Metabolite and Transcriptome Profiles of Proanthocyanidin Biosynthesis in the Development of Litchi Fruit
Abstract
1. Introduction
2. Results
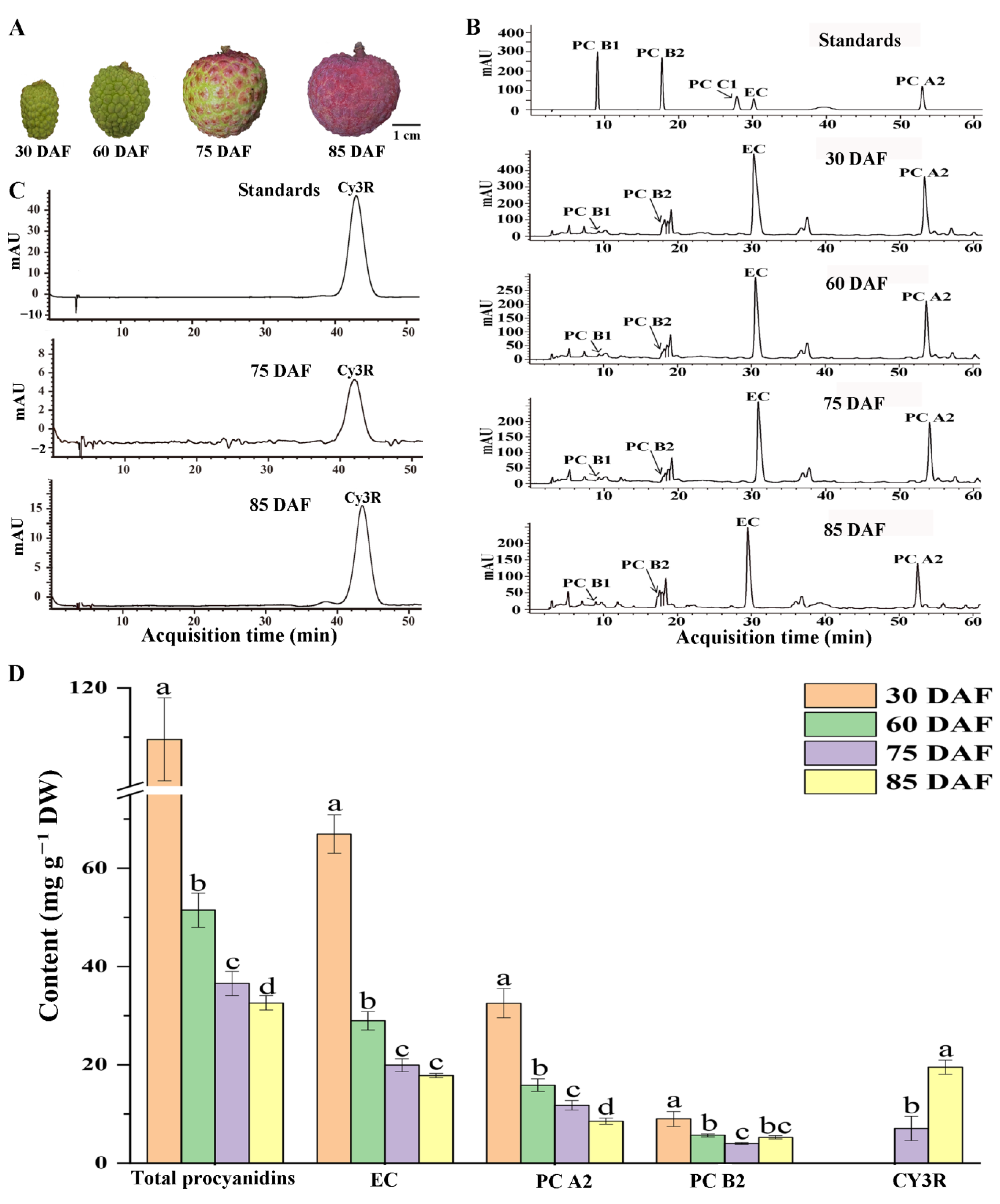
2.1. Changes in the Proanthocyanidin and Anthocyanin Content of the Pericarp during Fruit Development
2.2. Analysis of Transcriptome Sequencing Data of Litchi Pericarp during Fruit Development
2.3. GO and KEGG Enrichment Analysis of the DEGs
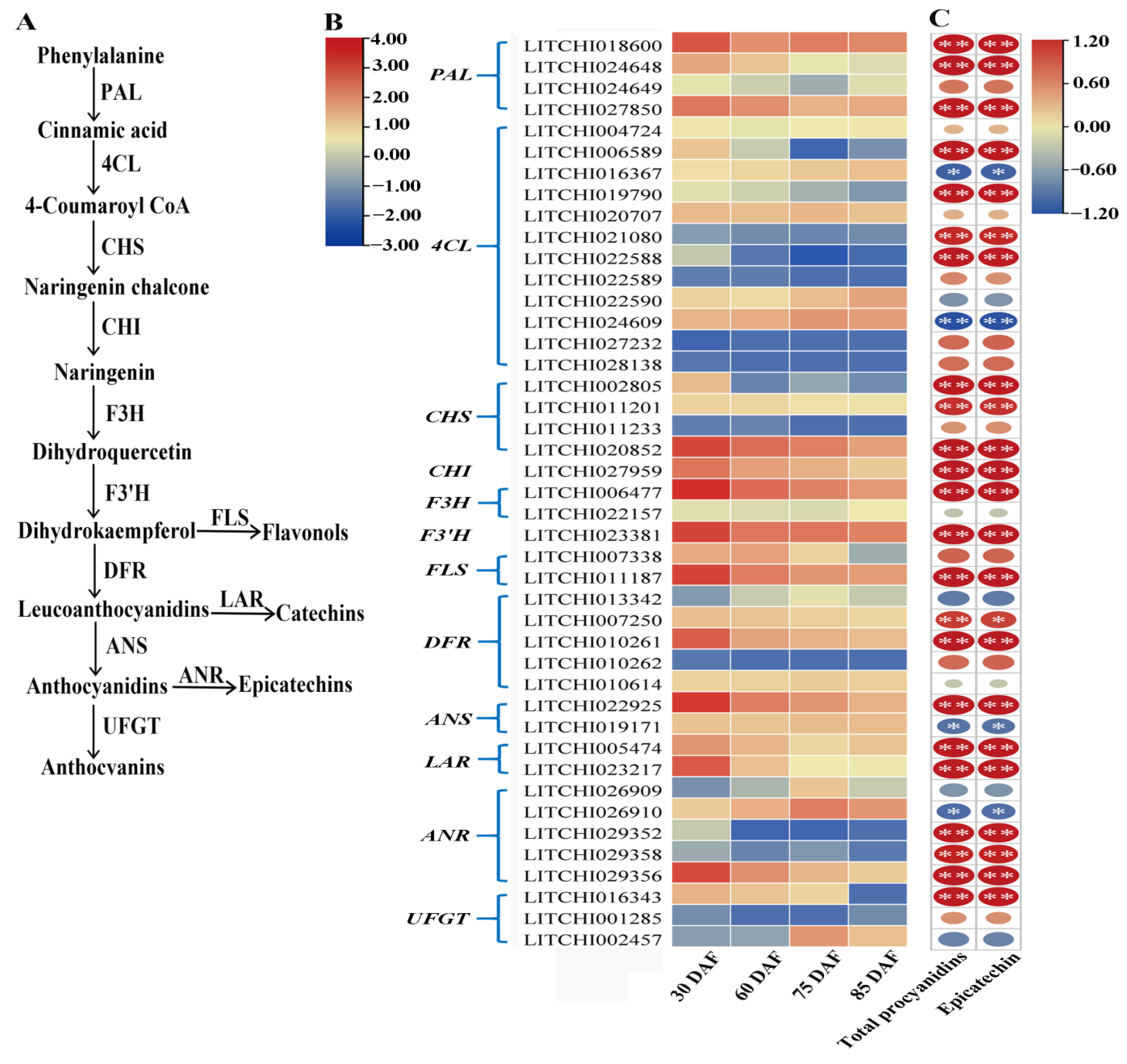
2.4. Expression of Flavonoid/PA Biosynthesis-Related Genes in Litchi Pericarp during Fruit Development
2.5. Verification of the Expression of the Genes Involved in Catechin Biosynthesis
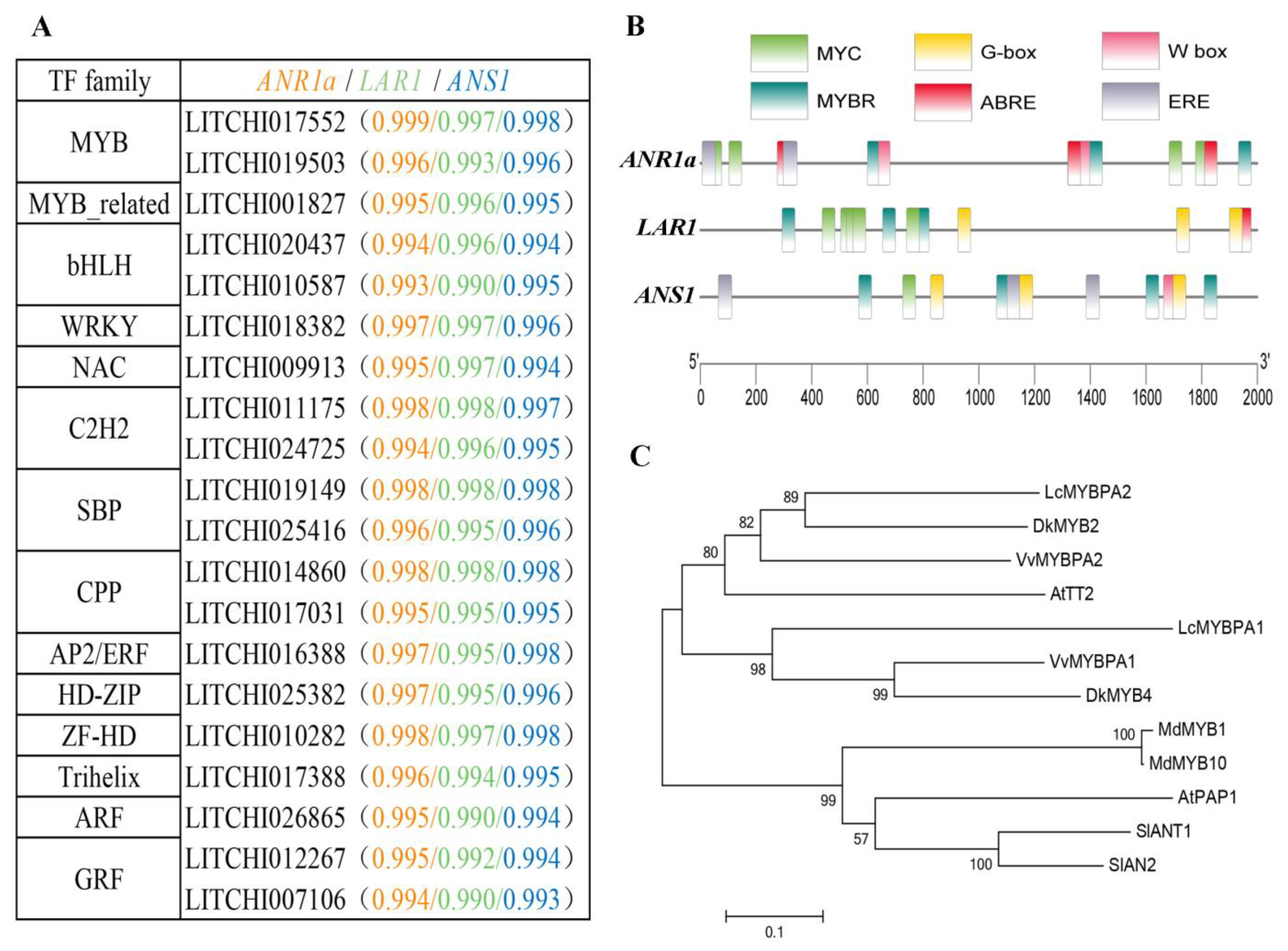
2.6. Prediction of the Biochemical Characteristics of ANR, LAR and ANS Proteins
2.7. Phylogenetic Trees of Litchi ANR, LAR and ANS Proteins
2.8. Screening of Potential Catechin Biosynthesis-Related Transcription Factor (TF) Genes
3. Discussion
3.1. PAs Highly Accumulate in the Young Fruit Stage and Decline with Fruit Development
3.2. Identification of Key Genes for PA Biosynthesis in Litchi Pericarp
3.3. Candidate Transcription Factors Involved in the Regulation of Catechin Biosynthesis in Litchi Pericarp
4. Materials and Methods
4.1. Plant Materials
4.2. Extraction and Analysis of PAs and Anthocyanins in the Pericarp
4.3. RNA Preparation and Transcriptome Analysis
4.4. Bioinformatic and Phylogenetic Analyses of ANR, LAR and ANS Genes
4.5. Quantitative Real-Time PCR (qRT-PCR) Analysis
4.6. Analysis of Cis-Acting Regulatory Elements in the Promoter
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jiang, Y.M.; Wang, Y.; Song, L.; Liu, H.; Lichter, A.; Kerdchoechuen, O.; Joyce, D.C.; Shi, J. Postharvest characteristics and handling of litchi fruit-an overview. Aust. J. Exp. Agr. 2006, 46, 1541. [Google Scholar] [CrossRef]
- Akamine, E.K. Preventing the darkening of fresh lychees prepared for export. Tech. Prog. Rep. Hawaii Agric. Exp. Stn. Univ. Hawaii 1960, 127, 17–21. [Google Scholar] [CrossRef]
- Zhang, X.; Fang, F.; He, Q.; Zhang, X.; Shi, N.; Song, J.; Zhang, Z.; Pang, X. Enzymatic characterization of a laccase from lychee pericarp in relation to browning reveals the mechanisms for fruit color protection. J. Food Process. Pres. 2017, 42, e13515. [Google Scholar] [CrossRef]
