Brain Cholesterol Biosynthetic Pathway Is Altered in a Preclinical Model of Fragile X Syndrome
Abstract
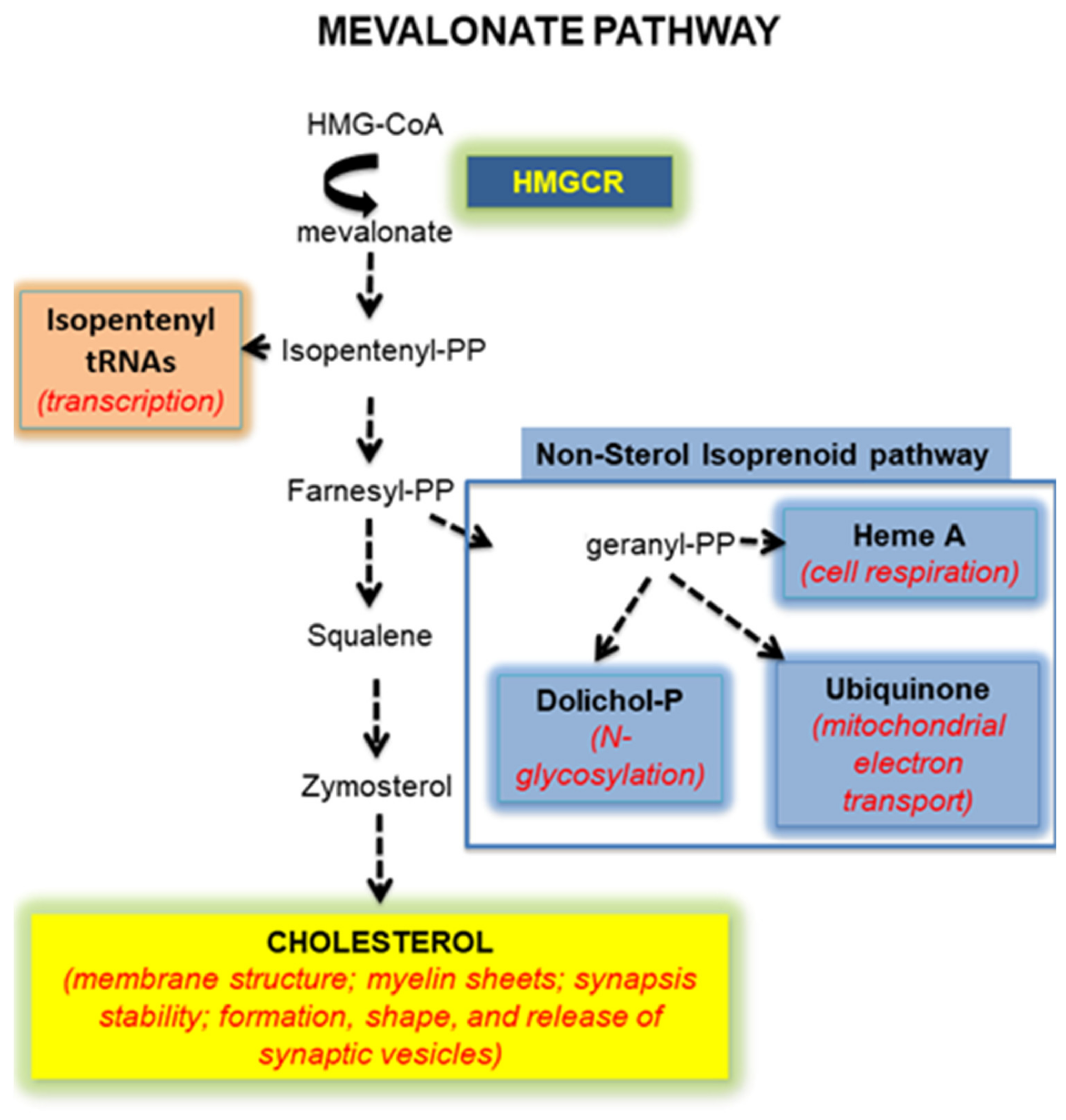
:1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Animals and Sample Collection
4.2. Measurement of Cholesterol in Brain, Liver and Serum Samples
4.3. Total Lysate and Membranes Preparation for Western Blot Analysis
4.4. Immunoblotting
4.5. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Musetti, A.; Manari, T.; Dioni, B.; Raffin, C.; Bravo, G.; Mariani, R.; Esposito, G.; Dimitriou, D.; Plazzi, G.; Franceschini, C.; et al. Parental Quality of Life and Involvement in Intervention for Children or Adolescents with Autism Spectrum Disorders: A Systematic Review. J. Pers. Med. 2021, 11, 894. [Google Scholar] [CrossRef] [PubMed]
- Prieto, M.; Folci, A.; Poupon, G.; Schiavi, S.; Buzzelli, V.; Pronot, M.; François, U.; Pousinha, P.; Lattuada, N.; Abelanet, S.; et al. Missense Mutation of Fmr1 Results in Impaired AMPAR-Mediated Plasticity and Socio-Cognitive Deficits in Mice. Nat. Commun. 2021, 12, 1557. [Google Scholar] [CrossRef] [PubMed]
- Santoro, M.R.; Bray, S.M.; Warren, S.T. Molecular Mechanisms of Fragile X Syndrome: A Twenty-Year Perspective. Annu. Rev. Pathol. 2012, 7, 219–245. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maurin, T.; Zongaro, S.; Bardoni, B. Fragile X Syndrome: From Molecular Pathology to Therapy. Neurosci. Biobehav. Rev. 2014, 46, 242–255. [Google Scholar] [CrossRef]
- Penagarikano, O.; Mulle, J.G.; Warren, S.T. The Pathophysiology of Fragile x Syndrome. Annu. Rev. Genomics Hum. Genet. 2007, 8, 109–129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalinowska, M.; Castillo, C.; Francesconi, A. Quantitative Profiling of Brain Lipid Raft Proteome in a Mouse Model of Fragile X Syndrome. PLoS ONE 2015, 10, e0121464. [Google Scholar] [CrossRef] [PubMed]
- Prieto, M.; Folci, A.; Martin, S. Post-Translational Modifications of the Fragile X Mental Retardation Protein in Neuronal Function and Dysfunction. Mol. Psychiatry 2020, 25, 1688–1703. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.; Kim, E.; Datta, D.; Lewis, D.A.; Soderling, S.H. Synaptic Actin Dysregulation, a Convergent Mechanism of Mental Disorders? J. Neurosci. 2016, 36, 11411–11417. [Google Scholar] [CrossRef] [Green Version]
- Threadgill, R.; Bobb, K.; Ghosh, A. Regulation of Dendritic Growth and Remodeling by Rho, Rac, and Cdc42. Neuron 1997, 19, 625–634. [Google Scholar] [CrossRef] [Green Version]
- Cartocci, V.; Segatto, M.; Di Tunno, I.; Leone, S.; Pfrieger, F.W.; Pallottini, V. Modulation of the Isoprenoid/Cholesterol Biosynthetic Pathway During Neuronal Differentiation In Vitro: MVA Pathway In Neuronal Differentiation. J. Cell. Biochem. 2016, 117, 2036–2044. [Google Scholar] [CrossRef] [PubMed]
