Pharmacotechnical, Physico-Chemical, and Antioxidant Evaluation of Newly Developed Capsule Formulations
Abstract
:1. Introduction
2. Results
2.1. Physical and Chemical Evaluation
2.1.1. FTIR Analysis
2.1.2. XRD Analysis
2.1.3. Thermogravimetric Analysis
2.1.4. SEM Analysis
2.2. Preformulation and Formulation Studies of the Capsules
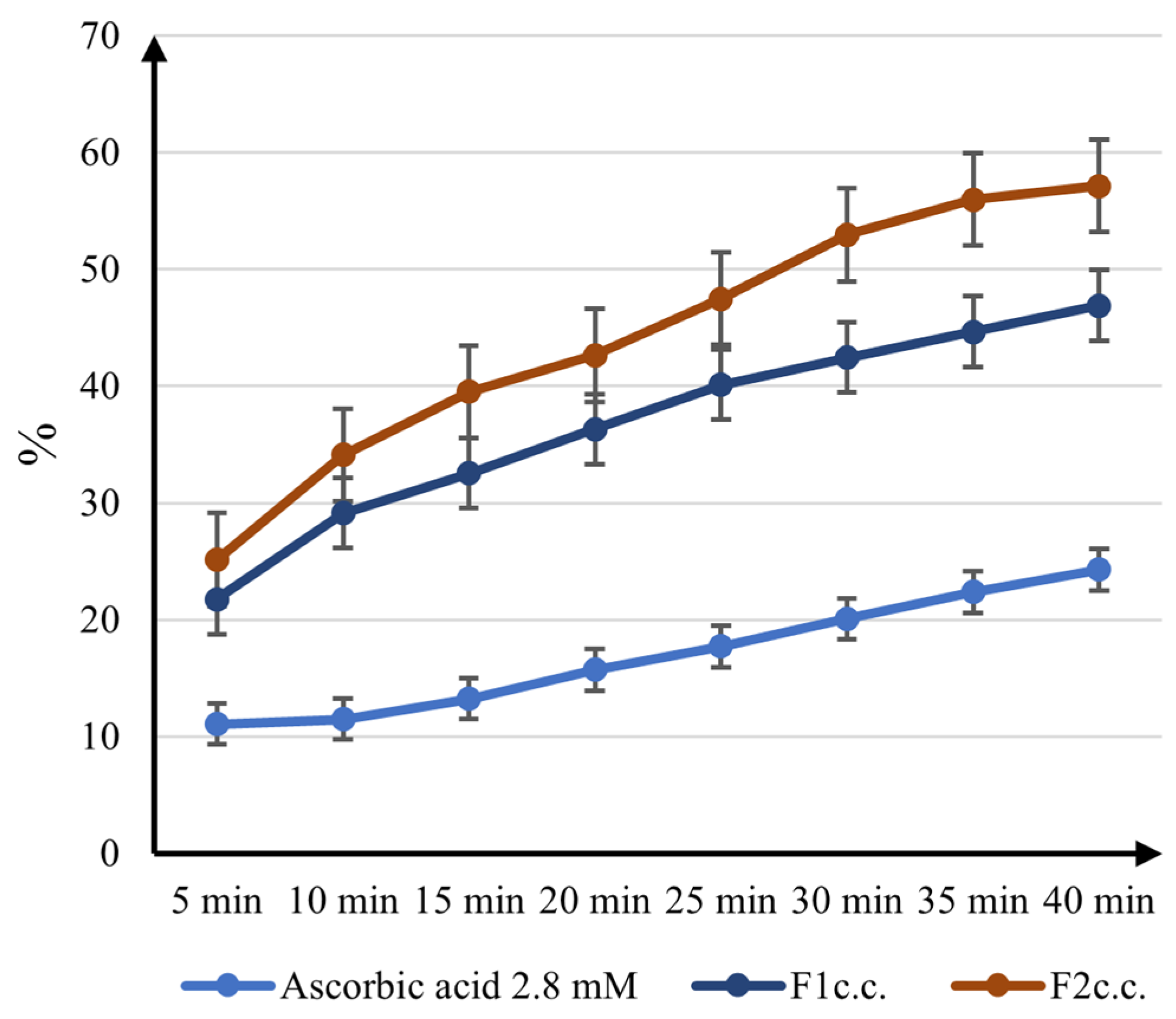
2.3. Capsules Content Antioxidant Activity
2.3.1. DPPH Radical Scavenging Activity
2.3.2. ABTS Radical Scavenging Activity
2.3.3. FRAP Assay
2.3.4. Capsules Quality Control
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Methods
4.2.1. Preformulation Studies of the Capsules
4.2.2. Formulation and Preparation of the Filling Materials for Capsules
4.2.3. Physical and Chemical Characterization
4.2.4. Capsules Content Properties
ABTS Radical Scavenging Activity
DPPH Radical Scavenging Activity
FRAP Assay
4.2.5. Final Capsules Manufacturing and Quality Control
Mass Uniformity
In Vitro Disintegration Time
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chiozzi, V.; Agriopoulou, S.; Varzakas, T. Advances, Applications, and Comparison of Thermal (Pasteurization, Sterilization, and Aseptic Packaging) against Non-Thermal (Ultrasounds, UV Radiation, Ozonation, High Hydrostatic Pressure) Technologies in Food Processing. Appl. Sci. 2022, 12, 2202. [Google Scholar] [CrossRef]
- Ziani, K.; Ioniță-Mîndrican, C.-B.; Mititelu, M.; Neacșu, S.M.; Negrei, C.; Moroșan, E.; Drăgănescu, D.; Preda, O.-T. Microplastics: A Real Global Threat for Environment and Food Safety: A State of the Art Review. Nutrients 2023, 15, 617. [Google Scholar] [CrossRef]
- Simioni, C.; Zauli, G.; Martelli, A.M.; Vitale, M.; Sacchetti, G.; Gonelli, A.; Neri, L.M. Oxidative stress: Role of physical exercise and antioxidant nutraceuticals in adulthood and aging. Oncotarget 2018, 9, 17181–17198. [Google Scholar] [CrossRef] [Green Version]
- Chaudhary, P.; Janmeda, P.; Docea, A.O.; Yeskaliyeva, B.; Abdull Razis, A.F.; Modu, B.; Calina, D.; Sharifi-Rad, J. Oxidative stress, free radicals and antioxidants: Potential crosstalk in the pathophysiology of human diseases. Front. Chem. 2023, 11, 1158198. [Google Scholar] [CrossRef] [PubMed]
- Saha, S.K.; Lee, S.B.; Won, J.; Choi, H.Y.; Kim, K.; Yang, G.-M.; Dayem, A.A.; Cho, S.-G. Correlation between Oxidative Stress, Nutrition, and Cancer Initiation. Int. J. Mol. Sci. 2017, 18, 1544. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Strath, L.J.; Sorge, R.E. Racial Differences in Pain, Nutrition, and Oxidative Stress. Pain Ther. 2022, 11, 37–56. [Google Scholar] [CrossRef] [PubMed]
- Pizzino, G.; Irrera, N.; Cucinotta, M.; Pallio, G.; Mannino, F.; Arcoraci, V.; Squadrito, F.; Altavilla, D.; Bitto, A. Oxidative Stress: Harms and Benefits for Human Health. Oxidative Med. Cell Longev. 2017, 2017, 8416763. [Google Scholar] [CrossRef] [Green Version]
- Juan, C.A.; Pérez de la Lastra, J.M.; Plou, F.J.; Pérez-Lebeña, E. The Chemistry of Reactive Oxygen Species (ROS) Revisited: Outlining Their Role in Biological Macromolecules (DNA, Lipids and Proteins) and Induced Pathologies. Int. J. Mol. Sci. 2021, 22, 4642. [Google Scholar] [CrossRef]
- Pilipović, K.; Jurišić Grubešić, R.; Dolenec, P.; Kučić, N.; Juretić, L.; Mršić-Pelčić, J. Plant-Based Antioxidants for Prevention and Treatment of Neurodegenerative Diseases: Phytotherapeutic Potential of Laurus nobilis, Aronia melanocarpa, and Celastrol. Antioxidants 2023, 12, 746. [Google Scholar] [CrossRef]