- Fang, F.; Zhang, X.; Luo, H.; Zhou, J.; Gong, Y.; Li, W.; Shi, Z.; He, Q.; Wu, Q.; Li, L.; et al. An intracellular laccase is responsible for the epicatechin mediated anthocyanin degradation in litchi fruit pericarp. Plant Physiol. 2015, 169, 2391–2408. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Zhang, X.; Zhong, R.; Liu, B.; Zhang, X.; Fang, F.; Zhang, Z.; Pang, X. Laccase-mediated flavonoid polymerization leads to the pericarp browning of litchi fruit. J. Agr. Food Chem. 2021, 69, 15218–15230. [Google Scholar] [CrossRef] [PubMed]
- Winkel-Shirley, B. Flavonoid Biosynthesis. A colorful model for genetics, biochemistry, cell biology, and biotechnology. Plant Physiol. 2001, 126, 485–493. [Google Scholar] [CrossRef]
- Mannino, G.; Chinigò, G.; Serio, G.; Genova, T.; Gentile, C.; Munaron, L.; Bertea, C.M. Proanthocyanidins and where to find them: A Meta-Analytic approach to investigate their chemistry, biosynthesis, distribution, and Effect on Human Health. Antioxidants 2021, 10, 1229. [Google Scholar] [CrossRef]
- Yu, C. Molecular mechanism of manipulating seed coat coloration in oilseed brassica species. J. Appl. Genet. 2013, 54, 135–145. [Google Scholar] [CrossRef]
- Hibi, Y.; Yanase, E. Oxidation of procyanidins with various degrees of condensation: Influence on the color-deepening phenomenon. J. Agr. Food Chem. 2019, 67, 4940–4946. [Google Scholar] [CrossRef]
- Pourcel, L.; Routaboul, J.; Kerhoas, L.; Caboche, M.; Lepiniec, L.; Debeaujon, I. Transparent testa10 encodes a laccase-like enzyme involved in oxidative polymerization of flavonoids in arabidopsis seed coat. Plant Cell 2005, 17, 2966–2980. [Google Scholar] [CrossRef]
- Prasad, K.N.; Yang, B.; Zhao, M.; Ruenroengklin, N.; Jiang, Y. Application of ultrasonication or high-pressure extraction of flavonoids from litchi fruit pericarp. J. Food Process Eng. 2009, 32, 828–843. [Google Scholar] [CrossRef]
- Zhang, D.; Quantick, P.C.; Grigor, J.M. Changes in phenolic compounds in litchi (Litchi chinensis Sonn.) fruit during postharvest storage. Postharvest Biol. Tec. 2000, 19, 165–172. [Google Scholar] [CrossRef]
- Sarni-Manchado, P.; Le Roux, E.; Le Guernevé, C.; Lozano, Y.; Cheynier, V. Phenolic composition of litchi fruit pericarp. J. Agr. Food Chem. 2000, 48, 5995–6002. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Liang, H.; Zhang, M.; Zhang, R.; Deng, Y.; Wei, Z.; Zhang, Y.; Tang, X. Phenolic profiles and antioxidant activity of litchi (Litchi Chinensis Sonn.) fruit pericarp from different commercially available cultivars. Molecules 2012, 17, 14954–14967. [Google Scholar] [CrossRef] [PubMed]
