A Potential ABA Analog to Increase Drought Tolerance in Arabidopsis thaliana
Abstract
:1. Introduction
2. Results
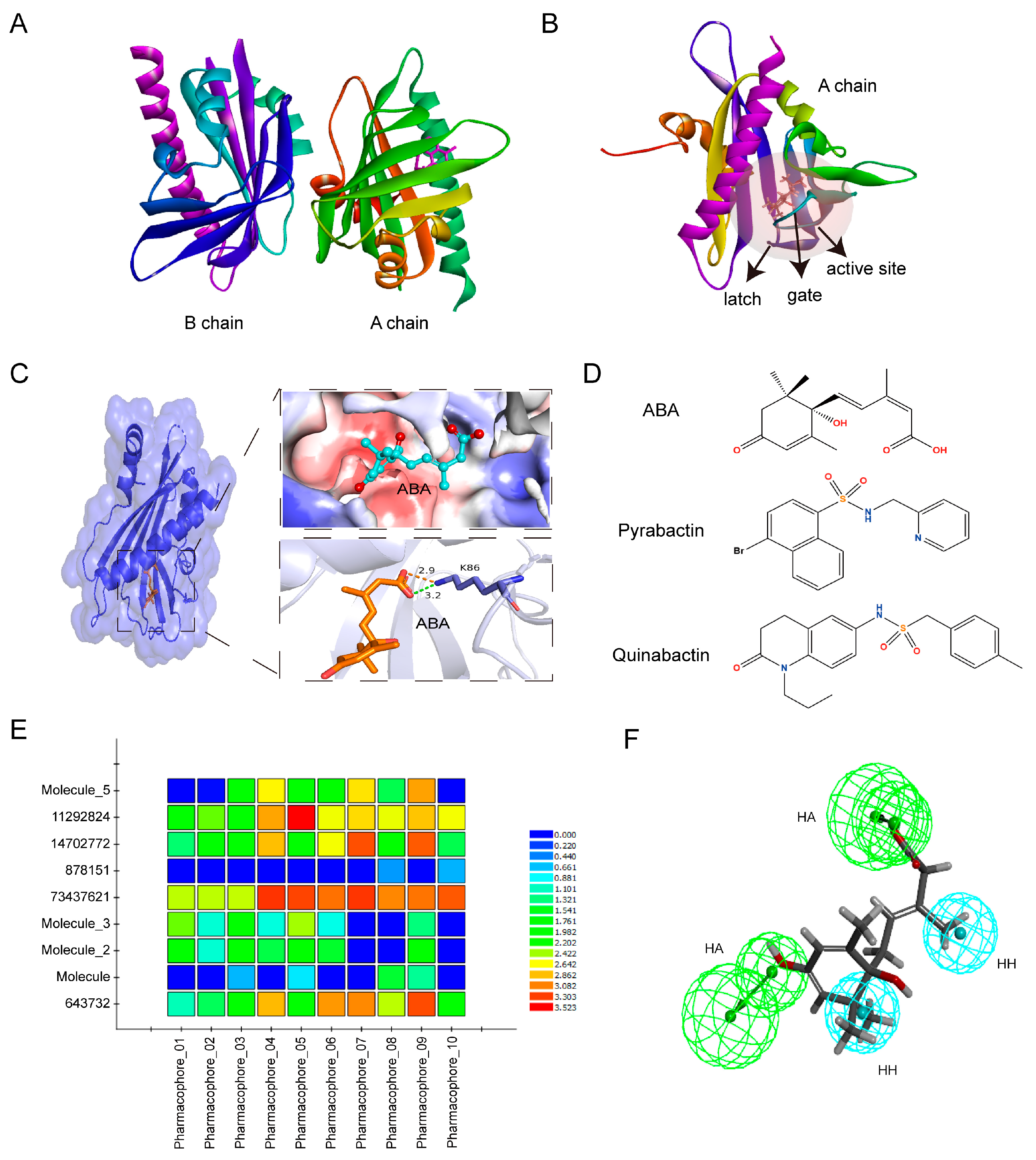
2.1. Ten Small Molecule Compounds Are Screened as Potential ABA Analogues through Virtual Screening Techniques
2.2. SLG1 Inhibits the Seedlings Growth of A. thaliana and Activates the Expression of ABA-Related Genes
2.3. SLG1 Reduces Water Loss and Enhances Drought Resistance
2.4. SLG1 Is a Potential agonist of Multiple ABA Receptors
2.5. Structural Basis of SLG1 Recognition by ABA Receptors
2.6. Molecular Dynamics Simulations of SLG1 Recognition by ABA Receptors
3. Discussion
4. Materials and Methods
4.1. Preparation Crystal Structure of PYL1 Protein
4.2. Preparation of Small Molecule Compounds
4.3. Molecular Docking Screening
4.4. Pharmacophore Screening
4.5. Phenotypic Assays
4.6. Gene Expression Analysis
4.7. Stability Measurement of ABA and SLG1
4.8. Yeast Two-Hybrid Assay
4.9. Agonist/Receptor-Mediated Phosphatase Inhibition Assays
4.10. Molecular Docking
4.11. Molecular Dynamics Simulations
4.12. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhu, J.K. Salt and drought stress signal transduction in plants. Annu. Rev. Plant Biol. 2002, 53, 247–273. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Li, G.-J.; Bressan, R.A.; Song, C.-P.; Zhu, J.-K.; Zhao, Y. Abscisic acid dynamics, signaling, and functions in plants. J. Integr. Plant Biol. 2020, 62, 25–54. [Google Scholar] [CrossRef] [PubMed]
- Kishor, P.B.K.; Tiozon, R.N.; Fernie, A.R.; Sreenivasulu, N. Abscisic acid and its role in the modulation of plant growth, development, and yield stability. Trends Plant Sci. 2022, 27, 1283–1295. [Google Scholar] [CrossRef] [PubMed]
- Chater, C.C.C.; Oliver, J.; Casson, S.; Gray, J.E. Putting the brakes on: Abscisic acid as a central environmental regulator of stomatal development. New Phytol. 2014, 202, 376–391. [Google Scholar] [CrossRef]
- Park, S.Y.; Fung, P.; Nishimura, N.; Jensen, D.R.; Fujii, H.; Zhao, Y.; Lumba, S.; Santiago, J.; Rodrigues, A.; Chow, T.F.; et al. Abscisic acid inhibits type 2C protein phosphatases via the PYR/PYL family of START proteins. Science 2009, 324, 1068–1071. [Google Scholar] [CrossRef]
- Melcher, K.; Ng, L.-M.; Zhou, X.E.; Soon, F.-F.; Xu, Y.; Suino-Powell, K.M.; Park, S.-Y.; Weiner, J.J.; Fujii, H.; Chinnusamy, V.; et al. A gate–latch–lock mechanism for hormone signalling by abscisic acid receptors. Nature 2009, 462, 602–608. [Google Scholar] [CrossRef]
- Yin, P.; Fan, H.; Hao, Q.; Yuan, X.; Wu, D.; Pang, Y.; Yan, C.; Li, W.; Wang, J.; Yan, N. Structural insights into the mechanism of abscisic acid signaling by PYL proteins. Nat. Struct. Mol. Biol. 2009, 16, 1230–1236. [Google Scholar] [CrossRef]
