Serum Interleukin-36 α as a Candidate Biomarker to Distinguish Behçet’s Syndrome and Psoriatic Arthritis
Abstract
:1. Introduction
2. Results
2.1. Patients Characteristics
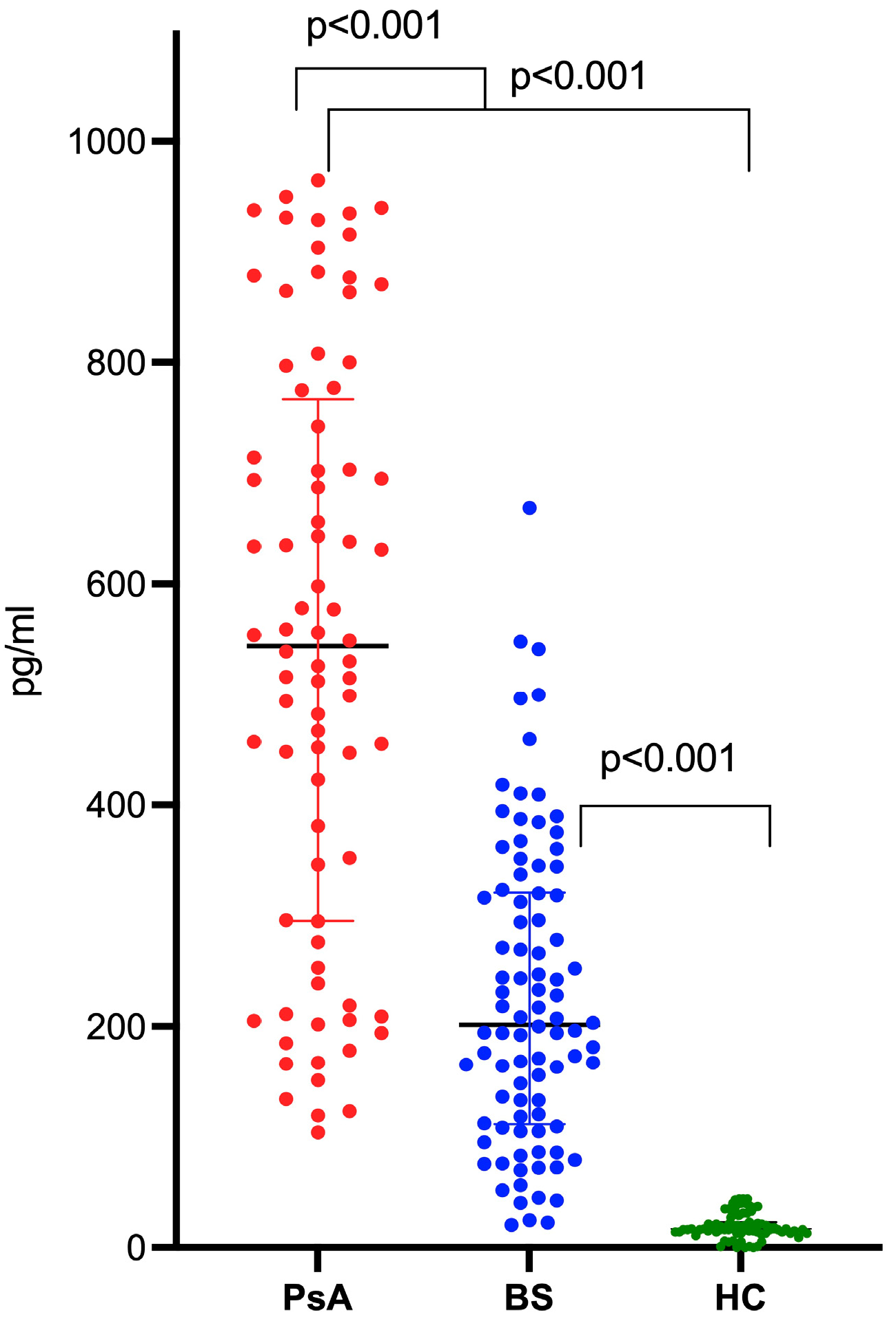
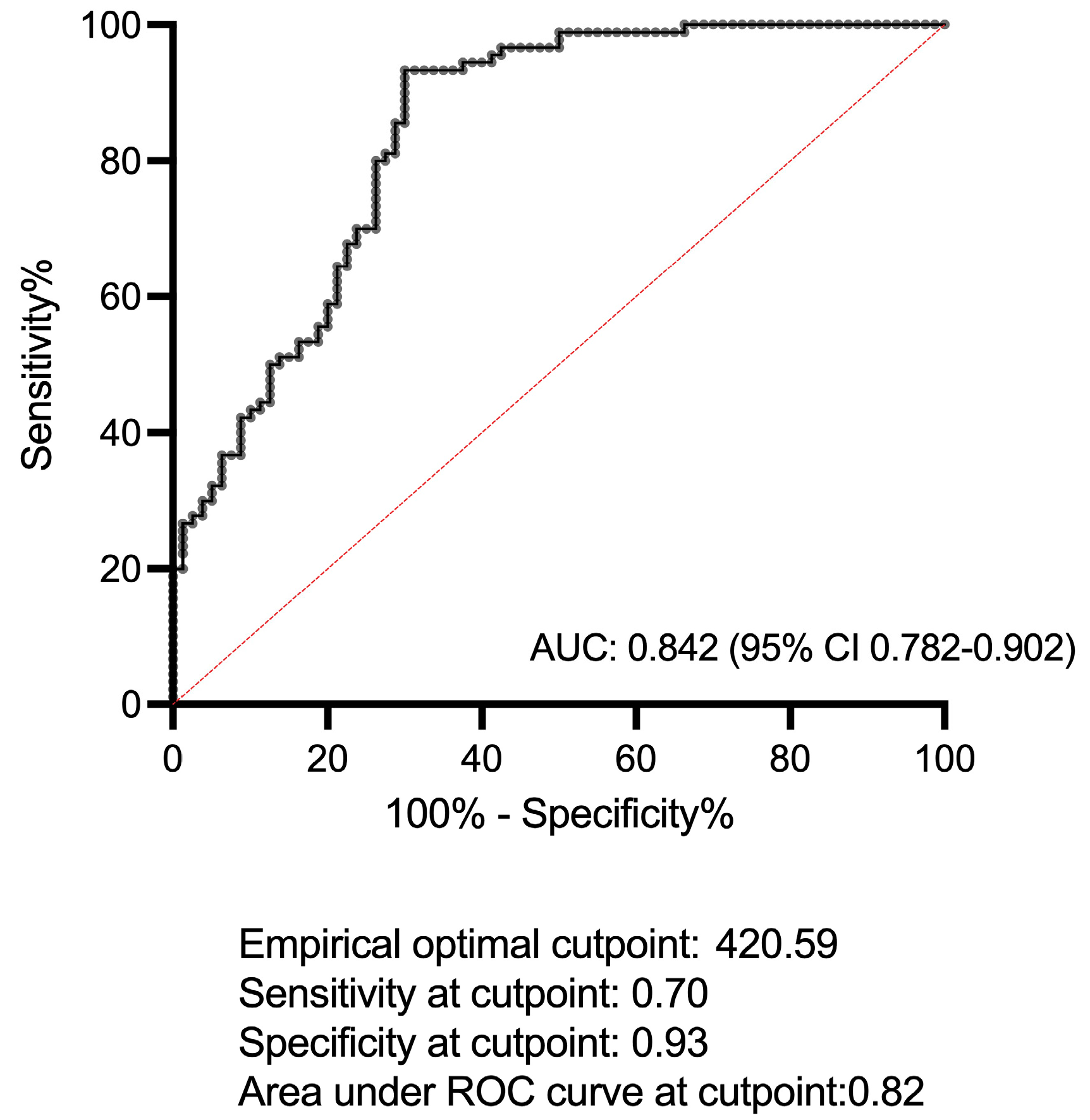
2.2. IL-36α Levels in Patients with BS, PsA, and in HCs
2.3. Variations in IL-36α Levels among BS Patients
3. Discussion
4. Materials and Methods
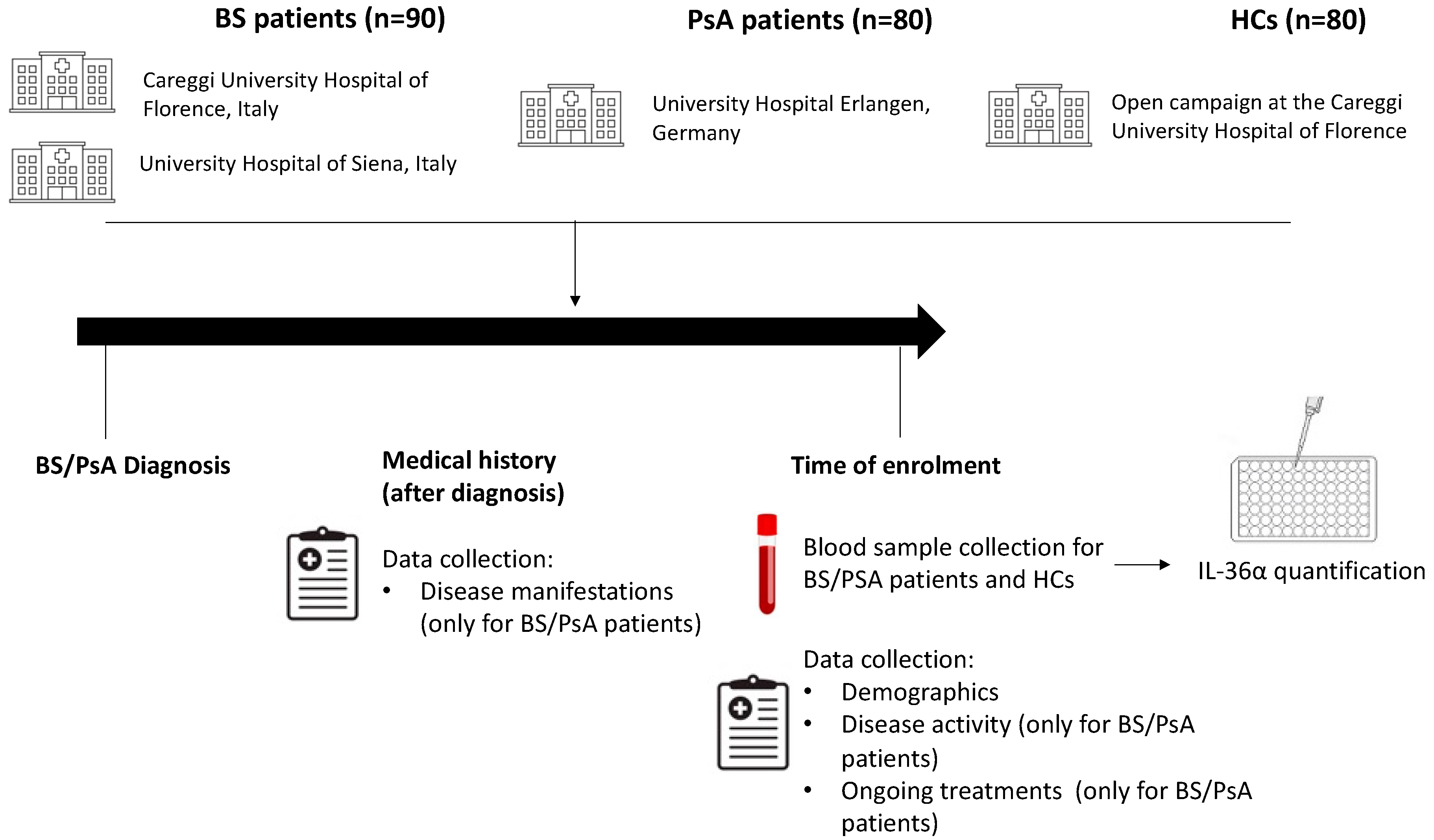
4.1. Study Design and Population
4.2. Data and Sample Collection
4.3. IL-36α Quantification
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| ID, Sex | Age | Disease Manifestations | HLAB51 | Active Disease | Ongoing Therapy | IL-36α Levels (pg/mL) |
|---|---|---|---|---|---|---|
| 1, M | 36 | OA, cutaneous, articular, anterior uveitis, gastrointestinal | Positive | Yes | csDMARDs | 278.3 |
| 2, F | 49 | OA, cutaneous, articular, anterior uveitis | Positive | No | csDMARDs | 233.2 |
| 3, F | 42 | OA, cutaneous, articular, gastrointestinal, positive pathergy test | Positive | No | Corticosteroids | 155.9 |
| 4, M | 56 | OA, cutaneous, articular, anterior uveitis | Positive | No | bDMARDs | 164.1 |
| 5, F | 33 | OA, cutaneous, articular, gastrointestinal, positive pathergy test | Positive | No | Corticosteroids | 360.4 |
| 6, M | 64 | OA, cutaneous, articular, anterior uveitis, gastrointestinal | Positive | Yes | csDMARDs | 387.5 |
| 7, F | 38 | OA, cutaneous, articular, positive pathergy test | Negative | Yes | csDMARDs | 186.7 |
| 8, F | 59 | OA, cutaneous, articular, anterior uveitis | Negative | No | Corticosteroids | 203.2 |
References
- Bettiol, A.; Hatemi, G.; Vannozzi, L.; Barilaro, A.; Prisco, D.; Emmi, G. Treating the Different Phenotypes of Behcet’s Syndrome. Front. Immunol. 2019, 10, 2830. [Google Scholar] [CrossRef] [PubMed]
- Bettiol, A.; Prisco, D.; Emmi, G. Behcet: The syndrome. Rheumatology 2020, 59, iii101–iii107. [Google Scholar] [CrossRef]
- Bettiol, A.; Alibaz-Oner, F.; Direskeneli, H.; Hatemi, G.; Saadoun, D.; Seyahi, E.; Prisco, D.; Emmi, G. Vascular Behcet syndrome: From pathogenesis to treatment. Nat. Rev. Rheumatol. 2023, 19, 111–126. [Google Scholar] [CrossRef] [PubMed]
