Plants as the Extended Phenotype of Endophytes—The Actual Source of Bioactive Compounds
Abstract
:1. Introduction
2. Are Endophytes Crucial for Plant Existence?
3. The Actual Source of Plant Bioactive Molecules
4. Molecular Background of Host-Specific Compounds Synthesis
5. Co-Evolution of Plant and Its Endophytes
6. Future Prospects
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dawkins, R. The Extended Phenotype: The Long Reach of the Gene; Oxford University Press: Oxford, UK, 1982. [Google Scholar]
- Rosell, F.; Bozsér, O.; Collen, P.; Parker, H. Ecological Impact of Beavers Castor Fiber and Castor Canadensis and Their Ability to Modify Ecosystems. Mamm. Rev. 2005, 35, 248–276. [Google Scholar] [CrossRef]
- Thomas, F.; Schmidt-Rhaesa, A.; Martin, G.; Manu, C.; Durand, P.; Renaud, F. Do Hairworms (Nematomorpha) Manipulate the Water Seeking Behaviour of Their Terrestrial Hosts? J. Evol. Biol. 2002, 15, 356–361. [Google Scholar] [CrossRef]
- Kilner, R.M.; Langmore, N.E. Cuckoos versus Hosts in Insects and Birds: Adaptations, Counter-Adaptations and Outcomes. Biol. Rev. 2011, 86, 836–852. [Google Scholar] [CrossRef] [PubMed]
- Hunter, P. Extended Phenotype Redux. How Far Can the Reach of Genes Extend in Manipulating the Environment of an Organism? EMBO Rep. 2009, 10, 212–215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hunter, P. The Revival of the Extended Phenotype. EMBO Rep. 2018, 19, 5–8. [Google Scholar] [CrossRef]
- Bailey, N.W. Evolutionary Models of Extended Phenotypes. Trends Ecol. Evol. 2012, 27, 561–569. [Google Scholar] [CrossRef]
- Lev-Yadun, S.; Halpern, M. Extended Phenotype in Action. Two Possible Roles for Silica Needles in Plants: Not Just Injuring Herbivores but Also Inserting Pathogens into Their Tissues. Plant Signal. Behav. 2019, 14, 1–5. [Google Scholar] [CrossRef]
- De la Fuente Canto, C.; Simonin, M.; King, E. An Extended Root Phenotype: The Rhizosphere, Its Formation and Impacts on Plant Fitness. Plant J. 2020, 103, 951–964. [Google Scholar] [CrossRef] [Green Version]
- Neal, A.L.; Bacq-Labreuil, A.; Zhang, X.; Clark, I.M.; Coleman, K.; Mooney, S.J.; Ritz, K.; Crawford, J.W. Soil as an Extended Composite Phenotype of the Microbial Metagenome. Sci. Rep. 2020, 10, 10649. [Google Scholar] [CrossRef]
- Herrera Paredes, S.; Lebeis, S.L. Giving Back to the Community: Microbial Mechanisms of Plant–Soil Interactions. Funct. Ecol. 2016, 30, 1043–1052. [Google Scholar] [CrossRef] [Green Version]
- Preston, G.M. Profiling the Extended Phenotype of Plant Pathogens. Mol. Plant Pathol. 2017, 18, 443–456. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruiz-González, M.X.; Vicente, O. The Microbially Extended Phenotype of Plants, a Keystone against Abiotic Stress. EuroBiotech J. 2022, 6, 174–182. [Google Scholar] [CrossRef]
- Partida-Martínez, L.P.; Heil, M. The Microbe-Free Plant: Fact or Artifact? Front. Plant Sci. 2011, 2, 100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lourenço, S.C.; Moldão-Martins, M.; Alves, V.D. Antioxidants of Natural Plant Origins: From Sources to Food Industry Applications. Molecules 2019, 24, 4132. [Google Scholar] [CrossRef] [Green Version]
- Greenwell, M.; Rahman, P.K.S.M. Medicinal Plants: Their Use in Anticancer Treatment. Int. J. Pharm. Sci. Res. 2015, 6, 4103–4112. [Google Scholar] [CrossRef]
- Mallavadhani, U.V.; Chandrashekhar, M.; Nayak, V.L.; Ramakrishna, S. Synthesis and Anticancer Activity of Novel Fused Pyrimidine Hybrids of Myrrhanone C, a Bicyclic Triterpene of Commiphora Mukul Gum Resin. Mol. Divers. 2015, 19, 745–757. [Google Scholar] [CrossRef]
- Reis Nunes, C.; Barreto Arantes, M.; Menezes de Faria Pereira, S.; Leandro da Cruz, L.; De Souza Passos, M.; Pereira de Moraes, L.; Curcino Vieira, I.J.; Barros de Oliveira, D. Plants as Sources of Anti-Inflammatory Agents. Molecules 2020, 25, 3726. [Google Scholar] [CrossRef]
- Madasu, C.; Gudem, S.; Sistla, R.; Uppuluri, V.M. Synthesis and Anti-Inflammatory Activity of Some Novel Pyrimidine Hybrids of Myrrhanone A, a Bicyclic Triterpene of Commiphora Mukul Gum Resin. Mon. Fur Chem. 2017, 148, 2183–2193. [Google Scholar] [CrossRef]
- Savoia, D. Plant-Derived Antimicrobial Compounds: Alternatives to Antibiotics. Future Microbiol. 2012, 7, 979–990. [Google Scholar] [CrossRef] [Green Version]
- Gomes, N.G.M.; Campos, M.G.; Órfão, J.M.C.; Ribeiro, C.A.F. Plants with Neurobiological Activity as Potential Targets for Drug Discovery. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2009, 33, 1372–1389. [Google Scholar] [CrossRef]
- Ali, M.; Khan, T.; Fatima, K.; Ali, Q.A.; Ovais, M.; Khalil, A.T.; Ullah, I.; Raza, A.; Shinwari, Z.K.; Idrees, M. Selected Hepatoprotective Herbal Medicines: Evidence from Ethnomedicinal Applications, Animal Models, and Possible Mechanism of Actions. Phyther. Res. 2018, 32, 199–215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petrovska, B.B. Historical Review of Medicinal Plants’ Usage. Pharmacogn. Rev. 2012, 6, 1–5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Global Botanical and Plant-Derived Drugs Market 2022–2026; February 2022. Available online: https://www.Technavio.Com/Report/Botanical-and-Plant-Derived-Drugs-Market-Industry-Analysis (accessed on 25 March 2023).
- Kumar, A.; Patil, D.; Rajamohanan, P.R.; Ahmad, A. Isolation, Purification and Characterization of Vinblastine and Vincristine from Endophytic Fungus Fusarium Oxysporum Isolated from Catharanthus Roseus. PLoS ONE 2013, 8, e0189308. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mishra, S.; Priyanka; Sharma, S. Metabolomic Insights into Endophyte-Derived Bioactive Compounds. Front. Microbiol. 2022, 13, 416. [Google Scholar] [CrossRef]
- Wilson, D. Endophyte: The Evolution of a Term, and Clarification of Its Use and Definition. Oikos 1995, 73, 274. [Google Scholar] [CrossRef]
- Köberl, M.; Schmidt, R.; Ramadan, E.M.; Bauer, R.; Berg, G. The Microbiome of Medicinal Plants: Diversity and Importance for Plant Growth, Quality, and Health. Front. Microbiol. 2013, 4, 400. [Google Scholar] [CrossRef] [Green Version]
- Sharma, H.; Rai, A.K.; Dahiya, D.; Chettri, R.; Singh, P. Exploring Endophytes for in Vitro Synthesis of Bioactive Compounds Similar to Metabolites Produced in Vivo by Host Plants. AIMS Microbiol. 2021, 7, 175–199. [Google Scholar] [CrossRef]
- Link, H.F. Observationes in Ordines Plantarum Naturales, Dissertatio Prima, Complectens Anandrarum Ordines Epiphytas, Mucedines, Gastromycos et Fungos; Der Gesellschaft Naturforschender Freunde zu Berlin: Berlin, Germany, 1809. [Google Scholar]
- Galippe, V. Note Sur La Présence de Micro Organismes Dans Les Tissus Végétaux (Deuxième Note). CR Seances Soc. Biol. Fil. 1887, 39, 557–560. [Google Scholar]
- Compant, S.; Sessitsch, A.; Mathieu, F. The 125th Anniversary of the First Postulation of the Soil Origin of Endophytic Bacteria—A Tribute to M.L.V. Galippe. Plant Soil 2012, 356, 299–301. [Google Scholar] [CrossRef] [Green Version]
- Laurent, E. Sur l’existence de Microbes Dans Les Tissus Des Plantes Supérieures. Bull. Soc. R. Bot. Bel. 1889, 28, 233–244. [Google Scholar]
- Hardoim, P.R.; van Overbeek, L.S.; van Elsas, J.D. Properties of Bacterial Endophytes and Their Proposed Role in Plant Growth. Trends Microbiol. 2008, 16, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.; Bhattacharjee, A.; Sharma, S. An Ecological Insight into the Multifaceted World of Plant-Endophyte Association. Crit. Rev. Plant Sci. 2021, 40, 127–146. [Google Scholar] [CrossRef]
- Petrini, O. Fungal Endophytes of Tree Leaves. In Microbial Ecology of Leaves. Brock/Springer Series in Contemporary Bioscience; Andrews, J.H., Hirano, S.S., Eds.; Springer: New York, NY, USA, 1991; pp. 179–197. [Google Scholar]
- Hardoim, P.R.; van Overbeek, L.S.; Berg, G.; Pirttilä, A.M.; Compant, S.; Campisano, A.; Döring, M.; Sessitsch, A. The Hidden World within Plants: Ecological and Evolutionary Considerations for Defining Functioning of Microbial Endophytes. Microbiol. Mol. Biol. Rev. 2015, 79, 293–320. [Google Scholar] [CrossRef] [Green Version]
- Clay, K.; Schardl, C. Evolutionary Origins and Ecological Consequences of Endophyte Symbiosis with Grasses. Am. Nat. 2002, 160, S99–S127. [Google Scholar] [CrossRef] [PubMed]
- Herre, E.A.; Knowlton, N.; Mueller, U.G.; Rehner, S.A. Patterns of Ecological Transmission and Evolutionary Association. Trends Ecol. Evol. 1999, 14, 49–53. [Google Scholar] [CrossRef]
