Physiological and Pathological Significance of Esophageal TRP Channels: Special Focus on TRPV4 in Esophageal Epithelial Cells
Abstract
:1. Introduction
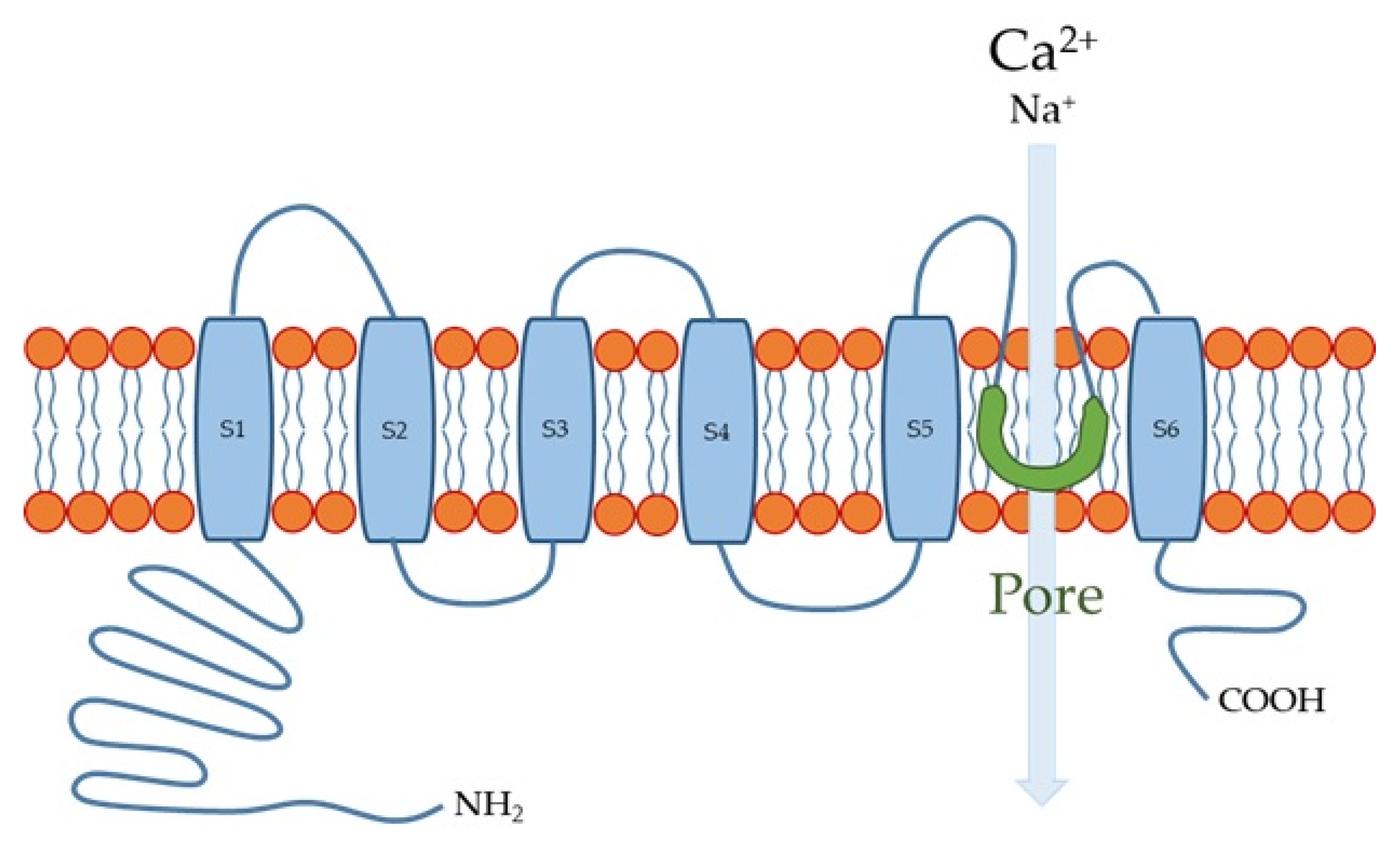
2. TRP Channels in the Esophageal Wall
3. TRPV4 in Mechanosensation
4. TRPV4 in Cell Proliferation and Migration
5. TRPV4 in Esophageal Inflammation and Tumors
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Greenwood-Van Meerveld, B.; Johnson, A.C.; Grundy, D. Gastrointestinal Physiology and Function. In Handbook of Experimental Pharmacology; Springer: Berlin/Heidelberg, Germany, 2017; pp. 1–16. ISBN 978-3-319-29806-1. [Google Scholar]
- Furness, J.B. The enteric nervous system and neurogastroenterology. Nat. Rev. Gastroenterol. Hepatol. 2012, 9, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Becskeházi, E.; Korsós, M.M.; Erőss, B.; Hegyi, P.; Venglovecz, V. OEsophageal Ion Transport Mechanisms and Significance Under Pathological Conditions. Front. Physiol. 2020, 11, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Von Furstenberg, R.J.; Li, J.; Stolarchuk, C.; Feder, R.; Campbell, A.; Kruger, L.; Gonzalez, L.M.; Blikslager, A.T.; Cardona, D.M.; McCall, S.J.; et al. Porcine Esophageal Submucosal Gland Culture Model Shows Capacity for Proliferation and Differentiation. Cmgh 2017, 4, 385–404. [Google Scholar] [CrossRef] [Green Version]
- Beyder, A.; Farrugia, G. Targeting ion channels for the treatment of gastrointestinal motility disorders. Therap. Adv. Gastroenterol. 2012, 5, 5–21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Justinich, C.J.; Mak, N.; Pacheco, I.; Mulder, D.; Wells, R.W.; Blennerhassett, M.G.; MacLeod, R.J. The extracellular calcium-sensing receptor (CaSR) on human esophagus and evidence of expression of the CaSR on the esophageal epithelial cell line (HET-1A). Am. J. Physiol.Gastrointest. Liver Physiol. 2008, 294, G120–G129. [Google Scholar] [CrossRef]
- Yoshida, N.; Katada, K.; Handa, O.; Takagi, T.; Kokura, S.; Naito, Y.; Mukaida, N.; Soma, T.; Shimada, Y.; Yoshikawa, T.; et al. Interleukin-8 production via protease-activated receptor 2 in human esophageal epithelial cells. Int. J. Mol. Med. 2007, 19, 335–340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Awayda, M.S.; Bengrine, A.; Tobey, N.A.; Stockand, J.D.; Orlando, R.C. Nonselective cation transport in native esophageal epithelia. Am. J. Physiol. Cell Physiol. 2004, 287, C395–C402. [Google Scholar] [CrossRef] [Green Version]
- Yamamura, H.; Ugawa, S.; Ueda, T.; Nagao, M.; Joh, T.; Shimada, S. Epithelial Na+ channel δ subunit is an acid sensor in the human oesophagus. Eur. J. Pharmacol. 2008, 600, 32–36. [Google Scholar] [CrossRef]
- Wu, L.; Oshima, T.; Shan, J.; Sei, H.; Tomita, T.; Ohda, Y.; Fukui, H.; Watari, J.; Miwa, H. PAR-2 activation enhances weak acid-induced ATP release through TRPV1 and ASIC sensitization in human esophageal epithelial cells. Am. J. Physiol. Gastrointest. Liver Physiol. 2015, 309, G695–G702. [Google Scholar] [CrossRef] [Green Version]
- Guarino, M.P.L.; Cheng, L.; Ma, J.; Harnett, K.; Biancani, P.; Altomare, A.; Panzera, F.; Behar, J.; Cicala, M. Increased TRPV1 gene expression in esophageal mucosa of patients with non-erosive and erosive reflux disease. Neurogastroenterol. Motil. 2010, 22, 746–752. [Google Scholar] [CrossRef]
- Mihara, H.; Boudaka, A.; Sugiyama, T.; Moriyama, Y.; Tominaga, M. Transient receptor potential vanilloid 4 (TRPV4)-dependent calcium influx and ATP release in mouse oesophageal keratinocytes. J. Physiol. 2011, 589, 3471–3482. [Google Scholar] [CrossRef] [Green Version]
- Holzer, P. TRP channels in the digestive system. Curr. Pharm. Biotechnol. 2011, 12, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Nilius, B.; Owsianik, G. The transient receptor potential family of ion channels. Genome Biol. 2011, 12, 218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caterina, M.J.; Schumacher, M.A.; Tominaga, M.; Rosen, T.A.; Levine, J.D.; Julius, D. The capsaicin receptor: A heat-activated ion channel in the pain pathway. Nature 1997, 389, 816–824. [Google Scholar] [CrossRef]
