An Injectable Hydrogel Scaffold Loaded with Dual-Drug/Sustained-Release PLGA Microspheres for the Regulation of Macrophage Polarization in the Treatment of Intervertebral Disc Degeneration
Abstract
:1. Introduction
2. Results and Discussion
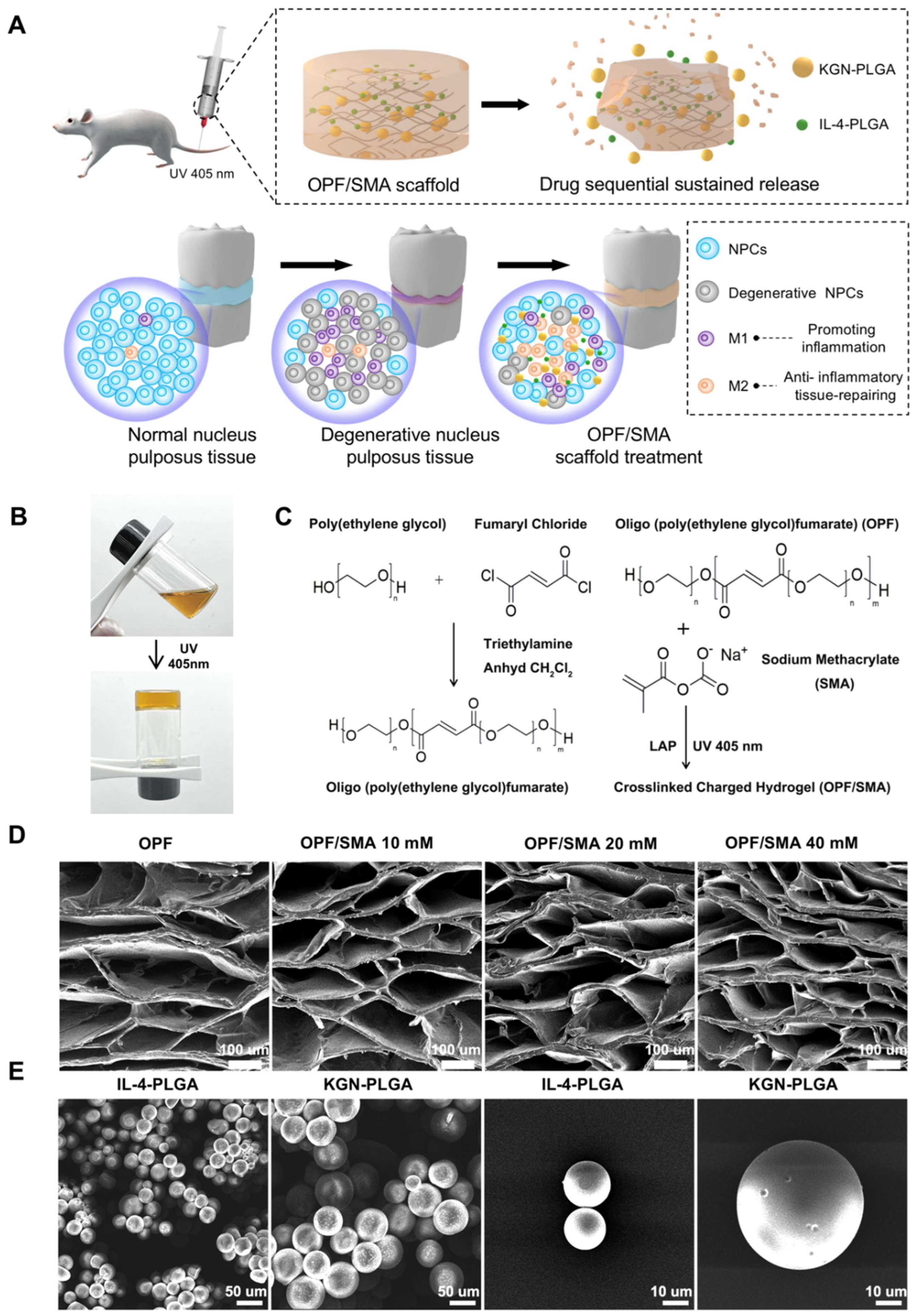
2.1. Fabrication and Characterization of OPF/SMA Composite Hydrogel Scaffold
2.2. The Cytocompatibility of the OPF/SMA Composite Hydrogel Scaffold
2.3. Regulation of Intervertebral Disc Immunity by the OPF/SMA Hydrogel Scaffold Loaded with IL-4-PLGA Microspheres
2.4. The Effect of Sequential Drug Release from the OPF/SMA Hydrogel Scaffold on the Repair of Degenerative Nucleus Pulposus Cells
2.5. The Reparative Effect of the OPF/SMA Hydrogel Scaffold on Degenerative Intervertebral Discs in Rats In Vivo
3. Materials and Methods
3.1. Reagents and Antibodies
3.2. Preparation of the OPF/SMA Hydrogel Scaffold
3.3. Preparation of the IL-4-PLGA and KGN-PLGA Microspheres
3.4. Scanning Electron Microscopy (SEM)
3.5. Fourier Transform Infrared (FTIR) Spectroscopy
3.6. Measurement of Pore Size, Porosity, and Particle Size
3.7. Mechanical Properties of the Hydrogel Scaffold
3.8. Swelling Rate of the Hydrogel Scaffold
3.9. Degradation of Hydrogel Scaffold In Vitro
3.10. Entrapment Efficiency (EE%) and In Vitro Sustained Release of PLGA Microspheres
3.11. Cell Culture
3.12. Cell Proliferation and Cell Viability Staining Experiments
3.13. Macrophage Polarization Assay
3.14. Realtime Fluorescence Quantitative PCR (qRT-PCR)
3.15. Western Blot (WB)
3.16. Animal Model
3.17. MRI and Micro-CT Evaluation
3.18. ELISA Assay
3.19. Macroscopic and Histological Assessments
3.20. Tissue Immunohistochemistry
3.21. Tissue Immunofluorescence
3.22. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jarrah, R.; Reardon, T.; Warner, T.; De Stefano, F.; Fiani, B. Spinal intervertebral disc regeneration versus repair: Cost, outcomes and future considerations. Regen. Med. 2022, 17, 337–340. [Google Scholar] [CrossRef] [PubMed]
- Isa, I.L.M.; Mokhtar, S.A.; Abbah, S.A.; Fauzi, M.B.; Devitt, A.; Pandit, A. Intervertebral Disc Degeneration: Biomaterials and Tissue Engineering Strategies toward Precision Medicine. Adv. Healthc. Mater. 2022, 11, e2102530. [Google Scholar] [CrossRef]
- Kasamkattil, J.; Gryadunova, A.; Martin, I.; Barbero, A.; Schären, S.; Krupkova, O.; Mehrkens, A. Spheroid-Based Tissue Engineering Strategies for Regeneration of the Intervertebral Disc. Int. J. Mol. Sci. 2022, 23, 2530. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, D.M.R.; Black, C.R.M.; Dawson, J.I.; Oreffo, R.O.C. A review of hydrogel use in fracture healing and bone regeneration: Hydrogel use in fracture healing and bone regeneration. J. Tissue Eng. Regen. Med. 2014, 10, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Schmocker, A.; Khoushabi, A.; Frauchiger, D.A.; Gantenbein, B.; Schizas, C.; Moser, C.; Bourban, P.-E.; Pioletti, D.P. A photopolymerized composite hydrogel and surgical implanting tool for a nucleus pulposus replacement. Biomaterials 2016, 88, 110–119. [Google Scholar] [CrossRef] [Green Version]
