcna) | Drosophila | PCNA | Grh binds to PCNA to regulate cellular differentiation | [50] |
| Castor (Cas) | Drosophila | CASZ1 | Grh may be the final step of a signalling cascade involving Cas that regulates the differentiation of embryonic neuroblasts | [51] |
| Suppressor of Hairless (Su(H)) | Drosophila | RBPJ | Su (H) and Grh may regulate both kni and knrl in regulating dorso-ventral patterning of the primitive gut tube. | [52] |
| Knirps (kni) | RAR related orphan receptor A | |||
| Knirps-related (knrl) | NR1H2 | |||
| Krotzkopf verkehrt (kkv) | Drosophila | HAS1/2/3 | These genes, when inactivated in Drosophila, lead to a “blimp” phenotype following removal of the vitelline membrane of the eggshell. | [53] |
| Knickkopf (knk), | THBD | |||
| Retroactive (rtv), | No known orthologues | Phenotypically similar to Grh mutants. | ||
| Zeppelin (zep) [renamed Gfat1] | GFPT1/2 | |||
| Fasciclin II (FasII) | Drosophila | NCAM1/2 | Compound mutants of Grh, FasII and/or ATPa show additive defects in epithelial integrity/cohesion. | [54] |
| ATPase a sub-unit (ATPa) | ATPA family | |||
| Branchless (Bnl; upstream) | Drosophila | FGF family | An upstream signalling factor that upregulates Grhl expression in Drosophila airway development | [55] |
| Ribbon (rib) | Drosophila | BTBD18/ZBTB9/ZBTB12 | Hypothesised that interaction between Grh and Rib may regulate tracheal branching and apical membrane growth; possibly via Bnl signalling. | [54] |
| Mab-5 (Ubx orthologue) | C. elegans | HoxB6 | Promoter binding analyses show that Grh can bind to gene promoters, as in Drosophila, in post-embryonic development | [4] |
| dbl-1 (dpp orthologue) | BMP Family | |||
| Aromatic L-aa decarboxylase (Ddc orthologue) | DDC | |||
| Starry Night/Flamingo (stan/flam) | Drosophila | Celsr1-3 | Grh regulates stan/flam levels in Drosophila wings during development. | [55] |
| Frizzled (Fz) | Frizzled | Fz is an upstream factor of Grh, and Grh is crucial for Fz function in apical accumulation of Fz. Interacts genetically with Grh during wing development. | ||
| Optomotor-blind (Omb) | Tbx2 | Predicted Grh target (in silico). Mutant shows defects in cuticle pattern formation, cell shape, pigmentation and hair-organisation. Slight interaction with Grh in wing development. | ||
| Ovo/Shaven Baby(ovo/svb) | Ovol1 | Predicted Grh target (in silico). Mutant shows imaginal disc morphogenesis defects; no interaction with Grh observed. | ||
| Scabrous (sca) | partial homology to fibrinogen | Predicted Grh target (in silico). Mutant shows imaginal disc morphogenesis defects; no interaction with Grh observed. | ||
| Pipsqueak (psq) | BACH2/BTBD18/DNAJC14 | Predicted Grh target (in silico). Mutant shows defects in early egg and embryo patterning; no interaction with Grh observed. | ||
| Ventral veins | POU3F2 | Predicted Grh target (in silico). Binds to a DNA sequence element required for expression of the Ddc gene. Could play a role in early ectodermal cells; no interaction with Grh observed. | ||
| lacking/Drifter (vvl) | ||||
| Abdominal A (Abd A) | Drosophila | HoxA6/B6/C6 | Grh is required to maintain AbdA expression in neuroblasts in late larval phase, although it is not required for initial AbdA initiation in these cells. | [56] |
| Pale (ple) | Drosophila | Tyrosine Hydroxylase | Ple contains 3 Grh-binding sites (as well as others) in 2 enhancer regions within the promoter. Potential role of Grh-Ple in wound repair &/or cross-linking of epidermal genes. | [46] |
| E-Cadherin (shotgun) | Drosophila | E-Cadherin | E-Cadherin is greatly reduced in Grh-deficient post-embryonic neuroblasts, ectopic Grh expression increases E-Cadherin expression, and the E-Cadherin promoter contains binding sites for Grh. | [57] |
| Pleiohomeotic (Pho) | Drosophila | ZFP42 | Grh may be responsible for targeting this Polycomb group (PcG) gene to its specific binding sites in target promoters. Polycomb genes are involved in silencing genes that control development | [58] |