- Schardl, C.L.; Craven, K.D.; Speakman, S.; Stromberg, A.; Lindstrom, A.; Yoshida, R. A Novel Test for Host-Symbiont Codivergence Indicates Ancient Origin of Fungal Endophytes in Grasses. Syst. Biol. 2008, 57, 483–498. [Google Scholar] [CrossRef] [Green Version]
- Lyu, D.; Smith, D.L. The Root Signals in Rhizospheric Inter-Organismal Communications. Front. Plant Sci. 2022, 13, 1064058. [Google Scholar] [CrossRef]
- Pinski, A.; Betekhtin, A.; Hupert-Kocurek, K.; Mur, L.A.J.; Hasterok, R. Defining the Genetic Basis of Plant–Endophytic Bacteria Interactions. Int. J. Mol. Sci. 2019, 20, 1947. [Google Scholar] [CrossRef] [Green Version]
- Tian, T.; Sun, B.; Shi, H.; Gao, T.; He, Y.; Li, Y.; Liu, Y.; Li, X.; Zhang, L.; Li, S.; et al. Sucrose Triggers a Novel Signaling Cascade Promoting Bacillus Subtilis Rhizosphere Colonization. ISME J. 2021, 15, 2723–2737. [Google Scholar] [CrossRef]
- Dekak, A.; Menasria, T.; Benhizia, Y.; Chenchouni, H. Endophytic Passenger Bacteria Associated with Genista Cinerea Nodules Growing in North African Drylands. Rhizosphere 2020, 14, 100205. [Google Scholar] [CrossRef]
- Leon, V.C.; Raja, M.; Pandian, R.T.P.; Kumar, A.; Sharma, P. Opportunistic Endophytism of Trichoderma Species in Rice Pusa Basmati-1 (PB1). Ind. J. Exp. Biol. 2018, 56, 121–128. [Google Scholar]
- Bacon, C.W.; Glenn, A.E.; Yates, I.E. Fusarium Verticillioides: Managing the Endophytic Association with Maize for Reduced Fumonisins Accumulation. Toxin Rev. 2008, 27, 411–446. [Google Scholar] [CrossRef]
- Azuddin, N.F.; Mohamad Noor Azmy, M.S.; Zakaria, L. Molecular Identification of Endophytic Fungi in Lawn Grass (Axonopus compressus) and Their Pathogenic Ability. Sci. Rep. 2023, 13, 4239. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Loayza, P.; White, J.F.; Torres, M.S.; Balslev, H.; Kristiansen, T.; Svenning, J.C.; Gil, N. Light Converts Endosymbiotic Fungus to Pathogen, Influencing Seedling Survival and Niche-Space Filling of a Common Tropical Tree, Iriartea Deltoidea. PLoS ONE 2011, 6, e16386. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Xie, J.; Fu, Y.; Cheng, J.; Qu, Z.; Zhao, Z.; Cheng, S.; Chen, T.; Li, B.; Wang, Q.; et al. A 2-Kb Mycovirus Converts a Pathogenic Fungus into a Beneficial Endophyte for Brassica Protection and Yield Enhancement. Mol. Plant 2020, 13, 1420–1433. [Google Scholar] [CrossRef]
- Raza, A.; Razzaq, A.; Mehmood, S.S.; Zou, X.; Zhang, X.; Lv, Y.; Xu, J. Impact of Climate Change on Crops Adaptation and Strategies to Tackle Its Outcome: A Review. Plants 2019, 8, 34. [Google Scholar] [CrossRef] [Green Version]
- Saharan, B.S.; Brar, B.; Duhan, J.S.; Kumar, R.; Marwaha, S.; Rajput, V.D.; Minkina, T. Molecular and Physiological Mechanisms to Mitigate Abiotic Stress Conditions in Plants. Life 2022, 12, 1634. [Google Scholar] [CrossRef]
- Mathivanan, S. Abiotic Stress-Induced Molecular and Physiological Changes and Adaptive Mechanisms in Plants. In Abiotic Stress in Plants; Fahad, S., Saud, S., Chen, Y., Wu, C., Wang, D., Eds.; IntechOpen: London, UK, 2021; ISBN 0000957720. [Google Scholar]
- Baron, N.C.; Rigobelo, E.C. Endophytic Fungi: A Tool for Plant Growth Promotion and Sustainable Agriculture. Mycology 2022, 13, 39–55. [Google Scholar] [CrossRef]
- Chow, C.; Padda, K.P.; Puri, A.; Chanway, C.P. An Archaic Approach to a Modern Issue: Endophytic Archaea for Sustainable Agriculture. Curr. Microbiol. 2022, 79, 322. [Google Scholar] [CrossRef]
- Ryan, R.P.; Germaine, K.; Franks, A.; Ryan, D.J.; Dowling, D.N. Bacterial Endophytes: Recent Developments and Applications. FEMS Microbiol. Lett. 2008, 278, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Márquez, L.M.; Redman, R.S.; Rodriguez, R.J.; Roossinck, M.J. A Virus in a Fungus in a Plant: Three-Way Symbiosis Required for Thermal Tolerance. Science 2007, 315, 513–515. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Redman, R.S.; Sheehan, K.B.; Stout, R.G.; Rodriguez, R.J.; Henson, J.M. Thermotolerance Generated by Plant/Fungal Symbiosis. Science 2002, 298, 1581. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, R.J.; Henson, J.; Van Volkenburgh, E.; Hoy, M.; Wright, L.; Beckwith, F.; Kim, Y.O.; Redman, R.S. Stress Tolerance in Plants via Habitat-Adapted Symbiosis. ISME J. 2008, 2, 404–416. [Google Scholar] [CrossRef] [Green Version]
- Redman, R.S.; Kim, Y.O.; Woodward, C.J.D.A.; Greer, C.; Espino, L.; Doty, S.L.; Rodriguez, R.J. Increased Fitness of Rice Plants to Abiotic Stress via Habitat Adapted Symbiosis: A Strategy for Mitigating Impacts of Climate Change. PLoS ONE 2011, 6, e14823. [Google Scholar] [CrossRef] [PubMed]
- Hosseyni Moghaddam, M.S.; Safaie, N.; Soltani, J.; Hagh-Doust, N. Desert-Adapted Fungal Endophytes Induce Salinity and Drought Stress Resistance in Model Crops. Plant Physiol. Biochem. 2021, 160, 225–238. [Google Scholar] [CrossRef]
- Hamdan, A. Psychrophiles: Ecological Significance and Potential Industrial Application. S. Afr. J. Sci. 2018, 114, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Araya, M.A.; Valenzuela, T.; Inostroza, N.G.; Maruyama, F.; Jorquera, M.A.; Acuña, J.J. Isolation and Characterization of Cold-Tolerant Hyper-ACC-Degrading Bacteria from the Rhizosphere, Endosphere, and Phyllosphere of Antarctic Vascular Plants. Microorganisms 2020, 8, 1788. [Google Scholar] [CrossRef]
- Vanhaelewyn, L.; Van Der Straeten, D.; De Coninck, B.; Vandenbussche, F. Ultraviolet Radiation From a Plant Perspective: The Plant-Microorganism Context. Front. Plant Sci. 2020, 11, 597642. [Google Scholar] [CrossRef]
- Barrera, A.; Hereme, R.; Ruiz-Lara, S.; Larrondo, L.F.; Gundel, P.E.; Pollmann, S.; Molina-Montenegro, M.A.; Ramos, P. Fungal Endophytes Enhance the Photoprotective Mechanisms and Photochemical Efficiency in the Antarctic Colobanthus Quitensis (Kunth) Bartl. Exposed to UV-B Radiation. Front. Ecol. Evol. 2020, 8, 122. [Google Scholar] [CrossRef]
- Ramos, P.; Rivas, N.; Pollmann, S.; Casati, P.; Molina-Montenegro, M.A. Hormonal and Physiological Changes Driven by Fungal Endophytes Increase Antarctic Plant Performance under UV-B Radiation. Fungal Ecol. 2018, 34, 76–82. [Google Scholar] [CrossRef]
- Oses-Pedraza, R.; Torres-Díaz, C.; Lavín, P.; Retamales-Molina, P.; Atala, C.; Gallardo-Cerda, J.; Acuña-Rodríguez, I.S.; Molina-Montenegro, M.A. Root Endophytic Penicillium Promotes Growth of Antarctic Vascular Plants by Enhancing Nitrogen Mineralization. Extremophiles 2020, 24, 721–732. [Google Scholar] [CrossRef] [PubMed]
- Molina-Montenegro, M.A.; Acuña-Rodríguez, I.S.; Torres-Díaz, C.; Gundel, P.E.; Dreyer, I. Antarctic Root Endophytes Improve Physiological Performance and Yield in Crops under Salt Stress by Enhanced Energy Production and Na+ Sequestration. Sci. Rep. 2020, 10, 5819. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haruma, T.; Yamaji, K.; Masuya, H.; Hanyu, K. Root Endophytic Chaetomium Cupreum Promotes Plant Growth and Detoxifies Aluminum in Miscanthus Sinensis Andersson Growing at the Acidic Mine Site. Plant Species Biol. 2018, 33, 109–122. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Yang, R.; Häggblom, M.M.; Li, M.; Guo, L.; Li, B.; Kolton, M.; Cao, Z.; Solemani, M.; Chen, Z.; et al. Characterization of Diazotrophic Root Endophytes in Chinese Silvergrass (Miscanthus sinensis). Microbiome 2022, 10, 186. [Google Scholar] [CrossRef]
- Salazar-Ramírez, G.; Flores-Vallejo, R.D.C.; Rivera-Leyva, J.C.; Tovar-Sánchez, E.; Sánchez-Reyes, A.; Mena-Portales, J.; Sánchez-Carbente, M.D.R.; Gaitán-Rodríguez, M.F.; Batista-García, R.A.; Villarreal, M.L.; et al. Characterization of Fungal Endophytes Isolated from the Metal Hyperaccumulator Plant Vachellia Farnesiana Growing in Mine Tailings. Microorganisms 2020, 8, 162931. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, Y.; Oliveira, R.S.; Nai, F.; Rajkumar, M.; Luo, Y.; Rocha, I.; Freitas, H. The Hyperaccumulator Sedum Plumbizincicola Harbors Metal-Resistant Endophytic Bacteria That Improve Its Phytoextraction Capacity in Multi-Metal Contaminated Soil. J. Environ. Manag. 2015, 156, 62–69. [Google Scholar] [CrossRef] [Green Version]