- Lam, J.; Lu, S.; Meretoja, V.V.; Tabata, Y.; Mikos, A.G.; Kasper, F.K. Generation of osteochondral tissue constructs with chondrogenically and osteogenically predifferentiated mesenchymal stem cells encapsulated in bilayered hydrogels. Acta Biomater. 2014, 10, 1112–1123. [Google Scholar] [CrossRef] [Green Version]
- Li, L.; Liu, X.; Gaihre, B.; Park, S.; Li, Y.; Terzic, A.; Lu, L. SDF-1α/OPF/BP Composites Enhance the Migrating and Osteogenic Abilities of Mesenchymal Stem Cells. Stem Cells Int. 2021, 2021, 1938819. [Google Scholar] [CrossRef]
- Frauchiger, D.; Tekari, A.; Wöltje, M.; Fortunato, G.; Benneker, L.; Gantenbein, B. A review of the application of reinforced hydrogels and silk as biomaterials for intervertebral disc repair. Eur. Cells Mater. 2017, 34, 271–290. [Google Scholar] [CrossRef]
- Chen, L.; McCrate, J.M.; Lee, J.C.-M.; Li, H. The role of surface charge on the uptake and biocompatibility of hydroxyapatite nanoparticles with osteoblast cells. Nanotechnology 2011, 22, 105708. [Google Scholar] [CrossRef] [Green Version]
- Iatridis, J.C.; Nicoll, S.B.; Michalek, A.J.; Walter, B.A.; Gupta, M.S. Role of biomechanics in intervertebral disc degeneration and regenerative therapies: What needs repairing in the disc and what are promising biomaterials for its repair? Spine J. 2013, 13, 243–262. [Google Scholar] [CrossRef]
- Tan, F.; Liu, J.; Liu, M.; Wang, J. Charge density is more important than charge polarity in enhancing osteoblast-like cell attachment on poly(ethylene glycol)-diacrylate hydrogel. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 76, 330–339. [Google Scholar] [CrossRef] [PubMed]
- Dadsetan, M.; Pumberger, M.; Casper, M.E.; Shogren, K.; Giuliani, M.; Ruesink, T.; Hefferan, T.E.; Currier, B.L.; Yaszemski, M.J. The effects of fixed electrical charge on chondrocyte behavior. Acta Biomater. 2011, 7, 2080–2090. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xue, X.; Hu, Y.; Wang, S.; Chen, X.; Jiang, Y.; Su, J. Fabrication of physical and chemical crosslinked hydrogels for bone tissue engineering. Bioact. Mater. 2022, 12, 327–339. [Google Scholar] [CrossRef] [PubMed]
- Fisher, L.E.; Kämmerling, L.; Alexander, M.R.; Ghaemmaghami, A.M. Immune-instructive materials as new tools for immunotherapy. Curr. Opin. Biotechnol. 2022, 74, 194–203. [Google Scholar] [CrossRef] [PubMed]