- Kiafar, M.; Faezi, S.T.; Kasaeian, A.; Baghdadi, A.; Kakaei, S.; Mousavi, S.A.; Nejadhosseinian, M.; Shahram, F.; Ghodsi, S.Z.; Shams, H.; et al. Diagnosis of Behcet’s disease: Clinical characteristics, diagnostic criteria, and differential diagnoses. BMC Rheumatol. 2021, 5, 2. [Google Scholar] [CrossRef] [PubMed]
- Tursen, U.; Gurler, A.; Boyvat, A. Evaluation of clinical findings according to sex in 2313 Turkish patients with Behcet’s disease. Int. J. Dermatol. 2003, 42, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Hatemi, G.; Christensen, R.; Bang, D.; Bodaghi, B.; Celik, A.F.; Fortune, F.; Gaudric, J.; Gul, A.; Kotter, I.; Leccese, P.; et al. 2018 update of the EULAR recommendations for the management of Behcet’s syndrome. Ann. Rheum. Dis. 2018, 77, 808–818. [Google Scholar] [CrossRef]
- Seyahi, E. Phenotypes in Behcet’s syndrome. Intern. Emerg. Med. 2019, 14, 677–689. [Google Scholar] [CrossRef]
- International Team for the Revision of the International Criteria for Behcet’s, D. The International Criteria for Behcet’s Disease (ICBD): A collaborative study of 27 countries on the sensitivity and specificity of the new criteria. J. Eur. Acad. Dermatol. Venereol. 2014, 28, 338–347. [Google Scholar] [CrossRef]
- Di Scala, G.; Bettiol, A.; Cojan, R.D.; Finocchi, M.; Silvestri, E.; Emmi, G. Efficacy of the anti-IL 17 secukinumab in refractory Behcet’s syndrome: A preliminary study. J. Autoimmun. 2019, 97, 108–113. [Google Scholar] [CrossRef]
- Fagni, F.; Bettiol, A.; Talarico, R.; Lopalco, G.; Silvestri, E.; Urban, M.L.; Russo, P.A.J.; Di Scala, G.; Emmi, G.; Prisco, D. Long-term effectiveness and safety of secukinumab for treatment of refractory mucosal and articular Behcet’s phenotype: A multicentre study. Ann. Rheum. Dis. 2020, 79, 1098–1104. [Google Scholar] [CrossRef]
- Olivieri, I.; Padula, A.; D’Angelo, S.; Cutro, M.S. Psoriatic arthritis sine psoriasis. J. Rheumatol. Suppl. 2009, 83, 28–29. [Google Scholar] [CrossRef] [PubMed]
- Peluso, R.; Iervolino, S.; Vitiello, M.; Bruner, V.; Lupoli, G.; Di Minno, M.N. Extra-articular manifestations in psoriatic arthritis patients. Clin. Rheumatol. 2015, 34, 745–753. [Google Scholar] [CrossRef] [PubMed]
