Frequent Spindle Assembly Errors Require Structural Rearrangement to Complete Meiosis in Zea mays
Abstract
:1. Introduction
2. Results
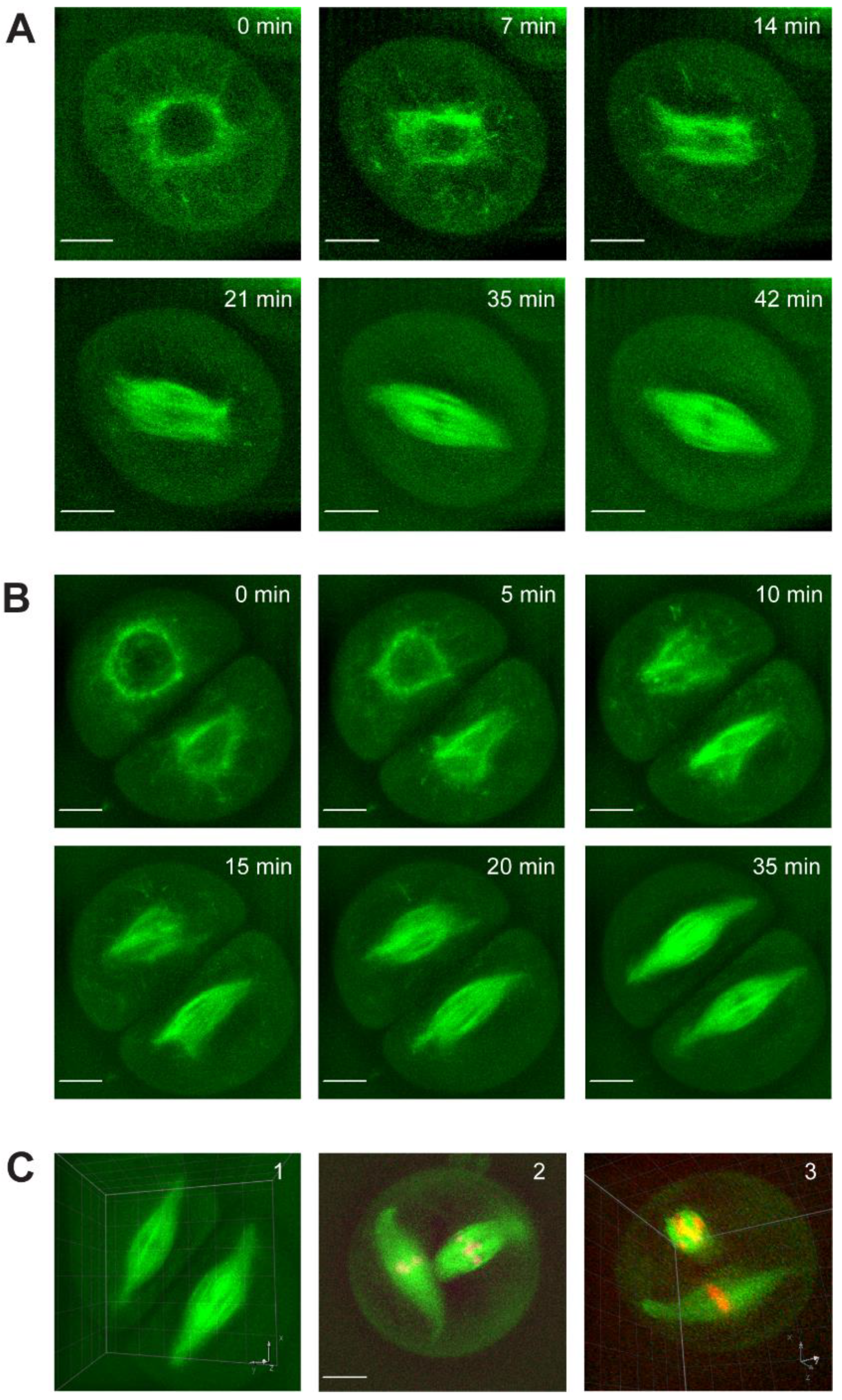
2.1. Spindle Assembly Dynamics in Male Meiosis I and Meiosis II
2.2. Meiotic Chromosome Congression Dynamics
2.3. Spindle Disassembly Dynamics and Phragmoplast Expansion in Meiosis I and II
2.4. Frequent Errors in Spindle Assembly Require Active Correction
3. Discussion
4. Materials and Methods
4.1. Maize Lines and Genotyping
4.2. Live Imaging
4.3. Image Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nagaoka, S.I.; Hassold, T.J.; Hunt, P.A. Human Aneuploidy: Mechanisms and New Insights into an Age-Old Problem. Nat Rev. Genet. 2012, 13, 493–504. [Google Scholar] [CrossRef] [Green Version]
- Bakhoum, S.F.; Ngo, B.; Laughney, A.M.; Cavallo, J.; Murphy, C.J.; Ly, P.; Shah, P.; Sriram, R.K.; Watkins, T.B.; Taunk, N.K. Chromosomal Instability Drives Metastasis through a Cytosolic DNA Response. Nature 2018, 553, 467–472. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taylor, A.M.; Shih, J.; Ha, G.; Gao, G.F.; Zhang, X.; Berger, A.C.; Schumacher, S.E.; Wang, C.; Hu, H.; Liu, J. Genomic and Functional Approaches to Understanding Cancer Aneuploidy. Cancer Cell 2018, 33, 676–689. e3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hassold, T.; Hall, H.; Hunt, P. The Origin of Human Aneuploidy: Where we have been, Where we are Going. Hum. Mol. Genet. 2007, 16, R203–R208. [Google Scholar] [CrossRef] [PubMed]
- Nazockdast, E.; Redemann, S. Mechanics of the Spindle Apparatus. Semin. Cell Dev. Biol. 2020, 107, 91–102. [Google Scholar] [CrossRef]
- Maiato, H.; Logarinho, E. Mitotic Spindle Multipolarity without Centrosome Amplification. Nat. Cell Biol. 2014, 16, 386–394. [Google Scholar] [CrossRef]
- Kapoor, T.M. Metaphase Spindle Assembly. Biology 2017, 6, 8. [Google Scholar] [CrossRef]
- Holubcová, Z.; Blayney, M.; Elder, K.; Schuh, M. Error-Prone Chromosome-Mediated Spindle Assembly Favors Chromosome Segregation Defects in Human Oocytes. Science 2015, 348, 1143–1147. [Google Scholar] [CrossRef] [Green Version]
- Petry, S.; Vale, R.D. Microtubule Nucleation at the Centrosome and Beyond. Nat. Cell Biol. 2015, 17, 1089–1093. [Google Scholar] [CrossRef] [Green Version]
- Heald, R.; Khodjakov, A. Thirty Years of Search and Capture: The Complex Simplicity of Mitotic Spindle Assembly. J. Cell Biol. 2015, 211, 1103–1111. [Google Scholar] [CrossRef]
- Schatten, H.; Sun, Q. The Functional Significance of Centrosomes in Mammalian Meiosis, Fertilization, Development, Nuclear Transfer, and Stem Cell Differentiation. Environ. Mol. Mutagen. 2009, 50, 620–636. [Google Scholar] [CrossRef] [PubMed]
