Proteomic Assessment of Extracellular Vesicles from Canine Tissue Explants as a Pipeline to Identify Molecular Targets in Osteosarcoma: PSMD14/Rpn11 as a Proof of Principle
Abstract
:1. Introduction
2. Results
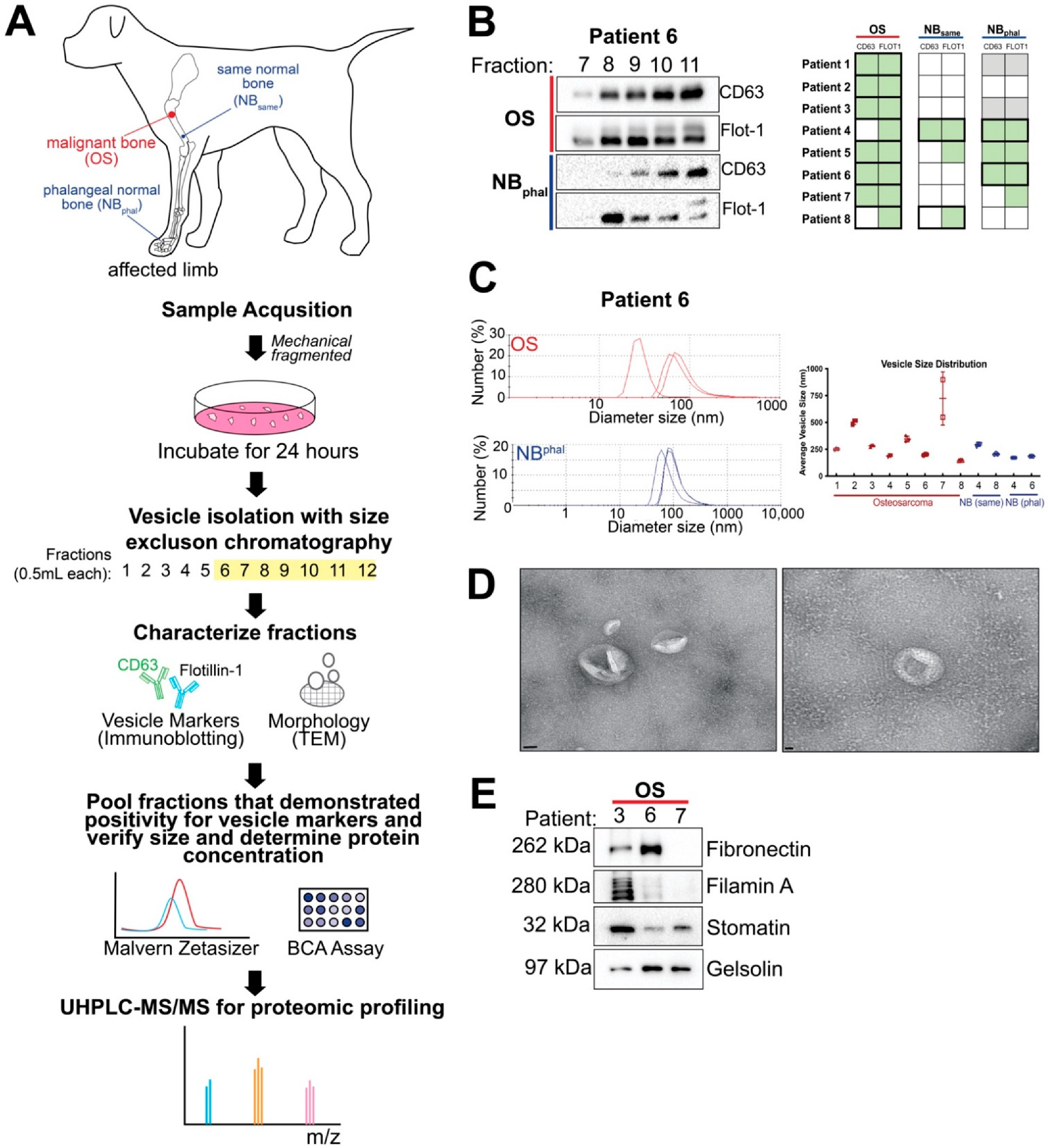
2.1. Extracellular Vesicles Can Be Successfully Isolated from OS and Normal Bone Tissue Explants
2.2. Twenty-Nine Proteins Are Significantly Increased in OS-Derived Vesicles Compared to Normal Bone-Derived Vesicles
2.3. Proteasome Components Are Clinically Relevant and of Current Interest in the Field of OS
2.4. OS Cell Lines Are Sensitive to Capzimin Treatment and Demonstrate Increased Ubiquitinated Products following Treatment
2.5. Capzimin Induces Apoptosis in a Dose-Dependent Manner in Canine OS Cells
2.6. Capzimin Reduces Clonogenic Survival, Cell Growth, and Migration
2.7. Capzimin Reduces Viability Compared to Doxorubicin Alone at Low Doses
2.8. Capzimin Reduces Viability of Metastatic Human OS Cells in 3D Conditions Resembling Secondary Sites
3. Discussion
4. Materials and Methods
4.1. Sample Acquisition and Explant Culture
4.2. Vesicle Isolation
4.3. Vesicle Lysis
4.4. Dynamic Light Scattering
4.5. Transmission Electron Micrscopy
4.6. Vesicle Protein Quantification
4.7. Ultra High Performance Liquid Chromatography and Tandem Mass Spectrometry (UHPLC-MS/MS)
4.8. Associations of mRNA Expression of Identified Proteins with Clinical Outcome
4.9. OS Cell Culture Conditions
4.10. Capzimin IC50 Dose Response
4.11. Capzimin Treatment & Lysate Collection for the Detection of Apoptosis
4.12. Cell Lysate Protein Quantification
4.13. Immunoblotting of Vesicle Fractions and Capzimin Treated Cells
4.14. Ubiqutinated Protein Enrichment
4.15. Annexin-V FITC and Propidium Iodide (PI) Staining and Flow Cytometry
4.16. Clonogenic Survival Assay
4.17. Phosphorylated Histone-H3 (pHH3) Immunoblabelling and Quantification
4.18. Cell Cycle Analysis
4.19. Transwell Assay
4.20. Combination Treatment: Capzimin and Doxorubicin
4.21. Assessment of MG63.3 Cytotoxicity in 2D and 3D Culture
4.22. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fenger, J.M.; London, C.A.; Kisseberth, W.C. Canine Osteosarcoma: A Naturally Occurring Disease to Inform Pediatric Oncology. ILAR J. 2014, 55, 69–85. [Google Scholar] [CrossRef] [Green Version]
- Dernell, W.; Ehrhart, N.; Straw, R.; Vail, D. Tumors of the Skeletal System. In Withrow & MacEwen’s Small Animal Clinical Oncology; Elsevier: Amsterdam, The Netherlands, 2007; Volume 23, pp. 540–582. [Google Scholar]
- Mirabello, L.; Troisi, R.J.; Savage, S.A. Osteosarcoma incidence and survival rates from 1973 to 2004. Cancer 2009, 115, 1531–1543. [Google Scholar] [CrossRef] [Green Version]
- Simpson, S.; Dunning, M.D.; de Brot, S.; Grau-Roma, L.; Mongan, N.P.; Rutland, C.S. Comparative review of human and canine osteosarcoma: Morphology, epidemiology, prognosis, treatment and genetics. Acta Vet. Scand. 2017, 59, 71. [Google Scholar] [CrossRef]
- Anfinsen, K.P.; Grotmol, T.; Bruland, O.S.; Jonasdottir, T.J. Breed-specific incidence rates of canine primary bone tumors-a population based survey of dogs in Norway. Can. J. Vet. Res. 2011, 75, 209–215. [Google Scholar] [PubMed]
- Wolke, R.E.; Nielsin, S.W. Site Incidence of Canine Osteosarcoma. J. Small Anim. Pract. 1966, 7, 489–492. [Google Scholar] [CrossRef] [PubMed]
- Straw, R.C.; Withrow, S.J.; Powers, B.E. Management of Canine Appendicular Osteosarcoma. Vet. Clin. N. Am. Small Anim. Pract. 1990, 20, 1141–1161. [Google Scholar] [CrossRef]