- Hahn, H.J.; Kwak, S.G.; Kim, D.K.; Kim, J.Y. Association of Behcet disease with psoriasis and psoriatic arthritis. Sci. Rep. 2021, 11, 2531. [Google Scholar] [CrossRef] [PubMed]
- Taylor, W.; Gladman, D.; Helliwell, P.; Marchesoni, A.; Mease, P.; Mielants, H.; Group, C.S. Classification criteria for psoriatic arthritis: Development of new criteria from a large international study. Arthritis Rheum. 2006, 54, 2665–2673. [Google Scholar] [CrossRef] [PubMed]
- Novelli, L.; Lubrano, E.; Venerito, V.; Perrotta, F.M.; Marando, F.; Curradi, G.; Iannone, F. Extra-Articular Manifestations and Comorbidities in Psoriatic Disease: A Journey Into the Immunologic Crosstalk. Front. Med. 2021, 8, 737079. [Google Scholar] [CrossRef] [PubMed]
- van der Houwen, T.B.; van Hagen, P.M.; van Laar, J.A.M. Immunopathogenesis of Behcet’s disease and treatment modalities. Semin. Arthritis Rheum. 2022, 52, 151956. [Google Scholar] [CrossRef]
- Gossec, L.; Baraliakos, X.; Kerschbaumer, A.; de Wit, M.; McInnes, I.; Dougados, M.; Primdahl, J.; McGonagle, D.G.; Aletaha, D.; Balanescu, A.; et al. EULAR recommendations for the management of psoriatic arthritis with pharmacological therapies: 2019 update. Ann. Rheum. Dis. 2020, 79, 700–712. [Google Scholar] [CrossRef]
- Hu, C.J.; Pan, J.B.; Song, G.; Wen, X.T.; Wu, Z.Y.; Chen, S.; Mo, W.X.; Zhang, F.C.; Qian, J.; Zhu, H.; et al. Identification of Novel Biomarkers for Behcet Disease Diagnosis Using Human Proteome Microarray Approach. Mol. Cell. Proteom. 2017, 16, 147–156. [Google Scholar] [CrossRef]
- Cavalli, G.; Colafrancesco, S.; Emmi, G.; Imazio, M.; Lopalco, G.; Maggio, M.C.; Sota, J.; Dinarello, C.A. Interleukin 1alpha: A comprehensive review on the role of IL-1alpha in the pathogenesis and treatment of autoimmune and inflammatory diseases. Autoimmun. Rev. 2021, 20, 102763. [Google Scholar] [CrossRef]
- Magne, D.; Palmer, G.; Barton, J.L.; Mezin, F.; Talabot-Ayer, D.; Bas, S.; Duffy, T.; Noger, M.; Guerne, P.A.; Nicklin, M.J.; et al. The new IL-1 family member IL-1F8 stimulates production of inflammatory mediators by synovial fibroblasts and articular chondrocytes. Arthritis Res. Ther. 2006, 8, R80. [Google Scholar] [CrossRef]
