Mesenchymal Stem Cells and Extracellular Vesicles Derived from Canine Adipose Tissue Ameliorates Inflammation, Skin Barrier Function and Pruritus by Reducing JAK/STAT Signaling in Atopic Dermatitis
Abstract
:1. Introduction
2. Results
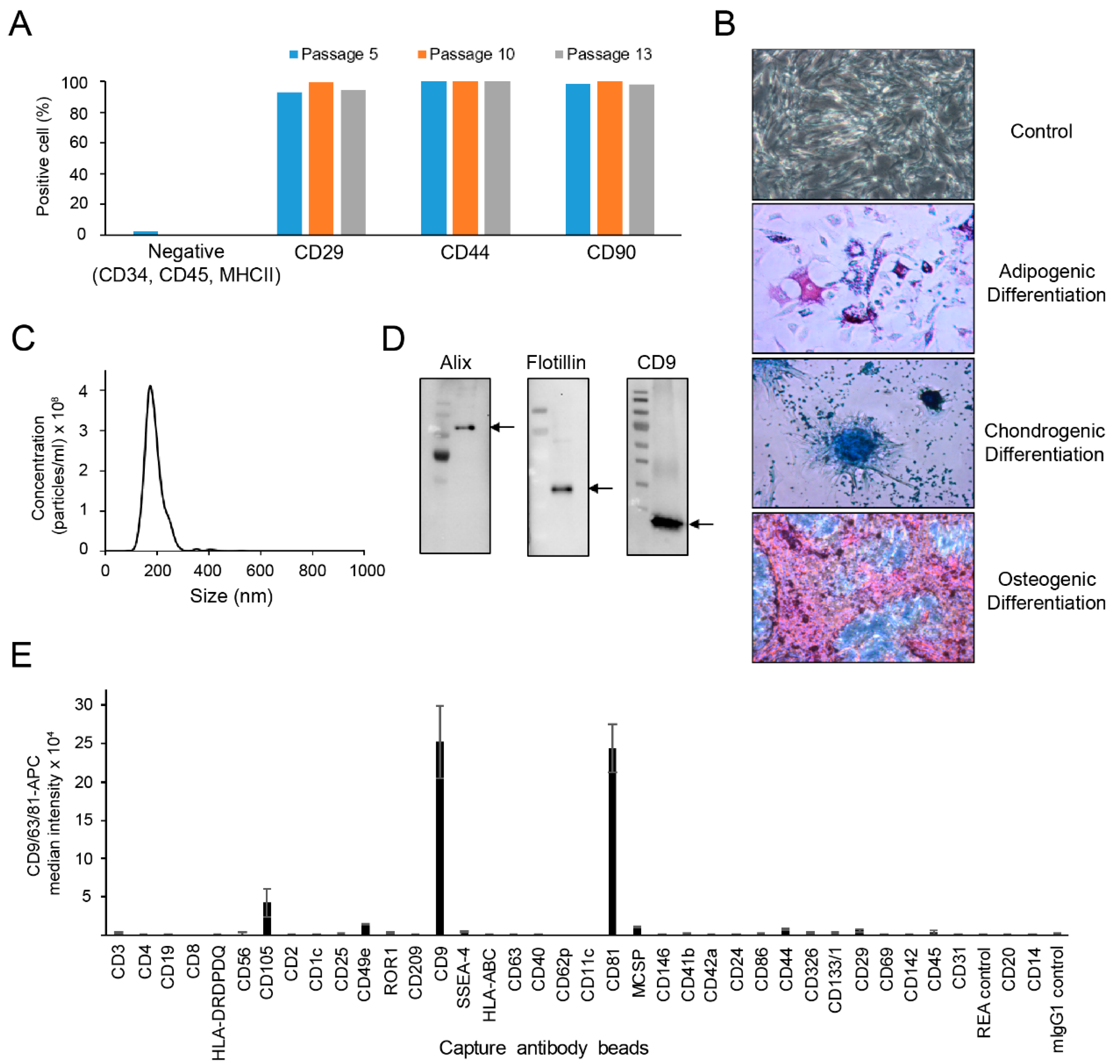
2.1. Characterization of cASCs and cASC-EVs
2.2. cASCs and cASC-EVs Alleviate Atopic Dermatitis Induced by DNCB Treatment
2.3. cASCs and cASC-EVs Ameliorated Skin Inflammation in DNCB-Induced Atopic Dermatitis Mice
2.4. cASCs and cASC-EVs Promote Skin Barrier Repair in DNCB-Induced Atopic Dermatitis Mice
2.5. cASCs and cASC-EVs Suppressed IL-31-Mediated Pruritus and Phosphorylated STATs in DNCB-Induced Atopic Dermatitis Mice
3. Discussion
4. Materials and Methods
4.1. Isolation, Culture, and Characterization of cASC
4.2. Isolation and Characterization of cASC-EV
4.3. Animal Care and Induction of AD Model
4.4. Measurement of Dermatitis Score and Epidermal Thickness
4.5. Measurement of Total Serum IgE and Inflammatory Cytokines
4.6. Histological Examination
4.7. RNA Extraction and Real-Time PCR
4.8. Western Blot Analysis
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Anjos-Afonso, F.; Siapati, E.K.; Bonnet, D. In vivo contribution of murine mesenchymal stem cells into multiple cell-types under minimal damage conditions. J. Cell Sci. 2004, 117, 5655–5664. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murphy, M.B.; Moncivais, K.; Caplan, A.I. Mesenchymal stem cells: Environmentally responsive therapeutics for regenerative medicine. Exp. Mol. Med. 2013, 45, e54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kapur, S.K.; Katz, A.J. Review of the adipose derived stem cell secretome. Biochimie 2013, 95, 2222–2228. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.W.; Kang, K.-S.; Koo, H.C.; Park, J.R.; Choi, E.W.; Park, Y.H. Soluble factors–mediated immunomodulatory effects of canine adipose tissue–derived mesenchymal stem cells. Stem Cells Dev. 2008, 17, 681–694. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carrade, D.D.; Borjesson, D.L. Immunomodulation by mesenchymal stem cells in veterinary species. Comp. Med. 2013, 63, 207–217. [Google Scholar]
- Zappia, E.; Casazza, S.; Pedemonte, E.; Benvenuto, F.; Bonanni, I.; Gerdoni, E.; Giunti, D.; Ceravolo, A.; Cazzanti, F.; Frassoni, F. Mesenchymal stem cells ameliorate experimental autoimmune encephalomyelitis inducing T-cell anergy. Blood 2005, 106, 1755–1761. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ohnishi, S.; Yanagawa, B.; Tanaka, K.; Miyahara, Y.; Obata, H.; Kataoka, M.; Kodama, M.; Ishibashi-Ueda, H.; Kangawa, K.; Kitamura, S. Transplantation of mesenchymal stem cells attenuates myocardial injury and dysfunction in a rat model of acute myocarditis. J. Mol. Cell. Cardiol. 2007, 42, 88–97. [Google Scholar] [CrossRef]
