Role of Histone Deacetylases in the Pathogenesis of Salivary Gland Tumors and Therapeutic Targeting Options
Abstract
1. Introduction
2. Molecular Aspects of SGT Pathology
2.1. Genetic Changes in SGTs
| Tumor Behavior | Tumor Type | Genetic Aberration | Prevalence | Clinical Course |
|---|---|---|---|---|
| Malignant | MECs | MECT1/MAML2 fusions | 40–80% | Favorable prognosis [25] |
| CRTC3/MAML2 fusions | 6%/younger ages | Favorable prognosis [24] | ||
| AdCCs | MYB/NFIB gene fusions | 50% | Unfavorable prognosis [25] | |
| AciCCs | HTN3/MSANTD3 fusions | <5% | Indolent course [26] | |
| MSANTD3 aberrations | 4–15% | |||
| MASCs | ETV6/NTRK3 fusions | Majority | Favorable prognosis [34] | |
| (Hyalinizing) CCCs | EWSR1/ATF1 fusions | 93% | Indolent course [35] | |
| EWSR1 rearrangement | 82% | |||
| Intraductal carcinoma | NCOA4-RET | 35% | Mean OS: 5 years [36] | |
| TRIM27-RET | ~10% | Aggressive/apocrine variant [36] | ||
| Microsecretory adenocarcinoma | MEF2C-SS18 fusion | Majority | Malignant nature [37] | |
| PAC and CASG | PRKD1/2/3 translocations or mutations | PRKD1/2/3 fusions: 13% of PACs; PRKD1 mutations: 56–73% PACs | Indolent course [38] | |
| PRKD1/2/3 fusions: 43–80% CASGs; PRKD1 mutations: 20% CASGs | ||||
| BCAC | PIK3CA mutations | ~10–30% | Unfavorable prognosis [39,40] | |
| CAexPA | PLAG1/HMGA2 fusions | 36% | Indolent course [41] | |
| Benign | PAs | PLAG1 translocations | 50% | Indolent course [42] |
| HMGA2 translocations | 34–40% | |||
| BCA | CTNNB1 mutations | 60% | Indolent course [43] |
2.2. Epigenetic Changes in SGTs
| SG Entity | Aberration Form | Related Molecule | References |
|---|---|---|---|
| AdCCs | ↑ methylation | EN1, RASSF1A, RECK, Stratifin (14-3-3 s), RUNX3 | [63,66,83,84,85] |
| ↓ methylation | SBSN, AQP1 | [65] | |
| ↑ Promoter methylation | E-cadherin, RASSF1A, RARb2 | [54,59,67,86] | |
| Histone trimethylation | H3K9me3 | [64,87] | |
| ↑ acetylation | H3K9Ac, H3K18Ac | [71,88] | |
| miR-17-92 ↑regulation | miR-17/-20a | [81] | |
| MECs | Aberrant expression of ncRNAs | NONHSAT154433.1, has-circ-0012342 | [76,82] |
| ↑ methylation | RUNX3, H3K9Me3 | [52,85,88] | |
| ↑ acetylation | H3K9Ac, H3K18Ac | [71,88] | |
| SDCs | ↑ methylation | GALR-1/-2 | [56] |
| ↑ Promoter methylation | RASSF1A, RARb2, | [67] | |
| PAs | Dysregulated expression (PLAG1 gene related) | 9110 lncRNAs/7750 mRNAs | [80] |
| CAexPA | ↑ Promoter methylation | CDH1 (E-cadherin) | [49,89] |
| ACCs | ↑ Promoter methylation | RASSF1A, RARb2 | [67] |
3. Role of Histone Deacetylation in SGTs
3.1. Biochemical and Functional Aspects of Histone Acetylation/Deacetylation
3.2. Role of HDACs in Different SGT Types
4. Targeting Options of HDACis in SGTs
4.1. HDAC Inhibition
4.1.1. HDACis in Mucoepidermoid Carcinoma (MEC)
4.1.2. HDACis in Adenoid Cystic Carcinoma (AdCC)
4.1.3. HDACis in Salivary Gland Ductal Adenocarcinoma (SGDA)
4.2. Phase-I Clinical Studies of HDACis in SGTs
4.2.1. Vorinostat in a Phase I Study
4.2.2. KA2507 Selective HDAC6 Inhibitor
4.3. Phase-II Trials of HDACis in SGTs
4.3.1. SAHA Treatment in AdCC
4.3.2. Combination of Pembrolizumab and Vorinostat in SGTs
5. Conclusions—Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Cunha, J.L.S.; Hernandez-Guerrero, J.C.; de Almeida, O.P.; Soares, C.D.; Mosqueda-Taylor, A. Salivary Gland Tumors: A Retrospective Study of 164 Cases from a Single Private Practice Service in Mexico and Literature Review. Head Neck Pathol. 2021, 15, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.; Hao, Y.; Huang, M.X.; Ma, D.Q.; Chen, Y.; Luo, H.Y.; Gao, Y.; Cao, Z.Q.; Peng, X.; Yu, G.Y. Salivary gland tumours in a northern Chinese population: A 50-year retrospective study of 7190 cases. Int. J. Oral Maxillofac. Surg. 2017, 46, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Sentani, K.; Ogawa, I.; Ozasa, K.; Sadakane, A.; Utada, M.; Tsuya, T.; Kajihara, H.; Yonehara, S.; Takeshima, Y.; Yasui, W. Characteristics of 5015 Salivary Gland Neoplasms Registered in the Hiroshima Tumor Tissue Registry over a Period of 39 Years. J. Clin. Med. 2019, 8, 566. [Google Scholar] [CrossRef] [PubMed]
- Lawal, A.O.; Adisa, A.O.; Kolude, B.; Adeyemi, B.F.; Olajide, M.A. A review of 413 salivary gland tumours in the head and neck region. J. Clin. Exp. Dent. 2013, 5, e218–e222. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Rooper, L.M. Challenges in Minor Salivary Gland Biopsies: A Practical Approach to Problematic Histologic Patterns. Head Neck Pathol. 2019, 13, 476–484. [Google Scholar] [CrossRef]
- Skalova, A.; Hyrcza, M.D.; Leivo, I. Update from the 5th Edition of the World Health Organization Classification of Head and Neck Tumors: Salivary Glands. Head Neck Pathol. 2022, 16, 40–53. [Google Scholar] [CrossRef]
- da Silva, L.P.; Serpa, M.S.; Viveiros, S.K.; Sena, D.A.C.; de Carvalho Pinho, R.F.; de Abreu Guimaraes, L.D.; de Sousa Andrade, E.S.; Dias Pereira, J.R.; Silveira, M.; Sobral, A.P.V.; et al. Salivary gland tumors in a Brazilian population: A 20-year retrospective and multicentric study of 2292 cases. J. Craniomaxillofac. Surg. 2018, 46, 2227–2233. [Google Scholar] [CrossRef]
- Mariz, B.; do Socorro Queiroz Feio, P.; Roza, A.; de Andrade, B.A.B.; Agostini, M.; Romanach, M.J.; Fonseca, F.P.; Pontes, H.A.R.; Ribeiro, A.C.P.; Brandao, T.B.; et al. Clinical predictors of malignancy in palatal salivary gland tumors. Oral Dis. 2019, 25, 1919–1924. [Google Scholar] [CrossRef]
- Vielh, P.; Klijanienko, J. Salivary Gland Tumours; S. Karger AG: Basel, Switzerland, 2000; ISBN 978-3-318-00544-8. [Google Scholar] [CrossRef]
- Habermann, C.R.; Gossrau, P.; Graessner, J.; Arndt, C.; Cramer, M.C.; Reitmeier, F.; Jaehne, M.; Adam, G. Diffusion-weighted echo-planar MRI: A valuable tool for differentiating primary parotid gland tumors? Rofo 2005, 177, 940–945. [Google Scholar] [CrossRef]
- Skalova, A.; Leivo, I.; Hellquist, H.; Agaimy, A.; Simpson, R.H.W.; Stenman, G.; Vander Poorten, V.; Bishop, J.A.; Franchi, A.; Hernandez-Prera, J.C.; et al. High-grade Transformation/Dedifferentiation in Salivary Gland Carcinomas: Occurrence Across Subtypes and Clinical Significance. Adv. Anat. Pathol. 2021, 28, 107–118. [Google Scholar] [CrossRef]
- Alfieri, S.; Granata, R.; Bergamini, C.; Resteghini, C.; Bossi, P.; Licitra, L.F.; Locati, L.D. Systemic therapy in metastatic salivary gland carcinomas: A pathology-driven paradigm? Oral Oncol. 2017, 66, 58–63. [Google Scholar] [CrossRef]
- Hellquist, H.; Skalova, A.; Barnes, L.; Cardesa, A.; Thompson, L.D.; Triantafyllou, A.; Williams, M.D.; Devaney, K.O.; Gnepp, D.R.; Bishop, J.A.; et al. Cervical Lymph Node Metastasis in High-Grade Transformation of Head and Neck Adenoid Cystic Carcinoma: A Collective International Review. Adv. Ther. 2016, 33, 357–368. [Google Scholar] [CrossRef]
- Gatta, G.; Guzzo, M.; Locati, L.D.; McGurk, M.; Prott, F.J. Major and minor salivary gland tumours. Crit. Rev. Oncol. Hematol. 2020, 152, 102959. [Google Scholar] [CrossRef]
- Herman, M.P.; Werning, J.W.; Morris, C.G.; Kirwan, J.M.; Amdur, R.J.; Mendenhall, W.M. Elective neck management for high-grade salivary gland carcinoma. Am. J. Otolaryngol. 2013, 34, 205–208. [Google Scholar] [CrossRef] [PubMed]