- Sumiyoshi, E.; Sugimoto, A.; Yamamoto, M. Protein Phosphatase 4 is Required for Centrosome Maturation in Mitosis and Sperm Meiosis in C. Elegans. J. Cell. Sci. 2002, 115, 1403–1410. [Google Scholar] [CrossRef] [PubMed]
- Casal, J.; Gonzalez, C.; Ripoll, P. Spindles and Centrosomes during Male Meiosis in Drosophila Melanogaster. Eur. J. Cell Biol. 1990, 51, 38–44. [Google Scholar]
- Kalt, M.R. Ultrastructural Observations on the Germ Line of Xenopus Laevis. Z. Zellforsch Mikrosk. Anat. 1973, 138, 41–62. [Google Scholar] [CrossRef] [PubMed]
- Yamada, M.; Goshima, G. Mitotic Spindle Assembly in Land Plants: Molecules and Mechanisms. Biology 2017, 6, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schuh, M.; Ellenberg, J. Self-Organization of MTOCs Replaces Centrosome Function during Acentrosomal Spindle Assembly in Live Mouse Oocytes. Cell 2007, 130, 484–498. [Google Scholar] [CrossRef] [Green Version]
- Clift, D.; Schuh, M. A Three-Step MTOC Fragmentation Mechanism Facilitates Bipolar Spindle Assembly in Mouse Oocytes. Nat. Commun. 2015, 6, 7217. [Google Scholar] [CrossRef] [Green Version]
- Xue, Z.; Liu, C.; Shi, W.; Miao, Y.; Shen, Y.; Tang, D.; Li, Y.; You, A.; Xu, Y.; Chong, K. OsMTOPVIB is Required for Meiotic Bipolar Spindle Assembly. Proc. Natl. Acad. Sci. USA 2019, 116, 15967–15972. [Google Scholar] [CrossRef] [Green Version]
- Skold, H.N.; Komma, D.J.; Endow, S.A. Assembly Pathway of the Anastral Drosophila Oocyte Meiosis I Spindle. J. Cell. Sci. 2005, 118, 1745–1755. [Google Scholar] [CrossRef] [Green Version]
- Baumann, C.; Wang, X.; Yang, L.; Viveiros, M.M. Error-Prone Meiotic Division and Subfertility in Mice with Oocyte-Conditional Knockdown of Pericentrin. J. Cell. Sci. 2017, 130, 1251–1262. [Google Scholar] [CrossRef] [Green Version]
- Lane, S.I.; Yun, Y.; Jones, K.T. Timing of Anaphase-Promoting Complex Activation in Mouse Oocytes is Predicted by Microtubule-Kinetochore Attachment but Not by Bivalent Alignment or Tension. Development 2012, 139, 1947–1955. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paz, J.; Lüders, J. Microtubule-Organizing Centers: Towards a Minimal Parts List. Trends Cell Biol. 2018, 28, 176–187. [Google Scholar] [CrossRef] [PubMed]
- Dumont, J.; Desai, A. Acentrosomal Spindle Assembly and Chromosome Segregation during Oocyte Meiosis. Trends Cell Biol. 2012, 22, 241–249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burbank, K.S.; Mitchison, T.J.; Fisher, D.S. Slide-and-Cluster Models for Spindle Assembly. Curr. Biol. 2007, 17, 1373–1383. [Google Scholar] [CrossRef] [Green Version]
- Wasteneys, G.O. Microtubule Organization in the Green Kingdom: Chaos Or Self-Order? J. Cell. Sci. 2002, 115, 1345–1354. [Google Scholar] [CrossRef]
- Zhang, H.; Dawe, R.K. Mechanisms of Plant Spindle Formation. Chromosome Res. 2011, 19, 335–344. [Google Scholar] [CrossRef]