- Hauser, F.; Waadt, R.; Schroeder, J.I. Evolution of abscisic acid synthesis and signaling mechanisms. Curr. Biol. 2011, 21, R346–R355. [Google Scholar] [CrossRef]
- Dittrich, M.; Mueller, H.M.; Bauer, H.; Peirats-Llobet, M.; Rodriguez, P.L.; Geilfus, C.-M.; Carpentier, S.C.; Al Rasheid, K.A.S.; Kollist, H.; Merilo, E.; et al. The role of Arabidopsis ABA receptors from the PYR/PYL/RCAR family in stomatal acclimation and closure signal integration. Nat. Plants 2019, 5, 1002–1011. [Google Scholar] [CrossRef]
- Li, W.; Wang, L.; Sheng, X.; Yan, C.; Zhou, R.; Hang, J.; Yin, P.; Yan, N. Molecular basis for the selective and ABA-independent inhibition of PP2CA by PYL13. Cell Res. 2013, 23, 1369–1379. [Google Scholar] [CrossRef]
- Hao, Q.; Yin, P.; Li, W.; Wang, L.; Yan, C.; Lin, Z.; Wu, J.Z.; Wang, J.; Yan, S.F.; Yan, N. The Molecular Basis of ABA-Independent Inhibition of PP2Cs by a Subclass of PYL Proteins. Mol. Cell 2011, 42, 662–672. [Google Scholar] [CrossRef]
- Cutler, A.J.; Krochko, J.E. Formation and breakdown of ABA. Trends Plant Sci. 1999, 4, 472–478. [Google Scholar] [CrossRef] [PubMed]
- Hewage, K.A.H.; Yang, J.F.; Wang, D.; Hao, G.F.; Yang, G.F.; Zhu, J.K. Chemical Manipulation of Abscisic Acid Signaling: A New Approach to Abiotic and Biotic Stress Management in Agriculture. Adv. Sci. 2020, 7, 2001265. [Google Scholar] [CrossRef] [PubMed]
- Cao, M.; Liu, X.; Zhang, Y.; Xue, X.; Zhou, X.E.; Melcher, K.; Gao, P.; Wang, F.; Zeng, L.; Zhao, Y.; et al. An ABA-mimicking ligand that reduces water loss and promotes drought resistance in plants. Cell Res. 2013, 23, 1043–1054. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, M.; Peterson, F.C.; Defries, A.; Park, S.Y.; Endo, A.; Nambara, E.; Volkman, B.F.; Cutler, S.R. Activation of dimeric ABA receptors elicits guard cell closure, ABA-regulated gene expression, and drought tolerance. Proc. Natl. Acad. Sci. USA 2013, 110, 12132–12137. [Google Scholar] [CrossRef] [PubMed]
- Min, M.K.; Kim, R.; Moon, S.J.; Lee, Y.; Han, S.; Lee, S.; Kim, B.G. Selection and functional identification of a synthetic partial ABA agonist, S7. Sci. Rep. 2020, 10, 4. [Google Scholar] [CrossRef]
- Vora, J.; Patel, S.; Athar, M.; Sinha, S.; Chhabria, M.T.; Jha, P.C.; Shrivastava, N. Pharmacophore modeling, molecular docking and molecular dynamics simulation for screening and identifying anti-dengue phytocompounds. J. Biomol. Struct. Dyn. 2020, 38, 1726–1740. [Google Scholar] [CrossRef] [PubMed]
- Vaidya, A.S.; Helander, J.D.M.; Peterson, F.C.; Elzinga, D.; Dejonghe, W.; Kaundal, A.; Park, S.-Y.; Xing, Z.; Mega, R.; Takeuchi, J.; et al. Dynamic control of plant water use using designed ABA receptor agonists. Science 2019, 366, eaaw8848. [Google Scholar] [CrossRef]
