Abstract
Flavonoids are polyphenolic phytochemical compounds found in many plants, fruits, vegetables, and leaves. They have a multitude of medicinal applications due to their anti-inflammatory, antioxidative, antiviral, and anticarcinogenic properties. Furthermore, they also have neuroprotective and cardioprotective effects. Their biological properties depend on the chemical structure of flavonoids, their mechanism of action, and their bioavailability. The beneficial effects of flavonoids have been proven for a variety of diseases. In the last few years, it is demonstrated that the effects of flavonoids are mediated by inhibiting the NF-κB (Nuclear Factor-κB) pathway. In this review, we have summarized the effects of some flavonoids on the most common diseases, such as cancer, cardiovascular, and human neurodegenerative diseases. Here, we collected all recent studies describing the protective and prevention role of flavonoids derived from plants by specifically focusing their action on the NF-κB signaling pathway.
1. Introduction
Flavonoids are a class of natural compounds characterized by a polyphenolic group and contained in different plants, fruits, grains, and flowers, as well as in beverages such as tea or wine [1]. A healthy diet is recognized all over the world as fundamental to preventing or ameliorating several diseases, such as cardiovascular and neurodegenerative diseases or cancer and its progression [2,3,4]. Hence, they are recognized as dietary flavonoids.
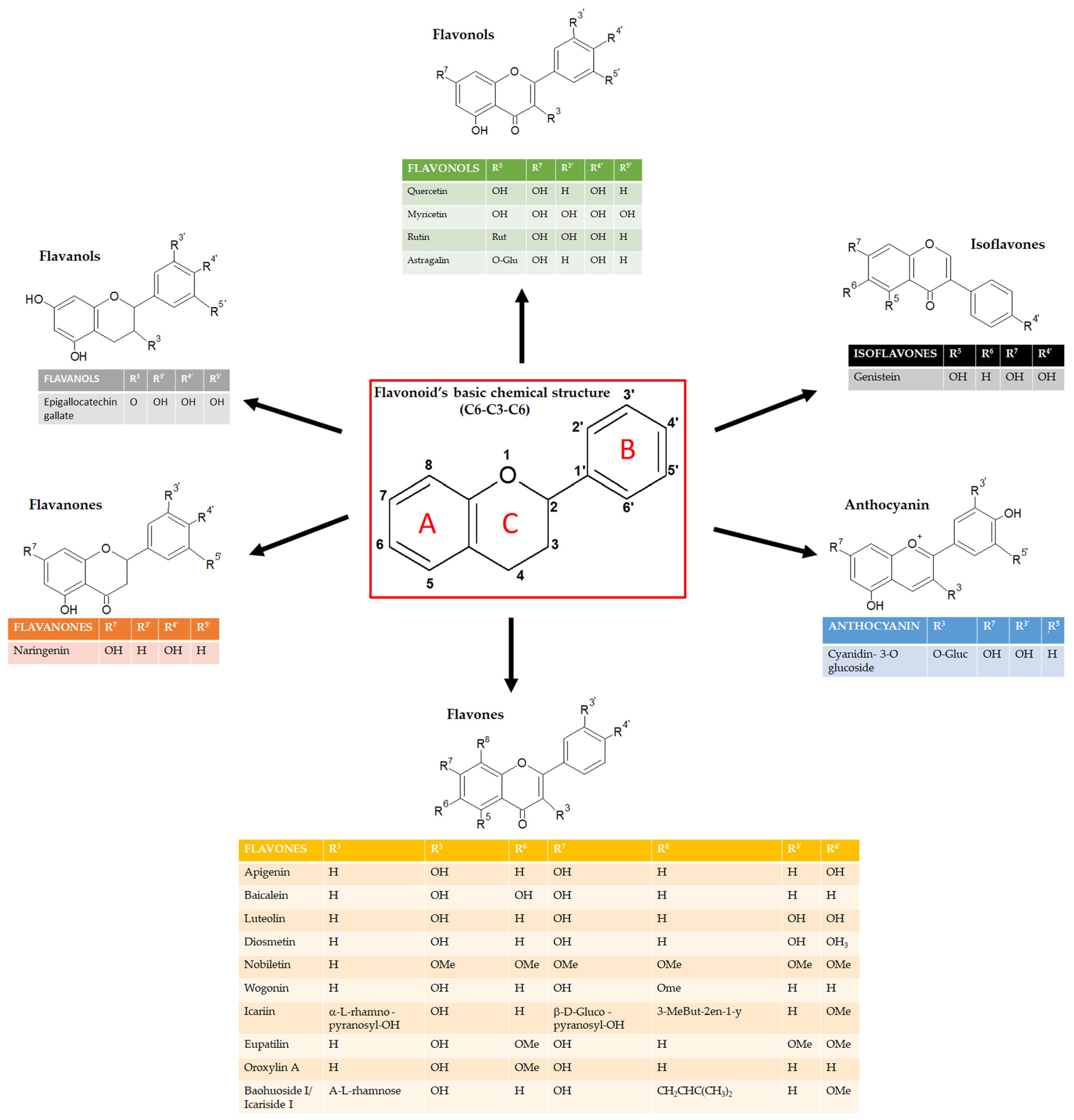
Flavonoids can be classified into several subgroups depending on the carbon of the C ring on which the B ring is attached and the degree of unsaturation and oxidation of the C ring [5]. According to these differences, we can distinguish isoflavones, neoflavonoids, flavones, flavonols, flavanones, flavanonols, flavanols or catechins, anthocyanins, and chalcones (Figure 1). Some natural compounds contain specific subgroups, e.g., onions and tea are major dietary sources of flavonols and flavones; isoflavones are mainly found in leguminous species; proanthocyanidins or condensed tannins are found in flowers, seeds, and fruits of diverse plant species, such as grapes, apples, blueberry, and some cereals; flavanols (epicatechin), flavonols (e.g., myricetin and quercetin) and anthocyanins (e.g., malvidin-3-glucoside) are enriched in red wine together with the resveratrol, well known for its antioxidant properties [6,7]. For the specific concentration of each flavonoid in the most common foods, refer to the USDA (United States Department of Agriculture) database for Flavonoid Content of Selected Foods (https://www.ars.usda.gov/ARSUserFiles/80400535/Data/Flav/Flav3.3.pdf, accessed on 17 May 2023).
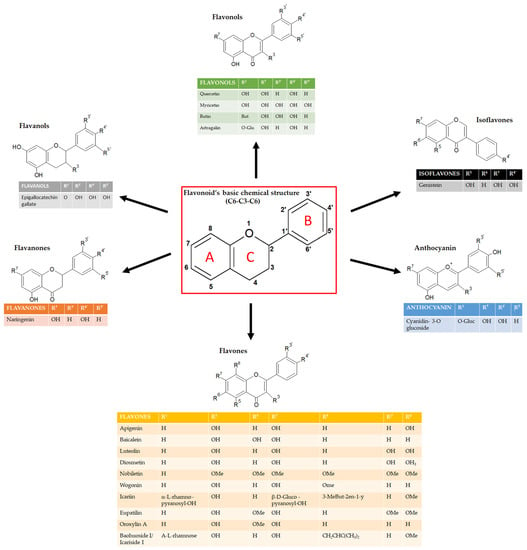
Figure 1.
Schematic representation of the chemical structure of flavonoids and its subgroups. The general structure of flavonoids consists of two benzene rings (A and B) linked via a heterocyclic pyrane ring (C).
In the last decade, the scientific community’s interest in these compounds has increased, and their beneficial or protective effects against different human diseases have been widely recognized.
Nutraceutical compounds have multiple biological activities, such as antioxidant, anti-inflammatory, or antiplatelet properties. The plant-derived flavonoids have a wide range of activities that could make them particularly effective against cardiovascular and neurodegenerative diseases [2,3]. Moreover, many scientists focused on the anticancer activities of several flavonoids and demonstrated their beneficial effects [8,9,10].
For these properties and together with absent or very low toxicity, they can be a valuable resource for the prevention of several diseases or to reduce tumor progression.
Nowadays, flavonoids are essential components of nutraceutical, pharmaceutical, and cosmetic applications because they have a huge spectrum of biological activity. However, the main mechanism through which they exert these effects is not well known. Recent manuscripts describe the role of flavonoids as anti-inflammatory agents targeting the NF-κB pathway in specific diseases such as cardiovascular diseases or cancer [11,12,13]. The Global Burden of Disease summarizes the main causes of death worldwide, the mortality across countries for a single disease, and the possible correlations with age and sex (https://www.thelancet.com/gbd#healthSummaries, accessed on 17 May 2023). Except for underdeveloped countries, these data, together with that by the global health observatory of the World Health Organization, showed that cardiovascular disease (CVD) is the top cause of death globally, especially in most industrialized countries such as North America and Europe. CVD refers to a plethora of pathologies that include hypertension, coronary heart disease (heart attack), and cerebrovascular disease (stroke), as discussed below. Cancers are nowadays defined as the second cause of death. Cancer is characterized by the fact the cells grow and spread in the body very fast through the blood and lymph systems, becoming a very aggressive and uncontrollable pathology [14]. The Global Burden of Disease recognizes dementia as the third cause of death worldwide. Dementia is a complex disease that includes several forms, such as Alzheimer’s disease, that result in the loss of cognitive capacity, including memory, language, and learning (https://www.who.int/data/gho/data/themes/mortality-and-global-health-estimates, accessed on 17 May 2023).
This review intends to summarize and discuss the results published in the last five years regarding the role of flavonoids on the diseases that affect most people around the world nowadays: cancer, cardiovascular, and neurodegenerative diseases. Moreover, we decided to focus only on the flavonoids that exert their protective or preventive role by affecting the NF-κB (Nuclear Factor-κB) signaling pathway.
2. The NF-κB Pathway
The NF-κB proteins can regulate the expression of hundreds of genes, which affect important physiological processes such as inflammation, immunity, proliferation, and cell death [15]. Since NF-κB activity is spontaneously regulated by several different stimuli, NF-κB proteins can be considered important regulators of cellular homeostasis [16,17].
NF-κB is a key mediator in the inflammatory response; its activation promotes the transcription of several target genes, many of which are pro-inflammatory [18]. Two pathways lead to the activation of the NF-κB signaling: the canonical and the non-canonical pathways [18,19]. In both pathways, the activation of the enzyme IκB Kinase (IKK) represents the common regulatory step. Notably, the canonical pathway is the most studied and plays a critical role in inflammatory responses. In this pathway, the NF-κB (p65/p50) dimer, in its inactive state, is sequestered in the cytoplasm by IκB (inhibitor of NF-κB) that covers the nuclear localization signal (NLS) of NF- κB protein.
Upon exposure to pro-inflammatory stimuli, such as cytokines, pathogens, and danger-associated molecular patterns, the p65/p50 dimer is released from IκB due to the phosphorylation cascade resulting in the proteasomal degradation of IκB. Subsequently, p65/p50 translocates into the nucleus, where it promotes the activation and expression of NF-κB target genes [18]. On the other hand, the non-canonical pathway is activated through a subset of Tumor Necrosis Factor Receptor (TNFR) superfamily members, leading to the activation of NF-κB-inducing kinase (NIK). NIK phosphorylates IKKa, which phosphorylates the C-terminal of NF-κB, allowing it to translocate into the nucleus. Once into the nucleus, NF-κB induces the expression of its target genes that play a role in cell proliferation and survival, as well as in immune cells’ development and inflammation, finally inducing the progression of several diseases, as discussed below [18].
3. Flavonoids and Cancer: Role of NF-κB Signaling Pathway
Cancer is one of the major health problems in the twenty-first century due to its high incidence rate and the second leading cause of mortality globally. Approximately 15 million people die every year due to the persistence of malignant cells, and the number of cases significantly increases day by day. For these reasons, cancer diagnosis and treatment require great effort [20,21].
Surgery, chemotherapy, radiation, and hormone and targeted therapies are examples of traditional cancer treatment approaches. However, multiple short- and long-term adverse effects may significantly affect patient prognosis depending on treatment-associated clinical factors. Several natural compounds have been studied to find new therapeutic agents, among which the bioactive elements derived from plants have gained the greatest interest in the scientific community.
The scientific community currently considers chemoprevention one of the main topics in order to counteract the continuous increase of neoplasms. Natural compounds are largely diffuse due to their anticancerous ability and their potential to overcome resistance. Many scientists focused on the anticancer properties of several flavonoids. Several in vitro studies showed the activities of flavonoids on tumor cells, such as inhibition of cell growth and alteration of tumor invasive behavior [22]. Fernández et al. studied the beneficial effects of five flavonoids, together with conventional chemotherapy, on three colorectal cancer cell lines (HCT116, HT-29, and T84) [23]. Hatono’s group demonstrated the effect of isoflavones on breast cancer cell development and their impact on breast cancer treatments [24]. Goh and collaborators demonstrated the chemopreventive role of nobiletin, extracted from citrus fruit, in colon cancer cells [25].