- Caterina, M.J. Transient receptor potential ion channels as participants in thermosensation and thermoregulation. AJP Regul. Integr. Comp. Physiol. 2006, 292, R64–R76. [Google Scholar] [CrossRef] [PubMed]
- Venkatachalam, K.; Montell, C. TRP Channels. Annu. Rev. Biochem. 2007, 76, 387–417. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, L.; Sweet, T.; Clapham, D.E. International Union of Basic and Clinical Pharmacology. LXXVI. Current Progress in the Mammalian TRP Ion Channel Family. Pharmacol. Rev. 2010, 62, 381–404. [Google Scholar] [CrossRef] [PubMed]
- Mihara, H.; Suzuki, N.; Boudaka, A.A.; Muhammad, J.S.; Tominaga, M.; Tabuchi, Y.; Sugiyama, T. Transient receptor potential vanilloid 4-dependent calcium influx and ATP release in mouse and rat gastric epithelia. World J. Gastroenterol. 2016, 22, 5512. [Google Scholar] [CrossRef]
- Mihara, H.; Boudaka, A.; Shibasaki, K.; Yamanaka, A.; Sugiyama, T.; Tominaga, M. Involvement of TRPV2 activation in intestinal movement through nitric oxide production in mice. J. Neurosci. 2010, 30, 16536–16544. [Google Scholar] [CrossRef] [PubMed]
- Boudaka, A.; Wörl, J.; Shiina, T.; Neuhuber, W.L.; Kobayashi, H.; Shimizu, Y.; Takewaki, T. Involvement of TRPV1-dependent and -independent components in the regulation of vagally induced contractions in the mouse esophagus. Eur. J. Pharmacol. 2007, 556, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Boudaka, A.; Wörl, J.; Shiina, T.; Saito, S.; Atoji, Y.; Kobayashi, H.; Shimizu, Y.; Takewaki, T. Key role of mucosal primary afferents in mediating the inhibitory influence of capsaicin on vagally mediated contractions in the mouse esophagus. J. Vet. Med. Sci. 2007, 69, 365–372. [Google Scholar] [CrossRef] [Green Version]
- Boudaka, A.; Wörl, J.; Shiina, T.; Shimizu, Y.; Takewaki, T.; Neuhuber, W.L. Galanin modulates vagally induced contractions in the mouse oesophagus. Neurogastroenterol. Motil. 2009, 21, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Alaimo, A.; Rubert, J. The pivotal role of TRP channels in homeostasis and diseases throughout the gastrointestinal tract. Int. J. Mol. Sci. 2019, 20, 5277. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holzer, P. Transient receptor potential (TRP) channels as drug targets for diseases of the digestive system. Pharmacol. Ther. 2011, 131, 142–170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boudaka, A.; Saito, C.T.; Tominaga, M. Deletion of TRPV4 enhances in vitro wound healing of murine esophageal keratinocytes. Sci. Rep. 2020, 10, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Boesmans, W.; Owsianik, G.; Tack, J.; Voets, T.; Vanden Berghe, P. TRP channels in neurogastroenterology: Opportunities for therapeutic intervention. Br. J. Pharmacol. 2010, 162, 18–37. [Google Scholar] [CrossRef] [Green Version]
- Bellono, N.W.; Bayrer, J.R.; Leitch, D.B.; Castro, J.; Zhang, C.; O’Donnell, T.A.; Brierley, S.M.; Ingraham, H.A.; Julius, D. Enterochromaffin Cells Are Gut Chemosensors that Couple to Sensory Neural Pathways. Cell 2017, 170, 185–198.e16. [Google Scholar] [CrossRef] [Green Version]
- Szallasi, A.; Cortright, D.N.; Blum, C.A.; Eid, S.R. The vanilloid receptor TRPV1: 10 years from channel cloning to antagonist proof-of-concept. Nat. Rev. Drug Discov. 2007, 6, 357–372. [Google Scholar] [CrossRef]
- Viana, F. TRPA1 channels: Molecular sentinels of cellular stress and tissue damage. J. Physiol. 2016, 594, 4151–4169. [Google Scholar] [CrossRef] [Green Version]
- Bezençon, C.; le Coutre, J.; Damak, S. Taste-signaling proteins are coexpressed in solitary intestinal epithelial cells. Chem. Senses 2007, 32, 41–49. [Google Scholar] [CrossRef] [Green Version]
- Hantute-Ghesquier, A.; Haustrate, A.; Prevarskaya, N.; Lehen’kyi, V. TRPM family channels in cancer. Pharmaceuticals 2018, 11, 58. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kobayashi, K.; Fukuoka, T.; Obata, K.; Yamanaka, H.; Dai, Y.; Tokunaga, A.; Noguchi, K. Distinct expression of TRPM8, TRPA1, and TRPV1 mRNAs in rat primary afferent neurons with Aδ/C-fibers and colocalization with Trk receptors. J. Comp. Neurol. 2005, 493, 596–606. [Google Scholar] [CrossRef] [PubMed]