- Tovar-Sanchez, E.; Concepcion-Acosta, C.M.; Sanchez-Reyes, A.; Sanchez-Cruz, R.; Folch-Mallol, J.L.; Mussali-Galante, P. Aspergillus Luchuensis, an Endophyte Fungus from the Metal Hyperaccumulator Plant Prosopis Laevigata, Promotes Its Growth and Increases Metal Translocation. Plants 2023, 12, 1338. [Google Scholar] [CrossRef]
- Tufail, M.A.; Ayyub, M.; Irfan, M.; Shakoor, A.; Chibani, C.M.; Schmitz, R.A. Endophytic Bacteria Perform Better than Endophytic Fungi in Improving Plant Growth under Drought Stress: A Meta-Comparison Spanning 12 Years (2010–2021). Physiol. Plant. 2022, 174, e13806. [Google Scholar] [CrossRef]
- Venugopalan, A.; Srivastava, S. Endophytes as in Vitro Production Platforms of High Value Plant Secondary Metabolites. Biotechnol. Adv. 2015, 33, 873–887. [Google Scholar] [CrossRef]
- Zhao, J.; Zhou, L.; Wang, J.; Shan, T. Endophytic Fungi for Producing Bioactive Compounds Originally from Their Host Plants Endophytic Fungi for Producing Bioactive Compounds Originally from Their Host Plants. Curr. Res. Technol. Educ. Trop. Appl. Microbiol. Microbial. Biotechnol. 2010, 1, 567–576. [Google Scholar]
- Altemimi, A.; Lakhssassi, N.; Baharlouei, A.; Watson, D.G.; Lightfoot, D.A. Phytochemicals: Extraction, Isolation, and Identification of Bioactive Compounds from Plant Extracts. Plants 2017, 6, 42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stierle, A.; Strobel, G.; Stierle, D. Taxol and Taxane Production by Taxomyces Andreanae, an Endop. Science 1993, 260, 214–216. [Google Scholar] [CrossRef] [PubMed]
- Gangadevi, V.; Muthumary, J. Taxol, an Anticancer Drug Produced by an Endophytic Fungus Bartalinia Robillardoides Tassi, Isolated from a Medicinal Plant, Aegle Marmelos Correa Ex Roxb. World J. Microbiol. Biotechnol. 2008, 24, 717–724. [Google Scholar] [CrossRef]
- Gallego-Jara, J.; Lozano-Terol, G.; Alba, R.; Manuel, C.; Puente, T.D.D. A Compressive Review about Taxol: History and Future Challenges. Molecules 2020, 25, 5986. [Google Scholar] [CrossRef] [PubMed]
- Barrales-Cureño, H.J.; Ramos Valdivia, A.C.; Soto Hernández, M. Increased Production of Taxoids in Suspension Cultures of Taxus Globosa after Elicitation. Futur. Pharmacol. 2022, 2, 45–54. [Google Scholar] [CrossRef]
- El-Sayed, A.S.A.; El Sayed, M.T.; Rady, A.; Zein, N.; Enan, G.; Shindia, A.; El-Hefnawy, S.; Sitohy, M.; Sitohy, B. Exploiting the Biosynthetic Potency of Taxol from Fungal Endophytes of Conifers Plants; Genome Mining and Metabolic Manipulation. Molecules 2020, 25, 3000. [Google Scholar] [CrossRef]
- Wu, Q.Y.; Huang, Z.Y.; Wang, J.Y.; Yu, H.L.; Xu, J.H. Construction of an Escherichia coli Cell Factory to Synthesize Taxadien-5α-Ol, the Key Precursor of Anti-Cancer Drug Paclitaxel. Bioresour. Bioprocess. 2022, 9, 82. [Google Scholar] [CrossRef]
- Huang, J.J.; Wei, T.; Ye, Z.W.; Zheng, Q.W.; Jiang, B.H.; Han, W.F.; Ye, A.Q.; Han, P.Y.; Guo, L.Q.; Lin, J.F. Microbial Cell Factory of Baccatin III Preparation in Escherichia coli by Increasing DBAT Thermostability and in Vivo Acetyl-CoA Supply. Front. Microbiol. 2022, 12, 803490. [Google Scholar] [CrossRef]
- Wall, M.E.; Wani, M.C.; Cook, C.E.; Palmer, K.H.; McPhail, A.T.; Sim, G.A. Plant Antitumor Agents. I. The Isolation and Structure of Camptothecin, a Novel Alkaloidal Leukemia and Tumor Inhibitor from Camptotheca acuminata. J. Am. Chem. Soc. 1966, 88, 3888–3890. [Google Scholar] [CrossRef]
- Pandey, S.S.; Singh, S.; Babu, C.S.V.; Shanker, K.; Srivastava, N.K.; Shukla, A.K.; Kalra, A. Fungal Endophytes of Catharanthus Roseus Enhance Vindoline Content by Modulating Structural and Regulatory Genes Related to Terpenoid Indole Alkaloid Biosynthesis. Sci. Rep. 2016, 6, 26583. [Google Scholar] [CrossRef] [Green Version]
- Shweta, S.; Zuehlke, S.; Ramesha, B.T.; Priti, V.; Mohana Kumar, P.; Ravikanth, G.; Spiteller, M.; Vasudeva, R.; Uma Shaanker, R. Endophytic Fungal Strains of Fusarium Solani, from Apodytes Dimidiata E. Mey. Ex Arn (Icacinaceae) Produce Camptothecin, 10-Hydroxycamptothecin and 9-Methoxycamptothecin. Phytochemistry 2010, 71, 117–122. [Google Scholar] [CrossRef]
- Kusari, S.; Zühlke, S.; Spiteller, M. An Endophytic Fungus from Camptotheca Acuminata That Produces Camptothecin and Analogues. J. Nat. Prod. 2009, 72, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Ran, X.; Zhang, G.; Li, S.; Wang, J. Characterization and Antitumor Activity of Camptothecin from Endophytic Fungus Fusarium Solani Isolated from Camptotheca Acuminate. Afr. Health Sci. 2017, 17, 566–574. [Google Scholar] [CrossRef] [Green Version]
- Gurudatt, P.S.; Priti, V.; Shweta, S.; Ramesha, B.T.; Ravikanth, G.; Vasudeva, R.; Amna, T.; Deepika, S.; Ganeshaiah, K.N.; Uma Shaanker, R.; et al. Attenuation of Camptothecin Production and Negative Relation between Hyphal Biomass and Camptothecin Content in Endophytic Fungal Strains Isolated from Nothapodytes Nimmoniana Grahm (Icacinaceae). Curr. Sci. 2010, 98, 1006–1010. [Google Scholar]
- Shweta, S.; Bindu, J.H.; Raghu, J.; Suma, H.K.; Manjunatha, B.L.; Kumara, P.M.; Ravikanth, G.; Nataraja, K.N.; Ganeshaiah, K.N.; Uma Shaanker, R. Isolation of Endophytic Bacteria Producing the Anti-Cancer Alkaloid Camptothecine from Miquelia Dentata Bedd. (Icacinaceae). Phytomedicine 2013, 20, 913–917. [Google Scholar] [CrossRef]
- Puri, S.C.; Nazir, A.; Chawla, R.; Arora, R.; Riyaz-Ul-Hasan, S.; Amna, T.; Ahmed, B.; Verma, V.; Singh, S.; Sagar, R.; et al. The Endophytic Fungus Trametes Hirsuta as a Novel Alternative Source of Podophyllotoxin and Related Aryl Tetralin Lignans. J. Biotechnol. 2006, 122, 494–510. [Google Scholar] [CrossRef]
- Xu, X.-D.; Liang, W.-X.; Yao, L.; Paek, K.Y.; Wang, Y.; Gao, W.-Y. Production of Ginsenoside by Chaetomium Sp. and Its Effect on Enhancing the Contents of Ginsenosides in Panax Ginseng Adventitious Roots. Biochem. Eng. J. 2021, 174, 108100. [Google Scholar] [CrossRef]
- Fu, Y.; Yin, Z.; Yin, C. Biotransformation of Ginsenoside Rb1 to Ginsenoside Rg3 by Endophytic Bacterium burkholderia sp. GE 17-7 Isolated from Panax Ginseng. J. Appl. Microbiol. 2017, 122, 1579–1585. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Jin, H.; Fu, Y.; Yin, Z.; Yin, C. Production of Rare Ginsenosides Rg3 and Rh2 by Endophytic Bacteria from Panax Ginseng. J. Agric. Food Chem. 2019, 67, 8493–8499. [Google Scholar] [CrossRef]
- Nadeem, M.; Ram, M.; Alam, P.; Ahmad, M.M.; Mohammad, A.; Al-Qurainy, F.; Khan, S.; Abdin, M.Z. Fusarium solani, P1, a new endophytic podophyllotoxin-producing fungus from roots of Podophyllum hexandrum. Afr. J. Microbiol. Res. 2012, 6, 2493–2499. [Google Scholar] [CrossRef]
- Huang, J.X.; Zhang, J.; Zhang, X.R.; Zhang, K.; Zhang, X.; He, X.R. Mucor Fragilis as a Novel Source of the Key Pharmaceutical Agents Podophyllotoxin and Kaempferol. Pharm. Biol. 2014, 52, 1237–1243. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Zhou, Y.; Zhou, X.; Xia, X.; Wei, Y.; He, L.; Tang, H.; Yu, L. Diversity and Bioactive Potential of Culturable Fungal Endophytes of Dysosma versipellis; A Rare Medicinal Plant Endemic to China. Sci. Rep. 2018, 8, 5929. [Google Scholar] [CrossRef] [PubMed]
- Kour, A.; Shawl, A.S.; Rehman, S.; Sultan, P.; Qazi, P.H.; Suden, P.; Khajuria, R.K.; Verma, V. Isolation and Identification of an Endophytic Strain of Fusarium Oxysporum Producing Podophyllotoxin from Juniperus Recurva. World J. Microbiol. Biotechnol. 2008, 24, 1115–1121. [Google Scholar] [CrossRef]
- Eyberger, A.; Dondapati, R.; Porter, J.R. Endophyte Fungal Isolates from Podophyllum Peltatum Produce Podophyllotoxin. J. Nat. Prod. 2006, 69, 1121–1124. [Google Scholar] [CrossRef] [PubMed]
- Dwibedi, V.; Saxena, S. Diversity and Phylogeny of Resveratrol-Producing Culturable Endophytic Fungi from Vitis Species in India. 3 Biotech 2019, 9, 182. [Google Scholar] [CrossRef]
- Dwibedi, V.; Saxena, S. Arcopilus Aureus, a Resveratrol-Producing Endophyte from Vitis Vinifera. Appl. Biochem. Biotechnol. 2018, 186, 476–495. [Google Scholar] [CrossRef]
- Meshram, V.; Kapoor, N.; Dwibedi, V.; Srivastava, A.; Saxena, S. Extracellular Resveratrol Producing Endophytic Fungus, Quambalaria Cyanenscens. Afr. J. Bot. 2022, 146, 409–4016. [Google Scholar] [CrossRef]
- Shi, J.; Zeng, Q.; Liu, Y.; Pan, Z. Alternaria Sp. MG1, a Resveratrol-Producing Fungus: Isolation, Identification, and Optimal Cultivation Conditions for Resveratrol Production. Appl. Microbiol. Biotechnol. 2012, 95, 369–379. [Google Scholar] [CrossRef]