- Chan, A.; Cande, W.Z. Maize Meiotic Spindles Assemble Around Chromatin and do Not Require Paired Chromosomes. J. Cell. Sci. 1998, 111 Pt 23, 3507–3515. [Google Scholar] [CrossRef]
- Smirnova, E.A.; Bajer, A.S. Microtubule Converging Centers and Reorganization of the Interphase Cytoskeleton and the Mitotic Spindle in Higher Plant Haemanthus. Cell Motil. Cytoskeleton 1994, 27, 219–233. [Google Scholar] [CrossRef]
- Smirnova, E.A.; Bajer, A.S. Early Stages of Spindle Formation and Independence of Chromosome and Microtubule Cycles in Haemanthus Endosperm. Cell Motil. Cytoskeleton 1998, 40, 22–37. [Google Scholar] [CrossRef]
- Martinez, P.; Luo, A.; Sylvester, A.; Rasmussen, C.G. Proper Division Plane Orientation and Mitotic Progression Together Allow Normal Growth of Maize. Proc. Natl. Acad. Sci. USA 2017, 114, 2759–2764. [Google Scholar] [CrossRef] [Green Version]
- Rasmussen, C.G. Using Live-Cell Markers in Maize to Analyze Cell Division Orientation and Timing. Plant Cell Div. Methods Protoc. 2016, 2016, 209–225. [Google Scholar]
- Chan, J.; Calder, G.; Fox, S.; Lloyd, C. Localization of the Microtubule End Binding Protein EB1 Reveals Alternative Pathways of Spindle Development in Arabidopsis Suspension Cells. Plant Cell 2005, 17, 1737–1748. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lloyd, C.; Chan, J. Not so Divided: The Common Basis of Plant and Animal Cell Division. Nat. Rev. Mol. Cell Biol. 2006, 7, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Nakaoka, Y.; Miki, T.; Fujioka, R.; Uehara, R.; Tomioka, A.; Obuse, C.; Kubo, M.; Hiwatashi, Y.; Goshima, G. An Inducible RNA Interference System in Physcomitrella Patens Reveals a Dominant Role of Augmin in Phragmoplast Microtubule Generation. Plant Cell 2012, 24, 1478–1493. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sheehan, M.J.; Pawlowski, W.P. Live Imaging of Rapid Chromosome Movements in Meiotic Prophase I in Maize. Proc. Natl. Acad. Sci. USA 2009, 106, 20989–20994. [Google Scholar] [CrossRef] [Green Version]
- Sheehan, M.J.; Pawlowski, W.P. Imaging Chromosome Dynamics in Meiosis in Plants. Meth. Enzymol. 2012, 505, 125. [Google Scholar]
- Yu, H.; Hiatt, E.N.; Chan, A.; Sweeney, M.; Dawe, R.K. Neocentromere-Mediated Chromosome Movement in Maize. J. Cell Biol. 1997, 139, 831–840. [Google Scholar] [CrossRef] [Green Version]
- Nannas, N.J.; Higgins, D.M.; Dawe, R.K. Anaphase Asymmetry and Dynamic Repositioning of the Division Plane during Maize Meiosis. J. Cell Sci. 2016, 21, 4014–4024. [Google Scholar] [CrossRef] [Green Version]
- Nannas, N.J.; Dawe, R.K. Live Cell Imaging of Meiotic Spindle and Chromosome Dynamics in Maize (Zea Mays). Curr. Protoc. Plant Biol. 2016, 4, 546–565. [Google Scholar] [CrossRef]
- Rasmussen, C.G.; Wright, A.J.; Müller, S. The Role of the Cytoskeleton and Associated Proteins in Determination of the Plant Cell Division Plane. Plant J. 2013, 75, 258–269. [Google Scholar] [CrossRef] [Green Version]