- McKemy, D.D.; Neuhausser, W.M.; Julius, D. Identification of a cold receptor reveals a general role for TRP channels in thermosensation. Nature 2002, 416, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Gao, G.; Peterson, B.Z.; Ouyang, A. TRPA1 in mast cell activation-induced long-lasting mechanical hypersensitivity of vagal afferent C-fibers in guinea pig esophagus. Am. J. Physiol. Gastrointest. Liver Physiol. 2009, 297, G34–G42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brozmanova, M.; Ru, F.; Surdenikova, L.; Mazurova, L.; Taylor-Clark, T.; Kollarik, M. Preferential activation of the vagal nodose nociceptive subtype by TRPA1 agonists in the guinea pig esophagus. Neurogastroenterol. Motil. 2011, 23, 437–445. [Google Scholar] [CrossRef] [Green Version]
- Liu, Z.; Hu, Y.; Yu, X.; Xi, J.; Fan, X.; Tse, C.M.; Myers, A.C.; Pasricha, P.J.; Li, X.; Yu, S. Allergen challenge sensitizes TRPA1 in vagal sensory neurons and afferent C-fiber subtypes in guinea pig esophagus. Am. J. Physiol. Gastrointest. Liver Physiol. 2015, 308, G482–G488. [Google Scholar] [CrossRef] [Green Version]
- Csekő, K.; Pécsi, D.; Kajtár, B.; Hegedűs, I.; Bollenbach, A.; Tsikas, D.; Szabó, I.L.; Szabó, S.; Helyes, Z. Upregulation of the trpa1 ion channel in the gastric mucosa after iodoacetamide-induced gastritis in rats: A potential new therapeutic target. Int. J. Mol. Sci. 2020, 21, 5591. [Google Scholar] [CrossRef]
- Yu, X.; Hu, Y.; Ru, F.; Kollarik, M.; Undem, B.J.; Yu, S. TRPM8 function and expression in vagal sensory neurons and afferent nerves innervating guinea pig esophagus. Am. J. Physiol. Gastrointest. Liver Physiol. 2015, 308, 489–496. [Google Scholar] [CrossRef] [Green Version]
- Banovcin, P.; Duricek, M.; Zatko, T.; Liptak, P.; Hyrdel, R.; Kollarik, M. The infusion of menthol into the esophagus evokes cold sensations in healthy subjects but induces heartburn in patients with gastroesophageal reflux disease (GERD). Dis. Esophagus 2019, 32, 1–6. [Google Scholar] [CrossRef]
- Kindt, S.; Vos, R.; Blondeau, K.; Tack, J. Influence of intra-oesophageal capsaicin instillation on heartburn induction and oesophageal sensitivity in man. Neurogastroenterol. Motil. 2009, 21, 1032–1039. [Google Scholar] [CrossRef]
- Kishimoto, E.; Naito, Y.; Handa, O.; Okada, H.; Mizushima, K.; Hirai, Y.; Nakabe, N.; Uchiyama, K.; Ishikawa, T.; Takagi, T.; et al. Oxidative stress-induced posttranslational modification of TRPV1 expressed in esophageal epithelial cells. Am. J. Physiol. Gastrointest. Liver Physiol. 2011, 301, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Wu, X.; Zhang, L.; Mao, A.; Ma, X.; He, D. Menthol relieves acid reflux inflammation by regulating TRPV1 in esophageal epithelial cells. Biochem. Biophys. Res. Commun. 2020, 525, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Mihara, H.; Suzuki, N.; Yamawaki, H.; Tominaga, M.; Sugiyama, T. TRPV2 ion channels expressed in inhibitory motor neurons of gastric myenteric plexus contribute to gastric adaptive relaxation and gastric emptying in mice. Am. J. Physiol. Gastrointest. Liver Physiol. 2013, 304, 22–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watanabe, H.; Davis, J.B.; Smart, D.; Jerman, J.C.; Smith, G.D.; Hayes, P.; Vriens, J.; Cairns, W.; Wissenbach, U.; Prenen, J.; et al. Activation of TRPV4 channels (hVRL-2/mTRP12) by phorbol derivatives. J. Biol. Chem. 2002, 277, 13569–13577. [Google Scholar] [CrossRef] [Green Version]
- Zhuang, L.; Peng, J.B.; Tou, L.; Takanaga, H.; Adam, R.M.; Hediger, M.A.; Freeman, M.R. Calcium-selective ion channel, CaT1, is apically localized in gastrointestinal tract epithelia and is aberrantly expressed in human malignancies. Lab. Investig. 2002, 82, 1755–1764. [Google Scholar] [CrossRef] [Green Version]
- Matthews, P.J.; Aziz, Q.; Facer, P.; Davis, J.B.; Thompson, D.G.; Anand, P. Increased capsaicin receptor TRPV1 nerve fibres in the inflamed human oesophagus. Eur. J. Gastroenterol. Hepatol. 2004, 16, 897–902. [Google Scholar] [CrossRef]
