Emerging Antifungal Targets and Strategies
Abstract
:1. Fungi and Fungal Infections
- (a)
- Superficial (superficial, cutaneous, and mucocutaneous). Infections are most common on the nail (onychomycosis), eye (keratomycosis), and ear (otomycosis).
- (b)
- Deep/invasive—fungi are localized and multiply in deep tissues and organs (lungs, liver, spleen, bones, brain, heart, and blood). These infections have a complex clinical course, poor prognosis, and are difficult to diagnose. They are most often caused by fungi from the genera Aspergillus and Candida [1,3,4].
2. Current Antifungals in Use and Their Limitations
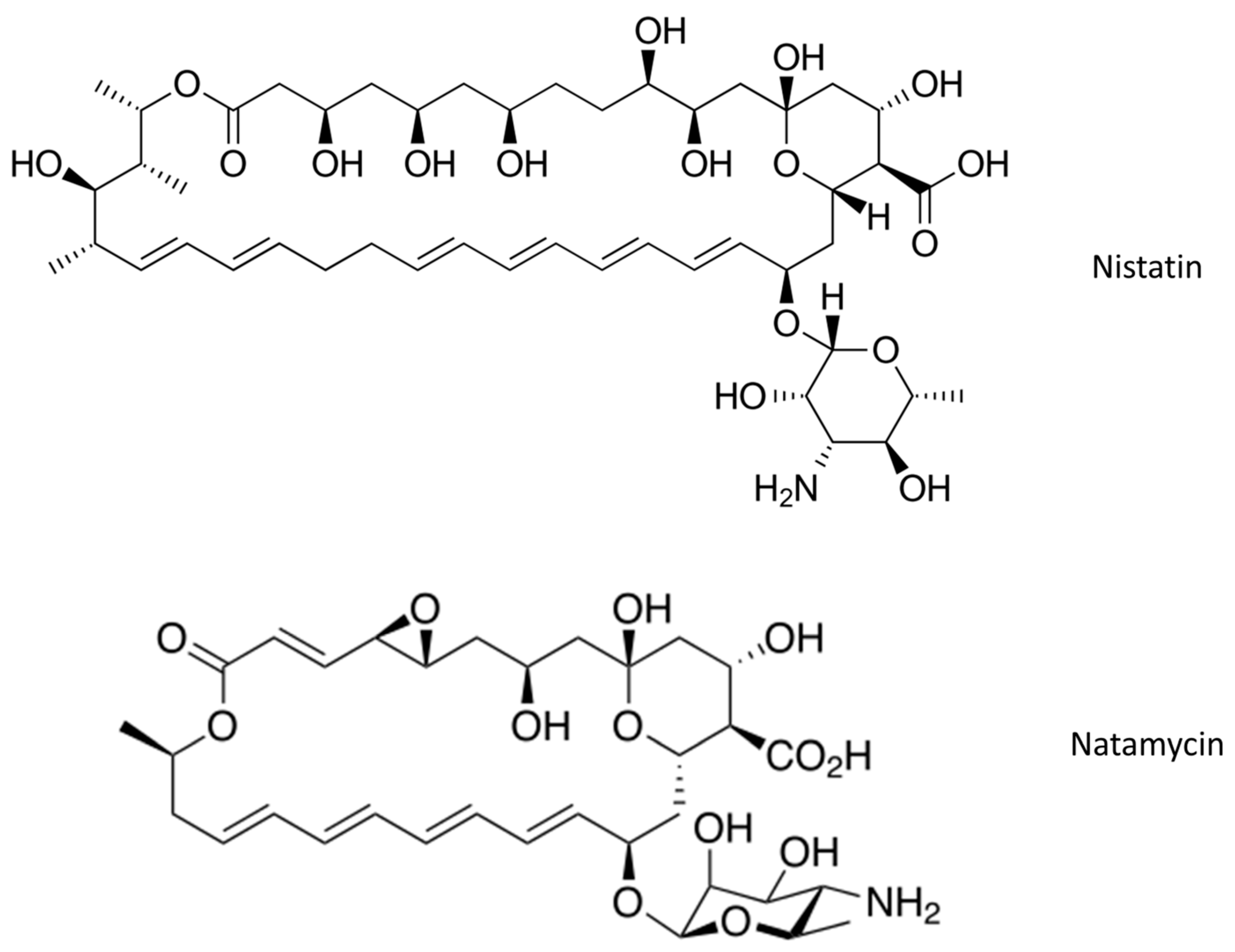
- Polyenes (amphotericin B, natamycin, and nystatin).
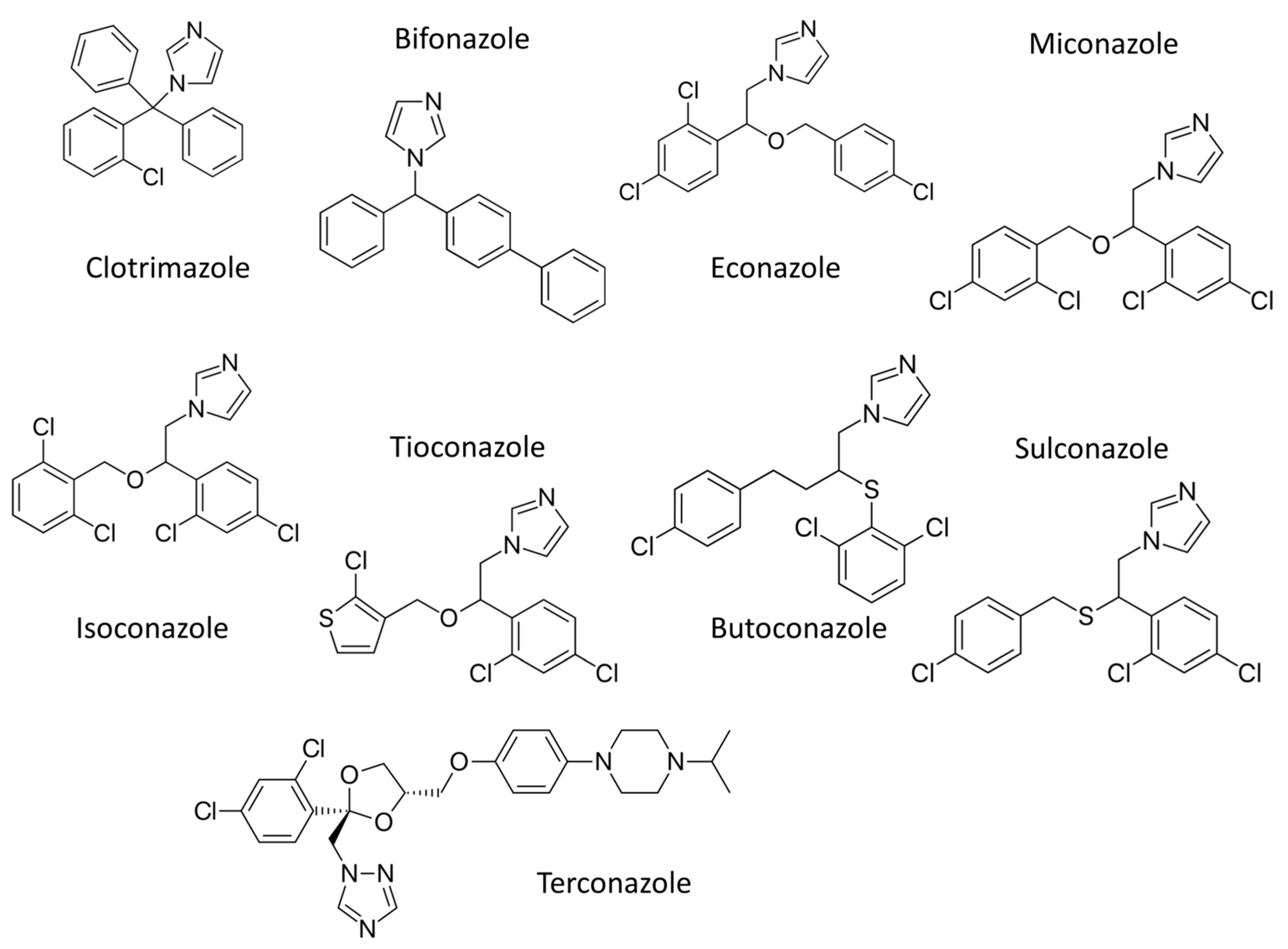
- Azoles—imidazoles (ketoconazole and miconazole) and triazoles (fluconazole, itraconazole, voriconazole, etc.).
- Echinocandines.