- Gonzalez-Rey, E.; Anderson, P.; González, M.A.; Rico, L.; Büscher, D.; Delgado, M. Human adult stem cells derived from adipose tissue protect against experimental colitis and sepsis. Gut 2009, 58, 929–939. [Google Scholar] [CrossRef] [PubMed]
- Kunter, U.; Rong, S.; Djuric, Z.; Boor, P.; Müller-Newen, G.; Yu, D.; Floege, J. Transplanted mesenchymal stem cells accelerate glomerular healing in experimental glomerulonephritis. J. Am. Soc. Nephrol. 2006, 17, 2202–2212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aggarwal, S.; Pittenger, M.F. Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood 2005, 105, 1815–1822. [Google Scholar] [CrossRef] [Green Version]
- Batten, P.; Sarathchandra, P.; Antoniw, J.W.; Tay, S.S.; Lowdell, M.W.; Taylor, P.M.; Yacoub, M.H. Human mesenchymal stem cells induce T cell anergy and downregulate T cell allo-responses via the TH2 pathway: Relevance to tissue engineering human heart valves. Tissue Eng. 2006, 12, 2263–2273. [Google Scholar] [CrossRef] [PubMed]
- Le Blanc, K.; Tammik, C.; Rosendahl, K.; Zetterberg, E.; Ringdén, O. HLA expression and immunologic propertiesof differentiated and undifferentiated mesenchymal stem cells. Exp. Hematol. 2003, 31, 890–896. [Google Scholar] [CrossRef]
- Le Blanc, K.; Frassoni, F.; Ball, L.; Locatelli, F.; Roelofs, H.; Lewis, I.; Lanino, E.; Sundberg, B.; Bernardo, M.E.; Remberger, M. Mesenchymal stem cells for treatment of steroid-resistant, severe, acute graft-versus-host disease: A phase II study. Lancet 2008, 371, 1579–1586. [Google Scholar] [CrossRef]
- Ringdén, O.; Uzunel, M.; Rasmusson, I.; Remberger, M.; Sundberg, B.; Lönnies, H.; Marschall, H.-U.; Dlugosz, A.; Szakos, A.; Hassan, Z. Mesenchymal stem cells for treatment of therapy-resistant graft-versus-host disease. Transplantation 2006, 81, 1390–1397. [Google Scholar] [CrossRef] [PubMed]
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): A position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 2018, 7, 1535750. [Google Scholar] [CrossRef] [Green Version]
- Février, B.; Raposo, G. Exosomes: Endosomal-derived vesicles shipping extracellular messages. Curr. Opin. Cell Biol. 2004, 16, 415–421. [Google Scholar] [CrossRef]
- Ratajczak, J.; Wysoczynski, M.; Hayek, F.; Janowska-Wieczorek, A.; Ratajczak, M. Membrane-derived microvesicles: Important and underappreciated mediators of cell-to-cell communication. Leukemia 2006, 20, 1487–1495. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Yin, Y.; Lai, R.C.; Tan, S.S.; Choo, A.B.H.; Lim, S.K. Mesenchymal stem cells secrete immunologically active exosomes. Stem Cells Dev. 2014, 23, 1233–1244. [Google Scholar] [CrossRef]
