Low-Dose Non-Targeted Effects and Mitochondrial Control
Abstract
:1. Introduction
2. Low-Dose Radiation Effects
3. Hormesis
4. Adaptive Radiation Response and the Involvement of Mitochondrial Functions
5. Low Dose Hyper-Radiation Sensitivity (HRS) and Induction of Radioresistance (IRR)
5.1. IRR
5.2. Involvement of Immune Functions
5.3. Involvement of Mitochondria
6. Bystander Effects: Short Distance NTEs
7. Long Distance NTEs: Abscopal Effects
8. Genomic Instability
9. Mitochondria and Innate and Adaptive Immunity Induced by IR
9.1. Low-Dose Immune Effects
9.2. Role of mtDNA in Innate and Adaptive Immune Responses
9.3. Immunogenic Effects in Antitumor RT
10. Concluding Remarks
10.1. RIBE and NTE and the Role of Mitochondria
| IR Exposure Exp. Device | Biological System | Observation | References |
|---|---|---|---|
| Low LET photons (γ-rays, X-rays) (0.5, 5 and 7.5 Gy) ICCM | Mammalian cells, Chinese hamster (CHO-K1) Human keratinocytes (HGV-G) Medium transfer | Absence of RIBE in CHO-K1 mutants with nonfunctional glucose-6-phosphate dehydrogenase (G6PD) involved in mitochondrial metabolism in HGV-G by inhibition of apoptosis and lactate metabolism. Alteration of calcium fluxes and loss of mitochondrial membrane potential (MMP). Involvement of mitochondrial ROS. | [318,319,323] |
| Low LET IR 5 mGy and 0.5 Gy ICCM | Human keratinocytes (HGV-G) Medium transfer | Reduction of clonogenicity. Induction of increases in mitochondrial mass and low Bcl-2 expression in bystander cells after 5 mGy in ICCM, but increased expression after 5 Gy. Decrease in survival. | [312] |
| X-rays 1Gy ICCM | Human hybrids cells: Chinese hamster ovary (CHO) cells GM10115 + human chromosome 4, Medium transfer | IR-induced mitochondrial dysfunction results in persistent high levels of ROS perpetuating genomic instability plus clastogenic and transgenerational effects. | [249,250] |
| IR: γ-rays 5 Gy ICCM | Chinese hamster (CHO-K1) Human keratinocytes (HGV-G) Medium transfer | Increase in mitochondrial mass, dysfunctional mitochondria in BE. | [324] |
| IR: γ-rays (5 mGy, 0.5 Gy, 5 Gy) ICCM | Chinese hamster (CHO-K1) Human keratinocytes (HGV-G) Medium transfer | Mitochondria are sensitive to LDIR and ICCM, loss of enzymatic functions (OXPHOS), and altered mtDNA-directed protein synthesis. | [325] |
| γ-rays or 160 kV X-rays (0.5 Gy) | Human mammary epithelial cells (HMEC); Balb/c mice TGF-β1 +/− and +/+ | Increased centrosome deregulation as a function of time. After IR, clonal expansion CA increased in HMEC, but unstable cells could be deleted by TGF-β1 via p53-dependent apoptosis (involving mitochondrial signaling) TGF-β1 that can also suppress EMT. | [326] |
| Microbeam IR with Carbon ions or X-rays | Mammalian cells Murine lymphoma L5178Y in co-culture with irradiated neoplastic epithelial cells. Co-culture experiments | Cytoplasmic and cell irradiation affects mitochondria and calcium fluxes in targeted glioma and fibroblast cells. Cytoplasmic IR involved mitochondrial damage and RIBE response. | [244] |
| Microbeam with α−particles | T98G glioma cells and AG01522 fibroblasts ICCM or Co-culture experiments | Calcium signaling occurs early (RIBE). NO and mito-chondrial ROS lead to chromosomal damage (MN). | [243] |
| 241AM source α-particles (100 mGy) ICCM | Hamster normal AL cells ρ+ and mtDNA-depletedAL cells (ρ0) (donor) and normal human fibroblasts (AG1522) (receptor cells). Medium transfer | Mitochondria-derived NO and O2− play an important role in the initiation and activation of RIBE. IR-induced intracellular factors derived from mitochondria and calcium-dependent mitochondrial NOS. Mitochondria intercellular signaling from irradiated cells participates in ROS-mediated genotoxicity. | [327,328] |
| Microbeam IR with 1–10 protons ICCM | Human keratinocytes HGV-G Medium transfer | ROS levels increased in bystander cells. Apoptosis induced was associated with a decrease in MMP and increased intracellular Ca2+ levels. | [242] |
| Microbeam IR with α-particles | Cervical cancer cells (HeLa) and mitochondria depleted pseudo-ρ0 cells | No RIBE in the absence of mtDNA. Signaling is inhibited by ROS and RNA inhibitors. Mt-dependent 53BP1 delocalization. BE involves intact mt signaling from targeted cytoplasm to the nucleus. | [196] |
| Microbeam IR with 4He ions (120 keV/μm) α-rays | Human fibroblast cells ρ0 and ρ+ | High BE mutagenic response in mtDNA depleted ρ0 cells. BE involved mt-dependent NF-κB/iNOS/NO and NF-κB/COX-2/prostaglandin E2 signaling and NOS and COX2 signaling. | [245] |
| Low-dose a-particles (0.29 mGy–25 mGy) and γ-rays (2 mGy–50 mGy | 208F and v-src trans-formed 208Fsrc3 rat fibroblast cell lines. Co-culture experiments | Low-dose IR of non-transformed cells can induce apoptosis in precancerous cells through RIBE involving ROS/NOS signaling and cytokines, such as TGF-β. The stimulatory effect saturates at 50 mGy for γ-rays and at 25 mGy for α-particles. | [191] |
| 1 GeV/u iron ions (LET~151 keV/μm), 600 MeV/u; silicon ions (LET~51 keV/μm), or 1 GeV protons (LET~0.2 keV/μm). | Normal human fibroblasts (AG1522) Test of progeny: co-cultures of cells exposed to low or high doses of high LET IR | RIBE depends on radiation quality and dose, and oxidative stress involving mitochondria. | [186] |
| γ-rays (0.05 and 0.5 Gy) ICCM | Human keratinocyte cell line (HaCaT) Medium transfer | Low-dose expression of genes involved in mitochondria-driven intrinsic apoptosis induced in bystander cells at low-dose (50 mGy). | [190] |
| γ-rays, α-particles and HZE particles (500 mGy) | Normal human FB (AG1522 cells) co-cultured with a-irradiated HeLa cells (500 mGy) (connexin 32) Co-culturing | Increased induction of MN and GI in bystander cells. | [224] |
| Tritium (β-radiation) induced UV biophoton emission | Human colon carcinoma cell line, HCT116 p53 +/+ Biophoton emission involvement in BE Exosomes | Biophoton electromagnetic bystander signaling compromises mitochondrial complex V (ATP production) and may be involved in the human fatigue syndrome. Exosomes extracted from UV-ICCM modulates clonogenic survival and MPP in bystander cells. | [38] |
| γ-rays (22 mGy) and biophoton emission | Cells: HCT116 p53 +/+ Test involvement of cellular emissions of biophotons in gamma radiation that is induced bystander cells | Low-dose biophoton emission from irradiated human cells may cause detrimental low-dose RIBE. | [39] |
| 6 MeV photons (Clinac 600), 2 Gy | Fadu cells derived from HNSCC Secretion of exosomes in RIBE | NTE is propagated by mtDNA and RNA in vesicles similar to exosomes. | [28] |
| X-rays (0.1, 0.25, 2 Gy) Extracelluar vesicles (EVs) | C57BL/6 mice Total body IR, Extracellular vesicles (EVs) | A panel of miRNA are involved in EVs bystander effects, differently at low and high dose, IR induced systemic effects. | [329] |
| X-rays 4 Gy CCCM/ICCM | Seven-week-old male ICR mice: ELV from irradiated mouse serum and ICCM | Absence of DNA damage in CCCM ELV or ICCM ELV from mt-depleted. ρ0 normal human fibroblasts. Secretion of mtDNA via exosomes is involved in mediating RIBE signals. | [32] |
| X-Rays Partial and whole body exposure 2 Gy | C57Bl/6 female mice of eight weeks of age Analysis of ‘Out of field ‘effects partial body IR in mice Exosomes | Deregulation of many proteins and miRNAs. Some miRNA, proteomic changes, and exosomes are involved in anti-apoptotic effects. Injection of exosomes from irradiated mice can prevent apoptosis. | [226,227] |
| γ-rays, high doses (2–8 Gy) ICCM | Human HepT2 cells Medium transfer from irradiated cells | Induction of Bax, Bcl2, caspases and γ-H2AX DNA damage in bystander HepT2 cells. | [330] |
|
200 kV X–rays (6 Gy) |
Human pancreatic cancer cells (MiaPaCa–2), wild–type (wt) and ATM−/− fibroblasts Co-culturing | Healthy ATM+/+ cells modify the DDR of irradiated cells by a microtubule- and ATM-dependent exchange of healthy mitochondria. | [201] |
10.2. Factors Possibly Contributing Adaptive Beneficial and Armful Effects of Low Doses
10.3. LDIR Adaptive Responses
10.4. Immune and Anti-Tumor LDIR Effects
10.5. Biopositive Effects of LDIR
10.6. Biopositive Effects of LDIR on the Immune System
10.7. Bionegative Effects of LDIR
10.8. Low Dose-Rate Effects
10.9. Relationship of LDIR and LDRIR to DDR and Mitochondrial ROS
10.10. Role of Mitochondria in Radioresistance
10.11. Conclusive Thoughts
Funding
Institutional Review Board Statement
Informed Consent statement:
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nagasawa, H.; Little, J.B. Induction of sister chromatid exchanges by extremely low doses of alpha-particles. Cancer Res. 1992, 52, 6394–6396. [Google Scholar] [PubMed]
- Morgan, W.F. Non-targeted and delayed effects of exposure to ionizing radiation: I. Radiation-induced genomic instability and bystander effects in vitro. Radiat. Res. 2003, 159, 567–580. [Google Scholar] [CrossRef]
- Morgan, W.F. Non-targeted and delayed effects of exposure to ionizing radiation: II. Radiation-induced genomic instability and bystander effects in vivo, clastogenic factors and transgenerational effects. Radiat. Res. 2003, 159, 581–596. [Google Scholar] [CrossRef] [PubMed]
- Averbeck, D. Non-targeted effects as a paradigm breaking evidence. Mutat. Res. 2010, 687, 7–12. [Google Scholar] [CrossRef]
- Morgan, W.F.; Sowa, M.B. Non-targeted effects induced by ionizing radiation: Mechanisms and potential impact on radiation induced health effects. Cancer Lett. 2015, 356, 17–21. [Google Scholar] [CrossRef] [Green Version]
- Baulch, J.E. Radiation-induced genomic instability, epigenetic mechanisms and the mitochondria: A dysfunctional ménage à trois? Int. J. Radiat. Biol. 2019, 95, 516–525. [Google Scholar] [CrossRef]
- Mothersill, C.; Cocchetto, A.; Seymour, C. Low dose and non-targeted radiation effects in environmental protection and medicine-A new model focusing on electromagnetic signaling. Int. J. Mol. Sci. 2022, 23, 11118. [Google Scholar] [CrossRef]
- ICRP. The 2007 Recommendations of the International Commission on Radiological Protection; ICRP Publication 103. Ann. ICRP 37 (2-4); ICRP: Ottawa, ON, Canada, 2007. [Google Scholar]
- Demaria, S.; Formenti, S.C. The abscopal effect 67 years later: From a side story to center stage. Br. J. Radiol. 2020, 93, 20200042. [Google Scholar] [CrossRef]
- Yin, T.; Xin, H.; Yu, J.; Teng, F. The role of exosomes in tumour immunity under radiotherapy: Eliciting abscopal effects? Biomark. Res. 2021, 9, 22. [Google Scholar] [CrossRef]
- Xue, L.Y.; Butler, N.J.; Makrigiorgos, G.M.; Adelstein, S.J.; Kassis, A.I. Bystander effect produced by radiolabeled tumor cells in vivo. Proc. Natl. Acad. Sci. USA 2002, 99, 13765–13770. [Google Scholar] [CrossRef]
- Ye, F.; Ning, J.; Liu, X.; Jin, X.; Wang, T.; Li, Q. The influence of non-DNA-targeted effects on carbon ion-induced low-dose hyper-radiosensitivity in MRC-5 cells. J. Radiat. Res. 2016, 57, 103–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buonanno, M.; Gonon, G.; Pandey, B.N.; Azzam, E.I. The intercellular communications mediating radiation-induced bystander effects and their relevance to environmental, occupational, and therapeutic exposures. Int. J. Radiat. Biol. 2022, 27, 964–982. [Google Scholar] [CrossRef] [PubMed]
- Azzam, E.I.; Jay-Guerin, J.-P.; Pain, D. Ionizing radiation-induced metabolic oxidative stress and prolonged cell injury. Cancer Lett. 2012, 32, 48–60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spitz, D.R.; Azzam, E.I.; Li, J.J.; Gius, D. Metabolic oxidation/reduction reactions and cellular responses to ionizing radiation: A unifying concept in stress response biology. Cancer Metastasis Rev. 2004, 23, 311–322. [Google Scholar] [CrossRef]
- Hei, T.K.; Zhou, H.; Ivanov, V.N.; Hong, M.; Lieberman, H.B.; Brenner, D.J.; Amundson, S.A.; Geard, C.R. Mechanism of radiation-induced bystander effects: A unifying model. J. Pharm. Pharmacol. 2008, 60, 943–950. [Google Scholar] [CrossRef] [Green Version]
- Weinberg, S.E.; Sena, L.A.; Chandel, N.S. Mitochondria in the regulation of innate and adaptive immunity. Immunity 2015, 42, 406–417. [Google Scholar] [CrossRef] [Green Version]
- Sia, J.; Hagekyriakou, J.; Chindris, I.; Albarakati, H.; Leong, T.; Schlenker, R.; Keam, S.P.; Williams, S.G.; Neeson, P.J.; Johnstone, R.W.; et al. Regulatory T cells shape the differential impact of radiation dose-fractionation schedules on host innate and adaptive antitumor immune defenses. Int. J. Radiat. Oncol. Biol. Phys. 2021, 111, 502–514. [Google Scholar] [CrossRef]