| Thelytoky (th) | A. mellifera (Honeybee) | Grhl family | The th gene may be responsible for regulating reproduction and sterility | [59] |
| FasciclinIII (FasIII) | Drosophila | Nectin3 | Flies over-expressing Grh show strong up-regulation of FasIII, coracle and sinuous and these pathways contribute to the formation of apical barriers in the epidermis. | [60] |
| Coracle (cor) | EPB41L3 | |||
| Sinuous (sinu) | Claudin Family | |||
| Neurexin (nxr) | Neurexin Family | Potential role for interaction of these genes with Grh in the formation of septate junctions in flies (analogous to adherens junctions in mammals). | ||
| Discs-large (Dlg) | Dlg Family | |||
| Tailless (tll) | Drosophila | NR2E1 | Ras-signalling, possibly involving removal of Grh repressors, can transactivate the expression of these two genes. Grh can also bind tll regulatory regions. | [61] |
| Hueckebein (hkb) | KLF1/14/16 | |||
| Intermediate Neuroblasts Defective (Ind) | Drosophila | GSX1/GSX2 | Grh binds to ind to putatively regulate dorso-ventral patterning | [62] |
| Flotillin-2 (flot-2) | Drosophila | Flot-2 | Grh binding sites are present on flot-2 and may contribute to epidermal wound repair. | [63] |
| GENE(S) | SPECIES | BIOLOGICAL ROLE | REFERENCE(S) |
|---|---|---|---|
| Cytokeratin 1 (XK81A1) | Xenopus | xGrhl1 down-regulation results in downregulation of XK8IAI with concomitant inhibition of terminal epidermal differentiation. | [64] |
| Transcription factor AP2 (AP-2) | AP-2 co-operates with xGrhl1 to transactivate the xKI81A reporter. | ||
| Distal-less Homeobox 3 (Dlx3) | Dlx3 and Dlx5 may be induced by xGrhl1 over-expression. | ||
| Distal-less Homeobox 5 (Dlx5) | |||
| Claudin B (cldnb) | Zebrafish | Grhl2b binds to claudin B and epcam in the otic vesicles in zebrafish. Loss of grhl2b leads to loss of both genes, and manifests as loss of balance and hearing behaviour in zebrafish. | [6] |
| epithelial cell adhesion molecule (epCAM) | |||
| Engrailed 2a (eng2a) | Zebrafish | Grhl2b mediated regulation of eng2a drives patterning and survival of neuroblasts at the midbrain-hindbrain boundary. | [13] |
| Small protein effector of cdc42 1 (spec1) | Bound by grhl2b to drive the morphogenesis of the midbrain-hindbrain boundary. | ||
| Endothelin 1 (edn1) | Zebrafish | Grhl3 regulation of Edn1 is critical for maintaining neural crest cell (NCC) fidelity in the branchial arches, and consequently, for lower jaw development. | [7] |
| IRF6 | Zebrafish/Xenopus (upstream regulator) | Irf6 binds to the grhl3 promoter and drives periderm differentiation. Grhl3 rescues Irf6 deficiency in both zebrafish and Xenopus | [65] |
| Rho Guanine Nucleotide Exchange Factor 19 (arhgef19) | Zebrafish | grhl3 regulates arhgef19 in the morphogenesis of the midbrain-hindbrain boundary. | [66] |
| GENE(S) | SPECIES | BIOLOGICAL ROLE | REFERENCE(S) |
|---|---|---|---|
| LIM Domain Only 4 (Lmo4) | Mouse | Lmo4 is co-expressed in several of the same regions as Grhl3 during mouse development, esp. the ectoderm, oral and dental epithelium. Lmo4 also dimerizes with Grhl3. | [67] |
| Protein Kinase C isoforms (PKCa, bI, bII, d, e, g) | Mouse (Upstream regulators) | Involved in mediating the inositol-responsive rescue of neural-tube defects in curly-tail mutants. Action is potentially via stimulating cell proliferation (particularly in hindgut). | [68] |
| Transglutaminase 1 (Tgm1) | Mouse | Involved in epithelialisation through cross-linking barrier components. | [20] |
| Tumor protein 53 (Tp53) | PC12 cells (Rat) | Activation of p53 by NGF during neurite differentiation results in up-regulated Grhl3 expression, and p53 binds to the Grhl3 promoter. | [69] |
| Desmoglein1 (Dsg1) | Mouse | Dsg1 is a direct target of Grhl1, but not Grhl3. Involved in mediating hair anchorage in epidermis through regulation of desmosomes. | [70] |