- Yu, X.; Yu, M.; Liu, Y.; Yu, S. TRP channel functions in the gastrointestinal tract. Semin. Immunopathol. 2016, 38, 385–396. [Google Scholar] [CrossRef]
- Ustaoglu, A.; Sawada, A.; Lee, C.; Lei, W.Y.; Chen, C.L.; Hackett, R.; Sifrim, D.; Peiris, M.; Woodland, P. Heartburn sensation in nonerosive reflux disease: Pattern of superficial sensory nerves expressing TRPV1 and epithelial cells expressing ASIC3 receptors. Am. J. Physiol. Gastrointest. Liver Physiol. 2021, 320, G804–G815. [Google Scholar] [CrossRef]
- Silva, R.O.; Bingana, R.D.; Sales, T.M.A.L.; Moreira, R.L.R.; Costa, D.V.S.; Sales, K.M.O.; Brito, G.A.C.; Santos, A.A.; Souza, M.Â.N.; Soares, P.M.G.; et al. Role of TRPV1 receptor in inflammation and impairment of esophageal mucosal integrity in a murine model of nonerosive reflux disease. Neurogastroenterol. Motil. 2018, 30, e13340. [Google Scholar] [CrossRef]
- Matsumoto, K.; Suenaga, M.; Mizutani, Y.; Matsui, K.; Yoshida, A.; Nakamoto, T.; Kato, S. Role of transient receptor potential vanilloid subtype 2 in lower oesophageal sphincter in rat acid reflux oesophagitis. J. Pharmacol. Sci. 2021, 146, 125–135. [Google Scholar] [CrossRef]
- Stokłosa, P.; Borgström, A.; Kappel, S.; Peinelt, C. TRP channels in digestive tract cancers. Int. J. Mol. Sci. 2020, 21, 1877. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shi, Y.; Ding, X.; He, Z.H.; Zhou, K.C.; Wang, Q.; Wang, Y.Z. Critical role of TRPC6 channels in G2 phase transition and the development of human oesophageal cancer. Gut 2009, 58, 1443–1450. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Xiao, Y.; Huang, M.; Shen, B.; Xue, H.; Wu, K. Effect of TRPM2-Mediated Calcium Signaling on Cell Proliferation and Apoptosis in Esophageal Squamous Cell Carcinoma. Technol. Cancer Res. Treat. 2021, 20, 153303382110452. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, S.; Shiozaki, A.; Ichikawa, D.; Hikami, S.; Kosuga, T.; Konishi, H.; Komatsu, S.; Fujiwara, H.; Okamoto, K.; Kishimoto, M.; et al. Transient receptor potential melastatin 7 as an independent prognostic factor in human esophageal squamous cell carcinoma. Anticancer Res. 2017, 37, 1161–1167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lan, X.; Zhao, J.; Song, C.; Yuan, Q.; Liu, X. TRPM8 facilitates proliferation and immune evasion of esophageal cancer cells. Biosci. Rep. 2019, 39, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Huang, R.; Wang, F.; Yang, Y.; Ma, W.; Lin, Z.; Cheng, N.; Long, Y.; Deng, S.; Li, Z. Recurrent activations of transient receptor potential vanilloid-1 and vanilloid-4 promote cellular proliferation and migration in esophageal squamous cell carcinoma cells. FEBS Open Bio 2019, 9, 206–225. [Google Scholar] [CrossRef] [Green Version]
- Kudou, M.; Shiozaki, A.; Yamazato, Y.; Katsurahara, K.; Kosuga, T.; Shoda, K.; Arita, T.; Konishi, H.; Komatsu, S.; Kubota, T.; et al. The expression and role of TRPV2 in esophageal squamous cell carcinoma. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Zhang, S.S.; Xie, X.; Wen, J.; Luo, K.J.; Liu, Q.W.; Yang, H.; Hu, Y.; Fu, J.H. TRPV6 plays a new role in predicting survival of patients with esophageal squamous cell carcinoma. Diagn. Pathol. 2016, 11, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Owsianik, G.; D’Hoedt, D.; Voets, T.; Nilius, B. Structure-function relationship of the TRP channel superfamily. Rev. Physiol. Biochem. Pharmacol. 2006, 156, 61–90. [Google Scholar]
- Liedtke, W.; Choe, Y.; Martí-Renom, M.A.; Bell, A.M.; Denis, C.S.; AndrejŠali; Hudspeth, A.J.; Friedman, J.M.; Heller, S. Vanilloid receptor-related osmotically activated channel (VR-OAC), a candidate vertebrate osmoreceptor. Cell 2000, 103, 525–535. [Google Scholar] [CrossRef] [Green Version]
- Strotmann, R.; Harteneck, C.; Nunnenmacher, K.; Schultz, G.; Plant, T.D. OTRPC4, a nonselective cation channel that confers sensitivity to extracellular osmolarity. Nat. Cell Biol. 2000, 2, 695–702. [Google Scholar] [CrossRef]