- Roat, C.; Saraf, M. Isolation and Characterization of T-Resveratrol and α-Viniferin, a Bioactive Secondary Metabolite of an Endophytic Fungus Aspergillus Stellifer Ab4, from Vitis Vinifera. J. Microbiol. Biotechnol. Food Sci. 2020, 9, 708–713. [Google Scholar] [CrossRef]
- Parthasarathy, R.; Shanmuganathan, R.; Pugazhendhi, A. Vinblastine Production by the Endophytic Fungus Curvularia Verruculosa from the Leaves of Catharanthus Roseus and Its in Vitro Cytotoxicity against HeLa Cell Line. Anal. Biochem. 2020, 593, 113530. [Google Scholar] [CrossRef]
- Kuriakose, G.C.; Palem, P.P.C.; Jayabaskaran, C. Fungal Vincristine from Eutypella spp.—CrP14 Isolated from Catharanthus Roseus Induces Apoptosis in Human Squamous Carcinoma Cell Line -A431. BMC Complement. Altern. Med. 2016, 16, 302. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, A.; Abnave, P.; Ahmad, A. Cultural, Morphological and Molecular Characterization of Vinca Alkaloids Producing Endophytic Fungus Fusarium Solani Isolated from Catharanthus Roseus. Int. J. Bot. Res. 2013, 3, 1–12. [Google Scholar]
- Yang, K.; Liang, J.; Li, Q.; Kong, X.; Chen, R.; Jin, Y. Cladosporium Cladosporioides XJ-AC03, an Aconitine-Producing Endophytic Fungus Isolated from Aconitum Leucostomum. World J. Microbiol. Biotechnol. 2013, 29, 933–938. [Google Scholar] [CrossRef]
- Aly, A.H.; Edrada-Ebel, R.A.; Indriani, I.D.; Wray, V.; Müller, W.E.G.; Totzke, F.; Zirrgiebel, U.; Schächtele, C.; Kubbutat, M.H.G.; Lin, W.H.; et al. Cytotoxic Metabolites from the Fungal Endophyte Alternaria sp. and Their Subsequent Detection in Its Host Plant Polygonum Senegalense. J. Nat. Prod. 2008, 71, 972–980. [Google Scholar] [CrossRef]
- Li, N.; Xu, D.; Huang, R.H.; Zheng, J.Y.; Liu, Y.Y.; Hu, B.S.; Gu, Y.Q.; Du, Q. A New Source of Diterpene Lactones From Andrographis paniculata (Burm. f.) Nees—Two Endophytic Fungi of Colletotrichum Sp. With Antibacterial and Antioxidant Activities. Front. Microbiol. 2022, 13, 819770. [Google Scholar] [CrossRef] [PubMed]
- Nischitha, R.; Shivanna, M.B. Metabolite Fingerprinting, in Vitro Antimicrobial and Antioxidant Activities and in-Silico Docking in Alloteropsis Cimicina and Its Endophytic Fungus Penicillium Pinophilum. Mol. Biol. Rep. 2021, 48, 4021–4037. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Bhatt, P.; Chaturvedi, P. Determination and Quantification of Asiaticoside in Endophytic Fungus from Centella asiatica (L.) Urban. World J. Microbiol. Biotechnol. 2018, 34, 111. [Google Scholar] [CrossRef]
- Kusari, S.; Verma, V.C.; Lamshoeft, M.; Spiteller, M. An Endophytic Fungus from Azadirachta Indica A. Juss. That Produces Azadirachtin. World J. Microbiol. Biotechnol. 2012, 28, 1287–1294. [Google Scholar] [CrossRef]
- Zhao, J.; Fu, Y.; Luo, M.; Zu, Y.; Wang, W.; Zhao, C.; Gu, C. Endophytic Fungi from Pigeon Pea [Cajanus cajan (L.) Millsp.] Produce Antioxidant Cajaninstilbene Acid. J. Agric. Food Chem. 2012, 60, 4314–4319. [Google Scholar] [CrossRef]
- Mohinudeen, I.A.H.K.; Kanumuri, R.; Soujanya, K.N.; Shaanker, R.U.; Rayala, S.K.; Srivastava, S. Sustainable Production of Camptothecin from an Alternaria sp. Isolated from Nothapodytes Nimmoniana. Sci. Rep. 2021, 11, 1478. [Google Scholar] [CrossRef]
- Devari, S.; Jaglan, S.; Kumar, M.; Deshidi, R.; Guru, S.; Bhushan, S.; Kushwaha, M.; Gupta, A.P.; Gandhi, S.G.; Sharma, J.P.; et al. Capsaicin Production by Alternaria Alternata, an Endophytic Fungus from Capsicum Annum; LC-ESI-MS/MS Analysis. Phytochemistry 2014, 98, 183–189. [Google Scholar] [CrossRef]
- Deepika, V.B.; Vohra, M.; Mishra, S.; Dorai, K.; Rai, P.; Satyamoorthy, K.; Murali, T.S. DNA Demethylation Overcomes Attenuation of Colchicine Biosynthesis in an Endophytic Fungus Diaporthe. J. Biotechnol. 2020, 323, 33–41. [Google Scholar] [CrossRef]
- Kusari, S.; Lamshöft, M.; Spiteller, M. Aspergillus Fumigatus Fresenius, an Endophytic Fungus from Juniperus communis L. Horstmann as a Novel Source of the Anticancer pro-Drug Deoxypodophyllotoxin. J. Appl. Microbiol. 2009, 107, 1019–1030. [Google Scholar] [CrossRef] [PubMed]
- Sarsaiya, S.; Jain, A.; Fan, X.; Jia, Q.; Xu, Q.; Shu, F.; Zhou, Q.; Shi, J.; Chen, J. New Insights into Detection of a Dendrobine Compound from a Novel Endophytic Trichoderma Longibrachiatum Strain and Its Toxicity against Phytopathogenic Bacteria. Front. Microbiol. 2020, 11, 337. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaul, S.; Ahmed, M.; Zargar, K.; Sharma, P.; Dhar, M.K. Prospecting Endophytic Fungal Assemblage of Digitalis lanata Ehrh. (Foxglove) as a Novel Source of Digoxin: A Cardiac Glycoside. 3 Biotech 2013, 3, 335–340. [Google Scholar] [CrossRef] [Green Version]
- Xiang, H.; Zhang, T.; Pang, X.; Wei, Y.; Liu, H.; Zhang, Y.; Ma, B.; Yu, L. Isolation of Endophytic Fungi from Dioscorea Zingiberensis C. H. Wright and Application for Diosgenin Production by Solid-State Fermentation. Appl. Microbiol. Biotechnol. 2018, 102, 5519–5532. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.-D.; Li, J.-R.; Zhou, L.-G.; Xu, L.-J.; Li, J.; Zhao, J.-L. Determination of Diosgenin Content of the Endophytic Fungi from Paris polyphylla Var. Yunnanensis by Using an Optimum ELISA. Nat. Prod. Res. Dev. 2007, 19, 1020–1023. [Google Scholar]
- Vigneshwari, A.; Rakk, D.; Németh, A.; Kocsubé, S.; Kiss, N.; Csupor, D.; Papp, T.; Škrbić, B.; Vágvölgyi, C.; Szekeres, A. Host Metabolite Producing Endophytic Fungi Isolated from Hypericum Perforatum. PLoS ONE 2019, 14, e217060. [Google Scholar] [CrossRef]
- Kusari, S.; Zühlke, S.; Košuth, J.; Čellárová, E.; Spiteller, M. Light-Independent Metabolomics of Endophytic Thielavia Subthermophila Provides Insight into Microbial Hypericin Biosynthesis. J. Nat. Prod. 2009, 72, 1825–1835. [Google Scholar] [CrossRef]
- Mir, R.A.; Kaushik, P.S.; Chowdery, R.A.; Anuradha, M. Elicitation of Forskolin in Cultures of Rhizactonia Bataticola-a Phytochemical Synthesizing Endophytic Fungi. Int. J. Pharm. Pharm. Sci. 2015, 7, 185–189. [Google Scholar]
- Yin, H.; Zhao, Q.; Sun, F.M.; An, T. Gentiopicrin-Producing Endophytic Fungus Isolated from Gentiana macrophylla. Phytomedicine 2009, 16, 793–797. [Google Scholar] [CrossRef] [PubMed]
- Parthasarathy, R.; Sathiyabama, M. Gymnemagenin-Producing Endophytic Fungus Isolated from a Medicinal Plant Gymnema Sylvestre R.Br. Appl. Biochem. Biotechnol. 2014, 172, 3141–3152. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Zhou, J.; Lin, Z.; Hu, Z. Study on Fermentation for Production of Huperzine A from Endophytic Fungus 2F09P03B of Huperzia serrata. Chin. Med. Biotechnol. 2007, 2, 254–259. [Google Scholar]
- Zhu, D.; Wang, J.; Zeng, Q.; Zhang, Z.; Yan, R. A Novel Endophytic Huperzine A-Producing Fungus, Shiraia Sp. Slf14, Isolated from Huperzia Serrata. J. Appl. Microbiol. 2010, 109, 1469–1478. [Google Scholar] [CrossRef] [PubMed]
- Kusari, S.; Lamshöft, M.; Zühlke, S.; Spiteller, M. An Endophytic Fungus from Hypericum Perforatum That Produces Hypericin. J. Nat. Prod. 2008, 71, 26–29. [Google Scholar] [CrossRef]
- Pan, B.F.; Su, X.; Hu, B.; Yang, N.; Chen, Q.; Wu, W. Fusarium Redolens 6WBY3, an Endophytic Fungus Isolated from Fritillaria unibracteata Var. Wabuensis, Produces Peimisine and Imperialine-3β-d-Glucoside. Fitoterapia 2015, 103, 213–221. [Google Scholar] [CrossRef]
- Blankenship, J.D.; Spiering, M.J.; Wilkinson, H.H.; Fannin, F.F.; Bush, L.P.; Schardl, C.L. Production of Loline Alkaloids by the Grass Endophyte, Neotyphodium Uncinatum, in Defined Media. Phytochemistry 2001, 58, 395–401. [Google Scholar] [CrossRef]
- Nicoletti, R.; Ferranti, P.; Caira, S.; Misso, G.; Castellano, M.; Di Lorenzo, G.; Caraglia, M. Myrtucommulone Production by a Strain of Neofusicoccum Australe Endophytic in Myrtle (Myrtus Communis). World J. Microbiol. Biotechnol. 2014, 30, 1047–1052. [Google Scholar] [CrossRef] [Green Version]
- Qin, D.; Wang, L.; Han, M.; Wang, J.; Song, H.; Yan, X.; Duan, X.; Dong, J. Effects of an Endophytic Fungus Umbelopsis Dimorphaon the Secondary Metabolites of Host-Plant Kadsura Angustifolia. Front. Microbiol. 2018, 9, 2845. [Google Scholar] [CrossRef] [Green Version]
- Pan, F.; Hou, K.; Gao, F.; Hu, B.; Chen, Q.; Wu, W. Peimisine and Peiminine Production by Endophytic Fungus fusarium sp. Isolated from Fritillaria unibracteata Var. Wabensis. Phytomedicine 2014, 21, 1104–1109. [Google Scholar] [CrossRef]
