TGF-β Type I Receptor Signaling in Melanoma Liver Metastases Increases Metastatic Outgrowth
Abstract
:Simple Summary
Abstract
1. Introduction
2. Results
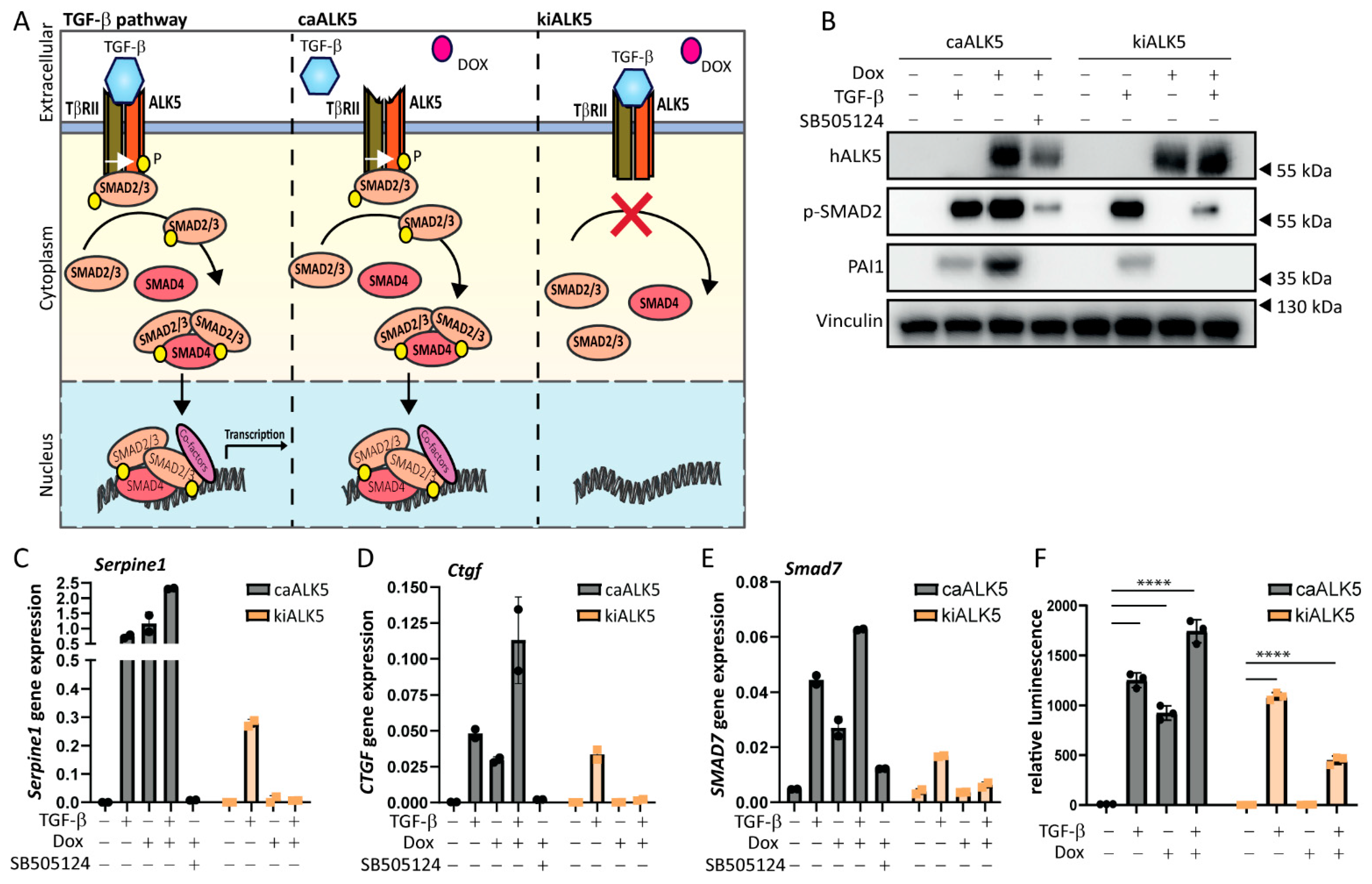
2.1. An Inducible Cellular Model to Activate and Repress TGF-β Type I Receptor Signaling
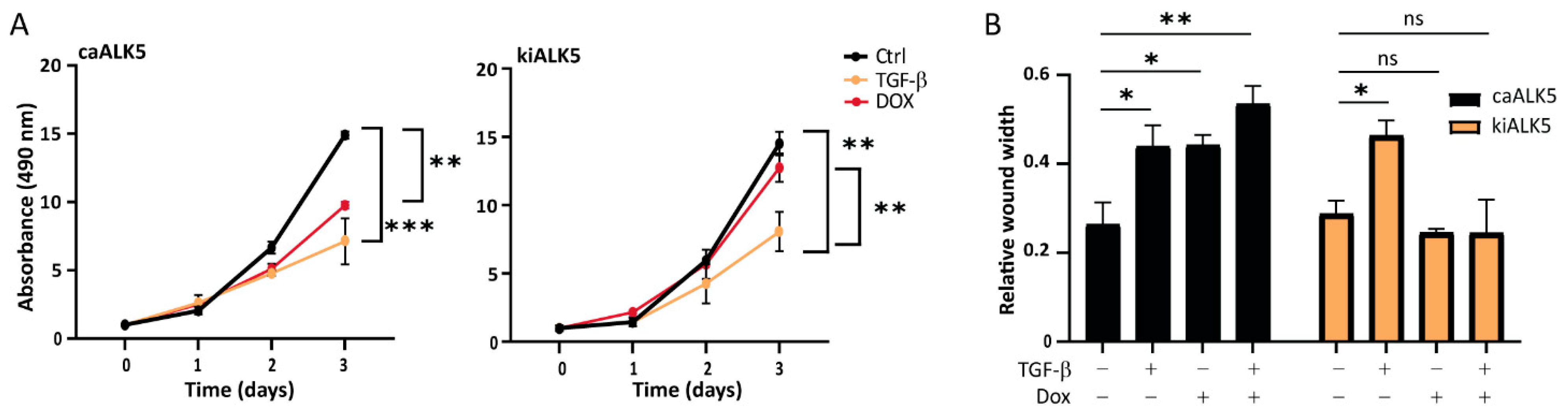
2.2. TGF-β Decreases B16F10 Cell Proliferation and Migration In Vitro
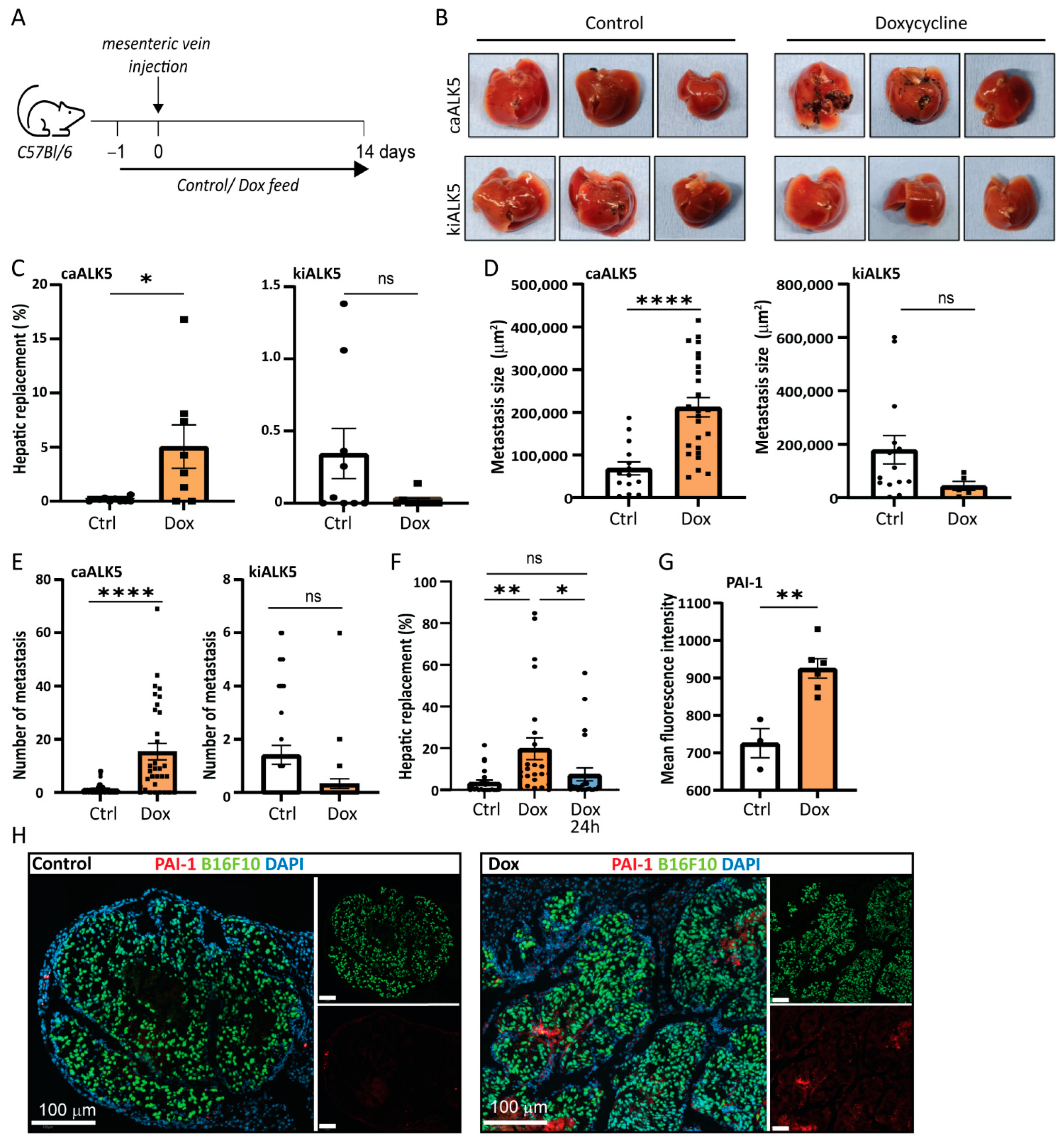
2.3. Metastatic Liver Outgrowth of B16F10 Cells Is Enhanced upon TGF-β Signaling in Tumor Cells
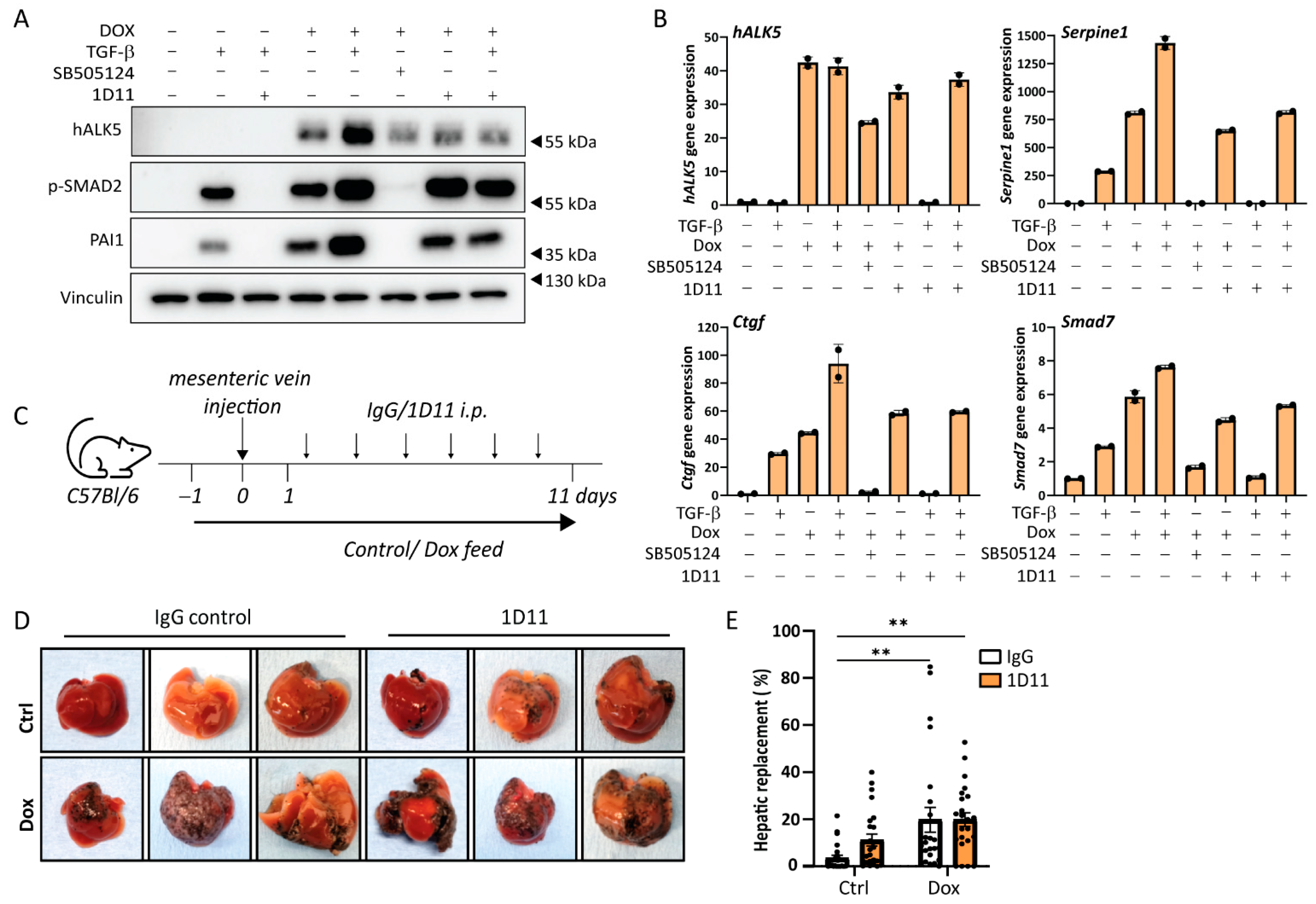
2.4. Metastatic Liver Outgrowth of Control and caALK5 Expressing B16F10 Cells Is Not Affected by TGF-β Neutralizing Antibody
2.5. Changes in Immune Cell Presence upon caALK5 Expression in B16F10 Metastasis
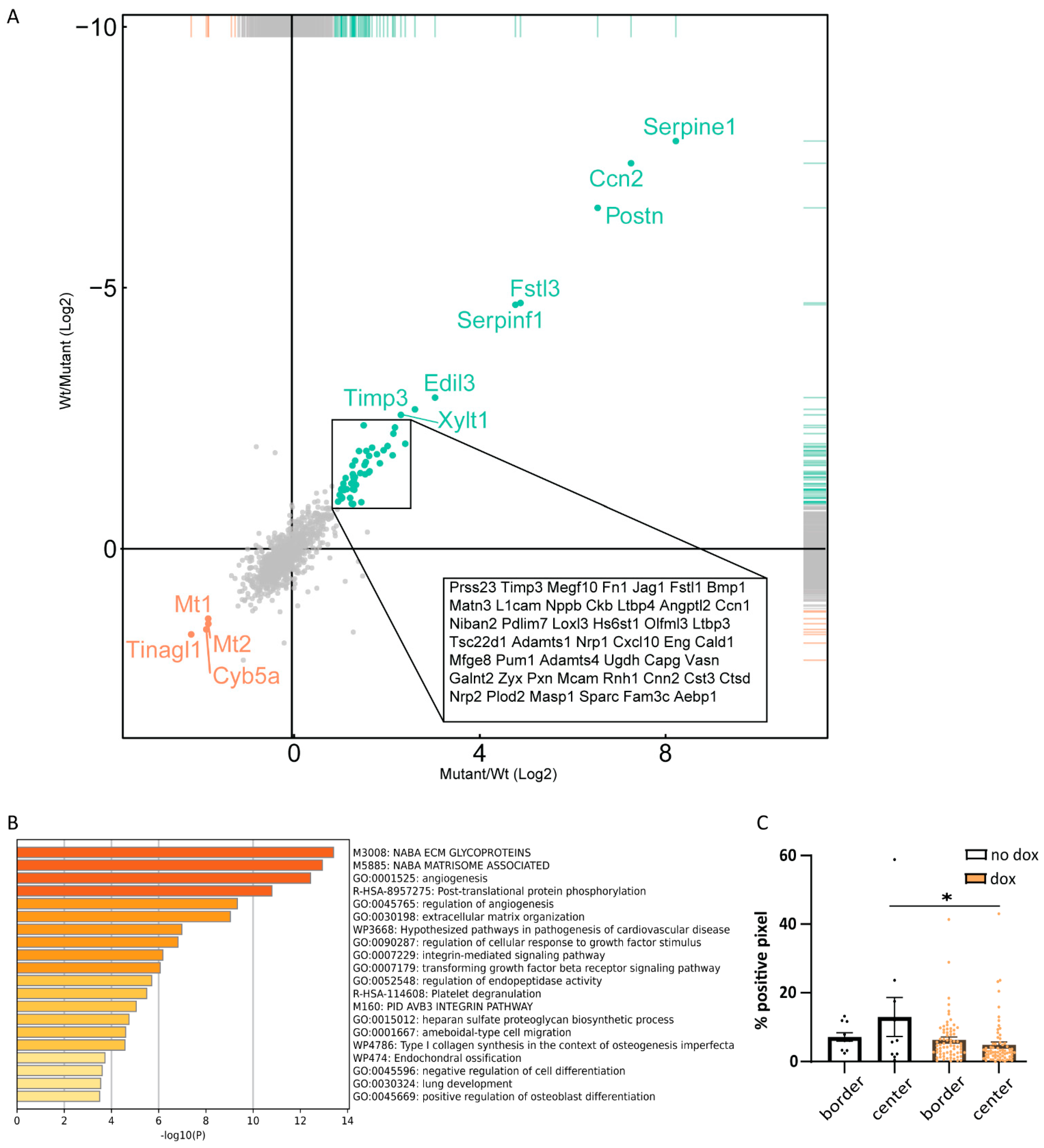
2.6. Secretome Analysis Suggests Tumor Microenvironmental Changes upon caALK5 Expression
3. Discussion
4. Methods
4.1. Reagents
4.2. Generation of Plasmids
4.3. Cell Culture
4.4. Western Blot Analysis
4.5. RT-qPCR
4.6. Animal Studies
4.7. Immunofluorescence Staining
4.8. Fluorescence-Activated Single Cell Sorting (FACS)
4.9. Incucyte® Proliferation Assay
4.10. MTS Assay
4.11. Colony Formation Assay
4.12. Wound Healing and Invasion Assay
4.13. Random Migration Assay
4.14. Mass Spectrometry of B16F10 Secretome
4.15. Gene Set Enrichment Analysis
4.16. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- DaCosta Byfield, S.; Major, C.; Laping, N.J.; Roberts, A.B. SB-505124 Is a Selective Inhibitor of Transforming Growth Factor-β Type I Receptors ALK4, ALK5, and ALK7. Mol. Pharmacol. 2004, 65, 744–752. [Google Scholar] [CrossRef] [PubMed]
- Siegel, P.M.; Massagué, J. Cytostatic and apoptotic actions of TGF-β in homeostasis and cancer. Nat. Rev. Cancer 2003, 3, 807–820. [Google Scholar] [CrossRef] [PubMed]