| Transforming growth factor alpha (TGF-a) | Mouse | Grhl3 acts upstream of TGF-a and is required for the formation of the eyelids in mouse. No evidence of direct binding. | [71] |
| Uroplakin II (Upk2) | Mouse | Regulates differentiation of umbrella cells and urinary bladder barrier formation. | [72] |
| E-cadherin (Cdh1) | Mouse | Grhl2 is responsible for directly regulating the expression of these genes in various epithelial tissues. Downregulated in Grhl2−/− collecting ducts. | [15,73,74] |
| Claudin 4 (Cldn4) | |||
| Rho Guanine Nucleotide Exchange Factor 19 (Arhgef19) | Mouse | Arhgef19 is bound by Grhl3 and acts as part of the Planar Cell Polarity (PCP) pathway in keratinocytes. | [75] |
| Phosphatase and tensin homolog (Pten) | Mouse | Pten acts downstream of Grhl3 to suppress squamous cell carcinomas. | [26] |
| Lamin B1 | Mouse | Lamin expression modifies the spina bifida presentation in the grhl3-hypomorphic model curly tail. No direct binding demonstrated. | [76] |
| Homeobox Protein Nkx2-1 | Mouse | Grhl2 and Nkx2-1 form a reciprocal regulatory loop in mouse lung epithelium. | [77] |
| Forkhead box M1b (FoxM1b) | Mouse | Grhl2 induces FoxM1B downstream of HPV-16 in the context of oral cancer in human oral keratinocytes. | [78] |
| micro-RNA-21 (miR-21) | Mouse | Decreased levels of Grhl3 contributed to squamous cell carcinoma via an upregulation of miR-21, contributing to a feedback loop involving Msh2. | [29] |
| Erb-B2 Receptor Tyrosine Kinase 3 (erbb3) | Mouse | Grhl2-mediated direct regulation of Erbb3 regulates proliferation, EMT and morphology in breast cancer cell lines. | [32] |
| Member RAS Oncogene Family 25 (Rab25) | Mouse | Grhl2-mediated regulation of Rab25 promotes localisation of Cldn4 at tight junction in cholangiocytes. Regulates lumen expansion and barrier formation in kidney epithelia. Downregulated in Grhl2−/− collecting ducts. | [73,74,79] |
| Histone deacetylase 3 (HDAC3) | Mouse | HDAC3 and MYCN directly inhibit Grhl1, therein promoting tumourigenesis in neuroblastoma cell lines. | [39] |
| Myc Family Member (MYCN) | |||
| Calcium channel, voltage-dependent, L type, alpha 1D subunit (Cacna1d) | Mouse | Cacnd1 and Xpo1 are predicted targets based on the presence of a conserved Grhl-binding site in their promoters; Ddc and Flot2 are orthologues of known Drosophila grh targets. Expression of all was reduced in the kidney of Grhl1−/− mice. Direct binding not shown. | [80] |
| Ddc (Dopa decarboxylase) | |||
| Flotillin 2 (Flot2) | |||
| Exportin 1 (Xpo1) | |||
| Ovo-like 2 transcription factor 2 (Ovol2) | Mouse | Grhl2 positively regulates Ovol2, and Ovol2 can rescue lumen defects in Grhl2−/− mice, via regulation of Cldn4 and Rab25. Downregulated in Grhl2−/− collecting ducts. | [73,74] |
| Potassium Two Pore Domain Channel Subfamily K Member 1 (Kcnk1) | Mouse | Grhl2 target genes involved in placental development in mice, as verified by direct promoter binding (ChIP). | [81,82] |
| Ladinin 1 (Lad1) | |||
| Leucine Zipper Down-Regulated In Cancer 1 (Ldoc1) | |||
| Placenta-enriched 1 (Plac1) | |||
| Prominin 2 (Prom2) | |||
| Member RAS Oncogene Family 15 (Rab15) | |||
| Sphingomyelin Phosphodiesterase Acid Like 3B (Smpdl3b) | |||
| Serine Peptidase Inhibitor, Kunitz Type 1 (Spint1) | Additionally, Grhl2 driving Spint1 may play a role in branching morphogenesis and development of the salivary gland. | ||
| Testis Expressed 19 (Tex19.1) | |||
| Transcription Factor AP-2 Alpha (Tfap2a) | |||
| Transmembrane Protein 54 (Tmem54) | |||
| Tyrosine Kinase Non Receptor 1 (Tnk1) | |||
| 1600014K23Rik (possibly ferritin, heavy polypeptide-like 17, member A; Fthl17a) | |||
| Glycogen synthase kinase 3 (Gsk3b) | Mouse (conditional deletion model) | Direct binding validated by ChIP and EMSA in oral epithelial cells. Grhl3-GSK3b pathway regulates proliferation in both normal and oral-epithelial cancer cells. | [27] |