- Chithra, S.; Jasim, B.; Sachidanandan, P.; Jyothis, M.; Radhakrishnan, E.K. Piperine Production by Endophytic Fungus Colletotrichum Gloeosporioides Isolated from Piper Nigrum . Phytomedicine 2014, 21, 534–540. [Google Scholar] [CrossRef] [PubMed]
- Verma, V.C.; Lobkovsky, E.; Gange, A.C.; Singh, S.K.; Prakash, S. Piperine Production by Endophytic Fungus periconia sp. Isolated from Piper longum L. J. Antibiot. 2011, 64, 427–431. [Google Scholar] [CrossRef] [PubMed]
- Ebada, S.S.; Eze, P.; Okoye, F.B.C.; Esimone, C.O.; Proksch, P. The Fungal Endophyte Nigrospora Oryzae Produces Quercetin Monoglycosides Previously Known Only from Plants. ChemistrySelect 2016, 1, 2767–2771. [Google Scholar] [CrossRef]
- Maehara, S.; Agusta, A.; Kitamura, C.; Ohashi, K.; Shibuya, H. Composition of the Endophytic Filamentous Fungi Associated with Cinchona Ledgeriana Seeds and Production of Cinchona Alkaloids. J. Nat. Med. 2016, 70, 271–275. [Google Scholar] [CrossRef] [PubMed]
- You, X.; Feng, S.; Luo, S.; Cong, D.; Yu, Z.; Yang, Z.; Zhang, J. Studies on a Rhein-Producing Endophytic Fungus Isolated from Rheum palmatum L. Fitoterapia 2013, 85, 161–168. [Google Scholar] [CrossRef]
- Cheng, Y.; Liu, G.; Li, Z.; Zhou, Y.; Gao, N. Screening Saikosaponin d (SSd)-Producing Endophytic Fungi from Bupleurum scorzonerifolium Willd. World J. Microbiol. Biotechnol. 2022, 38, 242. [Google Scholar] [CrossRef]
- Cui, J.; Guo, T.; Chao, J.; Wang, M.; Wang, J. Potential of the Endophytic Fungus Phialocephala Fortinii Rac56 Found in Rhodiola Plants to Produce Salidroside and P-Tyrosol. Molecules 2016, 21, 502. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Zhai, X.; Shu, Z.; Dong, R.; Ming, Q.; Qin, L.; Zheng, C. Phoma Glomerata D14: An Endophytic Fungus from Salvia Miltiorrhiza That Produces Salvianolic Acid C. Curr. Microbiol. 2016, 73, 31–37. [Google Scholar] [CrossRef]
- Wu, H.; Yang, H.Y.; You, X.L.; Li, Y.H. Diversity of Endophytic Fungi from Roots of Panax Ginseng and Their Saponin Yield Capacities. Springerplus 2013, 2, 107. [Google Scholar] [CrossRef] [Green Version]
- El-Elimat, T.; Raja, H.A.; Graf, T.N.; Faeth, S.H.; Cech, N.B.; Oberlies, N.H. Flavonolignans from Aspergillus Iizukae, a Fungal Endophyte of Milk Thistle (Silybum marianum). J. Nat. Prod. 2014, 77, 193–199. [Google Scholar] [CrossRef]
- Yin, H.; Chen, J.L. Sipeimine-Producing Endophytic Fungus Isolated from Fritillaria Ussuriensis. Z. Für Nat. C 2008, 63, 789–793. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El-Hawary, S.S.; Mohammed, R.; Abouzid, S.F.; Bakeer, W.; Ebel, R.; Sayed, A.M.; Rateb, M.E. Solamargine Production by a Fungal Endophyte of Solanum Nigrum. J. Appl. Microbiol. 2016, 120, 900–911. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ming, Q.; Han, T.; Li, W.; Zhang, Q.; Zhang, H.; Zheng, C.; Huang, F.; Rahman, K.; Qin, L. Tanshinone IIA and Tanshinone i Production by Trichoderma Atroviride D16, an Endophytic Fungus in Salvia Miltiorrhiza. Phytomedicine 2012, 19, 330–333. [Google Scholar] [CrossRef] [PubMed]
- Gauchan, D.P.; Vélëz, H.; Acharya, A.; Östman, J.R.; Lundén, K.; Elfstrand, M.; García-Gil, M.R. Annulohypoxylon sp. Strain MUS1, an Endophytic Fungus Isolated from Taxus wallichiana Zucc., Produces Taxol and Other Bioactive Metabolites. 3 Biotech 2021, 11, 152. [Google Scholar] [CrossRef]
- Kumar, P.; Singh, B.; Thakur, V.; Thakur, A.; Thakur, N.; Pandey, D.; Chand, D. Hyper-Production of Taxol from Aspergillus Fumigatus, an Endophytic Fungus Isolated from Taxus sp. of the Northern Himalayan Region. Biotechnol. Rep. 2019, 24, e00395. [Google Scholar] [CrossRef]
- Bashyal, B. Seimatoantlerium nepalense, an endophytic taxol producing coelomycete from Himalayan yew (Taxus wallachiana). Mycotaxonomy 1999, 72, 33–42. [Google Scholar]
- Strobel, G.A.; Ford, E.; Li, J.Y.; Sears, J.; Sidhu, R.S.; Hess, W.M. Seimatoantlerium Tepuiense Gen. Nov., a Unique Epiphytic Fungus Producing Taxol from the Venezuelan Guyana. Syst. Appl. Microbiol. 1999, 22, 426–433. [Google Scholar] [CrossRef]
- Palem, P.P.C.; Kuriakose, G.C.; Jayabaskaran, C. An Endophytic Fungus, Talaromyces Radicus, Isolated from Catharanthus Roseus, Produces Vincristine and Vinblastine, Which Induce Apoptotic Cell Death. PLoS ONE 2015, 10, e0144476. [Google Scholar] [CrossRef]
- Na, R.; Jiajia, L.; Dongliang, Y.; Yingzi, P.; Juan, H.; Xiong, L.; Nana, Z.; Jing, Z.; Yitian, L. Indentification of Vincamine Indole Alkaloids Producing Endophytic Fungi Isolated from Nerium Indicum, Apocynaceae. Microbiol. Res. 2016, 192, 114–121. [Google Scholar] [CrossRef]
- Yin, H.; Sun, Y.H. Vincamine-Producing Endophytic Fungus Isolated from Vinca Minor. Phytomedicine 2011, 18, 802–805. [Google Scholar] [CrossRef]
- Sathiyabama, M.; Parthasarathy, R. Withanolide Production by Fungal Endophyte Isolated from Withania Somnifera. Nat. Prod. Res. 2018, 32, 1573–1577. [Google Scholar] [CrossRef] [PubMed]
- Ghiasvand, M.; Makhdoumi, A.; Matin, M.M.; Vaezi, J. Exploring the Bioactive Compounds from Endophytic Bacteria of a Medicinal Plant: Ephedra Foliata (Ephedrales: Ephedraceae). Orient. Pharm. Exp. Med. 2019, 20, 61–70. [Google Scholar] [CrossRef]
- Soujanya, K.N.; Siva, R.; Mohana Kumara, P.; Srimany, A.; Ravikanth, G.; Mulani, F.A.; Aarthy, T.; Thulasiram, H.V.; Santhoshkumar, T.R.; Nataraja, K.N.; et al. Camptothecin-Producing Endophytic Bacteria from Pyrenacantha Volubilis Hook. (Icacinaceae): A Possible Role of a Plasmid in the Production of Camptothecin. Phytomedicine 2017, 36, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.S.; Liu, J.M.; Sun, J.; Huang, Y.T.; Jin, N.; Li, M.M.; Liang, Y.T.; Fan, B.; Wang, F.Z. Analysis of Endophytic Bacterial Diversity from Different Dendrobium Stems and Discovery of an Endophyte Produced Dendrobine-Type Sesquiterpenoid Alkaloids. Front. Microbiol. 2022, 12, 775665. [Google Scholar] [CrossRef] [PubMed]
- Jasim, B.; Geethu, P.R.; Mathew, J.; Radhakrishnan, E.K. Effect of Endophytic Bacillus Sp. from Selected Medicinal Plants on Growth Promotion and Diosgenin Production in Trigonella Foenum-Graecum. Plant Cell. Tissue Organ Cult. 2015, 122, 565–572. [Google Scholar] [CrossRef]
- Ptak, A.; Morańska, E.; Warchoł, M.; Gurgul, A.; Skrzypek, E.; Dziurka, M.; Laurain-Mattar, D.; Spina, R.; Jaglarz, A.; Simlat, M. Endophytic Bacteria from in Vitro Culture of Leucojum aestivum L. a New Source of Galanthamine and Elicitor of Alkaloid Biosynthesis. Sci. Rep. 2022, 12, 13700. [Google Scholar] [CrossRef]
- Taheri, S.; Mirjalili, M.; Farzaneh, M.; Rezadoost, H. Isolation and Characterization of Endophytic Galanthamin-Producing Bacteria from Narcissus Tazzeta L. Growing Wildly in Iran. In Proceedings of the Conference: 5th National Congress on Medicinal Plants, Isfahan, Iran, 18–19 May 2016. [Google Scholar]
- Yin, D.D.; Xu, F.; Wang, Y.L.; Yang, M.; Yin, D.K.; Wang, G.K. Analysis of Chuanxiong Rhizoma Substrate on Production of Ligustrazine in Endophytic Bacillus Subtilis by Ultra High Performance Liquid Chromatography with Quadrupole Time-of-Flight Mass Spectrometry. J. Sep. Sci. 2019, 42, 3067–3076. [Google Scholar] [CrossRef]
- Kusari, S.; Lamshöft, M.; Kusari, P.; Gottfried, S.; Zühlke, S.; Louven, K.; Hentschel, U.; Kayser, O.; Spiteller, M. Endophytes Are Hidden Producers of Maytansine in Putterlickia Roots. J. Nat. Prod. 2014, 77, 2577–2584. [Google Scholar] [CrossRef] [Green Version]
- Trapp, M.A.; Kai, M.; Mithöfer, A.; Rodrigues-Filho, E. Antibiotic Oxylipins from Alternanthera Brasiliana and Its Endophytic Bacteria. Phytochemistry 2015, 110, 72–82. [Google Scholar] [CrossRef]
- Caruso, M.; Colombo, A.L.; Fedeli, L.; Pavesi, A.; Quaroni, S.; Saracchi, M.; Ventrella, G. Isolation of Endophytic Fungi and Actinomycetes Taxane Producers. Ann. Microbiol. 2000, 50, 3–13. [Google Scholar]
- Anjum, N.; Chandra, R. Endophytic Bacteria of Catharanthus Roseus as an Alternative Source of Vindoline and Application of Response Surface Methodology to Enhance Its Production. Arch. Biol. Sci. 2019, 71, 27–38. [Google Scholar] [CrossRef]
- Zhai, X.; Jia, M.; Chen, L.; Zheng, C.J.; Rahman, K.; Han, T.; Qin, L.P. The Regulatory Mechanism of Fungal Elicitor-Induced Secondary Metabolite Biosynthesis in Medical Plants. Crit. Rev. Microbiol. 2017, 43, 238–261. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.; Thapa, S.; Mahawar, H.; Kumar, D.; Geat, N.; Singh, S.K. Prospecting Potential of Endophytes for Modulation of Biosynthesis of Therapeutic Bioactive Secondary Metabolites and Plant Growth Promotion of Medicinal and Aromatic Plants; Springer: Berlin/Heidelberg, Germany, 2022; Volume 115, ISBN 0123456789. [Google Scholar]