- Ohuchi, K.; Kambayashi, Y.; Hidaka, T.; Fujimura, T. Plasminogen Activating Inhibitor-1 Might Predict the Efficacy of Anti-PD1 Antibody in Advanced Melanoma Patients. Front. Oncol. 2021, 11, 798385. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Sun, S.; Xiang, H.; Yang, J.; Peng, M.; Gao, Q. Liver metastases and the efficacy of the PD-1 or PD-L1 inhibitors in cancer: A meta-analysis of randomized controlled trials. Oncoimmunology 2020, 9, 1746113. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Green, M.D.; Li, S.; Sun, Y.; Journey, S.N.; Choi, J.E.; Rizvi, S.M.; Qin, A.; Waninger, J.J.; Lang, X.; et al. Liver metastasis restrains immunotherapy efficacy via macrophage-mediated T cell elimination. Nat. Med. 2021, 27, 152–164. [Google Scholar] [CrossRef]
- Massagué, J. TGFβ signalling in context. Nat. Rev. Mol. Cell Biol. 2012, 13, 616–630. [Google Scholar] [CrossRef]
- Marvin, D.L.; Heijboer, R.; ten Dijke, P.; Ritsma, L. TGF-β signaling in liver metastasis. Clin. Transl. Med. 2020, 10, e160. [Google Scholar] [CrossRef]
- Krasagakis, K.; Fimmel, S.; Von Der Ohe, M.; Mansmann, U.; Orfanos, C.E. Desensitization of melanoma cells to autocrine TGF-? isoforms. J. Cell. Physiol. 1999, 178, 179–187. [Google Scholar] [CrossRef]
- Krasagakis, K.; Thölke, D.; Farthmann, B.; Eberle, J.; Mansmann, U.; Orfanos, C.E. Elevated plasma levels of transforming growth factor (TGF)-beta1 and TGF-beta2 in patients with disseminated malignant melanoma. Br. J. Cancer 1998, 77, 1492–1494. [Google Scholar] [CrossRef]
- Reed, J.A.; McNutt, N.S.; Prieto, V.G.; Albino, A.P. Expression of transforming growth factor-β2 in malignant melanoma correlates with the depth of tumor invasion: Implications for tumor progression. Am. J. Pathol. 1994, 145, 97–104. [Google Scholar]