- Boutet, M.A.; Bart, G.; Penhoat, M.; Amiaud, J.; Brulin, B.; Charrier, C.; Morel, F.; Lecron, J.C.; Rolli-Derkinderen, M.; Bourreille, A.; et al. Distinct expression of interleukin (IL)-36alpha, beta and gamma, their antagonist IL-36Ra and IL-38 in psoriasis, rheumatoid arthritis and Crohn’s disease. Clin. Exp. Immunol. 2016, 184, 159–173. [Google Scholar] [CrossRef]
- Zhou, Z.Y.; Chen, S.L.; Shen, N.; Lu, Y. Cytokines and Behcet’s disease. Autoimmun. Rev. 2012, 11, 699–704. [Google Scholar] [CrossRef]
- Emmi, G.; Talarico, R.; Lopalco, G.; Cimaz, R.; Cantini, F.; Viapiana, O.; Olivieri, I.; Goldoni, M.; Vitale, A.; Silvestri, E.; et al. Efficacy and safety profile of anti-interleukin-1 treatment in Behcet’s disease: A multicenter retrospective study. Clin. Rheumatol. 2016, 35, 1281–1286. [Google Scholar] [CrossRef]
- Fabiani, C.; Vitale, A.; Emmi, G.; Lopalco, G.; Vannozzi, L.; Guerriero, S.; Gentileschi, S.; Bacherini, D.; Franceschini, R.; Frediani, B.; et al. Interleukin (IL)-1 inhibition with anakinra and canakinumab in Behcet’s disease-related uveitis: A multicenter retrospective observational study. Clin. Rheumatol. 2017, 36, 191–197. [Google Scholar] [CrossRef]
- Fabiani, C.; Vitale, A.; Rigante, D.; Emmi, G.; Lopalco, G.; Di Scala, G.; Sota, J.; Orlando, I.; Franceschini, R.; Frediani, B.; et al. The Presence of Uveitis Is Associated with a Sustained Response to the Interleukin (IL)-1 Inhibitors Anakinra and Canakinumab in Behcet’s Disease. Ocul. Immunol. Inflamm. 2020, 28, 298–304. [Google Scholar] [CrossRef]
- Bettiol, A.; Silvestri, E.; Di Scala, G.; Amedei, A.; Becatti, M.; Fiorillo, C.; Lopalco, G.; Salvarani, C.; Cantarini, L.; Soriano, A.; et al. The right place of interleukin-1 inhibitors in the treatment of Behcet’s syndrome: A systematic review. Rheumatol. Int. 2019, 39, 971–990. [Google Scholar] [CrossRef]
- Milora, K.A.; Fu, H.; Dubaz, O.; Jensen, L.E. Unprocessed Interleukin-36alpha Regulates Psoriasis-Like Skin Inflammation in Cooperation With Interleukin-1. J. Investig. Dermatol. 2015, 135, 2992–3000. [Google Scholar] [CrossRef]