- Ponnampalam, E.N.; Kiani, A.; Santhiravel, S.; Holman, B.W.B.; Lauridsen, C.; Dunshea, F.R. The Importance of Dietary Antioxidants on Oxidative Stress, Meat and Milk Production, and Their Preservative Aspects in Farm Animals: Antioxidant Action, Animal Health, and Product Quality—Invited Review. Animals 2022, 12, 3279. [Google Scholar] [CrossRef]
- Morosan, E.; Mititelu, M.; Drăgănescu, D.; Lupuliasa, D.; Ozon, E.A.; Karampelas, O.; Gîrd, C.E.; Aramă, C.; Hovanet, M.V.; Musuc, A.M.; et al. Investigation into the Antioxidant Activity of Standardized Plant Extracts with Pharmaceutical Potential. Appl. Sci. 2021, 11, 8685. [Google Scholar] [CrossRef]
- Morosan, E.; Secăreanu, A.A.; Musuc, A.M.; Mititelu, M.; Ioniță, A.C.; Ozon, E.A.; Dărăban, A.M.; Karampelas, O. Advances on the Antioxidant Activity of a Phytocomplex Product Containing Berry Extracts from Romanian Spontaneous Flora. Processes 2022, 10, 646. [Google Scholar] [CrossRef]
- Ioniță-Mîndrican, C.-B.; Ziani, K.; Mititelu, M.; Oprea, E.; Neacșu, S.M.; Moroșan, E.; Dumitrescu, D.-E.; Roșca, A.C.; Drăgănescu, D.; Negrei, C. Therapeutic Benefits and Dietary Restrictions of Fiber Intake: A State of the Art Review. Nutrients 2022, 14, 2641. [Google Scholar] [CrossRef]
- Mititelu, M.; Udeanu, D.I.; Nedelescu, M.; Neacsu, S.M.; Nicoara, A.C.; Oprea, E.; Ghica, M. Quality Control of Different Types of Honey and Propolis Collected from Romanian Accredited Bee-Keepers and Consumer’s Risk Assessment. Crystals 2022, 12, 87. [Google Scholar] [CrossRef]
- Mititelu, M.; Moroșan, E.; Nicoară, A.C.; Secăreanu, A.A.; Musuc, A.M.; Atkinson, I.; Cusu, J.P.; Nițulescu, G.M.; Ozon, E.A.; Sarbu, I.; et al. Development of immediate release tablets containing calcium lactate synthetized from Black Sea mussel shells. Mar. Drugs 2022, 20, 45. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Ren, Z.; Zhang, J.; Chuang, C.C.; Kandaswamy, E.; Zhou, T.; Zuo, L. Role of ROS and Nutritional Antioxidants in Human Diseases. Front. Physiol. 2018, 9, 477. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anand, S.; Bharadvaja, N. Potential Benefits of Nutraceuticals for Oxidative Stress Management. Rev. Bras. Farmacogn. 2022, 32, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Meulmeester, F.L.; Luo, J.; Martens, L.G.; Mills, K.; van Heemst, D.; Noordam, R. Antioxidant Supplementation in Oxidative Stress-Related Diseases: What Have We Learned from Studies on Alpha-Tocopherol? Antioxidants 2022, 11, 2322. [Google Scholar] [CrossRef]
- Năstăsescu, V.; Mititelu, M.; Stanciu, T.I.; Drăgănescu, D.; Grigore, N.D.; Udeanu, D.I.; Stanciu, G.; Neacșu, S.M.; Dinu-Pîrvu, C.E.; Oprea, E.; et al. Food Habits and Lifestyle of Romanians in the Context of the COVID-19 Pandemic. Nutrients 2022, 14, 504. [Google Scholar] [CrossRef]
- Tarocco, A.; Caroccia, N.; Morciano, G.; Pinton, P. Melatonin as a master regulator of cell death and inflammation: Molecular mechanisms and clinical implications for newborn care. Cell Death Dis. 2019, 10, 317. [Google Scholar] [CrossRef] [Green Version]
- Muñoz-Jurado, A.; Escribano, B.M.; Caballero-Villarraso, J.; Galván, A.; Agüera, E.; Santamaría, A.; Túnez, I. Melatonin and multiple sclerosis: Antioxidant, anti-inflammatory and immunomodulator mechanism of action. Inflammopharmacology 2022, 30, 1569–1596. [Google Scholar] [CrossRef] [PubMed]
- Zozina, V.I.; Covantev, S.; Goroshko, O.A.; Krasnykh, L.M.; Kukes, V.G. Coenzyme Q10 in Cardiovascular and Metabolic Diseases: Current State of the Problem. Curr. Cardiol. Rev. 2018, 14, 164–174. [Google Scholar] [CrossRef] [PubMed]