- Alexander, J.W.; Patton, C.S. Primary Tumors of the Skeletal System. Vet. Clin. N. Am. Small Anim. Pract. 1983, 13, 181–195. [Google Scholar] [CrossRef]
- Armbrust, L.J.; Biller, D.S.; Bamford, A.; Chun, R.; Garrett, L.D.; Sanderson, M.W. Comparison of three-view thoracic radiography and computed tomography for detection of pulmonary nodules in dogs with neoplasia. J. Am. Vet. Med. Assoc. 2012, 240, 1088–1094. [Google Scholar] [CrossRef]
- Mueller, F.; Fuchs, B.; Kaser-Hotz, B. Comparative biology of human and canine osteosarcoma. Anticancer Res. 2007, 27, 155–164. [Google Scholar]
- MacEwen, E.G.; Kurzman, I.D. Canine osteosarcoma. Amputation and chemoimmunotherapy. Vet. Clin. N. Am.-Small Anim. Pract. 1996, 26, 123–133. [Google Scholar] [CrossRef]
- Thompson, J.P.; Fugent, M.J. Evaluation of survival times after limb amputation, with and without subsequent administration of cisplatin, for treatment of appendicular osteosarcoma in dogs: 30 cases (1979–1990). J. Am. Vet. Med. Assoc. 1992, 200, 531–533. [Google Scholar]
- Shapiro, W.; Fossum, T.W.; Kitchell, B.E.; Couto, C.G.; Theilen, G.H. Use of cisplatin for treatment of appendicular osteosarcoma in dogs. J. Am. Vet. Med. Assoc. 1988, 192, 507–511. [Google Scholar] [PubMed]
- Chun, R.; Garrett, L.D.; Henry, C.; Wall, M.; Smith, A.; Azene, N.M. Toxicity and Efficacy of Cisplatin and Doxorubicin Combination Chemotherapy for the Treatment of Canine Osteosarcoma. J. Am. Anim. Hosp. Assoc. 2005, 41, 382–387. [Google Scholar] [CrossRef]
- Mauldin, G.N.; Matus, R.E.; Withrow, S.J.; Patnaik, A.K. Canine osteosarcoma. Treatment by amputation versus amputation and adjuvant chemotherapy using doxorubicin and cisplatin. J. Vet. Intern. Med. 1988, 2, 177–180. [Google Scholar] [CrossRef] [PubMed]
- Straw, R.C.; Withrow, S.J.; Richter, S.L.; Powers, B.E.; Klein, M.K.; Postorino, N.C.; LaRue, S.M.; Ogilvie, G.K.; Vail, D.M.; Morrison, W.B.; et al. Amputation and Cisplatin for Treatment of Canine Osteosarcoma. J. Vet. Intern. Med. 1991, 5, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Lane, A.; Black, M.; Wyatt, K. Toxicity and efficacy of a novel doxorubicin and carboplatin chemotherapy protocol for the treatment of canine appendicular osteosarcoma following limb amputation. Aust. Vet. J. 2012, 90, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Berg, J.; Weinstein, M.J.; Schelling, S.H.; Rand, W.M. Treatment of dogs with osteosarcoma by administration of cisplatin after amputation or limb-sparing surgery: 22 cases (1987–1990). J. Am. Vet. Med. Assoc. 1992, 200, 2005–2008. [Google Scholar]
- Phillips, B.; Powers, B.E.; Dernell, W.S.; Straw, R.C.; Khanna, C.; Hogge, G.S.; Vail, D.M. Use of Single-Agent Carboplatin as Adjuvant or Neoadjuvant Therapy in Conjunction with Amputation for Appendicular Osteosarcoma in Dogs. J. Am. Anim. Hosp. Assoc. 2009, 45, 33–38. [Google Scholar] [CrossRef]
- Bergman, P.J.; MacEwen, E.G.; Kurzman, I.D.; Henry, C.J.; Hammer, A.S.; Knapp, D.W.; Hale, A.; Kruth, S.A.; Klein, M.K.; Klausner, J.; et al. Amputation and Carboplatin for Treatment of Dogs with Osteosarcoma: 48 Cases (1991 to 1993). J. Vet. Intern. Med. 1996, 10, 76–81. [Google Scholar] [CrossRef]