- Foster, D.S.; Marshall, C.D.; Gulati, G.S.; Chinta, M.S.; Nguyen, A.; Salhotra, A.; Jones, R.E.; Burcham, A.; Lerbs, T.; Cui, L.; et al. Elucidating the fundamental fibrotic processes driving abdominal adhesion formation. Nat. Commun. 2020, 11, 4061. [Google Scholar] [CrossRef] [PubMed]
- Ji, X.; Shao, H.; Li, X.; Ullah, M.W.; Luo, G.; Xu, Z.; Ma, L.; He, X.; Lei, Z.; Li, Q.; et al. Injectable immunomodulation-based porous chitosan microspheres/HPCH hydrogel composites as a controlled drug delivery system for osteochondral regeneration. Biomaterials 2022, 285, 121530. [Google Scholar] [CrossRef] [PubMed]
- Cha, B.; Shin, S.R.; Leijten, J.; Li, Y.; Singh, S.; Liu, J.C.; Annabi, N.; Abdi, R.; Dokmeci, M.R.; Vrana, N.E.; et al. Integrin-Mediated Interactions Control Macrophage Polarization in 3D Hydrogels. Adv. Healthc. Mater. 2017, 6, 1700289. [Google Scholar] [CrossRef] [Green Version]
- Bai, J.; Zhang, Y.; Fan, Q.; Xu, J.; Shan, H.; Gao, X.; Ma, Q.; Sheng, L.; Zheng, X.; Cheng, W.; et al. Reactive Oxygen Species-Scavenging Scaffold with Rapamycin for Treatment of Intervertebral Disk Degeneration. Adv. Healthc. Mater. 2020, 9, 1901186. [Google Scholar] [CrossRef]
- Ren, B.; Lu, J.; Li, M.; Zou, X.; Liu, Y.; Wang, C.; Wang, L. Anti-inflammatory effect of IL-1ra-loaded dextran/PLGA microspheres on Porphyromonas gingivalis lipopolysaccharide-stimulated macrophages in vitro and in vivo in a rat model of periodontitis. Biomed. Pharmacother. 2021, 134, 111171. [Google Scholar] [CrossRef]
- Wen, K.; Na, X.; Yuan, M.; Bazybek, N.; Li, X.; Wei, Y.; Ma, G. Preparation of novel ropivacaine hydrochloride-loaded PLGA microspheres based on post-loading mode and efficacy evaluation. Colloids Surf. B Biointerfaces 2022, 210, 112215. [Google Scholar] [CrossRef]
- Ochi, M.; Wan, B.; Bao, Q.; Burgess, D.J. Influence of PLGA molecular weight distribution on leuprolide release from microspheres. Int. J. Pharm. 2021, 599, 120450. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.; Xu, X.; Xu, Q.; Sun, Z.; Lv, Z.; Wu, R.; Yan, W.; Jiang, Q.; Shi, D. An Injectable Hydrogel Scaffold With Kartogenin-Encapsulated Nanoparticles for Porcine Cartilage Regeneration: A 12-Month Follow-up Study. Am. J. Sports Med. 2020, 48, 3233–3244. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.-J.; Chan, Y.-C.; Su, Z.-C.; Yeh, W.-L.; Lai, P.-L.; Chu, I.-M. Growth factor-loaded microspheres in mPEG-polypeptide hydrogel system for articular cartilage repair. J. Biomed. Mater. Res. Part A 2021, 109, 2516–2526. [Google Scholar] [CrossRef]
- Su, Y.; Liu, J.; Tan, S.; Liu, W.; Wang, R.; Chen, C. PLGA sustained-release microspheres loaded with an insoluble small-molecule drug: Microfluidic-based preparation, optimization, characterization, and evaluation in vitro and in vivo. Drug Deliv. 2022, 29, 1437–1446. [Google Scholar] [CrossRef] [PubMed]