- Lozano-Juste, J.; Infantes, L.; Garcia-Maquilon, I.; Ruiz-Partida, R.; Merilo, E.; Benavente, J.L.; Velazquez-Campoy, A.; Coego, A.; Bono, M.; Forment, J.; et al. Structure-guided engineering of a receptor-agonist pair for inducible activation of the ABA adaptive response to drought. Sci. Adv. 2023, 9, eade9948. [Google Scholar] [CrossRef]
- Choi, H.-I.; Hong, J.-H.; Ha, J.-O.; Kang, J.-Y.; Kim, S.Y. ABFs, a Family of ABA-responsive Element Binding Factors. J. Biol. Chem. 2000, 275, 1723–1730. [Google Scholar] [CrossRef]
- Yamaguchi-Shinozaki, K.; Shinozaki, K. Arabidopsis DNA Encoding Two Desiccation-Responsive rd29 Genes. Plant Physiol. 1993, 101, 1119–1120. [Google Scholar] [CrossRef] [PubMed]
- Kurkela, S.; Franck, M. Cloning and characterization of a cold-and ABA-inducible Arabidopsis gene. Plant Mol. Biol. 1990, 15, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Strizhov, N.; Ábrahám, E.; Ökrész, L.; Blickling, S.; Zilberstein, A.; Schell, J.; Koncz, C.; Szabados, L. Differential expression of two P5CS genes controlling proline accumulation during salt-stress requires ABA and is regulated by ABA1, ABI1 and AXR2 in Arabidopsis. Plant J. 1997, 12, 557–569. [Google Scholar] [PubMed]
- Zhang, X.L.; Jiang, L.; Xin, Q.; Liu, Y.; Tan, J.X.; Chen, Z.Z. Structural basis and functions of abscisic acid receptors PYLs. Front. Plant Sci. 2015, 6, 88. [Google Scholar] [CrossRef]
- Muhseen, Z.T.; Hameed, A.R.; Al-Bhadly, O.; Ahmad, S.; Li, G. Natural products for treatment of Plasmodium falciparum malaria: An integrated computational approach. Comput. Biol. Med. 2021, 134, 104415. [Google Scholar] [CrossRef]
- Zhao, Y.; Chow, T.F.; Puckrin, R.S.; Alfred, S.E.; Korir, A.K.; Larive, C.K.; Cutler, S.R. Chemical genetic interrogation of natural variation uncovers a molecule that is glycoactivated. Nat. Chem. Biol. 2007, 3, 716–721. [Google Scholar] [CrossRef]
- Cao, M.-J.; Zhang, Y.-L.; Liu, X.; Huang, H.; Zhou, X.E.; Wang, W.-L.; Zeng, A.; Zhao, C.-Z.; Si, T.; Du, J.; et al. Combining chemical and genetic approaches to increase drought resistance in plants. Nat. Commun. 2017, 8, 1183. [Google Scholar] [CrossRef]
- Lv, N.; Kong, Q.; Zhang, H.; Li, J. Discovery of novel Staphylococcus aureus penicillin binding protein 2a inhibitors by multistep virtual screening and biological evaluation. Bioorganic Med. Chem. Lett. 2021, 41, 128001. [Google Scholar] [CrossRef]
- Liu, C.; Yin, J.; Yao, J.; Xu, Z.; Tao, Y.; Zhang, H. Pharmacophore-Based Virtual Screening Toward the Discovery of Novel Anti-echinococcal Compounds. Front. Cell. Infect. Microbiol. 2020, 10, 118. [Google Scholar] [CrossRef]
- Ye, Y.; Zhou, L.; Liu, X.; Liu, H.; Li, D.; Cao, M.; Chen, H.; Xu, L.; Zhu, J.K.; Zhao, Y. A Novel Chemical Inhibitor of ABA Signaling Targets All ABA Receptors. Plant Physiol. 2017, 173, 2356–2369. [Google Scholar] [CrossRef]