- Tiwari, R.; Awasthi, A.; Mall, M.; Shukla, A.K.; Srinivas, K.V.N.S.; Syamasundar, K.V.; Kalra, A. Bacterial Endophyte-Mediated Enhancement of in Planta Content of Key Terpenoid Indole Alkaloids and Growth Parameters of Catharanthus Roseus. Ind. Crops Prod. 2013, 43, 306–310. [Google Scholar] [CrossRef]
- Singh, S.; Pandey, S.S.; Tiwari, R.; Pandey, A.; Shanker, K.; Kalra, A. Endophytic Consortium with Growth-Promoting and Alkaloid Enhancing Capabilities Enhance Key Terpenoid Indole Alkaloids of Catharanthus Roseus in the Winter and Summer Seasons. Ind. Crops Prod. 2021, 166, 113437. [Google Scholar] [CrossRef]
- Ming, Q.; Su, C.; Zheng, C.; Jia, M.; Zhang, Q.; Zhang, H.; Rahman, K.; Han, T.; Qin, L. Elicitors from the Endophytic Fungus Trichoderma Atroviride Promote Salvia Miltiorrhiza Hairy Root Growth and Tanshinone Biosynthesis. J. Exp. Bot. 2013, 64, 5687–5694. [Google Scholar] [CrossRef] [Green Version]
- Heinig, U.; Scholz, S.; Jennewein, S. Getting to the Bottom of Taxol Biosynthesis by Fungi. Fungal Divers. 2013, 60, 161–170. [Google Scholar] [CrossRef] [Green Version]
- Kusari, S.; Spiteller, M. Are We Ready for Industrial Production of Bioactive Plant Secondary Metabolites Utilizing Endophytes? Nat. Prod. Rep. 2011, 28, 1203–1207. [Google Scholar] [CrossRef] [PubMed]
- Li, J.Y.; Sidhu, R.S.; Ford, E.J.; Long, D.M.; Hess, W.M.; Strobel, G.A. The Induction of Taxol Production in the Endophytic Fungus—Periconia sp. from Torreya Grandifolia. J. Ind. Microbiol. Biotechnol. 1998, 20, 259–264. [Google Scholar] [CrossRef]
- Vasanthakumari, M.M.; Jadhav, S.S.; Sachin, N.; Vinod, G.; Shweta, S.; Manjunatha, B.L.; Kumara, P.M.; Ravikanth, G.; Nataraja, K.N.; Uma Shaanker, R. Restoration of Camptothecine Production in Attenuated Endophytic Fungus on Re-Inoculation into Host Plant and Treatment with DNA Methyltransferase Inhibitor. World J. Microbiol. Biotechnol. 2015, 31, 1629–1639. [Google Scholar] [CrossRef]
- Kusari, S.; Zühlke, S.; Spiteller, M. Effect of Artificial Reconstitution of the Interaction between the Plant Camptotheca Acuminata and the Fungal Endophyte Fusarium Solani on Camptothecin Biosynthesis. J. Nat. Prod. 2011, 74, 764–775. [Google Scholar] [CrossRef]
- Vasundhara, M.; Kumar, A.; Reddy, M.S. Molecular Approaches to Screen Bioactive Compounds from Endophytic Fungi. Front. Microbiol. 2016, 7, 1774. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sachin, N.; Manjunatha, B.L.; Kumara, P.M.; Ravikanth, G.; Shweta, S.; Suryanarayanan, T.S.; Ganeshaiah, K.N.; Shaanker, R.U. Do Endophytic Fungi Possess Pathway Genes for Plant Secondary Metabolites? Curr. Sci. 2013, 104, 178–182. [Google Scholar]
- Deepika, V.B.; Murali, T.S.; Satyamoorthy, K. Modulation of Genetic Clusters for Synthesis of Bioactive Molecules in Fungal Endophytes: A Review. Microbiol. Res. 2016, 182, 125–140. [Google Scholar] [CrossRef]
- Harrison, E.; Brockhurst, M.A. Plasmid-Mediated Horizontal Gene Transfer Is a Coevolutionary Process. Trends Microbiol. 2012, 20, 262–267. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arora, P.; Riyaz-Ul-Hassan, S. Endohyphal Bacteria; the Prokaryotic Modulators of Host Fungal Biology. Fungal Biol. Rev. 2019, 33, 72–81. [Google Scholar] [CrossRef]
- Hoffman, M.T.; Arnold, A.E. Diverse Bacteria Inhabit Living Hyphae of Phylogenetically Diverse Fungal Endophytes. Appl. Environ. Microbiol. 2010, 76, 4063–4075. [Google Scholar] [CrossRef] [Green Version]
- MacDonald, R.M.; Chandler, M.R. Bacterium-like Organelles in the Vesicular-Arbuscular Mycorrhizal Fungus Glomus Caledonius. New Phytol. 1981, 89, 165–360. [Google Scholar] [CrossRef]
- Hoffman, M.T.; Gunatilaka, M.K.; Wijeratne, K.; Gunatilaka, L.; Arnold, A.E. Endohyphal Bacterium Enhances Production of Indole-3-Acetic Acid by a Foliar Fungal Endophyte. PLoS ONE 2013, 8, 31–33. [Google Scholar] [CrossRef]
- Shaffer, J.P.; Zalamea, P.C.; Sarmiento, C.; Gallery, R.E.; Dalling, J.W.; Davis, A.S.; Baltrus, D.A.; Arnold, A.E. Context-Dependent and Variable Effects of Endohyphal Bacteria on Interactions between Fungi and Seeds. Fungal Ecol. 2018, 36, 117–127. [Google Scholar] [CrossRef]
- Araldi-Brondolo, S.J.; Spraker, J.; Shaffer, J.P.; Woytenko, E.H.; Baltrus, D.A.; Gallery, R.E.; Arnold, A.E. Bacterial Endosymbionts: Master Modulators of Fungal Phenotypes. Fungal Kingd. 2017, 5, 981–1004. [Google Scholar] [CrossRef]
- Pakvaz, S.; Soltani, J. Endohyphal Bacteria from Fungal Endophytes of the Mediterranean Cypress (Cupressus sempervirens) Exhibit in Vitro Bioactivity. For. Pathol. 2016, 46, 569–581. [Google Scholar] [CrossRef]
- Shaffer, J.P.; Carter, M.E.; Spraker, J.E.; Clark, M.; Smith, B.A.; Baltrus, D.A.; Elizabeth, A. Transcriptional Profiles of a Foliar Fungal Endophyte Growth Regulation during Early Phases of Symbiotic Interaction. mSystems 2022, 7, e0009122. [Google Scholar] [CrossRef] [PubMed]
- Krings, M.; Taylor, T.N.; Dotzler, N. Fungal Endophytes as a Driving Force in Land Plant Evolution: Evidence from the Fossil Record. In Biocomplexity of Plant-Fungal Interactions; Southworth, D., Ed.; John Wiley & Sons, Ltd: Hoboken, NJ, USA, 2012; pp. 5–27. [Google Scholar] [CrossRef]
- Goldenfeld, N.; Woese, C. Biology’s next Revolution. Nature 2007, 445, 369. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dunning Hotopp, J.C. Horizontal Gene Transfer between Bacteria and Animals. Trends Genet. 2011, 27, 157–163. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, D. Pull in and Push out: Mechanisms of Horizontal Gene Transfer in Bacteria. Front. Microbiol. 2018, 9, 2154. [Google Scholar] [CrossRef] [PubMed]
- Sobecky, P.A.; Hazen, T.H. Horizontal Gene Transfer and Mobile Genetic Elements in Marine Systems. Methods Mol. Biol. 2009, 532, 435–453. [Google Scholar] [CrossRef]
- Ma, J.; Wang, S.; Zhu, X.; Sun, G.; Chang, G.; Li, L.; Hu, X.; Zhang, S.; Zhou, Y.; Song, C.P.; et al. Major Episodes of Horizontal Gene Transfer Drove the Evolution of Land Plants. Mol. Plant 2022, 15, 857–871. [Google Scholar] [CrossRef]
- Tiwari, P.; Bae, H. Horizontal Gene Transfer and Endophytes: An Implication for the Acquisition of Novel Traits. Plants 2020, 9, 305. [Google Scholar] [CrossRef] [Green Version]
- Taghavi, S.; Barac, T.; Greenberg, B.; Borremans, B.; Vangronsveld, J.; Van Der Lelie, D. Horizontal Gene Transfer to Endogenous Endophytic Bacteria from Poplar Improves Phytoremediation of Toluene. Appl. Environ. Microbiol. 2005, 71, 8500–8505. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Li, H.; Zhao, W.; He, X.; Chen, J.; Geng, X.; Xiao, M. Induction of Toluene Degradation and Growth Promotion in Corn and Wheat by Horizontal Gene Transfer within Endophytic Bacteria. Soil Biol. Biochem. 2010, 42, 1051–1057. [Google Scholar] [CrossRef]
- Zou, K.; Liu, X.; Hu, Q.; Zhang, D.; Fu, S.; Zhang, S.; Huang, H.; Lei, F.; Zhang, G.; Miao, B.; et al. Root Endophytes and Ginkgo Biloba Are Likely to Share and Compensate Secondary Metabolic Processes, and Potentially Exchange Genetic Information by LTR-RTs. Front. Plant Sci. 2021, 12, 704985. [Google Scholar] [CrossRef] [PubMed]
- Franco, M.E.E.; Wisecaver, J.H.; Arnold, A.E.; Ju, Y.M.; Slot, J.C.; Ahrendt, S.; Moore, L.P.; Eastman, K.E.; Scott, K.; Konkel, Z.; et al. Ecological Generalism Drives Hyperdiversity of Secondary Metabolite Gene Clusters in Xylarialean Endophytes; Wiley Publishing: Hoboken, NJ, USA, 2022; Volume 233, ISBN 0000000249591. [Google Scholar]
- Bielecka, M.; Pencakowski, B.; Nicoletti, R. Using Next-Generation Sequencing Technology to Explore Genetic Pathways in Endophytic Fungi in the Syntheses of Plant Bioactive Metabolites. Agriculture 2022, 12, 187. [Google Scholar] [CrossRef]
- Scott, K.; Konkel, Z.; Gluck-Thaler, E.; Valero David, G.E.; Farinas Simmt, C.; Grootmyers, D.; Chaverri, P.; Slot, J. Endophyte Genomes Support Greater Metabolic Gene Cluster Diversity Compared with Non-Endophytes in Trichoderma. bioRxiv 2023. [Google Scholar] [CrossRef]
- Howitz, K.T.; Sinclair, D.A. Xenohormesis: Sensing the Chemical Cues of Other Species. Cell 2008, 133, 387–391. [Google Scholar] [CrossRef] [Green Version]