Xhantohumol and nobiletin, are the most abundant flavonoids extracted respectively from the hop plant and red-orange. Recently, authors showed the effects of these two compounds on colon cancer cells, demonstrating that they sensitize colorectal cancer stem cells to 5-fluorouracil (5-Fu) and oxaliplatin (FOX)-based chemotherapy and affect angiogenesis [26,27].
It has been demonstrated that inflammation favors the development of cancer and metastasis. Inflammation represents the first immune response to exogenous or endogenous damage and includes the release of inflammatory cytokines and mediators such as the interleukins IL-1, IL-6, IL-10, TNF-α (Tumor Necrosis Factor α), NF-κB, NO (Nitric Oxide), iNOS (inducible Nitric Oxide Synthase), and COX (CycloOXygenase) [28]. Constitutive induction of the NF-κB pathway has been found in lung, breast, and leukemia cells, and its up-regulation correlates with tumor progression and poor prognosis [29,30].
For this reason, in the last few years, researchers’ attention has been focused on identifying natural compounds, including flavonoids, that selectively affect or inhibit the NF-κB pathway [31,32,33].
Quercetin is a 7-hydroxyflavonol isolated from numerous plant species like horse chestnut, calendula, chamomile, and Ginkgo biloba. Moreover, quercetin is contained in several fruits, vegetables, green tea, and in red wine [34]. It has beneficial effects on human health by mediating antioxidant activities and having a role in the modulation of metabolic pathways [35,36]. Several in vitro studies have verified the antiproliferative effects of quercetin and its effect on the expression of apoptotic genes and cell cycle-related genes. Specifically, it has been well-documented that quercetin, by modulating PI3K/AKT/NF-κB (Phosphoinositide 3-kinases/Protein kinase B/NF-κB) or STAT3 (Signal transducer and activator of transcription 3), could exert antiproliferative, anti-inflammatory, and anticancer effects and can modulate the expression of many genes [37,38].
Quercetin produces a beneficial effect through the activation of Nrf2/ARE (Nuclear factor erythroid 2-related factor 2/ARE) that promotes the expression of antioxidant enzymes like superoxide dismutase. Moreover, quercetin inhibits the expression of pro-apoptotic and pro-inflammatory genes regulated by NF-κB [39,40]. Constitutive activation of the NF-κB transcription factor was detected in acute myeloid leukemia blasts and other hematopoietic cancers as well as in various solid tumors [41]. Quercetin shows inhibitory effects on the growth of malignant cells such as ovary, liver, bladder, or colorectal cancer cells [42,43]. More recently, Rubio and colleagues showed that in vitro leukemia cells’ treatment with 25 μM of quercetin for 24 h affects leukemia cell proliferation by modulating the NF-κB/Nrf2 pathway [44]. Furthermore, quercetin, by interfering with the NF-κB signaling pathway, induces apoptosis in colon cancer cells and decreases prostate cancer cells’ survival [45,46]. More recently, Sahyon and coworkers proved in vitro that combination therapy comprising quercetin (27.98 ± 1.74 μg/mL) and sulfamethoxazole exerts anticancer effects by inducing apoptosis via caspase-3 and NF-κB gene regulation and exhibits selective toxicity against cancer cells [47]. Moreover, the authors obtained the same results in vivo by using EAC (Ehrlich ascites carcinoma)-inoculated mice as animal models. EAC is a carcinoma that can spread in the abdominal region, affecting both the liver and kidney. For their experiment, the authors treated EAC-inoculated mice with a combination of sulfamethoxazole and quercetin (200 mg/kg/day) for 14 days.
It was reported that a common dietary flavonoid, apigenin, a trihydroxyflavone extracted from plants such as parsley (Petroselinum crispum), celery (Apium graveolens), and chamomile (Matricaria chamomilla), may prevent cancer therapy. A recent study conducted by Shukla and coworkers revealed that in TRAMP (TRansgenic Adenocarcinoma Mouse Prostate) mouse model, treatment with apigenin (20 and 50 μg/mouse/day for 20 days) inhibits phosphorylation and degradation of IκB, leading to suppression of NF-κB activation and finally affect prostate tumorigenesis. Moreover, they observed, after apigenin treatment, a downregulation of different NF-κB-regulated genes involved in proliferation (COX-2 and cyclin D1), angiogenesis (vascular endothelial growth factor), and apoptosis (B-cell lymphoma 2, Bcl-2, and B-cell lymphoma-extra-large, Bcl-xL) [48]. Another study conducted by the same group, using human prostate cancer cell lines, demonstrated the role of apigenin cells’ treatment (2.5 to 20 μM up to 16 h) in the suppression of NF-κB signaling through direct binding and consequent inhibition of IKKa [49]. It was also found that NF-κB expression and activity decreased following apigenin treatment in colon carcinoma cells and in non-small lung cancer cells. Tong et al. showed that, in colon carcinoma cell lines, 20 μM apigenin also inhibited epithelial-mesenchymal transition by blocking NF-κB translocation to the nucleus [50]. Moreover, their in vivo experiments showed the efficacy of apigenin (300 mg/kg for two weeks) in the inhibition of the metastasis of HCT-116 cells (colorectal cancer cell line) in a xenograft model of nude mice, mediated by NF-κB signaling pathway. Recently, Chen and colleagues proved that 20 μM apigenin enhanced TRAIL-induced apoptosis by suppressing the NF-κB nuclear translocation in non-small lung cancer cells [51]. In addition, in malignant mesothelioma, apigenin treatment (50 μM for 24 h) showed anticancer effects in vitro and in vivo by inhibiting NF-κB nuclear translocation [52].
Another interesting compound is represented by Diosmetin, a monomethoxyflavone isolated from citrus fruits that are known for its anti-inflammatory and antioxidant properties [53]. Several studies proved that diosmetin has antiproliferative, antimetastasis, and pro-apoptotic effects on breast cancer and hepatocellular carcinoma cells [53,54,55]. Moreover, the cytotoxic effect of diosmetin against Colo205, HT-29, and Caco-2 cells demonstrates that it has antitumorigenesis properties against human colorectal cancer [53,56]. Koosha et al. demonstrated that diosmetin inhibits cell proliferation and activates apoptosis signaling pathways in human colorectal cancer through the inhibition of the BMP (Bone morphogenetic proteins) and NF-κB pathways [57]. They showed that in diosmetin-treated cells (3.58 µg/mL for 48 h), TNF-a receptors expression was increased, which in turn activates the extrinsic apoptosis pathway. It is well known that TNF-a is an NF-κB inducer [58,59]. In their study, despite the upregulation of TNF-a receptors, translocation of NF-κB was inhibited via overexpression of IκB-a. Moreover, the reduction in protein expression of survivin confirms NF-κB inhibition [57]. Hence, diosmetin activates apoptosis both through the engagement of apoptotic factors and inhibition of NF-κB.
Sun et al. in their study evaluated the possible anticancer effects of Baohuoside-I or Icariside II, a glycosyloxyflavone isolated from Epimedium koreanum Nakai I, in contrasting hepatocellular carcinoma progression [60]. They proved that the treatment with 5–10 μM for 24 h of baohuoside-I significantly inhibited the proliferation of hepatocellular carcinoma cells by inducing apoptosis and downregulating the NF-κB signaling pathway. Thus, Baohuoside-I could be a potentially novel drug, opening new possibilities in the treatment of hepatocellular carcinoma [60].
El-Shitany’s group studied the role of an interesting compound, Icariin [61]. It is a prenylated flavonol glucoside, extracted from Epimedium koreanum Nakai, commonly known as horny goat weed, which offers different pharmacological properties, including reduction of inflammation and antioxidant effect. This group evaluated the anti-inflammatory mechanisms of icariin, focusing on both HO-1 (heme oxygenase-1)/Nrf2 and NF-κB pathways. They evidenced that in rats with induced acute inflammation, the pre-treatment with icariin (50 mg/kg) reduced levels of several inflammatory cytokines, COX-2 gene expression, and NF-κB activation [61].
Baicalein is a trihydroxyflavone extracted from the roots of Scutellaria baicalensis and showed antitumor activity in many cancer types. The anticancer potential of Baicalein has been investigated in a great number of studies [62,63,64,65]. It inhibits the proliferation of human cervical cancer cells by inducing apoptosis [66]. Similarly, it was also found to be active in inhibiting human prostate and ovarian cancer cells [67,68]. In the manuscript of Sheatta and colleagues, for their in vivo experiments, EAC-inoculated mice were treated with baicalin (50 mg/kg/day for three weeks) and with baicalin and/or 5-FU. Their results proved that baicalein significantly reduced inflammation and angiogenesis via suppression of NF-κB/IL-1b and VEGF (Vascular Endothelial Growth factor) as well as apoptotic pathways activation through the up-regulation of pro-apoptotic and downregulation of antiapoptotic genes [69].
Luteolin is a tetrahydroxyflavone found in plant leaves, fruits, cloves, and foods such as chamomile tea and green pepper [70]. Several studies showed that it is involved in sensitizing cancer cells to therapeutic-induced cytotoxicity by suppressing cell survival regulated by the NF-κB pathway [10]. The study conducted by Huang and coworkers showed that treatment with 10 and 30 µM of luteolin for 24 h blocked invasion and cell cycle progression of breast cancer cell lines inhibiting the NF-κB pathway and, as a consequence, reducing c-Myc proto-oncogene and hTERT (human telomerase reverse transcriptase) expression levels [71].
Nobiletin is a natural polymethoxy flavone extracted from the fruit peel of citrus. Nobiletin is well known to show anti-inflammatory properties, and in addition, it has been revealed to suppress the proliferation of human skin, prostate, colon, and breast carcinoma cell lines [72,73]. Jiang’s group found that nobiletin was actively involved in inducing autophagy, G0/G1 cell cycle arrest, and NF-κB signaling pathway inhibition, along with reducing cell migration and invasion in human pancreatic carcinoma cells [74]. Specifically, in their experiments, the authors treated the cells with different doses of nobiletin (6.12, 12.5 and 25 μM) for 24 h, and they found a dose-dependent reduction of the invasion capability of the cells and the inhibition of the NF-κB protein expression [74]. Based on these results, nobiletin could be evaluated as a potential lead molecule for the systemic treatment of cancer and merits more investigations.
Several experimental evidence suggested that the extracts of Litchi seeds have anticancer effects and can inhibit metastatic tumor growth [75,76]. It was proved that Litchi seed extracts, particularly enriched in flavonoids, show antiprostate cancer properties both in vitro and in vivo [77]. Recently, Chang and coworkers, evaluating the effects of the treatment with 100 μg/mL of total flavonoids of litchi seeds (TFLS) for 48 h on prostate cancer cell lines, found that this compound inhibited the NF-κB signaling pathway by decreasing the phosphorylation of IκBα and NF-κB nuclear expression. Moreover, their results illustrated that NF-κB influenced EMT (epithelial-mesenchymal transition). In addition, they showed that TFLS increased the level of cleaved-PARP (Poli ADP-ribose polymerase) and Bax (Bcl-2 associated X protein) and reduced the level of Bcl-2 (B-cell lymphoma 2). Hence, their results demonstrated that TFLS exerted pro-apoptotic and antimetastatic effects in prostate cancer cells by inhibiting the activation of the AKT/NF-κB signaling pathway [78].
Recently, the possible antitumoral role of Genistein was evaluated, a 7-hydroxyisoflavone in soybeans with different biological activities [79]. It was proved that genistein inhibits carcinogenesis in animal models [80]. Research results obtained by Xie and colleagues revealed that 20 µM genistein induces multiple myeloma cell proliferation inhibition and apoptosis by suppressing the NF-κB signaling pathway [81]. More recently, the Ozturk group revealed a role of genistein in thyroid cancer cell apoptosis through the downregulation of the NF-κB pathway [82].
In recent years, it was proved the anticancer role of Wogonin, a dihydroxy- and monomethoxy-flavone extracted from Scutellaria baicalensis Georgi, by inhibiting NF-κB activity. In particular, results obtained by Liu and colleagues demonstrated that treatment with increasing doses of wogonin (0, 25, 50 and 100 μM, for 24 h) can act as a natural sensitizer of human chemoresistant myelogenous leukemia cells through inhibition of Nrf2 via NF-κB signaling [83].
The major polyphenolic component of dried green tea extracts is epigallocatechin-gallate (EGCG), the ester of gallic acid and epigallocatechin, which belong to the family of flavanol [84]. Several in vitro, in vivo, and clinical studies have shown multiple EGCG anticancer actions. Among them, there are antiproliferative, pro-apoptotic, antiangiogenic, and anti-invasive functions [85]. Furthermore, EGCG has been observed to impair other processes that are involved in carcinogenesis as inflammation, oxidative stress, and hypoxia, and to target tumor microenvironment components [86]. EGCG negatively modulates the expression of various transcription factors such as Sp1 (Specific protein 1), AP-1 (activator protein 1), and NF-κB, finally preventing cancer formation [87,88]. More recently, it was revealed by Tian and colleagues that 20 μM EGCG may significantly sensitize ovarian cancer cells to cisplatin through modulation of the c-Myb-induced NF-κB -STAT3 signaling pathway [89].