- Lauden, L.; Siewiera, J.; Boukouaci, W.; Ramgolam, K.; Mourah, S.; Lebbe, C.; Charron, D.; Aoudjit, F.; Jabrane-Ferrat, N.; Al-Daccak, R. TGF-β-Induced (TGFBI) Protein in Melanoma: A Signature of High Metastatic Potential. J. Investig. Dermatol. 2014, 134, 1675–1685. [Google Scholar] [CrossRef]
- Cantelli, G.; Orgaz, J.L.; Rodriguez-Hernandez, I.; Karagiannis, P.; Maiques, O.; Matias-Guiu, X.; Nestle, F.O.; Marti, R.M.; Karagiannis, S.N.; Sanz-Moreno, V. TGF-β-Induced Transcription Sustains Amoeboid Melanoma Migration and Dissemination. Curr. Biol. 2015, 25, 2899–2914. [Google Scholar] [CrossRef] [PubMed]
- Evans, R.A.; Tian, Y.C.; Steadman, R.; Phillips, A.O. TGF-beta1-mediated fibroblast-myofibroblast terminal differ-entiation-the role of Smad proteins. Exp. Cell Res. 2003, 282, 90–100. [Google Scholar] [CrossRef] [PubMed]
- Yoon, H.; Tang, C.M.; Banerjee, S.; Delgado, A.L.; Yebra, M.; Davis, J.; Sicklick, J.K. TGF-β1-mediated transition of resident fibroblasts to cancer-associated fibroblasts promotes cancer me-tastasis in gastrointestinal stromal tumor. Oncogenesis 2021, 10, 13. [Google Scholar] [CrossRef]
- Batlle, E.; Massagué, J. Transforming Growth Factor-β Signaling in Immunity and Cancer. Immunity 2019, 50, 924–940. [Google Scholar] [CrossRef]
- Wieser, R.; Wrana, J.L.; Massagué, J. GS domain mutations that constitutively activate TβR-I, the downstream signaling component in the TGF-β receptor complex. EMBO J. 1995, 14, 2199–2208. [Google Scholar] [CrossRef] [PubMed]
- Meerbrey, K.L.; Hu, G.; Kessler, J.D.; Roarty, K.; Li, M.Z.; Fang, J.E.; Herschkowitz, J.I.; Burrows, A.E.; Ciccia, A.; Sun, T.; et al. The pINDUCER lentiviral toolkit for inducible RNA interference in vitro and in vivo. Proc. Natl. Acad. Sci. USA 2011, 108, 3665–3670. [Google Scholar] [CrossRef]