- Lai, P.; Chen, X.; Guo, L.; Wang, Y.; Liu, X.; Liu, Y.; Zhou, T.; Huang, T.; Geng, S.; Luo, C. A potent immunomodulatory role of exosomes derived from mesenchymal stromal cells in preventing cGVHD. J. Hematol. Oncol. 2018, 11, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Cosgrove, S.B.; Cleaver, D.M.; King, V.L.; Gilmer, A.R.; Daniels, A.E.; Wren, J.A.; Stegemann, M.R. Long-term compassionate use of oclacitinib in dogs with atopic and allergic skin disease: Safety, efficacy and quality of life. Vet. Dermatol. 2015, 26, 171-e35. [Google Scholar] [CrossRef]
- Pucheu-Haston, C.M. Atopic dermatitis in the domestic dog. Clin. Dermatol. 2016, 34, 299–303. [Google Scholar] [CrossRef] [PubMed]
- Jassies-van der Lee, A. T Cells and Immunomodulation in Canine Atopic Dermatitis; Utrecht University: Utrecht, The Netherlands, 2014. [Google Scholar]
- Schlotter, Y.M.; Rutten, V.P.; Riemers, F.M.; Knol, E.F.; Willemse, T. Lesional skin in atopic dogs shows a mixed Type-1 and Type-2 immune responsiveness. Vet. Immunol. Immunopathol. 2011, 143, 20–26. [Google Scholar] [CrossRef]
- Roesner, L.M.; Werfel, T.; Heratizadeh, A. The adaptive immune system in atopic dermatitis and implications on therapy. Expert Rev. Clin. Immunol. 2016, 12, 787–796. [Google Scholar] [CrossRef]
- Majewska, A.; Gajewska, M.; Dembele, K.; Maciejewski, H.; Prostek, A.; Jank, M. Lymphocytic, cytokine and transcriptomic profiles in peripheral blood of dogs with atopic dermatitis. BMC Vet. Res. 2016, 12, 174. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ding, Y.; Shao, X.; Li, X.; Zhai, Y.; Zhang, Y.; Wang, S.; Fang, H. Identification of candidate genes in atopic dermatitis based on bioinformatic methods. Int. J. Dermatol. 2016, 55, 791–800. [Google Scholar] [CrossRef] [PubMed]
- Lourenço, A.M.; Schmidt, V.; Sao Braz, B.; Nobrega, D.; Nunes, T.; Duarte-Correia, J.H.; Matias, D.; Maruhashi, E.; Reme, C.A.; Nuttall, T. Efficacy of proactive long-term maintenance therapy of canine atopic dermatitis with 0.0584% hydrocortisone aceponate spray: A double-blind placebo controlled pilot study. Vet. Dermatol. 2016, 27, 88-e25. [Google Scholar] [CrossRef] [Green Version]
- Little, P.R.; King, V.L.; Davis, K.R.; Cosgrove, S.B.; Stegemann, M.R. A blinded, randomized clinical trial comparing the efficacy and safety of oclacitinib and ciclosporin for the control of atopic dermatitis in client-owned dogs. Vet. Dermatol. 2015, 26, 23-e28. [Google Scholar] [CrossRef]
- Kisiel, A.H.; McDuffee, L.A.; Masaoud, E.; Bailey, T.R.; Esparza Gonzalez, B.P.; Nino-Fong, R. Isolation, characterization, and in vitro proliferation of canine mesenchymal stem cells derived from bone marrow, adipose tissue, muscle, and periosteum. Am. J. Vet. Res. 2012, 73, 1305–1317. [Google Scholar] [CrossRef] [PubMed]