- Güler, A.D.; Lee, H.; Iida, T.; Shimizy, I.; Tominaga, M.; Caterina, M. Heat-Evoked Activation of the Ion Channel, TRPV4. J. Neurosci. 2002, 22, 6408–6414. [Google Scholar] [CrossRef] [PubMed]
- Moore, C.; Cevikbas, F.; Pasolli, H.A.; Chen, Y.; Kong, W.; Kempkes, C.; Parekh, P.; Lee, S.H.; Kontchou, N.-A.; Yeh, I.; et al. UVB radiation generates sunburn pain and affects skin by activating epidermal TRPV4 ion channels and triggering endothelin-1 signaling. Proc. Natl. Acad. Sci. USA 2013, 110, E3225–E3234. [Google Scholar] [CrossRef] [Green Version]
- Boudaka, A.; Al-Suleimani, M.; Al-Lawati, I.; Baomar, H.; Al-Siyabi, S. Downregulation of endothelial transient receptor potential vanilloid type 4 channel underlines impaired endothelial nitric oxide-mediated relaxation in the mesenteric arteries of hypertensive rats. Physiol. Res. 2019, 68, 219–231. [Google Scholar] [CrossRef] [PubMed]
- Kida, N.; Sokabe, T.; Kashio, M.; Haruna, K.; Mizuno, Y.; Suga, Y.; Nishikawa, K.; Kanamaru, A.; Hongo, M.; Oba, A.; et al. Importance of transient receptor potential vanilloid 4 (TRPV4) in epidermal barrier function in human skin keratinocytes. Pflugers Arch. Eur. J. Physiol. 2012, 463, 715–725. [Google Scholar] [CrossRef] [PubMed]
- Shibasaki, K.; Tominaga, M.; Ishizaki, Y. Hippocampal neuronal maturation triggers post-synaptic clustering of brain temperature-sensor TRPV4. Biochem. Biophys. Res. Commun. 2015, 458, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Shikano, M.; Ueda, T.; Kamiya, T.; Ishida, Y.; Yamada, T.; Mizushima, T.; Shimura, T.; Mizoshita, T.; Tanida, S.; Kataoka, H.; et al. Acid inhibits TRPV4-mediated Ca 2+ influx in mouse esophageal epithelial cells. Neurogastroenterol. Motil. 2011, 23, 1020. [Google Scholar] [CrossRef]
- Lawhorn, B.G.; Brnardic, E.J.; Behm, D.J. Recent advances in TRPV4 agonists and antagonists. Bioorganic Med. Chem. Lett. 2020, 30, 127022. [Google Scholar] [CrossRef] [PubMed]
- Kuebler, W.M.; Jordt, S.E.; Liedtke, W.B. Urgent reconsideration of lung edema as a preventable outcome in COVID-19: Inhibition of TRPV4 represents a promising and feasible approach. Am. J. Physiol. Lung Cell. Mol. Physiol. 2020, 318, L1239–L1243. [Google Scholar] [CrossRef]
- Rosenbaum, T.; Benítez-Angeles, M.; Sánchez-Hernández, R.; Morales-Lázaro, S.L.; Hiriart, M.; Morales-Buenrostro, L.E.; Torres-Quiroz, F. TRPV4: A Physio and Pathophysiologically Significant Ion Channel. Int. J. Mol. Sci. 2020, 21, 3837. [Google Scholar] [CrossRef]
- Lawhorn, B.G.; Brnardic, E.J.; Behm, D.J. TRPV4 antagonists: A patent review (2015–2020). Expert Opin. Ther. Pat. 2021, 31, 773–784. [Google Scholar] [CrossRef] [PubMed]
- Kittaka, H.; Yamanoi, Y.; Tominaga, M. Transient receptor potential vanilloid 4 (TRPV4) channel as a target of crotamiton and its bimodal effects. Pflugers Arch. Eur. J. Physiol. 2017, 469, 1313–1323. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.L.; Maloney, K.N.; Pothen, R.G.; Clardy, J.; Clapham, D.E. Bisandrographolide from Andrographis paniculata activates TRPV4 channels. J. Biol. Chem. 2006, 281, 29897–29904. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vriens, J. Modulation of the Ca2 Permeable Cation Channel TRPV4 by Cytochrome P450 Epoxygenases in Vascular Endothelium. Circ. Res. 2005, 97, 908–915. [Google Scholar] [CrossRef] [Green Version]
- Vriens, J.; Owsianik, G.; Janssens, A.; Voets, T.; Nilius, B. Determinants of 4α-phorbol sensitivity in transmembrane domains 3 and 4 of the cation channel TRPV4. J. Biol. Chem. 2007, 282, 12796–12803. [Google Scholar] [CrossRef] [Green Version]
- Watanabe, H.; Vriens, J.; Prenen, J.; Droogmans, G.; Voets, T.; Nillus, B. Anandamide and arachidonic acid use epoxyeicosatrienoic acids to activate TRPV4 channels. Nature 2003, 424, 434–438. [Google Scholar] [CrossRef]
- Bang, S.; Yoo, S.; Yang, T.J.; Cho, H.; Hwang, S.W. Nociceptive and pro-inflammatory effects of dimethylallyl pyrophosphate via TRPV4 activation. Br. J. Pharmacol. 2012, 166, 1433–1443. [Google Scholar] [CrossRef] [Green Version]
