Expression of Autophagic and Inflammatory Markers in Normal Mucosa of Individuals with Colorectal Adenomas: A Cross Sectional Study among Italian Outpatients Undergoing Colonoscopy
Abstract
1. Introduction
2. Results
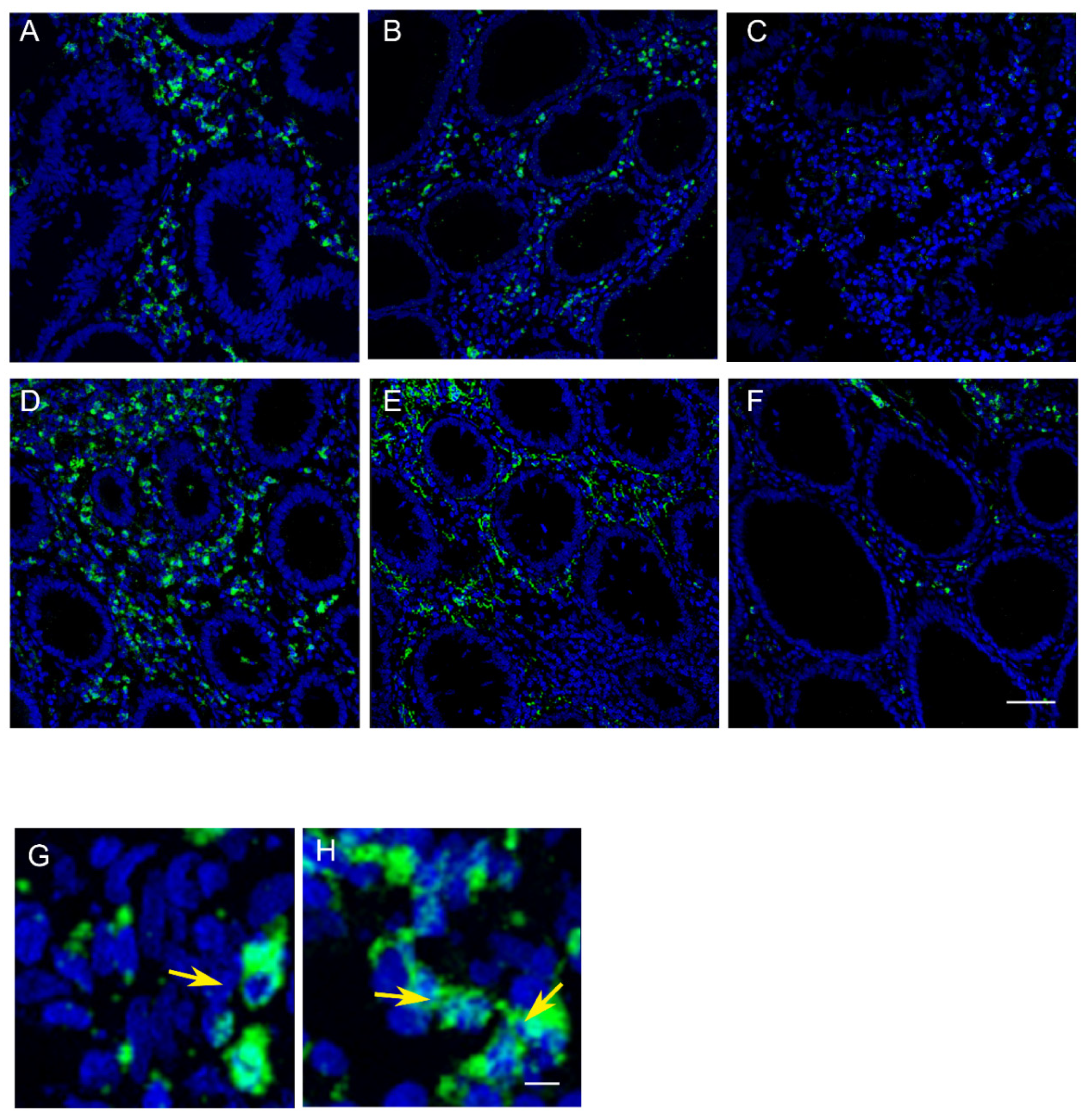
2.1. Patterns of Myeloperoxidase (MPO) Expression in Normal Mucosa Samples (Right Colon, Left Colon and Rectum)
2.2. MAP-LC3 Autophagic Activity in Normal Mucosa Samples (Right Colon, Left Colon, and Rectum)
2.3. Demographic, Anthropometric, and Clinical Features of Patients According to the Presence of Colorectal Adenomas
2.4. IFIS for MAP-LC3 and MPO Protein Expression in Patients with Colorectal Adenomas
2.5. IFIS for MAP-LC3 and MPO Protein Expression, and Clinical Characteristics
2.6. Multivariate Analysis
3. Discussion
4. Materials and Methods
4.1. Study Population
4.2. Statement of Ethics
4.3. Data Collection
4.4. Lipid Profile, C-Reactive Protein and IL-6
4.5. Evaluation of Immunofluorescence by Confocal Microscopy on Colonic Biopsies
4.6. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Grady, W.M.; Markowitz, S.D. The Molecular Pathogenesis of Colorectal Cancer and Its Potential Application to Colorectal. Cancer Screen. Dig. Dis. Sci. 2015, 60, 762–772. [Google Scholar] [CrossRef] [PubMed]
- Wolf, A.M.D.; Fontham, E.T.H.; Church, T.R.; Flowers, C.R.; Guerra, C.E.; LaMonte, S.J.; Etzioni, R.; McKenna, M.T.; Oeffinger, K.C.; Shih, Y.T.; et al. Colorectal cancer screening for average-risk adults: 2018 guideline update from the American Cancer Society. CA Cancer J. Clin. 2018, 68, 250–281. [Google Scholar] [CrossRef]
- Ladabaum, U.; Dominitz, J.A.; Kahi, C.; Schoen, R.E. Strategies for Colorectal Cancer Screening. Gastroenterology 2020, 158, 418–432. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Kalady, M.F.; Church, J. Young age of onset colorectal cancers. Int. J. Colorectal Dis. 2015, 30, 1653–1657. [Google Scholar] [CrossRef] [PubMed]
- Haslam, A.; Wagner Robb, S.; Hébert, J.R.; Huang, H.; Wirth, M.D.; Shivappa, N.; Ebell, M.H. The association between Dietary Inflammatory Index scores and the prevalence of colorectal adenoma. Public Health Nutr. 2017, 20, 1609–1616. [Google Scholar] [CrossRef] [PubMed]
- Bibbins-Domingo, K.; Grossman, D.C.; Curry, S.J.; Davidson, K.W.; Epling, J.W., Jr.; García, F.A.R.; Gillman, M.W.; Harper, D.M.; Kemper, A.R.; Krist, A.H.; et al. Screening for Colorectal Cancer: US Preventive Services Task Force Recommendation Statement. JAMA 2016, 315, 2564–2575. [Google Scholar] [CrossRef]