- Bourtchuladze, R.; Frenguelli, B.; Blendy, J.; Cioffi, D.; Schutz, G.; Silva, A.J. Deficient Long-Term Memory in Mice with a Targeted Mutation of the CAMP-Responsive Element-Binding Protein. Cell 1994, 79, 59–68. [Google Scholar] [CrossRef]
- Kandel, E.R. The Molecular Biology of Memory: CAMP, PKA, CRE, CREB-1, CREB-2, and CPEB. Mol. Brain 2012, 5, 14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carlezon, W.A.; Duman, R.S.; Nestler, E.J. The Many Faces of CREB. Trends Neurosci. 2005, 28, 436–445. [Google Scholar] [CrossRef] [PubMed]
- Segatto, M.; Manduca, A.; Lecis, C.; Rosso, P.; Jozwiak, A.; Swiezewska, E.; Moreno, S.; Trezza, V.; Pallottini, V. Simvastatin Treatment Highlights a New Role for the Isoprenoid/Cholesterol Biosynthetic Pathway in the Modulation of Emotional Reactivity and Cognitive Performance in Rats. Neuropsychopharmacology 2014, 39, 841–854. [Google Scholar] [CrossRef] [Green Version]
- Tonini, C.; Schiavi, S.; Macca, F.; Segatto, M.; Trezza, V.; Pallottini, V. Long-Lasting Impact of Perinatal Dietary Supplementation of Omega 3 Fatty Acids on Mevalonate Pathway: Potential Role on Neuron Trophism in Male Offspring Hippocampal Formation. Nutr. Neurosci. 2020, 25, 110–121. [Google Scholar] [CrossRef]
- Reddy, J.M.; Raut, N.G.R.; Seifert, J.L.; Hynds, D.L. Regulation of Small GTPase Prenylation in the Nervous System. Mol. Neurobiol. 2020, 57, 2220–2231. [Google Scholar] [CrossRef] [PubMed]
- Gendaszewska-Darmach, E.; Garstka, M.A.; Błażewska, K.M. Targeting Small GTPases and Their Prenylation in Diabetes Mellitus. J. Med. Chem. 2021, 64, 9677–9710. [Google Scholar] [CrossRef]
- Segatto, M.; Di Giovanni, A.; Marino, M.; Pallottini, V. Analysis of the Protein Network of Cholesterol Homeostasis in Different Brain Regions: An Age and Sex Dependent Perspective. J. Cell. Physiol. 2013, 228, 1561–1567. [Google Scholar] [CrossRef]
- Espenshade, P.J.; Hughes, A.L. Regulation of Sterol Synthesis in Eukaryotes. Annu. Rev. Genet. 2007, 41, 401–427. [Google Scholar] [CrossRef]
- Ikonen, E. Mechanisms for Cellular Cholesterol Transport: Defects and Human Disease. Physiol. Rev. 2006, 86, 1237–1261. [Google Scholar] [CrossRef]
- Segatto, M.; Leboffe, L.; Trapani, L.; Pallottini, V. Cholesterol Homeostasis Failure in the Brain: Implications for Synaptic Dysfunction and Cognitive Decline. Curr. Med. Chem. 2014, 21, 2788–2802. [Google Scholar] [CrossRef]
- Cartocci, V.; Catallo, M.; Tempestilli, M.; Segatto, M.; Pfrieger, F.W.; Bronzuoli, M.R.; Scuderi, C.; Servadio, M.; Trezza, V.; Pallottini, V. Altered Brain Cholesterol/Isoprenoid Metabolism in a Rat Model of Autism Spectrum Disorders. Neuroscience 2018, 372, 27–37. [Google Scholar] [CrossRef]