- Tsao, R.; Yang, R.; Young, J.C.; Zhu, H. Polyphenolic profiles in eight Apple cultivars using High-Performance Liquid Chromatography (HPLC). J. Agr. Food Chem. 2003, 51, 6347–6353. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Li, L.; You, X.; Li, C.; Zhang, E.; Li, Z.; Chen, G.; Peng, H. Phenolics and polysaccharides in major tropical fruits: Chemical compositions, analytical methods and bioactivities. Anal. Methods 2011, 3, 2212. [Google Scholar] [CrossRef]
- Gong, Y.; Fang, F.; Zhang, X.; Liu, B.; Luo, H.; Li, Z.; Zhang, X.; Zhang, Z.; Pang, X. B type and complex A/B type epicatechin trimers isolated from litchi pericarp aqueous extract show high antioxidant and anticancer activity. Int. J. Mol. Sci. 2018, 19, 301. [Google Scholar] [CrossRef]
- Xie, D.; Dixon, R.A. Proanthocyanidin biosynthesis-still more questions than answers? Phytochemistry 2005, 66, 2127–2144. [Google Scholar] [CrossRef]
- Tanner, G.J.; Francki, K.T.; Abrahams, S.; Watson, J.M.; Larkin, P.J.; Ashton, A.R. Proanthocyanidin biosynthesis in Plants. J. Biol. Chem. 2003, 278, 31647–31656. [Google Scholar] [CrossRef]
- Xie, D.; Sharma, S.B.; Paiva, N.L.; Ferreira, D.; Dixon, R.A. Role of anthocyanidin reductase, encoded by banyuls in plant flavonoid biosynthesis. Science 2003, 299, 396–399. [Google Scholar] [CrossRef]
- Zhao, J.; Pang, Y.; Dixon, R.A. The mysteries of proanthocyanidin transport and polymerization. Plant Physiol. 2010, 153, 437–443. [Google Scholar] [CrossRef] [PubMed]
- Dixon, R.A.; Xie, D.; Sharma, S.B. Proanthocyanidins-a final frontier in flavonoid research? New Phytol. 2005, 165, 9–28. [Google Scholar] [CrossRef] [PubMed]
- Marles, M.A.S.; Ray, H.; Gruber, M.Y. New perspectives on proanthocyanidin biochemistry and molecular regulation. Phytochemistry 2003, 64, 367–383. [Google Scholar] [CrossRef] [PubMed]
- Honda, C.; Kotoda, N.; Wada, M.; Kondo, S.; Kobayashi, S.; Soejima, J.; Zhang, Z.; Tsuda, T.; Moriguchi, T. Anthocyanin biosynthetic genes are coordinately expressed during red coloration in apple skin. Plant Physiol. Bioch. 2002, 40, 955–962. [Google Scholar] [CrossRef]
- Hu, G.; Feng, J.; Xiang, X.; Wang, J.; Salojärvi, J.; Liu, C.; Wu, Z.; Zhang, J.; Liang, X.; Jiang, Z.; et al. Two divergent haplotypes from a highly heterozygous lychee genome suggest independent domestication events for early and late-maturing cultivars. Nat. Genet. 2022, 54, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Saito, K.; Yonekura-Sakakibara, K.; Nakabayashi, R.; Higashi, Y.; Yamazaki, M.; Tohge, T.; Fernie, A.R. The flavonoid biosynthetic pathway in arabidopsis: Structural and genetic diversity. Plant Physiol. Bioch. 2013, 72, 21–34. [Google Scholar] [CrossRef]