- Naahidi, S.; Jafari, M.; Logan, M.; Wang, Y.; Yuan, Y.; Bae, H.; Dixon, B.; Chen, P. Biocompatibility of hydrogel-based scaffolds for tissue engineering applications. Biotechnol. Adv. 2017, 35, 530–544. [Google Scholar] [CrossRef]
- Witherel, C.E.; Sao, K.; Brisson, B.K.; Han, B.; Volk, S.W.; Petrie, R.J.; Han, L.; Spiller, K.L. Regulation of extracellular matrix assembly and structure by hybrid M1/M2 macrophages. Biomaterials 2021, 269, 120667. [Google Scholar] [CrossRef]
- Zhang, J.; Shi, H.; Zhang, N.; Hu, L.; Jing, W.; Pan, J. Interleukin-4-loaded hydrogel scaffold regulates macrophages polarization to promote bone mesenchymal stem cells osteogenic differentiation via TGF-β1/Smad pathway for repair of bone defect. Cell Prolif. 2020, 53, e12907. [Google Scholar] [CrossRef]
- Ji, X.; Lei, Z.; Yuan, M.; Zhu, H.; Yuan, X.; Liu, W.; Pu, H.; Jiang, J.; Zhang, Y.; Jiang, X.; et al. Cartilage repair mediated by thermosensitive photocrosslinkable TGFβ1-loaded GM-HPCH via immunomodulating macrophages, recruiting MSCs and promoting chondrogenesis. Theranostics 2020, 10, 2872–2887. [Google Scholar] [CrossRef]
- Xu, X.; Shi, D.; Shen, Y.; Xu, Z.; Dai, J.; Chen, D.; Teng, H.; Jiang, Q. Full-thickness cartilage defects are repaired via a microfracture technique and intraarticular injection of the small-molecule compound kartogenin. Arthritis Res. Ther. 2015, 17, 20. [Google Scholar] [CrossRef] [Green Version]
- Shi, D.; Xu, X.; Ye, Y.; Song, K.; Cheng, Y.; Di, J.; Hu, Q.; Li, J.; Ju, H.; Jiang, Q.; et al. Photo-Cross-Linked Scaffold with Kartogenin-Encapsulated Nanoparticles for Cartilage Regeneration. ACS Nano 2016, 10, 1292–1299. [Google Scholar] [CrossRef]
- Wang, S.-J.; Qin, J.-Z.; Zhang, T.-E.; Xia, C. Intra-articular Injection of Kartogenin-Incorporated Thermogel Enhancing Osteoarthritis Treatment. Front. Chem. 2019, 7, 677. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kwon, J.Y.; Lee, S.H.; Na, H.-S.; Jung, K.; Choi, J.; Cho, K.H.; Lee, C.-Y.; Kim, S.J.; Park, S.-H.; Shin, D.-Y.; et al. Kartogenin inhibits pain behavior, chondrocyte inflammation, and attenuates osteoarthritis progression in mice through induction of IL-10. Sci. Rep. 2018, 8, 13832. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zoetebier, B.; Schmitz, M.T.; Ito, K.; Karperien, M.; Tryfonidou, M.A.; Paez, J.I. Injectable Hydrogels for Articular Cartilage and Nucleus Pulposus Repair: Status Quo and Prospects. Tissue Eng. Part A 2022, 28, 478–499. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wang, D.; Luo, B.; Wang, D.; Jia, H.; Peng, P.; Shang, Q.; Mao, J.; Gao, C.; Peng, Y.; et al. Decoding the annulus fibrosus cell atlas by scRNA-seq to develop an inducible composite hydrogel: A novel strategy for disc reconstruction. Bioact. Mater. 2022, 14, 350–363. [Google Scholar] [CrossRef]