- Averbeck, D.; Rodriguez-Lafrasse, C. Role of mitochondria in IR responses: Epigenetic, metabolic, and signaling impacts. Int. J. Mol. Sci. 2021, 22, 11047. [Google Scholar] [CrossRef]
- Le, M.; McNeill, F.E.; Seymour, C.B.; Rusin, A.; Diamond, K.; Rainbow, A.J.; Murphy, J.; Mothersill, C.E. Modulation of oxidative phosphorylation (OXPHOS) by radiation- induced biophotons. Environ. Res. 2018, 163, 80–87. [Google Scholar] [CrossRef]
- Wang, X.; Gerdes, H.H. Transfer of mitochondria via tunneling nanotubes rescues apoptotic PC12 cells. Cell Death Differ. 2015, 22, 1181–1191. [Google Scholar] [CrossRef] [Green Version]
- Saha, T.; Dash, C.; Jayabalan, R.; Khiste, S.; Kulkarni, A.; Kurmi, K.; Mondal, J.; Majumder, P.K.; Bardia, A.; Jang, H.L.; et al. Intercellular nanotubes mediate mitochondrial trafficking between cancer and immune cells. Nat. Nanotechnol. 2022, 17, 98–106. [Google Scholar] [CrossRef]
- Martinez-Outschoorn, U.E.; Balliet, R.M.; Rivadeneira, D.B.; Chiavarina, B.; Pavlides, S.; Wang, C.; Whitaker-Menezes, D.; Daumer, K.M.; Lin, Z.; Witkiewicz, A.K.; et al. Oxidative stress in cancer associated fibroblasts drives tumor-stroma co-evolution: A new paradigm for understanding tumor metabolism, the field effect and genomic instability in cancer cells. Cell Cycle 2010, 9, 3256–3276. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamazaki, T.; Kirchmair, A.; Sato, A.; Buqué, A.; Rybstein, M.; Petroni, G.; Bloy, N.; Finotello, F.; Stafford, L.; Navarro Manzano, E.; et al. Mitochondrial DNA drives abscopal responses to radiation that are inhibited by autophagy. Nat. Immunol. 2020, 21, 1160–1171. [Google Scholar] [CrossRef] [PubMed]
- Fischietti, M.; Fratini, E.; Verzella, D.; Vecchiotti, D.; Capece, D.; Di Francesco, B.; Esposito, G.; Balata, M.; Ioannuci, L.; Sykes, P.; et al. Low radiation environment witches the overgrowth-induced cell apoptosis toward autophagy. Front. Public Health 2021, 8, 594789. [Google Scholar] [CrossRef] [PubMed]
- Meng, Q.; Zaharieva, E.K.; Sasatani, M.; Kobayashi, J. Possible relationship between mitochondrial changes and oxidative stress under low dose-rate irradiation. Redox Rep. 2021, 26, 160–169. [Google Scholar] [CrossRef]
- Du, Y.; Du, S.; Liu, L.; Gan, F.; Jiang, X.; Wangrao, K.; Lyu, P.; Gong, P.; Yao, Y. Radiation-induced bystander effect can be transmitted through exosomes using miRNAs as effector molecules. Radiat. Res. 2020, 194, 89–100. [Google Scholar] [CrossRef]
- Jelonek, K.; Wojakowska, A.; Marczak, L.; Muer, A.; Tinhofer-Keilholz, I.; Lysek-Gladysinska, M.; Widlak, P.; Pietrowska, M. Ionizing radiation affects protein composition of exosomes secreted in vitro from head and neck squamous cell carcinoma. Acta Biochim. Pol. 2015, 62, 265–272. [Google Scholar] [CrossRef] [Green Version]
- Shan, Z.; Wang, H.; Zhang, Y.; Min, W. The role of tumor-derived exosomes in the abscopal effect and immunotherapy. Life 2021, 11, 381. [Google Scholar] [CrossRef]
- Daguenet, E.; Louati, S.; Wozny, A.S.; Vial, N.; Gras, M.; Guy, J.B.; Vallard, A.; Rodriguez-Lafrasse, C.; Magné, N. Radiation induced bystander and abscopal effects: Important lessons from preclinical models. Br. J. Cancer 2020, 123, 339–348. [Google Scholar] [CrossRef]
- He, D.; Zhao, Z.; Fu, B.; Li, X.; Zhao, L.; Chen, Y.; Liu, L.; Liu, R.; Li, J. Exosomes participate in the radiotherapy resistance of cancers. Radiat. Res. 2022, 197, 559–565. [Google Scholar] [CrossRef]
- Ariyoshi, K.; Miura, T.; Kasai, K.; Fujishima, Y.; Nakata, A.; Yoshida, M. Radiation-induced bystander effect is mediated by mitochondrial DNA in exosome-ike vesicles. Sci. Rep. 2019, 9, 9103. [Google Scholar] [CrossRef] [Green Version]
- Jokar, S.; Marques, I.A.; Khazaei, S.; Martins-Marques, T.; Girao, H.; Laranjo, M.; Botelho, M.F. The footprint of exosomes in the radiation-induced bystander effects. Bioengineering 2022, 9, 243. [Google Scholar] [CrossRef]
- Miranda, S.; Correia, M.; Dias, A.G.; Pestana, A.; Soares, P.; Nunes, J.; Lima, J.; Máximo, V.; Boaventura, P. Evaluation of the role of mitochondria in the non-targeted effects of ionizing radiation using cybrid cellular models. Sci. Rep. 2020, 10, 6131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gorman, S.; Fox, E.; O’Donoghue, D.; Sheahan, K.; Hyland, J.; Mulcahy, H.; Loeb, L.A.; O’Sullivan, J. Mitochondrial mutagenesis induced by tumor-specific radiation bystander effects. J. Mol. Med. 2010, 88, 701–708. [Google Scholar] [CrossRef] [PubMed]
- Heeran, A.B.; Berrigan, H.P.; Buckley, C.E.; Bottu, H.M.; Prendiville, O.; Buckley, A.M.; Clarke, N.; Donlon, N.E.; Nugent, T.S.; Durand, M.; et al. Radiation-induced bystander effect (RIBE) alters mitochondrial metabolism using a human rectal cancer ex vivo explant model. Transl. Oncol. 2021, 14, 100882. [Google Scholar] [CrossRef] [PubMed]
- Le, M.; Fernandez-Palomo, C.; McNeill, F.E.; Seymour, C.B.; Rainbow, A.J.; Mothersill, C.E. Exosomes are released by bystander cells exposed to radiation-induced biophoton signals: Reconciling the mechanisms mediating the bystander effect. PLoS ONE 2017, 12, e0173685. [Google Scholar] [CrossRef] [Green Version]
- Le, M.; Mothersill, C.E.; Seymour, C.B.; Rainbow, A.J.; McNeill, F.E. An observed effect of p53 status on the bystander response to radiation-induced cellular photon emission. Radiat Res. 2017, 187, 169–185. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J.; Vo, N.T.K.; Chettle, D.R.; McNeill, F.E.; Seymour, C.B.; Mothersill, C.E. Quantifying biophoton emissions from human cells directly exposed to low-dose gamma radiation. Dose Response 2020, 18, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Mothersill, C.; Seymour, C. Low dose radiation mechanisms: The certainty of uncertainty. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2022, 876–877, 503451. [Google Scholar] [CrossRef]
- Matsumoto, H.; Tomita, M.; Otsuka, K.; Hatashita, M.; Hamada, N. Nitric oxide is a key molecule serving as a bridge between radiation-induced bystander and adaptive responses. Curr. Mol. Pharmacol. 2011, 4, 126–134. [Google Scholar] [CrossRef]
- Mothersill, C.; Seymour, C.B.; Joiner, M.C. Relationship between radiation-induced low-dose hypersensitivity and the bystander effect. Radiat. Res. 2002, 157, 526–532. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Palomo, C.; Seymour, C.; Mothersill, C. Inter-Relationship between low-dose hyper-radiosensitivity and radiation-induced bystander effects in the human T98G Glioma and the epithelial HaCaT cell line. Radiat. Res. 2016, 185, 124–133. [Google Scholar] [CrossRef] [PubMed]
- Pierce, D.A.; Preston, D.L. Radiation-related cancer risks at low doses among atomic bomb survivors. Radiat. Res. 2000, 154, 178–186. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brenner, D.J.; Doll, R.; Goodhead, D.T.; Hall, E.J.; Land, C.E.; Little, J.B.; Lubin, J.H.; Preston, D.L.; Preston, R.J.; Puskin, J.S.; et al. Cancer risks attributable to low doses of ionizing radiation: Assessing what we really know. Proc. Natl. Acad. Sci. USA 2003, 100, 13761–13766. [Google Scholar] [CrossRef]
- Wakeford, R.; Little, M.P. Childhood cancer after low-level intrauterine exposure to radiation. J. Radiol. Prot. 2002, 22, A123–A127. [Google Scholar] [CrossRef]
- Richardson, D.; Sugiyama, H.; Nishi, N.; Sakata, R.; Shimizu, Y.; Grant, E.J.; Soda, M.; Hsu, W.L.; Suyama, A.; Kodama, K.; et al. Ionizing radiation and leukemia mortality among Japanese Atomic Bomb Survivors, 1950–2000. Radiat. Res. 2009, 172, 368–382. [Google Scholar] [CrossRef]
- Little, M.P.; Wakeford, R.; Borrego, D.; French, B.; Zablotska, L.B.; Adams, M.J.; Allodji, R.; de Vathaire, F.; Lee, C.; Brenner, A.V.; et al. Leukaemia and myeloid malignancy among people exposed to low doses (<100 mSv) of ionising radiation during childhood: A pooled analysis of nine historical cohort studies. Lancet Haematol. 2018, 5, e346–e358. [Google Scholar] [CrossRef]
- Hsu, W.L.; Preston, D.L.; Soda, M.; Sugiyama, H.; Funamoto, S.; Kodama, K.; Kimura, A.; Kamada, N.; Dohy, H.; Tomonaga, M.; et al. The incidence of leukemia, lymphoma and multiple myeloma among atomic bomb survivors: 1950–2001. Radiat. Res. 2013, 179, 361–382. [Google Scholar] [CrossRef] [Green Version]
- Darby, S. Residential radon, smoking and lung cancer. Radiat Res. 2005, 163, 696. [Google Scholar]
- Pearce, M.S.; Salotti, J.A.; Little, M.P.; McHugh, K.; Lee, C.; Kim, K.P.; Howe, N.L.; Ronckers, C.M.; Rajaraman, P.; Sir Craft, A.W.; et al. Radiation exposure from CT scans in childhood and subsequent risk of leukaemia and brain tumours: A retrospective cohort study. Lancet 2012, 380, 499–505. [Google Scholar] [CrossRef] [Green Version]
- Berrington de Gonzalez, A.; Salotti, J.A.; McHugh, K.; Little, M.P.; Harbron, R.W.; Lee, C.; Ntowe, E.; Braganza, M.Z.; Parker, L.; Rajaraman, P.; et al. Relationship between paediatric CT scans and subsequent risk of leukaemia and brain tumours: Assessment of the impact of underlying conditions. Br. J. Cancer 2016, 114, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Meulepas, J.M.; Ronckers, C.M.; Smets, A.M.J.B.; Nievelstein, R.A.J.; Gradowska, P.; Lee, C.; Jahnen, A.; van Straten, M.; de Wit, M.Y.; Zonnenberg, B.; et al. Radiation exposure from pediatric CT scans and subsequent cancer risk in the Netherlands. J. Natl. Cancer Inst. 2019, 111, 256–263. [Google Scholar] [CrossRef] [PubMed]
- Hauptmann, M.; Byrnes, G.; Cardis, E.; Bernier, M.O.; Blettner, M.; Dabin, J.; Engels, H.; Istad, T.S.; Johansen, C.; Kaijser, M.; et al. Brain cancer after radiation exposure from CT examinations of children and young adults: Results from the EPI-CT cohort study. Lancet Oncol. 2023, 24, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Leuraud, K.; Richardson, D.B.; Cardis, E.; Daniels, R.D.; Gillies, M.; O’Hagan, J.A.; Hamra, G.B.; Haylock, R.; Laurier, D.; Moissonnier, M.; et al. Ionising radiation and risk of death from leukaemia and lymphoma in radiation-monitored workers (INWORKS): An international cohort study. Lancet Haematol. 2015, 2, e276–e281. [Google Scholar] [CrossRef] [Green Version]
- Laurier, D.; Richardson, D.B.; Cardis, E.; Daniels, R.D.; Gillies, M.; O’Hagan, J.; Hamra, G.B.; Haylock, R.; Leuraud, K.; Moissonnier, M.; et al. The International Nuclear Workers Study (Inworks): A Collaborative Epidemiological Study to Improve Knowledge about Health Effects of Protracted Low-Dose Exposure. Radiat. Prot. Dosim. 2017, 173, 21–25. [Google Scholar] [CrossRef] [Green Version]
- Richardson, D.B.; Cardis, E.; Daniels, R.D.; Gillies, M.; O’Hagan, J.A.; Hamra, G.B.; Haylock, R.; Laurier, D.; Leuraud, K.; Moissonnier, M.; et al. Risk of cancer from occupational exposure to ionising radiation: Retrospective cohort study of workers in France, the United Kingdom, and the United States (INWORKS). BMJ 2015, 351, h5359. [Google Scholar] [CrossRef] [Green Version]
- Brenner, D.J.; Sachs, R.K. Estimating radiation-induced cancer risks at very low doses: Rationale for using a linear no-threshold approach. Radiat. Environ. Biophys. 2006, 44, 253–256. [Google Scholar] [CrossRef]
- Preston, D.L.; Shimizu, Y.; Pierce, D.A.; Suyama, A.; Mabuchi, K. Studies of mortality of atomic bomb survivors. Report 13: Solid cancer and noncancer disease mortality: 1950–1997. Radiat. Res. 2003, 160, 381–407. [Google Scholar] [CrossRef]
- Calabrese, E.J. Flaws in the LNT single-hit model for cancer risk: An historical assessment. Environ. Res. 2017, 158, 773–788. [Google Scholar] [CrossRef]
- Tubiana, M.; Aurengo, A.; Averbeck, D.; Masse, R. The debate on the use of linear no threshold for assessing the effects of low doses. J. Radiol. Prot. 2006, 26, 317–324. [Google Scholar] [CrossRef]
- Averbeck, D.; Salomaa, S.; Bouffler, S.; Ottolenghi, A.; Smyth, V.; Sabatier, L. Progress in low dose health risk research: Novel effects and new concepts in low dose radiobiology. Mutat. Res. Rev. Mutat. Res. 2018, 776, 46–69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sykes, P.J. Until there is a resolution of the pro-LNT/anti-LNT debate, we should head toward a more sensible graded approach for protection from low-dose ionizing radiation. Dose Response 2020, 18, 1559325820921651. [Google Scholar] [CrossRef]
- Tharmalingam, S.; Sreetharan, S.; Brooks, A.L.; Boreham, D.R. Re-evaluation of the linear no-threshold (LNT) model using newparadigms and modern molecular studies. Chem. Biol. Interact. 2019, 301, 54–67. [Google Scholar] [CrossRef] [PubMed]
- Brooks, A.L. 2012 USA DOE Program 1999. Available online: http://lowdose.energy.gov/ (accessed on 13 April 2023).