- Saarilahti, K.; Kouri, M.; Collan, J.; Hamalainen, T.; Atula, T.; Joensuu, H.; Tenhunen, M. Intensity modulated radiotherapy for head and neck cancer: Evidence for preserved salivary gland function. Radiother. Oncol. 2005, 74, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Sato, M.; Yamamoto, H.; Hatanaka, Y.; Nishijima, T.; Jiromaru, R.; Yasumatsu, R.; Taguchi, K.; Masuda, M.; Nakagawa, T.; Oda, Y. Wnt/beta-catenin signal alteration and its diagnostic utility in basal cell adenoma and histologically similar tumors of the salivary gland. Pathol. Res. Pract. 2018, 214, 586–592. [Google Scholar] [CrossRef] [PubMed]
- Wemmert, S.; Willnecker, V.; Sauter, B.; Schuh, S.; Brunner, C.; Bohle, R.M.; Urbschat, S.; Schick, B. Genomic alterations in Warthin tumors of the parotid gland. Oncol. Rep. 2014, 31, 1899–1904. [Google Scholar] [CrossRef] [PubMed]
- Mito, J.K.; Jo, V.Y.; Chiosea, S.I.; Dal Cin, P.; Krane, J.F. HMGA2 is a specific immunohistochemical marker for pleomorphic adenoma and carcinoma ex-pleomorphic adenoma. Histopathology 2017, 71, 511–521. [Google Scholar] [CrossRef]
- Stenman, G. Fusion oncogenes in salivary gland tumors: Molecular and clinical consequences. Head Neck Pathol. 2013, 7 (Suppl. 1), S12–S19. [Google Scholar] [CrossRef]
- Skalova, A.; Vanecek, T.; Simpson, R.H.; Vazmitsel, M.A.; Majewska, H.; Mukensnabl, P.; Hauer, L.; Andrle, P.; Hosticka, L.; Grossmann, P.; et al. CRTC1-MAML2 and CRTC3-MAML2 fusions were not detected in metaplastic Warthin tumor and metaplastic pleomorphic adenoma of salivary glands. Am. J. Surg. Pathol. 2013, 37, 1743–1750. [Google Scholar] [CrossRef] [PubMed]
- Seethala, R.R.; Dacic, S.; Cieply, K.; Kelly, L.M.; Nikiforova, M.N. A reappraisal of the MECT1/MAML2 translocation in salivary mucoepidermoid carcinomas. Am. J. Surg. Pathol. 2010, 34, 1106–1121. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, T.; Miyabe, S.; Okabe, M.; Sakuma, H.; Ijichi, K.; Hasegawa, Y.; Nagatsuka, H.; Shimozato, K.; Inagaki, H. Clinicopathological significance of the CRTC3-MAML2 fusion transcript in mucoepidermoid carcinoma. Mod. Pathol. 2009, 22, 1575–1581. [Google Scholar] [CrossRef]
- Yin, L.X.; Ha, P.K. Genetic alterations in salivary gland cancers. Cancer 2016, 122, 1822–1831. [Google Scholar] [CrossRef]
- Andreasen, S.; Varma, S.; Barasch, N.; Thompson, L.D.R.; Miettinen, M.; Rooper, L.; Stelow, E.B.; Agander, T.K.; Seethala, R.R.; Chiosea, S.I.; et al. The HTN3-MSANTD3 Fusion Gene Defines a Subset of Acinic Cell Carcinoma of the Salivary Gland. Am. J. Surg. Pathol. 2019, 43, 489–496. [Google Scholar] [CrossRef] [PubMed]
- Andersson, M.K.; Stenman, G. The landscape of gene fusions and somatic mutations in salivary gland neoplasms—Implications for diagnosis and therapy. Oral Oncol. 2016, 57, 63–69. [Google Scholar] [CrossRef]
- Steiner, P.; Andreasen, S.; Grossmann, P.; Hauer, L.; Vanecek, T.; Miesbauerova, M.; Santana, T.; Kiss, K.; Slouka, D.; Skalova, A. Prognostic significance of 1p36 locus deletion in adenoid cystic carcinoma of the salivary glands. Virchows Arch. 2018, 473, 471–480. [Google Scholar] [CrossRef]
- Papadaki, H.; Finkelstein, S.D.; Kounelis, S.; Bakker, A.; Swalsky, P.A.; Kapadia, S.B. The role of p53 mutation and protein expression in primary and recurrent adenoid cystic carcinoma. Hum. Pathol. 1996, 27, 567–572. [Google Scholar] [CrossRef]
- Skalova, A.; Vanecek, T.; Sima, R.; Laco, J.; Weinreb, I.; Perez-Ordonez, B.; Starek, I.; Geierova, M.; Simpson, R.H.; Passador-Santos, F.; et al. Mammary analogue secretory carcinoma of salivary glands, containing the ETV6-NTRK3 fusion gene: A hitherto undescribed salivary gland tumor entity. Am. J. Surg. Pathol. 2010, 34, 599–608. [Google Scholar] [CrossRef]
- Chapman, E.; Skalova, A.; Ptakova, N.; Martinek, P.; Goytain, A.; Tucker, T.; Xiong, W.; Leader, M.; Kudlow, B.A.; Haimes, J.D.; et al. Molecular Profiling of Hyalinizing Clear Cell Carcinomas Revealed a Subset of Tumors Harboring a Novel EWSR1-CREM Fusion: Report of 3 Cases. Am. J. Surg. Pathol. 2018, 42, 1182–1189. [Google Scholar] [CrossRef]
- Garcia-Manero, G.; Issa, J.P. Histone deacetylase inhibitors: A review of their clinical status as antineoplastic agents. Cancer Investig. 2005, 23, 635–642. [Google Scholar] [CrossRef] [PubMed]
- Toyota, M.; Issa, J.P. Epigenetic changes in solid and hematopoietic tumors. Semin. Oncol. 2005, 32, 521–530. [Google Scholar] [CrossRef] [PubMed]
- Khalele, B.A. Systematic review of mammary analog secretory carcinoma of salivary glands at 7 years after description. Head Neck 2017, 39, 1243–1248. [Google Scholar] [CrossRef]
- Antonescu, C.R.; Katabi, N.; Zhang, L.; Sung, Y.S.; Seethala, R.R.; Jordan, R.C.; Perez-Ordoñez, B.; Have, C.; Asa, S.L.; Leong, I.T. EWSR1-ATF1 fusion is a novel and consistent finding in hyalinizing clear-cell carcinoma of salivary gland. Genes Chromosomes Cancer 2011, 50, 559–570. [Google Scholar] [CrossRef]
- Skalova, A.; Vanecek, T.; Uro-Coste, E.; Bishop, J.A.; Weinreb, I.; Thompson, L.D.R.; de Sanctis, S.; Schiavo-Lena, M.; Laco, J.; Badoual, C.; et al. Molecular Profiling of Salivary Gland Intraductal Carcinoma Revealed a Subset of Tumors Harboring NCOA4-RET and Novel TRIM27-RET Fusions: A Report of 17 cases. Am. J. Surg. Pathol. 2018, 42, 1445–1455. [Google Scholar] [CrossRef]
- Bishop, J.A.; Weinreb, I.; Swanson, D.; Westra, W.H.; Qureshi, H.S.; Sciubba, J.; MacMillan, C.; Rooper, L.M.; Dickson, B.C. Microsecretory Adenocarcinoma: A Novel Salivary Gland Tumor Characterized by a Recurrent MEF2C-SS18 Fusion. Am. J. Surg. Pathol. 2019, 43, 1023–1032. [Google Scholar] [CrossRef]
- de Jager, V.D.; de Visscher, S.A.; Schuuring, E.; Doff, J.J.; van Kempen, L.C. A novel PPP2R2A:: PRKD1 fusion in a cribriform adenocarcinoma of salivary gland. Genes Chromosomes Cancer 2023, 62, 297–300. [Google Scholar] [CrossRef]
- Jo, V.Y.; Sholl, L.M.; Krane, J.F. Distinctive Patterns of CTNNB1 (beta-Catenin) Alterations in Salivary Gland Basal Cell Adenoma and Basal Cell Adenocarcinoma. Am. J. Surg. Pathol. 2016, 40, 1143–1150. [Google Scholar] [CrossRef]
- Wilson, T.C.; Ma, D.; Tilak, A.; Tesdahl, B.; Robinson, R.A. Next-generation sequencing in salivary gland basal cell adenocarcinoma and basal cell adenoma. Head Neck Pathol. 2016, 10, 494–500. [Google Scholar]
- de Brito, B.S.; Giovanelli, N.; Egal, E.S.; Sanchez-Romero, C.; Nascimento, J.S.; Martins, A.S.; Tincani, A.J.; Del Negro, A.; Gondak, R.O.; Almeida, O.P.; et al. Loss of expression of Plag1 in malignant transformation from pleomorphic adenoma to carcinoma ex pleomorphic adenoma. Hum. Pathol. 2016, 57, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Owosho, A.A.; Adesina, O.M.; Odujoko, O.; Akinyemi, H.; Komolafe, A.; Tadros, S.; Bauer, R.; Summersgill, K.F. HMGA2 Immunoexpression is frequent in salivary gland pleomorphic adenoma: Immunohistochemical and molecular analyses of PLAG1 and HMGA2 in 25 cases. Int. J. Clin. Exp. Pathol. 2022, 15, 63–71. [Google Scholar]
- Lee, Y.-H.; Huang, W.-C.; Hsieh, M.-S. CTNNB1 mutations in basal cell adenoma of the salivary gland. J. Formos. Med. Assoc. 2018, 117, 894–901. [Google Scholar] [PubMed]
- Waddington, C.H. The epigenotype. 1942. Int. J. Epidemiol. 2012, 41, 10–13. [Google Scholar] [CrossRef]