- Truyens, S.; Weyens, N.; Cuypers, A.; Vangronsveld, J. Bacterial Seed Endophytes: Genera, Vertical Transmission and Interaction with Plants. Environ. Microbiol. Rep. 2015, 7, 40–50. [Google Scholar] [CrossRef]
- O’Brien, A.M.; Jack, C.N.; Friesen, M.L.; Frederickson, M.E. Whose Trait Is It Anyways? Coevolution of Joint Phenotypes and Genetic Architecture in Mutualisms. Proc. R. Soc. B Biol. Sci. 2021, 288, 20202483. [Google Scholar] [CrossRef]
- Tudzynski, B. Gibberellin Biosynthesis in Fungi: Genes, Enzymes, Evolution, and Impact on Biotechnology. Appl. Microbiol. Biotechnol. 2005, 66, 597–611. [Google Scholar] [CrossRef]
- Salazar-Cerezo, S.; Martínez-Montiel, N.; García-Sánchez, J.; Pérez-y-Terrón, R.; Martínez-Contreras, R.D. Gibberellin Biosynthesis and Metabolism: A Convergent Route for Plants, Fungi and Bacteria. Microbiol. Res. 2018, 208, 85–98. [Google Scholar] [CrossRef]
- Gupta, R.; Chakrabarty, S.K. Gibberellic Acid in Plant: Still a Mystery Unresolved. Plant Signal. Behav. 2013, 8, e25504. [Google Scholar] [CrossRef] [Green Version]
- Bömke, C.; Tudzynski, B. Diversity, Regulation, and Evolution of the Gibberellin Biosynthetic Pathway in Fungi Compared to Plants and Bacteria. Phytochemistry 2009, 70, 1876–1893. [Google Scholar] [CrossRef]
- Morrone, D.; Chambers, J.; Lowry, L.; Kim, G.; Anterola, A.; Bender, K.; Peters, R.J. Gibberellin Biosynthesis in Bacteria: Separate Ent-Copalyl Diphosphate and Ent-Kaurene Synthases in Bradyrhizobium Japonicum. FEBS Lett. 2009, 583, 475–480. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hedden, P. The Current Status of Research on Gibberellin Biosynthesis. Plant Cell Physiol. 2020, 61, 1832–1849. [Google Scholar] [CrossRef]
- Zhang, P.; Zhou, P.P.; Yu, L.J. An Endophytic Taxol-Producing Fungus from Taxus Media, Cladosporium Cladosporioides MD2. Curr. Microbiol. 2009, 59, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Z.Q.; Yang, Y.Y.; Zhao, N.; Wang, Y. Diversity of Endophytic Fungi and Screening of Fungal Paclitaxel Producer from Anglojap Yew, Taxus x Media. BMC Microbiol. 2013, 13, 71. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.; Zhao, H.; Barrero, R.A.; Zhang, B.; Sun, G.; Wilson, I.W.; Xie, F.; Walker, K.D.; Parks, J.W.; Bruce, R.; et al. Genome Sequencing and Analysis of the Paclitaxel-Producing Endophytic Fungus Penicillium Aurantiogriseum NRRL 62431. BMC Genom. 2014, 15, 69. [Google Scholar] [CrossRef] [Green Version]
- Sah, B.; Subban, K.; Chelliah, J. Cloning and Sequence Analysis of 10-Deacetylbaccatin III-10-O-Acetyl Transferase Gene and WRKY1 Transcription Factor from Taxol-Producing Endophytic Fungus Lasiodiplodia Theobromea. FEMS Microbiol. Lett. 2017, 364, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Miao, L.Y.; Mo, X.C.; Xi, X.Y.; Zhou, L.; De, G.; Ke, Y.S.; Liu, P.; Song, F.J.; Jin, W.W.; Zhang, P. Transcriptome Analysis of a Taxol-Producing Endophytic Fungus Cladosporium Cladosporioides MD2. AMB Express 2018, 8, 41. [Google Scholar] [CrossRef] [Green Version]
- Qiao, W.; Tang, T.; Ling, F. Comparative Transcriptome Analysis of a Taxol-Producing Endophytic Fungus, Aspergillus Aculeatinus Tax-6, and Its Mutant Strain. Sci. Rep. 2020, 10, 10558. [Google Scholar] [CrossRef] [PubMed]
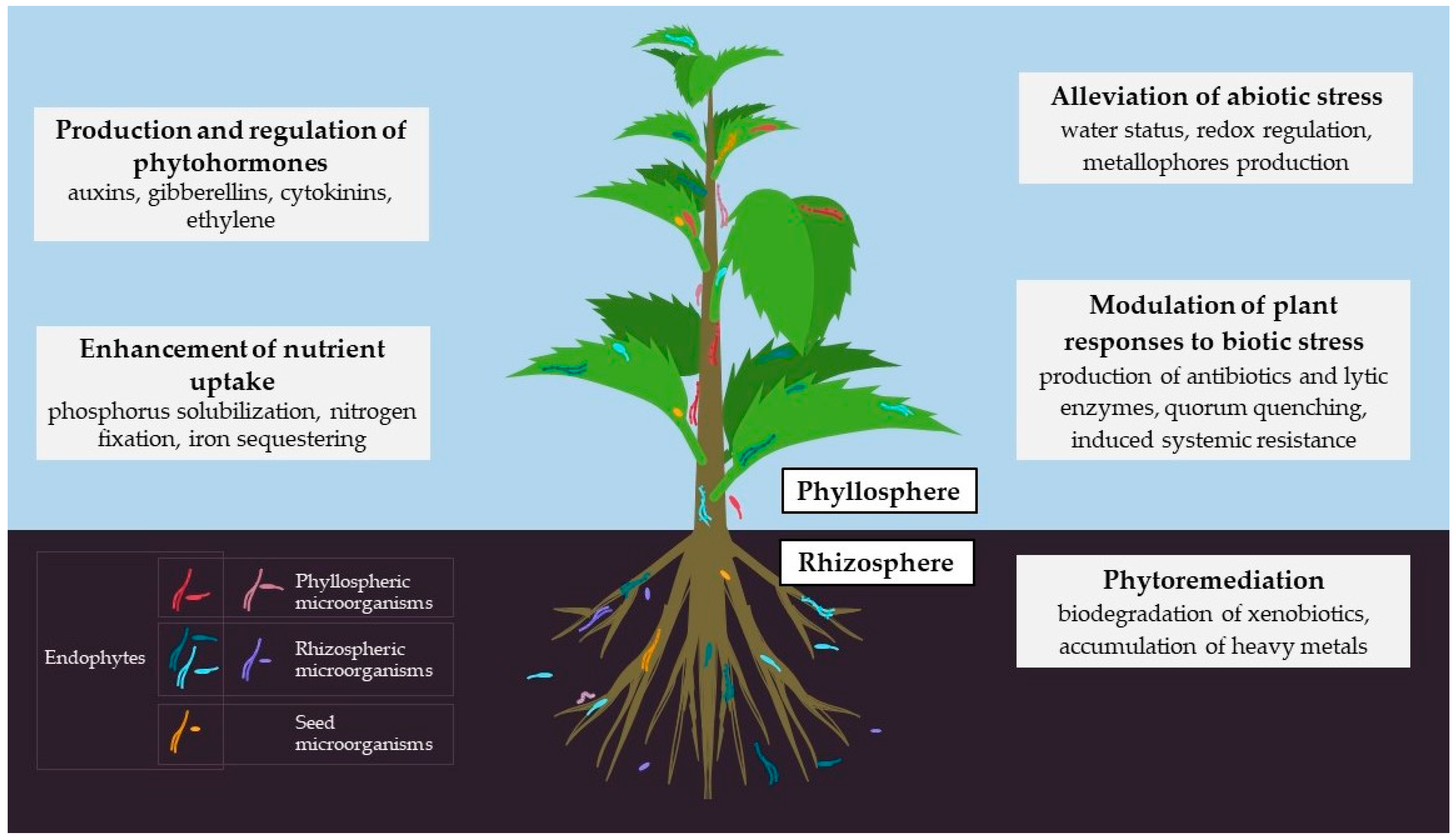
| Microorganisms | Living Conditions/Specifics |
|---|---|
| Rhizospheric | soil in close proximity to roots and its exudates |
| Rhizoplanic | surface of plant roots |
| Phyllospheric (=epiphytes) | surface of aerial parts of a plant |
| Endophytic (=endophytes) | inside plant tissues without causing any apparent harm to plant host |
| Obligate | living inside a plant during their entire lifespan |
| Facultative | optionally living inside of a plant |
| Systemic (true) | strictly symbiotic, non-pathogenic at any stage of its lifespan |
| Non-systemic (transient) | live asymptomatically within plant for a part of their lifespan, can turn into pathogen when plant host is stressed or resource-limited |
| Competent | successfully colonize plant, can alter its physiology and be selectively favored |
| Opportunistic | occasionally enter a plant and benefit from its internal environment |
| Passenger | enter a plant accidently in the absence of selective forces for efficient root colonization |
| Compound | Properties | Endophyte | Plant Host | Amount Produced by Endophyte Ex Planta | Reference |
|---|---|---|---|---|---|
| Fungi | |||||
| Aconitine | Analgesic, anti-inflammatory, anti-tumor | Cladosporium cladosporioides XJ-A C03 | Aconitum leucostomum | 236.4 µg/g 1 | [108] |
| Alternariol | Cytotoxic | Alternaria sp. | Polygonum senegalense | ND | [109] |
| Andrographolide | Anti-inflammatory, antineoplastic, anti-platelet aggregation | Colletotrichum sp. (AP-4, AP-12) | Andrographis paniculata | 30.089 ± 0.992 mg/g 1 (AP-4), 28.617 ± 0.641 mg/g 1 (AP-12) | [110] |
| Asarone | Anticonvulsant, GABA modulator | Penicillium pinophilum | Alloteropsis cimicina | ND | [111] |
| Asiaticoside | Antioxidant, anti-inflammatory, antirheumatic | Colletotrichum gloeosporioides | Centella asiactica | 62.29 ± 3.36 µg/100 mL 2 | [112] |
| Azadirachtin | Hepatoprotective, insecticide | Eupenicillium parvum | Azadirachta indica | 0.4 µg/100 g 1; 43 µg/L 2 | [113] |
| Cajaninstilbene acid | Analgesic, antioxidant, anti-inflammatory, hypoglycemic, neuroprotective | Fusarium proliferatum | Cajanus cajan | 100.5 ± 9.4 µg/g 1; 504.8 ± 20.1 µg/mL 2 | [114] |
| Camptothecin | Anticancer (inhibition of DNA topoisomerase I), potential antineoplastic agent | Fusarium solani (MTCC 9667, 9668) | Apodytes dimidiata | 37 µg/100 g 1; 53 µg/100 g 1 | [86] |
| Fusarium solani | Camptotheca acuminata | 150 ± 20 µg/L 2 | [88] | ||
| Alternaria alstroemeriae (NCIM1408) | Nothapodytes nimmoniana | 426.7 ± 33.6 µg/g 1 | [115] | ||
| Alternaria burnsii (NCIM1409) | 403.3 ± 41.6 µg/g 1 | ||||
| Fusarium sp. | 2.17 µg/100 mg 1 | [89] | |||
| Capsaicin | Analgesic | Alternaria alternata | Capsicum annuum | 8.30 µg/L 2 | [116] |
| Colchicine | Anti-gout, anti-inflammatory | Diaporthe perseae | Glorosa superba | 55.25 uµg/g 1 | [117] |
| Deoxypodophyllotoxin | Precursor for podophyllotoxin, anticancer | Aspergillus fumigatus | Juniperus communis | 4 ± 2 µg/100 g 1; 3 ± 2 µg/L 2 | [118] |
| Dendrobine | Anti-cataract, anti-influenza A virus, and anti-tumor, promising therapeutical effects on Alzheimer’s disease | Trichoderma longibrachiatum MD33 | Dendrobium nobile | ND | [119] |
| Digoxin | Anti-arrhythmia drug, cardiotonic drug | Unidentified | Digitalis lanata | ND | [120] |
| Diosgenin | Anticancer, antiatherogenic, antineoplastic, and antiviral agent | Aspergillus flavus | Dioscorea zingiberensis | ND | [121] |
| Curvularia lunata | |||||
| Fusarium sp. | |||||
| Paecilomyces sp. | Paris podophylla | ND | [122] | ||
| Emodin | Antioxidant, anti-inflammatory, antimicrobial, hepatoprotective, precursor to hypericin | Epicoccum nigrum | Hypericum perforatum | 87.7 µg/mL 3 | [123] |
| Thielavia subthermophilia | 113 ± 1 µg/100 g 1 | [124] | |||
| Forskolin | Antihypertensive, anti-HIV, platelet aggregation inhibitor | Rhizactonia bataticola | Coleus forskoholii | 0.5 mg 2 | [125] |
| Gentiopicrin | Antifungal, antihistamine, anti-inflammatory | Unidentified (QJ18) | Gentiana macrophylla | ND | [126] |
| Ginsenoside Rg3 | Antioxidant, antidiabetic, antineoplastic agent, apoptosis inducer | Chaetomium sp. | Panax ginseng | 5.60 ± 1.17 mg/g 1 | [92] |
| Gymnemagenin | Antidiabetic, antiviral | Penicillium oxalicum | Gymnema sylvestre | ND | [127] |
| Huperzine A | Neuroprotective, treatment of Alzheimer’s disease | Acremonium sp. | Huperzia serrata | 8.32 µg/L 2 | [128] |
| Shiraia sp. Slf14 | 327.8 µg/L 2 | [129] | |||
| Hypericin | Antidepressant, anti-inflammatory, antineoplastic, immunostimulating | Epicoccum nigrum | Hypericum perforatum | 117.1 µg/mL 3 | [123] |
| Thielavia subthermophilia | 35 ± 2 µg/100 g 1 | [124,130] | |||
| Imperialine-3β-D-glucoside | Anti-tumor | Fusarium redolens 6WBY3 | Fritillaria unibracteata | 18.8 µg/L 2 | [131] |
| Loline alkaloids | Insecticidal | Neotyphodium uncinatum | Lolium pratense | 700 mg/L 2 | [132] |
| Kaempferol | Antioxidant, antibacterial | Mucor fragilis | Sinopodophyllum hexandrum | ND | [96] |