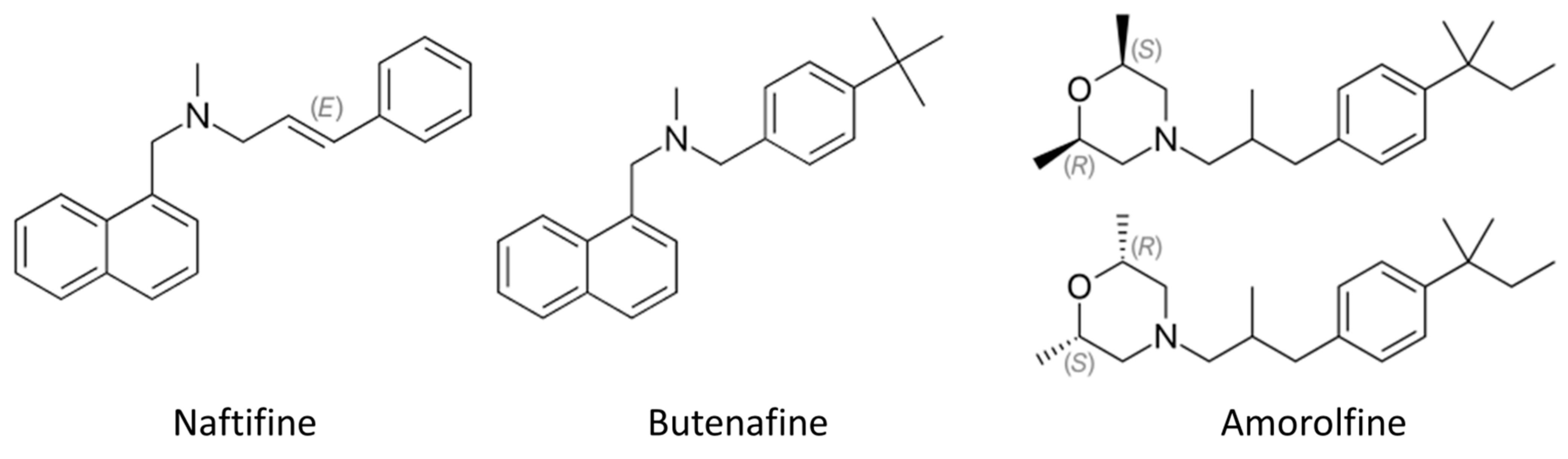
- Amines (terbinafine, etc.).
2.1. The Most Common Antifungal Drugs for Topical Applications
2.2. The Most Common Antifungal Drugs for Systemic Applications
3. Recent Update on Antifungal Targets and Strategies
3.1. Interference with Fungal Antimicrobial Resistance
3.1.1. Cell Efflux as Antifungal Target
3.1.2. Ergosterol Biosynthesis and ERG11 Expression
3.1.3. Heat Shock Protein 90 (Hsp90)
3.2. Tackling Different Virulence Aspects—Targeting Biofilm and Hyphae
3.3. Mitochondria as Antifungal Targets
3.4. Affecting Different Metabolic Pathways and Enzymes in Fungi
3.5. Fungal Cell Wall
3.6. Antifungal Vaccines
4. In Vivo and Clinical and Studies of Products Employing Novel Antifungal Pathways
5. Conclusions and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Mitchell, T.G. Fundamental Medical Mycology. Emerg. Infect. Dis. 2012, 18, 1393. [Google Scholar] [CrossRef] [Green Version]
- Janbon, G.; Quintin, J.; Lanternier, F.; d’Enfert, C. Studying fungal pathogens of humans and fungal infections: Fungal diversity and diversity of approaches. Microbes Infect. 2019, 21, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, T.G. Overview of basic medical mycology. Otolaryngol. Clin. N. Am. 2000, 33, 237–249. [Google Scholar] [CrossRef]
- McGinnis, M.R.; Tyring, S.K. Introduction to Mycology. Methods Gen. Mol. Microbiol. 1996, 925–928. [Google Scholar] [CrossRef]
- Feucht, C.; Patel, D.R. Principles of pharmacology. Pediatr. Clin. N. Am. 2011, 58, 11–19. [Google Scholar] [CrossRef]
- Wall, G.; Lopez-Ribot, J.L. Current Antimycotics, New Prospects, and Future Approaches to Antifungal Therapy. Antibiotics 2020, 9, 445. [Google Scholar] [CrossRef]
- Millikan, L.E. Current concepts in systemic and topical therapy for superficial mycoses. Clin. Dermatol. 2010, 28, 212–216. [Google Scholar] [CrossRef]
- Zhang, A.Y.; Camp, W.L.; Elewski, B.E. Advances in Topical and Systemic Antifungals. Dermatol. Clin. 2007, 25, 165–183. [Google Scholar] [CrossRef]
- Gupta, A.K.; Einarson, T.R.; Summerbell, R.C.; Shear, N.H. An overview of topical antifungal therapy in dermatomycoses. A North American perspective. Drugs 1998, 55, 645–674. [Google Scholar] [CrossRef]
- Lyu, X.; Zhao, C.; Yan, Z.M.; Hua, H. Efficacy of nystatin for the treatment of oral candidiasis: A systematic review and meta-analysis. Drug Des. Devel. Ther. 2016, 10, 1161–1171. [Google Scholar] [CrossRef] [Green Version]
- Te Welscher, Y.M.; Ten Napel, H.H.; Balagué, M.M.; Souza, C.M.; Riezman, H.; De Kruijff, B.; Breukink, E. Natamycin Blocks Fungal Growth by Binding Specifically to Ergosterol without Permeabilizing the Membrane. J. Biol. Chem. 2008, 283, 6393–6401. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Te Welscher, Y.M.; Van Leeuwen, M.R.; De Kruijff, B.; Dijksterhuis, J.; Breukink, E. Polyene antibiotic that inhibits membrane transport proteins. Proc. Natl. Acad. Sci. USA 2012, 109, 11156–11159. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, A.; Taniguchi, J. Review: Emerging strategies for antimicrobial drug delivery to the ocular surface: Implications for infectious keratitis. Ocul. Surf. 2017, 15, 670–679. [Google Scholar] [CrossRef] [PubMed]
- Parker, J.E.; Warrilow, A.G.S.; Price, C.L.; Mullins, J.G.L.; Kelly, D.E.; Kelly, S.L. Resistance to antifungals that target CYP51. J. Chem. Biol. 2014, 7, 143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spampinato, C.; Leonardi, D. Candida infections, causes, targets, and resistance mechanisms: Traditional and alternative antifungal agents. Biomed Res. Int. 2013, 2013, 204237. [Google Scholar] [CrossRef] [Green Version]
- Buil, J.B.; Snelders, E.; Denardi, L.B.; Melchers, W.J.G.; Verweij, P.E. Trends in Azole Resistance in Aspergillus fumigatus, the Netherlands, 1994–2016. Emerg. Infect. Dis. 2019, 25, 176. [Google Scholar] [CrossRef] [Green Version]
- Crowley, P.D.; Gallagher, H.C. Clotrimazole as a pharmaceutical: Past, present and future. J. Appl. Microbiol. 2014, 117, 611–617. [Google Scholar] [CrossRef]
- Fothergill, A.W. Miconazole: A historical perspective. Expert Rev. Anti. Infect. Ther. 2006, 4, 171–175. [Google Scholar] [CrossRef]
- Gupta, A.K.; Lyons, D.C.A. The Rise and Fall of Oral Ketoconazole. J. Cutan. Med. Surg. 2015, 19, 352–357. [Google Scholar] [CrossRef] [PubMed]
- Nowosielski, M.; Hoffmann, M.; Wyrwicz, L.S.; Stepniak, P.; Plewczynski, D.M.; Lazniewski, M.; Ginalski, K.; Rychlewski, L. Detailed mechanism of squalene epoxidase inhibition by terbinafine. J. Chem. Inf. Model. 2011, 51, 455–462. [Google Scholar] [CrossRef]
- Birnbaum, J.E. Pharmacology of the allylamines. J. Am. Acad. Dermatol. 1990, 23, 782–785. [Google Scholar] [CrossRef]
- Campoy, S.; Adrio, J.L. Antifungals. Biochem. Pharmacol. 2017, 133, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.C.; Lamichhane, A.K.; Cai, H.; Walter, P.J.; Bennett, J.E.; Kwon-Chung, K.J. Moderate levels of 5-fluorocytosine cause the emergence of high frequency resistance in cryptococci. Nat. Commun. 2021, 12, 3418. [Google Scholar] [CrossRef] [PubMed]
- Lemke, A.; Kiderlen, A.F.; Kayser, O. Amphotericin B. Appl. Microbiol. Biotechnol. 2005, 68, 151–162. [Google Scholar] [CrossRef] [PubMed]
- Cavassin, F.B.; Baú-Carneiro, J.L.; Vilas-Boas, R.R.; Queiroz-Telles, F. Sixty years of Amphotericin B: An Overview of the Main Antifungal Agent Used to Treat Invasive Fungal Infections. Infect. Dis. Ther. 2021, 10, 115–147. [Google Scholar] [CrossRef] [PubMed]
- Anderson, T.M.; Clay, M.C.; Cioffi, A.G.; Diaz, K.A.; Hisao, G.S.; Tuttle, M.D.; Nieuwkoop, A.J.; Comellas, G.; Maryum, N.; Wang, S.; et al. Amphotericin forms an extramembranous and fungicidal sterol sponge. Nat. Chem. Biol. 2014, 10, 400–406. [Google Scholar] [CrossRef] [PubMed]
- Stone, N.R.H.; Bicanic, T.; Salim, R.; Hope, W. Liposomal Amphotericin B (AmBisome®): A review of the pharmacokinetics, pharmacodynamics, clinical experience and future directions. Drugs 2016, 76, 485. [Google Scholar] [CrossRef] [Green Version]
- Adler-moore, J.P.; Proffitt, R.T. Amphotericin B lipid preparations: What are the differences? Clin. Microbiol. Infect. 2008, 14, 25–36. [Google Scholar] [CrossRef] [Green Version]