- Lepiniec, L.; Debeaujon, I.; Routaboul, J.; Baudry, A.; Pourcel, L.; Nesi, N.; Caboche, M. Genetics and biochemistry of seed flavonoids. Annu. Rev. Plant Biol. 2006, 57, 405–430. [Google Scholar] [CrossRef]
- Liu, C.; Chi, C.; Jin, L.; Zhu, J.; Yu, J.; Zhou, Y. The bZip transcription factor HY5 mediates CRY1a-induced anthocyanin biosynthesis in tomato. Plant Cell Environ. 2018, 41, 1762–1775. [Google Scholar] [CrossRef]
- Mehrtens, F.; Kranz, H.; Bednarek, P.; Weisshaar, B. The Arabidopsis transcription factor MYB12 is a flavonol-specific regulator of phenylpropanoid biosynthesis. Plant Physiol. 2005, 138, 1083–1096. [Google Scholar] [CrossRef]
- Nesi, N.; Jond, C.; Debeaujon, I.; Caboche, M.; Lepiniec, L. The arabidopsis TT2 gene encodes an R2R3 MYB domain protein that acts as a key determinant for proanthocyanidin accumulation in developing seed. Plant Cell 2001, 13, 2099–2114. [Google Scholar] [CrossRef]
- Terrier, N.; Torregrosa, L.; Ageorges, A.; Vialet, S.; Verriès, C.; Cheynier, V.; Romieu, C. Ectopic expression of VvMYBPA2 promotes proanthocyanidin biosynthesis in grapevine and suggests additional targets in the pathway. Plant Physiol. 2009, 149, 1028–1041. [Google Scholar] [CrossRef] [PubMed]
- Bogs, J.; Jaffé, F.W.; Takos, A.M.; Walker, A.R.; Robinson, S.P. The grapevine transcription factor VvMYBPA1 regulates proanthocyanidin synthesis during fruit development. Plant Physiol. 2007, 143, 1347–1361. [Google Scholar] [CrossRef] [PubMed]
- Akagi, T.; Ikegami, A.; Yonemori, K. DkMYB2 wound-induced transcription factor of persimmon (Diospyros kaki Thunb.), contributes to proanthocyanidin regulation. Planta 2010, 232, 1045–1059. [Google Scholar] [CrossRef]
- Akagi, T.; Ikegami, A.; Tsujimoto, T.; Kobayashi, S.; Sato, A.; Kono, A.; Yonemori, K. DkMyb4 is a MYB transcription factor involved in proanthocyanidin biosynthesis in persimmon fruit. Plant Physiol. 2009, 151, 2028–2045. [Google Scholar] [CrossRef]
- Mellway, R.D.; Tran, L.T.; Prouse, M.B.; Campbell, M.M.; Constabel, C.P. The wound-, pathogen-, and ultraviolet B-responsive MYB134 gene encodes an R2R3 MYB transcription factor that regulates proanthocyanidin synthesis in poplar. Plant Physiol. 2009, 150, 924–941. [Google Scholar] [CrossRef]