- Marvin, D.L.; ten Dijke, P.; Ritsma, L. An Experimental Liver Metastasis Mouse Model Suitable for Short and Long-Term Intravital Imaging. Curr. Protoc. 2021, 1, e116. [Google Scholar] [CrossRef]
- Papaccio, F.; Kovacs, D.; Bellei, B.; Caputo, S.; Migliano, E.; Cota, C.; Picardo, M. Profiling Cancer-Associated Fibroblasts in Melanoma. Int. J. Mol. Sci. 2021, 22, 7255. [Google Scholar] [CrossRef]
- Wen, S.W.; Ager, E.I.; Christophi, C. Bimodal role of Kupffer cells during colorectal cancer liver metastasis. Cancer Biol. Ther. 2013, 14, 606–613. [Google Scholar] [CrossRef]
- Ceci, C.; Atzori, M.; Lacal, P.; Graziani, G. Targeting Tumor-Associated Macrophages to Increase the Efficacy of Immune Checkpoint Inhibitors: A Glimpse into Novel Therapeutic Approaches for Metastatic Melanoma. Cancers 2020, 12, 3401. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.J.; Jung, J.I.; Lim, D.Y.; Kwon, G.T.; Her, S.; Park, J.H.; Park, J.H.Y. Bone marrow-derived, alternatively activated macrophages enhance solid tumor growth and lung metastasis of mammary carcinoma cells in a Balb/C mouse orthotopic model. Breast Cancer Res. 2012, 14, R81. [Google Scholar] [CrossRef] [PubMed]
- Schaller, J.; Agudo, J. Metastatic Colonization: Escaping Immune Surveillance. Cancers 2020, 12, 3385. [Google Scholar] [CrossRef]
- Tauriello, D.V.F.; Palomo-Ponce, S.; Stork, D.; Berenguer-Llergo, A.; Badia-Ramentol, J.; Iglesias, M.; Sevillano, M.; Ibiza, S.; Cañellas, A.; Hernando-Momblona, X.; et al. TGFβ drives immune evasion in genetically reconstituted colon cancer metastasis. Nature 2018, 554, 538–543. [Google Scholar] [CrossRef]