- Manzar, H.; Abdulhussein, D.; Yap, T.E.; Cordeiro, M.F. Cellular Consequences of Coenzyme Q10 Deficiency in Neurodegeneration of the Retina and Brain. Int. J. Mol. Sci. 2020, 21, 9299. [Google Scholar] [CrossRef]
- Al-Zharani, M.; Mubarak, M.; Rudayni, H.A.; Al-Doaiss, A.A.; Abd-Elwahab, M.M.; Al-Eissa, M.S. Quercetin as a Dietary Supplementary Flavonoid Alleviates the Oxidative Stress Induced by Lead Toxicity in Male Wistar Rats. Nutrients 2023, 15, 1888. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Qian, M.; Jiang, Q.; Tan, B.; Yin, Y.; Han, X. Evidence of Flavonoids on Disease Prevention. Antioxidants 2023, 12, 527. [Google Scholar] [CrossRef]
- Rudrapal, M.; Khairnar, S.J.; Khan, J.; Dukhyil, A.B.; Ansari, M.A.; Alomary, M.N.; Alshabrmi, F.M.; Palai, S.; Deb, P.K.; Devi, R. Dietary Polyphenols and Their Role in Oxidative Stress-Induced Human Diseases: Insights into Protective Effects, Antioxidant Potentials and Mechanism(s) of Action. Front. Pharmacol. 2022, 13, 806470. [Google Scholar] [CrossRef]
- Koushki, M.; Amiri-Dashatan, N.; Ahmadi, N.; Abbaszadeh, H.A.; Rezaei-Tavirani, M. Resveratrol: A miraculous natural compound for diseases treatment. Food Sci. Nutr. 2018, 6, 2473–2490. [Google Scholar] [CrossRef] [Green Version]
- Fernandez-Mejia, C. Pharmacological effects of biotin. J. Nutr. Biochem. 2005, 16, 424–427. [Google Scholar] [CrossRef]
- Aguilera-Mendez, A.; Hernandez-Equihua, M.G.; Rueda-Rocha, A.C.; Guajardo-Lopez, C.; Nieto-Aguilar, R.; Serrato-Ochoa, D.; Ruiz Herrera, L.F.; Guzman-Nateras, J.A. Protective effect of supplementation with biotin against high-fructose-induced metabolic syndrome in rats. Nutr. Res. 2018, 57, 86–96. [Google Scholar] [CrossRef]
- Topal, B.; Altındal, D.Ç.; Gümüşderelioğlu, M. Melatonin/HPbCD complex: Microwave synthesis, integration with chitosan scaffolds and inhibitory effects on MG-63CELLS. Int. J. Pharm. 2015, 496, 801–811. [Google Scholar] [CrossRef]
- Luo, Q.; Wang, Y.; Han, Q.Q.; Ji, L.S.; Zhang, H.M.; Fei, Z.H.; Wang, Y.Q. Comparison of the physicochemical, rheological, and morphologic properties of chitosan from four insects. Carbohydr. Polym. 2019, 209, 266–275. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.Q.; Pitto-Barry, A.; Habtemariam, A.; Romero-Canelon, I.; Sadler, P.J.; Barry, N.P. Nanoparticles of chitosan conjugated to organo-ruthenium complexes. Inorg. Chem. Front. 2016, 3, 1058–1064. [Google Scholar] [CrossRef] [Green Version]
- Choi, J.; Park, J.; Park, S. Design of Coenzyme Q10 solid dispersion for improved solubilization and stability. Int. J. Pharm. 2019, 572, 118832. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, K.; Higashi, T.; Yoshimatsu, A.; Hirayama, F.; Uekama, K.; Arima, H. Pseudorotaxane-like supramolecular complex of coenzyme Q10 with γ-cyclodextrin formed by solubility method. Chem. Pharm. Bull. 2008, 56, 701–706. [Google Scholar] [CrossRef] [Green Version]
- Tackett, J.E. FT-IR Characterization of Metal Acetates in Aqueous Solution. Appl. Spectrosc. 1989, 43, 483–489. [Google Scholar] [CrossRef]
- Heneczkowski, M.; Kopacz, M.; Nowak, D.; Kuzniar, A. Infrared spectrum analysis of some flavonoids. Acta Pol. Pharm. 2001, 58, 415–420. [Google Scholar] [PubMed]
- Pralhad, T.; Rajendrakumar, K. Study of freeze-dried quercetin–cyclodextrin binary systems by DSC, FT-IR, X-ray diffraction and SEM analysis. J. Pharm. Biomed. Anal. 2004, 34, 333–339. [Google Scholar] [CrossRef]
- Shi, G.; Rao, L.; Yu, H.; Xiang, H.; Yang, H.; Ji, R. Stabilization and encapsulation of photosensitive resveratrol within yeast cell. Int. J. Pharm. 2008, 349, 83–93. [Google Scholar] [CrossRef]