| p300 | Dog (MDCK cells) | Grhl2-mediated suppression of p300 led to reduced induction of matrix metalloproteases and suppression of tubulogenesis. No direct binding shown. | [36] |
| Zinc Finger E-Box Binding Homeobox 1 (ZEB1) | Mouse | Grhl2 and Zeb1 form a double negative regulatory feedback loop in breast cancer cell lines. | [32,38] |
| Myosin Heavy Chain 10 (Myh10) | Mouse | These genes were down-regulated in kidney collecting ducts lacking Grhl2, and were confirmed as direct binding targets via ChIP. | [74] |
| Shroom Family Member 3 (Shroom3) | |||
| Tight Junction Protein 3 (Tjp3) | |||
| E74 Like ETS Transcription Factor 5 (Elf5) | Mouse | Grhl2 binds to the Elf5 promoter in whole embryonic lung tissue, and this pathway may regulate proliferation in distal progenitor cells. | [19] |
| Fascin Actin-Bundling Protein 1 (Fscn1) | Mouse | Grhl3 regulates Fscn1 in keratinocytes during wound healing. | [83] |
| GENE(S) | SPECIES | BIOLOGICAL ROLE | REFERENCE(S) |
|---|---|---|---|
| Engrailed 1 (En1) | Human | Grhl1 binds to and transactivates promoter of human En1 in vitro. | [2] |
| Tumour necrosis factor a (TNF-a) | MCF-7 cells (Human; upstream regulator) | TNF-a induces Grhl3 expression in MCF tumour cells. Hypothesised to contribute to endothelial cell migration and angiogenesis | [84] |
| Human telomerase reverse transcriptase (hTERT) | SCC4 cells – Human | Grhl2 knockdown has inhibitory effect on telomerase activity, thus potentially implicating it in tumourogenesis. | [85] |
| Protein Kinase Akt; endothelial Nitric Oxide Synthase (eNOS) | Human | akt and eNOS are induced by Grhl3 in endothelial cells; no direct binding demonstrated. | [86] |
| Zinc Finger Enhancer-Binding Protein 1 (Zeb1) | Human | Grhl2 directly repressed transcription of Zeb1 in a breast cancer cell line, resulting in reduced EMT, enhanced anoikis sensitivity and upregulation of Smad, mir200b/c and BMP2. | [10] |
| Claudin 3 (Cldn3) | Human | Regulation of tight junctions in the epithelial barrier relies on Grhl2 expression and downstream activation of claudin3, 4. No direct binding demonstrated. | [87] |
| Cyclin B2 (CCNB2) | SCC4 cells–Human | Grhl2 binds to the proximal promoter regions of these target genes in Squamous Cell Carcinoma (SCC) cell lines, and influences histone modification at the target gene promoters. | [78] |
| Proliferating Cell Nuclear Antigen (PCNA) | |||
| Ki-67 | |||
| Small Proline Rich Protein 2a (Sprr2a) | |||
| B-cell lymphoma 2 (BCL2) | Human | Overexpression of Grhl2 decreased c-Myc and Bcl-2 expression in SGC7901 gastric cancer cells. No direct binding demonstrated. | [88] |
| Myc Family Member (c-Myc) | |||
| Multiple putative target genes (n = 296) | Human | Multiple targets predicted by cross-referencing ChIP-SEQ data and RNA-SEQ data from human bronchial epithelial (HBE) cells expressing dominant-negative Grhl2 | [11] |
| Interferon Regulatory Factor 6 (IRF6; upstream) | Human | IRF6 binds to an enhancer element near the Grhl3 promoter, and may regulate the development of the periderm in both zebrafish and human keratinocytes. Mutations in Grhl3 and IRF6 are also both causative of the human palatal clefting condition Van der Woude Syndrome. | [12,65] |
| Micro-RNA-122 (miR-122) | Human | Grhl2 negatively regulates miR-122 in hepatocyte differentiation. | [89,90] |
| Small Cell Adhesion Glycoprotein (SMAGP) | Grhl2-mediated regulation of SMAGP and ZNF50 genes is predicted to regulate airway cell polarity, maintain barriers and cell differentiation. | ||
| Zinc Finger Protein 750 (ZNF750) | The Grhl2-ZNF50 pathway may regulate ciliogenesis. | ||
| Glycogen synthase kinase 3 (Gsk3b) | Oral epithelial and cancer cell lines, Human | Direct binding validated by ChIP and EMSA in oral epithelial cells. Grhl3-GSK3b pathway regulates proliferation in both normal and oral-epithelial cancer cells. | [27] |