- Prusicki, M.A.; Keizer, E.M.; van Rosmalen, R.P.; Komaki, S.; Seifert, F.; Müller, K.; Wijnker, E.; Fleck, C.; Schnittger, A. Live Cell Imaging of Meiosis in Arabidopsis Thaliana. Elife 2019, 8, e42834. [Google Scholar] [CrossRef] [PubMed]
- Cande, W.Z.; Golubovskaya, I.N.; Wang, C.; Harper, L.C. Meiotic Genes and Meiosis in Maize. In Handbook of Maize: Genetics and Genomics; Bennetzen, J., Hake, S., Eds.; Springer: New York, NY, USA, 2009; pp. 353–375. [Google Scholar]
- Golubovskaya, I.N.; Sheridan, W.F.; Harper, L.C.; Cande, W.Z. Novel Meiotic Mutants of Maize Identified from Mu Transposon and EMS Mutant Screens. Maize Genet. Coop. Newsl. 2003, 77, 10–12. [Google Scholar]
- Staiger, C.J.; Cande, W.Z. Cytoskeletal analysis of maize meiotic mutants. In Molecular and Cell Biology of the Plant Cell Cycle; Springer: Dordrecht, The Netherlands, 1993; pp. 157–171. [Google Scholar]
- Dixit, R.; Cyr, R.J. Spatio-Temporal Relationship between Nuclear-Envelope Breakdown and Preprophase Band Disappearance in Cultured Tobacco Cells. Protoplasma 2002, 219, 116–121. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, T.; Sano, T.; Kutsuna, N.; Kumagai-Sano, F.; Hasezawa, S. Contribution of Anaphase B to Chromosome Separation in Higher Plant Cells Estimated by Image Processing. Plant Cell Physiol. 2007, 48, 1509–1513. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Otegui, M.S.; Verbrugghe, K.J.; Skop, A.R. Midbodies and Phragmoplasts: Analogous Structures Involved in Cytokinesis. Trends Cell Biol. 2005, 15, 404–413. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, Y.; Buchwalter, R.A.; Zheng, C.; Wight, E.M.; Chen, J.V.; Megraw, T.L. A Perinuclear Microtubule-Organizing Centre Controls Nuclear Positioning and Basement Membrane Secretion. Nat. Cell Biol. 2020, 22, 297–309. [Google Scholar] [CrossRef] [PubMed]
- Buschmann, H.; Holtmannspötter, M.; Borchers, A.; O’Donoghue, M.; Zachgo, S. Microtubule Dynamics of the Centrosome-like Polar Organizers from the Basal Land Plant Marchantia Polymorpha. New Phytol. 2016, 209, 999–1013. [Google Scholar] [CrossRef] [Green Version]
- Komaki, S.; Takeuchi, H.; Hamamura, Y.; Heese, M.; Hashimoto, T.; Schnittger, A. Functional Analysis of the Plant Chromosomal Passenger Complex. Plant Physiol. 2020, 183, 1586–1599. [Google Scholar] [CrossRef]
- Lee, J.; Lee, H.; Wi, S.; Park, K.Y.; Schmit, A.; Pai, H. Dual Functions of Nicotiana Benthamiana Rae1 in Interphase and Mitosis. Plant J. 2009, 59, 278–291. [Google Scholar] [CrossRef]
- Park, G.T.; Frost, J.M.; Park, J.; Kim, T.H.; Lee, J.S.; Oh, S.A.; Twell, D.; Brooks, J.S.; Fischer, R.L.; Choi, Y. Nucleoporin MOS7/Nup88 is Required for Mitosis in Gametogenesis and Seed Development in Arabidopsis. Proc. Natl. Acad. Sci. USA 2014, 111, 18393–18398. [Google Scholar] [CrossRef] [Green Version]