Anticancer properties were also demonstrated for Eupatilin and Oroxylin natural flavonoids. Wang and colleagues showed that Eupatilin, a trimethoxyflavone mainly found in Artemisa asiatica, could be a good anticancer compound. They proved that Eupatilin plays a role in the prevention of gastric cancer. In particular, they demonstrated that eupatilin treatment (12.5, 25 and 50 µM of for 24 h) of gastric cancer cells can reduce NF-κB activity, as well as down-regulating pro-inflammatory cytokines and metalloproteinases (MMP-2, MMP-9) [90]. Sun’s group suggests a possible role of Oroxylin A, a dihydroxy- and monomethoxy-flavone extracted from the Oroxylum Indicum tree and the Scutellariae radix, as a therapeutic candidate for the treatment of breast cancer. Their data reported that 24 h treatments with 20 μM of Oroxylin A can inhibit the invasion, cell proliferation, migration, and EMT by inactivating the NF-κB signaling pathway in human breast cancer cells [91].
Recently, another natural flavonoid, the astragalin, a trihydroxyflavone extracted from different medicinal plants such as Eucommia ulmoides, Moringa oleifera, Morus alba, and Radix astragali, has been reported to induce cell death in a caspase-dependent way enhancing the sensibility of lung cancer cells to TNF-a and inhibiting NF-κB activation [92]. Specifically, Chen and coworkers showed, by treating lung cancer cells for 24 h with different astragalin doses (2.5, 5, 10, 20 μg/mL), that this compound inhibits NF-κB/p65 nuclear translocation and IκBa degradation in a dose-dependent manner.
The literature data cited in this paragraph and summarized in Table 1 let us suppose that natural compounds widely present in the daily human diet, such as flavones and flavonols, through modulation of the NF-κB signaling pathway, could play a critical role in inhibiting tumor growth and could be considered as natural chemopreventive compounds (Figure 2).

Table 1.
Summary of the effects of flavonoids on cancer mediated by the NF-κB signaling pathway.
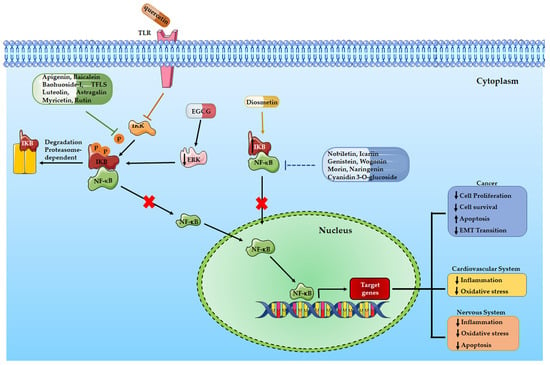
Figure 2.
A schematic illustration of the effects of flavonoids in different human diseases mediated by NF-κB signaling pathway.
4. Flavonoids and Cardiovascular Diseases: Role of NF-κB Signaling Pathway
Nowadays, cardiovascular disease (CVD) is the leading cause of death, especially in Europe, where it remains the primary cause of 42% of mortalities in men and 52% in women [93]. In the United States, it is estimated that 31% of all global deaths in 2016 were from CVDs, mainly heart attacks and stroke [94].
Inflammation is a key risk factor for CVD. A good deal of evidence demonstrates that chronic inflammation can affect atherosclerosis, myocardial infarction, and heart failure [95]. The upregulation of cytokines in the heart activates intracellular signaling pathways such as NF-κB, thus finally altering cardiac cell biology. It is well known that NF-κB activity is strictly correlated with the development and progression of inflammation and cardiovascular damage, as discussed by Fiordelisi and collaborators in their recent review [96].
Atherosclerosis (AS) is the primary cause of CVD that can determine myocardial infarction. AS can affect immune inflammatory activity through NF-κB activity, thus finally contributing to platelet migration and aggregation [97].
Platelet activation can induce aggregation and thrombus formation, ultimately contributing to ischemia and myocardial infarction. Several drugs are available to treat and prevent platelet hyperactivation, but they have many collateral negative effects. Diet and lifestyle have an impressive effect on LDL (Low-density lipoprotein)-cholesterol levels and CVD risk. For these reasons, recent studies reported the cardioprotective effects of medicinal plants and the Mediterranean diet in primary and secondary CVD prevention [98]. Hypertension is one of the most common diseases and a major risk factor for stroke and myocardial infarction; many patients affected by cardiovascular diseases are characterized by hypertension and/or cardiac dysfunction. In particular, hypertension can lead to a sustained increase in heart pressure overload, resulting in excessive proliferation and accumulation of collagen fibers in the interstitium and perivascular vessels, which can lead to myocardial fibrosis. For this reason, it is very important to decrease myocardial fibrosis, thus reducing the risk of cardiovascular disease.
Several epidemiological studies demonstrated the benefits of a diet enriched with flavonoids, especially for heart diseases.
The effect of Quercetin on CVD is well known; in fact, quercetin can reduce the expression of adhesion molecules and inflammatory markers [99]. It has been demonstrated that 25 μM of quercetin modulates TLR-NF-κB (Toll-Like Receptor-Nuclear Factor kappa B) signaling pathway on endothelial cells in vitro, thus affecting the expression of adhesion molecules involved in the leukocyte-endothelial interaction. Moreover, the authors demonstrated the anti-inflammatory activity of quercetin in rats treated for 60 days with 25 mg quercetin/kg body weight in a condition of a hypercholesterolemic diet [100]. Furthermore, by reducing MMP2 (Matrix Metalloproteinase 2) activity and oxidative stress, chronic treatment with quercetin (10 mg/kg/day) ameliorates hypertension-induced coronary hypertrophic remodeling in renovascular hypertensive rats, even if it does not reduce cardiac dysfunction [101]. An interesting study on patients with stable coronary artery disease demonstrated that quercetin therapy reduces the levels of IL-1b in the blood but also the level of IκBa mRNA, thus affecting the transcriptional activity of NF-κB signaling and finally ameliorating the indicators of chronic systemic inflammation after two-months of quercetin treatment at the daily dose of 120 mg [102].
Some studies suggested the involvement of the TGF-b (tumor growth factor b)/Smad and the NF-κB signaling pathways in the progression of myocardial fibrosis as well as the role of pro-inflammatory cytokines in the development and progression of myocardial fibrosis [95,96]. Therefore, as discussed before, an alternative strategy for the treatment of CVD, such as myocardial fibrosis, is the inhibition of inflammation.
Icariside II, the main metabolite of Icariin, is native to Korea and China, where it is well known as a nutraceutical compound in functional foods or as a phytopharmaceutical agent with anti-inflammatory and antioxidant activities that protects against cardiovascular diseases [103,104]. It is demonstrated that Icariin inhibits IL1β-induced activation of NF-κB-related signaling pathways, thus playing a significant anti-inflammatory role [105]. Fe Shu and collaborators demonstrated that Icariside II chronic treatment (4, 8, and 16 mg/kg) reduces myocardial fibrosis in spontaneously hypertensive rats through suppression of NF-κB signaling and the TGF-b1/Smad2 signaling pathways [106].
One of the major negative effects of Doxorubicin (DOXO) treatment in anticancer therapy is cardiotoxicity which can occur after 3 days (acute cardiotoxicity) or after 30 days of treatment (chronic cardiotoxicity) [107]. Doxorubicin is firstly responsible for inflammatory effects in the myocardium, characterized by the increased expression of NF-κB; moreover, cardiac inflammation contributes to cardiotoxicity. Proanthocyanidins are a class of oligomeric flavonoids derived from catechins and epicatechins, found in many plants, such as cranberry, blueberry, and grape seeds. Proanthocyanidins derived from grape seed extracts exert antioxidant effects, may reduce neutrophil infiltration and cyclooxygenase expression and, finally, reduce the adverse effects of doxorubicin treatment in anticancer therapy [108]. It was recently demonstrated that the NF-κB pathway is responsible for the prophylactic effects of proanthocyanidin extract against DOXO-induced heart injury and fibrosis [109]. Rats intraperitoneally injected with DOXO (10 mg/kg on Days 3, 9, 15, and 21 of the experiment) and proanthocyanidin extract (50 mg/kg/once daily) for 4 weeks showed a significant reduction of DOXO-induced cardiomyocyte inflammation, thus suggesting a possible role of proanthocyanidin extract against DOXO-induced cardiac toxicity [109].
Morin, a pentahydroxyflavone isolated from Chinese herbs of the Moraceae family and also present in many fruits and wines, can exert similar effects. Kuzu and collaborators demonstrated that Morin significantly decreases the expression of NF-κB, TNF-a, and IL-1β in DOXO-treated rats, inhibiting DOXO-induced oxidative stress and heart damage [110]. For the experiments reported in the manuscript, rats were treated with 50–100 mg/kg of morin (orally administered) for 10 days and with DOXO 40 mg/kg body weight by single dose intraperitoneal injection on the 8th day of the study. After analysis of different inflammation and cardiac markers,, the authors demonstrated the protective role of morin against DOXO-induced heart damage [110].
Curcumin, extracted from the rhizomes of Curcuma longa, which belongs to the ginger family, is well known as the main component of the dietary spice turmeric. Several studies demonstrated its antioxidant, anti-inflammatory and anticancer effects through inhibition of the NF-κB pathway [111,112,113]. Benzer et al. obtained similar results demonstrating the beneficial effect of curcumin against DOXO-induced cardiotoxicity in rats [114]. For their experiments, rats were treated orally for 7 days with 100–200 mg/kg body weight of curcumin, and the authors induced cardiotoxicity by injection of the single dose of 40 mg/kg of DOXO on the 5th day. Rats were sacrificed at day, and several analyses demonstrated the cardioprotective effects of curcumin after DOXO treatment [114].
Finally, we can conclude that flavonoids, as well as many other natural compounds, are able to decrease the risk of several CVD by reducing the activation of inflammatory pathways and especially of the NF-κB signaling pathway (Table 2 and Figure 2).

Table 2.
Summary of the effects of flavonoids on cardiovascular diseases mediated by the NF-κB signaling pathway.
5. Flavonoids and Neurodegenerative Diseases: Role of NF-κB Signaling Pathway
Neuroinflammation is one of the main causes of different neurodegenerative diseases such as dementia, atherosclerosis, and Alzheimer’s disease (AD). Several signals can interfere with the physiological communication between neurons and glial cells. NF-κB activation is mediated by Toll-like receptors (TLRs) that mediate the intracellular inflammatory response. Focusing on the brain, TLRs are mainly expressed in microglia and astrocytes. Once activated, the microglia can induce the expression of the NF-κB pathway and the subsequent synthesis of pro-inflammatory mediators. These pro-inflammatory cytokines finally activate kinases responsible for the hyperphosphorylation of proteins, such as tau protein, involved in the stability of cytotypes of the central nervous system. The tau proteins, in fact, become insoluble, aggregate, and form the neurofibrillary tangles that destroy the functionality of the cells. Dementia, AD, or Parkinson’s disease are specifically correlated with tau hyperphosphorylation.
Several studies described the role of NF-κB signaling in neurodegenerative diseases [115]. For example, in the brain section of Parkinson’s patients, it is demonstrated a huge increase in NF-κB activation and a strong nuclear immunoreactivity of p65 [116]. Recently, Debashis Dutta et al. demonstrated that, by specifically targeting the TLR2/NF-κB pathway, it is possible to reduce the a-synuclein spreading in vitro and in vivo, finally contributing to the treatment of neurodegenerative diseases such as Parkinson’s disease (PD) [117]. Clinical studies highlight the preventive effects of correct and healthy nutrition in the field of neurodegenerative diseases and especially AD or atherosclerosis [118,119]. Many nutraceutical bioactive principles have antiplatelet activity as well as anti-inflammatory activities [118].
It has recently been demonstrated that several flavonoids and tea catechins have a protective role toward neurons and tau protein hyperphosphorylation, mainly inhibiting the activity of MAPK (Mitogen-Activated Protein Kinase) and GSK3 pathways (Glycogen Synthase Kinase-3) [120,121,122,123]. Hung and collaborators demonstrated the antioxidant effects of quercetin. Specifically, quercetin treatment for 24 h (2.5–10 uM) and subsequent exposure to oxidized LDL protects endothelial cells from damage induced by oxidized LDL. Interestingly, quercetin exerts these effects through the activation of Sirtuin1 (SIRT1), which is involved in the regulation of several chronic conditions, such as neurodegenerative diseases [124].