- Jura, S.E.; Hillenbrand, K. Fluconazole. Pediatr. Rev. 2006, 27, 158–159. [Google Scholar] [CrossRef]
- Piérard, G.E.; Arrese, J.E.; Piérard-Franchimont, C. Itraconazole. Expert Opin. Pharmacother. 2000, 1, 287–304. [Google Scholar] [CrossRef]
- De Beule, K.; Van Gestel, J. Pharmacology of itraconazole. Drugs 2001, 61 (Suppl. 1), 27–37. [Google Scholar] [CrossRef] [PubMed]
- Wong, T.Y.; Loo, Y.S.; Veettil, S.K.; Wong, P.S.; Divya, G.; Ching, S.M.; Menon, R.K. Efficacy and safety of posaconazole for the prevention of invasive fungal infections in immunocompromised patients: A systematic review with meta-analysis and trial sequential analysis. Sci. Rep. 2020, 10, 14575. [Google Scholar] [CrossRef] [PubMed]
- Leung, S.; Poulakos, M.N.; Machin, J. Posaconazole: An Update of Its Clinical Use. Pharm. J. Pharm. Educ. Pract. 2015, 3, 210. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xing, Y.; Chen, L.; Feng, Y.; Zhou, Y.; Zhai, Y.; Lu, J. Meta-analysis of the safety of voriconazole in definitive, empirical, and prophylactic therapies for invasive fungal infections. BMC Infect. Dis. 2017, 17, 798. [Google Scholar] [CrossRef] [Green Version]
- Levine, M.T.; Chandrasekar, P.H. Adverse effects of voriconazole: Over a decade of use. Clin. Transplant. 2016, 30, 1377–1386. [Google Scholar] [CrossRef]
- Bhatia, A.; Kanish, B.; Badyal, D.; Kate, P.; Choudhary, S. Efficacy of oral terbinafine versus itraconazole in treatment of dermatophytic infection of skin—A prospective, randomized comparative study. Indian J. Pharmacol. 2019, 51, 116–119. [Google Scholar] [CrossRef]
- de los Ángeles Mejías Manzano, M.; Giráldez Gallego, Á.; Ontanilla Clavijo, G.; Sousa Martín, J.M. Terbinafine-induced hepatotoxicity. Gastroenterol. Hepatol. 2019, 42, 394–395. [Google Scholar] [CrossRef]
- Shapiro, M.; Li, L.J.; Miller, J. Terbinafine-induced neutropenia. Br. J. Dermatol. 1999, 140, 1196–1197. [Google Scholar]
- Hüttel, W. Echinocandins: Structural diversity, biosynthesis, and development of antimycotics. Appl. Microbiol. Biotechnol. 2021, 105, 55–66. [Google Scholar] [CrossRef]
- Pristov, K.E.; Ghannoum, M.A. Resistance of Candida to azoles and echinocandins worldwide. Clin. Microbiol. Infect. 2019, 25, 792–798. [Google Scholar] [CrossRef]
- Mota Fernandes, C.; Dasilva, D.; Haranahalli, K.; McCarthy, J.B.; Mallamo, J.; Ojima, I.; Del Poeta, M. The Future of Antifungal Drug Therapy: Novel Compounds and Targets. Antimicrob. Agents Chemother. 2021, 65, e01719-20. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, M.W.; Kontoyiannis, D.P.; Cornely, O.A.; Perfect, J.R.; Walsh, T.J. Novel Agents and Drug Targets to Meet the Challenges of Resistant Fungi. J. Infect. Dis. 2017, 216, S474–S483. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nguyen, S.; Truong, J.Q.; Bruning, J.B. Targeting Unconventional Pathways in Pursuit of Novel Antifungals. Front. Mol. Biosci. 2021, 7, 621366. [Google Scholar] [CrossRef] [PubMed]
- Bavaro, D.F.; Balena, F.; Ronga, L.; Signorile, F.; Romanelli, F.; Stolfa, S.; Sparapano, E.; De Carlo, C.; Mosca, A.; Monno, L.; et al. Emerging issue of fluconazole-resistant candidemia in a tertiary care hospital of southern italy: Time for antifungal stewardship program. J. Mycol. Med. 2021, 32, 101206. [Google Scholar] [CrossRef]
- Chakrabarti, A.; Sood, P. On the emergence, spread and resistance of Candida auris: Host, pathogen and environmental tipping points. J. Med. Microbiol. 2021, 70, 001318. [Google Scholar] [CrossRef]
- Hu, S.; Zhu, F.; Jiang, W.; Wang, Y.; Quan, Y.; Zhang, G.; Gu, F.; Yang, Y. Retrospective Analysis of the Clinical Characteristics of Candida auris Infection Worldwide from 2009 to 2020. Front. Microbiol. 2021, 12, 1278. [Google Scholar] [CrossRef]
- Arastehfar, A.; Gabaldón, T.; Garcia-Rubio, R.; Jenks, J.D.; Hoenigl, M.; Salzer, H.J.F.; Ilkit, M.; Lass-Flörl, C.; Perlin, D.S. Drug-Resistant Fungi: An Emerging Challenge Threatening Our Limited Antifungal Armamentarium. Antibiotics 2020, 9, 877. [Google Scholar] [CrossRef]
- Sanglard, D. Emerging threats in antifungal-resistant fungal pathogens. Front. Med. 2016, 3, 11. [Google Scholar] [CrossRef] [Green Version]
- Holmes, A.R.; Cardno, T.S.; Strouse, J.J.; Ivnitski-Steele, I.; Keniya, M.V.; Lackovic, K.; Monk, B.C.; Sklar, L.A.; Cannon, R.D. Targeting efflux pumps to overcome antifungal drug resistance. Future Med. Chem. 2016, 8, 1485. [Google Scholar] [CrossRef] [Green Version]
- Alcazar-Fuoli, L.; Mellado, E. Ergosterol biosynthesis in Aspergillus fumigatus: Its relevance as an antifungal target and role in antifungal drug resistance. Front. Microbiol. 2012, 3, 439. [Google Scholar] [CrossRef] [Green Version]
- Kuplińska, A.; Rząd, K. Molecular targets for antifungals in amino acid and protein biosynthetic pathways. Amino Acids 2021, 53, 961. [Google Scholar] [CrossRef] [PubMed]
- Rayens, E.; Rabacal, W.; Kang, S.E.; Celia, B.N.; Momany, M.; Norris, K.A. Vaccine-Induced Protection in Two Murine Models of Invasive Pulmonary Aspergillosis. Front. Immunol. 2021, 12, 1837. [Google Scholar] [CrossRef]
- Yang, L.B.; Guo, G.; Tian, Z.Q.; Zhou, L.X.; Zhu, L.J.; Peng, J.; Sun, C.Q.; Huang, M.J. TMT-based quantitative proteomic analysis of the effects of novel antimicrobial peptide AMP-17 against Candida albicans. J. Proteomics 2022, 250, 104385. [Google Scholar] [CrossRef] [PubMed]
- de Aguiar, F.L.L.; Santos, N.C.; de Paula Cavalcante, C.S.; Andreu, D.; Baptista, G.R.; Gonçalves, S. Antibiofilm Activity on Candida albicans and Mechanism of Action on Biomembrane Models of the Antimicrobial Peptide Ctn [15–34]. Int. J. Mol. Sci. 2020, 21, 8339. [Google Scholar] [CrossRef]
- Roscetto, E.; Contursi, P.; Vollaro, A.; Fusco, S.; Notomista, E.; Catania, M.R. Antifungal and anti-biofilm activity of the first cryptic antimicrobial peptide from an archaeal protein against Candida spp. clinical isolates. Sci. Rep. 2018, 8, 17570. [Google Scholar] [CrossRef]
- Ivanov, M.; Kannan, A.; Stojković, D.S.; Glamočlija, J.; Calhelha, R.C.; Ferreira, I.C.F.R.; Sanglard, D.; Soković, M. Flavones, flavonols, and glycosylated derivatives—Impact on Candida albicans growth and virulence, expression of cdr1 and erg11, cytotoxicity. Pharmaceuticals 2021, 14, 27. [Google Scholar] [CrossRef]