- Liu, B.; Marc, J.; Joshi, H.C.; Palevitz, B.A. A Gamma-Tubulin-Related Protein Associated with the Microtubule Arrays of Higher Plants in a Cell Cycle-Dependent Manner. J. Cell. Sci. 1993, 104, 1217–1228. [Google Scholar] [CrossRef] [PubMed]
- Vos, J.W.; Pieuchot, L.; Evrard, J.; Janski, N.; Bergdoll, M.; de Ronde, D.; Perez, L.H.; Sardon, T.; Vernos, I.; Schmit, A. The Plant TPX2 Protein Regulates Prospindle Assembly before Nuclear Envelope Breakdown. Plant Cell 2008, 20, 2783–2797. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mullen, T.J.; Davis-Roca, A.C.; Wignall, S.M. Spindle Assembly and Chromosome Dynamics during Oocyte Meiosis. Curr. Opin. Cell Biol. 2019, 60, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Kitajima, T.S.; Ohsugi, M.; Ellenberg, J. Complete Kinetochore Tracking Reveals Error-Prone Homologous Chromosome Biorientation in Mammalian Oocytes. Cell 2011, 146, 568–581. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Baumann, C.; De La Fuente, R.; Viveiros, M.M. CEP215 and AURKA Regulate Spindle Pole Focusing and aMTOC Organization in Mouse Oocytes. Reproduction 2020, 159, 261–274. [Google Scholar] [CrossRef]
- Higgins, D.M.; Nannas, N.J.; Dawe, R.K. The Maize Divergent Spindle-1 (Dv1) Gene Encodes a Kinesin-14A Motor Protein Required for Meiotic Spindle Pole Maturation. Front. Plant Sci. 2016, 7, 1277. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.; Marcus, A.; Li, W.; Hu, Y.; Calzada, J.V.; Grossniklaus, U.; Cyr, R.J.; Ma, H. The Arabidopsis ATK1 Gene is Required for Spindle Morphogenesis in Male Meiosis. Development 2002, 129, 2401–2409. [Google Scholar] [CrossRef]
- Quan, L.; Xiao, R.; Li, W.; Oh, S.; Kong, H.; Ambrose, J.C.; Malcos, J.L.; Cyr, R.; Twell, D.; Ma, H. Functional Divergence of the Duplicated AtKIN14a and AtKIN14b Genes: Critical Roles in Arabidopsis Meiosis and Gametophyte Development. Plant J. 2008, 53, 1013–1026. [Google Scholar] [CrossRef]
- Lawrence, C.J.; Morris, N.R.; Meagher, R.B.; Dawe, R.K. Dyneins have Run their Course in Plant Lineage. Traffic 2001, 2, 362–363. [Google Scholar] [CrossRef] [Green Version]
- Merdes, A.; Cleveland, D.W. Pathways of Spindle Pole Formation: Different Mechanisms; Conserved Components. J. Cell Biol. 1997, 138, 953–956. [Google Scholar] [CrossRef] [Green Version]