| Myrtucommulones | Anti-oxidant, anti-inflammatory, anti-tumor | Neofusicoccum australe | Myrtus communis | 0.2 mg/L 2 | [133] |
| Nigranoic acid | Anti-neoplastic agent, HIV-1 reverse transcriptase inhibitor | Umbelopsis dimorphia | Kadsura angustifolia | ND | [134] |
| Peiminine | Anticancer, anti-inflammatory | Fusarium sp. | Fritillaria unibracteata | 0.021 mg/L 2; 0.0054 mg/g 1 | [135] |
| Peimisine | Anticancer, anti-inflammatory, angiotensin-converting enzyme (ACE) inhibitor | Fusarium sp. | Fritillaria unibracteata | 0.09 mg/L2; 0.0023 mg/g 1 | [135] |
| Fusarium redolens 6WBY3 | 16.0 µg/L 2 | [131] | |||
| Piperine | Antioxidant, anti-inflammatory, antimycobacterial, insecticidial, increasing bioavailability of drugs | Colletotrichum gloeosporioides | Piper nigrum | ND | [136] |
| Periconia sp. | Piper longum | 750 mg 3 | [137] | ||
| Podophyllotoxin (PTOX) | Antimitotic, anti-tumor, precursor for anticancer drugs, e.g., etoposide and teniposide | Fusarium sp. (WB5121) | Dysosma versipellis | 277 µg/g 1 | [97] |
| Fusarium oxysporum | Juniper recurva | 28 µg/g 1 | [98] | ||
| Fusarium solani | Podophyllum hexandrum | 29.16 ± 0.57 µg/g 1 | [95] | ||
| Phialocephaa fortinii | Podophyllum peltatum | 189 µg/L 2 | [99] | ||
| Trametes hirsuta | Podophyllum hexandrum | 31 µg/g 1 | [91] | ||
| Mucor fragilis | Sinopodophyllum hexandrum | 49.3 µg/g 1 | [96] | ||
| Quercetin monoglycosides | Antioxidant, stimulating bacterial enzymatic activity | Nigrospora oryzae | Loranthus micranthus | ND | [138] |
| Quinidine | Antiarrhythmic, antimalarial | Diaporthe spp. (CLS-3) | Cinchona ledgeriana | 82.5 µg/L 2 | [139] |
| Resveratrol | Antioxidant, anti-inflammatory, antimutagen, antiviral, phytoestrogenic | Alternaria sp. MG1 | Vitis vinifera | 353 µg/L 2 | [103] |
| Arcopilus aureus | 89.1 µg/mL 2 | [101] | |||
| Botryosphaeria sp. | 37.3 µg/mL 2 | ||||
| Nigrospora sp. | 25.2 µg/mL 2 | ||||
| Aspergillus stellifer AB4 | 300 µg/L 2 | [104] | |||
| Fusarium equiseti | 52.3 µg/mL 2 | [100] | |||
| Quambalaria cyanescens | 40 mg/L 2 | [102] | |||
| Rhein | Anticancer, anti-inflammatory, antimicrobial, hemostatic | Fusarium solani | Rheum palmatum | 5.672 mg/L 2 | [140] |
| Saikosaponin d | Anti-inflammatory, anti-tumor, immunomodulatory | Fusarium acuminatum | Bupleurum scorzonerifolium | 2.40 µg/mL 2 | [141] |
| Fusarium oxysporum | 2.17 µg/mL 2 | ||||
| Salidroside | Adaptogenic, antioxidant, antidepressant, anti-inflammatory, neuroprotective | Phialocephala fortinii (Rac56) | Rhodiola angusta | 2.339 ± 0.1093 mg/mL 2 | [142] |
| Salvianolic acid C | Anticancer, antioxidant, treatment of cardiovascular and cerebrovascular diseases | Phoma glomerata D14 | Salvia miliorrhiza | 0.054 µg/mL 2; 47.67 ± 0.04 µg/g 3 | [143] |
| Saponin | Anti-inflammatory, antimicrobial, anti-ulcer, haemolytic, hepatoprotective | Fusarium sp. (Pg27) | Panax ginseng | 0.181 mg/mL 2 | [144] |
| Silybin A and B, isosilybin A | Anti-tumor, hepatoprotective | Aspergillus iizukae | Silybum marianum | 0.13–0.22 µg/g 3 | [145] |
| Sipeimine | Anti-tumor, antitussive | Unidentified (Fu7) | Fritillaria ussuriensis | ND | [146] |
| Solamargine | Antidiabetogenic, cytotoxic | Aspergillus flavus | Solanum nigrum | 250–300 µg/L2 | [147] |
| Tanshinone I and IIA | Anti-inflammatory, anticoronaviral, anticancer | Trichoderma atroviride D16 | Salvia miliorrhiza | 1.119 ± 0.008 µg/g 1 (I); 3.049 ± 0.001 µg/g 1 (IIA) | [148] |
| Taxol (paclitaxel) | Chemotherapy drug | Annulohypoxylon sp. MUS1 | Taxus wallichiana | 282.05 µg/L 2 | [149] |
| Aspergillus fumigatus | Taxus sp. | 1.60 g/L 2 | [150] | ||
| Seimatoantlerium nepalense | Taxus wallichiana | ND | [151] | ||
| Seimatoantlerium tepuisense | Venezuelan guyana | 250–350 ng/L 2 | [152] | ||
| Taxomyces andreanae | Taxus brevifolia | 24–50 ng/L 2 | [77] | ||
| Tyrosol | Antioxidant, cardioprotective | Phialocephala fortinii (Rac56) | Rhodiola angusta | 2.002 ± 0.0009 mg/mL 2 | [142] |
| Vinblastine | Antineoplastic, anti-tumor | Curvularia verruculosa | Catharanthus roseus | 182 µg/L 2 | [105] |
| Fusarium oxysporum | 76 µg/L 2 | [25] | |||
| Fusarium solani | ND | [107] | |||
| Talaromyces radicus | 70 µg/L 2 | [153] | |||
| Vincamine | Antihypertensive, vasodilator | Geomyces sp. | Nerium indicum | 1.279 mg/L 2 | [154] |
| Unidentified (Vm-J2) | Vinca minor | 0.1 mg/L 2 | [155] | ||
| Vincristine | Chemotherapy drug | Eutypella sp. | Catharanthus roseus | 53 ± 5.0 µg/L 2 | [106] |
| Fusarium oxysporum | 67 µg/L 2 | [25] | |||
| Fusarium solani | ND | [107] | |||
| Viniferin | Anti-inflammatory, anticancer, anti-angiogenic, antimicrobial, anthelminthic | Aspergillus stellifer AB4 | Vitis vinifera | 324 µg/L 2 | [104] |
| Withanolide | Antioxidant, anti-inflammatory, antistress, cardioprotective, neuroprotective | Taleromyces pinophilus | Withania somnifera | 360 mg/L 2 | [156] |
| Bacteria | |||||
| Achillin | Anti-inflammatory, antihypertensive, vasorelaxant | Microbacterium maritypicum | Ephedra foliata | ND | [157] |
| Berberine | Analgesic, anti-inflammatory, antimicrobial, hypolipidemic | Kytococcus schroeteri | Ephedra foliata | ND | [157] |
| Paenibacillus polymyxa | |||||
| Camptothecin | Anticancer (inhibition of DNA topoisomerase I), potential antineoplastic agent | Bacillus cereus ChST | Miquelia dentata | 1.177 µg/mL 2 | [90] |
| Bacillus subtilis PXJ-5 | 1.554 µg/mL 2 | ||||
| Bacillus subtilis | Pyrenacantha angustifolia | 0.18 µg/mL 2 | [158] | ||
| Kytococcus schroeteri | Ephedra foliata | ND | [157] | ||
| Daunorubicin | Antibiotic, antineoplastic, anticancer | Paenibacillus polymyxa | Ephedra foliata | ND | [157] |
| Dendrobine analogs | Anti-cataract, and anti-influenza A virus, anti-tumor, promising therapeutical effects for Alzheimer’s disease | Pseudomonas proteogens CHA0 | Dendrobium sp. | ND | [159] |
| Diosgenin | Anticancer, antiatherogenic, antineoplastic, and antiviral agent | Bacillus sp. | Trigonella foenum-graceum | 527.83 µg/L 2 | [160] |
| Bacillus cereus | 156.33 µg/L 2 | ||||
| Galanthamine | Treatment of Alzheimer’s disease | Paenibacillus lautus | Leucojum aestivum | 37.51 µg/g 1 | [161] |
| Burkholderia graminis | Narcissus tazetta | 37.8 µg/L 2 | [162] | ||
| Bacillus thuringiensis | 61.0 µg/L 2 | ||||
| Ginsenoside Rg3 | Antioxidant, antidiabetic, antineoplastic agent, apoptosis inducer | Burkholderia sp. | Panax ginseng | ND | [93] |
| Agrobacterium sp. (PDA-2) | 62.20 mg/L 2 | [94] | |||
| Harmine | Anti-inflammatory, anti-tumor, hallucinogen | Microbacterium maritypicum | Ephedra foliata | ND | [157] |
| Ligustrazine | Anti-inflammatory, nootropic | Bacillus subtilis | Ligusticum chuanxiong | 1.0268 mg/L 2 | [163] |
| Lycorine | Anticancer | Paenibacillus lautus | Leucojum aestivum | 37.51 µg/g 1 | [163] |
| Maytansine | Antimicrobial, antineoplastic | Unidentified | Putterlickia retrospinosa | ND | [164] |
| Putterlickia verrucosa | |||||
| Myricetin | Antioxidant, anti-inflammatory, antithrombotic, antidiabetic, neuroprotective | Microbacterium maritypicum | Ephedra foliata | ND | [157] |
| Oxylipins | Antibiotic | Bacillus sp. | Alternanthera brasiliana | ND | [165] |
| Taxol (paclitaxel) | Chemotherapy drug | Kitasatospora sp. | Taxus baccata | ND | [166] |
| Sanguinarine | Antibiotic, anticancer | Paenibacillus polymyxa | Ephedra foliata | ND | [157] |
| Vindoline | Antimitotic, precursor for vinblastine | Microbacterium sp. | Catharanthus roseus | 82 ug/L 2 | [167] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rutkowska, N.; Drożdżyński, P.; Ryngajłło, M.; Marchut-Mikołajczyk, O. Plants as the Extended Phenotype of Endophytes—The Actual Source of Bioactive Compounds. Int. J. Mol. Sci. 2023, 24, 10096. https://doi.org/10.3390/ijms241210096
Rutkowska N, Drożdżyński P, Ryngajłło M, Marchut-Mikołajczyk O. Plants as the Extended Phenotype of Endophytes—The Actual Source of Bioactive Compounds. International Journal of Molecular Sciences. 2023; 24(12):10096. https://doi.org/10.3390/ijms241210096
Chicago/Turabian StyleRutkowska, Natalia, Piotr Drożdżyński, Małgorzata Ryngajłło, and Olga Marchut-Mikołajczyk. 2023. "Plants as the Extended Phenotype of Endophytes—The Actual Source of Bioactive Compounds" International Journal of Molecular Sciences 24, no. 12: 10096. https://doi.org/10.3390/ijms241210096