- Cartocci, V.; Servadio, M.; Trezza, V.; Pallottini, V. Can Cholesterol Metabolism Modulation Affect Brain Function and Behavior? J. Cell. Physiol. 2017, 232, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Segatto, M.; Tonini, C.; Pfrieger, F.W.; Trezza, V.; Pallottini, V. Loss of Mevalonate/Cholesterol Homeostasis in the Brain: A Focus on Autism Spectrum Disorder and Rett Syndrome. Int. J. Mol. Sci. 2019, 20, 3317. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muscas, M.; Louros, S.R.; Osterweil, E.K. Lovastatin, Not Simvastatin, Corrects Core Phenotypes in the Fragile X Mouse Model. eNeuro 2019, 6, 2019. [Google Scholar] [CrossRef] [Green Version]
- Lisik, M.Z.; Gutmajster, E.; Sieroń, A.L. Low Levels of HDL in Fragile X Syndrome Patients. Lipids 2016, 51, 189–192. [Google Scholar] [CrossRef] [PubMed]
- Çaku, A.; Seidah, N.G.; Lortie, A.; Gagné, N.; Perron, P.; Dubé, J.; Corbin, F. New Insights of Altered Lipid Profile in Fragile X Syndrome. PLoS ONE 2017, 12, e0174301. [Google Scholar] [CrossRef] [Green Version]
- Schiavi, S.; Carbone, E.; Melancia, F.; Buzzelli, V.; Manduca, A.; Campolongo, P.; Pallottini, V.; Trezza, V. Perinatal Supplementation with Omega-3 Fatty Acids Corrects the Aberrant Social and Cognitive Traits Observed in a Genetic Model of Autism Based on FMR1 Deletion in Rats. Nutr. Neurosci. 2020, 11, 1–14. [Google Scholar] [CrossRef]
- Verkerk, A.J.; Pieretti, M.; Sutcliffe, J.S.; Fu, Y.H.; Kuhl, D.P.; Pizzuti, A.; Reiner, O.; Richards, S.; Victoria, M.F.; Zhang, F.P. Identification of a Gene (FMR-1) Containing a CGG Repeat Coincident with a Breakpoint Cluster Region Exhibiting Length Variation in Fragile X Syndrome. Cell 1991, 65, 905–914. [Google Scholar] [CrossRef]
- Oberlé, I.; Rousseau, F.; Heitz, D.; Kretz, C.; Devys, D.; Hanauer, A.; Boué, J.; Bertheas, M.F.; Mandel, J.L. Instability of a 550-Base Pair DNA Segment and Abnormal Methylation in Fragile X Syndrome. Science 1991, 252, 1097–1102. [Google Scholar] [CrossRef]
- Melancia, F.; Trezza, V. Modelling Fragile X Syndrome in the Laboratory Setting: A Behavioral Perspective. Behav. Brain Res. 2018, 350, 149–163. [Google Scholar] [CrossRef]
- Hamilton, S.M.; Green, J.R.; Veeraragavan, S.; Yuva, L.; McCoy, A.; Wu, Y.; Warren, J.; Little, L.; Ji, D.; Cui, X.; et al. Fmr1 and Nlgn3 Knockout Rats: Novel Tools for Investigating Autism Spectrum Disorders. Behav. Neurosci. 2014, 128, 103–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kenkel, W.M.; Yee, J.R.; Moore, K.; Madularu, D.; Kulkarni, P.; Gamber, K.; Nedelman, M.; Ferris, C.F. Functional Magnetic Resonance Imaging in Awake Transgenic Fragile X Rats: Evidence of Dysregulation in Reward Processing in the Mesolimbic/Habenular Neural Circuit. Transl. Psychiatry 2016, 6, e763. [Google Scholar] [CrossRef]