- Alvarez, P.; Jensen, L.E. Imiquimod Treatment Causes Systemic Disease in Mice Resembling Generalized Pustular Psoriasis in an IL-1 and IL-36 Dependent Manner. Mediators Inflamm. 2016, 2016, 6756138. [Google Scholar] [CrossRef]
- Sehat, M.; Talaei, R.; Dadgostar, E.; Nikoueinejad, H.; Akbari, H. Evaluating Serum Levels of IL-33, IL-36, IL-37 and Gene Expression of IL-37 in Patients with Psoriasis Vulgaris. Iran. J. Allergy Asthma Immunol. 2018, 17, 179–187. [Google Scholar]
- Boutet, M.A.; Nerviani, A.; Lliso-Ribera, G.; Lucchesi, D.; Prediletto, E.; Ghirardi, G.M.; Goldmann, K.; Lewis, M.; Pitzalis, C. Interleukin-36 family dysregulation drives joint inflammation and therapy response in psoriatic arthritis. Rheumatology 2020, 59, 828–838. [Google Scholar] [CrossRef]
- Boutet, M.A.; Nerviani, A.; Pitzalis, C. IL-36, IL-37, and IL-38 Cytokines in Skin and Joint Inflammation: A Comprehensive Review of Their Therapeutic Potential. Int. J. Mol. Sci. 2019, 20, 1257. [Google Scholar] [CrossRef]
- Towne, J.E.; Renshaw, B.R.; Douangpanya, J.; Lipsky, B.P.; Shen, M.; Gabel, C.A.; Sims, J.E. Interleukin-36 (IL-36) ligands require processing for full agonist (IL-36alpha, IL-36beta, and IL-36gamma) or antagonist (IL-36Ra) activity. J. Biol. Chem. 2011, 286, 42594–42602. [Google Scholar] [CrossRef]
- Macleod, T.; Doble, R.; McGonagle, D.; Wasson, C.W.; Alase, A.; Stacey, M.; Wittmann, M. Neutrophil Elastase-mediated proteolysis activates the anti-inflammatory cytokine IL-36 Receptor antagonist. Sci. Rep. 2016, 6, 24880. [Google Scholar] [CrossRef]
- Bettiol, A.; Becatti, M.; Silvestri, E.; Argento, F.R.; Fini, E.; Mannucci, A.; Galora, S.; Mattioli, I.; Urban, M.L.; Malandrino, D.; et al. Neutrophil-mediated mechanisms of damage and in-vitro protective effect of colchicine in non-vascular Behcet’s syndrome. Clin. Exp. Immunol. 2021, 206, 410–421. [Google Scholar] [CrossRef]
- Li, B.; Li, G.; Yang, X.; Song, Z.; Wang, Y.; Zhang, Z. NETosis in Psoriatic Arthritis: Serum MPO-DNA Complex Level Correlates With Its Disease Activity. Front. Immunol. 2022, 13, 911347. [Google Scholar] [CrossRef]