- Feinendegen, L.E. Evidence for beneficial low level radiation effects and radiation hormesis. Br. J. Radiol. 2005, 78, 3–7. [Google Scholar] [CrossRef]
- Planel, H.; Soleilhavoup, J.P.; Tixador, R.; Richoilley, G.; Conter, A.; Croute, F.; Caratero, C.; Gaubin, Y. Influence on cell proliferation of background radiation or exposure to very low, chronic gamma radiation. Health Phys. 1987, 52, 571–578. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J.; Dhawan, G.; Kapoor, R.; Iavicoli, I.; Calabrese, V. What is hormesis and its relevance to healthy aging and longevity? Biogerontology 2015, 16, 693–707. [Google Scholar] [CrossRef]
- Lau, Y.S.; Chew, M.T.; Alqahtani, A.; Jones, B.; Hill, M.A.; Nisbet, A.; Bradley, D.A. Low dose ionising radiation-induced hormesis: Therapeutic implications to human health. Appl. Sci. 2021, 11, 8909. [Google Scholar] [CrossRef]
- Tang, F.R.; Loke, W.K. Molecular mechanisms of low dose ionizing radiation-induced hormesis, adaptive responses, radioresistance, bystander effects, and genomic instability. Int. J. Radiat. Biol. 2015, 91, 13–27. [Google Scholar] [CrossRef]
- Polgár, S.; Schofield, P.N.; Madas, B.G. Datasets of in vitro clonogenic assays showing low dose hyper-radiosensitivity and induced radioresistance. Sci. Data 2022, 9, 555. [Google Scholar] [CrossRef]
- Dawood, A.; Mothersill, C.; Seymour, C. Low dose ionizing radiation and the immune response: What is the role of non-targeted effects? Int. J. Radiat. Biol. 2021, 97, 1368–1382. [Google Scholar] [CrossRef]
- Franco, N.; Lamartine, J.; Frouin, V.; Le Minter, P.; Petat, C.; Leplat, J.J.; Libert, F.; Gidrol, X.; Martin, M.T. Low-dose exposure to gamma rays induces specific gene regulations in normal human keratinocytes. Radiat. Res. 2005, 163, 623–635. [Google Scholar] [CrossRef] [PubMed]
- Sokolov, M.; Nguyen, V.; Neumann, R. Comparative analysis of whole-genome gene expression changes in cultured human embryonic stem cells in response to low, clinical diagnostic relevant, and high doses of ionizing radiation exposure. Int. J. Mol. Sci. 2015, 16, 14737–14748. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Belli, M.; Tabocchini, M.A. Ionizing radiation-induced epigenetic modifications and their relevance to radiation protection. Int. J. Mol. Sci. 2020, 21, 5993. [Google Scholar] [CrossRef] [PubMed]
- Sampadi, B.; Mullenders, L.H.F.; Vrieling, H. Low and high doses of ionizing radiation evoke discrete global (phospho)proteome responses. DNA Repair 2022, 113, 103305. [Google Scholar] [CrossRef]
- Rothkamm, K.; Löbrich, M. Evidence for a lack of DNA double-strand break repair in human cells exposed to very low X-ray doses. Proc. Natl. Acad. Sci. USA 2003, 100, 5057–5062. [Google Scholar] [CrossRef]
- Azzam, E.I.; de Toledo, S.M.; Gooding, T.; Little, J.B. Intercellular communication is involved in the bystander regulation of gene expression in human cells exposed to very low fluences of alpha particles. Radiat. Res. 1998, 150, 497–504. [Google Scholar] [CrossRef]
- Barcellos-Hoff, M.H.; Brooks, A.L. Extracellular signaling through the microenvironment: A hypothesis relating carcinogenesis, bystander effects, and genomic instability. Radiat. Res. 2001, 156, 618–627. [Google Scholar] [CrossRef]
- Foray, N.; Bourguignon, M.; Hamada, N. Individual response to ionizing radiation. Mutat. Res. Rev. Mutat. Res. 2016, 770, 369–386. [Google Scholar] [CrossRef]
- Tsukimoto, M.; Homma, T.; Ohshima, Y.; Kojima, S. Involvement of purinergic signaling in cellular response to gamma radiation. Radiat. Res. 2010, 173, 298–309. [Google Scholar] [CrossRef]
- Anuranjani; Bala, M. Concerted action of Nrf2-ARE pathway, MRN complex, HMGB1 and inflammatory cytokines—Implication in modification of radiation damage. Redox Biol. 2014, 2, 832–846. [Google Scholar] [CrossRef] [Green Version]
- Bensimon, A.; Aebersold, R.; Shiloh, Y. Beyond ATM: The protein kinase landscape of the DNA damage response. FEBS Lett. 2011, 585, 1625–1639. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maeda, M.; Tomita, M.; Maeda, M.; Matsumoto, H.; Usami, N.; Kume, K.; Kobayashi, K. Exposure of the cytoplasm to low-dose X-rays modifies ataxia telangiectasia mutated-mediated DNA damage responses. Sci. Rep. 2021, 11, 13113. [Google Scholar] [CrossRef] [PubMed]
- Berthel, E.; Foray, N.; Ferlazzo, M.L. The nucleoshuttling of the ATM protein: A unified model to describe the individual response to high- and low-dose of radiation? Cancers 2019, 11, 905. [Google Scholar] [CrossRef] [Green Version]
- Stagni, V.; Cirotti, C.; Barilà, D. Ataxia-telangiectasia mutated kinase in the control of oxidative stress, mitochondria, and autophagy in cancer: A maestro with a large orchestra. Front. Oncol. 2018, 8, 73. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J.; Baldwin, L.A. Defining hormesis. Hum. Exp. Toxicol. 2002, 21, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J. Linear non-threshold (LNT) fails numerous toxicological stress tests: Implications for continued policy use. Chem. Biol. Interact. 2022, 365, 110064. [Google Scholar] [CrossRef] [PubMed]
- Agathokleous, E.; Calabrese, E.J. Hormesis: A general biological principle. Chem. Res. Toxicol. 2022, 35, 547–549. [Google Scholar] [CrossRef]
- Sthijns, M.M.; Weseler, A.R.; Bast, A.; Haenen, G.R. Time in redox adaptation processes: From evolution to hormesis. Int. J. Mol. Sci. 2016, 17, 1649. [Google Scholar] [CrossRef] [Green Version]
- Vaiserman, A.M. Radiation hormesis: Historical perspective and implications for low-dose cancer risk assessment. Dose Response 2010, 8, 172–191. [Google Scholar] [CrossRef]
- Feinendegen, L.E. Quantification of adaptive protection following low-dose irradiation. Health Phys. 2016, 110, 276–280. [Google Scholar] [CrossRef]
- Sies, H.; Feinendegen, L.E. Radiation hormesis: The link to nanomolar hydrogen peroxide. Antioxid. Redox Signal. 2017, 27, 596–598. [Google Scholar] [CrossRef] [PubMed]
- Murray, D.; Mirzayans, R. Nonlinearities in the cellular response to ionizing radiation and the role of p53 therein. Int. J. Radiat. Biol. 2021, 97, 1088–1098. [Google Scholar] [CrossRef] [PubMed]
- Satta, L.; Augusti-Tocco, G.; Ceccarelli, R.; Esposito, A.; Fiore, M.; Paggi, P.; Poggesi, I.; Ricordy, R.; Scarsella, G.; Cundari, E. Low environmental radiation background impairs biological defence of the yeast Saccharomyces cerevisiae to chemical radiomimetic agents. Mutat. Res. 1995, 347, 129–133. [Google Scholar] [CrossRef] [PubMed]
- Satta, L.; Antonelli, F.; Belli, M.; Sapora, O.; Simone, G.; Sorrentino, E.; Tabocchini, M.A.; Amicarelli, F.; Ara, C.; Cerù, M.P.; et al. Influence of a low background radiation environment on biochemical and biological responses in V79 cells. Radiat. Environ. Biophys. 2002, 41, 217–224. [Google Scholar] [CrossRef]
- Fratini, E.; Carbone, C.; Capece, D.; Esposito, G.; Simone, G.; Tabocchini, M.A.; Tomasi, M.; Belli, M.; Satta, L. Low-radiation environment affects the development of protection mechanisms in V79 cells. Radiat. Environ. Biophys. 2015, 54, 183–194. [Google Scholar] [CrossRef]
- Hwang, S.; Jeong, H.; Hong, E.H.; Joo, H.M.; Cho, K.S.; Nam, S.Y. Low-dose ionizing radiation alleviates Aβ42-induced cell death via regulating AKT and p38 pathways in Drosophila Alzheimer’s disease models. Biol. Open 2019, 8, bio036657. [Google Scholar] [CrossRef] [Green Version]
- Lowe, X.R.; Bhattacharya, S.; Marchetti, F.; Wyrobek, A.J. Early brain response to low-dose radiation exposure involves molecular networks and pathways associated with cognitive functions, advanced aging and Alzheimer’s disease. Radiat. Res. 2009, 171, 53–65. [Google Scholar] [CrossRef] [Green Version]
- Cuttler, J.M.; Abdellah, E.; Goldberg, Y.; Al-Shamaa, S.; Symons, S.P.; Black, S.E.; Freedman, M. Low doses of ionizing radiation as a treatment for Alzheimer’s disease: A pilot study. J. Alzheimers Dis. 2021, 80, 1119–1128. [Google Scholar] [CrossRef]
- Cuttler, J.M.; Lamet, M.S.; Calabrese, E.J. Treatment of early-stage Alzheimer’s disease with CT scans of the brain: A case report. Dose Response 2022, 20, 15593258221078392. [Google Scholar] [CrossRef]
- Jebelli, J.; Hamper, M.C.; Van Quelef, D.; Caraballo, D.; Hartmann, J.; Kumi-Diaka, J. The potential therapeutic effects of low-dose ionizing radiation in Alzheimer’s disease. Cureus 2022, 14, e23461. [Google Scholar] [CrossRef]
- Bartolini, D.; Tew, K.D.; Marinelli, R.; Galli, F.; Wang, G.Y. Nrf2-modulation by seleno-hormetic agents and its potential for radiation protection. Biofactors 2020, 46, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Singla, N.; Chadha, V.D.; Dhawan, D.K. A concept of radiation hormesis: Stimulation of antioxidant machinery in rats by low dose ionizing radiation. Hell. J. Nucl. Med. 2019, 22, 43–48. [Google Scholar]
- Colceriu-Simon, I.M.; Hedesiu, M.; Toma, V.; Armencea, G.; Moldovan, A.; Stiufiuc, G.; Culic, B.; Tarmure, V.; Dinu, C.; Berindan-Neagoe, I.; et al. The Effects of Low-Dose Irradiation on Human Saliva: A Surface-Enhanced Raman Spectroscopy Study. Diagnostics 2019, 9, 101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaushik, N.; Kim, M.J.; Kaushik, N.K.; Myung, J.K.; Choi, M.Y.; Kang, J.H.; Cha, H.J.; Kim, C.S.; Nam, S.Y.; Lee, S.J. Low dose radiation regulates BRAF-induced thyroid cellular dysfunction and transformation. Cell Commun. Signal. 2019, 17, 12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Redpath, J.L.; Liang, D.; Taylor, T.H.; Christie, C.; Elmore, E. The shape of the dose-response curve for radiation-induced neoplastic transformation in vitro: Evidence for an adaptive response against neoplastic transformation at low doses of low-LET radiation. Radiat. Res. 2001, 156, 700–707. [Google Scholar] [CrossRef]
- Scott, B.R. Radiation-hormesis phenotypes, the related mechanisms and implications for disease prevention and therapy. J. Cell Commun. Signal. 2014, 8, 341–352. [Google Scholar] [CrossRef] [Green Version]
- Liu, S.Z. Nonlinear dose-response relationship in the immune system following exposure to ionizing radiation: Mechanisms and implications. Nonlinearity Biol. Toxicol. Med. 2003, 1, 71–92. [Google Scholar] [CrossRef] [Green Version]
- Liang, X.; So, Y.; Cui, J.; Ma, K.; Xu, X.; Zhao, Y.; Cai, L.; Li, W. The low-dose ionizing radiation stimulates cell proliferation via activation of the MAPK/ERK pathway in rat cultured mesenchymal stem cells. J. Radiat. Res. 2011, 52, 380–386. [Google Scholar] [CrossRef] [Green Version]