- Shen, J.; Chen, A.; Cai, Z.; Chen, Z.; Cao, R.; Liu, Z.; Li, Y.; Hao, J. Exhausted local lactate accumulation via injectable nanozyme-functionalized hydrogel microsphere for inflammation relief and tissue regeneration. Bioact. Mater. 2022, 12, 153–168. [Google Scholar] [CrossRef]
- Ligorio, C.; Vijayaraghavan, A.; Hoyland, J.A.; Saiani, A. Acidic and basic self-assembling peptide and peptide-graphene oxide hydrogels: Characterisation and effect on encapsulated nucleus pulposus cells. Acta Biomater. 2022, 143, 145–158. [Google Scholar] [CrossRef]
- Sivan, S.S.; Roberts, S.; Urban, J.P.G.; Menage, J.; Bramhill, J.; Campbell, D.; Franklin, V.J.; Lydon, F.; Merkher, Y.; Maroudas, A.; et al. Injectable hydrogels with high fixed charge density and swelling. pressure for nucleus pulposus repair: Biomimetic glycosaminoglycan analogues. Acta Biomater. 2014, 10, 1124–1133. [Google Scholar] [CrossRef]
- Dadsetan, M.; Liu, Z.; Pumberger, M.; Giraldo, C.V.; Ruesink, T.; Lu, L.; Yaszemski, M.J. A stimuli-responsive hydrogel for doxorubicin delivery. Biomaterials 2010, 31, 8051–8062. [Google Scholar] [CrossRef] [Green Version]
- Muir, V.G.; Qazi, T.H.; Shan, J.; Groll, J.; Burdick, J.A. Influence of Microgel Fabrication Technique on Granular Hydrogel Properties. ACS Biomater. Sci. Eng. 2021, 7, 4269–4281. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Lu, H.; Kawazoe, N.; Chen, G. Pore size effect of collagen scaffolds on cartilage regeneration. Acta Biomater. 2014, 10, 2005–2013. [Google Scholar] [CrossRef]
- Kim, H.Y.; Kim, H.N.; Lee, S.J.; Song, J.E.; Kwon, S.Y.; Chung, J.W.; Lee, D.; Khang, G. Effect of pore sizes of PLGA scaffolds on mechanical properties and cell behaviour for nucleus pulposus regeneration in vivo. J. Tissue Eng. Regen. Med. 2017, 11, 44–57. [Google Scholar] [CrossRef] [PubMed]
- Patterson, J.; Siew, R.; Herring, S.W.; Lin, A.S.P.; Guldberg, R.; Stayton, P.S. Hyaluronic acid hydrogels with controlled degradation properties for oriented bone regeneration. Biomaterials 2010, 31, 6772–6781. [Google Scholar] [CrossRef] [Green Version]
- Long, R.G.; Torre, O.M.; Hom, W.W.; Assael, D.J.; Iatridis, J.C. Design Requirements for Annulus Fibrosus Repair: Review of Forces, Displacements, and Material Properties of the Intervertebral Disk and a Summary of Candidate Hydrogels for Repair. J. Biomech. Eng. 2016, 138, 021007. [Google Scholar] [CrossRef]