| Tumor protein 63 (p63) | Human | Grhl2 and p63 form a regulatory feedback loop which governs epithelial traits in human keratinocytes. | [91] |
| Micro-RNA-217 (miR-217; upstream) | Human | miR-217 inhibits keratinocyte proliferation whilst promoting differentiation. | [92] |
| Micro-RNA-200 (miR-200) | Human | Grhl2 binds in the promoter region of miR-200; Grhl2 may regulate the promoter of a polycistronic primary transcript | [38] |
| P300 | Human | A small sequence of Grhl2 between aa 425-437 is responsible for inhibiting the C-terminal Domain of P300 and was postulated to contribute to the suppression of EMT and tubulogenesis. | [36] |
| Tetraspannin CD9 (CD9) | Human | CD9 is a direct target of Grhl1, and high CD9 expression was associated with favourable outcomes in neuroblastomas. | [39] |
| Octamer-binding transcription factor 4, (OCT-4) | Human | Grhl2 binds to the proximal region of the Oct-4 promotor, putatively regulating the transcription of Oct-4 in cell reprogramming and contributing to oral cell carcinoma. | [93] |
| Matrix Metalloproteinase 2, 7 and 9 (MMP 2/7/9) | Human | GRHL2 expression correlated with expression levels of these three MMPs in gastric cancer cells; no direct binding demonstrated. | [94] |
| Micro-RNA-194 (miR-194; upstream) | Human | Luciferase assays showed that miR-194 binds to the 3’UTR region of Grhl2. | [95] |
| Ras Homolog Family Member G (RhoG) | Human | RhoG possesses 3 GRHL2 binding sites, and Grhl2 inhibits RhoG expression | [96] |
| Micro-RNA-133a (miR-133a; upstream) | Human | miR-133a was demonstrated by Luciferase assay as binding to Grhl2 and subsequently regulating ESRP expression and EMT in the airway epithelium | [97] |
| Lysine Methyltransferase 2C (KMT2C) | Human | Grhl2 recruits KMTC and KMTD to interact with Grhl2-induced enhancers | [98] |
| Lysine Methyltransferase 2D (KMT2D) | |||
| Nitric Oxide Synthase 3 (NOS3) | Human | Grhl3 and eNOS co localise as observed by proximity ligation assays, however no direct demonstration of Grhl3 regulation of eNOS. | [99] |
| Extracellular signal-regulated kinase (ERK; upstream) | Human | Erk phosphorylates Grhl1, leading to translocation of Grhl1 to the nucleus and contributing to cell cycle proliferation in cancer cells. | [100] |
| S100 Calcium Binding Protein A10 (S100A10) | Human | Grhl2 binds to a site in the first intron of S100A10 and drives transcription in HEK-293 cells, and this pathway may drive extracellular matrix degradation in cancer. | [101] |
| Cell Division Cycle 27 (CDC27) | Human | GRHL1 binds to the promoters of these genes and directly regulates their expression in non-small cell lung cancer (NSCLC) cells. | [100] |
| RAD21 cohesive complex (RAD21) | |||
| Cell division cycle 7 (CDC7) | |||
| Anaphase promoting complex subunit 13 (ANAPC13) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gasperoni, J.G.; Fuller, J.N.; Darido, C.; Wilanowski, T.; Dworkin, S. Grainyhead-like (Grhl) Target Genes in Development and Cancer. Int. J. Mol. Sci. 2022, 23, 2735. https://doi.org/10.3390/ijms23052735
Gasperoni JG, Fuller JN, Darido C, Wilanowski T, Dworkin S. Grainyhead-like (Grhl) Target Genes in Development and Cancer. International Journal of Molecular Sciences. 2022; 23(5):2735. https://doi.org/10.3390/ijms23052735
Chicago/Turabian StyleGasperoni, Jemma G., Jarrad N. Fuller, Charbel Darido, Tomasz Wilanowski, and Sebastian Dworkin. 2022. "Grainyhead-like (Grhl) Target Genes in Development and Cancer" International Journal of Molecular Sciences 23, no. 5: 2735. https://doi.org/10.3390/ijms23052735
APA StyleGasperoni, J. G., Fuller, J. N., Darido, C., Wilanowski, T., & Dworkin, S. (2022). Grainyhead-like (Grhl) Target Genes in Development and Cancer. International Journal of Molecular Sciences, 23(5), 2735. https://doi.org/10.3390/ijms23052735