- Liang, X.; Gu, J.; Yu, D.; Wang, G.; Zhou, L.; Zhang, X.; Zhao, Y.; Chen, X.; Zheng, S.; Liu, Q.; et al. Low-dose radiation induces cell proliferation in human embryonic lung fibroblasts but not in lung cancer cells: Importance of ERK1/2 and AKT signaling pathways. Dose-Response 2016, 14, 1559325815622174. [Google Scholar] [CrossRef] [Green Version]
- Yang, G.; Yu, D.; Li, W.; Zhao, Y.; Wen, X.; Liang, X.; Zhang, X.; Zhou, L.; Hu, J.; Niu, C.; et al. Distinct biological effects of low-dose radiation on normal and cancerous human lung cells are mediated by ATM signaling. Oncotarget 2016, 7, 71856–71872. [Google Scholar] [CrossRef] [Green Version]
- Jiang, H.; Li, W.; Li, X.; Cai, L.; Wang, G. Low-dose radiation induces adaptive response in normal cells, but not in tumor cells: In vitro and in vivo studies. J. Radiat. Res. 2008, 49, 219–230. [Google Scholar] [CrossRef] [Green Version]
- Cheda, A.; Wrembel-Wargocka, J.; Lisiak, E.; Nowosielska, E.M.; Marciniak, M.; Janiak, M.K. Single low doses of x-rays inhibit the development of experimental tumor metastases and trigger the activities of NK cells in mice. Radiat. Res. 2004, 161, 335–340. [Google Scholar] [CrossRef]
- Hashimoto, S.; Shirato, H.; Hosokawa, M.; Nishioka, T.; Kuramitsu, Y.; Matushita, K.; Kobayashi, M.; Miyasaka, K. The suppression of metastases and the change in host immune response after low-dose total-body irradiation in tumor-bearing rats. Radiat. Res. 1999, 151, 717–724. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, M.; Kojima, S. Suppression of atopic dermatitis and tumor metastasis in mice by small amounts of radon. Radiat. Res. 2006, 165, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Scott, B.R.; Tharmalingam, S. The LNT model for cancer induction is not supported by radiobiological data. Chem. Biol. Interact. 2019, 301, 34–53. [Google Scholar] [CrossRef] [PubMed]
- Burtt, J.J.; Thompson, P.A.; Lafrenie, R.M. Non-targeted effects and radiation-induced carcinogenesis: A review. J. Radiol. Prot. 2016, 36, R23–R35. [Google Scholar] [CrossRef] [Green Version]
- Mitchel, R.E. Low doses of radiation are protective in vitro and in vivo: Evolutionary origins. Dose Response 2006, 4, 75–90. [Google Scholar] [CrossRef]
- Olivieri, G.; Bodycote, J.; Wolff, S. Adaptive response of human lymphocytes to low concentrations of radioactive thymidine. Science 1984, 223, 594–597. [Google Scholar] [CrossRef]
- Wolff, S.; Afzal, V.; Wiencke, J.K.; Olivieri, G.; Michaeli, A. Human lymphocytes exposed to low doses of ionizing radiations become refractory to high doses of radiation as well as to chemical mutagens that induce double-strand breaks in DNA. Int. J. Radiat. Biol. Relat. Stud. Phys. Chem. Med. 1988, 53, 39–47. [Google Scholar] [CrossRef]
- Wolff, S. Aspects of the adaptive response to very low doses of radiation and other agents. Mutat. Res. 1996, 358, 135–142. [Google Scholar] [CrossRef]
- Crawford, D.R.; Davies, K.J. Adaptive response and oxidative stress. Environ. Health Perspect. 1994, 102 (Suppl. S10), 25–28. [Google Scholar] [CrossRef]
- Kang, C.M.; Park, K.P.; Cho, C.K.; Seo, J.S.; Park, W.Y.; Lee, S.J.; Lee, Y.S. Hspa4 (HSP70) is involved in the radioadaptive response: Results from mouse splenocytes. Radiat. Res. 2002, 157, 650–655. [Google Scholar] [CrossRef]
- Lee, Y.J.; Park, G.H.; Cho, H.N.; Cho, C.K.; Park, Y.M.; Lee, S.J.; Lee, Y.S. Induction of adaptive response by low-dose radiation in RIF cells transfected with Hspb1 (Hsp25) or inducible Hspa (Hsp70). Radiat. Res. 2002, 157, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Varès, G.; Wang, B.; Tanaka, K.; Kakimoto, A.; Eguchi-Kasai, K.; Nenoi, M. Mutagenic adaptive response to high-LET radiation in human lymphoblastoid cells exposed to X-rays. Mutat. Res. 2011, 706, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Nenoi, M.; Wang, B.; Vares, G. In vivo radioadaptive response: A review of studies relevant to radiation-induced cancer risk. Hum. Exp. Toxicol. 2015, 34, 272–283. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Azzam, E.I.; Raaphorst, G.P.; Mitchel, R.E. Radiation-induced adaptive response for protection against micronucleus formation and neoplastic transformation in C3H 10T1/2 mouse embryo cells. Radiat. Res. 1994, 138, S28–S31. [Google Scholar] [CrossRef]
- Yu, H.; Liu, N.; Wang, H.; Shang, Q.; Jiang, P.; Zhang, Y. Different responses of tumor and normal cells to low-dose radiation. Contemp. Oncol. 2013, 17, 356–362. [Google Scholar] [CrossRef] [Green Version]
- Ryan, L.A.; Seymour, C.B.; Joiner, M.C.; Mothersill, C.E. Radiation-induced adaptive response is not seen in cell lines showing a bystander effect but is seen in lines showing HRS/IRR response. Int. J. Radiat. Biol. 2009, 85, 87–95. [Google Scholar] [CrossRef]
- Wang, B.; Ninomiya, Y.; Tanaka, K.; Maruyama, K.; Varès, G.; Eguchi-Kasai, K.; Nenoi, M. Adaptive response of low linear energy transfer X-rays for protection against high linear energy transfer accelerated heavy ion-induced teratogenesis. Birth Defects Res. B Dev. Reprod. Toxicol. 2012, 95, 379–385. [Google Scholar] [CrossRef]
- Katsube, T.; Wang, B.; Tanaka, K.; Ninomiya, Y.; Hirakawa, H.; Liu, C.; Maruyama, K.; Vares, G.; Liu, Q.; Kito, S.; et al. Synergistic effects of chronic restraint-induced stress and low-dose 56Fe-particle irradiation on induction of chromosomal aberrations in Trp53-heterozygous mice. Radiat. Res. 2021, 196, 100–112. [Google Scholar] [CrossRef]
- Dionet, C.; Müller-Barthélémy, M.; Marceau, G.; Denis, J.M.; Averbeck, D.; Gueulette, J.; Sapin, V.; Pereira, B.; Tchirkov, A.; Chautard, E.; et al. Different dose rate-dependent responses of human melanoma cells and fibroblasts to low dose fast neutrons. Int. J. Radiat. Biol. 2016, 92, 527–535. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Su, S.; Zhou, S.; Wen, C.; Zou, J.; Zhang, D.; Geng, J.; Yang, M.; Liu, M.; Li, L.; Wen, W. Evidence for adaptive response in a molecular epidemiological study of the inhabitants of a high background-radiation area of Yangjiang, China. Health Phys. 2018, 115, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, E.N.; Karuppasamy, C.V.; Kumar, V.A.; Soren, D.C.; Kumar, P.R.V.; Koya, P.K.M.; Jaikrishan, G.; Das, B. Radioadaptive response in peripheral blood lymphocytes of individuals residing in high-level natural radiation areas of Kerala in the southwest coast of India. Mutagenesis 2016, 32, 267–273. [Google Scholar] [CrossRef] [Green Version]
- Jain, V.; Saini, D.; Kumar, P.R.V.; Jaikrishan, G.; Das, B. Efficient repair of DNA double strand breaks in individuals from high level natural radiation areas of Kerala coast, south-west India. Mutat. Res. 2017, 806, 39–50. [Google Scholar] [CrossRef] [PubMed]
- de Toledo, S.M.; Asaad, N.; Venkatachalam, P.; Li, L.; Howell, R.W.; Spitz, D.R.; Azzam, E.I. Adaptive responses to low-dose/low-dose-rate gamma rays in normal human fibroblasts: The role of growth architecture and oxidative metabolism. Radiat. Res. 2006, 166, 849–857. [Google Scholar] [CrossRef] [PubMed]
- Bravard, A.; Luccioni, C.; Moustacchi, E.; Rigaud, O. Contribution of antioxidant enzymes to the adaptive response to ionizing radiation of human lymphoblasts. Int. J. Radiat. Biol. 1999, 75, 639–645. [Google Scholar] [CrossRef]
- Paraswani, N.; Thoh, M.; Bhilwade, H.N.; Ghosh, A. Early antioxidant responses via the concerted activation of NF-κB and Nrf2 characterize the gamma-radiation-induced adaptive response in quiescent human peripheral blood mononuclear cells. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2018, 831, 50–61. [Google Scholar] [CrossRef]
- Matsumoto, H.; Takahashi, A.; Ohnishi, T. Nitric oxide radicals choreograph a radioadaptive response. Cancer Res. 2007, 67, 8574–8579. [Google Scholar] [CrossRef] [Green Version]
- Lall, R.; Ganapathy, S.; Yang, M.; Xiao, S.; Xu, T.; Su, H.; Shadfan, M.; Asara, J.M.; Ha, C.S.; Ben-Sahra, I.; et al. Low-dose radiation exposure induces a HIF-1-mediated adaptive and protective metabolic response. Cell Death Differ. 2014, 21, 836–844. [Google Scholar] [CrossRef]
- Dieriks, B.; De Vos, W.; Baatout, S.; Van Oostveldt, P. Repeated exposure of human fibroblasts to ionizing radiation reveals an adaptive response that is not mediated by interleukin-6 or TGF-β. Mutat Res. 2011, 715, 19–24. [Google Scholar] [CrossRef]
- Barcellos-Hoff, M.H. The radiobiology of TGFβ. Semin. Cancer Biol. 2022, 86, 857–867. [Google Scholar] [CrossRef] [PubMed]
- Hooker, A.M.; Bhat, M.; Day, T.K.; Lane, J.M.; Swinburne, S.J.; Morley, A.A.; Sykes, P.J. The linear no-threshold model does not hold for low-dose ionizing radiation. Radiat. Res. 2004, 162, 447–452. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Day, T.K.; Hooker, A.M.; Zeng, G.; Sykes, P.J. Low dose X-radiation adaptive response in spleen and prostate of Atm knockout heterozygous mice. Int. J. Radiat. Biol. 2007, 83, 523–534. [Google Scholar] [CrossRef] [PubMed]
- Day, T.K.; Zeng, G.; Hooker, A.M.; Bhat, M.; Scott, B.R.; Turner, D.R.; Sykes, P.J. Extremely low priming doses of X radiation induce an adaptive response for chromosomal inversions in pKZ1 mouse prostate. Radiat. Res. 2006, 166, 757–766. [Google Scholar] [CrossRef] [Green Version]
- De Toledo, S.M.; Buonanno, M.; Li, M.; Asaad, N.; Qin, Y.; Gonon, G.; Shim, G.; Galdass, M.; Boateng, Y.; Zhang, J.; et al. The impact of adaptive and non-targeted effects in the biological responses to low dose/low fluence ionizing radiation: The modulating effect of linear energy transfer. Health Phys. 2011, 100, 290–292. [Google Scholar] [CrossRef] [Green Version]
- Guéguen, Y.; Bontemps, A.; Ebrahimian, T.G. Adaptive responses to low doses of radiation or chemicals: Their cellular and molecular mechanisms. Cell. Mol. Life Sci. 2019, 76, 1255–1273. [Google Scholar] [CrossRef]
- Devic, C.; Ferlazzo, M.L.; Berthel, E.; Foray, N. Influence of individual radiosensitivity on the hormesis phenomenon: Toward a mechanistic explanation based on the nucleoshuttling of ATM protein. Dose Response 2020, 18, 1559325820913784. [Google Scholar] [CrossRef]
- Kabilan, U.; Graber, T.E.; Alain, T.; Klokov, D. Ionizing radiation and translation control: A link to radiation hormesis? Int. J. Mol. Sci. 2020, 21, 6650. [Google Scholar] [CrossRef]