- Dell’olmo, E.; Gaglione, R.; Cesaro, A.; Cafaro, V.; Teertstra, W.R.; De Cock, H.; Notomista, E.; Haagsman, H.P.; Veldhuizen, E.J.A.; Arciello, A. Host defence peptides identified in human apolipoprotein B as promising antifungal agents. Appl. Microbiol. Biotechnol. 2015, 105, 1953–1964. [Google Scholar] [CrossRef] [PubMed]
- Bresciani, F.R.; Santi, L.; Beys-da-Silva, W.O.; Berger, M.; de A. Barcellos, V.; Schripsema, J.; von Poser, G.L.; Guimarães, J.A.; Vainstein, M.H. Antifungal activity of Allamanda polyantha seed extract and its iridoids promote morphological alterations in Cryptococcus spp. Arch. Pharm. 2020, 353, 2000133. [Google Scholar] [CrossRef]
- Ivanov, M.; Gašić, U.; Stojković, D.; Kostić, M.; Mišić, D.; Soković, M. New Evidence for Artemisia absinthium L. Application in Gastrointestinal Ailments: Ethnopharmacology, Antimicrobial Capacity, Cytotoxicity, and Phenolic Profile. Evid.-Based Complement Altern. Med. 2021, 2021, 9961089. [Google Scholar] [CrossRef]
- Yamashita, K.; Miyazaki, T.; Fukuda, Y.; Mitsuyama, J.; Saijo, T.; Shimamura, S.; Yamamoto, K.; Imamura, Y.; Izumikawa, K.; Yanagihara, K.; et al. The novel arylamidine T-2307 selectively disrupts yeast mitochondrial function by inhibiting respiratory chain complexes. Antimicrob. Agents Chemother. 2019, 63, e00374-19. [Google Scholar] [CrossRef] [Green Version]
- Ivanov, M.; Kannan, A.; Stojković, D.; Glamočlija, J.; Grdadolnik, S.G.; Sanglard, D.; Soković, M. Revealing the astragalin mode of anticandidal action. EXCLI J. 2020, 19, 1436–1445. [Google Scholar] [CrossRef] [PubMed]
- Staniszewska, M.; Kuryk, Ł.; Gryciuk, A.; Kawalec, J.; Rogalska, M.; Baran, J.; Łukowska-Chojnacka, E.; Kowalkowska, A. In Vitro Anti-Candida Activity and Action Mode of Benzoxazole Derivatives. Molecules 2021, 26, 5008. [Google Scholar] [CrossRef] [PubMed]
- Tong, Y.; Zhang, J.; Sun, N.; Wang, X.M.; Wei, Q.; Zhang, Y.; Huang, R.; Pu, Y.; Dai, H.; Ren, B.; et al. Berberine reverses multidrug resistance in Candida albicans by hijacking the drug efflux pump Mdr1p. Sci. Bull. 2021, 66, 1895–1905. [Google Scholar] [CrossRef]
- Zhang, M.; Chang, W.; Shi, H.; Zhou, Y.; Zheng, S.; Li, Y.; Li, L.; Lou, H. Biatriosporin D displays anti-virulence activity through decreasing the intracellular cAMP levels. Toxicol. Appl. Pharmacol. 2017, 322, 104–112. [Google Scholar] [CrossRef]
- Bapat, P.S.; Nobile, C.J. Photodynamic Therapy Is Effective Against Candida auris Biofilms. Front. Cell. Infect. Microbiol. 2021, 11, 819. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, M.; Kannan, A.; Stojković, D.S.; Glamočlija, J.; Calhelha, R.C.; Ferreira, I.C.F.R.; Sanglard, D.; Soković, M. Camphor and Eucalyptol-Anticandidal Spectrum, Antivirulence Effect, Efflux Pumps Interference and Cytotoxicity. Int. J. Mol. Sci. 2021, 22, 483. [Google Scholar] [CrossRef]
- Feldman, M.; Sionov, R.V.; Mechoulam, R.; Steinberg, D. Anti-Biofilm Activity of Cannabidiol against Candida albicans. Microorganisms 2021, 9, 441. [Google Scholar] [CrossRef]
- Zhang, T.; Chen, W. The Candida albicans Inhibitory Activity of the Extract from Papaya (Carica papaya L.) Seed Relates to Mitochondria Dysfunction. Int. J. Mol. Sci. 2017, 18, 1858. [Google Scholar] [CrossRef] [Green Version]
- Ravi, J.; Ravichellam, S.; Ponnuchamy, K.; Shunmugiah, K.P.; Shanmugaraj, G. Catechol thwarts virulent dimorphism in Candida albicans and potentiates the antifungal efficacy of azoles and polyenes. Sci. Rep. 2021, 11, 1–20. [Google Scholar] [CrossRef]
- Zheng, S.; Chang, W.; Zhang, M.; Shi, H.; Lou, H. Chiloscyphenol A derived from Chinese liverworts exerts fungicidal action by eliciting both mitochondrial dysfunction and plasma membrane destruction. Sci. Rep. 2018, 8, 326. [Google Scholar] [CrossRef] [Green Version]
- Rajasekar, V.; Darne, P.; Prabhune, A.; Kao, R.Y.T.; Solomon, A.P.; Ramage, G.; Samaranayake, L.; Neelakantan, P. A curcumin-sophorolipid nanocomplex inhibits Candida albicans filamentation and biofilm development. Colloids Surf. B Biointerfaces 2021, 200, 111617. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Liu, X.; Zhong, L.; Sui, Y.; Quan, G.; Huang, Y.; Wang, F.; Ma, T. Dioscin Inhibits Virulence Factors of Candida albicans. Biomed Res. Int. 2018, 2018, 4651726. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ko, H.T.; Hsu, L.H.; Yang, S.Y.; Chen, Y.L. Repurposing the thrombopoietin receptor agonist eltrombopag as an anticryptococcal agent. Med. Mycol. 2020, 58, 493–504. [Google Scholar] [CrossRef]
- Janeczko, M.; Masłyk, M.; Kubiński, K.; Golczyk, H. Emodin, a natural inhibitor of protein kinase CK2, suppresses growth, hyphal development, and biofilm formation of Candida albicans. Yeast 2017, 34, 253–265. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, R.H.; Shang, Z.C.; Li, T.X.; Yang, M.H.; Kong, L.Y. In Vitro Antibiofilm Activity of Eucarobustol E against Candida albicans. Antimicrob. Agents Chemother. 2017, 61, e02707-16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Wang, K.; Masso-Silva, J.A.; Rivera, A.; Xue, C. A heat-killed Cryptococcus mutant strain induces host protection against multiple invasive mycoses in a murine vaccine model. MBio 2019, 10, e02145-19. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Shan, M.; Zhu, Y.; Yao, H.; Li, H.; Gu, B.; Zhu, Z. Kalopanaxsaponin A induces reactive oxygen species mediated mitochondrial dysfunction and cell membrane destruction in Candida albicans. PLoS ONE 2020, 15, e0243066. [Google Scholar] [CrossRef]
- Kim, J.; Ha Quang Bao, T.; Shin, Y.K.; Kim, K.Y. Antifungal activity of magnoflorine against Candida strains. World J. Microbiol. Biotechnol. 2018, 34, 167. [Google Scholar] [CrossRef]
- Montoya, M.C.; Beattie, S.; Alden, K.M.; Krysan, D.J. Derivatives of the antimalarial drug mefloquine are broad-spectrum antifungal molecules with activity against drug-resistant clinical isolates. Antimicrob. Agents Chemother. 2020, 64, e02331-19. [Google Scholar] [CrossRef]
- Bae, M.; Kim, H.; Moon, K.; Nam, S.J.; Shin, J.; Oh, K.B.; Oh, D.C. Mohangamides A and B, new dilactone-tethered pseudo-dimeric peptides inhibiting Candida albicans isocitrate lyase. Org. Lett. 2015, 17, 712–715. [Google Scholar] [CrossRef]