- Saghatelian, A.; McKinney, M.K.; Bandell, M.; Patapoutian, A.; Cravatt, B.F. A FAAH-regulated class of N-acyl taurines that activates TRP ion channels. Biochemistry 2006, 45, 9007–9015. [Google Scholar] [CrossRef]
- Ma, X.; He, D.; Ru, X.; Chen, Y.; Cai, Y.; Bruce, I.C.; Xia, Q.; Yao, X.; Jin, J. Apigenin, a plant-derived flavone, activates transient receptor potential vanilloid 4 cation channel. Br. J. Pharmacol. 2012, 166, 349–358. [Google Scholar] [CrossRef] [Green Version]
- Klausen, T.K.; Pagani, A.; Minassi, A.; Ech-Chahad, A.; Prenen, J.; Owsianik, G.; Hoffmann, E.K.; Pedersen, S.F.; Appendino, G.; Nilius, B. Modulation of the transient receptor potential vanilloid channel TRPV4 by 4α-phorbol esters: A structure-activity study. J. Med. Chem. 2009, 52, 2933–2939. [Google Scholar] [CrossRef]
- Vincent, F.; Acevedo, A.; Nguyen, M.T.; Dourado, M.; DeFalco, J.; Gustafson, A.; Spiro, P.; Emerling, D.E.; Kelly, M.G.; Duncton, M.A.J. Identification and characterization of novel TRPV4 modulators. Biochem. Biophys. Res. Commun. 2009, 389, 490–494. [Google Scholar] [CrossRef] [PubMed]
- Dias, F.C.; Alves, V.S.; Matias, D.O.; Figueiredo, C.P.; Miranda, A.L.P.; Passos, G.F.; Costa, R. The selective TRPV4 channel antagonist HC-067047 attenuates mechanical allodynia in diabetic mice. Eur. J. Pharmacol. 2019, 856, 172408. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Fang, Q.; Wang, Z.; Zhang, J.Y.; MacLeod, A.S.; Hall, R.P.; Liedtke, W.B. Transient receptor potential vanilloid 4 ion channel functions as a pruriceptor in epidermal keratinocytes to evoke histaminergic itch. J. Biol. Chem. 2016, 291, 10252–10262. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gradilone, S.A.; Masyuk, T.V.; Huang, B.Q.; Banales, J.M.; Lehmann, G.L.; Radtke, B.N.; Stroope, A.; Masyuk, A.I.; Splinter, P.L.; LaRusso, N.F. Activation of Trpv4 Reduces the Hyperproliferative Phenotype of Cystic Cholangiocytes From an Animal Model of ARPKD. Gastroenterology 2010, 139, 304–314.e2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, J.H.; Park, C.; Kim, S.J.; Kim, H.J.; Oh, G.S.; Shen, A.; So, H.S.; Park, R. Different uptake of gentamicin through TRPV1 and TRPV4 channels determines cochlear hair cell vulnerability. Exp. Mol. Med. 2013, 45, e12. [Google Scholar] [CrossRef] [Green Version]
- Nilius, B.; Vriens, J.; Prenen, J.; Droogmans, G.; Voets, T. TRPV4 calcium entry channel: A paradigm for gating diversity. Am. J. Physiol. Cell Physiol. 2004, 286, 195–205. [Google Scholar] [CrossRef]
- Neuhuber, W.L.; Raab, M.; Berthoud, H.R.; Wörl, J. Advances in Anatomy Embryology and Cell Biology. In Innervation of the Mammalian Esophagus; Springer: Berlin/Heidelberg, Germany, 2007; Volume 185, pp. 1–73. ISBN 3540292055. [Google Scholar]
- Dütsch, M.; Eichhorn, U.; Wörl, J.; Wank, M.; Berthoud, H.R.; Neuhuber, W.L. Vagal and spinal afferent innervation of the rat esophagus: A combined retrograde tracing and immunocytochemical study with special emphasis on calcium-binding proteins. J. Comp. Neurol. 1998, 398, 289–307. [Google Scholar] [CrossRef]
- Wank, M.; Neuhuber, W.L. Local differences in vagal afferent innervation of the rat esophagus are reflected by neurochemical differences at the level of the sensory ganglia and by different brainstem projections. J. Comp. Neurol. 2001, 435, 41–59. [Google Scholar] [CrossRef]
- Berthoud, H.R.; Patterson, L.M.; Neumann, F.; Neuhuber, W.L. Distribution and structure of vagal afferent intraganglionic laminar endings (IGLEs) in the rat gastrointestinal tract. Anat. Embryol. 1997, 195, 183–191. [Google Scholar] [CrossRef]
- Wang, Z.J.; Neuhuber, W.L. Intraganglionic laminar endings in the rat esophagus contain purinergic P2X2 and P2X3 receptor immunoreactivity. Anat. Embryol. 2003, 207, 363–371. [Google Scholar] [CrossRef]
- Kestler, C.; Neuhuber, W.L.; Raab, M. Distribution of P2X 3 receptor immunoreactivity in myenteric ganglia of the mouse esophagus. Histochem. Cell Biol. 2009, 131, 13–27. [Google Scholar] [CrossRef] [PubMed]
- Burnstock, G. The past, present and future of purine nucleotides as signalling molecules. Neuropharmacology 1997, 36, 1127–1139. [Google Scholar] [PubMed]