- Bongiorno, D.; Ceraulo, L.; Ferrugia, M.; Filizzola, F.; Ruggirello, A.; Liveri, V.T. Inclusion complexes of cyclomaltooligosaccharides (cyclodextrins) with melatonin in solid phase. ARKIVOC 2005, 15, 118–130. [Google Scholar] [CrossRef]
- Wang, Z.; Hu, F.; Che, Z.H.; Song, Q.; Shen, B.D.; Yuan, H.L. Preparation and in vitro release of quercetin nanocrystals self-stabilized Pickering emulsion. China J. Chin. Mater. Med. 2022, 47, 1230–1236. [Google Scholar]
- Sun, R.; Zhao, G.; Ni, S.; Xia, Q. Lipid based nanocarriers with different lipid compositions for topical delivery of resveratrol: Comparative analysis of characteristics and performance. J. Drug Del. Sci. Tech. 2014, 24, 591–600. [Google Scholar] [CrossRef]
- Babu, R.J. Effect of cyclodextrins on the complexation and nasal permeation of melatonin. Drug Deliv. 2008, 15, 381–388. [Google Scholar] [CrossRef] [Green Version]
- Han, Q.; Huang, L.; Luo, Q.; Wang, Y.; Wu, M.; Sun, S.; Zhang, H.; Wang, Y. Synthesis and biological evaluation of biotin-conjugated Portulaca oleracea polysaccharides. RSC Adv. 2021, 11, 18084–18092. [Google Scholar] [CrossRef]
- Muthurajan, T.; Rammanohar, P.; Rajendran, N.P.; Sethuraman, S.; Krishnan, U.M. Evaluation of a quercetin–gadolinium complex as an efficient positive contrast enhancer for magnetic resonance imaging. RSC Adv. 2015, 5, 86967–86979. [Google Scholar] [CrossRef]
- Bukhari, S.B.; Memon, S.; Mahroof-Tahir, M.; Bhanger, M.I. Synthesis, characterization and antioxidant activity copper–quercetin complex. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2009, 71, 1901–1906. [Google Scholar] [CrossRef] [PubMed]
- Sabzichi, M.; Samadi, N.; Mohammadian, J.; Hamishehkar, H.; Akbarzadeh, M.; Molavi, O. Sustained release of melatonin: A novel approach in elevating efficacy of tamoxifen in breast cancer treatment. Colloids Surf. B Biointerfaces 2016, 145, 64–71. [Google Scholar] [CrossRef]
- Hao, J.; Gao, Y.; Zhao, J.; Zhang, J.; Li, Q.; Zhao, Z.; Liu, J. Preparation and optimization of resveratrol nanosuspensions by antisolvent precipitation using Box-Behnken design. AAPS PharmSciTech. 2015, 16, 118–128. [Google Scholar] [CrossRef] [Green Version]
- Council of Europe. European Pharmacopoeia, 10th ed.; EDQM, Council of Europe: Strasbourg, France, 2019. [Google Scholar]
- Markl, D.; Zeitler, J.A. A Review of Disintegration Mechanisms and Measurement Techniques. Pharm. Res. 2017, 34, 890–917. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Desai, P.M.; Liew, C.V.; Heng, P.W.S. Review of Disintegrants and the Disintegration Phenomena. J. Pharm. Sci. 2016, 105, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Morrison, A.B.; Campbell, J.A. Tablet disintegration and physiological Availablity. J. Pharm. Sci. 1965, 54, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Desai, P.M.; Liew, C.V.; Heng, P.W.S. Understanding disintegrant action by visualization. J. Pharm. Sci. 2012, 101, 2155–2164. [Google Scholar] [CrossRef]
- Caramella, C.; Ferrari, F.; Bonferoni, M.C.; Ronchi, M. Disintegrant in solid dosage forms. Drug Dev. Ind. Pharm. 1990, 16, 1577–2561. [Google Scholar] [CrossRef]
- Janssen, P.H.M.; Depaifve, S.; Neveu, A.; Francqui, F.; Dickhoff, B.H.J. Impact of Powder Properties on the Rheological Behavior of Excipients. Pharmaceutics 2021, 13, 1198. [Google Scholar] [CrossRef]
- Jallo, L.J.; Chen, Y.; Bowen, J.; Etzler, F.; Dave, R. Prediction of interparticle adhesion force from surface energy and surface roughness. J. Adhes. Sci. Tech. 2011, 25, 367–384. [Google Scholar] [CrossRef]