- Fukuda, K.; Sugihara, E.; Ohta, S.; Izuhara, K.; Funakoshi, T.; Amagai, M.; Saya, H. Periostin Is a Key Niche Component for Wound Metastasis of Melanoma. PLoS ONE 2015, 10, e0129704. [Google Scholar] [CrossRef]
- Kotobuki, Y.; Yang, L.; Serada, S.; Tanemura, A.; Yang, F.; Nomura, S.; Kudo, A.; Izuhara, K.; Murota, H.; Fujimoto, M.; et al. Periostin accelerates human malignant melanoma progression by modifying the melanoma micro-environment. Pigment Cell Melanoma Res. 2014, 27, 630–639. [Google Scholar] [CrossRef] [PubMed]
- Ohno, F.; Nakahara, T.; Kido-Nakahara, M.; Ito, T.; Nunomura, S.; Izuhara, K.; Furue, M. Periostin Links Skin Inflammation to Melanoma Progression in Humans and Mice. Int. J. Mol. Sci. 2019, 20, 169. [Google Scholar] [CrossRef]
- Zhou, W.; Ke, S.Q.; Huang, Z.; Flavahan, W.; Fang, X.; Paul, J.; Wu, L.; Sloan, A.E.; McLendon, R.E.; Li, X.; et al. Periostin secreted by glioblastoma stem cells recruits M2 tumour-associated macrophages and pro-motes malignant growth. Nat. Cell Biol. 2015, 17, 170–182. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef]
- Mariathasan, S.; Turley, S.J.; Nickles, D.; Castiglioni, A.; Yuen, K.; Wang, Y.; Kadel, E.E., III; Koeppen, H.; Astarita, J.L.; Cubas, R.; et al. TGFβ attenuates tumour response to PD-L1 blockade by contributing to exclusion of T cells. Nature 2018, 554, 544–548. [Google Scholar] [CrossRef]
- Hanks, B.; Holtzhausen, A.; Evans, K.S.; Jamieson, R.; Gimpel, P.; Campbell, O.M.; Hector-Greene, M.; Sun, L.; Tewari, A.; George, A.; et al. Type III TGF-β receptor downregulation generates an immunotolerant tumor microenvironment. J. Clin. Investig. 2013, 123, 3925–3940. [Google Scholar] [CrossRef] [PubMed]
- Finger, E.C.; Cheng, C.-F.; Williams, T.R.; Rankin, E.B.; Bedogni, B.; Tachiki, L.; Spong, S.; Giaccia, A.J.; Powell, M.B. CTGF is a therapeutic target for metastatic melanoma. Oncogene 2013, 33, 1093–1100. [Google Scholar] [CrossRef]