- Balakier, H.; Sojecki, A.; Motamedi, G.; Librach, C. Time-dependent Capability of Human Oocytes for Activation and Pronuclear Formation during Metaphase II Arrest. Hum. Reprod. 2004, 19, 982–987. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Y.; Wang, L.; Zhang, L.; He, Z.; Feng, G.; Sun, H.; Wang, J.; Li, Z.; Liu, C.; Han, J.; et al. Cyclin B3 is Required for Metaphase to Anaphase Transition in Oocyte Meiosis I. J. Cell Biol. 2019, 218, 1553–1563. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, H.G.; Muszynski, M.G.; Kelly Dawe, R. The Maize Homologue of the Cell Cycle Checkpoint Protein MAD2 Reveals Kinetochore Substructure and Contrasting Mitotic and Meiotic Localization Patterns. J. Cell Biol. 1999, 145, 425–435. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shah, J.V.; Cleveland, D.W. Waiting for Anaphase: Mad2 and the Spindle Assembly Checkpoint. Cell 2000, 103, 997–1000. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Dawe, R.K. Fused Sister Kinetochores Initiate the Reductional Division in Meiosis, I. Nat. Cell Biol. 2009, 11, 1103–1108. [Google Scholar] [CrossRef]
- Wolff, I.D.; Tran, M.V.; Mullen, T.J.; Villeneuve, A.M.; Wignall, S.M. Assembly of Caenorhabditis Elegans Acentrosomal Spindles Occurs without Evident Microtubule-Organizing Centers and Requires Microtubule Sorting by KLP-18/Kinesin-12 and MESP-1. Mol. Biol. Cell 2016, 27, 3122–3131. [Google Scholar] [CrossRef]
- Fu, M.; Wang, C.; Xue, F.; Higgins, J.; Chen, M.; Zhang, D.; Liang, W. The DNA Topoisomerase VI–B Subunit OsMTOPVIB is Essential for Meiotic Recombination Initiation in Rice. Mol. Plant 2016, 9, 1539–1541. [Google Scholar] [CrossRef] [Green Version]
- Xue, Z.; Li, Y.; Zhang, L.; Shi, W.; Zhang, C.; Feng, M.; Zhang, F.; Tang, D.; Yu, H.; Gu, M. OsMTOPVIB Promotes Meiotic DNA Double-Strand Break Formation in Rice. Mol. Plant 2016, 9, 1535–1538. [Google Scholar] [CrossRef]
- Li, J.; Dukowic-Schulze, S.; Lindquist, I.E.; Farmer, A.D.; Kelly, B.; Li, T.; Smith, A.G.; Retzel, E.F.; Mudge, J.; Chen, C. The Plant-Specific Protein FEHLSTART Controls Male Meiotic Entry, Initializing Meiotic Synchronization in Arabidopsis. Plant J. 2015, 84, 659–671. [Google Scholar] [CrossRef]
- Vogt, E.; Kirsch-Volders, M.; Parry, J.; Eichenlaub-Ritter, U. Spindle Formation, Chromosome Segregation and the Spindle Checkpoint in Mammalian Oocytes and Susceptibility to Meiotic Error. Mutat. Res./Genet. Toxicol. Environ. Mutagenesis 2008, 651, 14–29. [Google Scholar] [CrossRef]
- Kwon, M.; Godinho, S.A.; Chandhok, N.S.; Ganem, N.J.; Azioune, A.; Thery, M.; Pellman, D. Mechanisms to Suppress Multipolar Divisions in Cancer Cells with Extra Centrosomes. Genes Dev. 2008, 22, 2189–2203. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, Z.; Lončarek, J.; Khodjakov, A.; Rieder, C.L. Extra Centrosomes and/Or Chromosomes Prolong Mitosis in Human Cells. Nat. Cell Biol. 2008, 10, 748–751. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Su, H.; Liu, Y.; Dong, Q.; Feng, C.; Zhang, J.; Liu, Y.; Birchler, J.A.; Han, F. Dynamic Location Changes of Bub1-phosphorylated-H2AThr133 with CENH3 Nucleosome in Maize Centromeric Regions. New Phytol. 2017, 214, 682–694. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, H.; Wu, H.; Pan, X.; Jin, W.; Li, X. Aberrant Meiotic Modulation Partially Contributes to the Lower Germination Rate of Pollen Grains in Maize (Zea Mays, L.) Under Low Nitrogen Supply. Plant Cell Physiol. 2017, 58, 342–353. [Google Scholar] [PubMed]