- Wan, B.; Bao, Q.; Burgess, D.J. In vitro-in vivo correlation of PLGA microspheres: Effect of polymer source variation and temperature. J. Control. Release 2022, 347, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, Y.; Kokubo, Y.; Nakajima, H.; Honjoh, K.; Watanabe, S.; Matsumine, A. Distribution and Polarization of Hematogenous Macrophages Associated with the Progression of Intervertebral Disc Degeneration. Spine 2021, 47, E149–E158. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Yin, H.; Yan, Z.; Li, H.; Wu, J.; Wang, Y.; Wei, F.; Tian, G.; Ning, C.; Li, H.; et al. The immune microenvironment in cartilage injury and repair. Acta Biomater. 2022, 140, 23–42. [Google Scholar] [CrossRef]
- Gong, L.; Li, J.; Zhang, J.; Pan, Z.; Liu, Y.; Zhou, F.; Hong, Y.; Hu, Y.; Gu, Y.; Ouyang, H.; et al. An interleukin-4-loaded bi-layer 3D printed scaffold promotes osteochondral regeneration. Acta Biomater. 2020, 117, 246–260. [Google Scholar] [CrossRef]
- Liu, H.; Kang, H.; Song, C.; Lei, Z.; Li, L.; Guo, J.; Xu, Y.; Guan, H.; Fang, Z.; Li, F. Urolithin A Inhibits the Catabolic Effect of TNFα on Nucleus Pulposus Cell and Alleviates Intervertebral Disc Degeneration in vivo. Front. Pharmacol. 2018, 9, 1043. [Google Scholar] [CrossRef]
- Sun, Y.; Lyu, M.; Lu, Q.; Cheung, K.; Leung, V. Current Perspectives on Nucleus Pulposus Fibrosis in Disc Degeneration and Repair. Int. J. Mol. Sci. 2022, 23, 6612. [Google Scholar] [CrossRef]
- Wang, L.; He, T.; Liu, J.; Tai, J.; Wang, B.; Zhang, L.; Quan, Z. Revealing the Immune Infiltration Landscape and Identifying Diagnostic Biomarkers for Lumbar Disc Herniation. Front. Immunol. 2021, 12, 666355. [Google Scholar] [CrossRef]
- Kinard, L.A.; Kasper, F.K.; Mikos, A.G. Synthesis of oligo(poly(ethylene glycol) fumarate). Nat. Protoc. 2012, 7, 1219–1227. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, H.; Temenoff, J.S.; Holland, T.A.; Tabata, Y.; Mikos, A.G. Delivery of TGF-β1 and chondrocytes via injectable, biodegradable hydrogels for cartilage tissue engineering applications. Biomaterials 2005, 26, 7095–7103. [Google Scholar] [CrossRef] [PubMed]
- Maged, A.; Abdelbaset, R.; Mahmoud, A.A.; Elkasabgy, N.A. Merits and advances of microfluidics in the pharmaceutical field: Design technologies and future prospects. Drug Deliv. 2022, 29, 1549–1570. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Wei, K.; Tu, J.; Chen, Y.; He, Y.; Ding, Y.; Xu, H.; Bao, X.; Xie, H.; Fang, H.; et al. Reducing Inflammation and Vascular Invasion in Intervertebral Disc Degeneration via Cystathionine-γ-Lyase Inhibitory Effect on E-Selectin. Front. Cell Dev. Biol. 2021, 9, 741046. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Wang, H.; Chen, J.; Zhang, Y.; Mo, Q.; Zhang, P.; Wang, M.; Liu, H.; Bao, X.; Sun, Y.; et al. Silk-based hydrogel incorporated with metal-organic framework nanozymes for enhanced osteochondral regeneration. Bioact. Mater. 2023, 20, 221–242. [Google Scholar] [CrossRef] [PubMed]