- Becatti, M.; Emmi, G.; Silvestri, E.; Bruschi, G.; Ciucciarelli, L.; Squatrito, D.; Vaglio, A.; Taddei, N.; Abbate, R.; Emmi, L.; et al. Neutrophil Activation Promotes Fibrinogen Oxidation and Thrombus Formation in Behcet Disease. Circulation 2016, 133, 302–311. [Google Scholar] [CrossRef]
- Becatti, M.; Emmi, G.; Bettiol, A.; Silvestri, E.; Di Scala, G.; Taddei, N.; Prisco, D.; Fiorillo, C. Behcet’s syndrome as a tool to dissect the mechanisms of thrombo-inflammation: Clinical and pathogenetic aspects. Clin. Exp. Immunol. 2019, 195, 322–333. [Google Scholar] [CrossRef]
- Emmi, G.; Silvestri, E.; Cameli, A.M.; Bacherini, D.; Vannozzi, L.; Squatrito, D.; Emmi, L. Anakinra for resistant Behcet uveitis: Why not? Clin. Exp. Rheumatol. 2013, 31, 152–153. [Google Scholar]
- Todorovic, V.; Su, Z.; Putman, C.B.; Kakavas, S.J.; Salte, K.M.; McDonald, H.A.; Wetter, J.B.; Paulsboe, S.E.; Sun, Q.; Gerstein, C.E.; et al. Small Molecule IL-36gamma Antagonist as a Novel Therapeutic Approach for Plaque Psoriasis. Sci. Rep. 2019, 9, 9089. [Google Scholar] [CrossRef]
- Wolf, J.; Ferris, L.K. Anti-IL-36R antibodies, potentially useful for the treatment of psoriasis: A patent evaluation of WO2013074569. Expert. Opin. Ther. Pat. 2014, 24, 477–479. [Google Scholar] [CrossRef]
- Scheibe, K.; Backert, I.; Wirtz, S.; Hueber, A.; Schett, G.; Vieth, M.; Probst, H.C.; Bopp, T.; Neurath, M.F.; Neufert, C. IL-36R signalling activates intestinal epithelial cells and fibroblasts and promotes mucosal healing in vivo. Gut 2017, 66, 823–838. [Google Scholar] [CrossRef]
- Medina-Contreras, O.; Harusato, A.; Nishio, H.; Flannigan, K.L.; Ngo, V.; Leoni, G.; Neumann, P.A.; Geem, D.; Lili, L.N.; Ramadas, R.A.; et al. Cutting Edge: IL-36 Receptor Promotes Resolution of Intestinal Damage. J. Immunol. 2016, 196, 34–38. [Google Scholar] [CrossRef]
- Frey, S.; Derer, A.; Messbacher, M.; Baeten, D.L.P.; Bugatti, S.; Montecucco, C.; Georg Schett, G.; Hueber, A.J. The novel cytokine interleukin-36α is expressed in psoriatic and rheumatoid arthritis synovium. Ann. Rheum. Dis. 2013, 72, 156974. [Google Scholar] [CrossRef]