- Savoian, M.S.; Rieder, C.L. Mitosis in Primary Cultures of Drosophila Melanogaster Larval Neuroblasts. J. Cell Sci. 2002, 115, 3061–3072. [Google Scholar] [CrossRef] [PubMed]
- Gatlin, J.C.; Matov, A.; Groen, A.C.; Needleman, D.J.; Maresca, T.J.; Danuser, G.; Mitchison, T.J.; Salmon, E.D. Spindle Fusion Requires Dynein-Mediated Sliding of Oppositely Oriented Microtubules. Curr. Biol. 2009, 19, 287–296. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Church, K.; Nicklas, R.B.; Lin, H.P. Micromanipulated Bivalents can Trigger Mini-Spindle Formation in Drosophila Melanogaster Spermatocyte Cytoplasm. J. Cell Biol. 1986, 103, 2765–2773. [Google Scholar] [CrossRef] [Green Version]
- Yu, H.; Li, J. Breeding Future Crops to Feed the World through De Novo Domestication. Nat. Commun. 2022, 13, 1171. [Google Scholar] [CrossRef]
- Tilman, D.; Balzer, C.; Hill, J.; Befort, B.L. Global Food Demand and the Sustainable Intensification of Agriculture. Proc. Natl. Acad. Sci. USA 2011, 108, 20260–20264. [Google Scholar] [CrossRef] [Green Version]
- Godfray, H.C.J.; Beddington, J.R.; Crute, I.R.; Haddad, L.; Lawrence, D.; Muir, J.F.; Pretty, J.; Robinson, S.; Thomas, S.M.; Toulmin, C. Food Security: The Challenge of Feeding 9 Billion People. Science 2010, 327, 812–818. [Google Scholar] [CrossRef] [Green Version]






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Weiss, J.D.; McVey, S.L.; Stinebaugh, S.E.; Sullivan, C.F.; Dawe, R.K.; Nannas, N.J. Frequent Spindle Assembly Errors Require Structural Rearrangement to Complete Meiosis in Zea mays. Int. J. Mol. Sci. 2022, 23, 4293. https://doi.org/10.3390/ijms23084293
Weiss JD, McVey SL, Stinebaugh SE, Sullivan CF, Dawe RK, Nannas NJ. Frequent Spindle Assembly Errors Require Structural Rearrangement to Complete Meiosis in Zea mays. International Journal of Molecular Sciences. 2022; 23(8):4293. https://doi.org/10.3390/ijms23084293
Chicago/Turabian StyleWeiss, Jodi D., Shelby L. McVey, Sarah E. Stinebaugh, Caroline F. Sullivan, R. Kelly Dawe, and Natalie J. Nannas. 2022. "Frequent Spindle Assembly Errors Require Structural Rearrangement to Complete Meiosis in Zea mays" International Journal of Molecular Sciences 23, no. 8: 4293. https://doi.org/10.3390/ijms23084293
APA StyleWeiss, J. D., McVey, S. L., Stinebaugh, S. E., Sullivan, C. F., Dawe, R. K., & Nannas, N. J. (2022). Frequent Spindle Assembly Errors Require Structural Rearrangement to Complete Meiosis in Zea mays. International Journal of Molecular Sciences, 23(8), 4293. https://doi.org/10.3390/ijms23084293