- Ansari, M.A.; Fatima, Z.; Ahmad, K.; Hameed, S. Monoterpenoid perillyl alcohol impairs metabolic flexibility of Candida albicans by inhibiting glyoxylate cycle. Biochem. Biophys. Res. Commun. 2018, 495, 560–566. [Google Scholar] [CrossRef] [PubMed]
- Abirami, G.; Alexpandi, R.; Durgadevi, R.; Kannappan, A.; Veera Ravi, A. Inhibitory Effect of Morin Against Candida albicans Pathogenicity and Virulence Factor Production: An in vitro and in vivo Approaches. Front. Microbiol. 2020, 11, 23020. [Google Scholar] [CrossRef] [PubMed]
- Tarang, S.; Kesherwani, V.; LaTendresse, B.; Lindgren, L.; Rocha-Sanchez, S.M.; Weston, M.D. In silico Design of a Multivalent Vaccine Against Candida albicans. Sci. Rep. 2020, 10, 1066. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Szerencsés, B.; Gácser, A.; Endre, G.; Domonkos, I.; Tiricz, H.; Vágvölgyi, C.; Szolomajer, J.; Howan, D.H.O.; Tóth, G.K.; Pfeiffer, I.; et al. Symbiotic NCR Peptide Fragments Affect the Viability, Morphology and Biofilm Formation of Candida Species. Int. J. Mol. Sci. 2021, 22, 3666. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Uppuluri, P.; Mamouei, Z.; Alqarihi, A.; Elhassan, H.; French, S.; Lockhart, S.R.; Chiller, T.; Edwards, J.E.; Ibrahim, A.S. The NDV-3A vaccine protects mice from multidrug resistant Candida auris infection. PLOS Pathog. 2019, 15, e1007460. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stojković, D.; Dias, M.I.; Drakulić, D.; Barros, L.; Stevanović, M.; Ferreira, I.C.F.R.; Soković, M.D. Methanolic extract of the herb ononis spinosa L. Is an antifungal agent with no cytotoxicity to primary human cells. Pharmaceuticals 2020, 13, 78. [Google Scholar] [CrossRef]
- Han, X.; Zhu, X.; Hong, Z.; Wei, L.; Ren, Y.; Wan, F.; Zhu, S.; Peng, H.; Guo, L.; Rao, L.; et al. Structure-Based Rational Design of Novel Inhibitors against Fructose-1,6-Bisphosphate Aldolase from Candida albicans. J. Chem. Inf. Model. 2017, 57, 1426–1438. [Google Scholar] [CrossRef]
- Shukla, M.; Rohatgi, S. Vaccination with secreted aspartyl proteinase 2 protein from candida parapsilosis can enhance survival of mice during c. tropicalis-mediated systemic candidiasis. Infect. Immun. 2020, 88, e00312-20. [Google Scholar] [CrossRef]
- Ivanov, M.; Kostić, M.; Stojković, D.; Soković, M. Rosmarinic acid–Modes of antimicrobial and antibiofilm activities of common plant polyphenol. S. Afr. J. Bot. 2022, 146, 521–527. [Google Scholar] [CrossRef]
- Yan, Y.; Tan, F.; Miao, H.; Wang, H.; Cao, Y.Y. Effect of Shikonin Against Candida albicans Biofilms. Front. Microbiol. 2019, 10, 1085. [Google Scholar] [CrossRef] [Green Version]
- Haque, F.; Alfatah, M.; Ganesan, K.; Bhattacharyya, M.S. Inhibitory Effect of Sophorolipid on Candida albicans Biofilm Formation and Hyphal Growth. Sci. Rep. 2016, 6, 23575. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Güzel-Akdemir, Ö.; Carradori, S.; Grande, R.; Demir-Yazıcı, K.; Angeli, A.; Supuran, C.T.; Akdemir, A. Development of Thiazolidinones as Fungal Carbonic Anhydrase Inhibitors. Int. J. Mol. Sci. 2020, 21, 2960. [Google Scholar] [CrossRef] [Green Version]
- Yan, T.; Li, F.; Li, J.; Chen, F. Antifungal Activity of ToAP2D Peptide Against Sporothrix globosa. Front. Bioeng. Biotechnol. 2021, 9, 874. [Google Scholar] [CrossRef]
- Nithyanand, P.; Beema Shafreen, R.M.; Muthamil, S.; Karutha Pandian, S. Usnic acid inhibits biofilm formation and virulent morphological traits of Candida albicans. Microbiol. Res. 2015, 179, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Folly, M.L.C.; Ferreira, G.F.; Salvador, M.R.; Sathler, A.A.; da Silva, G.F.; Santos, J.C.B.; dos Santos, J.R.A.; Nunes Neto, W.R.; Rodrigues, J.F.S.; Fernandes, E.S.; et al. Evaluation of in vitro Antifungal Activity of Xylosma prockia (Turcz.) Turcz. (Salicaceae) Leaves against Cryptococcus spp. Front. Microbiol. 2020, 10, 3114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jahanshiri, Z.; Shams-Ghahfarokhi, M.; Asghari-Paskiabi, F.; Saghiri, R.; Razzaghi-Abyaneh, M. α-Bisabolol inhibits Aspergillus fumigatus Af239 growth via affecting microsomal ∆24-sterol methyltransferase as a crucial enzyme in ergosterol biosynthesis pathway. World J. Microbiol. Biotechnol. 2017, 33, 55. [Google Scholar] [CrossRef]
- Horn, C.; Vediyappan, G. Anticapsular and Antifungal Activity of α-Cyperone. Antibiotics 2021, 10, 51. [Google Scholar] [CrossRef]
- De Micheli, M.; Bille, J.; Schueller, C.; Sanglard, D. A common drug-responsive element mediates the upregulation of the Candida albicans ABC transporters CDR1 and CDR2, two genes involved in antifungal drug resistance. Mol. Microbiol. 2002, 43, 1197–1214. [Google Scholar] [CrossRef]
- Shao, J.; Zhang, M.; Wang, T.; Li, Y.; Wang, C. The roles of CDR1, CDR2, and MDR1 in kaempferol-induced suppression with fluconazole-resistant Candida albicans. Pharm. Biol. 2015, 54, 984–992. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yong, J.; Zu, R.; Huang, X.; Ge, Y.; Li, Y. Synergistic Effect of Berberine Hydrochloride and Fluconazole Against Candida albicans Resistant Isolates. Front. Microbiol. 2020, 11, 1498. [Google Scholar] [CrossRef]
- Jamiu, A.T.; Albertyn, J.; Sebolai, O.; Gcilitshana, O.; Pohl, C.H. Inhibitory effect of polyunsaturated fatty acids alone or in combination with fluconazole on Candida krusei biofilms in vitro and in Caenorhabditis elegans. Med. Mycol. 2021, 59, 1225–1237. [Google Scholar] [CrossRef] [PubMed]
- Lv, Q.Z.; Yan, L.; Jiang, Y.Y. The synthesis, regulation, and functions of sterols in Candida albicans: Well-known but still lots to learn. Virulence 2016, 7, 649. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Flowers, S.A.; Colón, B.; Whaley, S.G.; Schuler, M.A.; David Rogers, P. Contribution of clinically derived mutations in ERG11 to azole resistance in Candida albicans. Antimicrob. Agents Chemother. 2015, 59, 450–460. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prasath, K.G.; Tharani, H.; Kumar, M.S.; Pandian, S.K. Palmitic Acid Inhibits the Virulence Factors of Candida tropicalis: Biofilms, Cell Surface Hydrophobicity, Ergosterol Biosynthesis, and Enzymatic Activity. Front. Microbiol. 2020, 11, 864. [Google Scholar] [CrossRef] [PubMed]