- Verdier, J.; Zhao, J.; Torres-Jerez, I.; Ge, S.; Liu, C.; He, X.; Mysore, K.S.; Dixon, R.A.; Udvardi, M.K. MtPAR MYB transcription factor acts as an on switch for proanthocyanidin biosynthesis in medicago truncatula. Proc. Natl. Acad. Sci. USA 2012, 109, 1766–1771. [Google Scholar] [CrossRef]
- Reichel, M.; Carle, R.; Sruamsiri, P.; Neidhart, S. Changes in flavonoids and nonphenolic pigments during on-tree maturation and postharvest pericarp browning of litchi (Litchi chinensis Sonn.) as shown by HPLC-MSn. J. Agric. Food Chem. 2011, 59, 3924–3939. [Google Scholar] [CrossRef]
- Arlotta, C.; Puglia, G.D.; Genovese, C.; Toscano, V.; Karlova, R.; Beekwilder, J.; Vos, R.C.H.D.; Raccuia, S.A. MYB5-like and bHLH influence flavonoid composition in pomegranate. Plant Sci. 2020, 298, 110563. [Google Scholar] [CrossRef]
- Wang, N.; Liu, W.; Zhang, T.; Jiang, S.; Xu, H.; Wang, Y.; Zhang, Z.; Wang, C.; Chen, X. Transcriptomic analysis of red-fleshed apples reveals the novel role of MdWRKY11 in flavonoid and anthocyanin biosynthesis. J. Agr. Food Chem. 2018, 66, 7076–7086. [Google Scholar] [CrossRef]
- Schreiner, M.; Mewis, I.; Huyskens-Keil, S.; Jansen, M.A.K.; Zrenner, R.; Winkler, J.B.; O’Brien, N.; Krumbein, A. UV-B-induced secondary plant metabolites-potential benefits for plant and human health. Crit. Rev. Plant Sci. 2012, 31, 229–240. [Google Scholar] [CrossRef]
- Dixon, R.A.; Steele, C.L. Flavonoids and isoflavonoids-a gold mine for metabolic engineering. Trends Plant Sci. 1999, 4, 394–400. [Google Scholar] [CrossRef]
- Kevan, P.; Giurfa, M.; Chittka, L. Why are there so many and so few white flowers? Trends Plant Sci. 1996, 1, 252. [Google Scholar] [CrossRef]
- Dixon, R.A.; Liu, C.; Jun, J.H. Metabolic engineering of anthocyanins and condensed tannins in plants. Curr. Opin. Biotech. 2013, 24, 329–335. [Google Scholar] [CrossRef]
- Boss, P.K.; Davies, C.; Robinson, S.P. Analysis of the expression of anthocyanin pathway genes in developing Vitis vinifera L. cv shiraz grape berries and the implications for pathway regdation. Plant Physiol. 1996, 111, 1059–1066. [Google Scholar] [CrossRef] [PubMed]
- Suvanto, J.; Karppinen, K.; Riihinen, K.; Jaakola, L.; Salminen, J. Changes in the proanthocyanidin composition and related gene expression in bilberry (Vaccinium Myrtillus L.) tissues. J. Agr. Food Chem. 2020, 68, 7378–7386. [Google Scholar] [CrossRef] [PubMed]
- Sivakumar, D.; Naudé, Y.; Rohwer, E.; Korsten, L. Volatile compounds, quality attributes, mineral composition and pericarp structure of south african litchi export cultivars Mauritius and Mclean’s red. J. Sci. Food Agr. 2008, 88, 1074–1081. [Google Scholar] [CrossRef]