- Podczeck, F.; Sharma, M. The influence of particle size and shape of components of binary powder mixtures on the maximum volume reduction due to packing. Int. J. Pharm. 1996, 137, 41–47. [Google Scholar] [CrossRef]
- Ian, J.H.; Fitzpatrick, S.S.; Booth, W. Rational design of powder formulations for tamp filling processes. JPP 2003, 55, 1593–1599. [Google Scholar] [CrossRef]
- Rowe, R.C.; Sheskey, P.J.; Willer, P.J. Handbook of Pharmaceutical Excipients; RPS: London, UK, 2009; pp. 651–653. [Google Scholar]
- Shah, R.B.; Tawakkul, M.A.; Khan, M.A. Comparative evaluation of flow for pharmaceutical powders and granules. AAPS PharmSciTech. 2008, 9, 250–258. [Google Scholar] [CrossRef] [Green Version]
- Švonja-Parezanović, G.; Lalić-Popović, M.; Goločorbin-Kon, S.; Todorović, N.; Pavlović, N.; Jovičić-Bata, J. The effect of magnesium stearate and sodium starch glycolate on powder flowability. Acta Period. Technol. 2019, 2019, 304–310. [Google Scholar] [CrossRef] [Green Version]
- Shi, L.; Chattoraj, S.; Sun, C.C. Reproducibility of flow properties of microcrystalline cellulose—Avicel PH102. Powder Technol. 2011, 212, 253–257. [Google Scholar] [CrossRef]
- Pop, A.L.; Crișan, S.; Bârcă, M.; Ciobanu, A.-M.; Varlas, V.N.; Pop, C.; Pali, M.-A.; Cauni, D.; Ozon, E.A.; Udeanu, D.; et al. Evaluation of Dissolution Profiles of a Newly Developed Solid Oral Immediate-Release Formula Containing Alpha-Lipoic Acid. Processes 2021, 9, 176. [Google Scholar] [CrossRef]
- Osorio, J.G.; Muzzio, F.J. Effects of powder flow properties on capsule filling weight uniformity. Drug Dev. Ind. Pharm. 2013, 39, 1464–1475. [Google Scholar] [CrossRef] [PubMed]
- Kurihara, K.; Ichikawa, I. Effect of Powder Flowability on Capsule-Filling-Weight-Variation. Chem. Pharm. Bull. 1978, 26, 1250–1256. [Google Scholar] [CrossRef] [Green Version]
- Chaerunisaa, A.Y.; Sriwidodo, S.; Abdassah, M. Microcrystalline cellulose as pharmaceutical excipient. In Pharmaceutical Formulation Design—Recent Practices; Ahmad, U., Akhtar, J., Eds.; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef] [Green Version]
- Hlinak, A.J.; Kuriyan, K.; Morris, K.R.; Reklaitis, G.V.; Basu, P.K. Understanding critical material properties for solid dosage form design. J. Pharm. Innov. 2006, 1, 12–17. [Google Scholar] [CrossRef]
- Nokhodchi, A. Effect of Moisture on compaction and compression. Pharm. Technol. 2005, 29, 46–66. [Google Scholar]
- Popescu, I.A.; Donea, C.; Lupulesa, D.; Nicoară, A.C.; Popa, D.E.; Miron, D.S.; Rădulescu, F.S. Comparative assessment of the in vitro performance for modified release tablets containing pentoxifylline using padlle method and flow-through cells. Farmacia 2015, 6, 903–908. [Google Scholar]
- Sadeer, N.B.; Montesano, D.; Albrizio, S.; Zengin, G.; Mahomoodally, M.F. The Versatility of Antioxidant Assays in Food Science and Safety—Chemistry, Applications, Strengths, and Limitations. Antioxidants 2020, 9, 709. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [Green Version]
- Carocho, M.; Ferreira, I.C. A review on antioxidants, prooxidants and related controversy: Natural and synthetic compounds, screening and analysis methodologies and future perspectives. Food Chem. Toxicol. 2013, 51, 15–25. [Google Scholar] [CrossRef]
- Ancerewicz, J.; Migliavacca, E.; Carrupt, P.A.; Testa, B.; Bree, F.; Zini, R.; Tillement, J.P.; Labidalle, S.; Guyot, D.; Chauvet-Monges, A.M.; et al. Structure-property relationships of trimetazidine derivatives and model compounds as potential antioxidants. Free Radic. Biol. Med. 1998, 25, 113–120. [Google Scholar] [CrossRef]