- Baechler, B.L.; Bloemberg, D.; Quadrilatero, J. Mitophagy regulates mitochondrial network signaling, oxidative stress, and apoptosis during myoblast differentiation. Autophagy 2019, 15, 1606–1619. [Google Scholar] [CrossRef]
- Ghafourifar, P.; Cadenas, E. Mitochondrial nitric oxide synthase. Trends Pharmacol. Sci. 2005, 26, 190–195. [Google Scholar] [CrossRef]
- Joiner, M.C.; Lambin, P.; Malaise, E.P.; Robson, T.; Arrand, J.E.; Skov, K.A.; Marples, B. Hypersensitivity to very-low single radiation doses: Its relationship to the adaptive response and induced radioresistance. Mutat. Res. 1996, 358, 171–183. [Google Scholar] [CrossRef] [PubMed]
- Joiner, M.C.; Lambin, P.; Marples, B. Adaptive response and induced resistance. Comptes Rendus Acad. Sci. III 1999, 322, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Joiner, M.C.; Marples, B.; Lambin, P.; Short, S.C.; Turesson, I. Low-dose hypersensitivity: Current status and possible mechanisms. Int. J. Radiat. Oncol. Biol. Phys. 2001, 49, 379–389. [Google Scholar] [CrossRef] [PubMed]
- Heuskin, A.C.; Michiels, C.; Lucas, S. Low dose hypersensitivity following in vitro cell irradiation with charged particles: Is the mechanism the same as with X-ray radiation? Int. J. Radiat. Biol. 2014, 90, 81–89. [Google Scholar] [CrossRef]
- Marples, B.; Lambin, P.; Skov, K.A.; Joiner, M.C. Low dose hyper-radiosensitivity and increased radioresistance in mammalian cells. Int. J. Radiat. Biol. 1997, 71, 721–735. [Google Scholar] [CrossRef]
- Park, S.Y.; Lee, C.J.; Choi, J.H.; Kim, J.H.; Kim, J.W.; Kim, J.Y.; Nam, J.S. The JAK2/STAT3/CCND2 axis promotes colorectal cancer stem cell persistence and radioresistance. J. Exp. Clin. Cancer Res. 2019, 38, 399. [Google Scholar] [CrossRef] [Green Version]
- Schulz, A.; Meyer, F.; Dubrovska, A.; Borgmann, K. Cancer stem cells and radioresistance: DNA Repair and beyond. Cancers 2019, 11, 862. [Google Scholar] [CrossRef] [Green Version]
- Shimura, T.; Sasatani, M.; Kawai, H.; Kamiya, K.; Kobayashi, J.; Komatsu, K.; Kunugita, N. A comparison of radiation-induced mitochondrial damage between neural progenitor stem cells and differentiated cells. Cell Cycle 2017, 16, 565–573. [Google Scholar] [CrossRef] [Green Version]
- Marples, B.; Adomat, H.; Koch, C.J.; Skov, K.A. Response of V79 cells to low doses of X-rays and negative pi-mesons: Clonogenic survival and DNA strand breaks. Int. J. Radiat. Biol. 1996, 70, 429–436. [Google Scholar] [CrossRef]
- Marples, B.; Wouters, B.G.; Collis, S.J.; Chalmers, A.J.; Joiner, M.C. Low-dose hyper-radiosensitivity: A consequence of ineffective cell cycle arrest of radiation-damaged G2-phase cells. Radiat. Res. 2004, 161, 247–255. [Google Scholar] [CrossRef]
- Marples, B.; Collis, S.J. Low-dose hyper-radiosensitivity: Past, present, and future. Int. J. Radiat. Oncol. Biol. Phys. 2008, 70, 1310–1318. [Google Scholar] [CrossRef] [PubMed]
- Krueger, S.A.; Wilson, G.D.; Piasentin, E.; Joiner, M.C.; Marples, B. The effects of G2-phase enrichment and checkpoint abrogation on low-dose hyper-radiosensitivity. Int. J. Radiat. Oncol. Biol. Phys. 2010, 77, 1509–1517. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernet, M.; Mégnin-Chanet, F.; Hall, J.; Favaudon, V. Control of the G2/M checkpoints after exposure to low doses of ionizing radiation: Implications for hyper-radiosensitivity. DNA Repair 2010, 9, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Olobatuyi, O.; de Vries, G.; Hillen, T. Effects of G2-checkpoint dynamics on low-dose hyperradiosensitivity. J. Math. Biol. 2018, 77, 1969–1997. [Google Scholar] [CrossRef] [PubMed]
- Krueger, S.A.; Joiner, M.C.; Weinfeld, M.; Piasentin, E.; Marples, B. Role of apoptosis in low-dose hyper-radiosensitivity. Radiat. Res. 2007, 167, 260–267. [Google Scholar] [CrossRef]
- Wykes, S.M.; Piasentin, E.; Joiner, M.C.; Wilson, G.D.; Marples, B. Low-dose hyper-radiosensitivity is not caused by a failure to recognize DNA double-strand breaks. Radiat. Res. 2006, 165, 516–524. [Google Scholar] [CrossRef]
- Xue, L.; Yu, D.; Furusawa, Y.; Okayasu, R.; Tong, J.; Cao, J.; Fan, S. Regulation of ATM in DNA double strand break repair accounts for the radiosensitivity in human cells exposed to high linear energy transfer ionizing radiation. Mutat. Res. 2009, 670, 15–23. [Google Scholar] [CrossRef]
- Xue, L.; Furusawa, Y.; Yu, D. ATR signaling cooperates with ATM in the mechanism of low dose hypersensitivity induced by carbon ion beam. DNA Repair 2015, 34, 1–8. [Google Scholar] [CrossRef]
- Enns, L.; Rasouli-Nia, A.; Hendze, L.M.; Marples, B.; Weinfeld, M. Association of ATM activation and DNA repair with induced radioresistance after low-dose irradiation. Radiat. Prot. Dosim. 2015, 166, 131–136. [Google Scholar] [CrossRef] [Green Version]
- Burdak-Rothkamm, S.; Rothkamm, K.; Prise, K.M. ATM acts downstream of ATR in the DNA damage response signaling of bystander cells. Cancer Res. 2008, 68, 7059–7065. [Google Scholar] [CrossRef] [Green Version]
- Shimura, T.; Kunugita, N. Mitochondrial reactive oxygen species-mediated genomic instability in low-dose irradiated human cells through nuclear retention of cyclin D1. Cell Cycle 2016, 15, 1410–1414. [Google Scholar] [CrossRef]
- Hanson, I.; Pitman, K.E.; Altanerova, U.; Altaner, Č.; Malinen, E.; Edin, N.F.J. Low-dose-rate radiation-induced secretion of TGF-β3 together with an activator in small extracellular vesicles modifies low-dose hyper-radiosensitivity through ALK1 binding. Int. J. Mol. Sci. 2022, 23, 8147. [Google Scholar] [CrossRef]
- Maeda, M.; Usami, N.; Kobayashi, K. Low-dose hypersensitivity in nucleus-irradiated V79 Cells studied with synchrotron X-ray microbeam. J. Radiat. Res. 2008, 49, 171–180. [Google Scholar] [CrossRef] [Green Version]
- Maeda, M.; Tomita, M.; Usami, N.; Kobayashi, K. Bystander cell death is modified by sites of energy deposition within cell irradiated with a synchrotron X-ray microbeam. Radiat. Res. 2010, 174, 37–45. [Google Scholar] [CrossRef]
- Zhang, B.; Davidson, M.M.; Zhou, H.; Wang, C.; Walker, W.F.; Hei, T.K. Cytoplasmic irradiation results in mitochondrial dysfunction and DRP1-dependent mitochondrial fission. Cancer Res. 2013, 73, 6700–6710. [Google Scholar] [CrossRef] [Green Version]
- Wu, J.; Zhang, B.; Wu, Y.R.; Davidson, M.M.; Hei, T.K. Targeted cytoplasmic irradiation and autophagy. Mutat. Res. 2017, 806, 88–97. [Google Scholar] [CrossRef]
- Chandna, S.; Dwarakanath, B.S.; Khaitan, D.; Mathew, T.L.; Jain, V. Low-dose radiation hypersensitivity in human tumor cell lines: Effects of cell-cell contact and nutritional deprivation. Radiat. Res. 2002, 157, 516–525. [Google Scholar] [CrossRef]
- Ghosh, S.; Kumar, A.; Chandna, S. Connexin-43 downregulation in G2/M phase enriched tumour cells causes extensive low-dose hyper-radiosensitivity (HRS) associated with mitochondrial apoptotic events. Cancer Lett. 2015, 363, 46–59. [Google Scholar] [CrossRef]
- Aasen, T.; Mesnil, M.; Naus, C.C.; Lampe, P.D.; Laird, D.W. Gap junctions and cancer: Communicating for 50 years. Nat. Rev. Cancer 2016, 16, 775–788. [Google Scholar] [CrossRef] [Green Version]
- Kutova, O.M.; Pospelov, A.D.; Balalaeva, I.V. The multifaceted role of connexins in tumor microenvironment initiation and maintenance. Biology 2023, 12, 204. [Google Scholar] [CrossRef]
- Mathur, A.; Kumar, A.; Babu, B.; Chandna, S. In vitro mesenchymal-epithelial transition in NIH3T3 fibroblasts results in onset of low-dose radiation hypersensitivity coupled with attenuated connexin-43 response. Biochim. Biophys. Acta Gen. Subj. 2018, 1862, 414–426. [Google Scholar] [CrossRef] [PubMed]
- Prise, K.M.; O’Sullivan, J.M. Radiation-induced bystander signaling in cancer therapy. Nat. Rev. Cancer 2009, 9, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Azzam, E.I.; de Toledo, S.M.; Little, J.B. Oxidative metabolism, gap junctions and the ionizing radiation-induced bystander effect. Oncogene 2003, 22, 7050–7057. [Google Scholar] [CrossRef] [Green Version]
- Buonanno, M.; de Toledo, S.M.; Pain, D.; Azzam, E.I. Long-term consequences of radiation-induced bystander effects depend on radiation quality and dose and correlate with oxidative stress. Radiat. Res. 2011, 175, 405–415. [Google Scholar] [CrossRef] [Green Version]
- Yu, Q.; Li, P.; Weng, M.; Wu, S.; Zhang, Y.; Chen, X.; Zhang, Q.; Shen, G.; Ding, X.; Fu, S. Nano-vesicles are a potential tool to monitor therapeutic efficacy of carbon ion radiotherapy in prostate cancer. J. Biomed. Nanotechnol. 2018, 14, 168–178. [Google Scholar] [CrossRef]
- He, C.; Li, L.; Wang, L.; Meng, W.; Hao, Y.; Zhu, G. Exosome-mediated cellular crosstalk within the tumor microenvironment upon irradiation. Cancer Biol. Med. 2021, 18, 21–33. [Google Scholar] [CrossRef]
- Belyakov, O.V.; Mitchell, S.A.; Parikh, D.; Randers-Pehrson, G.; Marino, S.A.; Amundson, S.A.; Geard, C.R.; Brenner, D.J. Biological effects in unirradiated human tissue induced by radiation damage up to 1 mm away. Proc. Natl. Acad. Sci. USA 2005, 102, 14203–14208. [Google Scholar] [CrossRef]
- Furlong, H.; Mothersill, C.; Lyng, F.M.; Howe, O. Apoptosis is signaled early by low doses of ionising radiation in a radiation induced bystander effect. Mutat. Res. 2013, 741–742, 35–43. [Google Scholar] [CrossRef]
- Portess, D.I.; Bauer, G.; Hill, M.A.; O’Neill, P. Low-dose irradiation of non-transformed cells stimulates the selective removal of precancerous cells via intercellular induction of apoptosis. Cancer Res. 2007, 67, 1246–1253. [Google Scholar] [CrossRef] [Green Version]
- Klammer, H.; Ladenov, E.; Li, F.; Iliakis, G. Bystander effects as manifestation of intercellular communication of DNA damage and of the cellular oxidative status. Cancer Lett. 2015, 356, 58–71. [Google Scholar] [CrossRef]
- Hei, T.K.; Zhou, H.; Chai, Y.; Ponnaiya, B.; Ivanov, V.N. Radiation induced non-targeted response: Mechanism and potential clinical implications. Curr. Mol. Pharmacol. 2011, 4, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Qin, F.; Chen, G.; Yu, K.N.; Yang, M.; Cao, W.; Kong, P.; Peng, S.; Sun, M.; Nie, L.; Han, W. Golgi phosphoprotein 3 mediates radiation-induced bystander effect via ERK/EGR1/TNF-α signal axis. Antioxidants 2022, 11, 2172. [Google Scholar] [CrossRef] [PubMed]
- Yokota, Y.; Funayama, T.; Mutou-Yoshihara, Y.; Ikeda, H.; Kobayashi, Y. The bystander cell-killing effect mediated by nitric oxide in normal human fibroblasts varies with irradiation dose but not with radiation quality. Int. J. Radiat. Biol. 2015, 91, 383–388. [Google Scholar] [CrossRef]