- Mochizuki, T.; Sokabe, T.; Araki, I.; Fujishita, K.; Shibasaki, K.; Uchida, K.; Naruse, K.; Koizumi, S.; Takeda, M.; Tominaga, M. The TRPV4 cation channel mediates stretch-evoked Ca2+ influx and ATP release in primary urothelial cell cultures. J. Biol. Chem. 2009, 284, 21257–21264. [Google Scholar] [CrossRef] [Green Version]
- Okada, S.F.; Nicholas, R.A.; Kreda, S.M.; Lazarowski, E.R.; Boucher, R.C. Physiological Regulation of ATP Release at the Apical Surface of Human Airway Epithelia. J. Biol. Chem. 2006, 281, 22992–23002. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McIlwrath, S.L.; Davis, B.M.; Bielefeldt, K. Deletion of P2X3 receptors blunts gastro-oesophageal sensation in mice. Neurogastroenterol. Motil. 2009. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Page, A.J.; Martin, C.M.; Blackshaw, L. A Vagal mechanoreceptors and chemoreceptors in mouse stomach and esophagus. J. Neurophysiol. 2002, 87, 2095–2103. [Google Scholar] [CrossRef] [Green Version]
- Mandadi, S.; Sokabe, T.; Shibasaki, K.; Katanosaka, K.; Mizuno, A.; Moqrich, A.; Patapoutian, A.; Fukumi-Tominaga, T.; Mizumura, K.; Tominaga, M. TRPV3 in keratinocytes transmits temperature information to sensory neurons via ATP. Pflugers Arch. Eur. J. Physiol. 2009, 458, 1093–1102. [Google Scholar] [CrossRef] [Green Version]
- Dos Santos-Silva, M.A.; Trajano, E.T.L.; Schanuel, F.S.; Monte-Alto-Costa, A. Heat delays skin wound healing in mice. Exp. Biol. Med. 2017, 242, 258–266. [Google Scholar] [CrossRef] [Green Version]
- Justet, C.; Chifflet, S.; Hernandez, J.A. Calcium Oscillatory Behavior and Its Possible Role during Wound Healing in Bovine Corneal Endothelial Cells in Culture. Biomed Res. Int. 2019, 2019, 1–16. [Google Scholar] [CrossRef]
- Borea, P.A.; Gessi, S.; Merighi, S.; Vincenzi, F.; Varani, K. Pharmacology of adenosine receptors: The state of the art. Physiol. Rev. 2018, 98, 1591–1625. [Google Scholar] [CrossRef]
- Zimmermann, H.; Zebisch, M.; Sträter, N. Cellular function and molecular structure of ecto-nucleotidases. Purinergic Signal. 2012, 8, 437–502. [Google Scholar] [CrossRef] [Green Version]
- Robson, S.C.; Sévigny, J.; Zimmermann, H. The E-NTPDase family of ectonucleotidases: Structure function relationships and pathophysiological significance. Purinergic Signal. 2006, 2, 409–430. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burnstock, G. Purines and Purinoceptors: Molecular Biology Overview. In Reference Module in Biomedical Sciences; Elsevier: Amsterdam, The Netherlands, 2014; pp. 1253–1262. ISBN 9780080450469. [Google Scholar]
- Armstrong, D.; Bennett, J.R.; Blum, A.L.; Dent, J.; De Dombal, F.T.; Galmiche, P.; Lundell, L.; Margulies, M.; Richter, J.E.; Spechler, S.J.; et al. The endoscopic assessment of esophagitis: A progress report on observer agreement. Gastroenterology 1996, 111, 85–92. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suzuki, N.; Mihara, H.; Nishizono, H.; Tominaga, M.; Sugiyama, T. Protease-Activated Receptor-2 Up-Regulates Transient Receptor Potential Vanilloid 4 Function in Mouse Esophageal Keratinocyte. Dig. Dis. Sci. 2015, 60, 3570–3578. [Google Scholar] [CrossRef] [PubMed]
- Mihara, H.; Boudaka, A.; Tominaga, M.; Sugiyama, T. Transient Receptor Potential Vanilloid 4 Regulation of Adenosine Triphosphate Release by the Adenosine Triphosphate Transporter Vesicular Nucleotide Transporter, a Novel Therapeutic Target for Gastrointestinal Baroreception and Chronic Inflammation. Digestion 2019, 101, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Furuta, G.T.; Katzka, D.A. Eosinophilic Esophagitis Definition and Differential Diagnosis. N. Engl. J. Med. 2016, 373, 1640–1648. [Google Scholar] [CrossRef] [Green Version]
- Spechler, S.J.; Souza, R.F. Barrett’s Esophagus. N. Engl. J. Med. 2014, 371, 836–845. [Google Scholar] [CrossRef]
- Yu, S.; Huang, S.; Ding, Y.; Wang, W.; Wang, A.; Lu, Y. Transient receptor potential ion-channel subfamily V member 4: A potential target for cancer treatment. Cell Death Dis. 2019, 10. [Google Scholar] [CrossRef] [Green Version]