- Nemoto, K.; Kagawa, M.; Nozawa, A.; Hasegawa, Y.; Hayashi, M.; Imai, K.; Tomii, K.; Sawasaki, T. Identification of new abscisic acid receptor agonists using a wheat cell-free based drug screening system. Sci. Rep. 2018, 8, 4268. [Google Scholar] [CrossRef] [PubMed]
- Saito, S.; Hirai, N.; Matsumoto, C.; Ohigashi, H.; Ohta, D.; Sakata, K.; Mizutani, M. Arabidopsis CYP707As encode (+)-abscisic acid 8’-hydroxylase, a key enzyme in the oxidative catabolism of abscisic acid. Plant Physiol. 2004, 134, 1439–1449. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, P.L. Abscisic Acid Catabolism Generates Phaseic Acid, a Molecule Able to Activate a Subset of ABA Receptors. Mol. Plant 2016, 9, 1448–1450. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Kong, X.; Huang, Q.; Zhang, Q.; Ge, H.; Zhang, L.; Li, G.; Peng, L.; Liu, Z.; Wang, J.; et al. CARK1 phosphorylates subfamily III members of ABA receptors. J. Exp. Bot. 2019, 70, 519–528. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Yue, X.; Wang, R.; Zhang, Y. Ethylene mediates UV-B-induced stomatal closure via peroxidase-dependent hydrogen peroxide synthesis in Vicia faba L. J. Exp. Bot. 2011, 62, 2657–2666. [Google Scholar] [CrossRef]
- Fu, Q.; Wang, Y.; Yang, J.; Jiao, Y.; Li, W.; Yang, F.; Yin, X.; Shang, B.; Liu, R.; Zhang, Y.; et al. Plasmopara viticola RxLR effector PvAvh77 triggers cell death and governs immunity responses in grapevine. J. Exp. Bot. 2023, 74, 2047–2066. [Google Scholar] [CrossRef]
- Ma, Y.; Szostkiewicz, I.; Korte, A.; Moes, D.; Yang, Y.; Christmann, A.; Grill, E. Regulators of PP2C Phosphatase Activity Function as Abscisic Acid Sensors. Science 2009, 324, 1064–1068. [Google Scholar] [CrossRef]
- Páll, S.; Zhmurov, A.; Bauer, P.; Abraham, M.; Lundborg, M.; Gray, A.; Hess, B.; Lindahl, E. Heterogeneous parallelization and acceleration of molecular dynamics simulations in GROMACS. J. Chem. Phys. 2020, 153, 134110. [Google Scholar] [CrossRef]






Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, R.; Liang, G.; Gong, J.; Wang, J.; Zhang, Y.; Hao, Z.; Li, G. A Potential ABA Analog to Increase Drought Tolerance in Arabidopsis thaliana. Int. J. Mol. Sci. 2023, 24, 8783. https://doi.org/10.3390/ijms24108783
Liu R, Liang G, Gong J, Wang J, Zhang Y, Hao Z, Li G. A Potential ABA Analog to Increase Drought Tolerance in Arabidopsis thaliana. International Journal of Molecular Sciences. 2023; 24(10):8783. https://doi.org/10.3390/ijms24108783
Chicago/Turabian StyleLiu, Ruiqi, Guoyan Liang, Jiaxin Gong, Jiali Wang, Yanjie Zhang, Zhiqiang Hao, and Guanglin Li. 2023. "A Potential ABA Analog to Increase Drought Tolerance in Arabidopsis thaliana" International Journal of Molecular Sciences 24, no. 10: 8783. https://doi.org/10.3390/ijms24108783