- Tartier, L.; Gilchrist, S.; Burdak-Rothkamm, S.; Folkard, M.; Prise, K.M. Cytoplasmic irradiation induces mitochondrial-dependent 53BP1 protein relocalization in irradiated and bystander cells. Cancer Res. 2007, 67, 5872–5879. [Google Scholar] [CrossRef] [Green Version]
- Rustom, A.; Saffrich, R.; Markovic, I.; Walther, P.; Gerdes, H.H. Nanotubular highways for intercellular organelle transport. Science 2004, 303, 1007–1010. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chauveau, A.; Aucher, A.; Eissmann, P.; Vivier, E.; Davis, D.M. Membrane nanotubes facilitate long-distance interactions between natural killer cells and target cells. Proc. Natl. Acad. Sci. USA 2010, 107, 5545–5550. [Google Scholar] [CrossRef]
- Lu, J.; Zheng, X.; Li, F.; Yu, Y.; Chen, Z.; Liu, Z.; Wang, Z.; Xu, H.; Yang, W. Tunneling nanotubes promote intercellular mitochondria transfer followed by increased invasiveness in bladder cancer cells. Oncotarget 2017, 8, 15539–15552. [Google Scholar] [CrossRef] [Green Version]
- Gong, W.; Pan, W.; He, W.; Huang, M.; Zhang, J.; Gu, Z.; Zhang, D.; Yang, Z.; Qu, J. Superresolution iagig of the dynaic cleavage of intercellular tunneling nanotubes. Front. Optoelectron. 2020, 13, 318–326. [Google Scholar] [CrossRef]
- Jin, S.; Cordes, N. ATM controls DNA repair and mitochondria transfer between neighboring cells. Cell Commun. Signal. 2019, 17, 144. [Google Scholar] [CrossRef] [Green Version]
- Choy, K.R.; Watters, D.J. Neurodegeneration in ataxia-telangiectasia: Multiple roles of ATM kinase in cellular homeostasis. Dev. Dyn. 2018, 247, 33–46. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Lee, J.H.; Paull, T.T.; Gehrke, S.; D’Alessandro, A.; Dou, Q.; Gladyshev, V.N.; Schroeder, E.A.; Steyl, S.K.; Christian, B.E.; et al. Mitochondrial redox sensing by the kinase ATM maintains cellular antioxidant capacity. Sci. Signal. 2018, 11, eaaq0702. [Google Scholar] [CrossRef] [Green Version]
- Saha, T.; Solomon, J.; Samson, A.O.; Gil-Henn, H. Invasion and Metastasis as a Central Hallmark of Breast Cancer. J. Clin. Med. 2021, 10, 3498. [Google Scholar] [CrossRef]
- Mori, D.; Miyagawa, S.; Kawamura, T.; Yoshioka, D.; Hata, H.; Ueno, T.; Toda, K.; Kuratani, T.; Oota, M.; Kawai, K.; et al. Mitochondrial transfer induced by adipose-derived mesenchymal stem cell transplantation improves cardiac function in rat models of ischemic cardiomyopathy. Cell. Transplant. 2023, 32, 9636897221148457. [Google Scholar] [CrossRef] [PubMed]
- Fan, Q.; Maejima, Y.; Wei, L.; Nakagama, S.; Shiheido-Watanabe, Y.; Sasano, T. The pathophysiological significance of “mitochondrial ejection” from cells. Biomolecules 2022, 12, 1770. [Google Scholar] [CrossRef] [PubMed]
- MacAskill, A.F.; Kittler, J.T. Control of mitochondrial transport and localization in neurons. Trends Cell Biol. 2010, 20, 102–112. [Google Scholar] [CrossRef]
- Pasquier, J.; Guerrouahen, B.S.; Al Thawadi, H.; Ghiabi, P.; Maleki, M.; Abu-Kaoud, N.; Jacob, A.; Mirshahi, M.; Galas, L.; Rafii, S.; et al. Preferential transfer of mitochondria from endothelial to cancer cells through tunneling nanotubes modulates chemoresistance. J. Transl. Med. 2013, 11, 94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spees, J.L.; Olson, S.D.; Whitney, M.J.; Prockop, D.J. Mitochondrial transfer between cells can rescue aerobic respiration. Proc. Natl. Acad. Sci USA 2006, 103, 1283–1288. [Google Scholar] [CrossRef]
- Van Steenbergen, V.; Lavoie-Cardinal, F.; Kazwiny, Y.; Decet, M.; Martens, T.; Verstreken, P.; Boesmans, W.; De Koninck, P.; Vanden Berghe, P. Nano-positioning and tubulin conformation contribute to axonal transport regulation of mitochondria along microtubules. Proc. Natl. Acad. Sci. USA 2022, 119, e2203499119. [Google Scholar] [CrossRef]
- Sheng, H.; Cai, Q. Mitochondrial transport in neurons: Impact on synaptic homeostasis and neurodegeneration. Nat. Rev. Neurosci. 2012, 13, 77–93. [Google Scholar] [CrossRef] [Green Version]
- Lou, E.; Fujisawa, S.; Morozov, A.; Barlas, A.; Romin, Y.; Dogan, Y.; Gholami, S.; Moreira, A.L.; Manova-Todorova, K.; Moore, M.A. Tunneling nanotubes provide a unique conduit for intercellular transfer of cellular contents in human malignant pleural mesothelioma. PLoS ONE 2012, 7, e3309. [Google Scholar]
- Weng, Z.; Zhang, B.; Tsilioni, I.; Theoharides, T.C. Nanotube formation: A rapid form of “alarm signaling”? Clin. Ther. 2016, 38, 1066–1072. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Subramaniam, M.D.; Iyer, M.; Nair, A.P.; Venkatesan, D.; Mathavan, S.; Eruppakotte, N.; Kizhakkillach, S.; Chandran, M.K.; Roy, A.; Gopalakrishnan, A.V.; et al. Oxidative stress and mitochondrial transfer: A new dimension towards ocular diseases. Genes Dis. 2020, 9, 610–637. [Google Scholar] [CrossRef] [PubMed]
- Naphade, S.; Sharma, J.; Gaide Chevronnay, H.P.; Shook, M.A.; Yeagy, B.A.; Rocca, C.J.; Ur, S.N.; Lau, A.J.; Courtoy, P.J.; Cherqui, S. Brief reports: Lysosomal cross-correction by hematopoietic stem cell-derived macrophages via tunneling nanotubes. Stem Cells 2015, 33, 301–309. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fairley, H.; Grimm, A.; Eckert, A. Mitochondria transfer in brain injury and disease. Cells 2022, 11, 3603. [Google Scholar] [CrossRef] [PubMed]
- Patel, P.S.; Castelow, C.; Patel, D.S.; Bhattacharya, S.K.; Kuscu, C.; Kuscu, C.; Makowski, L.; Eason, J.D.; Bajwa, A. Mitochondrial role in oncogenesis and potential chemotherapeutic strategy of mitochondrial infusion in breast cancer. Int. J. Mol. Sci. 2022, 23, 12993. [Google Scholar] [CrossRef] [PubMed]
- Weiner-Gorzel, K.; Murphy, M. Mitochondrial dynamics, a new therapeutic target for Triple Negative Breast Cancer. Biochim. Biophys. Acta Rev. Cancer 2021, 1875, 188518. [Google Scholar] [CrossRef]
- Xing, J.; Qi, L.; Liu, X.; Shi, G.; Sun, X.; Yang, Y. Roles of mitochondrial fusion and fission in breast cancer progression: A systematic review. World J. Surg. Oncol. 2022, 20, 331. [Google Scholar] [CrossRef]
- Song, I.S.; Jeong, Y.J.; Jeong, S.H.; Kim, J.E.; Han, J.; Kim, T.H.; Jang, S.W. Modulation of mitochondrial ERβ expression inhibits triple-negative breast cancer tumor progression by activating mitochondrial function. Cell. Physiol. Biochem. 2019, 52, 468–485. [Google Scholar] [CrossRef] [Green Version]
- Humphries, B.A.; Cutter, A.C.; Buschhaus, J.M.; Chen, Y.C.; Qyli, T.; Palagama, D.S.W.; Eckley, S.; Robison, T.H.; Bevoor, A.; Chiang, B.; et al. Enhanced mitochondrial fission suppresses signaling and metastasis in triple-negative breast cancer. Breast Cancer Res. 2020, 22, 60. [Google Scholar] [CrossRef]
- Demaria, S.; Ng, B.; Devitt, M.L.; Babb, J.S.; Kawashima, N.; Liebes, L.; Formenti, S.C. Ionizing radiation inhibition of distant untreated tumors (abscopal effect) is immune mediated. Int. J. Radiat. Oncol. Biol. Phys. 2004, 58, 862–870. [Google Scholar] [CrossRef]
- Kong, C.; Song, W.; Fu, T. Systemic inflammatory response syndrome is triggered by mitochondrial damage (Review). Mol. Med. Rep. 2022, 25, 147. [Google Scholar] [CrossRef] [PubMed]
- De Toledo, S.M.; Buonanno, M.; Harris, A.L.; Azzam, E.I. Genomic instability induced in distant progeny of bystander cells depends on the connexins expressed in the irradiated cells. Int. J. Radiat. Biol. 2017, 93, 1182–1194. [Google Scholar] [CrossRef]
- Mancuso, M.; Pasquali, E.; Leonardi, S.; Rebessi, S.; Tanori, M.; Giardullo, P.; Borra, F.; Pazzaglia, S.; Naus, C.C.; Di Majo, V.; et al. Role of connexin43 and ATP in long-range bystander radiation damage and oncogenesis in vivo. Oncogene 2011, 30, 4601–4608. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pazzaglia, S.; Tanno, B.; De Stefano, I.; Giardullo, P.; Leonardi, S.; Merla, C.; Babini, G.; Tuncay Cagatay, S.; Mayah, A.; Kadhim, M.; et al. Micro-RNA and proteomic profiles of plasma-derived exosomes from irradiated mice reveal molecular changes preventing apoptosis in neonatal cerebellum. Int. J. Mol. Sci. 2022, 23, 2169. [Google Scholar] [CrossRef]
- Pazzaglia, S.; Eidemüller, M.; Lumniczky, K.; Mancuso, M.; Ramadan, R.; Stolarczyk, L.; Moertl, S. Out-of-field effects: Lessons learned from partial body exposure. Radiat. Environ. Biophys. 2022, 61, 485–504. [Google Scholar] [CrossRef] [PubMed]
- Tanno, B.; Novelli, F.; Leonardi, S.; Merla, C.; Babini, G.; Giardullo, P.; Kadhim, M.; Traynor, D.; Medipally, D.K.R.; Meade, A.D.; et al. MiRNA-mediated fibrosis in the out-of-target heart following partial-body irradiation. Cancers 2022, 14, 3463. [Google Scholar] [CrossRef] [PubMed]
- Habets, T.H.; Oth, T.; Houben, A.W.; Huijskens, M.J.; Senden-Gijsbers, B.L.; Schnijderberg, M.C.; Brans, B.; Dubois, L.J.; Lambin, P.; De Saint-Hubert, M.; et al. Fractionated radiotherapy with 3 × 8 Gy induces systemic anti-tumour responses and abscopal tumour inhibition without modulating the humoral anti-tumour tesponse. PLoS ONE 2016, 11, e0159515. [Google Scholar] [CrossRef]
- Desai, S.; Kobayashi, A.; Konishi, T.; Oikawa, M.; Pandey, B.N. Damaging and protective bystander cross-talk between human lung cancer and normal cells after proton microbeam irradiation. Mutat. Res. 2014, 763–764, 39–44. [Google Scholar] [CrossRef]
- Diegeler, S.; Hellweg, C.E. Intercellular communication of tumor cells and immune cells after exposure to different ionizing radiation qualities. Front. Immunol. 2017, 8, 664. [Google Scholar] [CrossRef] [Green Version]
- Kadhim, M.; Tuncay Cagatay, S.; Elbakrawy, E.M. Non-targeted effects of radiation: A personal perspective on the role of exosomes in an evolving paradigm. Int. J. Radiat. Biol. 2022, 98, 410–420. [Google Scholar] [CrossRef]
- Al-Abedi, R.; Tuncay Cagatay, S.; Mayah, A.; Brooks, S.A.; Kadhim, M. Ionising radiation promotes invasive potential of breast cancer cells: The role of exosomes in the process. Int. J. Mol. Sci. 2021, 22, 11570. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Zhong, W.; Yang, L.; Wen, P.; Luo, Y.; Wu, C. The emerging role of exosomes in radiotherapy. Cell. Commun. Signal. 2022, 20, 171. [Google Scholar] [CrossRef] [PubMed]
- Jabbari, N.; Karimipour, M.; Khaksar, M.; Akbariazar, E.; Heidarzadeh, M.; Mojarad, B.; Aftab, H.; Rahbarghazi, R.; Rezaie, J. Tumor-derived extracellular vesicles: Insights into bystander effects of exosomes after irradiation. Lasers Med. Sci. 2020, 35, 531–545. [Google Scholar] [CrossRef] [PubMed]