- Vieira, N.; Brandalise, V.; Zucconi, E.; Secco, M.; Strauss, B.; Zatz, M. Isolation, characterization, and differentiation potential of canine adipose-derived stem cells. Cell Transplant. 2010, 19, 279–289. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, H.S.; Lee, J.H.; Roh, K.H.; Jun, H.J.; Kang, K.S.; Kim, T.Y. Clinical trial of human umbilical cord blood-derived stem cells for the treatment of moderate-to-severe atopic dermatitis: Phase I/IIa studies. Stem Cells 2017, 35, 248–255. [Google Scholar] [CrossRef] [Green Version]
- Na, K.; Yoo, H.; Zhang, Y.; Choi, M.; Lee, K.; Yi, T.; Song, S.; Jeon, M. Bone marrow-derived clonal mesenchymal stem cells inhibit ovalbumin-induced atopic dermatitis. Cell Death Dis. 2014, 5, e1345. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yi, Y.W.; Lee, J.H.; Kim, S.-Y.; Pack, C.-G.; Ha, D.H.; Park, S.R.; Youn, J.; Cho, B.S. Advances in analysis of biodistribution of exosomes by molecular imaging. Int. J. Mol. Sci. 2020, 21, 665. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Monroe, E.W.; Daly, A.F.; Shalhoub, R.F. Appraisal of the validity of histamine-induced wheal and flare to predict the clinical efficacy of antihistamines. J. Allergy Clin. Immunol. 1997, 99, S798–S806. [Google Scholar] [CrossRef]
- Bandell, M.; Story, G.M.; Hwang, S.W.; Viswanath, V.; Eid, S.R.; Petrus, M.J.; Earley, T.J.; Patapoutian, A. Noxious cold ion channel TRPA1 is activated by pungent compounds and bradykinin. Neuron 2004, 41, 849–857. [Google Scholar] [CrossRef] [Green Version]
- Oh, M.-H.; Oh, S.Y.; Lu, J.; Lou, H.; Myers, A.C.; Zhu, Z.; Zheng, T. TRPA1-dependent pruritus in IL-13–induced chronic atopic dermatitis. J. Immunol. 2013, 191, 5371–5382. [Google Scholar] [CrossRef] [Green Version]
- Cevikbas, F.; Wang, X.; Akiyama, T.; Kempkes, C.; Savinko, T.; Antal, A.; Kukova, G.; Buhl, T.; Ikoma, A.; Buddenkotte, J. A sensory neuron–expressed IL-31 receptor mediates T helper cell–dependent itch: Involvement of TRPV1 and TRPA1. J. Allergy Clin. Immunol. 2014, 133, 448–460.e447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dambacher, J.; Beigel, F.; Seiderer, J.; Haller, D.; Göke, B.; Auernhammer, C.J.; Brand, S. Interleukin 31 mediates MAP kinase and STAT1/3 activation in intestinal epithelial cells and its expression is upregulated in inflammatory bowel disease. Gut 2007, 56, 1257–1265. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Shea, J.J.; Plenge, R. JAK and STAT signaling molecules in immunoregulation and immune-mediated disease. Immunity 2012, 36, 542–550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jee, M.; Im, Y.; Choi, J.; Kang, S. Compensation of cATSCs-derived TGF β 1 and IL10 expressions was effectively modulated atopic dermatitis. Cell Death Dis. 2013, 4, e497. [Google Scholar] [CrossRef] [Green Version]
- Kavanagh, H.; Mahon, B.P. Allogeneic mesenchymal stem cells prevent allergic airway inflammation by inducing murine regulatory T cells. Allergy 2011, 66, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Yun, J.W.; Shin, T.H.; Lee, S.H.; Lee, B.C.; Yu, K.R.; Seo, Y.; Lee, S.; Kang, T.W.; Choi, S.W. Human Umbilical Cord Blood Mesenchymal Stem Cell-Derived PGE 2 and TGF-β1 Alleviate Atopic Dermatitis by Reducing Mast Cell Degranulation. Stem Cells 2015, 33, 1254–1266. [Google Scholar] [CrossRef] [PubMed]