- Golden, C.E.M.; Breen, M.S.; Koro, L.; Sonar, S.; Niblo, K.; Browne, A.; Burlant, N.; Di Marino, D.; De Rubeis, S.; Baxter, M.G.; et al. Deletion of the KH1 Domain of Fmr1 Leads to Transcriptional Alterations and Attentional Deficits in Rats. Cereb. Cortex 2019, 29, 2228–2244. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Till, S.M.; Asiminas, A.; Jackson, A.D.; Katsanevaki, D.; Barnes, S.A.; Osterweil, E.K.; Bear, M.F.; Chattarji, S.; Wood, E.R.; Wyllie, D.J.A.; et al. Conserved Hippocampal Cellular Pathophysiology but Distinct Behavioural Deficits in a New Rat Model of FXS. Hum. Mol. Genet. 2015, 24, 5977–5984. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tian, Y.; Yang, C.; Shang, S.; Cai, Y.; Deng, X.; Zhang, J.; Shao, F.; Zhu, D.; Liu, Y.; Chen, G.; et al. Loss of FMRP Impaired Hippocampal Long-Term Plasticity and Spatial Learning in Rats. Front. Mol. Neurosci. 2017, 10, 269. [Google Scholar] [CrossRef] [Green Version]
- Trapani, L. Regulation and Deregulation of Cholesterol Homeostasis: The Liver as a Metabolic "Power Station". World J. Hepatol. 2012, 4, 184. [Google Scholar] [CrossRef]
- Dichter, G.S.; Felder, J.N.; Green, S.R.; Rittenberg, A.M.; Sasson, N.J.; Bodfish, J.W. Reward Circuitry Function in Autism Spectrum Disorders. Soc. Cogn. Affect. Neurosci. 2012, 7, 160–172. [Google Scholar] [CrossRef] [Green Version]
- Wu, H.; Wang, X.; Gao, J.; Liang, S.; Hao, Y.; Sun, C.; Xia, W.; Cao, Y.; Wu, L. Fingolimod (FTY720) Attenuates Social Deficits, Learning and Memory Impairments, Neuronal Loss and Neuroinflammation in the Rat Model of Autism. Life Sci. 2017, 173, 43–54. [Google Scholar] [CrossRef]
- Mast, N.; White, M.A.; Bjorkhem, I.; Johnson, E.F.; Stout, C.D.; Pikuleva, I.A. Crystal Structures of Substrate-Bound and Substrate-Free Cytochrome P450 46A1, the Principal Cholesterol Hydroxylase in the Brain. Proc. Natl. Acad. Sci. USA 2008, 105, 9546–9551. [Google Scholar] [CrossRef] [Green Version]
- Zhang, C.; Jin, D.-D.; Wang, X.-Y.; Lou, L.; Yang, J. Key Enzymes for the Mevalonate Pathway in the Cardiovascular System. J. Cardiovasc. Pharmacol. 2021, 77, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Martini, C.; Pallottini, V. Cholesterol: From Feeding to Gene Regulation. Genes Nutr. 2007, 2, 181–193. [Google Scholar] [CrossRef] [Green Version]
- Long, J.-Y.; Jiang, W.; Xia, H.-B.; Fu, J.-Y.; Lu, P.; Hu, F.; Feng, W.-C.; Sun, W.-W.; Gao, M.-M.; Yi, Y.-H.; et al. FMRP-Absence-Induced up-Regulation of Hypothalamic MAP1B Expression Decreases AgRP Level Linking with Reduces in Food Intake and Body Weight. Neurochem. Int. 2020, 140, 104847. [Google Scholar] [CrossRef] [PubMed]