- Sharma, S.; Kelly, T.K.; Jones, P.A. Epigenetics in cancer. Carcinogenesis 2010, 31, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Williams, M.D.; Chakravarti, N.; Kies, M.S.; Maruya, S.; Myers, J.N.; Haviland, J.C.; Weber, R.S.; Lotan, R.; El-Naggar, A.K. Implications of methylation patterns of cancer genes in salivary gland tumors. Clin. Cancer Res. 2006, 12, 7353–7358. [Google Scholar] [CrossRef] [PubMed]
- Schache, A.G.; Hall, G.; Woolgar, J.A.; Nikolaidis, G.; Triantafyllou, A.; Lowe, D.; Risk, J.M.; Shaw, R.J.; Liloglou, T. Quantitative promoter methylation differentiates carcinoma ex pleomorphic adenoma from pleomorphic salivary adenoma. Br. J. Cancer 2010, 103, 1846–1851. [Google Scholar] [CrossRef]
- Hu, Y.H.; Zhang, C.Y.; Tian, Z.; Wang, L.Z.; Li, J. Aberrant protein expression and promoter methylation of p16 gene are correlated with malignant transformation of salivary pleomorphic adenoma. Arch. Pathol. Lab. Med. 2011, 135, 882–889. [Google Scholar] [CrossRef]
- Xia, L.; Hu, Y.; Gu, T.; Wang, L.; Tian, Z. Promoter hypermethylation may contribute to E-cadherin repression in the human salivary carcinoma ex pleomorphic adenoma. Int. J. Oncol. 2018, 52, 496–504. [Google Scholar] [CrossRef]
- Nikolic, N.; Carkic, J.; Ilic Dimitrijevic, I.; Eljabo, N.; Radunovic, M.; Anicic, B.; Tanic, N.; Falk, M.; Milasin, J. P14 methylation: An epigenetic signature of salivary gland mucoepidermoid carcinoma in the Serbian population. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2018, 125, 52–58. [Google Scholar] [CrossRef]
- Guo, X.L.; Sun, S.Z.; Wang, W.X.; Wei, F.C.; Yu, H.B.; Ma, B.L. Alterations of p16INK4a tumour suppressor gene in mucoepidermoid carcinoma of the salivary glands. Int. J. Oral Maxillofac. Surg. 2007, 36, 350–353. [Google Scholar] [CrossRef]
- Sasahira, T.; Kurihara, M.; Yamamoto, K.; Bhawal, U.K.; Kirita, T.; Kuniyasu, H. Downregulation of runt-related transcription factor 3 associated with poor prognosis of adenoid cystic and mucoepidermoid carcinomas of the salivary gland. Cancer Sci. 2011, 102, 492–497. [Google Scholar] [CrossRef] [PubMed]
- Maruya, S.; Kurotaki, H.; Shimoyama, N.; Kaimori, M.; Shinkawa, H.; Yagihashi, S. Expression of p16 protein and hypermethylation status of its promoter gene in adenoid cystic carcinoma of the head and neck. ORL J. Otorhinolaryngol. Relat. Spec. 2003, 65, 26–32. [Google Scholar] [CrossRef]
- Li, J.; El-Naggar, A.; Mao, L. Promoter methylation of p16INK4a, RASSF1A, and DAPK is frequent in salivary adenoid cystic carcinoma. Cancer 2005, 104, 771–776. [Google Scholar] [CrossRef] [PubMed]
- Daa, T.; Kashima, K.; Kondo, Y.; Yada, N.; Suzuki, M.; Yokoyama, S. Aberrant methylation in promoter regions of cyclin-dependent kinase inhibitor genes in adenoid cystic carcinoma of the salivary gland. APMIS 2008, 116, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Kanazawa, T.; Misawa, K.; Fukushima, H.; Misawa, Y.; Sato, Y.; Maruta, M.; Imayoshi, S.; Kusaka, G.; Kawabata, K.; Mineta, H. Epigenetic inactivation of galanin receptors in salivary duct carcinoma of the parotid gland: Potential utility as biomarkers for prognosis. Oncol. Lett. 2018, 15, 9043–9050. [Google Scholar] [CrossRef]
- Wang, Z.; Ling, S.; Rettig, E.; Sobel, R.; Tan, M.; Fertig, E.J.; Considine, M.; El-Naggar, A.K.; Brait, M.; Fakhry, C. Epigenetic screening of salivary gland mucoepidermoid carcinoma identifies hypomethylation of CLIC3 as a common alteration. Oral Oncol. 2015, 51, 1120. [Google Scholar] [CrossRef]
- Shieh, Y.S.; Shiah, S.G.; Jeng, H.H.; Lee, H.S.; Wu, C.W.; Chang, L.C. DNA methyltransferase 1 expression and promoter methylation of E-cadherin in mucoepidermoid carcinoma. Cancer 2005, 104, 1013–1021. [Google Scholar]
- Zhang, C.Y.; Mao, L.; Li, L.; Tian, Z.; Zhou, X.J.; Zhang, Z.Y.; Li, J. Promoter methylation as a common mechanism for inactivating E-cadherin in human salivary gland adenoid cystic carcinoma. Cancer 2007, 110, 87–95. [Google Scholar] [CrossRef]
- Bell, D.; Bell, A.H.; Bondaruk, J.; Hanna, E.Y.; Weber, R.S. In-depth characterization of the salivary adenoid cystic carcinoma transcriptome with emphasis on dominant cell type. Cancer 2016, 122, 1513–1522. [Google Scholar] [CrossRef]
- Bell, D.; Bell, A.; Roberts, D.; Weber, R.S.; El-Naggar, A.K. Developmental transcription factor EN1--a novel biomarker in human salivary gland adenoid cystic carcinoma. Cancer 2012, 118, 1288–1292. [Google Scholar] [CrossRef]
- Baba, S.; Akashi, T.; Kayamori, K.; Ohuchi, T.; Ogawa, I.; Kubota, N.; Nakano, K.; Nagatsuka, H.; Hasegawa, H.; Matsuzaka, K. Homeobox transcription factor engrailed homeobox 1 is a possible diagnostic marker for adenoid cystic carcinoma and polymorphous adenocarcinoma. Pathol. Int. 2021, 71, 113–123. [Google Scholar] [CrossRef]
- Zhang, C.-Y.; Zhao, Y.-X.; Xia, R.-H.; Han, J.; Wang, B.-S.; Tian, Z.; Wang, L.-Z.; Hu, Y.-H.; Li, J. RASSF1A promoter hypermethylation is a strong biomarker of poor survival in patients with salivary adenoid cystic carcinoma in a Chinese population. PLoS ONE 2014, 9, e110159. [Google Scholar]
- Xia, R.; Zhou, R.; Tian, Z.; Zhang, C.; Wang, L.; Hu, Y.; Han, J.; Li, J. High expression of H3K9me3 is a strong predictor of poor survival in patients with salivary adenoid cystic carcinoma. Arch. Pathol. Lab. Med. 2013, 137, 1761–1769. [Google Scholar] [CrossRef]
- Shao, C.; Sun, W.; Tan, M.; Glazer, C.A.; Bhan, S.; Zhong, X.; Fakhry, C.; Sharma, R.; Westra, W.H.; Hoque, M.O. Integrated, Genome-Wide Screening for Hypomethylated Oncogenes in Salivary Gland Adenoid Cystic CarcinomaScreening for Hypomethylated Oncogenes in Salivary Gland ACC. Clin. Cancer Res. 2011, 17, 4320–4330. [Google Scholar] [PubMed]
- Uchida, D.; Begum, N.; Almofti, A.; Kawamata, H.; Yoshida, H.; Sato, M. Frequent downregulation of 14-3-3 sigma protein and hypermethylation of 14-3-3 sigma gene in salivary gland adenoid cystic carcinoma. Br. J. Cancer 2004, 91, 1131–1138. [Google Scholar] [CrossRef]
- Lee, E.-S.; Issa, J.-P.; Roberts, D.B.; Williams, M.D.; Weber, R.S.; Kies, M.S.; El-Naggar, A.K. Quantitative promoter hypermethylation analysis of cancer-related genes in salivary gland carcinomas: Comparison with methylation-specific PCR technique and clinical significance. Clin. Cancer Res. 2008, 14, 2664–2672. [Google Scholar]
- Aniello, F.; Colella, G.; Muscariello, G.; Lanza, A.; Ferrara, D.; Branno, M.; Minucci, S. Expression of four histone lysine-methyltransferases in parotid gland tumors. Anticancer. Res. 2006, 26, 2063–2067. [Google Scholar]
- Wagner, V.P.; Martins, M.D.; Guimaraes, D.M.; Vasconcelos, A.C.; Meurer, L.; Vargas, P.A.; Fonseca, F.P.; Squarize, C.H.; Castilho, R.M. Reduced chromatin acetylation of malignant salivary gland tumors correlates with enhanced proliferation. J. Oral Pathol. Med. 2017, 46, 792–797. [Google Scholar] [CrossRef] [PubMed]
- Mat Lazim, N.; Yousaf, A.; Abusalah, M.A.H.; Sulong, S.; Mohd Ismail, Z.I.; Mohamud, R.; Abu-Harirah, H.A.; AlRamadneh, T.N.; Hassan, R.; Abdullah, B. The Epigenesis of Salivary Glands Carcinoma: From Field Cancerization to Carcinogenesis. Cancers 2023, 15, 2111. [Google Scholar]
- Lam-Ubol, A.; Phattarataratip, E. Distinct histone H3 modification profiles correlate with aggressive characteristics of salivary gland neoplasms. Sci. Rep. 2022, 12, 15063. [Google Scholar] [CrossRef] [PubMed]