- Usher-Smith, J.A.; Walter, F.M.; Emery, J.D.; Win, A.K.; Griffin, S.J. Risk Prediction Models for Colorectal Cancer: A Systematic Review. Cancer Prev. Res. Phila. 2016, 9, 13–26. [Google Scholar] [CrossRef]
- Degett, T.H.; Christensen, J.; Dalton, S.O.; Bossen, K.; Frederiksen, K.; Iversen, L.H.; Gögenur, I.I. Prediction of the postoperative 90-day mortality after acute colorectal cancer surgery: Development and temporal validation of the ACORCA model. Int. J. Color. Dis. 2021, 36, 1873–1883. [Google Scholar] [CrossRef]
- Keum, N.; Giovannucci, E. Global burden of colorectal cancer: Emerging trends, risk factors and prevention strategies. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 713–732. [Google Scholar] [CrossRef]
- Patel, S.G.; Ahnen, D.J. Colorectal Cancer in the Young. Curr. Gastroenterol. Rep. 2018, 20, 15. [Google Scholar] [CrossRef] [PubMed]
- Tuomisto, A.E.; Mäkinen, M.J.; Väyrynen, J.P. Systemic inflammation in colorectal cancer: Underlying factors, effects, and prognostic significance. World J. Gastroenterol. 2019, 25, 4383–4404. [Google Scholar] [CrossRef] [PubMed]
- Notarnicola, M.; Caruso, M.G.; Tutino, V.; De Nunzio, V.; Gigante, I.; De Leonardis, G.; Veronese, N.; Rotolo, O.; Reddavide, R.; Stasi, E.; et al. Nutrition and lipidomic profile in colorectal cancers. Acta Biomed. 2018, 89, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Peltonen, R.; Hagström, J.; Tervahartiala, T.; Sorsa, T.; Haglund, C.; Isoniemi, H. High Expression of MMP-9 in Primary Tumors and High Preoperative MPO in Serum Predict Improved Prognosis in Colorectal Cancer with Operable Liver Metastases. Oncology 2021, 99, 144–160. [Google Scholar] [CrossRef]
- Xie, Q.; Liu, Y.; Li, X.T. The interaction mechanism between autophagy and apoptosis in colon cancer. Transl. Oncol. 2020, 13, 100871. [Google Scholar] [CrossRef]
- Singh, B.; Bhaskar, S. Methods for Detection of Autophagy in Mammalian Cells. Methods Mol. Biol. 2019, 2045, 245–258. [Google Scholar] [CrossRef]
- Koustas, E.; Sarantis, P.; Papavassiliou, A.G.; Karamouzis, M.V. Upgraded role of autophagy in colorectal carcinomas. World J. Gastrointest. Oncol. 2018, 10, 367–369. [Google Scholar] [CrossRef]
- He, S.; Zhao, Z.; Yang, Y.; O’Connell, D.; Zhang, X.; Oh, S.; Ma, B.; Lee, J.H.; Zhang, T.; Varghese, B.; et al. Truncating mutation in the autophagy gene UVRAG confers oncogenic properties and chemosensitivity in colorectal cancers. Nat. Commun. 2015, 6, 7839. [Google Scholar] [CrossRef]
- Yun, C.W.; Jeon, J.; Go, G.; Lee, J.H.; Lee, S.H. The Dual Role of Autophagy in Cancer Development and a Therapeutic Strategy for Cancer by Targeting Autophagy. Int. J. Mol. Sci. 2020, 22, 179. [Google Scholar] [CrossRef]