- Fusi, C.; Materazzi, S.; Minocci, D.; Maio, V.; Oranges, T.; Massi, D.; Nassini, R. Transient receptor potential vanilloid 4 (TRPV4) is downregulated in keratinocytes in human non-melanoma skin cancer. J. Investig. Dermatol. 2014, 134, 2408–2417. [Google Scholar] [CrossRef] [Green Version]
- Olivan-Viguera, A.; Garcia-Otin, A.L.; Lozano-Gerona, J.; Abarca-Lachen, E.; Garcia-Malinis, A.J.; Hamilton, K.L.; Gilaberte, Y.; Pueyo, E.; Köhler, R. Pharmacological activation of TRPV4 produces immediate cell damage and induction of apoptosis in human melanoma cells and HaCaT keratinocytes. PLoS ONE 2018, 13, 1–19. [Google Scholar] [CrossRef]
- Ohsaki, A.; Tanuma, S.; Tsukimoto, M. TRPV4 Channel-Regulated ATP Release Contributes to γ-Irradiation-Induced Production of IL-6 and IL-8 in Epidermal Keratinocytes. Biol. Pharm. Bull. 2018, 41, 1620–1626. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| TRP Channel | Localization | Functional Role | Species | Reference |
|---|---|---|---|---|
| TRPA1 | Vagal sensory neurons and afferent nerves | Mediate long-lasting mechanical hypersensitivity | Guinea pig | [35,36,37] |
| TRPM8 | Jugular vagal C fibers | Esophageal sensation and nociception Cold sensation | Guinea pig Human | [39,40] |
| TRPV1 | Sensory neurons and afferent nerve fibers | Mediates capsaicin-induced heartburn and esophageal sensitivity | Guinea pig, Human, Mouse, Rat | [13,41] |
| Esophageal keratinocytes | Mediates IL8 production and induces intracellular production of reactive oxygen species | Human, Mouse | [42,43] | |
| TRPV2 | Nitrergic myenteric neurons | Possible modulation of esophageal motility via myenteric co-innervation of vagal efferent fibers | Mouse | [21,44] |
| TRPV4 | Esophageal keratinocytes | Mediates mechanosensation via ATP release Delays in vitro wound healing by contributing to increases in levels of adenosine, derived from TRPV4-mediated ATP release | Mouse | [12,45] |
| TRPV6 | Esophageal keratinocytes | Putative role in mediating cell survival and programmed cell death | Human, Mouse | [46] |
| Name | Selectivity | In Vivo/Route/Species | Reference |
|---|---|---|---|
| Agonist | |||
| GSK1016790A | Non-selective | +(IV, SC) mice | [45,73] |
| 4αPDD | Non- selective | In vivo | [45] |
| 4αLPDD | Non- selective | [45] | |
| 4αPD | Non- selective | [74] | |
| Phorbol 12 myristate 13-acetate | Non-selective | [45] | |
| 5,6-epoxyeicosatrienoic acids (5,6-EET) | Non-selective | [75,76,77] | |
| Dimethylallyl pyrophosphate (DMAPP) | Non-selective | +(intraplantar) mice | [78] |
| Bisandrographolide A (BAA) | Non-selective | [74] | |
| N-arachidonoyl taurine | Non-selective | [79] | |
| Apigenin | No evidence | [80] | |
| Cannabidivarin, Tetrahydrocannabidivarin | Non-selective | [45] | |
| RN-1747 | Non-selective | [81] | |
| Antagonist | |||
| HC-067047 | Selective | +(SC) mice | [82,83] |
| Citral | Selective | [82] | |
| RN-1734 | Selective | [82] | |
| GSK205 | Selective | +(topical) mice | [82,84] |
| GSK2193874 | Non-selective | +(IV, IP) mice and rats | [82,85] |
| Ruthenium red (RR) | Non-selective | [45,86,87] | |
| Butamben | Non-selective | [82] | |
| Capsazepine | Non-selective TRPV | [82] | |
| Gd3+ | Non-selective TRPV | [87] | |
| La3+ | Non-selective TRPV | [87] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boudaka, A.; Tominaga, M. Physiological and Pathological Significance of Esophageal TRP Channels: Special Focus on TRPV4 in Esophageal Epithelial Cells. Int. J. Mol. Sci. 2022, 23, 4550. https://doi.org/10.3390/ijms23094550
Boudaka A, Tominaga M. Physiological and Pathological Significance of Esophageal TRP Channels: Special Focus on TRPV4 in Esophageal Epithelial Cells. International Journal of Molecular Sciences. 2022; 23(9):4550. https://doi.org/10.3390/ijms23094550
Chicago/Turabian StyleBoudaka, Ammar, and Makoto Tominaga. 2022. "Physiological and Pathological Significance of Esophageal TRP Channels: Special Focus on TRPV4 in Esophageal Epithelial Cells" International Journal of Molecular Sciences 23, no. 9: 4550. https://doi.org/10.3390/ijms23094550