- Nandakumar, V.; Vaid, M.; Katiyar, S.K. (-)-Epigallocatechin-3-gallate reactivates silenced tumor suppressor genes, Cip1/p21 and p16INK4a, by reducing DNA methylation and increasing histones acetylation in human skin cancer cells. Carcinogenesis 2011, 32, 537–544. [Google Scholar] [CrossRef]
- Martin, C.; Chen, S.; Heilos, D.; Sauer, G.; Hunt, J.; Shaw, A.G.; Sims, P.F.; Jackson, D.A.; Lovric, J. Changed genome heterochromatinization upon prolonged activation of the Raf/ERK signaling pathway. PLoS ONE 2010, 5, e13322. [Google Scholar] [CrossRef]
- Mysliwiec, M.R.; Carlson, C.D.; Tietjen, J.; Hung, H.; Ansari, A.Z.; Lee, Y. Jarid2 (Jumonji, AT rich interactive domain 2) regulates NOTCH1 expression via histone modification in the developing heart. J. Biol. Chem. 2012, 287, 1235–1241. [Google Scholar] [CrossRef]
- Stephens, P.J.; Davies, H.R.; Mitani, Y.; Van Loo, P.; Shlien, A.; Tarpey, P.S.; Papaemmanuil, E.; Cheverton, A.; Bignell, G.R.; Butler, A.P.; et al. Whole exome sequencing of adenoid cystic carcinoma. J. Clin. Investig. 2013, 123, 2965–2968. [Google Scholar] [CrossRef]
- Lu, H.; Han, N.; Xu, W.; Zhu, Y.; Liu, L.; Liu, S.; Yang, W. Screening and bioinformatics analysis of mRNA, long non-coding RNA and circular RNA expression profiles in mucoepidermoid carcinoma of salivary gland. Biochem. Biophys. Res. Commun. 2019, 508, 66–71. [Google Scholar] [CrossRef]
- Chen, Z.; Lin, S.; Li, J.L.; Ni, W.; Guo, R.; Lu, J.; Kaye, F.J.; Wu, L. CRTC1-MAML2 fusion-induced lncRNA LINC00473 expression maintains the growth and survival of human mucoepidermoid carcinoma cells. Oncogene 2018, 37, 1885–1895. [Google Scholar] [CrossRef]
- Binmadi, N.O.; Basile, J.R.; Perez, P.; Gallo, A.; Tandon, M.; Elias, W.; Jang, S.I.; Alevizos, I. miRNA expression profile of mucoepidermoid carcinoma. Oral Dis. 2018, 24, 537–543. [Google Scholar] [CrossRef]
- Flores, B.C.; Lourenco, S.V.; Damascena, A.S.; Kowaslki, L.P.; Soares, F.A.; Coutinho-Camillo, C.M. Altered expression of apoptosis-regulating miRNAs in salivary gland tumors suggests their involvement in salivary gland tumorigenesis. Virchows. Arch. 2017, 470, 291–299. [Google Scholar] [CrossRef]
- Xu, W.; Liu, L.; Lu, H.; Fu, J.; Zhang, C.; Yang, W.; Shen, S. Dysregulated long non-coding RNAs in pleomorphic adenoma tissues of pleomorphic adenoma gene 1 transgenic mice. Mol. Med. Rep. 2019, 19, 4735. [Google Scholar] [CrossRef] [PubMed]
- Mitani, Y.; Roberts, D.B.; Fatani, H.; Weber, R.S.; Kies, M.S.; Lippman, S.M.; El-Naggar, A.K. MicroRNA profiling of salivary adenoid cystic carcinoma: Association of miR-17-92 upregulation with poor outcome. PLoS ONE 2013, 8, e66778. [Google Scholar]
- Ramírez-Martínez, C.M.; Jacinto-Alemán, L.F.; Cruz-Hervert, L.P.; Portilla-Robertson, J.; Leyva-Huerta, E.R. Bioinformatic Analysis for Mucoepidermoid and Adenoid Cystic Carcinoma of Therapeutic Targets. Vaccines 2022, 10, 1557. [Google Scholar] [CrossRef]
- Liu, J.; Shao, C.; Tan, M.L.; Mu, D.; Ferris, R.L.; Ha, P.K. Molecular biology of adenoid cystic carcinoma. Head Neck 2012, 34, 1665–1677. [Google Scholar] [CrossRef]
- Zhou, X.; Huang, S.; Zhang, D.; Zhang, S.; Li, W.; Chen, Z.; Wu, H. Effects of 5-aza-2′ deoxycytidine on RECK gene expression and tumor invasion in salivary adenoid cystic carcinoma. Braz. J. Med. Biol. Res. 2015, 48, 254. [Google Scholar] [CrossRef]
- Nikolic, N.; Carkic, J.; Jacimovic, J.; Jakovljevic, A.; Anicic, B.; Jezdic, Z.; Milasin, J. Methylation of tumour suppressor genes in benign and malignant salivary gland tumours: A systematic review and meta-analysis. Epigenetics. 2022, 17, 1661–1676. [Google Scholar] [CrossRef]
- Camuzi, D.; Simão, T.A.; Dias, F.; Ribeiro Pinto, L.F.; Soares-Lima, S.C. Head and Neck Cancers Are Not Alike When Tarred with the Same Brush: An Epigenetic Perspective from the Cancerization Field to Prognosis. Cancers. 2021, 13, 5630. [Google Scholar] [CrossRef]
- Huang, C.G.; Li, M.Z.; Wang, S.H.; Liu, Y.; Zhang, H.L.; Haybaeck, J.; Yang, Z.H. Analysis of Cytological Misdiagnosis and Oversight of Adenoid Cystic Carcinoma of Salivary Gland. Cancer Control. 2023, 30, 10732748221131652. [Google Scholar] [CrossRef] [PubMed]
- Nishida, H.; Kusaba, T.; Kawamura, K.; Oyama, Y.; Daa, T. Histopathological Aspects of the Prognostic Factors for Salivary Gland Cancers. Cancers. 2023, 15, 1236. [Google Scholar] [CrossRef]
- Kiwerska, K.; Kowal-Wisniewska, E.; Ustaszewski, A.; Bartkowiak, E.; Jarmuz-Szymczak, M.; Wierzbicka, M.; Giefing, M. Global DNA Methylation Profiling Reveals Differentially Methylated CpGs between Salivary Gland Pleomorphic Adenomas with Distinct Clinical Course. Int. J. Mol. Sci. 2022, 23, 5962. [Google Scholar] [CrossRef] [PubMed]
- Xia, C.; Tao, Y.; Li, M.; Che, T.; Qu, J. Protein acetylation and deacetylation: An important regulatory modification in gene transcription (Review). Exp. Ther. Med. 2020, 20, 2923–2940. [Google Scholar] [CrossRef] [PubMed]
- Meier, K.; Brehm, A. Chromatin regulation: How complex does it get? Epigenetics 2014, 9, 1485–1495. [Google Scholar] [CrossRef] [PubMed]
- Seto, E.; Yoshida, M. Erasers of histone acetylation: The histone deacetylase enzymes. Cold Spring Harb. Perspect. Biol. 2014, 6, a018713. [Google Scholar] [CrossRef]
- Ruijter, A.J.M.d.; Gennip, A.H.v.; Caron, H.N.; Kemp, S.; Kuilenburg, A.B.P.v. Histone deacetylases (HDACs): Characterization of the classical HDAC family. Biochem. J. 2003, 370, 737–749. [Google Scholar] [CrossRef]
- Haberland, M.; Montgomery, R.L.; Olson, E.N. The many roles of histone deacetylases in development and physiology: Implications for disease and therapy. Nat. Rev. Genet. 2009, 10, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Bruhn, C.; Bastianello, G.; Foiani, M. Cancer cell histone density links global histone acetylation, mitochondrial proteome and histone acetylase inhibitor sensitivity. Commun Biol. 2022, 5, 882. [Google Scholar] [CrossRef]
- Rettig, I.; Koeneke, E.; Trippel, F.; Mueller, W.C.; Burhenne, J.; Kopp-Schneider, A.; Fabian, J.; Schober, A.; Fernekorn, U.; von Deimling, A.; et al. Selective inhibition of HDAC8 decreases neuroblastoma growth in vitro and in vivo and enhances retinoic acid-mediated differentiation. Cell Death Dis. 2015, 6, e1657. [Google Scholar] [CrossRef]
- Cuttini, E.; Goi, C.; Pellarin, E.; Vida, R.; Brancolini, C. HDAC4 in cancer: A multitasking platform to drive not only epigenetic modifications. Front. Mol. Biosci. 2023, 10, 24. [Google Scholar] [CrossRef] [PubMed]
- Milde, T.; Oehme, I.; Korshunov, A.; Kopp-Schneider, A.; Remke, M.; Northcott, P.; Deubzer, H.E.; Lodrini, M.; Taylor, M.D.; Von Deimling, A. HDAC5 and HDAC9 in Medulloblastoma: Novel Markers for Risk Stratification and Role in Tumor Cell GrowthHDAC5 and HDAC9 in Medulloblastoma. Clin. Cancer Res. 2010, 16, 3240–3252. [Google Scholar] [CrossRef]
- Deskin, B.; Lasky, J.; Zhuang, Y.; Shan, B. Requirement of HDAC6 for activation of Notch1 by TGF-beta1. Sci. Rep. 2016, 6, 31086. [Google Scholar] [CrossRef]
- Yang, Y.; Huang, Y.; Wang, Z.; Wang, H.T.; Duan, B.; Ye, D.; Wang, C.; Jing, R.; Leng, Y.; Xi, J.; et al. HDAC10 promotes lung cancer proliferation via AKT phosphorylation. Oncotarget 2016, 7, 59388–59401. [Google Scholar] [CrossRef] [PubMed]