Naringenin is a flavanone found in different herbs and fruits, such as grapefruit, bergamot, tart cherries, and tomatoes. It is well known that naringenin has antibacterial activity but can also reduce hepatitis C virus production in liver cells in vitro [125,126]. Importantly, Naringenin represents a promising compound in the treatment of AD because it activates the expression of Amiloid-b degradation enzymes and induces M2 polarization of microglia [127]. Sarubbo and collaborators demonstrated that naringenin, as well as quercetin, negatively regulates inflammatory pathways, thus decreasing TNF-a, IL-6, and IL-1 expression through the regulation of the nuclear translocation of p65 and the increased expression of SIRT1 in the hippocampus of rat [128]. As discussed previously, SIRT1 has a neuroprotective function and regulates NF-κB expression, while aging reduces SIRT1 hippocampal expression. Old rats were subjected to chronic treatment with naringenin or quercetin (20 mg/kg/day) for 28 days. This chronic treatment, due to the antioxidant properties of polyphenolic compounds, stabilize hippocampal SIRT1 expression, thus affecting NF-κB signaling and the inflammation pathway [128].
Epigallocatechin is the most abundant component of tea catechins, found in solid green tea extract. For this reason, it is responsible for green tea’s antioxidant, anti-inflammatory, and antiapoptotic activities [129]. Epigallocatechin 3-gallate (EGCG) overpasses the blood–brain barrier and reduces the β-amyloid (Aβ) accumulation through the suppression of the ERK/NF-κB pathway, finally exerting a neuroprotective effect [130,131].
Chang and Rong demonstrated that EGCG inhibited neurotoxic Aβ accumulation in a model of a senescent mouse (SAMP8, Senescence-accelerated mouse prone 8) subjected to EGCG chronic treatment of 5 and 15 mg/kg for 60 days. Moreover, the authors demonstrated that EGCG induced the expression of neprilysin, a protease involved in brain Aβ metabolism in vivo, thus reducing the Aβ accumulation [132].
Myricetin is an hexahydroxyflavone present in several vegetables and fruits [133]. The compound exerts various beneficial effects such as antihypertensive, anti-inflammatory, and immunomodulatory [134,135]. As discussed, neuroinflammation is caused by microglia activation. Long Sun et al. utilized a rat model of permanent middle cerebral artery occlusion (pMCAO) and administered three different concentrations of myricetin (1–5–25 mg/kg) once per day for 1 week. They demonstrated that myricetin reduces ischemic cerebral injury in the rat middle cerebral artery occlusion model by decreasing the expression of several pro-inflammatory cytokines, such as IL-1b, IL-6, TNF-a, the phosphorylation of p38 MAPK, and the level of NF-κB/p65 [136]. Jang and collaborators have recently demonstrated the inhibitory effects of Myricetin on microglia neuroinflammation through the production of pro-inflammatory molecules and cytokines [137]. The authors demonstrated their results in vitro on microglial BV-2 cells treated with myricetin 10–25 μM for 1 h and then stimulated with LPS. Moreover, myricetin (50–100 mg/kg), once a day for 7 days, inhibited LPS-induced macrophages and microglial activation in the hippocampus and cortex of mice in vivo [137]. Kaewmool et al. obtained similar results studying the effects of Cyanidin 3-O-glucoside, anthocyanins responsible for the red and blue pigments of different fruits and well known for its neuroprotective effects [138]. Authors demonstrated that cyanidin treatment (2.5–5–10 μM) for 4 h inhibits the NF-κB and p38 pathways, thus affecting the expression of pro-inflammatory cytokines in microglial BV2 cells [138,139].
Recently, Chiocchio et al. demonstrated the neuroprotective role of a flavonoid blend contained in leaves of Castanea sativa and mainly enriched in astragalin, isorhamnetin glucoside, and myricetin. Authors demonstrated that chestnut extracts significantly decrease the mRNA expression of pro-inflammatory cytokines and the activation of the NF-κB pathway on BV2 microglial cells treated for 3 h with 0.5 mg/mL of the extract [140].
It is well known that PD patients have a high level of pro-inflammatory cytokines in their brains and that bacterial LPS (lipopolysaccharides) can induce microglia activation, inflammatory cytokines expression, and PD-like symptoms in animal models [141]. Hartmann and colleagues demonstrated that activation and nuclear translocation of NF-κB are specifically involved in this process [142]. Patel and Singh recently demonstrated that apigenin treatment (25–50 mg/kg) for 2 weeks attenuates LPS-induced parkinsonism in experimental rats targeting TLR/NF-κB and Nrf2/HO-1 signaling pathways. Apigenin reduces neuroinflammation in rat striatum as demonstrated by a significant decrease in TNF-a, IL-1b and IL-6 expression. Moreover, it reduces the nuclear translocation of phospho-NF-κB in the rat brain, finally exerting a neuroprotective effect [143].
Rutin, a natural flavonoid glycoside derived from quercetin, exerts similar neuroprotective effects on different neurodiseases, including AD and PD [144]. Sun and collaborators demonstrated that rutin treatment (100 mg/kg) for 30 days prevents neuroinflammation in mouse models of AD by decreasing IL-1b and TNF-a production. Interestingly, authors evaluated the level of IKK-b, p65, and phospho-p65 in the brain protein lysates of mice to demonstrate that the rutin’s neuroprotective effects are mediated by the downregulation of NF-κB activation [145].
In 2014, Wang and collaborators showed that Genistein, at the dose of 10 mg/kg once daily for 2 weeks, ameliorated brain damage [146]. Recently it has been demonstrated that genistein reduces the inflammation of the Hippocampus in cecal ligation and puncture (CLP)-induced septic rats treated with a dose of 15 mg/kg daily for 10 days. In this study, authors focused on the hippocampus NF-κB activation showing that genistein reduces the degradation of IκBa and the nuclear translocation of NF-κB [147].
It is known that nobiletin showed antidementia properties attenuating cognitive impairment in the AD mice model [148,149]. Qi et al. recently demonstrated that nobiletin treatment (25–50–100 μM) for 4 h counteracts the microglia activation and the secretion of pro-inflammatory mediators in BV2 microglia cells. Moreover, the authors demonstrated the protective effects of nobiletin on mice treated with a dose of 100 mg/kg once daily for 7 weeks. These effects are mediated by the modulation of MAPKs, PI3K/AKT, and NF-κB signaling pathways [150]. Ghasemi-Tarie showed similar results in the AD rat model where nobiletin treatment (mg/kg/day) for 7 days prevents amyloid-induced cognitive defects by blocking p65 nuclear translocation and IκBa phosphorylation [151].
Here, we summarize the role of some natural compounds that, through affecting inflammatory pathways, and specifically the NF-κB pathway, can reduce neuroinflammation and subsequent neuronal degeneration (Table 3 and Figure 2).

Table 3.
Summary of the effects of flavonoids on neurodegenerative diseases mediated by the NF-κB signaling pathway.
6. Methods
The literature review was conducted using recommended five-step scoping review guidelines [153]: (1) identify a research question (Roles of NF-κB signaling pathways in mediating the effects of flavonoids on human diseases); (2) identify relevant studies; (3) select relevant studies; (4) chart data from these studies; and (5) collate, summarize, and report the results.
We have identified studies from the PubMed/Medline database about flavonoids and NF-κB signaling pathways in human diseases. The aim was to access original research on subjects related to human pathology. Keywords used to search the database were Flavonoids, NF-κB, cancer, neurological diseases, and cardiovascular diseases. We have excluded non-English reviews, and we included papers published within recent 5 years (except for the cardinal papers on the topic).
7. Conclusions
To date, NF-κB represents a key signaling molecule that regulates the expression of a great number of genes, thus modulating several cell signaling pathways such as cell survival and proliferation pathways or immune regulatory and inflammation pathways. For this reason, in the last decades, researchers’ attention was focused on the possibility that NF-κB could be a good target for several diseases. Therefore, one of the researcher’s challenges is the development of NF-κB inhibitors to treat different diseases and avoid the risk of side effects related to conventional therapy. A healthy diet is recognized all over the world to prevent or ameliorate several diseases, such as cardiovascular and neurodegenerative diseases, or cancer and its progression. Several natural compounds are now being explored for their capability to affect and reduce the progression of many diseases with low collateral effects or toxicity. Moreover, recent manuscripts describe the role of flavonoids as negative regulators of the NF-κB signaling pathway. For these reasons, the scientific community is now focusing on flavonoids contained in many plants, fruits, and beverages as a possible alternative strategy to overcome or prevent different pathologies. The literature data reported in this review suggest the potential capability of flavonoids to modulate pathological diseases such as cancer, cardiovascular and neurodegenerative diseases by affecting directly or indirectly the NF-κB pathway (Figure 2). Furthermore, the researcher’s results might provide substantial support for further investigation to assess the efficacy and safety of flavonoid compounds as part of a healthy diet or as co-treatment of human cancers and cardiovascular and neurodegenerative diseases.
8. Limitations and Future Perspectives
It is well known that flavonoids are lipophilic compounds, but their interaction with biological membranes is not clear and needs to be better clarified. Many flavonoids interact with receptors on the membrane, protein components of the membrane, and often with lipid rafts. The interaction with the membrane and/or the speed of the diffusion depends on the different compositions of the membrane in each tissue and, on the other hand, on the specific chemical structure of each flavonoid [154]. The presence of definite groups facilitates the adsorption of the compounds, e.g., gallate groups in catechins make EGCG better adsorbed by the membrane [155,156,157]. Flavonols, because of their planar structure, interact with the membrane with higher affinity than flavanones, even if flavanones are more hydrophobic [158].
Another limitation, that we have to take into account if we want to consider flavonoids as food supplements, is the toxicity of these compounds. Even if the beneficial properties of all these flavonoids are clear, it is important to consider that they have to pass clinical trials for efficacy and toxicity in humans. Many flavonoids have been demonstrated to be mutagenic in mammalian cells or in mice [159,160,161]. Recently Zhang and colleagues analyzed the toxicities of four flavonoids (luteolin, apigenin, quercetin, and genistein), demonstrating, through in silico, in vitro, and in vivo analysis, the developmental and mutagenic toxicity together with estrogen activity that makes them dangerous, especially in young people [161].
Further investigations to optimize the bioavailability, biodistribution, and safety of the compounds for future human studies are needed. Specifically, it is necessary: (i) to clarify how each flavonoid, according to its specific chemical structure, can cross the membrane; (ii) that the scientific community deepens knowledge and clarifies the real toxicity of each compound.
Author Contributions
Conceptualization, M.M.B. and C.C.; writing—original draft preparation, M.M.B. and C.C.; writing—review and editing, M.M.B., R.A. and C.C.; supervision, C.C. and R.A. All authors have read and agreed to the published version of the manuscript.