- Smith, D.E.; Renshaw, B.R.; Ketchem, R.R.; Kubin, M.; Garka, K.E.; Sims, J.E. Four new members expand the interleukin-1 superfamily. J. Biol. Chem. 2000, 275, 1169–1175. [Google Scholar] [CrossRef]
- Queen, D.; Ediriweera, C.; Liu, L. Function and Regulation of IL-36 Signaling in Inflammatory Diseases and Cancer Development. Front. Cell. Dev. Biol. 2019, 7, 317. [Google Scholar] [CrossRef]
- Wang, Q.; Yi, S.; Su, G.; Du, Z.; Pa, S.; Huang, X.; Cao, Q.; Yuan, G.; Kijlstra, A.; Yang, P. Changes in the Gut Microbiome Contribute to the Development of Behcet’s Disease via Adjuvant Effects. Front. Cell Dev. Biol. 2021, 9, 716760. [Google Scholar] [CrossRef]
- Liu, Y.; Fan, Q.Y.; Zhang, N.; Mi, L.Y.; Zhao, X.; Xu, K. Characterisation of circulating endothelial microparticles in Behçet’s disease: New markers of chronic endothelial damage? Clin. Exp. Rheumatol. 2023. [Google Scholar] [CrossRef]
| BS N (% Out of 90) | PsA N (% Out of 80) | Healthy Controls N (% Out of 80) | |
|---|---|---|---|
| Sex—male | 51 (56.7) | 42 (52.5) | 42 (52.5) |
| Age, years (median, IQR) | 45 (36–55) | 50 (38–66) | 45 (38–52) |
| Disease manifestations | |||
| Oral aphthosis | 84 (93.3) | 2 (2.5) | |
| Genital aphthosis | 34 (37.8) | 0 (0) | |
| Cutaneous | 73 (81.1) | - | |
| Psoriasis | - | 72 (90.0) | |
| Arthritis | 47 (52.2) | 80 (100.0%) | |
| Gastrointestinal symptoms | 34 (38.2) | 9 (11.3%) | |
| Uveitis | 31 (34.4) | 5 (6.3%) | |
| Vascular | 17 (18.9) | 7 (8.8%) * | |
| Neurological | 16 (17.8) | - | |
| HLA-B51 positivity | 40 (44.4) | - | |
| Active disease | 44 (48.9) | 37 (46.3) | |
| BDCAF (median, IQR) | 0 (0–3) | - | |
| PASI (median, IQR) | - | 1.2 (0–2.1) | |
| Ongoing immunomodulating treatment | |||
| No treatment/only NSAIDs | 0 | 10 (12.5) | |
| Corticosteroids/colchicine | 36 (40.0) | 3 (3.8) | |
| csDMARDs | 29 (32.2) | 30 (38.5) | |
| Methotrexate | 0 | 28 (35.0) | |
| Leflunomide | 0 | 2 (3.5) | |
| Biologic (±cs) DMARDs | 25 (27.8) | 37 (46.3) | |
| TNFi | 21 (23.3) | 27 (33.8) | |
| IL12/23i | 0 | 3 (3.8) | |
| IL1i | 4 (4.4) |
| With the Considered Feature | Without the Considered Feature | p-Value # | |