- Bvumbi, C.; Chi, G.F.; Stevens, M.Y.; Mombeshora, M.; Mukanganyama, S. The Effects of Tormentic Acid and Extracts from Callistemon citrinus on Candida albicans and Candida tropicalis Growth and Inhibition of Ergosterol Biosynthesis in Candida albicans. Sci. World J. 2021, 2021, 8856147. [Google Scholar] [CrossRef]
- An, Y.; Liu, W.; Xie, H.; Fan, H.; Han, J.; Sun, B. Construction and activity evaluation of novel benzodioxane derivatives as dual-target antifungal inhibitors. Eur. J. Med. Chem. 2022, 227, 113950. [Google Scholar] [CrossRef] [PubMed]
- Trifan, A.; Bostănaru, A.C.; Luca, S.V.; Temml, V.; Akram, M.; Herdlinger, S.; Kulinowski, Ł.; Skalicka-Woźniak, K.; Granica, S.; Czerwińska, M.E.; et al. Honokiol and Magnolol: Insights into Their Antidermatophytic Effects. Plants 2021, 10, 2522. [Google Scholar] [CrossRef]
- Ebrahimi-Shaghaghi, F.; Noormohammadi, Z.; Atyabi, S.M.; Razzaghi-Abyaneh, M. Inhibitory effects of cold atmospheric plasma on the growth, virulence factors and HSP90 gene expression in Candida albicans. Arch. Biochem. Biophys. 2021, 700, 108772. [Google Scholar] [CrossRef]
- Robbins, N.; Uppuluri, P.; Nett, J.; Rajendran, R.; Ramage, G.; Lopez-Ribot, J.L.; Andes, D.; Cowen, L.E. Hsp90 Governs Dispersion and Drug Resistance of Fungal Biofilms. PLOS Pathog. 2011, 7, e1002257. [Google Scholar] [CrossRef]
- Whitesell, L.; Robbins, N.; Huang, D.S.; McLellan, C.A.; Shekhar-Guturja, T.; LeBlanc, E.V.; Nation, C.S.; Hui, R.; Hutchinson, A.; Collins, C.; et al. Structural basis for species-selective targeting of Hsp90 in a pathogenic fungus. Nat. Commun. 2019, 10, 402. [Google Scholar] [CrossRef] [Green Version]
- Yuan, R.; Tu, J.; Sheng, C.; Chen, X.; Liu, N. Effects of Hsp90 Inhibitor Ganetespib on Inhibition of Azole-Resistant Candida albicans. Front. Microbiol. 2021, 12, 1280. [Google Scholar] [CrossRef]
- Tripathi, S.K.; Feng, Q.; Liu, L.; Levin, D.E.; Roy, K.K.; Doerksen, R.J.; Baerson, S.R.; Shi, X.; Pan, X.; Xu, W.-H.; et al. Puupehenone, a Marine-Sponge-Derived Sesquiterpene Quinone, Potentiates the Antifungal Drug Caspofungin by Disrupting Hsp90 Activity and the Cell Wall Integrity Pathway. Msphere 2020, 5, e00818-19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tu, B.; Yin, G.; Li, H. Synergistic effects of vorinostat (SAHA) and azoles against Aspergillus species and their biofilms. BMC Microbiol. 2020, 20, 28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kernien, J.F.; Snarr, B.D.; Sheppard, D.C.; Nett, J.E. The interface between fungal biofilms and innate immunity. Front. Immunol. 2018, 8, 1968. [Google Scholar] [CrossRef] [Green Version]
- Silva, S.; Rodrigues, C.F.; Araújo, D.; Rodrigues, M.E.; Henriques, M. Candida Species Biofilms’ Antifungal Resistance. J. Fungi 2017, 3, 8. [Google Scholar] [CrossRef] [Green Version]
- Pierce, C.G.; Vila, T.; Romo, J.A.; Montelongo-Jauregui, D.; Wall, G.; Ramasubramanian, A.; Lopez-Ribot, J.L. The Candida albicans Biofilm Matrix: Composition, Structure and Function. J. Fungi 2017, 3, 14. [Google Scholar] [CrossRef] [Green Version]
- Li, Z.; Nielsen, K. Morphology Changes in Human Fungal Pathogens upon Interaction with the Host. J. Fungi 2017, 3, 66. [Google Scholar] [CrossRef] [Green Version]
- Bar-Yosef, H.; Vivanco Gonzalez, N.; Ben-Aroya, S.; Kron, S.J.; Kornitzer, D. Chemical inhibitors of Candida albicans hyphal morphogenesis target endocytosis. Sci. Rep. 2017, 7, 5692. [Google Scholar] [CrossRef] [Green Version]
- Ćirić, A.D.; Petrović, J.D.; Glamočlija, J.M.; Smiljković, M.S.; Nikolić, M.M.; Stojković, D.S.; Soković, M.D. Natural products as biofilm formation antagonists and regulators of quorum sensing functions: A comprehensive review update and future trends. S. Afr. J. Bot. 2019, 120, 65–80. [Google Scholar] [CrossRef]
- Smiljković, M.; Kostić, M.; Stojković, D.; Glamočlija, J.; Soković, M. Could Flavonoids Compete with Synthetic Azoles in Diminishing Candida albicans Infections? A Comparative Review Based on In Vitro Studies. Curr. Med. Chem. 2018, 26, 2536–2554. [Google Scholar] [CrossRef]
- Singh, B.N.; Upreti, D.K.; Singh, B.R.; Pandey, G.; Verma, S.; Roy, S.; Naqvi, A.H.; Rawat, A.K.S. Quercetin sensitizes fluconazole-resistant Candida albicans to induce apoptotic cell death by modulating quorum sensing. Antimicrob. Agents Chemother. 2015, 59, 2153–2168. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, M.; Wang, H.; Zhu, L. Quercetin Assists Fluconazole to Inhibit Biofilm Formations of Fluconazole-Resistant Candida albicans in In Vitro and In Vivo Antifungal Managements of Vulvovaginal Candidiasis. Cell. Physiol. Biochem. 2016, 40, 727–742. [Google Scholar] [CrossRef] [PubMed]
- Manoharan, R.K.; Lee, J.H.; Lee, J. Antibiofilm and antihyphal activities of cedar leaf essential oil, camphor, and fenchone derivatives against Candida albicans. Front. Microbiol. 2017, 8, 1476. [Google Scholar] [CrossRef] [Green Version]
- Bruni, N.; Capucchio, M.T.; Biasibetti, E.; Pessione, E.; Cirrincione, S.; Giraudo, L.; Corona, A.; Dosio, F. Antimicrobial Activity of Lactoferrin-Related Peptides and Applications in Human and Veterinary Medicine. Molecules 2016, 21, 752. [Google Scholar] [CrossRef] [PubMed]
- Zaragoza, O. Basic principles of the virulence of Cryptococcus. Virulence 2019, 10, 490–501. [Google Scholar] [CrossRef] [Green Version]
- Mayer, F.L.; Kronstad, J.W. Disarming Fungal Pathogens: Bacillus safensis Inhibits Virulence Factor Production and Biofilm Formation by Cryptococcus neoformans and Candida albicans. MBio 2017, 8, e01537-17. [Google Scholar] [CrossRef] [Green Version]
- Li, D.; Calderone, R. Exploiting mitochondria as targets for the development of new antifungals. Virulence 2017, 8, 159. [Google Scholar] [CrossRef] [Green Version]
- Pan, J.; Hu, C.; Yu, J.H. Lipid Biosynthesis as an Antifungal Target. J. Fungi 2018, 4, 50. [Google Scholar] [CrossRef] [Green Version]
- Wijnants, S.; Vreys, J.; Van Dijck, P. Interesting antifungal drug targets in the central metabolism of Candida albicans. Trends Pharmacol. Sci. 2022, 43, 69–79. [Google Scholar] [CrossRef]
- Ramakrishnan, J.; Rathore, S.S.; Raman, T. Review on fungal enzyme inhibitors—potential drug targets to manage human fungal infections. RSC Adv. 2016, 6, 42387–42401. [Google Scholar] [CrossRef]