Funding
This study was funded by the European Union 2014–2020 PON Ricerca e Innovazione grant from the Italian Ministry of Education, University and Research, entitled “PROGEMA-Processi Green per l’Estrazione di Principi Attivi e la Depurazione di Matrici di Scarto e Non” (ARS01_00432) to Riccardo Alessandro.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable since this study is a narrative review of the literature. All included studies were ethically conducted.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing does not apply to this review article.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| NF-κB | Nuclear Factor-κB |
| USDA | United States Department Of Agriculture |
| UV | Ultraviolet |
| IKK | IκB Kinase |
| IκB | Inhibitor of NF-κB |
| NLS | Nuclear localization signal |
| TNFR | Tumor Necrosis Factor Receptor |
| NIK | NF-κB-inducing kinase |
| 5-FU | 5-Fluorouracil |
| TNF-α | Tumor Necrosis Factor α |
| IL-1 | Interleukin-1 |
| IL-6 | Interleukin-6 |
| IL-10 | Interleukin-10 |
| NO | Nitric Oxide |
| iNOS | Inducible Nitric Oxide Synthase |
| COX | CycloOXygenase |
| PI3K | Phosphoinositide 3-kinases |
| AKT | Protein kinase B |
| STAT3 | Signal transducer and activator of transcription 3 |
| Nrf2 | Nuclear factor erythroid 2-related factor 2 |
| ARE | Antioxidant response element |
| EAC | Ehrlich ascites carcinoma |
| TRAMP | TRansgenic Adenocarcinoma Mouse Prostate |
| Bcl-2 | B-cell lymphoma 2 |
| Bcl-xL | B-cell lymphoma-extra-large |
| TRAIL | Tumor-necrosis factor-related apoptosis-inducing ligand, CD253 |
| BMP | Bone morphogenetic proteins |
| HO-1 | Heme oxygenase-1 |
| VEGF | Vascular Endothelial Growth factor |
| hTERT | Human telomerase reverse transcriptase |
| TFLS | Total flavonoids of litchi seeds |
| EMT | Epithelial-mesenchymal transition |
| PARP | Poli ADP-ribose polymerase |
| Bax | Bcl-2 associated X protein |
| EGCG | Epigallocatechin-gallate |
| Sp1 | Specific protein 1 |
| AP-1 | Activator protein 1 |
| MMP-2 | Metalloproteinase-2 |
| MMP-9 | Metalloproteinase-9 |
| CVD | Cardiovascular disease |
| AS | Atherosclerosis |
| LDL | Low-density lipoprotein |
| TLR | Toll-Like Receptor |
| TGF-β | Tumor growth factor-β |
| DOXO | Doxorubicin |
| AD | Alzheimer’s disease |
| PD | Parkinson’s disease |
| MAPK | Mitogen-Activated Protein Kinase |
| GSK3 | Glycogen Synthase Kinase-3 |
| SIRT1 | Sirtuin1 |
| Aβ | β-amyloid |
| Erk | Extracellular signal-regulated kinases |
| SAMP8 | Senescence-accelerated mouse prone 8 |
| pMCAO | Permanent middle cerebral artery occlusion |
| LPS | Lipopolysaccharide |
| CLP | Cecal ligation and puncture |
References
- Sun, W.; Shahrajabian, M.H. Therapeutic Potential of Phenolic Compounds in Medicinal Plants-Natural Health Products for Human Health. Molecules 2023, 28, 1845. [Google Scholar] [CrossRef] [PubMed]
- Calis, Z.; Mogulkoc, R.; Baltaci, A.K. The Roles of Flavonols/Flavonoids in Neurodegeneration and Neuroinflammation. Mini Rev. Med. Chem. 2020, 20, 1475–1488. [Google Scholar] [CrossRef] [PubMed]
- Micek, A.; Godos, J.; Del Rio, D.; Galvano, F.; Grosso, G. Dietary Flavonoids and Cardiovascular Disease: A Comprehensive Dose-Response Meta-Analysis. Mol. Nutr. Food Res. 2021, 65, e2001019. [Google Scholar] [CrossRef] [PubMed]
- Aiello, P.; Consalvi, S.; Poce, G.; Raguzzini, A.; Toti, E.; Palmery, M.; Biava, M.; Bernardi, M.; Kamal, M.A.; Perry, G.; et al. Dietary flavonoids: Nano delivery and nanoparticles for cancer therapy. Semin. Cancer Biol. 2021, 69, 150–165. [Google Scholar] [CrossRef]
- Shen, N.; Wang, T.; Gan, Q.; Liu, S.; Wang, L.; Jin, B. Plant flavonoids: Classification, distribution, biosynthesis, and antioxidant activity. Food Chem. 2022, 383, 132531. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, I.; Perez-Gregorio, R.; Soares, S.; Mateus, N.; de Freitas, V. Wine Flavonoids in Health and Disease Prevention. Molecules 2017, 22, 292. [Google Scholar] [CrossRef]
- Yang, L.; Xian, D.; Xiong, X.; Lai, R.; Song, J.; Zhong, J. Proanthocyanidins against Oxidative Stress: From Molecular Mechanisms to Clinical Applications. Biomed. Res. Int. 2018, 2018, 8584136. [Google Scholar] [CrossRef]
- Selvakumar, P.; Badgeley, A.; Murphy, P.; Anwar, H.; Sharma, U.; Lawrence, K.; Lakshmikuttyamma, A. Flavonoids and Other Polyphenols Act as Epigenetic Modifiers in Breast Cancer. Nutrients 2020, 12, 761. [Google Scholar] [CrossRef]
- Zhai, K.; Mazurakova, A.; Koklesova, L.; Kubatka, P.; Busselberg, D. Flavonoids Synergistically Enhance the Anti-Glioblastoma Effects of Chemotherapeutic Drugs. Biomolecules 2021, 11, 1841. [Google Scholar] [CrossRef]
- Imran, M.; Rauf, A.; Abu-Izneid, T.; Nadeem, M.; Shariati, M.A.; Khan, I.A.; Imran, A.; Orhan, I.E.; Rizwan, M.; Atif, M.; et al. Luteolin, a flavonoid, as an anticancer agent: A review. Biomed. Pharmacother. 2019, 112, 108612. [Google Scholar] [CrossRef]
- Khan, H.; Ullah, H.; Castilho, P.; Gomila, A.S.; D’Onofrio, G.; Filosa, R.; Wang, F.; Nabavi, S.M.; Daglia, M.; Silva, A.S.; et al. Targeting NF-κB signaling pathway in cancer by dietary polyphenols. Crit. Rev. Food Sci. Nutr. 2020, 60, 2790–2800. [Google Scholar] [CrossRef]
- Choy, K.W.; Murugan, D.; Leong, X.F.; Abas, R.; Alias, A.; Mustafa, M.R. Flavonoids as Natural Anti-Inflammatory Agents Targeting Nuclear Factor-Kappa B (NFκB) Signaling in Cardiovascular Diseases: A Mini Review. Front. Pharmacol. 2019, 10, 1295. [Google Scholar] [CrossRef]
- Kariagina, A.; Doseff, A.I. Anti-Inflammatory Mechanisms of Dietary Flavones: Tapping into Nature to Control Chronic Inflammation in Obesity and Cancer. Int. J. Mol. Sci. 2022, 23, 15753. [Google Scholar] [CrossRef]
- The global burden of cancer attributable to risk factors, 2010–2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 2022, 400, 563–591. [CrossRef] [PubMed]
- Giuliani, C.; Bucci, I.; Napolitano, G. The Role of the Transcription Factor Nuclear Factor-kappa B in Thyroid Autoimmunity and Cancer. Front. Endocrinol. 2018, 9, 471. [Google Scholar] [CrossRef] [PubMed]
- Barnabei, L.; Laplantine, E.; Mbongo, W.; Rieux-Laucat, F.; Weil, R. NF-κB: At the Borders of Autoimmunity and Inflammation. Front. Immunol. 2021, 12, 716469. [Google Scholar] [CrossRef] [PubMed]
- Nathan, C. Points of control in inflammation. Nature 2002, 420, 846–852. [Google Scholar] [CrossRef]
- Zinatizadeh, M.R.; Schock, B.; Chalbatani, G.M.; Zarandi, P.K.; Jalali, S.A.; Miri, S.R. The Nuclear Factor Kappa B (NF-kB) signaling in cancer development and immune diseases. Genes Dis. 2021, 8, 287–297. [Google Scholar] [CrossRef]
- Sun, S.C. Non-canonical NF-κB signaling pathway. Cell Res. 2011, 21, 71–85. [Google Scholar] [CrossRef]
- Block, K.I.; Gyllenhaal, C.; Lowe, L.; Amedei, A.; Amin, A.; Amin, A.; Aquilano, K.; Arbiser, J.; Arreola, A.; Arzumanyan, A.; et al. Designing a broad-spectrum integrative approach for cancer prevention and treatment. Semin. Cancer Biol. 2015, 35, S276–S304. [Google Scholar] [CrossRef]
- Bray, F.; Ren, J.S.; Masuyer, E.; Ferlay, J. Global estimates of cancer prevalence for 27 sites in the adult population in 2008. Int. J. Cancer 2013, 132, 1133–1145. [Google Scholar] [CrossRef] [PubMed]
- Cragg, G.M.; Pezzuto, J.M. Natural Products as a Vital Source for the Discovery of Cancer Chemotherapeutic and Chemopreventive Agents. Med. Princ. Pract. 2016, 25 (Suppl. S2), 41–59. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, J.; Silvan, B.; Entrialgo-Cadierno, R.; Villar, C.J.; Capasso, R.; Uranga, J.A.; Lombo, F.; Abalo, R. Antiproliferative and palliative activity of flavonoids in colorectal cancer. Biomed. Pharmacother. 2021, 143, 112241. [Google Scholar] [CrossRef]
- Hatono, M.; Ikeda, H.; Suzuki, Y.; Kajiwara, Y.; Kawada, K.; Tsukioki, T.; Kochi, M.; Suzawa, K.; Iwamoto, T.; Yamamoto, H.; et al. Effect of isoflavones on breast cancer cell development and their impact on breast cancer treatments. Breast Cancer Res. Treat. 2021, 185, 307–316. [Google Scholar] [CrossRef]
- Goh, J.X.H.; Tan, L.T.; Goh, J.K.; Chan, K.G.; Pusparajah, P.; Lee, L.H.; Goh, B.H. Nobiletin and Derivatives: Functional Compounds from Citrus Fruit Peel for Colon Cancer Chemoprevention. Cancers 2019, 11, 867. [Google Scholar] [CrossRef] [PubMed]
- Turdo, A.; Glaviano, A.; Pepe, G.; Calapa, F.; Raimondo, S.; Fiori, M.E.; Carbone, D.; Basilicata, M.G.; Di Sarno, V.; Ostacolo, C.; et al. Nobiletin and Xanthohumol Sensitize Colorectal Cancer Stem Cells to Standard Chemotherapy. Cancers 2021, 13, 3927. [Google Scholar] [CrossRef]
- Corrado, C.; Barreca, M.M.; Raimondo, S.; Diana, P.; Pepe, G.; Basilicata, M.G.; Conigliaro, A.; Alessandro, R. Nobiletin and xanthohumol counteract the TNFalpha-mediated activation of endothelial cells through the inhibition of the NF-κB signaling pathway. Cell. Biol. Int. 2023, 47, 634–647. [Google Scholar] [CrossRef]
- Pereira, M.F.; Martino, T.; Dalmau, S.R.; Paes, M.C.; Barja-Fidalgo, C.; Albano, R.M.; Coelho, M.G.; Sabino, K.C. Terpenic fraction of Pterodon pubescens inhibits nuclear factor kappa B and extracellular signal-regulated protein kinase 1/2 activation and deregulates gene expression in leukemia cells. BMC Complement. Altern. Med. 2012, 12, 231. [Google Scholar] [CrossRef]
- Mirzaei, S.; Saghari, S.; Bassiri, F.; Raesi, R.; Zarrabi, A.; Hushmandi, K.; Sethi, G.; Tergaonkar, V. NF-κB as a regulator of cancer metastasis and therapy response: A focus on epithelial-mesenchymal transition. J. Cell. Physiol. 2022, 237, 2770–2795. [Google Scholar] [CrossRef]
- Chauhan, A.; Islam, A.U.; Prakash, H.; Singh, S. Phytochemicals targeting NF-κB signaling: Potential anti-cancer interventions. J. Pharm. Anal. 2022, 12, 394–405. [Google Scholar] [CrossRef]
- Avci, N.G.; Ebrahimzadeh-Pustchi, S.; Akay, Y.M.; Esquenazi, Y.; Tandon, N.; Zhu, J.J.; Akay, M. NF-κB inhibitor with Temozolomide results in significant apoptosis in glioblastoma via the NF-κB(p65) and actin cytoskeleton regulatory pathways. Sci. Rep. 2020, 10, 13352. [Google Scholar] [CrossRef] [PubMed]
- Rasmi, R.R.; Sakthivel, K.M.; Guruvayoorappan, C. NF-κB inhibitors in treatment and prevention of lung cancer. Biomed. Pharmacother. 2020, 130, 110569. [Google Scholar] [CrossRef] [PubMed]
- Mates, L.; Rusu, M.E.; Popa, D.S. Phytochemicals and Biological Activities of Walnut Septum: A Systematic Review. Antioxidants 2023, 12, 604. [Google Scholar] [CrossRef]