- Tseng, Y.J.; Lee, C.H.; Chen, W.Y.; Yang, J.L.; Tzeng, H.T. Inhibition of PAI-1 Blocks PD-L1 Endocytosis and Improves the Response of Melanoma Cells to Immune Checkpoint Blockade. J. Investig. Dermatol. 2021, 141, 2690–2698.e6. [Google Scholar] [CrossRef]
- Chen, S.; Morine, Y.; Tokuda, K.; Yamada, S.; Saito, Y.; Nishi, M.; Ikemoto, T.; Shimada, M. Cancer-associated fibroblast-induced M2-polarized macrophages promote hepatocellular carcinoma progression via the plasminogen activator inhibitor-1 pathway. Int. J. Oncol. 2021, 59, 59. [Google Scholar] [CrossRef]
- Calon, A.; Espinet, E.; Palomo-Ponce, S.; Tauriello, D.V.; Iglesias, M.; Céspedes, M.V.; Sevillano, M.; Nadal, C.; Jung, P.; Zhang, X.H.-F.; et al. Dependency of Colorectal Cancer on a TGF-β-Driven Program in Stromal Cells for Metastasis Initiation. Cancer Cell 2012, 22, 571–584. [Google Scholar] [CrossRef]
- Lind, H.; Gameiro, S.R.; Jochems, C.; Donahue, R.N.; Strauss, J.; Gulley, J.L.; Palena, C.; Schlom, J. Dual targeting of TGF-β and PD-L1 via a bifunctional anti-PD-L1/TGF-βRII agent: Status of preclinical and clinical advances. J. Immunother. Cancer 2020, 8, 1426519. [Google Scholar] [CrossRef] [PubMed]
- Birrer, M.J.; Mileshkin, L.R.; Fujiwara, K.; Ray-Coquard, I.; Alexandre, J.; Okamoto, A.; Mirza, M.R.; Gulley, J.L.; Jehl, G.; Ramage, S.; et al. 879TiP Phase II study of bintrafusp alfa, a bifunctional fusion protein targeting TGF-β and PD-L1, in platinum-experienced advanced cervical cancer. Ann. Oncol. 2020, 31, S644–S645. [Google Scholar] [CrossRef]
- Pinner, S.; Jordan, P.; Sharrock, K.; Bazley, L.; Collinson, L.; Marais, R.; Bonvin, E.; Goding, C.; Sahai, E. Intravital imaging reveals transient changes in pigment production and Brn2 expression during meta-static melanoma dissemination. Cancer Res. 2009, 69, 7969–7977. [Google Scholar] [CrossRef]