- Kedare, S.B.; Singh, R.P. Genesis and development of DPPH method of antioxidant assay. J. Food Sci. Technol. 2011, 48, 412–422. [Google Scholar] [CrossRef] [Green Version]
- Schaich, K.M.; Tian, X.; Xie, J. Hurdles and pitfalls in measuring antioxidant efficacy: A critical evaluation of ABTS, DPPH, and ORAC assays. J. Funct. Foods. 2015, 14, 111–125. [Google Scholar] [CrossRef]
- Xie, J.; Schaich, K.M. Re-evaluation of the 2,2-Diphenyl-1-picrylhydrazyl Free Radical (DPPH) assay for Antioxidant Activity. J. Agric. Food Chem. 2014, 62, 4251–4260. [Google Scholar] [CrossRef] [PubMed]
- Ozcelik, B.; Lee, J.H.; Min, D.B. Effects of light, oxygen, and pH on the absorbance of 2,2-diphenyl-1-picrylhydrazyl. J. Food Sci. 2003, 68, 487–490. [Google Scholar] [CrossRef]
- Bonnefont-Rousselot, D.; Collin, F. Melatonin: Action as antioxidant and potential applications in human disease and aging. Toxicology 2010, 278, 55–67. [Google Scholar] [CrossRef]
- Cardinali, D.P.; Srinivasan, V.; Brzezinski, A.; Brown, G.M. Melatonin and its analogs in insomnia and depression. J. Pineal Res. 2012, 52, 365–375. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosales-Corral, S.A.; Acuña-Castroviejo, D.; Coto-Montes, A.; Boga, J.A.; Manchester, L.C.; Fuentes-Broto, L.; Korkmaz, A.; Ma, S.; Tan, D.X.; Reiter, R.J. Alzheimer’s disease: Pathological mechanisms and the beneficial role of melatonin. J. Pineal Res. 2012, 52, 67–202. [Google Scholar]
- Savu, O.; Elian, V.; Steriade, O.; Teodoru, I.; Mihut, S.; Tacu, C.; Covic, A.; Serafinceanu, C. The impact of basal insulin analogues on glucose variability in patients with type 2 diabetes undergoing renal replacement therapy for end-stage renal disease. Int. Urol. Nephrol. 2016, 48, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Motilva, V.; García-Mauriño, S.; Talero, E.; Illanes, M.J. New paradigms in chronic intestinal inflammation and colon cancer: Role of melatonin. Pineal Res. 2011, 51, 44–60. [Google Scholar] [CrossRef]
- Brzezinski, A. Melatonin in humans. N. Engl. J. Med. 1997, 336, 186–195. [Google Scholar] [CrossRef]
- Hardeland, R.; Madrid, J.A.; Tan, D.X.; Reiter, R.J. Kynuramines, metabolites of melatonin and other indoles: The resurrection of an almost forgotten class of biogenic amines. J. Pineal Res. 2012, 52, 139–166. [Google Scholar] [CrossRef]
- Ilie, I.; Ciubotaru, V.; Tulin, A.; Hortopan, D.; Caragheorgheopol, A.; Purice, M.; Neamtu, C.; Elian, V.I.; Banica, A.; Oprea, L.; et al. The Multifarious Cushing’s—Lessons from A Case Series. Acta Endocrinol. 2019, 15, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Korkmaz, A.; Reiter, R.J.; Topal, T.; Manchester, L.C.; Oter, S.; Tan, D.X. Melatonin: An established antioxidant worthy of use in clinical trials. Mol. Med. 2009, 15, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Johns, J.R.; Platts, J.A. Theoretical insight into the antioxidant properties of melatonin and derivatives. Org. Biomol. Chem. 2014, 12, 7820. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sedel, F.; Bernard, D.; Mock, D.M.; Tourbah, A. Targeting demyelination and virtual hypoxia with high-dose biotin as a treatment for progressive multiple sclerosis. Neuropharmacology 2016, 110, 644–653. [Google Scholar] [CrossRef] [Green Version]
- Sghaier, R.; Zarrouk, A.; Nury, T.; Badreddine, I.; O’Brien, N.; Mackrill, J.J.; Vejux, A.; Samadi, M.; Nasser, B.; Caccia, C.; et al. Biotin attenuation of oxidative stress, mitochondrial dysfunction, lipid metabolism alteration and 7β-hydroxycholesterol-induced cell death in 158N murine oligodendrocytes. Free Radic. Res. 2019, 53, 535–561. [Google Scholar] [CrossRef] [Green Version]