- Huffman, D.M.; Grizzle, W.E.; Bamman, M.M.; Kim, J.S.; Eltoum, I.A.; Elgavish, A.; Nagy, T.R. SIRT1 is significantly elevated in mouse and human prostate cancer. Cancer Res. 2007, 67, 6612–6618. [Google Scholar] [CrossRef]
- Bradbury, C.A.; Khanim, F.L.; Hayden, R.; Bunce, C.M.; White, D.A.; Drayson, M.T.; Craddock, C.; Turner, B.M. Histone deacetylases in acute myeloid leukaemia show a distinctive pattern of expression that changes selectively in response to deacetylase inhibitors. Leukemia 2005, 19, 1751–1759. [Google Scholar] [CrossRef]
- Singh, A.K.; Bishayee, A.; Pandey, A.K. Targeting Histone Deacetylases with Natural and Synthetic Agents: An Emerging Anticancer Strategy. Nutrients 2018, 10, 731. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, N.; Zino, S.; Macintyre, A.; Kingsmore, D.; Payne, A.P.; George, W.D.; Shiels, P.G. Altered sirtuin expression is associated with node-positive breast cancer. Br. J. Cancer 2006, 95, 1056–1061. [Google Scholar] [CrossRef]
- de Ceu Teixeira, M.; Sanchez-Lopez, E.; Espina, M.; Garcia, M.L.; Durazzo, A.; Lucarini, M.; Novellino, E.; Souto, S.B.; Santini, A.; Souto, E.B. Sirtuins and SIRT6 in Carcinogenesis and in Diet. Int. J. Mol. Sci. 2019, 20, 4945. [Google Scholar] [CrossRef]
- Chang, H.H.; Chiang, C.P.; Hung, H.C.; Lin, C.Y.; Deng, Y.T.; Kuo, M.Y. Histone deacetylase 2 expression predicts poorer prognosis in oral cancer patients. Oral Oncol. 2009, 45, 610–614. [Google Scholar] [CrossRef]
- Wawruszak, A.; Kalafut, J.; Okon, E.; Czapinski, J.; Halasa, M.; Przybyszewska, A.; Miziak, P.; Okla, K.; Rivero-Muller, A.; Stepulak, A. Histone Deacetylase Inhibitors and Phenotypical Transformation of Cancer Cells. Cancers 2019, 11, 148. [Google Scholar] [CrossRef] [PubMed]
- Chau, N.G.; Hurwitz, S.; Mitchell, C.M.; Aserlind, A.; Grunfeld, N.; Kaplan, L.; Hsi, P.; Bauer, D.E.; Lathan, C.S.; Rodriguez-Galindo, C.; et al. Intensive treatment and survival outcomes in NUT midline carcinoma of the head and neck. Cancer 2016, 122, 3632–3640. [Google Scholar] [CrossRef] [PubMed]
- Esteves, G.; Ferreira, J.; Afonso, R.; Martins, C.; Zagalo, C.; Felix, A. HDAC Overexpression in a NUT Midline Carcinoma of the Parotid Gland with Exceptional Survival: A Case Report. Head Neck Pathol. 2020, 14, 1117–1122. [Google Scholar] [CrossRef] [PubMed]
- Pouloudi, D.; Manou, M.; Sarantis, P.; Tsoukalas, N.; Tsourouflis, G.; Dana, E.; Karamouzis, M.V.; Klijanienko, J.; Theocharis, S. Clinical Significance of Histone Deacetylase (HDAC)-1, -2, -4 and -6 Expression in Salivary Gland Tumors. Diagnostics 2021, 11, 517. [Google Scholar] [CrossRef]
- Zhao, S.; Guo, J.; Zhao, Y.; Fei, C.; Zheng, Q.; Li, X.; Chang, C. Chidamide, a novel histone deacetylase inhibitor, inhibits the viability of MDS and AML cells by suppressing JAK2/STAT3 signaling. Am. J. Transl. Res. 2016, 8, 3169–3178. [Google Scholar]
- Dong, M.; Ning, Z.Q.; Xing, P.Y.; Xu, J.L.; Cao, H.X.; Dou, G.F.; Meng, Z.Y.; Shi, Y.K.; Lu, X.P.; Feng, F.Y. Phase I study of chidamide (CS055/HBI-8000), a new histone deacetylase inhibitor, in patients with advanced solid tumors and lymphomas. Cancer Chemother. Pharmacol. 2012, 69, 1413–1422. [Google Scholar] [CrossRef]
- Yang, S.; Nan, P.; Li, C.; Lin, F.; Li, H.; Wang, T.; Zhou, C.; Zhang, X.; Meng, X.; Qian, H.; et al. Inhibitory effect of chidamide on the growth of human adenoid cystic carcinoma cells. Biomed. Pharmacother. 2018, 99, 608–614. [Google Scholar] [CrossRef]
- Tsimberidou, A.M.; Beer, P.A.; Cartwright, C.A.; Haymaker, C.; Vo, H.H.; Kiany, S.; Cecil, A.R.L.; Dow, J.; Haque, K.; Silva, F.A.; et al. Preclinical Development and First-in-Human Study of KA2507, a Selective and Potent Inhibitor of Histone Deacetylase 6, for Patients with Refractory Solid Tumors. Clin. Cancer Res. 2021, 27, 3584–3594. [Google Scholar] [CrossRef]
- Ho, A.S.; Kannan, K.; Roy, D.M.; Morris, L.G.; Ganly, I.; Katabi, N.; Ramaswami, D.; Walsh, L.A.; Eng, S.; Huse, J.T.; et al. The mutational landscape of adenoid cystic carcinoma. Nat. Genet. 2013, 45, 791–798. [Google Scholar] [CrossRef]
- Ramalingam, S.S.; Kummar, S.; Sarantopoulos, J.; Shibata, S.; LoRusso, P.; Yerk, M.; Holleran, J.; Lin, Y.; Beumer, J.H.; Harvey, R.D.; et al. Phase I study of vorinostat in patients with advanced solid tumors and hepatic dysfunction: A National Cancer Institute Organ Dysfunction Working Group study. J. Clin. Oncol. 2010, 28, 4507–4512. [Google Scholar] [CrossRef]
- Goncalves, P.H.; Heilbrun, L.K.; Barrett, M.T.; Kummar, S.; Hansen, A.R.; Siu, L.L.; Piekarz, R.L.; Sukari, A.W.; Chao, J.; Pilat, M.J.; et al. A phase 2 study of vorinostat in locally advanced, recurrent, or metastatic adenoid cystic carcinoma. Oncotarget 2017, 8, 32918–32929. [Google Scholar] [CrossRef]
- Woods, D.M.; Sodré, A.L.; Villagra, A.; Sarnaik, A.; Sotomayor, E.M.; Weber, J. HDAC Inhibition Upregulates PD-1 Ligands in Melanoma and Augments Immunotherapy with PD-1 BlockadeHDAC Inhibition Upregulates PD-1 Ligands in Melanoma. Cancer Immunol. Res. 2015, 3, 1375–1385. [Google Scholar] [CrossRef] [PubMed]
- Hellmann, M.D.; Janne, P.A.; Opyrchal, M.; Hafez, N.; Raez, L.E.; Gabrilovich, D.I.; Wang, F.; Trepel, J.B.; Lee, M.J.; Yuno, A.; et al. Entinostat plus Pembrolizumab in Patients with Metastatic NSCLC Previously Treated with Anti-PD-(L)1 Therapy. Clin. Cancer Res. 2021, 27, 1019–1028. [Google Scholar] [CrossRef] [PubMed]
- Cohen, R.B.; Delord, J.P.; Doi, T.; Piha-Paul, S.A.; Liu, S.V.; Gilbert, J.; Algazi, A.P.; Damian, S.; Hong, R.L.; Le Tourneau, C.; et al. Pembrolizumab for the Treatment of Advanced Salivary Gland Carcinoma: Findings of the Phase 1b KEYNOTE-028 Study. Am. J. Clin. Oncol. 2018, 41, 1083–1088. [Google Scholar] [CrossRef]
- Goncalves, P.H.; Kummar, S.; Siu, L.L.; Hansen, A.R.; Savvides, P.; Sukari, A.; Chao, J.; Heilbrun, L.K.; Pilat, M.J.; Smith, D.W.; et al. A phase II study of suberoylanilide hydroxamic acid (SAHA) in subjects with locally advanced, recurrent, or metastatic adenoid cystic carcinoma (ACC). J. Clin. Oncol. 2013, 31, 6045. [Google Scholar] [CrossRef]
- Rodriguez, C.P.; Wu, Q.V.; Voutsinas, J.; Fromm, J.R.; Jiang, X.; Pillarisetty, V.G.; Lee, S.M.; Santana-Davila, R.; Goulart, B.; Baik, C.S.; et al. A Phase II Trial of Pembrolizumab and Vorinostat in Recurrent Metastatic Head and Neck Squamous Cell Carcinomas and Salivary Gland Cancer. Clin. Cancer Res. 2020, 26, 837–845. [Google Scholar] [CrossRef]
- Chang, K.; Creighton, C.J.; Davis, C.; Donehower, L.; Drummond, J.; Wheeler, D.; Ally, A.; Balasundaram, M.; Birol, I.; Butterfield, Y.S.N.; et al. The Cancer Genome Atlas Pan-Cancer analysis project. Nat. Genet. 2013, 45, 1113–1120. [Google Scholar] [CrossRef]
- Edgar, R.; Domrachev, M.; Lash, A.E. Gene Expression Omnibus: NCBI gene expression and hybridization array data repository. Nucleic Acids Res. 2002, 30, 207–210. [Google Scholar] [CrossRef]
- Tang, F.; Barbacioru, C.; Wang, Y.; Nordman, E.; Lee, C.; Xu, N.; Wang, X.; Bodeau, J.; Tuch, B.B.; Siddiqui, A.; et al. mRNA-Seq whole-transcriptome analysis of a single cell. Nat. Methods 2009, 6, 377–382. [Google Scholar] [CrossRef]
- Wang, Z.; Gerstein, M.; Snyder, M. RNA-Seq: A revolutionary tool for transcriptomics. Nat. Rev. Genet. 2009, 10, 57–63. [Google Scholar] [CrossRef]