- Su, W.R.; Zhang, Q.Z.; Shi, S.H.; Nguyen, A.L.; Le, A.D. Human gingiva-derived mesenchymal stromal cells attenuate contact hypersensitivity via prostaglandin E2-dependent mechanisms. Stem Cells 2011, 29, 1849–1860. [Google Scholar] [CrossRef]
- Sun, Y.Q.; Deng, M.X.; He, J.; Zeng, Q.X.; Wen, W.; Wong, D.S.; Tse, H.F.; Xu, G.; Lian, Q.; Shi, J. Human pluripotent stem cell-derived mesenchymal stem cells prevent allergic airway inflammation in mice. Stem Cells 2012, 30, 2692–2699. [Google Scholar] [CrossRef] [Green Version]
- Guo, L.; Lai, P.; Wang, Y.; Huang, T.; Chen, X.; Luo, C.; Geng, S.; Huang, X.; Wu, S.; Ling, W. Extracellular vesicles from mesenchymal stem cells prevent contact hypersensitivity through the suppression of Tc1 and Th1 cells and expansion of regulatory T cells. Int. Immunopharmacol. 2019, 74, 105663. [Google Scholar] [CrossRef] [PubMed]
- Shin, K.-O.; Ha, D.H.; Kim, J.O.; Crumrine, D.A.; Meyer, J.M.; Wakefield, J.S.; Lee, Y.; Kim, B.; Kim, S.; Kim, H.-k. Exosomes from human adipose tissue-derived mesenchymal stem cells promote epidermal barrier repair by inducing de novo synthesis of ceramides in atopic dermatitis. Cells 2020, 9, 680. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hall, M.; Rosenkrantz, W.S.; Griffin, C.; Mendelsohn, C. Evaluation of the potential use of adipose-derived mesenchymal stromal cells in the treatment of canine atopic dermatitis: A pilot study. Vet. Ther. 2010, 11, E1–E14. [Google Scholar]
- Villatoro, A.J.; Hermida-Prieto, M.; Fernández, V.; Fariñas, F.; Alcoholado, C.; Rodríguez-García, M.I.; Mariñas-Pardo, L.; Becerra, J. Allogeneic adipose-derived mesenchymal stem cell therapy in dogs with refractory atopic dermatitis: Clinical efficacy and safety. Vet. Rec. 2018, 183, 654. [Google Scholar] [CrossRef] [PubMed]
- Tarpataki, N. Recent developments in canine atopic dermatitis: A review. Acta Vet. Hung. 2006, 54, 473–484. [Google Scholar] [CrossRef] [PubMed]
- Pucheu-Haston, C.M.; Bizikova, P.; Marsella, R.; Santoro, D.; Nuttall, T.; Eisenschenk, M.N. Lymphocytes, cytokines, chemokines and the T-helper 1–T-helper 2 balance in canine atopic dermatitis. Vet. Dermatol. 2015, 26, 124-e132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olivry, T.; Mayhew, D.; Paps, J.S.; Linder, K.E.; Peredo, C.; Rajpal, D.; Hofland, H.; Cote-Sierra, J. Early activation of Th2/Th22 inflammatory and pruritogenic pathways in acute canine atopic dermatitis skin lesions. J. Investig. Dermatol. 2016, 136, 1961–1969. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gonzales, A.J.; Fleck, T.J.; Humphrey, W.R.; Galvan, B.A.; Aleo, M.M.; Mahabir, S.P.; Tena, J.K.; Greenwood, K.G.; McCall, R.B. IL-31-induced pruritus in dogs: A novel experimental model to evaluate anti-pruritic effects of canine therapeutics. Vet. Dermatol. 2016, 27, 34-e10. [Google Scholar] [CrossRef] [PubMed]