- Sharif, T.; Martell, E.; Dai, C.; Kennedy, B.E.; Murphy, P.; Clements, D.R.; Kim, Y.; Lee, P.W.; Gujar, S.A. Autophagic homeostasis is required for the pluripotency of cancer stem cells. Autophagy 2017, 13, 264–284. [Google Scholar] [CrossRef]
- Groulx, J.F.; Khalfaoui, T.; Benoit, Y.D.; Bernatchez, G.; Carrier, J.C.; Basora, N.; Beaulieu, J.F. Autophagy is active in normal colon mucosa. Autophagy 2012, 8, 893–902. [Google Scholar] [CrossRef] [PubMed]
- Ra, E.A.; Lee, T.A.; Won Kim, S.; Park, A.; Choi, H.J.; Jang, I.; Kang, S.; Hee Cheon, J.; Cho, J.W.; Eun Lee, J.; et al. TRIM31 promotes Atg5/Atg7-independent autophagy in intestinal cells. Nat. Commun. 2016, 7, 11726. [Google Scholar] [CrossRef] [PubMed]
- Yin, K.; Lee, J.; Liu, Z.; Kim, H.; Martin, D.R.; Wu, D.; Liu, M.; Xue, X. Mitophagy protein PINK1 suppresses colon tumor growth by metabolic reprogramming via p53 activation and reducing acetyl-CoA production. Cell Death Differ. 2021, 28, 2421–2435. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.; Chen, Y.; Cheng, D.; He, Z.; Shi, X.; Du, B.; Xi, X.; Gao, Y.; Guo, Y. YAP inhibits autophagy and promotes progression of colorectal cancer via upregulating Bcl-2 expression. Cell Death Dis. 2021, 12, 457. [Google Scholar] [CrossRef] [PubMed]
- Sena, P.; Mariani, F.; Mancini, S.; Benincasa, M.; Magnani, G.; Pedroni, M.; Palumbo, C.; Roncucci, L. Autophagy is upregulated during colorectal carcinogenesis, and in DNA microsatellite stable carcinomas. Oncol. Rep. 2015, 34, 3222–3230. [Google Scholar] [CrossRef][Green Version]
- Gupta, R.; Sinha, S.; Paul, R.N. The impact of microsatellite stability status in colorectal cancer. Curr. Probl. Cancer 2018, 42, 548–559. [Google Scholar] [CrossRef]
- Mehrvarz Sarshekeh, A.; Alshenaifi, J.; Roszik, J.; Manyam, G.C.; Advani, S.M.; Katkhuda, R.; Verma, A.; Lam, M.; Willis, J.; Shen, J.P.; et al. ARID1A Mutation May Define an Immunologically Active Subgroup in Patients with Microsatellite Stable Colorectal Cancer. Clin. Cancer Res. 2021, 27, 1663–1670. [Google Scholar] [CrossRef]
- Kim, K.; Kim, Y.W.; Shim, H.; Kim, B.R.; Kwon, H.Y. Differences in clinical features and oncologic outcomes between metastatic right and left colon cancer. J. BUON 2018, 23, 11–18. [Google Scholar]
- Petrelli, F.; Tomasello, G.; Borgonovo, K.; Ghidini, M.; Turati, L.; Dallera, P.; Passalacqua, R.; Sgroi, G.; Barni, S. Prognostic Survival Associated With Left-Sided vs Right-Sided Colon Cancer: A Systematic Review and Meta-analysis. JAMA Oncol. 2017, 3, 211–219. [Google Scholar] [CrossRef]