- Venkata, S.; Zeeshan, F.; Kamal, A.; Luqman, A.K.; Saif, H. Efficiency of vanillin in impeding metabolic adaptability and virulence of Candida albicans by inhibiting glyoxylate cycle, morphogenesis, and biofilm formation. Curr. Med. Mycol. 2020, 6, 1. [Google Scholar] [CrossRef] [PubMed]
- Cortés, J.C.G.; Curto, M.Á.; Carvalho, V.S.D.; Pérez, P.; Ribas, J.C. The fungal cell wall as a target for the development of new antifungal therapies. Biotechnol. Adv. 2019, 37, 107352. [Google Scholar] [CrossRef]
- Curto, M.Á.; Butassi, E.; Ribas, J.C.; Svetaz, L.A.; Cortés, J.C.G. Natural products targeting the synthesis of β(1,3)-D-glucan and chitin of the fungal cell wall. Existing drugs and recent findings. Phytomedicine 2021, 88, 153556. [Google Scholar] [CrossRef]
- Cabib, E.; Schmidt, M. Chitin synthase III activity, but not the chitin ring, is required for remedial septa formation in budding yeast. FEMS Microbiol. Lett. 2003, 224, 299–305. [Google Scholar] [CrossRef] [Green Version]
- Schmidt, M.; Bowers, B.; Varma, A.; Roh, D.H.; Cabib, E. In budding yeast, contraction of the actomyosin ring and formation of the primary septum at cytokinesis depend on each other. J. Cell Sci. 2002, 115, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Ibe, C.; Munro, C.A. Fungal cell wall: An underexploited target for antifungal therapies. PLOS Pathog. 2021, 17, e1009470. [Google Scholar] [CrossRef]
- Larwood, D.J. Nikkomycin Z—Ready to Meet the Promise? J. Fungi 2020, 6, 261. [Google Scholar] [CrossRef]
- Jallow, S.; Govender, N.P. Ibrexafungerp: A First-in-Class Oral Triterpenoid Glucan Synthase Inhibitor. J. Fungi 2021, 7, 163. [Google Scholar] [CrossRef]
- Ge, Z.; Ji, Q.; Chen, C.; Liao, Q.; Wu, H.; Liu, X.; Huang, Y.; Yuan, L.; Liao, F. Synthesis and biological evaluation of novel 3-substituted amino-4-hydroxylcoumarin derivatives as chitin synthase inhibitors and antifungal agents. J. Enzyme Inhib. Med. Chem. 2016, 31, 219–228. [Google Scholar] [CrossRef] [Green Version]
- Mann, P.A.; McLellan, C.A.; Koseoglu, S.; Si, Q.; Kuzmin, E.; Flattery, A.; Harris, G.; Sher, X.; Murgolo, N.; Wang, H.; et al. Chemical Genomics-Based Antifungal Drug Discovery: Targeting Glycosylphosphatidylinositol (GPI) Precursor Biosynthesis. ACS Infect. Dis. 2015, 1, 59–72. [Google Scholar] [CrossRef]
- Wan Tso, G.H.; Reales-Calderon, J.A.; Pavelka, N. The Elusive Anti-Candida Vaccine: Lessons from the past and opportunities for the future. Front. Immunol. 2018, 9, 897. [Google Scholar] [CrossRef]
- Oliveira, L.V.N.; Wang, R.; Specht, C.A.; Levitz, S.M. Vaccines for human fungal diseases: Close but still a long way to go. npj Vaccines 2021, 6, 33. [Google Scholar] [CrossRef] [PubMed]
- Madende, M.; Albertyn, J.; Sebolai, O.; Pohl, C.H. Caenorhabditis elegans as a model animal for investigating fungal pathogenesis. Med. Microbiol. Immunol. 2019, 209, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Pujol, N.; Zugasti, O.; Wong, D.; Couillault, C.; Kurz, C.L.; Schulenburg, H.; Ewbank, J.J. Anti-fungal innate immunity in C. elegans is enhanced by evolutionary diversification of antimicrobial peptides. PLoS Pathog. 2008, 4, e1000105. [Google Scholar] [CrossRef] [Green Version]
- Wurster, S.; Bandi, A.; Beyda, N.D.; Albert, N.D.; Raman, N.M.; Raad, I.I.; Kontoyiannis, D.P. Drosophila melanogaster as a model to study virulence and azole treatment of the emerging pathogen Candida auris. J. Antimicrob. Chemother. 2019, 74, 1904–1910. [Google Scholar] [CrossRef]
- Xin, H.; Mohiuddin, F.; Tran, J.; Adams, A.; Eberle, K. Experimental Mouse Models of Disseminated Candida auris Infection. mSphere 2019, 4, e00339-19. [Google Scholar] [CrossRef] [Green Version]
- Nett, J.E.; Andes, D.R. Fungal Biofilms: In Vivo Models for Discovery of Anti-Biofilm Drugs. Microbiol. Spectr. 2015, 3, 3–4. [Google Scholar] [CrossRef] [Green Version]
- Rauseo, A.M.; Coler-Reilly, A.; Larson, L.; Spec, A. Hope on the Horizon: Novel Fungal Treatments in Development. Open forum Infect. Dis. 2020, 7, ofaa016. [Google Scholar] [CrossRef] [Green Version]
- Shaw, K.J.; Ibrahim, A.S. Fosmanogepix: A Review of the First-in-Class Broad Spectrum Agent for the Treatment of Invasive Fungal Infections. J. Fungi 2020, 6, 239. [Google Scholar] [CrossRef]
- Wiederhold, N.P. Review of the Novel Investigational Antifungal Olorofim. J. Fungi 2020, 6, 122. [Google Scholar] [CrossRef]

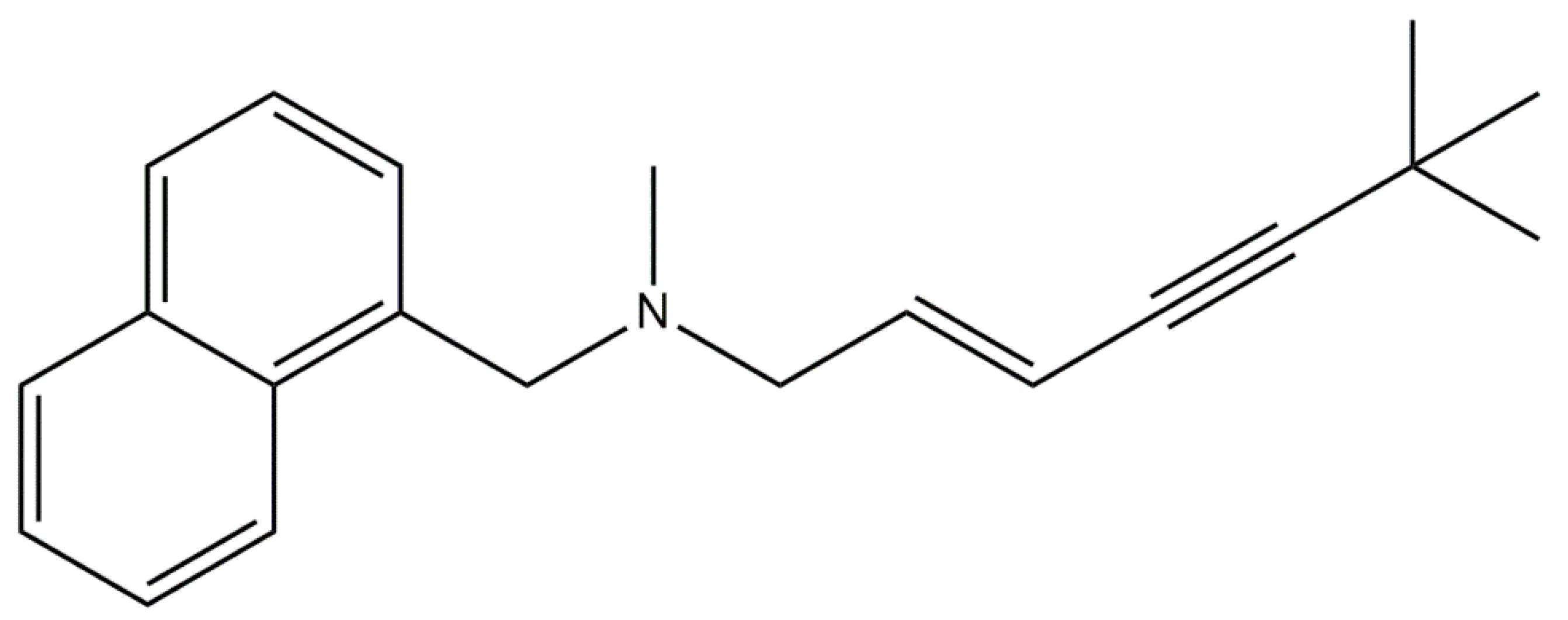
| Product | Target | Assay | Fungi | Active Concentration | Reference |
|---|---|---|---|---|---|
| (S)-2-amino-4-oxo-5-hydroxypentanoic acid | Homoserine dehydrogenase | Spectroscopic studies, and X-ray crystallography | Candida albicans, C. tropicalis, C. glabrata | 64–128 µg/mL | [51] |
| AF.KEX1 | Endoprotease | Mice in vivo study | Aspergillus fumigatus KexB | 50 µg | [52] |
| AMP-17 | Range of metabolic pathway | Proteomic analysis | C. albicans ATCC 10231 | 32 μg/mL | [53] |
| Antimicrobial peptide Ctn [15–34] | Biofilm, cell membrane | XTT and fluorescent dyes, CLSM, and atomic force microscopy; Fluorescence assays | C. albicans (ATCC 90028) and C. albicans LABMIC 0125 | 10 µM | [54] |
| Antimicrobial peptide VLL-28 | Damaging the cell wall/biofilm | XTT reduction assay, CV assay, CLSM | Range of Candida strains | 12.5 –25 µM | [55] |
| Apigenin, Apigetrin, Luteolin, Quercetin, Quercitrin, Isoquercitrin, Rutin | Hyphal growth, biofilm, CDR1 | Microscopic examination, CV antibiofilm assay, qRT-PCR | C. albicans (475/15) | 0.0375–0.075 mg/mL | [56] |
| ApoB-derived peptides | Cell membrane, metabolic activity | Propidium iodide intake; cell proliferator reagent WST-1 | C. albicans ATCC 10231, A. niger N402 | 5–40 µM | [57] |
| Aqueous seed extract from Allamanda polyantha | Capsule formation, size | Fluorescent microscopy | C. neoformans H99 | 281–1126 µg/mL | [58] |
| Artemisia absinthium extract | Biofilm, EPS, cell membrane | CV antibiofilm assay, Congo red assay, nucleotide leakage and CV uptake assays | C. albicans (475/15) | 0.5 mg/mL | [59] |
| Arylamidine T-2307 | Inhibition of respiratory chain complexes | Assessment of respiratory chain enzymatic activities, measurement of intracellular ATP levels | Saccharomyces cerevisiae 24657 (D273-10B) and C. albicans MYA-2876 (SC5314) | 220–344 µM | [60] |