- Fourcade, S.; Goicoechea, L.; Parameswaran, J.; Schlüter, A.; Launay, N.; Ruiz, M.; Seyer, A.; Colsch, B.; Calingasan, N.Y.; Ferrer, I.; et al. High-dose biotin restores redox balance, energy and lipid homeostasis, and axonal health in a model of adrenoleukodystrophy. Brain Pathol. 2020, 30, 945–963. [Google Scholar] [CrossRef]
- Littarru, G.P.; Tiano, L. Bioenergetic and antioxidant properties of coenzyme Q10: Recent developments. Mol Biotechnol. 2007, 37, 31–37. [Google Scholar] [CrossRef]
- Cirilli, I.; Damiani, E.; Dludla, P.V.; Hargreaves, I.; Marcheggiani, F.; Millichap, L.E.; Orlando, P.; Silvestri, S.; Tiano, L. Role of Coenzyme Q10 in Health and Disease: An Update on the Last 10 Years (2010–2020). Antioxidants 2021, 10, 1325. [Google Scholar] [CrossRef]
- Xu, D.; Hu, M.J.; Wang, Y.Q.; Cui, Y.L. Antioxidant Activities of Quercetin and Its Complexes for Medicinal Application. Molecules 2019, 24, 1123. [Google Scholar] [CrossRef] [Green Version]
- Liao, H.; Tang, M.; Luo, L.; Li, C.; Chiclana, F.; Zeng, X.J. A bibliometric analysis and visualization of medical big data research. Sustainability 2018, 10, 166. [Google Scholar] [CrossRef] [Green Version]
- Han, Q.S.; Wang, X.H.; Cai, S.F.; Liu, X.L.; Zhang, Y.F.; Yang, L.; Wang, C.; Yang, R. Quercetin nanoparticles with enhanced bioavailability as multifunctional agents toward amyloid induced neurotoxicity. J. Mater. Chem. B. 2018, 6, 1387–1393. [Google Scholar] [CrossRef]
- Wang, M.; Li, J.; Rangarajan, M.; Shao, Y.; LaVoie, E.J.; Huang, T.C.; Ho, C.T. Antioxidative phenolic compounds from sage (Salvia officinalis). J. Agric. Food Chem. 1998, 46, 4869–4873. [Google Scholar] [CrossRef]
- Stivala, L.A.; Savio, M.; Carafoli, F.; Perucca, P.; Bianchi, L.; Maga, G.; Forti, L.; Pagnoni, U.M.; Albini, A.; Prosperi, E.; et al. Specific structural determinants are responsible for the antioxidant activity and the cell cycle effects of resveratrol. J. Biol. Chem. 2001, 276, 22586–22594. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cao, H.; Pan, X.; Li, C.; Zhou, C.; Deng, F.; Li, T. Density functional theory calculations for resveratrol. Bioorg. Med. Chem. Lett. 2003, 13, 1869–1871. [Google Scholar] [CrossRef]
- Musuc, A.M.; Anuta, V.; Atkinson, I.; Sarbu, I.; Popa, V.T.; Munteanu, C.; Mircioiu, C.; Ozon, E.A.; Nitulescu, G.M.; Mitu, M.A. Formulation of Chewable Tablets Containing Carbamazepine-β-cyclodextrin Inclusion Complex and F-Melt Disintegration Excipient. The Mathematical Modeling of the Release Kinetics of Carbamazepine. Pharmaceutics 2021, 13, 915. [Google Scholar] [CrossRef]
- Burcea Dragomiroiu, G.T.A.; Miron, D.S.; Baloescu, C.; Bârcă, M.; Mitu, M.A.; Popa, D.E.; Rădulescu, F.Ș. Comparative in-vitro study of immediate and modified released oral dosage forms of cefaclor. Farmacia 2012, 60, 334–341. [Google Scholar]
- Balaci, T.; Velescu, B.; Karampelas, O.; Musuc, A.M.; Nitulescu, G.M.; Ozon, E.A.; Nitulescu, G.; Gird, C.E.; Fita, C.; Lupuliasa, D. Physico-Chemical and Pharmaco-Technical Characterization of Inclusion Complexes Formed by Rutoside with beta-Cyclodextrin and Hydroxypropyl-beta-Cyclodextrin Used to Develop Solid Dosage Forms. Processes 2021, 9, 26. [Google Scholar] [CrossRef]
- Carr, R.L. Evaluating flow properties of solids. Chem. Eng. 1965, 72, 69–72. [Google Scholar]
- Hausner, H.H. Friction conditions in a mass of metal powder. Int. J. Powder Metall. 1967, 3, 7–13. [Google Scholar]