- Natsume, S.; Yamaguchi, T.; Takao, M.; Iijima, T.; Wakaume, R.; Takahashi, K.; Matsumoto, H.; Nakano, D.; Horiguchi, S.I.; Koizumi, K.; et al. Clinicopathological and molecular differences between right-sided and left-sided colorectal cancer in Japanese patients. Jpn. J. Clin. Oncol. 2018, 48, 609–618. [Google Scholar] [CrossRef]
- Zhu, L.; Ling, C.; Xu, T.; Zhang, J.; Zhang, Y.; Liu, Y.; Fang, C.; Yang, L.; Zhuang, W.; Wang, R.; et al. Clinicopathological Features and Survival of Signet-Ring Cell Carcinoma and Mucinous Adenocarcinoma of Right Colon, Left Colon, and Rectum. Pathol. Oncol. Res. 2021, 27, 1609800. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, M.; Greten, F.R. The inflammatory pathogenesis of colorectal cancer. Nat. Rev. Immunol. 2021, 21, 653–667. [Google Scholar] [CrossRef] [PubMed]
- Hickey, M.J. MPO and neutrophils: A magnetic attraction. Blood J. Am. Soc. Hematol. 2011, 117, 1103–1104. [Google Scholar] [CrossRef]
- Kirilovsky, A.; Marliot, F.; El Sissy, C.; Haicheur, N.; Galon, J.; Pages, F. Rational bases for the use of the Immunoscore in routine clinical settings as a prognostic and predictive biomarker in cancer patients. Int. Immunol. 2016, 28, 373–382. [Google Scholar] [CrossRef]
- Wikberg, M.L.; Ling, A.; Li, X.; Öberg, Å.; Edin, S.; Palmqvist, R. Neutrophil infiltration is a favorable prognostic factor in early stages of colon cancer. Hum. Pathol. 2017, 68, 193–202. [Google Scholar] [CrossRef]
- Tosti, N.; Cremonesi, E.; Governa, V.; Basso, C.; Kancherla, V.; Coto-Llerena, M.; Amicarella, F.; Weixler, B.; Däster, S.; Sconocchia, G.; et al. Infiltration by IL22-Producing T Cells Promotes Neutrophil Recruitment and Predicts Favorable Clinical Outcome in Human Colorectal Cancer. Cancer Immunol. Res. 2020, 8, 1452–1462. [Google Scholar] [CrossRef]
- Rao, H.L.; Chen, J.W.; Li, M.; Xiao, Y.B.; Fu, J.; Zeng, Y.X.; Cai, M.Y.; Dan, X. Increased intratumoral neutrophil in colorectal carcinomas correlates closely with malignant phenotype and predicts patients’ adverse prognosis. PLoS ONE 2012, 7, e30806. [Google Scholar] [CrossRef]
- Lin, Y.; Cheng, L.; Liu, Y.; Wang, Y.; Wang, Q.; Wang, H.L.; Shi, G.; Li, J.S.; Wang, Q.N.; Yang, Q.M.; et al. Intestinal epithelium-derived BATF3 promotes colitis-associated colon cancer through facilitating CXCL5-mediated neutrophils recruitment. Mucosal Immunol. 2021, 14, 187–198. [Google Scholar] [CrossRef]
- Galli, C.; Basso, D.; Plebani, M. CA 19-9: Handle with care. Clin. Chem. Lab. Med. 2013, 51, 1369–1383. [Google Scholar] [CrossRef]