- Vargas, A.J.; Burd, R. Hormesis and synergy: Pathways and mechanisms of quercetin in cancer prevention and management. Nutr. Rev. 2010, 68, 418–428. [Google Scholar] [CrossRef]
- Ghafouri-Fard, S.; Shoorei, H.; Khanbabapour Sasi, A.; Taheri, M.; Ayatollahi, S.A. The impact of the phytotherapeutic agent quercetin on expression of genes and activity of signaling pathways. Biomed. Pharmacother. 2021, 141, 111847. [Google Scholar] [CrossRef]
- Kelly, G.S. Quercetin. Monograph. Altern. Med. Rev. 2011, 16, 172–194. [Google Scholar] [PubMed]
- Lai, W.W.; Hsu, S.C.; Chueh, F.S.; Chen, Y.Y.; Yang, J.S.; Lin, J.P.; Lien, J.C.; Tsai, C.H.; Chung, J.G. Quercetin inhibits migration and invasion of SAS human oral cancer cells through inhibition of NF-κB and matrix metalloproteinase-2/-9 signaling pathways. Anticancer Res. 2013, 33, 1941–1950. [Google Scholar]
- Anand David, A.V.; Arulmoli, R.; Parasuraman, S. Overviews of Biological Importance of Quercetin: A Bioactive Flavonoid. Pharmacogn. Rev. 2016, 10, 84–89. [Google Scholar] [CrossRef]
- Ramyaa, P.; Krishnaswamy, R.; Padma, V.V. Quercetin modulates OTA-induced oxidative stress and redox signalling in HepG2 cells—Up regulation of Nrf2 expression and down regulation of NF-κB and COX-2. Biochim. Biophys. Acta 2014, 1840, 681–692. [Google Scholar] [CrossRef] [PubMed]
- Acharya, A.; Das, I.; Chandhok, D.; Saha, T. Redox regulation in cancer: A double-edged sword with therapeutic potential. Oxid. Med. Cell. Longev. 2010, 3, 23–34. [Google Scholar] [CrossRef]
- Rushworth, S.A.; Zaitseva, L.; Murray, M.Y.; Shah, N.M.; Bowles, K.M.; MacEwan, D.J. The high Nrf2 expression in human acute myeloid leukemia is driven by NF-κB and underlies its chemo-resistance. Blood 2012, 120, 5188–5198. [Google Scholar] [CrossRef] [PubMed]
- Tao, S.F.; He, H.F.; Chen, Q. Quercetin inhibits proliferation and invasion acts by up-regulating miR-146a in human breast cancer cells. Mol. Cell. Biochem. 2015, 402, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Z.; Long, C.; Junming, T.; Qihuan, L.; Youshun, Z.; Chan, Z. Quercetin-induced apoptosis of HL-60 cells by reducing PI3K/Akt. Mol. Biol. Rep. 2012, 39, 7785–7793. [Google Scholar] [CrossRef]
- Rubio, V.; Garcia-Perez, A.I.; Herraez, A.; Diez, J.C. Different roles of Nrf2 and NFKB in the antioxidant imbalance produced by esculetin or quercetin on NB4 leukemia cells. Chem. Biol. Interact. 2018, 294, 158–166. [Google Scholar] [CrossRef]
- Zhang, X.A.; Zhang, S.; Yin, Q.; Zhang, J. Quercetin induces human colon cancer cells apoptosis by inhibiting the nuclear factor-kappa B Pathway. Pharmacogn. Mag. 2015, 11, 404–409. [Google Scholar] [CrossRef] [PubMed]
- Ward, A.B.; Mir, H.; Kapur, N.; Gales, D.N.; Carriere, P.P.; Singh, S. Quercetin inhibits prostate cancer by attenuating cell survival and inhibiting anti-apoptotic pathways. World J. Surg. Oncol. 2018, 16, 108. [Google Scholar] [CrossRef] [PubMed]
- Sahyon, H.A.E.; Ramadan, E.N.M.; Althobaiti, F.; Mashaly, M.M.A. Anti-proliferative effects of the combination of Sulfamethoxazole and Quercetin via caspase3 and NFkB gene regulation: An in vitro and in vivo study. Naunyn Schmiedebergs Arch. Pharmacol. 2022, 395, 227–246. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.; Shankar, E.; Fu, P.; MacLennan, G.T.; Gupta, S. Suppression of NF-κB and NF-κB-Regulated Gene Expression by Apigenin through IκBα and IKK Pathway in TRAMP Mice. PLoS ONE 2015, 10, e0138710. [Google Scholar] [CrossRef]
- Shukla, S.; Kanwal, R.; Shankar, E.; Datt, M.; Chance, M.R.; Fu, P.; MacLennan, G.T.; Gupta, S. Apigenin blocks IKKalpha activation and suppresses prostate cancer progression. Oncotarget 2015, 6, 31216–31232. [Google Scholar] [CrossRef]
- Tong, J.; Shen, Y.; Zhang, Z.; Hu, Y.; Zhang, X.; Han, L. Apigenin inhibits epithelial-mesenchymal transition of human colon cancer cells through NF-κB/Snail signaling pathway. Biosci. Rep. 2019, 39, BSR20190452. [Google Scholar] [CrossRef]
- Chen, M.; Wang, X.; Zha, D.; Cai, F.; Zhang, W.; He, Y.; Huang, Q.; Zhuang, H.; Hua, Z.C. Apigenin potentiates TRAIL therapy of non-small cell lung cancer via upregulating DR4/DR5 expression in a p53-dependent manner. Sci. Rep. 2016, 6, 35468. [Google Scholar] [CrossRef] [PubMed]
- Masuelli, L.; Benvenuto, M.; Mattera, R.; Di Stefano, E.; Zago, E.; Taffera, G.; Tresoldi, I.; Giganti, M.G.; Frajese, G.V.; Berardi, G.; et al. In Vitro and In Vivo Anti-tumoral Effects of the Flavonoid Apigenin in Malignant Mesothelioma. Front. Pharmacol. 2017, 8, 373. [Google Scholar] [CrossRef] [PubMed]
- Androutsopoulos, V.P.; Mahale, S.; Arroo, R.R.; Potter, G. Anticancer effects of the flavonoid diosmetin on cell cycle progression and proliferation of MDA-MB 468 breast cancer cells due to CYP1 activation. Oncol. Rep. 2009, 21, 1525–1528. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Shi, Y.; Peng, W.; Zhang, Q.; Liu, J.; Chen, N.; Zhu, R. Diosmetin induces apoptosis by upregulating p53 via the TGF-beta signal pathway in HepG2 hepatoma cells. Mol. Med. Rep. 2016, 14, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wen, X.; Liu, B.; Zhang, Q.; Zhang, J.; Miao, H.; Zhu, R. Diosmetin inhibits the metastasis of hepatocellular carcinoma cells by downregulating the expression levels of MMP-2 and MMP-9. Mol. Med. Rep. 2016, 13, 2401–2408. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.Y.; Yuan, D.; Yang, J.Y.; Wang, L.H.; Wu, C.F. Cytotoxic activity of flavonoids from the flowers of Chrysanthemum morifolium on human colon cancer Colon205 cells. J. Asian Nat. Prod. Res. 2009, 11, 771–778. [Google Scholar] [CrossRef]
- Koosha, S.; Mohamed, Z.; Sinniah, A.; Alshawsh, M.A. Investigation into the Molecular Mechanisms underlying the Anti-proliferative and Anti-tumorigenesis activities of Diosmetin against HCT-116 Human Colorectal Cancer. Sci. Rep. 2019, 9, 5148. [Google Scholar] [CrossRef]
- Schnare, M.; Barton, G.M.; Holt, A.C.; Takeda, K.; Akira, S.; Medzhitov, R. Toll-like receptors control activation of adaptive immune responses. Nat. Immunol. 2001, 2, 947–950. [Google Scholar] [CrossRef]
- Baldwin, A.S., Jr. Series introduction: The transcription factor NF-κB and human disease. J. Clin. Investig. 2001, 107, 3–6. [Google Scholar] [CrossRef]
- Sun, Y.; Pang, B.; Wang, Y.; Xiao, J.; Jiang, D. Baohuoside I Inhibits the Proliferation of Hepatocellular Carcinoma Cells via Apoptosis Signaling and NF-kB Pathway. Chem. Biodivers. 2021, 18, e2100063. [Google Scholar] [CrossRef]
- El-Shitany, N.A.; Eid, B.G. Icariin modulates carrageenan-induced acute inflammation through HO-1/Nrf2 and NF-kB signaling pathways. Biomed. Pharmacother. 2019, 120, 109567. [Google Scholar] [CrossRef] [PubMed]
- Ikemoto, S.; Sugimura, K.; Yoshida, N.; Yasumoto, R.; Wada, S.; Yamamoto, K.; Kishimoto, T. Antitumor effects of Scutellariae radix and its components baicalein, baicalin, and wogonin on bladder cancer cell lines. Urology 2000, 55, 951–955. [Google Scholar] [CrossRef]
- Li-Weber, M. New therapeutic aspects of flavones: The anticancer properties of Scutellaria and its main active constituents Wogonin, Baicalein and Baicalin. Cancer Treat. Rev. 2009, 35, 57–68. [Google Scholar] [CrossRef] [PubMed]
- Parajuli, P.; Joshee, N.; Rimando, A.M.; Mittal, S.; Yadav, A.K. In vitro antitumor mechanisms of various Scutellaria extracts and constituent flavonoids. Planta Med. 2009, 75, 41–48. [Google Scholar] [CrossRef]
- Chiu, Y.W.; Lin, T.H.; Huang, W.S.; Teng, C.Y.; Liou, Y.S.; Kuo, W.H.; Lin, W.L.; Huang, H.I.; Tung, J.N.; Huang, C.Y.; et al. Baicalein inhibits the migration and invasive properties of human hepatoma cells. Toxicol. Appl. Pharmacol. 2011, 255, 316–326. [Google Scholar] [CrossRef]
- Peng, Y.; Guo, C.; Yang, Y.; Li, F.; Zhang, Y.; Jiang, B.; Li, Q. Baicalein induces apoptosis of human cervical cancer HeLa cells in vitro. Mol. Med. Rep. 2015, 11, 2129–2134. [Google Scholar] [CrossRef] [PubMed]
- Miocinovic, R.; McCabe, N.P.; Keck, R.W.; Jankun, J.; Hampton, J.A.; Selman, S.H. In vivo and in vitro effect of baicalein on human prostate cancer cells. Int. J. Oncol. 2005, 26, 241–246. [Google Scholar] [CrossRef]
- Chen, J.; Li, Z.; Chen, A.Y.; Ye, X.; Luo, H.; Rankin, G.O.; Chen, Y.C. Inhibitory effect of baicalin and baicalein on ovarian cancer cells. Int. J. Mol. Sci. 2013, 14, 6012–6025. [Google Scholar] [CrossRef]
- Shehatta, N.H.; Okda, T.M.; Omran, G.A.; Abd-Alhaseeb, M.M. Baicalin; a promising chemopreventive agent, enhances the antitumor effect of 5-FU against breast cancer and inhibits tumor growth and angiogenesis in Ehrlich solid tumor. Biomed. Pharmacother. 2022, 146, 112599. [Google Scholar] [CrossRef]
- Lin, Y.; Shi, R.; Wang, X.; Shen, H.M. Luteolin, a flavonoid with potential for cancer prevention and therapy. Curr. Cancer Drug. Targets 2008, 8, 634–646. [Google Scholar] [CrossRef]
- Huang, L.; Jin, K.; Lan, H. Luteolin inhibits cell cycle progression and induces apoptosis of breast cancer cells through downregulation of human telomerase reverse transcriptase. Oncol. Lett. 2019, 17, 3842–3850. [Google Scholar] [CrossRef] [PubMed]
- Murakami, A.; Nakamura, Y.; Torikai, K.; Tanaka, T.; Koshiba, T.; Koshimizu, K.; Kuwahara, S.; Takahashi, Y.; Ogawa, K.; Yano, M.; et al. Inhibitory effect of citrus nobiletin on phorbol ester-induced skin inflammation, oxidative stress, and tumor promotion in mice. Cancer Res. 2000, 60, 5059–5066. [Google Scholar] [PubMed]
- Kandaswami, C.; Perkins, E.; Soloniuk, D.S.; Drzewiecki, G.; Middleton, E., Jr. Antiproliferative effects of citrus flavonoids on a human squamous cell carcinoma in vitro. Cancer Lett. 1991, 56, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Chen, H.; Jin, C.; Mo, J.; Wang, H. Nobiletin flavone inhibits the growth and metastasis of human pancreatic cancer cells via induction of autophagy, G0/G1 cell cycle arrest and inhibition of NF-kB signalling pathway. J. BUON 2020, 25, 1070–1075. [Google Scholar]
- Hsu, C.P.; Lin, C.C.; Huang, C.C.; Lin, Y.H.; Chou, J.C.; Tsia, Y.T.; Su, J.R.; Chung, Y.C. Induction of apoptosis and cell cycle arrest in human colorectal carcinoma by Litchi seed extract. J. Biomed. Biotechnol. 2012, 2012, 341479. [Google Scholar] [CrossRef]
- Emanuele, S.; Notaro, A.; Palumbo Piccionello, A.; Maggio, A.; Lauricella, M.; D’Anneo, A.; Cernigliaro, C.; Calvaruso, G.; Giuliano, M. Sicilian Litchi Fruit Extracts Induce Autophagy versus Apoptosis Switch in Human Colon Cancer Cells. Nutrients 2018, 10, 1490. [Google Scholar] [CrossRef]