- Frimberger, A.E.; Chan, C.M.; Moore, A.S. Canine Osteosarcoma Treated by Post-Amputation Sequential Accelerated Doxorubicin and Carboplatin Chemotherapy: 38 Cases. J. Am. Anim. Hosp. Assoc. 2016, 52, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Berg, J.; Weinstein, M.J.; Springfield, D.S.; Rand, W.M. Results of surgery and doxorubicin chemotherapy in dogs with osteosarcoma. J. Am. Vet. Med. Assoc. 1995, 206, 1555–1560. [Google Scholar] [PubMed]
- Spodnick, G.J.; Berg, J.; Rand, W.M.; Schelling, S.H.; Couto, G.; Harvey, H.J.; Henderson, R.A.; MacEwen, G.; Mauldin, N.; McCaw, D.L. Prognosis for dogs with appendicular osteosarcoma treated by amputation alone: 162 cases (1978–1988). J. Am. Vet. Med. Assoc. 1992, 200, 995–999. [Google Scholar] [PubMed]
- Kowal, J.; Arras, G.; Colombo, M.; Jouve, M.; Morath, J.P.; Primdal-Bengtson, B.; Dingli, F.; Loew, D.; Tkach, M.; Théry, C. Proteomic comparison defines novel markers to characterize heterogeneous populations of extracellular vesicle subtypes. Proc. Natl. Acad. Sci. USA 2016, 113, E968–E977. [Google Scholar] [CrossRef] [Green Version]
- Zaborowski, M.P.; Balaj, L.; Breakefield, X.O.; Lai, C.P. Extracellular Vesicles: Composition, Biological Relevance, and Methods of Study. Bioscience 2015, 65, 783–797. [Google Scholar] [CrossRef] [Green Version]
- Yoon, Y.J.; Kim, O.Y.; Gho, Y.S. Extracellular vesicles as emerging intercellular communicasomes. BMB Rep. 2014, 47, 531–539. [Google Scholar] [CrossRef]
- Mathivanan, S.; Ji, H.; Simpson, R.J. Exosomes: Extracellular organelles important in intercellular communication. J. Proteomics 2010, 73, 1907–1920. [Google Scholar] [CrossRef]
- Torreggiani, E.; Roncuzzi, L.; Perut, F.; Zini, N.; Baldini, N. Multimodal transfer of MDR by exosomes in human osteosarcoma. Int. J. Oncol. 2016, 49, 189–196. [Google Scholar] [CrossRef] [Green Version]
- Macklin, R.; Wang, H.; Loo, D.; Martin, S.; Cumming, A.; Cai, N.; Lane, R.; Ponce, N.S.; Topkas, E.; Inder, K.; et al. Extracellular vesicles secreted by highly metastatic clonal variants of osteosarcoma preferentially localize to the lungs and induce metastatic behaviour in poorly metastatic clones. Oncotarget 2016, 7, 43570–43587. [Google Scholar] [CrossRef] [Green Version]
- Wolf-Dennen, K.; Gordon, N.; Kleinerman, E.S. Exosomal communication by metastatic osteosarcoma cells modulates alveolar macrophages to an M2 tumor-promoting phenotype and inhibits tumoricidal functions. Oncoimmunology 2020, 9, 1747677. [Google Scholar] [CrossRef] [Green Version]
- Mazumdar, A.; Urdinez, J.; Boro, A.; Migliavacca, J.; Arlt, M.J.E.; Muff, R.; Fuchs, B.; Snedeker, J.G.; Gvozdenovic, A. Osteosarcoma-Derived Extracellular Vesicles Induce Lung Fibroblast Reprogramming. Int. J. Mol. Sci. 2020, 21, 5451. [Google Scholar] [CrossRef]
- Bandu, R.; Oh, J.W.; Kim, K.P. Mass spectrometry-based proteome profiling of extracellular vesicles and their roles in cancer biology. Exp. Mol. Med. 2019, 51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hurwitz, S.N.; Rider, M.A.; Bundy, J.L.; Liu, X.; Singh, R.K.; Meckes, D.G. Proteomic profiling of NCI-60 extracellular vesicles uncovers common protein cargo and cancer type-specific biomarkers. Oncotarget 2016, 7, 86999–87015. [Google Scholar] [CrossRef] [PubMed]
- Rontogianni, S.; Synadaki, E.; Li, B.; Liefaard, M.C.; Lips, E.H.; Wesseling, J.; Wu, W.; Altelaar, M. Proteomic profiling of extracellular vesicles allows for human breast cancer subtyping. Commun. Biol. 2019, 2, 325. [Google Scholar] [CrossRef] [Green Version]