- Lüönd, F.; Pirkl, M.; Hisano, M.; Prestigiacomo, V.; Kalathur, R.K.; Beerenwinkel, N.; Christofori, G. Hierarchy of TGFβ/SMAD, Hippo/YAP/TAZ, and Wnt/β-catenin signaling in melanoma phenotype switching. Life Sci. Alliance 2022, 5, 202101010. [Google Scholar] [CrossRef]
- Helfrich, I.; Ullrich, N.; Zigrino, P.; Schadendorf, D. Primary tumor versus metastasis: New experimental models for studies on cancer cell homing and metastasis in melanoma. Pigment. Cell Melanoma Res. 2014, 27, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Persson, U.; Izumi, H.; Souchelnytskyi, S.; Itoh, S.; Grimsby, S.; Engström, U.; Heldin, C.-H.; Funa, K.; ten Dijke, P. The L45 loop in type I receptors for TGF-β family members is a critical determinant in specifying Smad isoform activation. FEBS Lett. 1998, 434, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Bankhead, P.; Loughrey, M.B.; Fernández, J.A.; Dombrowski, Y.; McArt, D.G.; Dunne, P.D.; McQuaid, S.; Gray, R.T.; Murray, L.J.; Coleman, H.G.; et al. QuPath: Open source software for digital pathology image analysis. Sci. Rep. 2017, 7, 16878. [Google Scholar] [CrossRef] [PubMed]
- Dijkstra, J.J.; Neikes, H.K.; Rezaeifard, S.; Ma, X.; Voest, E.E.; Tauriello, D.V.; Vermeulen, M. Multiomics of Colorectal Cancer Organoids Reveals Putative Mediators of Cancer Progression Re-sulting from SMAD4 Inactivation. J. Proteome Res. 2023, 22, 138–151. [Google Scholar] [CrossRef]
- Rappsilber, J.; Mann, M.; Ishihama, Y. Protocol for micro-purification, enrichment, pre-fractionation and storage of peptides for proteomics using Stage Tips. Nat. Protoc. 2007, 2, 1896–1906. [Google Scholar] [CrossRef] [PubMed]