- Guo, H.; Luo, H.; Yuan, H.; Xia, Y.; Shu, P.; Huang, X.; Lu, Y.; Liu, X.; Keller, E.T.; Sun, D.; et al. Author Correction: Litchi seed extracts diminish prostate cancer progression via induction of apoptosis and attenuation of EMT through Akt/GSK-3beta signaling. Sci. Rep. 2021, 11, 15201. [Google Scholar] [CrossRef]
- Chang, M.; Zhu, D.; Chen, Y.; Zhang, W.; Liu, X.; Li, X.L.; Cheng, Z.; Su, Z.; Zhang, J.; Lu, Y.; et al. Total Flavonoids of Litchi Seed Attenuate Prostate Cancer Progression Via Inhibiting AKT/mTOR and NF-kB Signaling Pathways. Front. Pharmacol. 2021, 12, 758219. [Google Scholar] [CrossRef]
- Zamani-Garmsiri, F.; Emamgholipour, S.; Rahmani Fard, S.; Ghasempour, G.; Jahangard Ahvazi, R.; Meshkani, R. Polyphenols: Potential anti-inflammatory agents for treatment of metabolic disorders. Phytother. Res. 2022, 36, 415–432. [Google Scholar] [CrossRef]
- Li, H.Q.; Luo, Y.; Qiao, C.H. The mechanisms of anticancer agents by genistein and synthetic derivatives of isoflavone. Mini Rev. Med. Chem. 2012, 12, 350–362. [Google Scholar] [CrossRef]
- Xie, J.; Wang, J.; Zhu, B. Genistein inhibits the proliferation of human multiple myeloma cells through suppression of nuclear factor-kappaB and upregulation of microRNA-29b. Mol. Med. Rep. 2016, 13, 1627–1632. [Google Scholar] [CrossRef] [PubMed]
- Ozturk, S.A.; Alp, E.; Yar Saglam, A.S.; Konac, E.; Menevse, E.S. The effects of thymoquinone and genistein treatment on telomerase activity, apoptosis, angiogenesis, and survival in thyroid cancer cell lines. J. Cancer Res. Ther. 2018, 14, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Tian, S.; Liu, M.; Jian, L.; Zhao, L. Wogonin inhibits the proliferation and invasion, and induces the apoptosis of HepG2 and Bel7402 HCC cells through NF-κB/Bcl-2, EGFR and EGFR downstream ERK/AKT signaling. Int. J. Mol. Med. 2016, 38, 1250–1256. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Afaq, F.; Saleem, M.; Ahmad, N.; Mukhtar, H. Targeting multiple signaling pathways by green tea polyphenol (-)-epigallocatechin-3-gallate. Cancer Res. 2006, 66, 2500–2505. [Google Scholar] [CrossRef]
- Gan, R.Y.; Li, H.B.; Sui, Z.Q.; Corke, H. Absorption, metabolism, anti-cancer effect and molecular targets of epigallocatechin gallate (EGCG): An updated review. Crit. Rev. Food Sci. Nutr. 2018, 58, 924–941. [Google Scholar] [CrossRef]
- Zubair, H.; Azim, S.; Ahmad, A.; Khan, M.A.; Patel, G.K.; Singh, S.; Singh, A.P. Cancer Chemoprevention by Phytochemicals: Nature’s Healing Touch. Molecules 2017, 22, 395. [Google Scholar] [CrossRef]
- Shimizu, M.; Shirakami, Y.; Moriwaki, H. Targeting receptor tyrosine kinases for chemoprevention by green tea catechin, EGCG. Int. J. Mol. Sci. 2008, 9, 1034–1049. [Google Scholar] [CrossRef]
- Shimizu, M.; Weinstein, I.B. Modulation of signal transduction by tea catechins and related phytochemicals. Mutat. Res. 2005, 591, 147–160. [Google Scholar] [CrossRef]
- Tian, M.; Tian, D.; Qiao, X.; Li, J.; Zhang, L. Modulation of Myb-induced NF-kB -STAT3 signaling and resulting cisplatin resistance in ovarian cancer by dietary factors. J. Cell. Physiol. 2019, 234, 21126–21134. [Google Scholar] [CrossRef]
- Wang, Y.; Hou, H.; Li, M.; Yang, Y.; Sun, L. Anticancer effect of eupatilin on glioma cells through inhibition of the Notch-1 signaling pathway. Mol. Med. Rep. 2016, 13, 1141–1146. [Google Scholar] [CrossRef]
- Sun, X.; Chang, X.; Wang, Y.; Xu, B.; Cao, X. Oroxylin A Suppresses the Cell Proliferation, Migration, and EMT via NF-κB Signaling Pathway in Human Breast Cancer Cells. Biomed. Res. Int. 2019, 2019, 9241769. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Cai, F.; Zha, D.; Wang, X.; Zhang, W.; He, Y.; Huang, Q.; Zhuang, H.; Hua, Z.C. Astragalin-induced cell death is caspase-dependent and enhances the susceptibility of lung cancer cells to tumor necrosis factor by inhibiting the NF-small ka, CyrillicB pathway. Oncotarget 2017, 8, 26941–26958. [Google Scholar] [CrossRef] [PubMed]
- Townsend, N.; Nichols, M.; Scarborough, P.; Rayner, M. Cardiovascular disease in Europe--epidemiological update 2015. Eur. Heart J. 2015, 36, 2696–2705. [Google Scholar] [CrossRef]
- Micha, R.; Penalvo, J.L.; Cudhea, F.; Imamura, F.; Rehm, C.D.; Mozaffarian, D. Association Between Dietary Factors and Mortality from Heart Disease, Stroke, and Type 2 Diabetes in the United States. JAMA 2017, 317, 912–924. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhao, L.; Zhou, X.; Meng, X. Role of inflammation, immunity, and oxidative stress in hypertension: New insights and potential therapeutic targets. Front. Immunol. 2022, 13, 1098725. [Google Scholar] [CrossRef] [PubMed]
- Fiordelisi, A.; Iaccarino, G.; Morisco, C.; Coscioni, E.; Sorriento, D. NFκB is a Key Player in the Crosstalk between Inflammation and Cardiovascular Diseases. Int. J. Mol. Sci. 2019, 20, 1599. [Google Scholar] [CrossRef]
- Wolf, D.; Ley, K. Immunity and Inflammation in Atherosclerosis. Circ. Res. 2019, 124, 315–327. [Google Scholar] [CrossRef]
- Irfan, M.; Kwon, T.H.; Yun, B.S.; Park, N.H.; Rhee, M.H. Eisenia bicyclis (brown alga) modulates platelet function and inhibits thrombus formation via impaired P(2)Y(12) receptor signaling pathway. Phytomedicine 2018, 40, 79–87. [Google Scholar] [CrossRef]
- Patel, R.V.; Mistry, B.M.; Shinde, S.K.; Syed, R.; Singh, V.; Shin, H.S. Therapeutic potential of quercetin as a cardiovascular agent. Eur. J. Med. Chem. 2018, 155, 889–904. [Google Scholar] [CrossRef]
- Bhaskar, S.; Sudhakaran, P.R.; Helen, A. Quercetin attenuates atherosclerotic inflammation and adhesion molecule expression by modulating TLR-NF-κB signaling pathway. Cell. Immunol. 2016, 310, 131–140. [Google Scholar] [CrossRef]
- Da Rocha, E.V.; Falchetti, F.; Pernomian, L.; de Mello, M.M.B.; Parente, J.M.; Nogueira, R.C.; Gomes, B.Q.; Bertozi, G.; Sanches-Lopes, J.M.; Tanus-Santos, J.E.; et al. Quercetin decreases cardiac hypertrophic mediators and maladaptive coronary arterial remodeling in renovascular hypertensive rats without improving cardiac function. Naunyn Schmiedebergs Arch. Pharmacol. 2023, 396, 939–949. [Google Scholar] [CrossRef] [PubMed]
- Chekalina, N.; Burmak, Y.; Petrov, Y.; Borisova, Z.; Manusha, Y.; Kazakov, Y.; Kaidashev, I. Quercetin reduces the transcriptional activity of NF-kB in stable coronary artery disease. Indian. Heart J. 2018, 70, 593–597. [Google Scholar] [CrossRef]
- Fu, S.; Li, Y.; Wu, Y.; Yue, Y.; Yang, D. Icariside II improves myocardial fibrosis in spontaneously hypertensive rats by inhibiting collagen synthesis. J. Pharm. Pharmacol. 2020, 72, 227–235. [Google Scholar] [CrossRef]
- Irfan, M.; Kwon, T.H.; Lee, D.H.; Hong, S.B.; Oh, J.W.; Kim, S.D.; Rhee, M.H. Antiplatelet and Antithrombotic Effects of Epimedium koreanum Nakai. Evid. Based Complement. Alternat. Med. 2021, 2021, 7071987. [Google Scholar] [CrossRef]
- Hua, W.; Zhang, Y.; Wu, X.; Kang, L.; Tu, J.; Zhao, K.; Li, S.; Wang, K.; Song, Y.; Luo, R.; et al. Icariin Attenuates Interleukin-1beta-Induced Inflammatory Response in Human Nucleus Pulposus Cells. Curr. Pharm. Des. 2018, 23, 6071–6078. [Google Scholar] [CrossRef]
- Fu, S.; Li, Y.L.; Wu, Y.T.; Yue, Y.; Qian, Z.Q.; Yang, D.L. Icariside II attenuates myocardial fibrosis by inhibiting nuclear factor-κB and the TGF-beta1/Smad2 signalling pathway in spontaneously hypertensive rats. Biomed. Pharmacother. 2018, 100, 64–71. [Google Scholar] [CrossRef]
- Hao, G.; Yu, Y.; Gu, B.; Xing, Y.; Xue, M. Protective effects of berberine against doxorubicin-induced cardiotoxicity in rats by inhibiting metabolism of doxorubicin. Xenobiotica 2015, 45, 1024–1029. [Google Scholar] [CrossRef] [PubMed]
- Bagchi, D.; Swaroop, A.; Preuss, H.G.; Bagchi, M. Free radical scavenging, antioxidant and cancer chemoprevention by grape seed proanthocyanidin: An overview. Mutat. Res. 2014, 768, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Sadek, K.M.; Mahmoud, S.F.E.; Zeweil, M.F.; Abouzed, T.K. Proanthocyanidin alleviates doxorubicin-induced cardiac injury by inhibiting NF-kB pathway and modulating oxidative stress, cell cycle, and fibrogenesis. J. Biochem. Mol. Toxicol. 2021, 35, e22716. [Google Scholar] [CrossRef]
- Kuzu, M.; Kandemir, F.M.; Yildirim, S.; Kucukler, S.; Caglayan, C.; Turk, E. Morin attenuates doxorubicin-induced heart and brain damage by reducing oxidative stress, inflammation and apoptosis. Biomed. Pharmacother. 2018, 106, 443–453. [Google Scholar] [CrossRef]
- Li, W.; Jiang, L.; Lu, X.; Liu, X.; Ling, M. Curcumin protects radiation-induced liver damage in rats through the NF-κB signaling pathway. BMC Complement. Med. Ther. 2021, 21, 10. [Google Scholar] [CrossRef]
- Chung, S.S.; Dutta, P.; Chard, N.; Wu, Y.; Chen, Q.H.; Chen, G.; Vadgama, J. A novel curcumin analog inhibits canonical and non-canonical functions of telomerase through STAT3 and NF-κB inactivation in colorectal cancer cells. Oncotarget 2019, 10, 4516–4531. [Google Scholar] [CrossRef]
- Hassan, F.U.; Rehman, M.S.; Khan, M.S.; Ali, M.A.; Javed, A.; Nawaz, A.; Yang, C. Curcumin as an Alternative Epigenetic Modulator: Mechanism of Action and Potential Effects. Front. Genet. 2019, 10, 514. [Google Scholar] [CrossRef]
- Benzer, F.; Kandemir, F.M.; Ozkaraca, M.; Kucukler, S.; Caglayan, C. Curcumin ameliorates doxorubicin-induced cardiotoxicity by abrogation of inflammation, apoptosis, oxidative DNA damage, and protein oxidation in rats. J. Biochem. Mol. Toxicol. 2018, 32, e22030. [Google Scholar] [CrossRef]
- Singh, S.; Singh, T.G. Role of Nuclear Factor Kappa B (NF-κB) Signalling in Neurodegenerative Diseases: An Mechanistic Approach. Curr. Neuropharmacol. 2020, 18, 918–935. [Google Scholar] [CrossRef] [PubMed]
- Camandola, S.; Mattson, M.P. NF-κ B as a therapeutic target in neurodegenerative diseases. Expert. Opin. Ther. Targets 2007, 11, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Dutta, D.; Jana, M.; Majumder, M.; Mondal, S.; Roy, A.; Pahan, K. Selective targeting of the TLR2/MyD88/NF-κB pathway reduces alpha-synuclein spreading in vitro and in vivo. Nat. Commun. 2021, 12, 5382. [Google Scholar] [CrossRef]
- Calfio, C.; Gonzalez, A.; Singh, S.K.; Rojo, L.E.; Maccioni, R.B. The Emerging Role of Nutraceuticals and Phytochemicals in the Prevention and Treatment of Alzheimer’s Disease. J. Alzheimers Dis. 2020, 77, 33–51. [Google Scholar] [CrossRef]