- Hoshino, A.; Kim, H.S.; Bojmar, L.; Gyan, K.E.; Cioffi, M.; Hernandez, J.; Zambirinis, C.P.; Rodrigues, G.; Molina, H.; Heissel, S.; et al. Extracellular Vesicle and Particle Biomarkers Define Multiple Human Cancers. Cell 2020, 182, 1044–1061.e18. [Google Scholar] [CrossRef]
- Gu, Z.C.; Enenkel, C. Proteasome assembly. Cell. Mol. Life Sci. 2014, 71, 4729–4745. [Google Scholar] [CrossRef]
- Tanaka, K. The proteasome: Overview of structure and functions. Proc. Jpn. Acad. Ser. B 2009, 85, 12–36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Verma, R.; Aravind, L.; Oania, R.; McDonald, W.H.; Yates, J.R.; Koonin, E.V.; Deshaies, R.J. Role of Rpn11 Metalloprotease in Deubiquitination and Degradation by the 26 S Proteasome. Science 2002, 298, 611–615. [Google Scholar] [CrossRef]
- Patatsos, K.; Shekhar, T.M.; Hawkins, C.J. Pre-clinical evaluation of proteasome inhibitors for canine and human osteosarcoma. Vet. Comp. Oncol. 2018, 16, 544–553. [Google Scholar] [CrossRef] [PubMed]
- Harris, M.A.; Miles, M.A.; Shekhar, T.M.; Cerra, C.; Georgy, S.R.; Ryan, S.D.; Cannon, C.M.; Hawkins, C.J. The Proteasome Inhibitor Ixazomib Inhibits the Formation and Growth of Pulmonary and Abdominal Osteosarcoma Metastases in Mice. Cancers 2020, 12, 1207. [Google Scholar] [CrossRef]
- Somarelli, J.A.; Rupprecht, G.; Altunel, E.; Flamant, E.M.; Rao, S.; Sivaraj, D.; Lazarides, A.L.; Hoskinson, S.M.; Sheth, M.U.; Cheng, S.; et al. A Comparative Oncology Drug Discovery Pipeline to Identify and Validate New Treatments for Osteosarcoma. Cancers 2020, 12, 3335. [Google Scholar] [CrossRef]
- Li, J.; Yakushi, T.; Parlati, F.; Mackinnon, A.L.; Perez, C.; Ma, Y.; Carter, K.P.; Colayco, S.; Magnuson, G.; Brown, B.; et al. Capzimin is a potent and specific inhibitor of proteasome isopeptidase Rpn11. Nat. Chem. Biol. 2017, 13, 486–493. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rastogi, N.; Mishra, D.P. Therapeutic targeting of cancer cell cycle using proteasome inhibitors. Cell Div. 2012, 7, 26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jing, C.; Li, X.; Zhou, M.; Zhang, S.; Lai, Q.; Liu, D.; Ye, B.; Li, L.; Wu, Y.; Li, H.; et al. The PSMD14 inhibitor Thiolutin as a novel therapeutic approach for esophageal squamous cell carcinoma through facilitating SNAIL degradation. Theranostics 2021, 11, 5847–5862. [Google Scholar] [CrossRef]
- Sun, F.; Zhang, Y.; Xu, L.; Li, S.; Chen, X.; Zhang, L.; Wu, Y.; Li, J. Proteasome Inhibitor MG132 Enhances Cisplatin-Induced Apoptosis in Osteosarcoma Cells and Inhibits Tumor Growth. Oncol. Res. Featur. Preclin. Clin. Cancer Ther. 2018, 26, 655–664. [Google Scholar] [CrossRef]
- Liu, Y.; Raina, D.B.; Sebastian, S.; Nagesh, H.; Isaksson, H.; Engellau, J.; Lidgren, L.; Tägil, M. Sustained and controlled delivery of doxorubicin from an in-situ setting biphasic hydroxyapatite carrier for local treatment of a highly proliferative human osteosarcoma. Acta Biomater. 2021, 131, 555–571. [Google Scholar] [CrossRef] [PubMed]
- Boston, S.E.; Ehrhart, N.P.; Dernell, W.S.; Lafferty, M.; Withrow, S.J. Evaluation of survival time in dogs with stage III osteosarcoma that undergo treatment: 90 cases (1985–2004). J. Am. Vet. Med. Assoc. 2006, 228, 1905–1908. [Google Scholar] [CrossRef]
- Schott, C.R.; Ludwig, L.; Mutsaers, A.J.; Foster, R.A.; Wood, G.A. The autophagy inhibitor spautin-1, either alone or combined with doxorubicin, decreases cell survival and colony formation in canine appendicular osteosarcoma cells. PLoS ONE 2018, 13, e0206427. [Google Scholar] [CrossRef]