- Mitu, M.A.; Cretu, E.A.; Novac, M.; Karampelas, O.; Nicoara, A.; Nitulescu, G.; Lupuleasa, D. The flowing characteristics of some composed powders containing inclusion complexes in beta-cyclodextrin. In Proceedings of the Romanian National Congress of Pharmacy, 17th Edition: 21st Century Pharmacy—Between Intelligent Specialization and Social Responsibility 2018, Bucharest, Romania, 26–29 September 2018; pp. 129–133. [Google Scholar]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cationic decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Margina, D.; Olaru, O.T.; Ilie, M.; Grădinaru, D.; Guțu, C.; Voicu, S.; Dinischiotu, A.; Spandidos, D.A.; Tsatsakis, A.M. Assessment of the potential health benefits of certain total extracts from Vitisvinifera, Aesculushyppocastanum and Curcuma longa. Exp. Ther. Med. 2015, 10, 1681–1688. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nair, V.D.P.; Dairam, A.; Agbonon, A.; Arnason, J.T.; Foster, B.C.; Kanfer, I. Investigation of the antioxidant activity of African potato (Hypoxis hemerocallidea). J. Agric. Food Chem. 2007, 55, 1707–1711. [Google Scholar] [CrossRef] [PubMed]




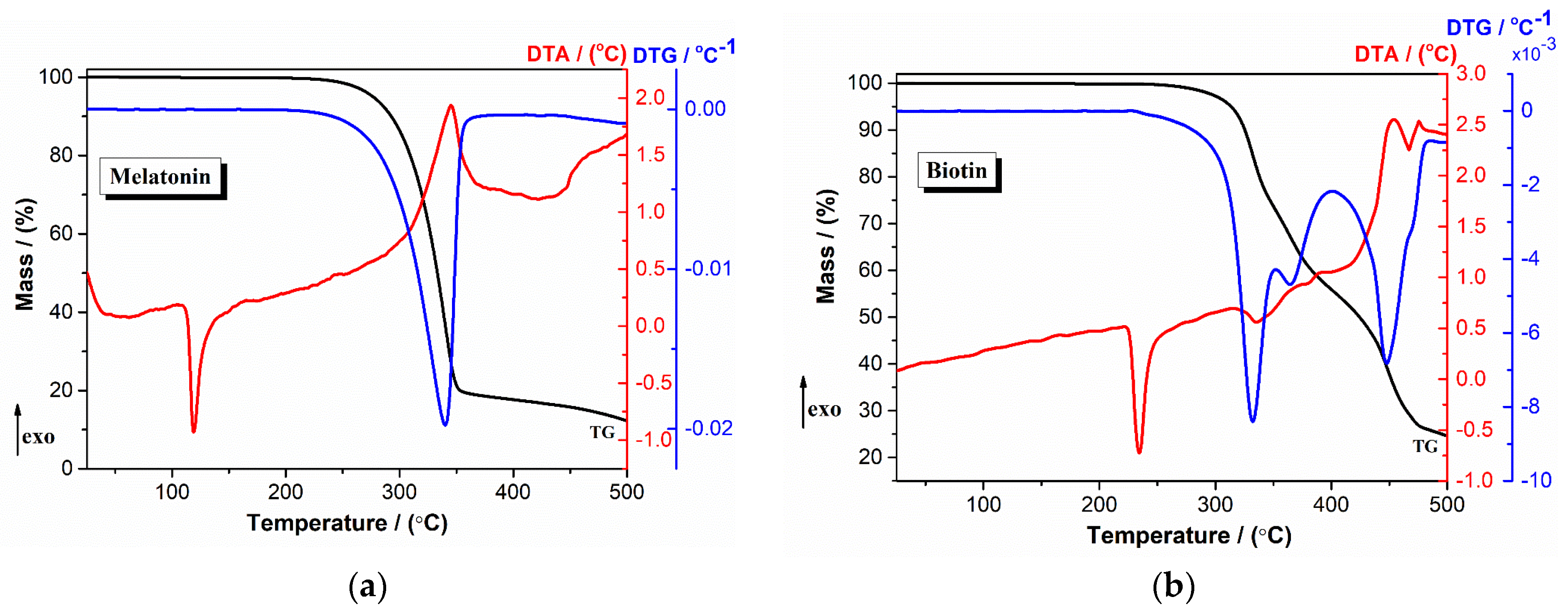





| Parameter | F1a.i. | F2a.i. | F1c.c. | F2c.c. |
|---|---|---|---|---|
| Flow time (s) * | 21.4 ± 1.76 | 23.6 ± 3.21 | 14.7 ± 2.37 | 15.3 ± 2.04 |
| Angle of repose (θ°) * | 40.26 ± 2.69 | 43.12 ± 3.16 | 27.85 ± 2.12 | 28.34 ± 1.97 |
| Flow rate (g/s) * | 2.336 | 2.118 | 3.401 | 3.267 |
| Bulk density (g/mL) | 0.588 ± 0.26 | 0.589 ± 0.55 | 0.551 ± 0.18 | 0.602 ± 0.25 |
| Tapped density (g/mL) | 0.833 ± 0.34 | 0.884 ± 0.41 | 0.713 ± 0.22 | 0.806 ± 0.12 |
| Carr Index (CI) (%) | 29.411 | 33.48 | 22.72 | 25.31 |
| Hausner’s ratio (HR) | 1.417 | 1.500 | 1.29 | 1.33 |
| Moisture content (%) | 3.95 ± 1.12 | 5.67 ± 2.23 | 3.04 ± 0.85 | 5.18 ± 1.93 |
| Tested Parameters * | Formulation Code | |
|---|---|---|
| F1c.c. | F2c.c. | |
| Mass uniformity (mg) | 483.17 ± 3.64 | 480.31 ± 4.29 |
| In vitro disintegration time (s) | 458 ± 3 | 672 ± 7 |
| Ingredients | Quantity (mg)/Capsule | Role in Formulation | |
|---|---|---|---|
| F1c.c. | F2c.c. | ||
| Melatonin | 5 | - | Active ingredient |
| Biotin | 10 | 10 | Active ingredient |
| Coenzyme Q10 | 300 | 100 | Active ingredient |
| Quercetin | - | 100 | Active ingredient |
| Resveratrol | - | 100 | Active ingredient |
| Avicel PH 102—Microcrystalline cellulose | 155 | 160 | Filler |
| Explotab®—Sodium starch glycolate | 10 | 10 | Superdisintegrant |
| Ligamed® MF-2-V—Magnesium stearate | 5 | 5 | Lubricant |
| Total | 485 | 485 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ozon, E.A.; Iuga, I.D.M.; Mititelu, M.; Musuc, A.M.; Manolescu, B.N.; Petrescu, S.; Cusu, J.P.; Rusu, A.; Surdu, V.-A.; Oprea, E.; et al. Pharmacotechnical, Physico-Chemical, and Antioxidant Evaluation of Newly Developed Capsule Formulations. Int. J. Mol. Sci. 2023, 24, 11426. https://doi.org/10.3390/ijms241411426
Ozon EA, Iuga IDM, Mititelu M, Musuc AM, Manolescu BN, Petrescu S, Cusu JP, Rusu A, Surdu V-A, Oprea E, et al. Pharmacotechnical, Physico-Chemical, and Antioxidant Evaluation of Newly Developed Capsule Formulations. International Journal of Molecular Sciences. 2023; 24(14):11426. https://doi.org/10.3390/ijms241411426
Chicago/Turabian StyleOzon, Emma Adriana, Izabela Dana Maria Iuga, Magdalena Mititelu, Adina Magdalena Musuc, Bogdan Nicolae Manolescu, Simona Petrescu, Jeanina Pandele Cusu, Adriana Rusu, Vasile-Adrian Surdu, Eliza Oprea, and et al. 2023. "Pharmacotechnical, Physico-Chemical, and Antioxidant Evaluation of Newly Developed Capsule Formulations" International Journal of Molecular Sciences 24, no. 14: 11426. https://doi.org/10.3390/ijms241411426