- Maccioni, R.B.; Calfio, C.; Gonzalez, A.; Luttges, V. Novel Nutraceutical Compounds in Alzheimer Prevention. Biomolecules 2022, 12, 249. [Google Scholar] [CrossRef]
- Zaplatic, E.; Bule, M.; Shah, S.Z.A.; Uddin, M.S.; Niaz, K. Molecular mechanisms underlying protective role of quercetin in attenuating Alzheimer’s disease. Life Sci. 2019, 224, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Atrahimovich, D.; Avni, D.; Khatib, S. Flavonoids-Macromolecules Interactions in Human Diseases with Focus on Alzheimer, Atherosclerosis and Cancer. Antioxidants 2021, 10, 423. [Google Scholar] [CrossRef] [PubMed]
- Maher, P. The Potential of Flavonoids for the Treatment of Neurodegenerative Diseases. Int. J. Mol. Sci. 2019, 20, 3056. [Google Scholar] [CrossRef]
- Zu, G.; Sun, K.; Li, L.; Zu, X.; Han, T.; Huang, H. Mechanism of quercetin therapeutic targets for Alzheimer disease and type 2 diabetes mellitus. Sci. Rep. 2021, 11, 22959. [Google Scholar] [CrossRef] [PubMed]
- Hung, C.H.; Chan, S.H.; Chu, P.M.; Tsai, K.L. Quercetin is a potent anti-atherosclerotic compound by activation of SIRT1 signaling under oxLDL stimulation. Mol. Nutr. Food Res. 2015, 59, 1905–1917. [Google Scholar] [CrossRef]
- Bouchelaghem, S. Propolis characterization and antimicrobial activities against Staphylococcus aureus and Candida albicans: A review. Saudi J. Biol. Sci. 2022, 29, 1936–1946. [Google Scholar] [CrossRef] [PubMed]
- Nahmias, Y.; Goldwasser, J.; Casali, M.; van Poll, D.; Wakita, T.; Chung, R.T.; Yarmush, M.L. Apolipoprotein B-dependent hepatitis C virus secretion is inhibited by the grapefruit flavonoid naringenin. Hepatology 2008, 47, 1437–1445. [Google Scholar] [CrossRef]
- Yang, Z.; Kuboyama, T.; Tohda, C. Naringenin promotes microglial M2 polarization and Abeta degradation enzyme expression. Phytother. Res. 2019, 33, 1114–1121. [Google Scholar] [CrossRef]
- Sarubbo, F.; Ramis, M.R.; Kienzer, C.; Aparicio, S.; Esteban, S.; Miralles, A.; Moranta, D. Chronic Silymarin, Quercetin and Naringenin Treatments Increase Monoamines Synthesis and Hippocampal Sirt1 Levels Improving Cognition in Aged Rats. J. Neuroimmune Pharmacol. 2018, 13, 24–38. [Google Scholar] [CrossRef]
- Khalatbary, A.R.; Khademi, E. The green tea polyphenolic catechin epigallocatechin gallate and neuroprotection. Nutr. Neurosci. 2020, 23, 281–294. [Google Scholar] [CrossRef]
- Calderaro, A.; Patane, G.T.; Tellone, E.; Barreca, D.; Ficarra, S.; Misiti, F.; Lagana, G. The Neuroprotective Potentiality of Flavonoids on Alzheimer’s Disease. Int. J. Mol. Sci. 2022, 23, 14835. [Google Scholar] [CrossRef]
- Arafa, M.H.; Atteia, H.H. Protective Role of Epigallocatechin Gallate in a Rat Model of Cisplatin-Induced Cerebral Inflammation and Oxidative Damage: Impact of Modulating NF-κB and Nrf2. Neurotox. Res. 2020, 37, 380–396. [Google Scholar] [CrossRef]
- Chang, X.; Rong, C.; Chen, Y.; Yang, C.; Hu, Q.; Mo, Y.; Zhang, C.; Gu, X.; Zhang, L.; He, W.; et al. (-)-Epigallocatechin-3-gallate attenuates cognitive deterioration in Alzheimer’s disease model mice by upregulating neprilysin expression. Exp. Cell Res. 2015, 334, 136–145. [Google Scholar] [CrossRef]
- Nardini, M.; Garaguso, I. Characterization of bioactive compounds and antioxidant activity of fruit beers. Food Chem. 2020, 305, 125437. [Google Scholar] [CrossRef]
- Taheri, Y.; Suleria, H.A.R.; Martins, N.; Sytar, O.; Beyatli, A.; Yeskaliyeva, B.; Seitimova, G.; Salehi, B.; Semwal, P.; Painuli, S.; et al. Myricetin bioactive effects: Moving from preclinical evidence to potential clinical applications. BMC Complement. Med. Ther. 2020, 20, 241. [Google Scholar] [CrossRef] [PubMed]
- Imran, M.; Saeed, F.; Hussain, G.; Imran, A.; Mehmood, Z.; Gondal, T.A.; El-Ghorab, A.; Ahmad, I.; Pezzani, R.; Arshad, M.U.; et al. Myricetin: A comprehensive review on its biological potentials. Food Sci. Nutr. 2021, 9, 5854–5868. [Google Scholar] [CrossRef]
- Sun, L.; Xu, P.; Fu, T.; Huang, X.; Song, J.; Chen, M.; Tian, X.; Yin, H.; Han, J. Myricetin against ischemic cerebral injury in rat middle cerebral artery occlusion model. Mol. Med. Rep. 2018, 17, 3274–3280. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.H.; Lee, S.H.; Jung, K.; Yoo, H.; Park, G. Inhibitory Effects of Myricetin on Lipopolysaccharide-Induced Neuroinflammation. Brain Sci. 2020, 10, 32. [Google Scholar] [CrossRef]
- Kaewmool, C.; Udomruk, S.; Phitak, T.; Pothacharoen, P.; Kongtawelert, P. Cyanidin-3-O-Glucoside Protects PC12 Cells Against Neuronal Apoptosis Mediated by LPS-Stimulated BV2 Microglial Activation. Neurotox. Res. 2020, 37, 111–125. [Google Scholar] [CrossRef]
- Wang, W.; Zhu, G.; Wang, Y.; Li, W.; Yi, S.; Wang, K.; Fan, L.; Tang, J.; Chen, R. Multi-Omics Integration in Mice With Parkinson’s Disease and the Intervention Effect of Cyanidin-3-O-Glucoside. Front. Aging Neurosci. 2022, 14, 877078. [Google Scholar] [CrossRef]
- Chiocchio, I.; Prata, C.; Mandrone, M.; Ricciardiello, F.; Marrazzo, P.; Tomasi, P.; Angeloni, C.; Fiorentini, D.; Malaguti, M.; Poli, F.; et al. Leaves and Spiny Burs of Castanea Sativa from an Experimental Chestnut Grove: Metabolomic Analysis and Anti-Neuroinflammatory Activity. Metabolites 2020, 10, 408. [Google Scholar] [CrossRef] [PubMed]
- Badanjak, K.; Fixemer, S.; Smajic, S.; Skupin, A.; Grunewald, A. The Contribution of Microglia to Neuroinflammation in Parkinson’s Disease. Int. J. Mol. Sci. 2021, 22, 4676. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, A. Postmortem studies in Parkinson’s disease. Dialogues Clin. Neurosci. 2004, 6, 281–293. [Google Scholar] [CrossRef]
- Patel, M.; Singh, S. Apigenin Attenuates Functional and Structural Alterations via Targeting NF-kB/Nrf2 Signaling Pathway in LPS-Induced Parkinsonism in Experimental Rats: Apigenin Attenuates LPS-Induced Parkinsonism in Experimental Rats. Neurotox. Res. 2022, 40, 941–960. [Google Scholar] [CrossRef]
- Budzynska, B.; Faggio, C.; Kruk-Slomka, M.; Samec, D.; Nabavi, S.F.; Sureda, A.; Devi, K.P.; Nabavi, S.M. Rutin as Neuroprotective Agent: From Bench to Bedside. Curr. Med. Chem. 2019, 26, 5152–5164. [Google Scholar] [CrossRef]
- Sun, X.Y.; Li, L.J.; Dong, Q.X.; Zhu, J.; Huang, Y.R.; Hou, S.J.; Yu, X.L.; Liu, R.T. Rutin prevents tau pathology and neuroinflammation in a mouse model of Alzheimer’s disease. J. Neuroinflamm. 2021, 18, 131. [Google Scholar] [CrossRef]
- Wang, S.; Wei, H.; Cai, M.; Lu, Y.; Hou, W.; Yang, Q.; Dong, H.; Xiong, L. Genistein attenuates brain damage induced by transient cerebral ischemia through up-regulation of ERK activity in ovariectomized mice. Int. J. Biol. Sci. 2014, 10, 457–465. [Google Scholar] [CrossRef]
- Shi, B.; Chen, M.; Xia, Z.; Tang, W.; Li, Y.; Qin, C.; Ahmadi, A.; Huang, C.; Xu, H. Genistein attenuates neuroinflammation and oxidative stress and improves cognitive impairment in a rat model of sepsis-associated encephalopathy: Potential role of the Nrf2 signaling pathway. Metab. Brain Dis. 2023, 38, 339–347. [Google Scholar] [CrossRef]
- Nakajima, A.; Ohizumi, Y. Potential Benefits of Nobiletin, A Citrus Flavonoid, against Alzheimer’s Disease and Parkinson’s Disease. Int. J. Mol. Sci. 2019, 20, 3380. [Google Scholar] [CrossRef] [PubMed]
- Kimura, J.; Shimizu, K.; Kajima, K.; Yokosuka, A.; Mimaki, Y.; Oku, N.; Ohizumi, Y. Nobiletin Reduces Intracellular and Extracellular beta-Amyloid in iPS Cell-Derived Alzheimer’s Disease Model Neurons. Biol. Pharm. Bull. 2018, 41, 451–457. [Google Scholar] [CrossRef] [PubMed]
- Qi, G.; Mi, Y.; Fan, R.; Li, R.; Liu, Z.; Liu, X. Nobiletin Protects against Systemic Inflammation-Stimulated Memory Impairment via MAPK and NF-κB Signaling Pathways. J. Agric. Food Chem. 2019, 67, 5122–5134. [Google Scholar] [CrossRef]
- Ghasemi-Tarie, R.; Kiasalari, Z.; Fakour, M.; Khorasani, M.; Keshtkar, S.; Baluchnejadmojarad, T.; Roghani, M. Nobiletin prevents amyloid beta(1-40)-induced cognitive impairment via inhibition of neuroinflammation and oxidative/nitrosative stress. Metab. Brain Dis. 2022, 37, 1337–1349. [Google Scholar] [CrossRef] [PubMed]
- Hua, F.Z.; Ying, J.; Zhang, J.; Wang, X.F.; Hu, Y.H.; Liang, Y.P.; Liu, Q.; Xu, G.H. Naringenin pre-treatment inhibits neuroapoptosis and ameliorates cognitive impairment in rats exposed to isoflurane anesthesia by regulating the PI3/Akt/PTEN signalling pathway and suppressing NF-κB-mediated inflammation. Int. J. Mol. Med. 2016, 38, 1271–1280. [Google Scholar] [CrossRef]
- Levac, D.; Colquhoun, H.; O’Brien, K.K. Scoping studies: Advancing the methodology. Implement. Sci. 2010, 5, 69. [Google Scholar] [CrossRef]
- Zhang, H.; Hassan, Y.I.; Liu, R.; Mats, L.; Yang, C.; Liu, C.; Tsao, R. Molecular Mechanisms Underlying the Absorption of Aglycone and Glycosidic Flavonoids in a Caco-2 BBe1 Cell Model. ACS Omega 2020, 5, 10782–10793. [Google Scholar] [CrossRef] [PubMed]
- Kosinova, P.; Berka, K.; Wykes, M.; Otyepka, M.; Trouillas, P. Positioning of antioxidant quercetin and its metabolites in lipid bilayer membranes: Implication for their lipid-peroxidation inhibition. J. Phys. Chem. B. 2012, 116, 1309–1318. [Google Scholar] [CrossRef]
- Raghunathan, M.; Zubovski, Y.; Venable, R.M.; Pastor, R.W.; Nagle, J.F.; Tristram-Nagle, S. Structure and elasticity of lipid membranes with genistein and daidzein bioflavinoids using X-ray scattering and MD simulations. J. Phys. Chem. B. 2012, 116, 3918–3927. [Google Scholar] [CrossRef]
- Tarahovsky, Y.S.; Kim, Y.A.; Yagolnik, E.A.; Muzafarov, E.N. Flavonoid-membrane interactions: Involvement of flavonoid-metal complexes in raft signaling. Biochim. Biophys. Acta 2014, 1838, 1235–1246. [Google Scholar] [CrossRef] [PubMed]
- Hendrich, A.B. Flavonoid-membrane interactions: Possible consequences for biological effects of some polyphenolic compounds. Acta Pharmacol. Sin. 2006, 27, 27–40. [Google Scholar] [CrossRef]
- Middleton, E., Jr.; Kandaswami, C.; Theoharides, T.C. The effects of plant flavonoids on mammalian cells: Implications for inflammation, heart disease, and cancer. Pharmacol. Rev. 2000, 52, 673–751. [Google Scholar]
- Singh, P.; Mishra, S.K.; Noel, S.; Sharma, S.; Rath, S.K. Acute exposure of apigenin induces hepatotoxicity in Swiss mice. PLoS ONE 2012, 7, e31964. [Google Scholar] [CrossRef]
- Zhang, Z.; Chen, S.; Mei, H.; Xuan, J.; Guo, X.; Couch, L.; Dobrovolsky, V.N.; Guo, L.; Mei, N. Ginkgo biloba leaf extract induces DNA damage by inhibiting topoisomerase II activity in human hepatic cells. Sci. Rep. 2015, 5, 14633. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).