- Morelli, P.; Gaspari, M.; Gabriele, C.; Dastoli, S.; Bennardo, L.; Pavel, A.B.; Patruno, C.; Del Duca, E.; Nistico, S.P. Proteomic analysis from skin swabs reveals a new set of proteins identifying skin impairment in atopic dermatitis. Exp. Derm. 2021, 30, 811–819. [Google Scholar] [CrossRef]
- Nettis, E.; Ferrucci, S.M.; Pellacani, G.; Di Leo, E.; Argenziano, G.; Foti, C.; Rongioletti, F.; Patruno, C.; Ortoncelli, M.; Macchia, L.; et al. Dupilumab in atopic dermatitis: Predictors of treatment outcome and time to response. J. Eur. Acad. Derm. Venereol. 2021, 35, e896–e898. [Google Scholar] [CrossRef] [PubMed]
- Potestio, L.; Napolitano, M.; Bennardo, L.; Fabbrocini, G.; Patruno, C. Atopic dermatitis exacerbation after COVID-19 vaccination in Dupilumab-treated patients. J. Eur. Acad. Derm. Venereol. 2022. [Google Scholar] [CrossRef]
- Arzi, B.; Clark, K.C.; Sundaram, A.; Spriet, M.; Verstraete, F.J.; Walker, N.J.; Loscar, M.R.; Fazel, N.; Murphy, W.J.; Vapniarsky, N. Therapeutic efficacy of fresh, allogeneic mesenchymal stem cells for severe refractory feline chronic gingivostomatitis. Stem Cells Transl. Med. 2017, 6, 1710–1722. [Google Scholar] [CrossRef] [PubMed]
- Leibacher, J.; Henschler, R. Biodistribution, migration and homing of systemically applied mesenchymal stem/stromal cells. Stem Cell Res. Ther. 2016, 7, 7. [Google Scholar] [CrossRef] [Green Version]
- Ra, J.C.; Kang, S.K.; Shin, I.S.; Park, H.G.; Joo, S.A.; Kim, J.G.; Kang, B.-C.; Lee, Y.S.; Nakama, K.; Piao, M. Stem cell treatment for patients with autoimmune disease by systemic infusion of culture-expanded autologous adipose tissue derived mesenchymal stem cells. J. Transl. Med. 2011, 9, 181. [Google Scholar] [CrossRef] [Green Version]
- Furue, M. Regulation of Filaggrin, Loricrin, and Involucrin by IL-4, IL-13, IL-17A, IL-22, AHR, and NRF2: Pathogenic Implications in Atopic Dermatitis. Int. J. Mol. Sci. 2020, 21, 5382. [Google Scholar] [CrossRef]
- Kanwal, S.; Singh, S.K.; Soman, S.P.; Choudhury, S.; Kumari, P.; Ram, P.K.; Garg, S.K. Expression of barrier proteins in the skin lesions and inflammatory cytokines in peripheral blood mononuclear cells of atopic dogs. Sci. Rep. 2021, 11, 11418. [Google Scholar] [CrossRef]
- Furue, M.; Yamamura, K.; Kido-Nakahara, M.; Nakahara, T.; Fukui, Y. Emerging role of interleukin-31 and interleukin-31 receptor in pruritus in atopic dermatitis. Allergy 2018, 73, 29–36. [Google Scholar] [CrossRef] [Green Version]