- Huang, X.; Yuan, W.; Wang, H.; Li, J.; Huang, H.; Shi, L.; Jinhua, Y. Linking cracking resistance and fruit desiccation rate to pericarp structure in litchi (Litchi chinensis Sonn.). J. Hortic. Sci. Biotech. 2004, 79, 897–905. [Google Scholar] [CrossRef]
- Hammerbacher, A.; Paetz, C.; Wright, L.P.; Fischer, T.C.T.; Bohlmann, J.; Davis, A.J.; Fenning, T.M.; Gershenzon, J.; Schmidt, A. Flavan-3-ols in norway spruce: Biosynthesis, accumulation, and function in response to attack by the bark beetle-associated fungus ceratocystis polonica. Plant Physiol. 2014, 164, 2107–2122. [Google Scholar] [CrossRef]
- Vrhovsek, U.; Rigo, A.; Tonon, D.; Mattivi, F. Quantitation of polyphenols in different apple varieties. J. Agr. Food Chem. 2004, 52, 6532–6538. [Google Scholar] [CrossRef]
- Xia, H.; Zhang, X.; Shen, Y.; Guo, Y.; Wang, T.; Wang, J.; Lin, L.; Deng, H.; Deng, Q.; Xu, K.; et al. Comparative analysis of flavonoids in white and red table grape cultivars during ripening by widely targeted metabolome and transcript levels. J. Food Sci. 2022, 87, 1650–1661. [Google Scholar] [CrossRef]
- Lai, B.; Hu, B.; Qin, Y.; Zhao, J.; Wang, H.; Hu, G. Transcriptomic analysis of Litchi chinensis pericarp during maturation with a focus on chlorophyll degradation and flavonoid biosynthesis. BMC Genom. 2015, 16, 225. [Google Scholar] [CrossRef]
- Wei, Y.; Hu, F.; Hu, G.; Li, X.; Huang, X.; Wang, H. Differential expression of anthocyanin biosynthetic genes in relation to anthocyanin accumulation in the pericarp of litchi chinensis sonn. PLoS ONE 2011, 6, e19455. [Google Scholar] [CrossRef]
- Bogs, J.; Downey, M.O.; Harvey, J.S.; Ashton, A.R.; Tanner, G.J.; Robinson, S.P. Proanthocyanidin synthesis and expression of genes encoding leucoanthocyanidin reductase and anthocyanidin reductase in developing grape berries and grapevine leaves. Plant Physiol. 2005, 139, 652–663. [Google Scholar] [CrossRef]
- Cantín, C.M.; Moreno, M.A.; Gogorcena, Y. Evaluation of the antioxidant capacity, phenolic compounds, and vitamin C content of different peach and nectarine [Prunus persica (L.) Batsch] breeding progenies. J. Agr. Food Chem. 2009, 57, 4586–4592. [Google Scholar] [CrossRef]
- Liao, L.; Vimolmangkang, S.; Wei, G.; Zhou, H.; Korban, S.S.; Han, Y. Molecular characterization of genes encoding leucoanthocyanidin reductase involved in proanthocyanidin biosynthesis in apple. Front. Plant Sci. 2015, 6, 135. [Google Scholar] [CrossRef]