- LeBlanc, A.K.; Mazcko, C.N. Improving human cancer therapy through the evaluation of pet dogs. Nat. Rev. Cancer 2020, 20, 727–742. [Google Scholar] [CrossRef]
- Koons, N.; Amato, N.; Sauer, S.; Warshawsky, D.; Barkan, D.; Khanna, C. Assessing a Novel 3D Assay System for Drug Screening against OS Metastasis. Pharmaceuticals 2021, 14, 971. [Google Scholar] [CrossRef] [PubMed]
- Bhat, M.; Robichaud, N.; Hulea, L.; Sonenberg, N.; Pelletier, J.; Topisirovic, I. Targeting the translation machinery in cancer. Nat. Rev. Drug Discov. 2015, 14, 261–278. [Google Scholar] [CrossRef] [PubMed]
- Morrow, J.J.; Mendoza, A.; Koyen, A.; Lizardo, M.M.; Ren, L.; Waybright, T.J.; Hansen, R.J.; Gustafson, D.L.; Zhou, M.; Fan, T.M.; et al. mTOR Inhibition Mitigates Enhanced mRNA Translation Associated with the Metastatic Phenotype of Osteosarcoma Cells In Vivo. Clin. Cancer Res. 2016, 22, 6129–6141. [Google Scholar] [CrossRef] [Green Version]
- Osborne, T.S.; Khanna, C. A Review of the Association between Osteosarcoma Metastasis and Protein Translation. J. Comp. Pathol. 2012, 146, 132–142. [Google Scholar] [CrossRef] [Green Version]
- Moriarity, B.S.; Otto, G.M.; Rahrmann, E.P.; Rathe, S.K.; Wolf, N.K.; Weg, M.T.; Manlove, L.A.; LaRue, R.S.; Temiz, N.A.; Molyneux, S.D.; et al. A Sleeping Beauty forward genetic screen identifies new genes and pathways driving osteosarcoma development and metastasis. Nat. Genet. 2015, 47, 615–624. [Google Scholar] [CrossRef]
- LeBlanc, A.K.; Mazcko, C.N.; Cherukuri, A.; Berger, E.P.; Kisseberth, W.C.; Brown, M.E.; Lana, S.E.; Weishaar, K.; Flesner, B.K.; Bryan, J.N.; et al. Adjuvant Sirolimus Does Not Improve Outcome in Pet Dogs Receiving Standard-of-Care Therapy for Appendicular Osteosarcoma: A Prospective, Randomized Trial of 324 Dogs. Clin. Cancer Res. 2021, 27, 3005–3016. [Google Scholar] [CrossRef]
- Nunes, A.T.; Annunziata, C.M. Proteasome inhibitors: Structure and function. Semin. Oncol. 2017, 44, 377–380. [Google Scholar] [CrossRef]
- Deshaies, R.J. Proteotoxic crisis, the ubiquitin-proteasome system, and cancer therapy. BMC Biol. 2014, 12, 94. [Google Scholar] [CrossRef]
- Adams, J. The proteasome: A suitable antineoplastic target. Nat. Rev. Cancer 2004, 4, 349–360. [Google Scholar] [CrossRef]
- Kubiczkova, L.; Pour, L.; Sedlarikova, L.; Hajek, R.; Sevcikova, S. Proteasome inhibitors-molecular basis and current perspectives in multiple myeloma. J. Cell. Mol. Med. 2014, 18, 947–961. [Google Scholar] [CrossRef]
- Park, J.E.; Miller, Z.; Jun, Y.; Lee, W.; Kim, K.B. Next-generation proteasome inhibitors for cancer therapy. Transl. Res. 2018, 198, 1–16. [Google Scholar] [CrossRef]
- Gupta, N.; Hanley, M.J.; Venkatakrishnan, K.; Perez, R.; Norris, R.E.; Nemunaitis, J.; Yang, H.; Qian, M.G.; Falchook, G.; Labotka, R.; et al. Pharmacokinetics of ixazomib, an oral proteasome inhibitor, in solid tumour patients with moderate or severe hepatic impairment. Br. J. Clin. Pharmacol. 2016, 82, 728–738. [Google Scholar] [CrossRef] [Green Version]
- Huang, Z.; Wu, Y.; Zhou, X.; Xu, J.; Zhu, W.; Shu, Y.; Liu, P. Efficacy of therapy with bortezomib in solid tumors: A review based on 32 clinical trials. Futur. Oncol. 2014, 10, 1795–1807. [Google Scholar] [CrossRef]
- Ge, M.; Qiao, Z.; Kong, Y.; Liang, H.; Sun, Y.; Lu, H.; Xu, Z.; Liu, H. Modulating proteasome inhibitor tolerance in multiple myeloma: An alternative strategy to reverse inevitable resistance. Br. J. Cancer 2021, 124, 770–776. [Google Scholar] [CrossRef]