- Ferretti, E.; Corcione, A.; Pistoia, V. The IL-31/IL-31 receptor axis: General features and role in tumor microenvironment. J. Leukoc. Biol. 2017, 102, 711–717. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ha, D.H.; Kim, H.-k.; Lee, J.; Kwon, H.H.; Park, G.-H.; Yang, S.H.; Jung, J.Y.; Choi, H.; Lee, J.H.; Sung, S. Mesenchymal Stem/Stromal Cell-Derived Exosomes for Immunomodulatory Therapeutics and Skin Regeneration. Cells 2020, 9, 1157. [Google Scholar] [CrossRef] [PubMed]
- Seo, Y.; Kim, H.-S.; Hong, I.-S. Stem cell-derived extracellular vesicles as immunomodulatory therapeutics. Stem Cells Int. 2019. [Google Scholar] [CrossRef] [PubMed]
- Shin, T.-H.; Kim, H.-S.; Choi, S.W.; Kang, K.-S. Mesenchymal stem cell therapy for inflammatory skin diseases: Clinical potential and mode of action. Int. J. Mol. Sci. 2017, 18, 244. [Google Scholar] [CrossRef] [Green Version]
- Pérez-Merino, E.; Usón-Casaús, J.; Duque-Carrasco, J.; Zaragoza-Bayle, C.; Mariñas-Pardo, L.; Hermida-Prieto, M.; Vilafranca-Compte, M.; Barrera-Chacón, R.; Gualtieri, M. Safety and efficacy of allogeneic adipose tissue-derived mesenchymal stem cells for treatment of dogs with inflammatory bowel disease: Endoscopic and histological outcomes. Vet. J. 2015, 206, 391–397. [Google Scholar] [CrossRef] [PubMed]
- Villatoro, A.J.; Fernández, V.; Claros, S.; Rico-Llanos, G.A.; Becerra, J.; Andrades, J.A. Use of adipose-derived mesenchymal stem cells in keratoconjunctivitis sicca in a canine model. BioMed Res. Int. 2015. [Google Scholar] [CrossRef]
- Cho, B.S.; Kim, J.O.; Ha, D.H.; Yi, Y.W. Exosomes derived from human adipose tissue-derived mesenchymal stem cells alleviate atopic dermatitis. Stem Cell Res. Ther. 2018, 9, 187. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jeong, H.; Shin, J.Y.; Kim, M.-J.; Na, J.; Ju, B.-G. Activation of Aryl Hydrocarbon Receptor Negatively Regulates Thymic Stromal Lymphopoietin Gene Expression via Protein Kinase Cδ-p300–NF-κB Pathway in Keratinocytes under Inflammatory Conditions. J. Investig. Dermatol. 2019, 139, 1098–1109. [Google Scholar] [CrossRef] [PubMed]




| Mean ± SD | Group 1 (Normal) | Group 2 (DNCB + Vehicle) | Group 3 (DNCB + EV) | Group 4 (DNCB + cASC) | p-Value * (Group 2–3) | p-Value * (Group 2–4) |
|---|---|---|---|---|---|---|
| Epidermal Thickness (Skin, μm) | 23.8 ± 3.2 | 130.0 ± 23.9 | 88.4 ± 13.5 | 83.3 ± 20.8 | 0.002 | 0.003 |
| Epidermal Thickness (Ear, μm) | 13.7 ± 3.5 | 76.5 ± 36.8 | 37.0 ± 14.7 | 24.4 ± 3.9 | 0.044 | 0.018 |
| Serum IgE (μg/mL) | 42.4 ± 10.9 | 867.5 ± 194.6 | 588.9 ± 95.8 | 437.6 ± 123.6 | 0.016 | 0.002 |
| Mast cell count (Skin, ea) | 12.7 ± 5.3 | 57.0 ± 8.2 | 32.7 ± 12.4 | 34.7 ± 11.9 | 0.003 | 0.004 |
| Mast cell count (Ear, ea) | 11.7 ± 4.2 | 31.3 ± 4.2 | 20.8 ± 2.4 | 20.5 ± 2.3 | <0.001 | <0.001 |
| IL-4 mRNA (fold) | 1.0 ± 0.5 | 2.5 ± 1.4 | 1.4 ± 0.1 | 1.7 ± 0.2 | 0.094 | 0.197 |
| IL-13 mRNA (fold) | 1.0 ± 0.6 | 2.8 ± 1.3 | 1.3 ± 0.3 | 1.9 ± 0.6 | 0.039 | 0.163 |