- Pang, Y.; Abeysinghe, I.S.B.; He, J.; He, X.; Huhman, D.; Mewan, K.M.; Sumner, L.W.; Yun, J.; Dixon, R.A. Functional characterization of proanthocyanidin pathway enzymes from tea and their application for metabolic engineering. Plant Physiol. 2013, 161, 1103–1116. [Google Scholar] [CrossRef]
- Maugé, C.; Granier, T.; D’Estaintot, B.L.; Gargouri, M.; Manigand, C.; Schmitter, J.; Chaudière, J.; Gallois, B. Crystal structure and catalytic mechanism of leucoanthocyanidin reductase from Vitis vinifera. J. Mol. Biol. 2010, 397, 1079–1091. [Google Scholar] [CrossRef]
- Dixon, R.A.; Sarnala, S. Proanthocyanidin biosynthesis-a matter of protection. Plant Physiol. 2020, 184, 579–591. [Google Scholar] [CrossRef]
- Zhang, J.; Xu, H.; Wang, N.; Jiang, S.; Fang, H.; Zhang, Z.; Yang, G.; Wang, Y.; Su, M.; Xu, L.; et al. The ethylene response factor MdERF1B regulates anthocyanin and proanthocyanidin biosynthesis in apple. Plant Mol. Biol. 2018, 98, 205–218. [Google Scholar] [CrossRef]
- Schaart, J.G.; Dubois, C.; Romero De La Fuente, I.; Houwelingen, A.M.M.L.; Vos, R.C.H.D.; Jonker, H.H.; Xu, W.; Routaboul, J.; Lepinice, L.; Bovy, A.G. Identification and characterization of MYB-bHLH-WD40 regulatory complexes controlling proanthocyanidin biosynthesis in strawberry (Fragaria × ananassa) fruits. New Phytol. 2013, 197, 454–467. [Google Scholar] [CrossRef]
- Cheng, J.; Yu, K.; Shi, Y.; Wang, J.; Duan, C. Transcription factor VviMYB86 oppositely regulates proanthocyanidin and anthocyanin biosynthesis in grape berries. Front. Plant Sci. 2021, 11, 613677. [Google Scholar] [CrossRef] [PubMed]
- Espley, R.V.; Hellens, R.P.; Putterill, J.; Stevenson, D.E.; Kutty-Amma, S.; Allan, A.C. Red colouration in apple fruit is due to the activity of the MYB transcription factor, MdMYB10. Plant J. 2007, 49, 414–427. [Google Scholar] [CrossRef]
- Xie, D.; Sharma, S.B.; Wright, E.; Wang, Z.; Dixon, R.A. Metabolic engineering of proanthocyanidins through co-expression of anthocyanidin reductase and the PAP1 MYB transcription factor. Plant J. 2006, 45, 895–907. [Google Scholar] [CrossRef]
- Mao, Z.; Jiang, H.; Wang, S.; Wang, Y.; Yu, L.; Zou, Q.; Liu, W.; Jiang, S.; Wang, N.; Zhang, Z.; et al. The MdHY5-MdWRKY41-MdMYB transcription factor cascade regulates the anthocyanin and proanthocyanidin biosynthesis in red-fleshed apple. Plant Sci. 2021, 306, 110848. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Zhu, Q.; Zhou, X.; Zhang, X.; Dang, Z.; Liang, S.; Li, G.; Zhang, Z.; Fang, F.; Pang, X. Characterization of a pericarp browning related Laccase 14-4 from longan fruit with a focus on (epi)catechin oxidative polymerization. Postharvest Biol. Tec. 2022, 185, 111802. [Google Scholar] [CrossRef]