- Siemian, J.N.; Arenivar, M.A.; Sarsfield, S.; Aponte, Y. Hypothalamic Control of Interoceptive Hunger. Curr. Biol. 2021, 31, 3797–3809.e5. [Google Scholar] [CrossRef] [PubMed]
- Pfrieger, F.W.; Ungerer, N. Cholesterol Metabolism in Neurons and Astrocytes. Prog. Lipid Res. 2011, 50, 357–371. [Google Scholar] [CrossRef]
- Cartocci, V.; Tonini, C.; Di Pippo, T.; Vuono, F.; Schiavi, S.; Marino, M.; Trezza, V.; Pallottini, V. Prenatal Exposure to Valproate Induces Sex-, Age-, and Tissue-Dependent Alterations of Cholesterol Metabolism: Potential Implications on Autism. J. Cell Physiol. 2019, 234, 4362–4374. [Google Scholar] [CrossRef]
- Osterweil, E.K.; Chuang, S.-C.; Chubykin, A.A.; Sidorov, M.; Bianchi, R.; Wong, R.K.S.; Bear, M.F. Lovastatin Corrects Excess Protein Synthesis and Prevents Epileptogenesis in a Mouse Model of Fragile X Syndrome. Neuron 2013, 77, 243–250. [Google Scholar] [CrossRef] [Green Version]
- Pikuleva, I.A.; Cartier, N. Cholesterol Hydroxylating Cytochrome P450 46A1: From Mechanisms of Action to Clinical Applications. Front. Aging Neurosci. 2021, 13, 696778. [Google Scholar] [CrossRef]
- Grayaa, S.; Zerbinati, C.; Messedi, M.; HadjKacem, I.; Chtourou, M.; Ben Touhemi, D.; Naifar, M.; Ayadi, H.; Ayedi, F.; Iuliano, L. Plasma Oxysterol Profiling in Children Reveals 24-Hydroxycholesterol as a Potential Marker for Autism Spectrum Disorders. Biochimie 2018, 153, 80–85. [Google Scholar] [CrossRef]
- Wang, H. Lipid Rafts: A Signaling Platform Linking Cholesterol Metabolism to Synaptic Deficits in Autism Spectrum Disorders. Front. Behav. Neurosci. 2014, 8, 104. [Google Scholar] [CrossRef] [Green Version]
- Martín, M.G.; Pfrieger, F.; Dotti, C.G. Cholesterol in Brain Disease: Sometimes Determinant and Frequently Implicated. EMBO Rep. 2014, 15, 1036–1052. [Google Scholar] [CrossRef] [Green Version]
- Moutinho, M.; Nunes, M.J.; Rodrigues, E. The Mevalonate Pathway in Neurons: It’s Not Just about Cholesterol. Exp. Cell Res. 2017, 360, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Lingor, P.; Teusch, N.; Schwarz, K.; Mueller, R.; Mack, H.; Bähr, M.; Mueller, B.K. Inhibition of Rho Kinase (ROCK) Increases Neurite Outgrowth on Chondroitin Sulphate Proteoglycan in Vitro and Axonal Regeneration in the Adult Optic Nerve in Vivo. J. Neurochem. 2007, 103, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Gopalakrishnan, S.M.; Teusch, N.; Imhof, C.; Bakker, M.H.M.; Schurdak, M.; Burns, D.J.; Warrior, U. Role of Rho Kinase Pathway in Chondroitin Sulfate Proteoglycan-Mediated Inhibition of Neurite Outgrowth in PC12 Cells. J. Neurosci. Res. 2008, 86, 2214–2226. [Google Scholar] [CrossRef] [PubMed]
- Linseman, D.A.; Loucks, F.A. Diverse Roles of Rho Family GTPases in Neuronal Development, Survival, and Death. Front. Biosci. 2008, 13, 657–676. [Google Scholar] [CrossRef]