- Tumay, V.; Guner, O.S. The utility and prognostic value of CA 19-9 and CEA serum markers in the long-term follow up of patients with colorectal cancer. A single-center experience over 13 years. Ann. Ital. Chir. 2020, 91, 494–503. [Google Scholar]
- Jelski, W.; Mroczko, B. Biochemical Markers of Colorectal Cancer-Present and Future. Cancer Manag. Res. 2020, 12, 4789–4797. [Google Scholar] [CrossRef] [PubMed]
- Łukaszewicz-Zając, M.; Mroczko, B. Circulating Biomarkers of Colorectal Cancer (CRC)-Their Utility in Diagnosis and Prognosis. J. Clin. Med. 2021, 10, 2391. [Google Scholar] [CrossRef] [PubMed]
- Thanikachalam, K.; Khan, G. Colorectal Cancer and Nutrition. Nutrients 2019, 11, 164. [Google Scholar] [CrossRef] [PubMed]
- Passarelli, M.N.; Newcomb, P.A. Blood Lipid Concentrations and Colorectal Adenomas: A Systematic Review and Meta-Analysis of Colonoscopy Studies in Asia, 2000–2014. Am. J. Epidemiol. 2016, 183, 691–700. [Google Scholar] [CrossRef]
- Tian, Y.; Wang, K.; Li, J.; Wang, J.; Wang, Z.; Fan, Y.; Ye, Y.; Ji, G.; Li, Y. The association between serum lipids and colorectal neoplasm: A systemic review and meta-analysis. Public Health Nutr. 2015, 18, 3355–3370. [Google Scholar] [CrossRef]
- Hanyuda, A.; Lee, D.H.; Ogino, S.; Wu, K.; Giovannucci, E.L. Long-term status of predicted body fat percentage, body mass index and other anthropometric factors with risk of colorectal carcinoma: Two large prospective cohort studies in the US. Int. J. Cancer 2020, 146, 2383–2393. [Google Scholar] [CrossRef]
- Nguyen, S.P.; Bent, S.; Chen, Y.-H.; Terdiman, J.P. Gender as a risk factor for advanced neoplasia and colorectal cancer: A systematic review and meta-analysis. Clin. Gastroenterol. Hepatol. 2009, 7, 676–681.e3. [Google Scholar] [CrossRef]
- Brenner, H.; Kloor, M.; Pox, C.P. Colorectal cancer. Lancet 2014, 383, 1490–1502. [Google Scholar] [CrossRef]
- Wong, M.C.S.; Huang, J.; Huang, J.L.W.; Pang, T.W.Y.; Choi, P.; Wang, J.; Chiang, J.I.; Jiang, J.Y. Global Prevalence of Colorectal Neoplasia: A Systematic Review and Meta-Analysis. Clin. Gastroenterol. Hepatol. 2020, 18, 553–561.e10. [Google Scholar] [CrossRef]
- Ku, M.S.; Chiu, S.Y.; Chien, K.L.; Lee, Y.C.; Chen, S.L.; Chen, C.D. Gender difference in metabolic syndrome and incident colorectal adenoma: A prospective observational study (KCIS No.42). Med. Baltim. 2021, 100, e26121. [Google Scholar] [CrossRef]
- Kim, H.M.; Kim, H.S. Gender-specific colorectal cancer: Epidemiologic difference and role of estrogen. Korean J. Gastroenterol. 2014, 63, 201–208. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Viggiani, M.T.; Polimeno, L.; Di Leo, A.; Barone, M. Phytoestrogens: Dietary Intake, Bioavailability, and Protective Mechanisms against Colorectal Neoproliferative Lesions. Nutrients 2019, 11, 1709. [Google Scholar] [CrossRef] [PubMed]