| Astragalin | Hyphal growth, cell membrane integrity | Microscopic examination, nucleotide leakage assay | C. albicans (475/15) | 0.075 mg/mL | [61] |
| Benzoxazole derivatives | Ergosterol, cell efflux | Ergosterol estimation assay using spectrophotometry and HPLC, Rhodamine 123 efflux | C. albicans (SC5314) | 0.125–160 µg/mL | [62] |
| Berberine | Mitochondrial function and Mdr1p | RNA-seq | C. albicans CaS and CaR | 32 μg/mL | [63] |
| Biatriosporin D | Hyphal growth | Microscopy, qRT-PCR | C. albicans (SC5314 and BWP17-DPP3-GFP) | 0.5–4.0 μg/ml | [64] |
| Blue light | Biofilm viability | Adherence inhibition, developmental inhibition, and disruption biofilm assays (CFU count LIVE/DEAD BacLight viability staining) | C. albicans (SC5314) and C. auris (AR0383, AR0389, and AR0390) | - | [65] |
| Camphor Eucalyptol | Hyphal growth and biofilm | Microscopy, CV antibiofilm assay | C. albicans (475/15) | 0.125 mg/mL and 23 mg/mL | [66] |
| Cannabidiol | EPS production | Confocal laser scanning microscopy (CLSM), qRT-PCR | C. albicans SC5314 | 50 µg/mL | [67] |
| Carica papaya seeds extract | Mitochondrial function | Measurement of mitochondrial membrane potential, assay for mitochondrial enzyme activities | C. albicans ATCC | 5–25 µg/mL | [68] |
| Catechol | Hyphal growth, biofilm, downregulation of RAS1, HWP1 and ALS3 | Microscopy, CV assay, RT-PCR | C. albicans (ATCC 10231) | 2–256 μg/mL | [69] |
| Chiloscyphenol A | Mitochondrial function | Analysis of mitochondrial membrane potential, measurement of intracellular ATP production, observation of the localization of Tom70-GFP | C. albicans (SC5314) | Up to 64 μg/mL | [70] |
| Curcumin-sophorolipid nanocomplex | Biofilm and hyphae | CV assay, CLSM, qRT-PCR | C. albicans (SC5314) | 9.37 μg/mL | [71] |
| Dioscin | Hyphal growth, biofilm, production of phospholipase | Microscopy, XTT reduction assay, CLSM, Egg yolk phospholipase assay | C. albicans (SC5314) | 4 μg/mL | [72] |
| Eltrombopag | Capsule and biofilm formation, melanin production | ImageJ of stained cells, XTT reduction assay, L-DOPA melanization assay | C. neoformans H99 | 0.06 mg/l | [73] |
| Emodin | Hyphal growth and biofilm formation | Morphology on Spider medium, MTT assay | C. albicans (ATCC 10231) | 12.50 μg/mL | [74] |
| Eucarobustol E | Hyphae, biofilm | Filmentation assay on hypaae inducing medium, XTT reduction assay, CLSM, SEM, RNA-seq, RT-PCR | C. albicans (SC5314 and ATCC 24433) | 8.0–16.0 μg/mL | [75] |
| Heat-killed fbp1Δ cells | F-box protein Fbp1, ligase subunit | Mice in vivo study | Cryptococcus neoformans H99 and its mutants, C. gattii R265 and C. albicans SC5314, A. fumigatus R21 | 5 × 107 heat-killed fungal cells | [76] |
| Kalopanaxsaponin A | Mitochondrial function and cell membrane | Asessment of alteration of mtΔψ and ATP content, PI staining, TEM | C. albicans (SC5314) | 8–32 µg/mL | [77] |
| Magnoflorine | Alpha-glucosidase | Spectrophotometric alpha-glucosidase inhibition assay | C. albicans (ATCC 10231) | 50 µg/mL | [78] |
| Mefloquine derivatives | Mitochondrial and vacuolar function | MitoTracker Red uptake | C. albicans (SC5314), C. neoformans H99 | 1–64 μg/mL | [79] |
| Mohangamide A | Isocitrate lyase in glyoxylate pathway | High-Throughput Screening | C. albicans (SC5314) | 4.4 µM | [80] |
| Monoterpenoid perillyl alcohol | Glyoxylate cycle inhibitor | Isocitrate lyase enzyme assay | C. albicans (SC5314) | 320 µg/mL | [81] |
| Morin | Hyphal growth, biofilm formation, phospholipase and exopolysaccharide production | Microscopic analysis; CV antibiofilm assay; Egg yolk assay; Fourier Transform Infrared Spectroscopy Analysis | C. albicans (ATCC 90028) | 150 μg/ml | [82] |
| mvPC | Multivalent | In silico study | C. albicans (SC5314) | - | [83] |
| NCR Peptide Fragments | Biofilm, hyphae | XTT reduction assay; Microscopy | C. albicans ATCC 10231, C. tropicalis CBS 94 | 0.78–12.5 µM | [84] |
| NDV-3A | Adhesins | In silico and in vivo mice studies | C. auris strains (CAU-01, CAU-03, CAU-05, CAU-07, and CAU-09) | 300 μg | [85] |
| Ononis spinosa methanolic extract | Biofilm, cell membrane | CV antibiofilm assay, nucleotide leakage assay | C. albicans (ATCC 10231) | 10 mg/mL | [86] |
| Phenylhydrazones | Fructose-1,6-bisphosphate aldolase | Molecular dynamics, enzyme inhibition assay | C. albicans (SC5314) | 2.7–4.1 µM | [87] |
| Recombinant Sap2 | Secreted aspartyl protease | Mice in vivo study | C. tropicalis (ATCC 750) | 10 μg | [88] |
| Rosmarinic acid | Biofilm, EPS production, mitochondrial activity | CV antibiofilm assay, Congo red binding assay, MTT assay | C. albicans (475/15) | 0.025–0.1 mg/mL | [89] |
| Shikonin | Hyphal growth, biofilm formation | Microscopy, XTT reduction assay, CLSM | C. albicans (SC5314) | 0.5–4 μg/mL | [90] |
| Sophorolipids | Biofilm and hyphae | XTT reduction assay, SEM and CLSM analysis, qRT-PCR | C. albicans (SC5314) | 15.0–30.0 μg/ml | [91] |
| Thiazolidinones | Carbonic anhydrase | Molecular docking and molecular dynamics, enzyme inhibition assay | Candida glabrata | 0.1–10 µM | [92] |
| ToAP2D Peptide | Mitochondria | JC-1 kit | Sporothrix globosa | 4 mg/mL | [93] |
| Usnic acid | EPS production | Spectrophotometrically, FT-IR analysis | C. albicans (ATCC 90028) | 100 μg/ml | [94] |
| Xylosma prockia ethyl acetate fraction | Ergosterol | Spectrophotometric quantification | C. gattii (ATCC 24065 and ATCC 32608) C. neoformans (ATCC 24067, ATCC 28957, ATCC 62066, and ATCC H99) | 1–16 mg/L | [95] |
| α-bisabolol | Ergosterol production | Spectrophotometry method | A. fumigatus (Af 239) | 0.28–9.0 mM | [96] |
| α-Cyperone | Capsule | Microscopic examination | C. neoformans | 16 µg/mL | [97] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ivanov, M.; Ćirić, A.; Stojković, D. Emerging Antifungal Targets and Strategies. Int. J. Mol. Sci. 2022, 23, 2756. https://doi.org/10.3390/ijms23052756
Ivanov M, Ćirić A, Stojković D. Emerging Antifungal Targets and Strategies. International Journal of Molecular Sciences. 2022; 23(5):2756. https://doi.org/10.3390/ijms23052756
Chicago/Turabian StyleIvanov, Marija, Ana Ćirić, and Dejan Stojković. 2022. "Emerging Antifungal Targets and Strategies" International Journal of Molecular Sciences 23, no. 5: 2756. https://doi.org/10.3390/ijms23052756