- Ling, Q.; Tejada-Simon, M.V. Statins and the Brain: New Perspective for Old Drugs. Prog. Neuropsychopharmacol. Biol. Psychiatry 2016, 66, 80–86. [Google Scholar] [CrossRef]
- Avan, R.; Sahebnasagh, A.; Hashemi, J.; Monajati, M.; Faramarzi, F.; Henney, N.C.; Montecucco, F.; Jamialahmadi, T.; Sahebkar, A. Update on Statin Treatment in Patients with Neuropsychiatric Disorders. Life 2021, 11, 1365. [Google Scholar] [CrossRef]
- Sodero, A.O.; Barrantes, F.J. Pleiotropic Effects of Statins on Brain Cells. Biochim. Biophys. Acta. Biomembr. 2020, 1862, 183340. [Google Scholar] [CrossRef]
- Abbasi, D.A.; Nguyen, T.T.A.; Hall, D.A.; Robertson-Dick, E.; Berry-Kravis, E.; Cologna, S.M. Characterization of the Cerebrospinal Fluid Proteome in Patients with Fragile X-Associated Tremor/Ataxia Syndrome. Cerebellum 2022, 21, 86–98. [Google Scholar] [CrossRef]
- Kulinski, J.; Besack, D.; Oleykowski, C.A.; Godwin, A.K.; Yeung, A.T. CEL I Enzymatic Mutation Detection Assay. Biotechniques 2000, 29, 44–48. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein Measurement with the Folin Phenol Reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef]





| Serum Cholesterol mg/dL | |||
|---|---|---|---|
| Groups | WT | Fmr1-Δexon 8 | p |
| PND35 | 55.37 ± 7.98 | 44.53 ± 13.14 * | 0.0233 |
| PND90 | 73.38 ± 13.76 | 51.51 ± 4.55 ** | 0.0097 |
| Liver Cholesterol Content mg/g Tissue | |||
|---|---|---|---|
| Groups | WT | Fmr1-Δexon 8 | p |
| PND35 | 1.22 ± 0.28 | 1.08 ± 0.13 | 0.364 |
| PND90 | 0.87 ± 0.10 | 1.04 ± 0.17 | 0.098 |
| Brain Cholesterol Content mg/g Tissue | |||
|---|---|---|---|
| Groups | WT | Fmr1-Δexon 8 | p |
| Nucleus accumbens | 1.22 ± 0.28 | 1.08 ± 0.13 | 0.364 |
| Cortex | 6.02 ± 0.90 | 5.77 ± 0.80 | 0.644 |
| Amygdala | 5.30 ± 0.81 | 9.66 ± 1.97 ** | 0.004 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Parente, M.; Tonini, C.; Buzzelli, V.; Carbone, E.; Trezza, V.; Pallottini, V. Brain Cholesterol Biosynthetic Pathway Is Altered in a Preclinical Model of Fragile X Syndrome. Int. J. Mol. Sci. 2022, 23, 3408. https://doi.org/10.3390/ijms23063408
Parente M, Tonini C, Buzzelli V, Carbone E, Trezza V, Pallottini V. Brain Cholesterol Biosynthetic Pathway Is Altered in a Preclinical Model of Fragile X Syndrome. International Journal of Molecular Sciences. 2022; 23(6):3408. https://doi.org/10.3390/ijms23063408
Chicago/Turabian StyleParente, Martina, Claudia Tonini, Valeria Buzzelli, Emilia Carbone, Viviana Trezza, and Valentina Pallottini. 2022. "Brain Cholesterol Biosynthetic Pathway Is Altered in a Preclinical Model of Fragile X Syndrome" International Journal of Molecular Sciences 23, no. 6: 3408. https://doi.org/10.3390/ijms23063408