|---|---|---|---|
| IL-36 (pg/mL) | IL-36 (pg/mL) | ||
| Overall | 201.7 (112.7–320.2) | ||
| Disease manifestations | |||
| Genital aphthosis | 269.4 (173.2–384.5) | 176.1 (109.2–266.3) | 0.015 * |
| Cutaneous | 207.2 (132.9–323.4) | 192.2 (94.9–271.2) | 0.284 |
| Arthritis | 201.7 (120.2–320.2) | 194.2 (104.8–344.2) | 0.784 |
| Intestinal symptoms | 212.2 (104.8–360.4) | 200.2 (118.2–316.5) | 0.950 |
| Uveitis | 243.4 (192.2–345.1) | 173.2 (94.9–312.3) | 0.029 * |
| Vascular | 269.4 (208.2–351.4) | 192.2 (108.1–296.2) | 0.072 |
| Neurological | 228.3 (171.1–375.2) | 194.3 (108.1–318.3) | 0.240 |
| HLA-B51 | 229.7 (152.0–340.8) | 193.2 (94.9–312.3) | 0.237 |
| Active disease | 200.7 (106.5–379.9) | 201.7 (133.0–296.2) | 0.529 |
| BDCAF | Spearman’s rho: 0.492 | <0.001 * | |
| Ongoing immunomodulating treatment | |||
| Corticosteroids/colchicine | 245.7 (160.5–348.3) | 0.138 | |
| csDMARDs | 192.2 (120.2–252.2) | ||
| Biologic (±cs) DMARDs | 176.1 (82.9–294.4) | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bettiol, A.; Fagni, F.; Mattioli, I.; Bagni, G.; Vitiello, G.; Grassi, A.; Della Bella, C.; Benagiano, M.; Troilo, A.; Holownia, K.S.; et al. Serum Interleukin-36 α as a Candidate Biomarker to Distinguish Behçet’s Syndrome and Psoriatic Arthritis. Int. J. Mol. Sci. 2023, 24, 8817. https://doi.org/10.3390/ijms24108817
Bettiol A, Fagni F, Mattioli I, Bagni G, Vitiello G, Grassi A, Della Bella C, Benagiano M, Troilo A, Holownia KS, et al. Serum Interleukin-36 α as a Candidate Biomarker to Distinguish Behçet’s Syndrome and Psoriatic Arthritis. International Journal of Molecular Sciences. 2023; 24(10):8817. https://doi.org/10.3390/ijms24108817
Chicago/Turabian StyleBettiol, Alessandra, Filippo Fagni, Irene Mattioli, Giacomo Bagni, Gianfranco Vitiello, Alessia Grassi, Chiara Della Bella, Marisa Benagiano, Arianna Troilo, Katarzyna Stella Holownia, and et al. 2023. "Serum Interleukin-36 α as a Candidate Biomarker to Distinguish Behçet’s Syndrome and Psoriatic Arthritis" International Journal of Molecular Sciences 24, no. 10: 8817. https://doi.org/10.3390/ijms24108817