- Strybel, U.; Marczak, L.; Zeman, M.; Polanski, K.; Mielańczyk, Ł.; Klymenko, O.; Samelak-Czajka, A.; Jackowiak, P.; Smolarz, M.; Chekan, M.; et al. Molecular composition of serum exosomes could discriminate rectal cancer patients with different responses to neoadjuvant radiotherapy. Cancers 2022, 14, 993. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Ma, H.; Lv, C.; Lan, F.; Wang, Y.; Deng, Y. Exosomes and exosomal microRNA in non-targeted radiation bystander and abscopal effects in the central nervous system. Cancer Lett. 2021, 499, 73–84. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhao, Y.; Han, W.; Chiu, S.K.; Zhu, L.; Wu, L.; Yu, K.N. Rescue effects in radiobiology: Unirradiated bystander cells assist irradiated cells through intercellular signal feedback. Mutat. Res. 2011, 706, 59–64. [Google Scholar] [CrossRef]
- Yu, K.N. Radiation-induced rescue effect: Insights from microbeam Experiments. Biology 2022, 11, 1548. [Google Scholar] [CrossRef]
- Widel, M.; Przybyszewski, W.M.; Cieslar-Pobuda, A.; Saenko, Y.V.; Rzeszowska-Wolny, J. Bystander normal human fibroblasts reduce damage response in radiation targeted cancer cells through intercellular ROS level modulation. Mutat. Res. 2012, 731, 117–124. [Google Scholar] [CrossRef]
- Hamada, N.; Matsumoto, H.; Hara, T.; Kobayashi, Y. Intercellular and intracellular signaling pathways mediating ionizing radiation-induced bystander effects. J. Radiat. Res. 2007, 48, 87–95. [Google Scholar] [CrossRef] [Green Version]
- Lyng, F.M.; Maguire, P.; Kilmurray, N.; Mothersill, C.; Shao, C.; Folkard, M.; Prise, K.M. Apoptosis is initiated in human keratinocytes exposed to signalling factors from microbeam irradiated cells. Int. J. Radiat. Biol. 2006, 82, 393–399. [Google Scholar] [CrossRef]
- Shao, C.; Lyng, F.M.; Folkard, M.; Prise, K.M. Calcium fluxes modulate the radiation-induced bystander responses in targeted glioma and fibroblast cells. Radiat. Res. 2006, 166, 479–487. [Google Scholar] [CrossRef]
- Shao, C.; Folkard, M.; Michael, B.D.; Prise, K.M. Targeted cytoplasmic irradiation induces bystander responses. Proc. Natl. Acad. Sci. USA 2004, 101, 13495–13500. [Google Scholar] [CrossRef]
- Zhou, H.; Ivanov, V.N.; Lien, Y.C.; Davidson, M.; Hei, T.K. Mitochondrial function and nuclear factor-kappa B-mediated signaling in radiation-induced bystander effects. Cancer Res. 2008, 68, 2233–2240. [Google Scholar] [CrossRef] [Green Version]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morgan, W.F. Is there a common mechanism underlying genomic instability, bystander effects and other nontargeted effects of exposure to ionizing radiation? Oncogene 2003, 22, 7094–7099. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morgan, W.F. Radiation-induced genomic instability. Health Phys. 2011, 100, 280–281. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.J.; Chandrasekaran, K.; Morgan, W.F. Mitochondrial dysfunction, persistently elevated levels of reactive oxygen species and radiation-induced genomic instability: A review. Mutagenesis 2006, 21, 361–367. [Google Scholar] [CrossRef] [Green Version]
- Kim, G.J.; Fiskum, G.M.; Morgan, W.F. A role for mitochondrial dysfunction in perpetuating radiation-induced genomic instability. Cancer Res. 2006, 66, 10377–10383. [Google Scholar] [CrossRef] [Green Version]
- Dayal, D.; Martin, S.M.; Owens, K.M.; Aykin-Burns, N.; Zhu, Y.; Boominathan, A.; Pain, D.; Limoli, C.L.; Goswami, P.C.; Domann, F.E.; et al. Mitochondrial complex II dysfunction can contribute significantly to genomic instability after exposure to ionizing radiation. Radiat. Res. 2009, 172, 737–745. [Google Scholar] [CrossRef] [Green Version]
- Szumiel, I. Ionizing radiation-induced oxidative stress, epigenetic changes and genomic instability: The pivotal role of mitochondria. Int. J. Radiat. Biol. 2015, 91, 1–12. [Google Scholar] [CrossRef]
- Tewari, S.; Khan, K.; Husain, N.; Rastogi, M.; Mishra, S.P.; Srivastav, A.K. Peripheral blood lymphocytes as in vitro model to evaluate genomic instability caused by low dose radiation. Asian Pac. J. Cancer Prev. 2016, 17, 1773–1777. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shimura, T.; Kobayashi, J.; Komatsu, K.; Kunugita, N. DNA damage signaling guards against perturbation of cyclin D1 expression triggered by low-dose long-term fractionated radiation. Oncogenesis. 2014, 3, e132. [Google Scholar] [CrossRef] [PubMed]
- Shimura, T. ATM-mediated mitochondrial radiation responses of human fibroblasts. Genes 2021, 12, 1015. [Google Scholar] [CrossRef] [PubMed]
- Shiloh, Y. The cerebellar degeneration in ataxia-telangiectasia: A case for genome instability. DNA Repair 2020, 95, 102950. [Google Scholar] [CrossRef] [PubMed]
- Mitiagin, Y.; Barzilai, A. Ataxia-telangiectasia mutated plays an important role in cerebellar integrity and functionality. Neural Regen. Res. 2023, 18, 497–502. [Google Scholar] [CrossRef]
- Subramanian, G.N.; Yeo, A.J.; Gatei, M.H.; Coman, D.J.; Lavin, M.F. Metabolic stress and mitochondrial dysfunction in ataxia-telangiectasia. Antioxidants 2022, 11, 653. [Google Scholar] [CrossRef]
- Lee, J.H.; Paull, T.T. Mitochondria at the crossroads of ATM-mediated stress signaling and regulation of reactive oxygen species. Redox Biol. 2020, 32, 101511. [Google Scholar] [CrossRef]
- Valentin-Vega, Y.A.; Maclean, K.H.; Tait-Mulder, J.; Milasta, S.; Steeves, M.; Dorsey, F.C.; Cleveland, J.L.; Green, D.R.; Kastan, M.B. Mitochondrial dysfunction in ataxia-telangiectasia. Blood 2012, 119, 1490–1500. [Google Scholar] [CrossRef] [Green Version]
- Valentin-Vega, Y.A.; Kastan, M.B. A new role for ATM: Regulating mitochondrial function and mitophagy. Autophagy 2012, 8, 840–841. [Google Scholar] [CrossRef] [Green Version]
- Stagni, V.; Ferri, A.; Cirotti, C.; Barilà, D. ATM kinase-dependent regulation of autophagy: A key player in senescence? Front. Cell. Dev. Biol. 2021, 8, 599048. [Google Scholar] [CrossRef]
- Chao, T.; Shih, H.T.; Hsu, S.C.; Chen, P.J.; Fan, Y.S.; Jeng, Y.M.; Shen, Z.Q.; Tsai, T.F.; Chang, Z.F. Autophagy restricts mitochondrial DNA damage-induced release of ENDOG (endonuclease G) to regulate genome stability. Autophagy 2021, 17, 3444–3460. [Google Scholar] [CrossRef] [PubMed]
- Morita, A.; Tanimoto, K.; Murakami, T.; Morinaga, T.; Hosoi, Y. Mitochondria are required for ATM activation by extranuclear oxidative stress in cultured human hepatoblastoma cell line Hep G2 cells. Biochem. Biophys. Res. Commun. 2014, 443, 1286–1290. [Google Scholar] [CrossRef] [PubMed]
- Fakouri, N.B.; Hou, Y.; Demarest, T.G.; Christiansen, L.S.; Okur, M.N.; Mohanty, J.G.; Croteau, D.L.; Bohr, V.A. Toward understanding genomic instability, mitochondrial dysfunction and aging. FEBS J. 2019, 286, 1058–1073. [Google Scholar] [CrossRef] [PubMed]
- Lozoya, O.A.; Xu, F.; Grenet, D.; Wang, T.; Grimm, S.A.; Godfrey, V.; Waidyanatha, S.; Woychik, R.P.; Santos, J.H. Single nucleotide resolution analysis reveals pervasive, long-lasting DNA methylation changes by developmental exposure to a mitochondrial toxicant. Cell. Rep. 2020, 32, 108131. [Google Scholar] [CrossRef]
- Breda, C.N.S.; Davanzo, G.G.; Basso, P.J.; Saraiva Câmara, N.O.; Moraes-Vieira, P.M.M. Mitochondria as central hub of the immune system. Redox Biol. 2019, 26, 101255. [Google Scholar] [CrossRef]
- Nadalutti, C.A.; Ayala-Peña, S.; Santos, J.H. Mitochondrial DNA damage as driver of cellular outcomes. Am. J. Physiol. Cell. Physiol. 2022, 322, C136–C150. [Google Scholar] [CrossRef]
- Raval, P.K.; Garg, S.G.; Gould, S.B. Endosymbiotic selective pressure at the origin of eukaryotic cell biology. eLife 2022, 11, e81033. [Google Scholar] [CrossRef]
- Su, Y.J.; Wang, P.W.; Weng, S.W. The role of mitochondria in immune-cell-mediated tissue regeneration and ageing. Int. J. Mol. Sci. 2021, 22, 2668. [Google Scholar] [CrossRef]
- Bagheri, H.S.; Bani, F.; Tasoglu, S.; Zarebkohan, A.; Rahbarghazi, R.; Sokullu, E. Mitochondrial donation in translational medicine; from imagination to reality. J. Transl. Med. 2020, 18, 367. [Google Scholar] [CrossRef]
- Elsässer, T.; Weyrather, W.K.; Friedrich, T.; Durante, M.; Iancu, G.; Krämer, M.; Kragl, G.; Brons, S.; Winter, M.; Weber, K.J.; et al. Quantification of the relative biological effectiveness for ion beam radiotherapy: Direct experimental comparison of proton and carbon ion beams and a novel approach for treatment planning. Int. J. Radiat. Oncol. Biol. Phys. 2010, 78, 1177–1183. [Google Scholar] [CrossRef]
- Lumniczky, K.; Impens, N.; Armengol, G.; Candéias, S.; Georgakilas, A.G.; Hornhardt, S.; Martin, O.A.; Rödel, F.; Schaue, D. Low dose ionizing radiation effects on the immune system. Environ. Int. 2021, 149, 106212. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.J.; Kang, H.; Hong, E.H.; Kim, J.Y.; Nam, S.Y. Transcriptome analysis of low-dose ionizing radiation-impacted genes in CD4+ T-cells undergoing activation and regulation of their expression of select cytokines. J. Immunotoxicol. 2018, 15, 137–146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, G.; Kong, Q.; Wang, G.; Jin, H.; Zhou, L.; Yu, D.; Niu, C.; Han, W.; Li, W.; Cui, J. Low-dose ionizing radiation induces direct activation of natural killer cells and provides a novel approach for adoptive cellular immunotherapy. Cancer Biother. Radiopharm. 2014, 29, 428–434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klug, F.; Prakash, H.; Huber, P.E.; Seibel, T.; Bender, N.; Halama, N.; Pfirschke, C.; Voss, R.H.; Timke, C.; Umansky, L.; et al. Low-dose irradiation programs macrophage differentiation to an iNOS+/M1 phenotype that orchestrates effective T cell immunotherapy. Cancer Cell 2013, 24, 589–602. [Google Scholar] [CrossRef] [Green Version]
- Rödel, F.; Frey, B.; Gaipl, U.; Keilholz, L.; Fournier, C.; Manda, K.; Schöllnberger, H.; Hildebrandt, G.; Rödel, C. Modulation of inflammatory immune reactions by low-dose ionizing radiation: Molecular mechanisms and clinical application. Curr. Med. Chem. 2012, 19, 1741–1750. [Google Scholar] [CrossRef] [PubMed]
- Frey, B.; Hehlgans, S.; Rödel, F.; Gaipl, U.S. Modulation of inflammation by low and high doses of ionizing radiation: Implications for benign and malign diseases. Cancer Lett. 2015, 368, 230–237. [Google Scholar] [CrossRef]