| IL-31 mRNA (fold) | 1.0 ± 0.4 | 4.2 ± 2.2 | 1.0 ± 0.5 | 1.5 ± 0.5 | 0.016 | 0.030 |
| TSLP mRNA (fold) | 1.0 ± 0.4 | 5.6 ± 2.3 | 1.3 ± 1.1 | 0.6 ± 0.4 | 0.002 | 0.003 |
| K1 mRNA (fold) | 1.0 ± 0.2 | 0.6 ± 0.2 | 0.9 ± 0.1 | 1.0 ± 0.2 | 0.005 | 0.002 |
| RANTES mRNA (fold) | 1.0 ± 0.5 | 3.6 ± 1.1 | 1.6 ± 1.0 | 1.2 ± 0.3 | 0.007 | 0.002 |
| TEWL (g/m2/h) | 11.8 ± 2.0 | 39.7 ± 6.9 | 26.5 ± 3.6 | 22.5 ± 3.5 | 0.002 | <0.001 |
| Hydration (A.U.) | 49.4 ± 5.6 | 33.5 ± 4.1 | 39.6 ± 7.9 | 41.4 ± 2.5 | 0.121 | 0.002 |
| FLG mRNA (fold) | 1 ± 0.1 | 0.5 ± 0.1 | 0.9 ± 0.3 | 0.6 ± 0.1 | 0.009 | 0.062 |
| LOR mRNA (fold) | 1 ± 0.3 | 0.6 ± 0.2 | 0.8 ± 0.1 | 0.8 ± 0.1 | 0.039 | 0.072 |
| INV mRNA (fold) | 1.0 ± 0.4 | 0.5 ± 0.2 | 0.7 ± 0.1 | 0.8 ± 0.2 | 0.068 | 0.063 |
| TRPA1 mRNA (fold) | 1.0 ± 0.4 | 2.1 ± 0.3 | 1.4 ± 0.1 | 1.5 ± 0.1 | 0.003 | 0.008 |
| IL-31RA mRNA (fold) | 1.0 ± 0.4 | 1.7 ± 0.4 | 1.3 ± 0.1 | 1.4 ± 0.1 | 0.038 | 0.128 |
| OSMR mRNA (fold) | 1.0 ± 0.6 | 3.3 ± 1.7 | 0.9 ± 0.5 | 0.6 ± 0.2 | 0.016 | 0.012 |
| p-STAT1/STAT1 (fold) | 1.0 ± 0.1 | 4.0 ± 0.4 | 0.7 ± 0.2 | 0.8 ± 0.2 | <0.001 | <0.001 |
| p-STAT3/STAT3 (fold) | 1.0 ± 0.1 | 3.1 ± 0.2 | 2.0 ± 0.5 | 1.3 ± 0.7 | 0.031 | 0.013 |
| Median [IQR] (Min–Max) | Group 1 (Normal) | Group 2 (DNCB + Vehicle) | Group 3 (DNCB + EV) | Group 4 (DNCB + cASC) | p-Value ** (Group 2–3) | p-Value ** (Group 2–4) |
|---|---|---|---|---|---|---|
| Dermatitis Score | 0 | 8.5 [8.0–9.0] (3.0–10.0) | 4.0 [4.0–4.0] (3.0–6.0) | 3 [3.0–3.8] (3.0–4.0) | 0.040 | 0.019 |
| TARC mRNA (fold) | 0.9 [0.8–1.2] (0.5–1.7) | 5.0 [3.4–5.4] (1.8–8.3) | 0.7 [0.6–1.4] (0.3–3.9) | 1.8 [1.7–1.8] (1.6–1.9) | 0.010 | 0.016 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, S.Y.; Yoon, T.H.; Na, J.; Yi, S.J.; Jin, Y.; Kim, M.; Oh, T.-H.; Chung, T.-W. Mesenchymal Stem Cells and Extracellular Vesicles Derived from Canine Adipose Tissue Ameliorates Inflammation, Skin Barrier Function and Pruritus by Reducing JAK/STAT Signaling in Atopic Dermatitis. Int. J. Mol. Sci. 2022, 23, 4868. https://doi.org/10.3390/ijms23094868
Kim SY, Yoon TH, Na J, Yi SJ, Jin Y, Kim M, Oh T-H, Chung T-W. Mesenchymal Stem Cells and Extracellular Vesicles Derived from Canine Adipose Tissue Ameliorates Inflammation, Skin Barrier Function and Pruritus by Reducing JAK/STAT Signaling in Atopic Dermatitis. International Journal of Molecular Sciences. 2022; 23(9):4868. https://doi.org/10.3390/ijms23094868
Chicago/Turabian StyleKim, Sung Youl, Tae Hong Yoon, Jungtae Na, Seong Joon Yi, Yunseok Jin, Minji Kim, Tae-Ho Oh, and Tae-Wook Chung. 2022. "Mesenchymal Stem Cells and Extracellular Vesicles Derived from Canine Adipose Tissue Ameliorates Inflammation, Skin Barrier Function and Pruritus by Reducing JAK/STAT Signaling in Atopic Dermatitis" International Journal of Molecular Sciences 23, no. 9: 4868. https://doi.org/10.3390/ijms23094868