- Kim, D.; Langmead, B.; Salzberg, S.L. HISAT: A fast spliced aligner with low memory requirements. Nat. Methods 2015, 12, 357–360. [Google Scholar] [CrossRef]
- Langfelder, P.; Horvath, S. WGCNA: An R package for weighted correlation network analysis. BMC Bioinform. 2008, 9, 559. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using Real-Time Quantitative PCR and the 2−ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]




| Gene ID | Gene Name | Number of Amino Acids | Molecular Weight (kD) | Isoelectric Point | Signal Peptide | N-Glycosyl Sites |
|---|---|---|---|---|---|---|
| LITCHI029356 | LcANR1a | 336 | 36.45 | 6.55 | No | 2 |
| LITCHI029352 | LcANR1b | 334 | 36.3 | 6.55 | No | 2 |
| LITCHI026910 | LcANR2a | 349 | 38.89 | 5.72 | No | 2 |
| LITCHI026909 | LcANR2b | 343 | 38.01 | 5.35 | No | 3 |
| LITCHI029358 | LcANR3 | 281 | 30.18 | 8.87 | No | 2 |
| LITCHI023217 | LcLAR1 | 357 | 39.54 | 5.77 | No | 1 |
| LITCHI005474 | LcLAR2 | 343 | 38.05 | 6.13 | No | 3 |
| LITCHI022925 | LcANS1 | 357 | 40.56 | 5.59 | No | 1 |
| LITCHI019171 | LcANS2 | 345 | 38.29 | 6.21 | No | 2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhong, R.; Wei, J.; Liu, B.; Luo, H.; Zhang, Z.; Pang, X.; Fang, F. Metabolite and Transcriptome Profiles of Proanthocyanidin Biosynthesis in the Development of Litchi Fruit. Int. J. Mol. Sci. 2023, 24, 532. https://doi.org/10.3390/ijms24010532
Zhong R, Wei J, Liu B, Luo H, Zhang Z, Pang X, Fang F. Metabolite and Transcriptome Profiles of Proanthocyanidin Biosynthesis in the Development of Litchi Fruit. International Journal of Molecular Sciences. 2023; 24(1):532. https://doi.org/10.3390/ijms24010532
Chicago/Turabian StyleZhong, Ruihao, Junbin Wei, Bin Liu, Honghui Luo, Zhaoqi Zhang, Xuequn Pang, and Fang Fang. 2023. "Metabolite and Transcriptome Profiles of Proanthocyanidin Biosynthesis in the Development of Litchi Fruit" International Journal of Molecular Sciences 24, no. 1: 532. https://doi.org/10.3390/ijms24010532
APA StyleZhong, R., Wei, J., Liu, B., Luo, H., Zhang, Z., Pang, X., & Fang, F. (2023). Metabolite and Transcriptome Profiles of Proanthocyanidin Biosynthesis in the Development of Litchi Fruit. International Journal of Molecular Sciences, 24(1), 532. https://doi.org/10.3390/ijms24010532