- Mancini, S.; Alboni, S.; Mattei, G.; Rioli, G.; Sena, P.; Marchi, M.; Sacchetti, A.; Boarino, V.; Roncucci, L.; Galeazzi, G.M.; et al. Preliminary results of a multidisciplinary Italian study adopting a Psycho-Neuro-Endocrine-Immunological (PNEI) approach to the study of colorectal adenomas. Acta Biomed. 2020, 92, e2021014. [Google Scholar] [CrossRef] [PubMed]
- Sena, P.; Mancini, S.; Benincasa, M.; Mariani, F.; Palumbo, C.; Roncucci, L. Metformin Induces Apoptosis and Alters Cellular Responses to Oxidative Stress in Ht29 Colon Cancer Cells: Preliminary Findings. Int. J. Mol. Sci. 2018, 19, 1478. [Google Scholar] [CrossRef]

| Groups | n | IFIS Mean (SD) | p |
|---|---|---|---|
| NMRC | 116 | 101.5 (10.9) | <0.001 |
| NMLC | 116 | 69.9 (13.9) | |
| NMR | 116 | 13.5 (9.7) |
| Groups | n | IFIS Mean (SD) | p |
|---|---|---|---|
| NMRC | 116 | 57.7 (27.9) | <0.001 |
| NMLC | 116 | 27.6 (19.8) | |
| NMR | 116 | 2.3 (4.9) |
| Continuous Variables | Size | Missing | Mean | Std Dev | Max | Min | |
|---|---|---|---|---|---|---|---|
| Age in years | 116 | 0 | 60.844 | 9.264 | 82 | 44 | |
| Weight Kg | 116 | 0 | 77.766 | 17.618 | 125 | 49 | |
| Height in meters | 116 | 0 | 1.678 | 0.0884 | 1.91 | 1.5 | |
| BMI Kg (m−2) | 116 | 0 | 27.416 | 4.583 | 38.1 | 18.8 | |
| Waist cm | 116 | 0 | 99.516 | 15.489 | 129 | 65 | |
| Hip cm | 116 | 0 | 102.547 | 9.791 | 120 | 81 | |
| w/h ratio | 116 | 0 | 0.968 | 0.0999 | 1.17 | 0.75 | |
| SBP mmHg | 116 | 0 | 146.719 | 18.457 | 180 | 110 | |
| DBP mmHg | 116 | 0 | 83.359 | 10.429 | 105 | 60 | |
| Glyc mg/dL | 116 | 0 | 93.594 | 18.281 | 194 | 64 | |
| Chol tot mg/dL | 116 | 0 | 196.75 | 19.948 | 243 | 156 | |
| HDL mg/dL | 116 | 1 | 49.873 | 10.723 | 77 | 32 | |
| LDL mg/dL | 116 | 1 | 120.349 | 16.805 | 154 | 84 | |
| TRGL mg/dL | 116 | 0 | 133.156 | 45.543 | 220 | 57 | |
| CRP mg/dL | 116 | 0 | 0.573 | 0.444 | 2.1 | 0.2 | |
| IL-6 | 116 | 0 | 4.215 | 3.831 | 27.649 | 0.84 | |
| Dichotomous Variables | Number | % | Variables | M/F(%) | |||
| Sex M/F | 64/52 | 51.6/48.4 | Presence of adenomas | 37/14(72.6/27.4) | |||
| Adenoma Y/N | 51/116 | 43.9/56.1 | |||||
| Smoke Y/N | 43/73 | 37.5/62.5 | |||||
| Alcohol Y/N | 77/39 | 67.2/32.8 | |||||
| Sedentary Y/N | 49/67 | 42.2/57.8 | |||||
| Dichotomous Variables | Number | % | Variables | M/F(%) | |||
| Sex M/F | 64/52 | 51.6/48.4 | Presence of adenomas | 37/14(72.6/27.4) | |||