- Issy, A.C.; Castania, V.; Castania, M.; Salmon, C.E.G.; Nogueira-Barbosa, M.H.; Bel, E.D.; Defino, H.L.A. Experimental model of intervertebral disc degeneration by needle puncture in Wistar rats. Braz. J. Med. Biol. Res. 2013, 46, 235–244. [Google Scholar] [CrossRef]
- Pan, Z.; Sun, H.; Xie, B.; Xia, D.; Zhang, X.; Yu, D.; Li, J.; Xu, Y.; Wang, Z.; Wu, Y.; et al. Therapeutic effects of gefitinib-encapsulated thermosensitive injectable hydrogel in intervertebral disc degeneration. Biomaterials 2018, 160, 56–68. [Google Scholar] [CrossRef] [PubMed]
- Norcross, J.P.; Lester, G.E.; Weinhold, P.; Dahners, L.E. An in vivo model of degenerative disc disease. J. Orthop. Res. 2003, 21, 183–188. [Google Scholar] [CrossRef]






| Entrapment Efficiency (EE%) | Standard Error | Particle Size (μm) | |
|---|---|---|---|
| IL-4-PLGA | 91.94 | 1.81 | 20.06 ± 3.87 |
| KGN-PLGA | 93.57 | 2.37 | 51.13 ± 6.21 |
| Target Gene | Species | Forward | Reverse |
|---|---|---|---|
| GAPDH | Mouse/Rat | CCGCATCTTCTTGTGCAGTG | CGATACGGCCAAATCGTTC |
| Aggrecan | Rat | GCTACGACGCCATCTGCTACAC | ATGTCCTCTTCACCACCCACTCC |
| Collagen II | Rat | GGAGCAGCAAGAGCAAGGAGAAG | GGAGCCCTCAGTGGACAGTAGAC |
| MMP13 | Rat | ATACGAGCATCCATCCCGAGACC | AACCGCAGCACTGAGCCTTTTC |
| CD86 | Mouse | ACGGAGTCAATGAAGATTTCCT | GATTCGGCTTCTTGTGACATAC |
| CD206 | Mouse | CCTATGAAAATTGGGCTTACGG | CTGACAAATCCAGTTGTTGAGG |
| TNF-α | Rat | ATGTCTCAGCCTCTTCTCATTC | GCTTGTCACTCGAATTTTGAGA |
| iNOS | Mouse | TCTAGTGAAGCAAAGCCCAACA | TGATGGACCCCAAGCAAGAC |
| IL-10 | Mouse | GGTTGCCAAGCCTTATCGGA | CACCTTGGTCTTGGAGCTTATT |
| IL-13 | Mouse | CACACAAGACCAGACTCCCCT | TGGCGAAACAGTTGCTTTGT |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, H.; Guo, Q.; Zhao, H.; Liu, K.; Kang, H.; Gao, F.; Guo, J.; Yuan, X.; Hu, S.; Li, F.; et al. An Injectable Hydrogel Scaffold Loaded with Dual-Drug/Sustained-Release PLGA Microspheres for the Regulation of Macrophage Polarization in the Treatment of Intervertebral Disc Degeneration. Int. J. Mol. Sci. 2023, 24, 390. https://doi.org/10.3390/ijms24010390
Cheng H, Guo Q, Zhao H, Liu K, Kang H, Gao F, Guo J, Yuan X, Hu S, Li F, et al. An Injectable Hydrogel Scaffold Loaded with Dual-Drug/Sustained-Release PLGA Microspheres for the Regulation of Macrophage Polarization in the Treatment of Intervertebral Disc Degeneration. International Journal of Molecular Sciences. 2023; 24(1):390. https://doi.org/10.3390/ijms24010390
Chicago/Turabian StyleCheng, Haozhe, Qian Guo, Hongjian Zhao, Kun Liu, Honglei Kang, Fang Gao, Jianfeng Guo, Xi Yuan, Shuang Hu, Feng Li, and et al. 2023. "An Injectable Hydrogel Scaffold Loaded with Dual-Drug/Sustained-Release PLGA Microspheres for the Regulation of Macrophage Polarization in the Treatment of Intervertebral Disc Degeneration" International Journal of Molecular Sciences 24, no. 1: 390. https://doi.org/10.3390/ijms24010390