- Lou, Z.; Ren, T.; Peng, X.; Sun, Y.; Jiao, G.; Lu, Q.; Zhang, S.; Lu, X.; Guo, W. Bortezomib induces apoptosis and autophagy in osteosarcoma cells through mitogen-activated protein kinase pathway in vitro. J. Int. Med. Res. 2013, 41, 1505–1519. [Google Scholar] [CrossRef] [Green Version]
- Meiners, S.; Heyken, D.; Weller, A.; Ludwig, A.; Stangl, K.; Kloetzel, P.-M.; Krüger, E. Inhibition of Proteasome Activity Induces Concerted Expression of Proteasome Genes and de Novo Formation of Mammalian Proteasomes. J. Biol. Chem. 2003, 278, 21517–21525. [Google Scholar] [CrossRef] [Green Version]
- Bernardi, R.; Liebermann, D.A.; Hoffman, B. Cdc25A stability is controlled by the ubiquitin-proteasome pathway during cell cycle progression and terminal differentiation. Oncogene 2000, 19, 2447–2454. [Google Scholar] [CrossRef] [Green Version]
- Huang, Q.; Li, X.; Huang, Z.; Yu, F.; Wang, X.; Wang, S.; He, Z.; Lin, J. ACTN4 Promotes the Proliferation, Migration, Metastasis of Osteosarcoma and Enhances its Invasive Ability through the NF-κB Pathway. Pathol. Oncol. Res. 2020, 26, 893–904. [Google Scholar] [CrossRef] [Green Version]
- Russo, A.; Fratto, M.E.; Bazan, V.; Schiró, V.; Agnese, V.; Cicero, G.; Vincenzi, B.; Tonini, G.; Santini, D. Targeting apoptosis in solid tumors: The role of bortezomib from preclinical to clinical evidence. Expert Opin. Ther. Targets 2007, 11, 1571–1586. [Google Scholar] [CrossRef]
- Traenckner, E.B.; Wilk, S.; Baeuerle, P.A. A proteasome inhibitor prevents activation of NF-kappa B and stabilizes a newly phosphorylated form of I kappa B-alpha that is still bound to NF-kappa B. EMBO J. 1994, 13, 5433–5441. [Google Scholar] [CrossRef]
- Zhang, W.; Zhao, J.-M.; Lin, J.; Hu, C.-Z.; Zhang, W.-B.; Yang, W.-L.; Zhang, J.; Zhang, J.-W.; Zhu, J. Adaptive Fibrogenic Reprogramming of Osteosarcoma Stem Cells Promotes Metastatic Growth. Cell Rep. 2018, 24, 1266–1277.e5. [Google Scholar] [CrossRef] [Green Version]
- Baritaki, S.; Chapman, A.; Yeung, K.; Spandidos, D.A.; Palladino, M.; Bonavida, B. Inhibition of epithelial to mesenchymal transition in metastatic prostate cancer cells by the novel proteasome inhibitor, NPI-0052: Pivotal roles of Snail repression and RKIP induction. Oncogene 2009, 28, 3573–3585. [Google Scholar] [CrossRef] [Green Version]
- Yu, D.; Kahen, E.; Cubitt, C.L.; McGuire, J.; Kreahling, J.; Lee, J.; Altiok, S.; Lynch, C.C.; Sullivan, D.M.; Reed, D.R. Identification of Synergistic, Clinically Achievable, Combination Therapies for Osteosarcoma. Sci. Rep. 2015, 5, 16991. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Butler, L.R.; Densham, R.M.; Jia, J.; Garvin, A.J.; Stone, H.R.; Shah, V.; Weekes, D.; Festy, F.; Beesley, J.; Morris, J.R. The proteasomal de-ubiquitinating enzyme POH1 promotes the double-strand DNA break response. EMBO J. 2012, 31, 3918–3934. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Kemp, C.J.; Henikoff, S. Anthracyclines induce double-strand DNA breaks at active gene promoters. Mutat. Res. Mol. Mech. Mutagen. 2015, 773, 9–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ogiso, Y.; Tomida, A.; Lei, S.; Omura, S.; Tsuruo, T. Proteasome inhibition circumvents solid tumor resistance to topoisomerase II-directed drugs. Cancer Res. 2000, 60, 2429–2434. [Google Scholar]