- Langfelder, P.; Horvath, S. WGCNA: An R package for weighted correlation network analysis. BMC Bioinform. 2008, 9, 559. [Google Scholar] [CrossRef]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 2009, 4, 44–57. [Google Scholar] [CrossRef]
- Szklarczyk, D.; Kirsch, R.; Koutrouli, M.; Nastou, K.; Mehryary, F.; Hachilif, R.; Gable, A.L.; Fang, T.; Doncheva, N.T.; Pyysalo, S.; et al. The STRING database in 2023: Protein-protein association networks and functional enrichment analyses for any sequenced genome of interest. Nucleic Acids Res. 2023, 51, D638–D646. [Google Scholar] [CrossRef] [PubMed]
- Kourou, K.; Exarchos, T.P.; Exarchos, K.P.; Karamouzis, M.V.; Fotiadis, D.I. Machine learning applications in cancer prognosis and prediction. Comput. Struct. Biotechnol. J. 2015, 13, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Hasin, Y.; Seldin, M.; Lusis, A. Multi-omics approaches to disease. Genome Biol. 2017, 18, 83. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.H.; Kim, D.W.; Lee, S.K.; Kim, S.G. Effects of 4-hexylresorcinol administration on the submandibular glands in a growing rat model. Head Face Med. 2022, 18, 16. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-Y.; Kweon, H.-Y.; Kim, D.-W.; Choi, J.-Y.; Kim, S.-G. 4-Hexylresorcinol Inhibits Class I Histone Deacetylases in Human Umbilical Cord Endothelial Cells. Appl. Sci. 2021, 11, 3486. [Google Scholar] [CrossRef]
- Qian, D.Z.; Kato, Y.; Shabbeer, S.; Wei, Y.; Verheul, H.M.; Salumbides, B.; Sanni, T.; Atadja, P.; Pili, R. Targeting tumor angiogenesis with histone deacetylase inhibitors: The hydroxamic acid derivative LBH589. Clin. Cancer Res. 2006, 12, 634–642. [Google Scholar] [CrossRef] [PubMed]
- Frohlich, L.F.; Mrakovcic, M.; Smole, C.; Zatloukal, K. Molecular mechanism leading to SAHA-induced autophagy in tumor cells: Evidence for a p53-dependent pathway. Cancer Cell Int. 2016, 16, 68. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.C.; Huang, H.H.; Lai, C.Y.; Lin, Y.J.; Liou, J.P.; Lai, M.J.; Li, Y.H.; Teng, C.M.; Yang, C.R. Novel histone deacetylase inhibitor MPT0G009 induces cell apoptosis and synergistic anticancer activity with tumor necrosis factor-related apoptosis-inducing ligand against human hepatocellular carcinoma. Oncotarget 2016, 7, 402–417. [Google Scholar] [CrossRef]
- Li, Y.; Liu, T.; Ivan, C.; Huang, J.; Shen, D.Y.; Kavanagh, J.J.; Bast, R.C., Jr.; Fu, S.; Hu, W.; Sood, A.K. Enhanced Cytotoxic Effects of Combined Valproic Acid and the Aurora Kinase Inhibitor VE465 on Gynecologic Cancer Cells. Front. Oncol. 2013, 3, 58. [Google Scholar] [CrossRef]
- Wu, P.; Tian, Y.; Chen, G.; Wang, B.; Gui, L.; Xi, L.; Ma, X.; Fang, Y.; Zhu, T.; Wang, D.; et al. Correction: Ubiquitin B: An essential mediator of trichostatin A-induced tumor-selective killing in human cancer cells. Cell Death Differ. 2022, 29, 1299. [Google Scholar] [CrossRef]
- Ramaiah, M.J.; Tangutur, A.D.; Manyam, R.R. Epigenetic modulation and understanding of HDAC inhibitors in cancer therapy. Life Sci. 2021, 277, 119504. [Google Scholar] [CrossRef]
- Kauh, J.; Fan, S.; Xia, M.; Yue, P.; Yang, L.; Khuri, F.R.; Sun, S.Y. c-FLIP degradation mediates sensitization of pancreatic cancer cells to TRAIL-induced apoptosis by the histone deacetylase inhibitor LBH589. PLoS ONE 2010, 5, e10376. [Google Scholar] [CrossRef]
- Liu, J.; Li, G.; Wang, X.; Wang, L.; Zhao, R.; Wang, J.; Kong, Y.; Ding, J.; Li, J.; Zhang, L. Droxinostat, a Histone Deacetylase Inhibitor, Induces Apoptosis in Hepatocellular Carcinoma Cell Lines via Activation of the Mitochondrial Pathway and Downregulation of FLIP. Transl. Oncol. 2016, 9, 70–78. [Google Scholar] [CrossRef]
- Chintakuntlawar, A.V.; Okuno, S.H.; Price, K.A. Systemic therapy for recurrent or metastatic salivary gland malignancies. Cancers Head Neck 2016, 1, 11. [Google Scholar] [CrossRef] [PubMed]
- Ahn, M.Y.; Ahn, J.W.; Kim, H.S.; Lee, J.; Yoon, J.H. Apicidin inhibits cell growth by downregulating IGF-1R in salivary mucoepidermoid carcinoma cells. Oncol. Rep. 2015, 33, 1899–1907. [Google Scholar] [CrossRef] [PubMed]
- Ahn, M.Y.; Yoon, J.H. Histone deacetylase 7 silencing induces apoptosis and autophagy in salivary mucoepidermoid carcinoma cells. J. Oral Pathol. Med. 2017, 46, 276–283. [Google Scholar] [CrossRef]
- Guimarães, D.M.; Almeida, L.O.; Martins, M.D.; Warner, K.A.; Silva, A.R.; Vargas, P.A.; Nunes, F.D.; Squarize, C.H.; Nör, J.E.; Castilho, R.M. Sensitizing mucoepidermoid carcinomas to chemotherapy by targeted disruption of cancer stem cells. Oncotarget 2016, 7, 42447. [Google Scholar] [CrossRef]
- Almeida, L.O.; Guimaraes, D.M.; Martins, M.D.; Martins, M.A.T.; Warner, K.A.; Nor, J.E.; Castilho, R.M.; Squarize, C.H. Unlocking the chromatin of adenoid cystic carcinomas using HDAC inhibitors sensitize cancer stem cells to cisplatin and induces tumor senescence. Stem. Cell Res. 2017, 21, 94–105. [Google Scholar] [CrossRef]
- Parag-Sharma, K.; Tasoulas, J.; Musicant, A.M.; do Nascimento-Filho, C.H.V.; Zhu, Z.; Twomey, C.; Liu, P.; Castilho, R.M.; Amelio, A.L. Synergistic efficacy of combined EGFR and HDAC inhibitors overcomes tolerance to EGFR monotherapy in salivary mucoepidermoid carcinoma. Oral Oncol. 2021, 115, 105166. [Google Scholar] [CrossRef] [PubMed]
- Jang, B.; Yang, I.H.; Cho, N.P.; Jin, B.; Lee, W.; Jung, Y.C.; Hong, S.D.; Shin, J.A.; Cho, S.D. Down-regulation and nuclear localization of survivin by sodium butyrate induces caspase-dependent apoptosis in human oral mucoepidermoid carcinoma. Oral Oncol. 2019, 88, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Nakano, M.; Ohwada, K.; Shindo, Y.; Konno, T.; Kohno, T.; Kikuchi, S.; Tsujiwaki, M.; Ishii, D.; Nishida, S.; Kakuki, T.; et al. Inhibition of HDAC and Signal Transduction Pathways Induces Tight Junctions and Promotes Differentiation in p63-Positive Salivary Duct Adenocarcinoma. Cancers 2022, 14, 2584. [Google Scholar] [CrossRef]
- Talevi, A. Multi-target pharmacology: Possibilities and limitations of the “skeleton key approach” from a medicinal chemist perspective. Front. Pharmacol. 2015, 6, 205. [Google Scholar] [CrossRef]
- Lai, C.J.; Bao, R.; Tao, X.; Wang, J.; Atoyan, R.; Qu, H.; Wang, D.G.; Yin, L.; Samson, M.; Forrester, J.; et al. CUDC-101, a multitargeted inhibitor of histone deacetylase, epidermal growth factor receptor, and human epidermal growth factor receptor 2, exerts potent anticancer activity. Cancer Res. 2010, 70, 3647–3656. [Google Scholar] [CrossRef]
- Wang, J.; Pursell, N.W.; Samson, M.E.; Atoyan, R.; Ma, A.W.; Selmi, A.; Xu, W.; Cai, X.; Voi, M.; Savagner, P.; et al. Potential advantages of CUDC-101, a multitargeted HDAC, EGFR, and HER2 inhibitor, in treating drug resistance and preventing cancer cell migration and invasion. Mol. Cancer Ther. 2013, 12, 925–936. [Google Scholar] [CrossRef]
- Galloway, T.J.; Wirth, L.J.; Colevas, A.D.; Gilbert, J.; Bauman, J.E.; Saba, N.F.; Raben, D.; Mehra, R.; Ma, A.W.; Atoyan, R.; et al. A Phase I Study of CUDC-101, a Multitarget Inhibitor of HDACs, EGFR, and HER2, in Combination with Chemoradiation in Patients with Head and Neck Squamous Cell Carcinoma. Clin. Cancer Res. 2015, 21, 1566–1573. [Google Scholar] [CrossRef]