- Lau, H.T.; Suh, H.W.; Golkowski, M.; Ong, S.E. Comparing SILAC- and stable isotope dimethyl-labeling ap-proaches for quantitative proteomics. J. Proteome Res. 2014, 13, 4164–4174. [Google Scholar] [CrossRef]
- Cox, J.; Mann, M. MaxQuant enables high peptide identification rates, individualized p.p.b.-range mass accuracies and proteome-wide protein quantification. Nat. Biotechnol. 2008, 26, 1367–1372. [Google Scholar] [CrossRef]
- Ritchie, M.E.; Phipson, B.; Wu, D.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015, 43, e47. [Google Scholar] [CrossRef]
- Kanehisa, M.; Furumichi, M.; Tanabe, M.; Sato, Y.; Morishima, K. KEGG: New perspectives on genomes, pathways, diseases and drugs. Nucleic Acids Res. 2017, 45, D353–D361. [Google Scholar] [CrossRef]
- Gene Ontology Consortium. The Gene Ontology Resource: 20 years and still GOing strong. Nucleic Acids Res. 2019, 47, D330–D338. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marvin, D.L.; Dijkstra, J.; Zulfiqar, R.M.; Vermeulen, M.; ten Dijke, P.; Ritsma, L. TGF-β Type I Receptor Signaling in Melanoma Liver Metastases Increases Metastatic Outgrowth. Int. J. Mol. Sci. 2023, 24, 8676. https://doi.org/10.3390/ijms24108676
Marvin DL, Dijkstra J, Zulfiqar RM, Vermeulen M, ten Dijke P, Ritsma L. TGF-β Type I Receptor Signaling in Melanoma Liver Metastases Increases Metastatic Outgrowth. International Journal of Molecular Sciences. 2023; 24(10):8676. https://doi.org/10.3390/ijms24108676
Chicago/Turabian StyleMarvin, Dieuwke L., Jelmer Dijkstra, Rabia M. Zulfiqar, Michiel Vermeulen, Peter ten Dijke, and Laila Ritsma. 2023. "TGF-β Type I Receptor Signaling in Melanoma Liver Metastases Increases Metastatic Outgrowth" International Journal of Molecular Sciences 24, no. 10: 8676. https://doi.org/10.3390/ijms24108676