| Adenoma Y/N | 51/116 | 43.9/56.1 | |||||
| Smoke Y/N | 43/73 | 37.5/62.5 | |||||
| Alcohol Y/N | 77/39 | 67.2/32.8 | |||||
| Sedentary Y/N | 49/67 | 42.2/57.8 | |||||
| Variables | Positive | Negative | p | |
|---|---|---|---|---|
| Adenoma Group | Cigarette smoking | 20 | 19 | 0.85 |
| Alcohol use | 44 | 17 | 0.28 | |
| Sedentary lifestyle | 30 | 28 | 0.25 | |
| No-Adenoma Group | Cigarette smoking | 24 | 21 | 0.85 |
| Alcohol use | 43 | 24 | 0.28 | |
| Sedentary lifestyle | 27 | 37 | 0.25 |
| Variable | Coefficient | Standard Error | p | Odds Ratio | 95% Confidence Interval |
|---|---|---|---|---|---|
| NMRC | 0.020 | 0.007 | 0.008 | 1.0202 | 1.0052–1.0354 |
| Constant | −1.409 | 0.493 | 0.004 | ||
| NMLC | 0.022 | 0.022 | 0.026 | 1.0225 | 1.0027–1.0427 |
| Constant | −0.869 | 0.435 | 0.012 |
| Variable | Coefficient | Standard Error | p |
|---|---|---|---|
| Constant | 15.547 | 15.490 | 0.318 |
| adenoma | 10.752 | 5.172 | 0.040 |
| Il-6 | 1.732 | 0.696 | 0.014 |
| LDL-Chol | −0.074 | 0.213 | 0.730 |
| Total Chol | 0.197 | 0.171 | 0.253 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sena, P.; Mancini, S.; Pedroni, M.; Reggiani Bonetti, L.; Carnevale, G.; Roncucci, L. Expression of Autophagic and Inflammatory Markers in Normal Mucosa of Individuals with Colorectal Adenomas: A Cross Sectional Study among Italian Outpatients Undergoing Colonoscopy. Int. J. Mol. Sci. 2022, 23, 5211. https://doi.org/10.3390/ijms23095211
Sena P, Mancini S, Pedroni M, Reggiani Bonetti L, Carnevale G, Roncucci L. Expression of Autophagic and Inflammatory Markers in Normal Mucosa of Individuals with Colorectal Adenomas: A Cross Sectional Study among Italian Outpatients Undergoing Colonoscopy. International Journal of Molecular Sciences. 2022; 23(9):5211. https://doi.org/10.3390/ijms23095211
Chicago/Turabian StyleSena, Paola, Stefano Mancini, Monica Pedroni, Luca Reggiani Bonetti, Gianluca Carnevale, and Luca Roncucci. 2022. "Expression of Autophagic and Inflammatory Markers in Normal Mucosa of Individuals with Colorectal Adenomas: A Cross Sectional Study among Italian Outpatients Undergoing Colonoscopy" International Journal of Molecular Sciences 23, no. 9: 5211. https://doi.org/10.3390/ijms23095211
APA StyleSena, P., Mancini, S., Pedroni, M., Reggiani Bonetti, L., Carnevale, G., & Roncucci, L. (2022). Expression of Autophagic and Inflammatory Markers in Normal Mucosa of Individuals with Colorectal Adenomas: A Cross Sectional Study among Italian Outpatients Undergoing Colonoscopy. International Journal of Molecular Sciences, 23(9), 5211. https://doi.org/10.3390/ijms23095211