- Parra, M. Class IIa HDACs—New insights into their functions in physiology and pathology. FEBS J. 2015, 282, 1736–1744. [Google Scholar] [CrossRef]
- Yu, Y.; Cao, F.; Yu, X.; Zhou, P.; Di, Q.; Lei, J.; Tai, Y.; Wu, H.; Li, X.; Wang, X.; et al. The expression of HDAC7 in cancerous gastric tissues is positively associated with distant metastasis and poor patient prognosis. Clin. Transl. Oncol. 2017, 19, 1045–1054. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.Y.; Fu, J.; Xiao, X.; Wu, J.; Wu, R.C. MiR-34a regulates therapy resistance by targeting HDAC1 and HDAC7 in breast cancer. Cancer Lett. 2014, 354, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Barneda-Zahonero, B.; Collazo, O.; Azagra, A.; Fernandez-Duran, I.; Serra-Musach, J.; Islam, A.B.; Vega-Garcia, N.; Malatesta, R.; Camos, M.; Gomez, A.; et al. The transcriptional repressor HDAC7 promotes apoptosis and c-Myc downregulation in particular types of leukemia and lymphoma. Cell Death Dis. 2015, 6, e1635. [Google Scholar] [CrossRef]
- Yu, X.; Wang, M.; Wu, J.; Han, Q.; Zhang, X. Correction to: ZNF326 promotes malignant phenotype of glioma by up-regulating HDAC7 expression and activating Wnt pathway. J. Exp. Clin. Cancer Res. 2020, 39, 14. [Google Scholar] [CrossRef]
- Sang, Y.; Sun, L.; Wu, Y.; Yuan, W.; Liu, Y.; Li, S.W. Histone deacetylase 7 inhibits plakoglobin expression to promote lung cancer cell growth and metastasis. Int. J. Oncol. 2019, 54, 1112–1122. [Google Scholar] [CrossRef] [PubMed]
- Witt, A.E.; Lee, C.W.; Lee, T.I.; Azzam, D.J.; Wang, B.; Caslini, C.; Petrocca, F.; Grosso, J.; Jones, M.; Cohick, E.B.; et al. Identification of a cancer stem cell-specific function for the histone deacetylases, HDAC1 and HDAC7, in breast and ovarian cancer. Oncogene 2017, 36, 1707–1720. [Google Scholar] [CrossRef]
- Li, Q.G.; Xiao, T.; Zhu, W.; Yu, Z.Z.; Huang, X.P.; Yi, H.; Lu, S.S.; Tang, Y.Y.; Huang, W.; Xiao, Z.Q. HDAC7 promotes the oncogenicity of nasopharyngeal carcinoma cells by miR-4465-EphA2 signaling axis. Cell Death Dis. 2020, 11, 322. [Google Scholar] [CrossRef]
- Li, S.; Wang, B.; Xu, Y.; Zhang, J. Autotaxin is induced by TSA through HDAC3 and HDAC7 inhibition and antagonizes the TSA-induced cell apoptosis. Mol. Cancer 2011, 10, 18. [Google Scholar] [CrossRef] [PubMed]
- Kwiecinska, P.; Wrobel, A.; Tauboll, E.; Gregoraszczuk, E.L. Valproic acid, but not levetiracetam, selectively decreases HDAC7 and HDAC2 expression in human ovarian cancer cells. Toxicol. Lett. 2014, 224, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Dokmanovic, M.; Perez, G.; Xu, W.; Ngo, L.; Clarke, C.; Parmigiani, R.B.; Marks, P.A. Histone deacetylase inhibitors selectively suppress expression of HDAC7. Mol. Cancer Ther. 2007, 6, 2525–2534. [Google Scholar] [CrossRef] [PubMed]
- Stenner, M.; Weinell, A.; Ponert, T.; Hardt, A.; Hahn, M.; Preuss, S.F.; Guntinas-Lichius, O.; Klussmann, J.P. Cytoplasmic expression of survivin is an independent predictor of poor prognosis in patients with salivary gland cancer. Histopathology 2010, 57, 699–706. [Google Scholar] [CrossRef]
- Wheatley, S.P.; Altieri, D.C. Survivin at a glance. J. Cell Sci. 2019, 132, jcs223826. [Google Scholar] [CrossRef]
- Stauber, R.H.; Mann, W.; Knauer, S.K. Nuclear and cytoplasmic survivin: Molecular mechanism, prognostic, and therapeutic potential. Cancer Res. 2007, 67, 5999–6002. [Google Scholar] [CrossRef]
- Lippert, B.M.; Knauer, S.K.; Fetz, V.; Mann, W.; Stauber, R.H. Dynamic survivin in head and neck cancer: Molecular mechanism and therapeutic potential. Int. J. Cancer 2007, 121, 1169–1174. [Google Scholar] [CrossRef]
- Wang, H.; Holloway, M.P.; Ma, L.; Cooper, Z.A.; Riolo, M.; Samkari, A.; Elenitoba-Johnson, K.S.; Chin, Y.E.; Altura, R.A. Acetylation directs survivin nuclear localization to repress STAT3 oncogenic activity. J. Biol. Chem. 2010, 285, 36129–36137. [Google Scholar] [CrossRef]
- Sen, N.; Che, X.; Rajamani, J.; Zerboni, L.; Sung, P.; Ptacek, J.; Arvin, A.M. Signal transducer and activator of transcription 3 (STAT3) and survivin induction by varicella-zoster virus promote replication and skin pathogenesis. Proc. Natl. Acad. Sci. USA 2012, 109, 600–605. [Google Scholar] [CrossRef]
- Hu, X.; Wang, L.; Lin, L.; Han, X.; Dou, G.; Meng, Z.; Shi, Y. A phase I trial of an oral subtype-selective histone deacetylase inhibitor, chidamide, in combination with paclitaxel and carboplatin in patients with advanced non-small cell lung cancer. Chin. J. Cancer Res. 2016, 28, 444–451. [Google Scholar] [CrossRef]
- Shi, Y.; Jia, B.; Xu, W.; Li, W.; Liu, T.; Liu, P.; Zhao, W.; Zhang, H.; Sun, X.; Yang, H.; et al. Chidamide in relapsed or refractory peripheral T cell lymphoma: A multicenter real-world study in China. J. Hematol. Oncol. 2017, 10, 69. [Google Scholar] [CrossRef]
- Gong, K.; Xie, J.; Yi, H.; Li, W. CS055 (Chidamide/HBI-8000), a novel histone deacetylase inhibitor, induces G1 arrest, ROS-dependent apoptosis and differentiation in human leukaemia cells. Biochem. J. 2012, 443, 735–746. [Google Scholar] [CrossRef]
- Zhou, Y.; Pan, D.S.; Shan, S.; Zhu, J.Z.; Zhang, K.; Yue, X.P.; Nie, L.P.; Wan, J.; Lu, X.P.; Zhang, W.; et al. Non-toxic dose chidamide synergistically enhances platinum-induced DNA damage responses and apoptosis in Non-Small-Cell lung cancer cells. Biomed. Pharmacother. 2014, 68, 483–491. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Dong, M.; Hong, X.; Zhang, W.; Feng, J.; Zhu, J.; Yu, L.; Ke, X.; Huang, H.; Shen, Z.; et al. Results from a multicenter, open-label, pivotal phase II study of chidamide in relapsed or refractory peripheral T-cell lymphoma. Ann. Oncol. 2015, 26, 1766–1771. [Google Scholar] [CrossRef] [PubMed]
- Koster, M.I.; Kim, S.; Mills, A.A.; DeMayo, F.J.; Roop, D.R. p63 is the molecular switch for initiation of an epithelial stratification program. Genes Dev. 2004, 18, 126–131. [Google Scholar] [CrossRef]
- Steurer, S.; Riemann, C.; Buscheck, F.; Luebke, A.M.; Kluth, M.; Hube-Magg, C.; Hinsch, A.; Hoflmayer, D.; Weidemann, S.; Fraune, C.; et al. p63 expression in human tumors and normal tissues: A tissue microarray study on 10,200 tumors. Biomark. Res. 2021, 9, 7. [Google Scholar] [CrossRef]
- Sivakumar, N.; Narwal, A.; Pandiar, D.; Devi, A.; Anand, R.; Bansal, D.; Kamboj, M. Diagnostic utility of p63/p40 in the histologic differentiation of salivary gland tumors: A systematic review. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2022, 133, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Dokmanovic, M.; Clarke, C.; Marks, P.A. Histone deacetylase inhibitors: Overview and perspectives. Mol. Cancer Res. 2007, 5, 981–989. [Google Scholar] [CrossRef] [PubMed]
- Boyault, C.; Sadoul, K.; Pabion, M.; Khochbin, S. HDAC6, at the crossroads between cytoskeleton and cell signaling by acetylation and ubiquitination. Oncogene 2007, 26, 5468–5476. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.S.; Lim, K.H.; Guo, X.; Kawaguchi, Y.; Gao, Y.; Barrientos, T.; Ordentlich, P.; Wang, X.F.; Counter, C.M.; Yao, T.P. The cytoplasmic deacetylase HDAC6 is required for efficient oncogenic tumorigenesis. Cancer Res. 2008, 68, 7561–7569. [Google Scholar] [CrossRef]
- Li, T.; Zhang, C.; Hassan, S.; Liu, X.; Song, F.; Chen, K.; Zhang, W.; Yang, J. Histone deacetylase 6 in cancer. J. Hematol. Oncol. 2018, 11, 111. [Google Scholar] [CrossRef] [PubMed]