- Nitiss, J.L. DNA topoisomerase II and its growing repertoire of biological functions. Nat. Rev. Cancer 2009, 9, 327–337. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marinello, J.; Delcuratolo, M.; Capranico, G. Anthracyclines as Topoisomerase II Poisons: From Early Studies to New Perspectives. Int. J. Mol. Sci. 2018, 19, 3480. [Google Scholar] [CrossRef] [Green Version]
- Froelich-Ammon, S.J.; Osheroff, N. Topoisomerase Poisons: Harnessing the Dark Side of Enzyme Mechanism. J. Biol. Chem. 1995, 270, 21429–21432. [Google Scholar] [CrossRef] [Green Version]
- Nitiss, J.L. Targeting DNA topoisomerase II in cancer chemotherapy. Nat. Rev. Cancer 2009, 9, 338–350. [Google Scholar] [CrossRef] [Green Version]
- Keerthikumar, S.; Gangoda, L.; Liem, M.; Fonseka, P.; Atukorala, I.; Ozcitti, C.; Mechler, A.; Adda, C.G.; Ang, C.-S.; Mathivanan, S. Proteogenomic analysis reveals exosomes are more oncogenic than ectosomes. Oncotarget 2015, 6, 15375–15396. [Google Scholar] [CrossRef] [Green Version]
- Chaiyawat, P.; Sungngam, P.; Teeyakasem, P.; Sirikaew, N.; Klangjorhor, J.; Settakorn, J.; Diskul-Na-Ayudthaya, P.; Chokchaichamnankit, D.; Srisomsap, C.; Svasti, J.; et al. Protein profiling of osteosarcoma tissue and soft callus unveils activation of the unfolded protein response pathway. Int. J. Oncol. 2019, 54, 1704–1718. [Google Scholar] [CrossRef] [Green Version]
- Nagao-Kitamoto, H.; Setoguchi, T.; Kitamoto, S.; Nakamura, S.; Tsuru, A.; Nagata, M.; Nagano, S.; Ishidou, Y.; Yokouchi, M.; Kitajima, S.; et al. Ribosomal protein S3 regulates GLI2-mediated osteosarcoma invasion. Cancer Lett. 2015, 356, 855–861. [Google Scholar] [CrossRef] [PubMed]
- Cheng, D.-D.; Zhu, B.; Li, S.-J.; Yuan, T.; Yang, Q.-C.; Fan, C.-Y. Down-regulation of RPS9 Inhibits Osteosarcoma Cell Growth through Inactivation of MAPK Signaling Pathway. J. Cancer 2017, 8, 2720–2728. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Z.; Huang, S.; Luo, Q.; Zhang, Z.; Long, X. Inhibiting valosin-containing protein suppresses osteosarcoma cell metastasis via AKT/nuclear factor of kappa B signaling pathway in vitro. Indian J. Pathol. Microbiol. 2013, 56, 190. [Google Scholar] [CrossRef]
- Wu, G.; Zhang, M. Identification of Biomarkers and Study of Mechanisms Related to Metastatic of Osteosarcoma Based on Integrated Bioinformatics Analyses Methods. Res. Sq. 2020, 1–13. [Google Scholar] [CrossRef]
- Rathore, R.; Caldwell, K.E.; Schutt, C.; Brashears, C.B.; Prudner, B.C.; Ehrhardt, W.R.; Leung, C.H.; Lin, H.; Daw, N.C.; Beird, H.C.; et al. Metabolic compensation activates pro-survival mTORC1 signaling upon 3-phosphoglycerate dehydrogenase inhibition in osteosarcoma. Cell Rep. 2021, 34, 108678. [Google Scholar] [CrossRef]
- Xu, C.; Song, J.; Su, Y.; Liu, X. Expression of b-FGF and endostatin and their clinical significance in human osteosarcoma. Orthop. Surg. 2010, 2, 291–298. [Google Scholar] [CrossRef]
- Shapovalov, Y.; Benavidez, D.; Zuch, D.; Eliseev, R.A. Proteasome inhibition with bortezomib suppresses growth and induces apoptosis in osteosarcoma. Int. J. Cancer 2010, 127, 67–76. [Google Scholar] [CrossRef]
- Xian, M.; Cao, H.; Cao, J.; Shao, X.; Zhu, D.; Zhang, N.; Huang, P.; Li, W.; Yang, B.; Ying, M.; et al. Bortezomib sensitizes human osteosarcoma cells to adriamycin-induced apoptosis through ROS-dependent activation of p-eIF2α/ATF4/CHOP axis. Int. J. Cancer 2017, 141, 1029–1041. [Google Scholar] [CrossRef]