- Schnekenburger, M.; Florean, C.; Dicato, M.; Diederich, M. Epigenetic alterations as a universal feature of cancer hallmarks and a promising target for personalized treatments. Curr. Top. Med. Chem. 2016, 16, 745–776. [Google Scholar] [CrossRef] [PubMed]
- Losson, H.; Schnekenburger, M.; Dicato, M.; Diederich, M. HDAC6-an Emerging Target Against Chronic Myeloid Leukemia? Cancers 2020, 12, 318. [Google Scholar] [CrossRef]
- Coca-Pelaz, A.; Rodrigo, J.P.; Bradley, P.J.; Vander Poorten, V.; Triantafyllou, A.; Hunt, J.L.; Strojan, P.; Rinaldo, A.; Haigentz, M., Jr.; Takes, R.P.; et al. Adenoid cystic carcinoma of the head and neck--An update. Oral Oncol. 2015, 51, 652–661. [Google Scholar] [CrossRef] [PubMed]
- Ellington, C.L.; Goodman, M.; Kono, S.A.; Grist, W.; Wadsworth, T.; Chen, A.Y.; Owonikoko, T.; Ramalingam, S.; Shin, D.M.; Khuri, F.R.; et al. Adenoid cystic carcinoma of the head and neck: Incidence and survival trends based on 1973-2007 Surveillance, Epidemiology, and End Results data. Cancer 2012, 118, 4444–4451. [Google Scholar] [CrossRef] [PubMed]
- Uijen, M.J.M.; Lassche, G.; van Engen-van Grunsven, A.C.H.; Driessen, C.M.L.; van Herpen, C.M.L. Case series of docetaxel, trastuzumab, and pertuzumab (DTP) as first line anti-HER2 therapy and ado-trastuzumab emtansine (T-DM1) as second line for recurrent or metastatic HER2-positive salivary duct carcinoma. Oral Oncol. 2022, 125, 105703. [Google Scholar] [CrossRef]
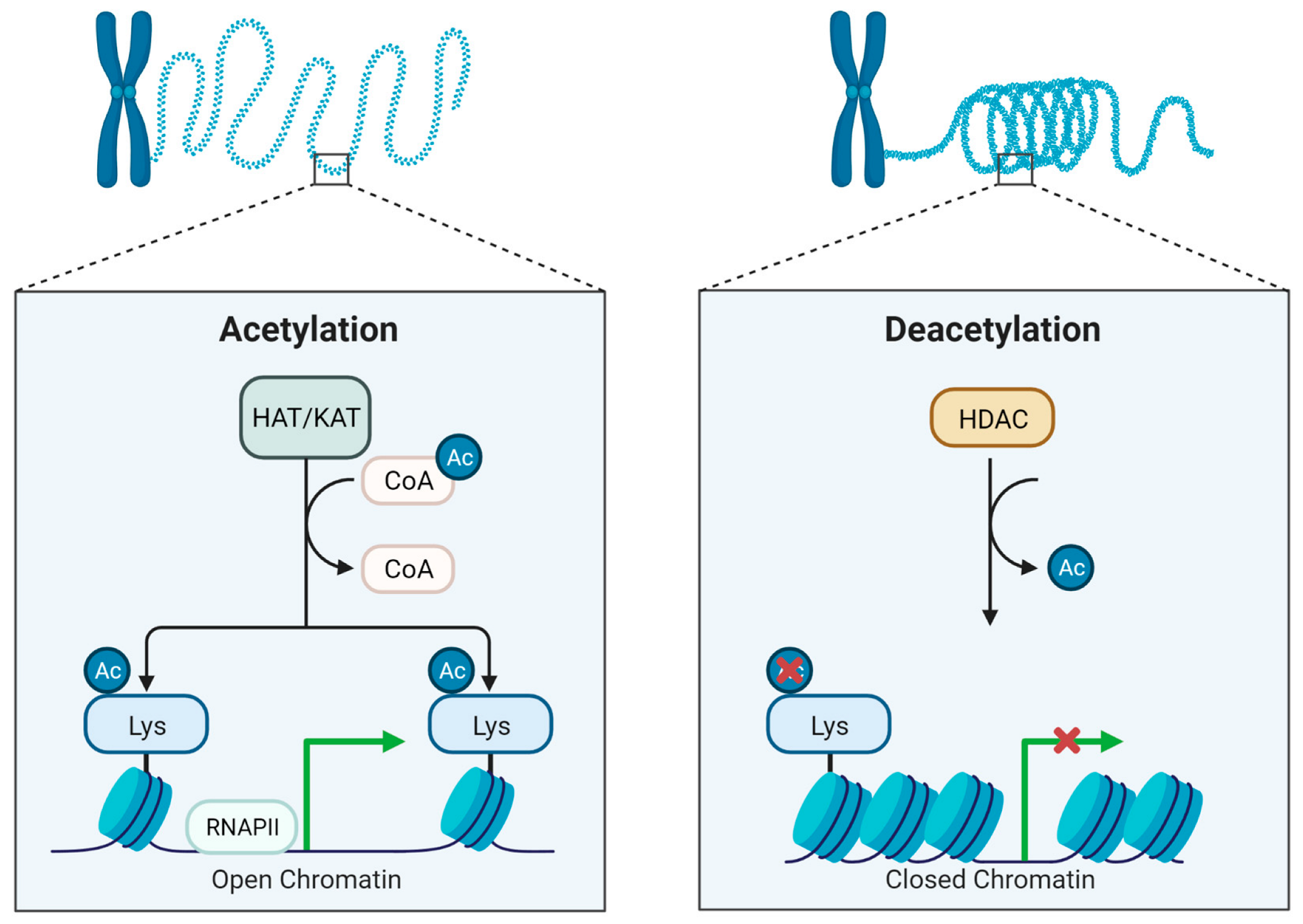
| Salivary Carcinomas Not Otherwise Specified (NOS) and Emerging Entities | Malignant Epithelial Tumors |
|---|---|
| Large-cell neuroendocrine carcinoma | Acinic cell carcinoma |
| Lymphoepithelial carcinoma | Adenoid cystic carcinoma |
| Oncocytic carcinoma | Basal cell adenocarcinoma |
| Small-cell neuroendocrine carcinoma | Carcinoma ex pleomorphic adenoma |
| Squamous cell carcinoma | Carcinosarcoma |
| Undifferentiated carcinoma | Clear cell carcinoma |
| Benign Epithelial Tumors | Epithelial–myoepithelial carcinoma |
| Basal cell adenoma | Intraductal carcinoma |
| Cystadenoma | Microsecretory adenocarcinoma * |
| Ductal papillomas | Mucinous adenocarcinoma * |
| Intercalated duct adenoma * | Mucoepidermoid carcinoma |
| Keratocystoma * | Myoepithelial carcinoma |
| Lymphadenoma | Polymorphous adenocarcinoma |
| Myoepithelioma | Salivary duct carcinoma |
| Oncocytoma | Sclerosing microcystic adenocarcinoma * |
| Pleomorphic adenoma | Sebaceous adenocarcinoma |
| Sclerosing polycystic adenoma * | Secretory carcinoma |
| Sialadenoma papilliferum | Uncertain Malignant Potentiality |
| Striated duct adenoma * | Sialoblastoma |
| Warthin tumor |
| HDAC Class | Subtypes | Cellular Location | Substrate Specificity | Functions and Targets |
|---|---|---|---|---|
| Class-I | HDAC1, HDAC2, HDAC3, HDAC8 | Nucleus | All four core histones (HDAC1,2); H3, H4 (HDAC8) | Gene repression; Cell-cycle regulation; Apoptosis; DNA repair [93] |
| Class-IIa | HDAC4, HDAC5, HDAC7, HDAC9 | Nucleus and Cytoplasm | All four core histones (HDAC4,5) | Cell differentiation; Apoptosis; Angiogenesis; Response to external signals [94] |
| Class-IIb | HDAC6, HDAC10 | Cytoplasm (HDAC6); Nucleus (HDAC10) | H3K9, H3K56, α-tubulin (HDAC6); Polyamine catabolism (HDAC10) | Regulation of microtubule stability; Aggresome formation [94] |
| Class-III | SIRT1, SIRT2 | Nucleus and Cytoplasm | H4K16, H3K9 (SIRT1); H4K16, H3K56 (SIRT2) | Aging; Metabolism; DNA repair; Stress resistance [93] |
| SIRT3, SIRT4, SIRT5 | Mitochondria | H4K16 (SIRT3); Mitochondrial metabolism (SIRT4,5) | Energy homeostasis; Oxidative stress response [93] | |
| SIRT6, SIRT7 | Nucleus (SIRT6); Nucleolus (SIRT7) | H3K9, H3K56 (SIRT6); H3K18 (SIRT7) | Genome stability; Ribosome biogenesis [93] | |
| Class-IV | HDAC11 | Nucleus and Cytoplasm | H3K9, H3K14 | DNA replication regulation; Immune regulation [93] |
| Categorization | Study Type/Method | HDAC Type | Expression Rate | Reference |
|---|---|---|---|---|
| HN malignancies | Tissue sections (FFPE)/IHC | HDAC2, -9, -10 | ~100% | [107] |
| SIRT3, -5, -6, -7 | ~100% | [107] | ||
| HDAC1, SIRT1 | ~75% | [107] | ||
| SGTs | Tissue sections (FFPE)/IHC | HDAC1 | Benign SGTs: 30% | [110] |
| Malignant SGTs: 14% | [110] | |||
| Tissue sections (FFPE)/IHC | HDAC2 | Benign SGTs: 86% | [110] | |
| Malignant SGTs: 82% | [110] | |||
| Tissue sections (FFPE)/IHC | HDAC4 | Benign SGTs: 44% | [110] | |
| Malignant SGTs: 36% | [110] | |||
| Tissue sections (FFPE)/IHC | HDAC6 | Benign SGTs: 11% | [110] | |
| Malignant SGTs: 18% | [110] |
| Type of Salivary Gland Malignancy | Substance | Main Outcome | Reference |
|---|---|---|---|
| MEC (cell line) | Apicidin (HDACi) | ↓ proliferative potential | [143,144] |
| MEC (cell line) | Vorinostat (HDACi) | ↓ cancer stem cell population | [145] |
| Vorinostat (HDACi) + cisplatin | ↑ sensitivity to cisplatin | ||
| AdCC (cell line) Mice transplanted with tumor cells | Vorinostat (HDACi) | ↓ population of cancer cells and stem cells | [146] |
| Vorinostat (HDACi) + cisplatin | Further ↓ cancer stem cell population | ||
| AdCC (cell line) | Chidamide (HDACi) | ↓ proliferative potential | [113] |
| Chidamide (HDACi) + cisplatin | Cell cycle arrest | ||
| MEC (cell line) | Vorinostat (HDACi) | ↓ cancer stem cell population | [69] |
| Vorinostat (HDACi) + Εmetin (anti-NF-κB) | Achieving combined therapeutic effect | ||
| MEC (cell line) | CUDC-101 (EGFRi + HDACi) | ↑ cytotoxicity ↓ cancer stem cell oncogenicity | [147] |
| MEC (cell line) Mice transplanted with tumor cells | Sodium butyrate (HDACi) | ↓ proliferative potential no organ toxicities in vivo | [148] |
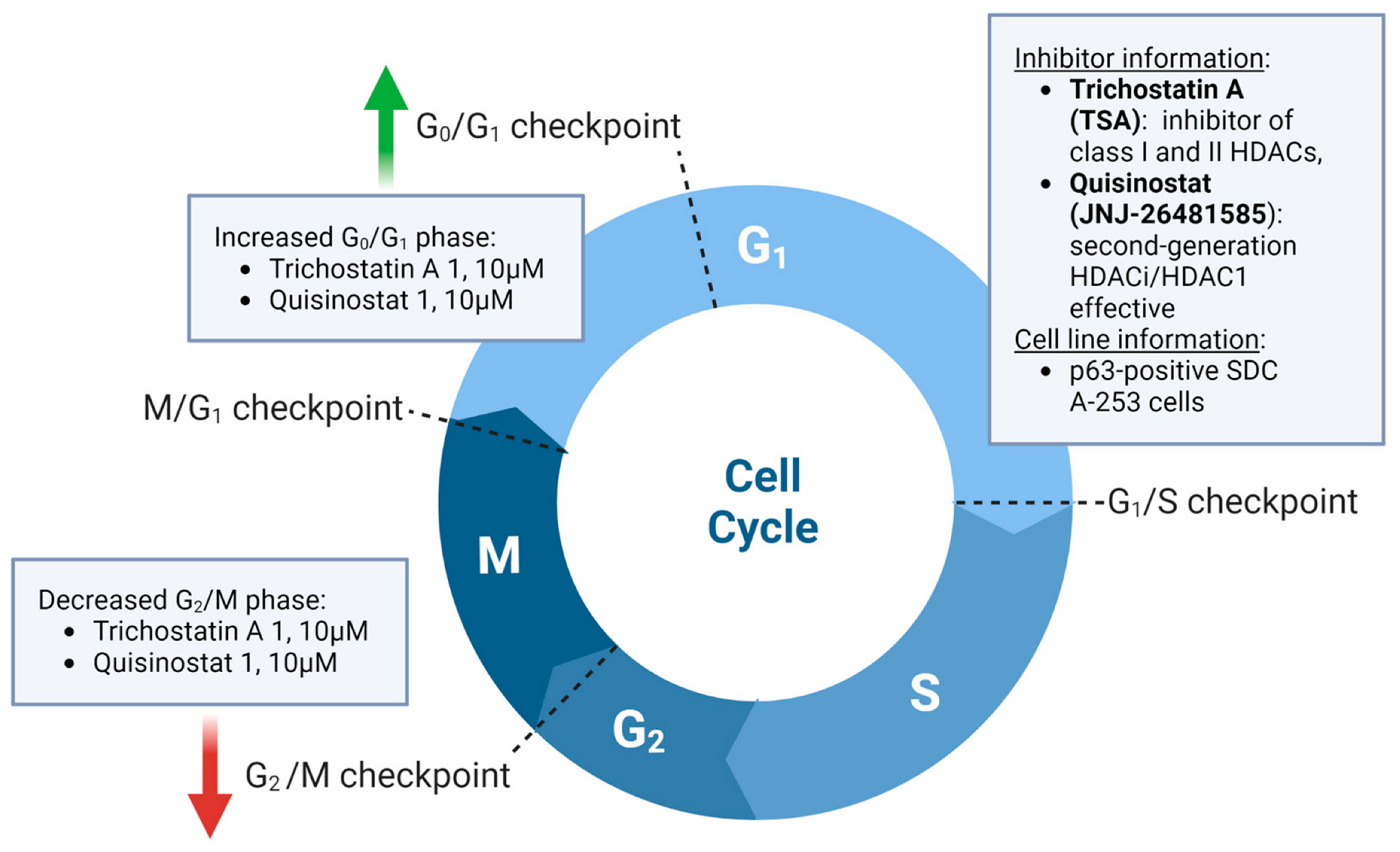
| SGDA (cell line) | HDACi (TSA + Quisinostat) | ↓ proliferation and migration potential ↑ epithelial barrier function | [149] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manou, M.; Kanakoglou, D.S.; Loupis, T.; Vrachnos, D.M.; Theocharis, S.; Papavassiliou, A.G.; Piperi, C. Role of Histone Deacetylases in the Pathogenesis of Salivary Gland Tumors and Therapeutic Targeting Options. Int. J. Mol. Sci. 2023, 24, 10038. https://doi.org/10.3390/ijms241210038
Manou M, Kanakoglou DS, Loupis T, Vrachnos DM, Theocharis S, Papavassiliou AG, Piperi C. Role of Histone Deacetylases in the Pathogenesis of Salivary Gland Tumors and Therapeutic Targeting Options. International Journal of Molecular Sciences. 2023; 24(12):10038. https://doi.org/10.3390/ijms241210038
Chicago/Turabian StyleManou, Maria, Dimitrios S. Kanakoglou, Theodoros Loupis, Dimitrios M. Vrachnos, Stamatios Theocharis, Athanasios G. Papavassiliou, and Christina Piperi. 2023. "Role of Histone Deacetylases in the Pathogenesis of Salivary Gland Tumors and Therapeutic Targeting Options" International Journal of Molecular Sciences 24, no. 12: 10038. https://doi.org/10.3390/ijms241210038
APA StyleManou, M., Kanakoglou, D. S., Loupis, T., Vrachnos, D. M., Theocharis, S., Papavassiliou, A. G., & Piperi, C. (2023). Role of Histone Deacetylases in the Pathogenesis of Salivary Gland Tumors and Therapeutic Targeting Options. International Journal of Molecular Sciences, 24(12), 10038. https://doi.org/10.3390/ijms241210038