- McGuire, J.J.; Nerlakanti, N.; Lo, C.H.; Tauro, M.; Utset-Ward, T.J.; Reed, D.R.; Lynch, C.C. Histone deacetylase inhibition prevents the growth of primary and metastatic osteosarcoma. Int. J. Cancer 2020, 147, 2811–2823. [Google Scholar] [CrossRef]
- Song, Y.; Li, S.; Ray, A.; Das, D.S.; Qi, J.; Samur, M.K.; Tai, Y.-T.; Munshi, N.; Carrasco, R.D.; Chauhan, D.; et al. Blockade of deubiquitylating enzyme Rpn11 triggers apoptosis in multiple myeloma cells and overcomes bortezomib resistance. Oncogene 2017, 36, 5631–5638. [Google Scholar] [CrossRef] [Green Version]
- Johnsen, K.B.; Gudbergsson, J.M.; Andresen, T.L.; Simonsen, J.B. What is the blood concentration of extracellular vesicles? Implications for the use of extracellular vesicles as blood-borne biomarkers of cancer. Biochim. Biophys. Acta-Rev. Cancer 2019, 1871, 109–116. [Google Scholar] [CrossRef] [Green Version]
- Poulet, G.; Massias, J.; Taly, V. Liquid Biopsy: General Concepts. Acta Cytol. 2019, 63, 449–455. [Google Scholar] [CrossRef] [PubMed]
- Brady, J.V.; Troyer, R.M.; Ramsey, S.A.; Leeper, H.; Yang, L.; Maier, C.S.; Goodall, C.P.; Ruby, C.E.; Albarqi, H.A.M.; Taratula, O.; et al. A Preliminary Proteomic Investigation of Circulating Exosomes and Discovery of Biomarkers Associated with the Progression of Osteosarcoma in a Clinical Model of Spontaneous Disease. Transl. Oncol. 2018, 11, 1137–1146. [Google Scholar] [CrossRef] [PubMed]
- Weinman, M.A.; Ramsey, S.A.; Leeper, H.J.; Brady, J.V.; Schlueter, A.; Stanisheuski, S.; Maier, C.S.; Miller, T.; Ruby, C.E.; Bracha, S. Exosomal proteomic signatures correlate with drug resistance and carboplatin treatment outcome in a spontaneous model of canine osteosarcoma. Cancer Cell Int. 2021, 21, 245. [Google Scholar] [CrossRef]
- Luu, A.K.; Wood, G.A.; Viloria-Petit, A.M. Recent Advances in the Discovery of Biomarkers for Canine Osteosarcoma. Front. Vet. Sci. 2021, 8, 734965. [Google Scholar] [CrossRef] [PubMed]
- Kornilov, R.; Puhka, M.; Mannerström, B.; Hiidenmaa, H.; Peltoniemi, H.; Siljander, P.; Seppänen-Kaijansinkko, R.; Kaur, S. Efficient ultrafiltration-based protocol to deplete extracellular vesicles from fetal bovine serum. J. Extracell. Vesicles 2018, 7, 1422674. [Google Scholar] [CrossRef] [Green Version]






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luu, A.K.; Cadieux, M.; Wong, M.; Macdonald, R.; Jones, R.; Choi, D.; Oblak, M.; Brisson, B.; Sauer, S.; Chafitz, J.; et al. Proteomic Assessment of Extracellular Vesicles from Canine Tissue Explants as a Pipeline to Identify Molecular Targets in Osteosarcoma: PSMD14/Rpn11 as a Proof of Principle. Int. J. Mol. Sci. 2022, 23, 3256. https://doi.org/10.3390/ijms23063256
Luu AK, Cadieux M, Wong M, Macdonald R, Jones R, Choi D, Oblak M, Brisson B, Sauer S, Chafitz J, et al. Proteomic Assessment of Extracellular Vesicles from Canine Tissue Explants as a Pipeline to Identify Molecular Targets in Osteosarcoma: PSMD14/Rpn11 as a Proof of Principle. International Journal of Molecular Sciences. 2022; 23(6):3256. https://doi.org/10.3390/ijms23063256
Chicago/Turabian StyleLuu, Anita K., Mia Cadieux, Mackenzie Wong, Rachel Macdonald, Robert Jones, Dongsic Choi, Michelle Oblak, Brigitte Brisson, Scott Sauer, James Chafitz, and et al. 2022. "Proteomic Assessment of Extracellular Vesicles from Canine Tissue Explants as a Pipeline to Identify Molecular Targets in Osteosarcoma: PSMD14/Rpn11 as a Proof of Principle" International Journal of Molecular Sciences 23, no. 6: 3256. https://doi.org/10.3390/ijms23063256