- Dove, A.P.H.; Cmelak, A.; Darrow, K.; McComas, K.N.; Chowdhary, M.; Beckta, J.; Kirschner, A.N. The use of low-dose radiation therapy in osteoarthritis: A review. Int. J. Radiat. Oncol. Biol. Phys. 2022, 114, 203–220. [Google Scholar] [CrossRef]
- Kim, B.H.; Bae, H.C.; Wang, S.Y.; Jang, B.S.; Chang, J.H.; Chie, E.K.; Yi, H.S.; Kwon, J.; Han, H.S.; Kim, H.J. Low-dose irradiation could mitigate osteoarthritis progression via anti-inflammatory action that modulates mitochondrial function. Radiother. Oncol. 2022, 170, 231–241. [Google Scholar] [CrossRef]
- Iannello, A.; Thompson, T.W.; Ardolino, M.; Lowe, S.W.; Raulet, D.H. p53-dependent chemokine production by senescent tumor cells supports NKG2D-dependent tumor elimination by natural killer cells. J. Exp. Med. 2013, 210, 2057–2069. [Google Scholar] [CrossRef]
- Pilones, K.A.; Charpentier, M.; Garcia-Martinez, E.; Daviaud, C.; Kraynak, J.; Aryankalayil, J.; Formenti, S.C.; Demaria, S. Radiotherapy cooperates with IL15 to induce antitumor immune responses. Cancer Immunol. Res. 2020, 8, 1054–1063. [Google Scholar] [CrossRef]
- Tong, L.; Jiménez-Cortegana, C.; Tay, A.H.M.; Wickström, S.; Galluzzi, L.; Lundqvist, A. NK cells and solid tumors: Therapeutic potential and persisting obstacles. Mol. Cancer. 2022, 21, 206. [Google Scholar] [CrossRef] [PubMed]
- Kirkby, C. A model of infection and immune response to low dose radiation. Int. J. Radiat. Biol. 2022, 98, 1243–1256. [Google Scholar] [CrossRef]
- Wen, K.; Bai, C.; Zhao, H.; Zhou, P.; Gao, S.; Guan, H.; Song, M. Low dose radiation therapy attenuates ACE2 depression and inflammatory cytokines induction by COVID-19 viral spike protein in human bronchial epithelial cells. Int. J. Radiat. Biol. 2022, 98, 1532–1541. [Google Scholar] [CrossRef] [PubMed]
- Schirrmacher, V. Mitochondria at work: New insights into regulation and dysregulation of cellular energy supply and metabolism. Biomedicines 2020, 8, 526. [Google Scholar] [CrossRef]
- Manukian, G.; Kivolowitz, C.; DeAngelis, T.; Shastri, A.A.; Savage, J.E.; Camphausen, K.; Rodeck, U.; Zarif, J.C.; Simone, N.L. Caloric restriction impairs regulatory T cells within the tumor microenvironment after radiation and primes effector T cells. Int. J. Radiat. Oncol. Biol. Phys. 2021, 110, 1341–1349. [Google Scholar] [CrossRef] [PubMed]
- Bao, X.; Xie, L. Targeting purinergic pathway to enhance radiotherapy-induced immunogenic cancer cell death. J. Exp. Clin. Cancer Res. 2022, 41, 222. [Google Scholar] [CrossRef]
- Alam, K.; Moinuddin; Jabeen, S. Immunogenicity of mitochondrial DNA modified by hydroxyl radical. Cell. Immunol. 2007, 247, 12–17. [Google Scholar] [CrossRef]
- Banoth, B.; Cassel, S.L. Mitochondria in innate immune signaling. Transl. Res. 2018, 202, 52–68. [Google Scholar] [CrossRef]
- Kawamura, K.F.; Qi, F.; Kobayashi, J. Potential relationship between the biological effects of low-dose irradiation and mitochondrial ROS production. J. Radiat. Res. 2018, 59 (Suppl. S2), ii91–ii97. [Google Scholar] [CrossRef] [Green Version]
- Van Houten, B.; Hunter, S.E.; Meyer, J.N. Mitochondrial DNA damage induced autophagy, cell death, and disease. Front. Biosci. (Landmark Ed) 2016, 21, 42–54. [Google Scholar] [CrossRef]
- Zhang, B.; Davidso, M.M.; Hei, T.K. Mitochondria regulate DNA damage and genomic instability induced by high LET radiation. Life Sci. Space Res. 2014, 1, 80–88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McArthur, K.; Whitehead, L.W.; Heddleston, J.M.; Li, L.; Padman, B.S.; Oorschot, V.; Geoghegan, N.D.; Chappaz, S.; Davidson, S.; San Chin, H.; et al. BAK/BAX macropores facilitate mitochondrial herniation and mtDNA efflux during apoptosis. Science 2018, 359, eaao6047. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Demaria, S.; Formenti, S.C. Radiation as an immunological adjuvant: Current evidence on dose and fractionation. Front. Oncol. 2012, 2, 153. [Google Scholar] [CrossRef] [Green Version]
- McLaughlin, M.; Patin, E.C.; Pedersen, M.; Wilkins, A.; Dillon, M.T.; Melcher, A.A.; Harrington, K.J. Inflammatory microenvironment remodelling by tumour cells after radiotherapy. Nat. Rev. Cancer 2020, 20, 203–217. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.; Tait, S.W.G. Targeting immunogenic cell death in cancer. Mol. Oncol. 2020, 14, 2994–3006. [Google Scholar] [CrossRef]
- Chan Wah Hak, C.M.L.; Rullan, A.; Patin, E.C.; Pedersen, M.; Melcher, A.A.; Harrington, K.J. Enhancing anti-tumour innate immunity by targeting the DNA damage response and pattern recognition receptors in combination with radiotherapy. Front. Oncol. 2022, 12, 971959. [Google Scholar] [CrossRef] [PubMed]
- Murao, A.; Aziz, M.; Wang, H.; Brenner, M.; Wang, P. Release mechanisms of major DAMPs. Apoptosis 2021, 26, 152–162. [Google Scholar] [CrossRef]
- Del Prete, A.; Salvi, V.; Soriani, A.; Laffranchi, M.; Sozio, F.; Bosisio, D.; Sozzani, S. Dendritic cell subsets in cancer immunity and tumor antigen sensing. Cell. Mol. Immunol. 2023, 20, 432–447. [Google Scholar] [CrossRef]
- Deng, L.; Liang, H.; Xu, M.; Yang, X.; Burnette, B.; Arina, A.; Li, X.D.; Mauceri, H.; Beckett, M.; Darga, T.; et al. STING-dependent cytosolic DNA sensing promotes radiation-induced type I interferon-dependent antitumor immunity in immunogenic tumors. Immunity 2014, 41, 843–852. [Google Scholar] [CrossRef] [Green Version]
- Cai, X.; Chiu, Y.H.; Chen, Z.J. The cGAS-cGAMP-STING pathway of cytosolic DNA sensing and signaling. Mol. Cell 2014, 54, 289–296. [Google Scholar] [CrossRef] [Green Version]
- Xu, M.M.; Pu, Y.; Han, D.; Shi, Y.; Cao, X.; Liang, H.; Chen, X.; Li, X.-D.; Deng, L.; Chen, Z.J.; et al. Dendritic cells but not macrophages sense tumor mitochondrial DNA for cross-priming through signal regulatory protein α signaling. Immunity 2017, 47, 363–373.e5. [Google Scholar] [CrossRef] [PubMed]
- Storozynsky, Q.; Hitt, M.M. The impact of radiation-induced DNA damage on cGAS-STING-mediated immune responses to cancer. Int. J. Mol. Sci. 2020, 21, 8877. [Google Scholar] [CrossRef] [PubMed]
- Mackenzie, K.J.; Carroll, P.; Martin, C.-A.; Murina, O.; Fluteau, A.; Simpson, D.J.; Olova, N.; Sutcliffe, H.; Rainger, J.K.; Leitch, A.; et al. cGAS surveillance of micronuclei links genome instability to innate immunity. Nature 2017, 548, 461–465. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsumura, S.; Wang, B.; Kawashima, N.; Braunstein, S.; Badura, M.; Cameron, T.O.; Babb, J.S.; Schneider, R.J.; Formenti, S.C.; Dustin, M.L.; et al. Radiation-induced CXCL16 release by breast cancer cells attracts effector T cells. J. Immunol. 2008, 181, 3099–3107. [Google Scholar] [CrossRef] [Green Version]
- Lin, W.; Xu, Y.; Chen, X.; Liu, J.; Weng, Y.; Zhuang, Q.; Lin, F.; Huang, Z.; Wu, S.; Ding, J.; et al. Radiation-induced small extracellular vesicles as “carriages” promote tumor antigen release and trigger antitumor immunity. Theranostics 2020, 10, 4871–4884. [Google Scholar] [CrossRef]
- Kaminski, J.M.; Shinohara, E.; Summers, J.B.; Niermann, K.J.; Morimoto, A.; Brousal, J. The controversial abscopal effect. Cancer Treat. Rev. 2005, 31, 159–172. [Google Scholar] [CrossRef]
- Wennerberg, E.; Vanpouille-Box, C.; Bornstein, S.; Yamazaki, T.; Demaria, S.; Galluzzi, L. Immune recognition of irradiated cancer cells. Immunol. Rev. 2017, 280, 220–230. [Google Scholar] [CrossRef]
- Liu, J.; Liu, M.; Zhang, H.; Guo, W. High-Contrast Fluorescence Diagnosis of Cancer Cells/Tissues Based on -Lapachone-Triggered ROS Amplification Specific in Cancer Cells. Angew. Chem. Int. Ed. Engl. 2021, 60, 12992–12998. [Google Scholar] [CrossRef]
- Reczek, C.R.; Chandel, N.S. The two faces of reactive oxygen species in cancer. Annu. Rev. Cancer Biol. 2017, 1, 79–98. [Google Scholar] [CrossRef]
- Maguire, P.; Mothersill, C.; Seymour, C.; Lyng, F.M. Medium from irradiated cells induces dose-dependent mitochondrial changes and BCL2 responses in unirradiated human keratinocytes. Radiat. Res. 2005, 163, 384–390. [Google Scholar] [CrossRef]
- Ludgate, C.M. Optimizing cancer treatments to induce an acute immune response: Radiation Abscopal effects, PAMPs, and DAMPs. Clin. Cancer Res. 2012, 18, 4522–4525. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bian, C.; Zheng, Z.; Su, J.; Wang, H.; Chang, S.; Xin, Y.; Jiang, X. Targeting Mitochondrial Metabolism to Reverse Radioresistance: An Alternative to Glucose Metabolism. Antioxidants 2022, 11, 2202. [Google Scholar] [CrossRef]
- Galluzzi, L.; Pedro, J.M.B.-S.; Levine, B.; Green, D.R.; Kroemer, G. Pharmacological modulation of autophagy: Therapeutic potential and persisting obstacles. Nat. Rev. Drug Discov. 2017, 16, 487–511. [Google Scholar] [CrossRef] [Green Version]
- Piskounova, E.; Agathocleous, M.; Murphy, M.M.; Hu, Z.; Huddlestun, S.E.; Zhao, Z.; Leitch, A.M.; Johnson, T.M.; DeBerardinis, R.J.; Morrison, S.J. Oxidative stress inhibits distant metastasis by human melanoma cells. Nature 2015, 527, 186–191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakamura, H.; Takada, K. Reactive oxygen species in cancer: Current findings and future directions. Cancer Sci. 2021, 112, 3945–3952. [Google Scholar] [CrossRef] [PubMed]
- Mothersill, C.; Stamato, T.D.; Perez, M.L.; Cummins, R.; Mooney, R.; Seymour, C.B. Involvement of energy metabolism in the production of ‘bystander effects’ by radiation. Br. J. Cancer 2000, 82, 1740–1746. [Google Scholar] [CrossRef] [Green Version]
- Lyng, F.M.; Seymour, C.B.; Mothersill, C. Production of a signal by irradiated cells which leads to a response in unirradiated cells characteristic of initiation of apoptosis. Br. J. Cancer 2000, 83, 1223–1230. [Google Scholar] [CrossRef]
- Maguire, P.; Mothersill, C.; McClean, B.; Seymour, C.; Lyng, F.M. Modulation of radiation responses by pre-exposure to irradiated cell conditioned medium. Radiat. Res. 2007, 167, 485–492. [Google Scholar] [CrossRef] [Green Version]
- Nikitaki, Z.; Mavragani, I.V.; Laskaratou, D.A.; Gika, V.; Moskvin, V.P.; Theofilatos, K.; Vougas, K.; Stewart, R.D.; Georgakilas, A.G. Systemic mechanisms and effects of ionizing radiation: A new ‘old’ paradigm of how the bystanders and distant can become the players. Semin. Cancer Biol. 2016, 37–38, 77–95. [Google Scholar] [CrossRef]
- Rajendran, S.; Harrison, S.H.; Thomas, R.A.; Tucker, J.D. The role of mitochondria in the radiation-induced bystander effect in human lymphoblastoid cells. Radiat. Res. 2011, 175, 159–171. [Google Scholar] [CrossRef] [Green Version]
- Lyng, F.M.; Seymour, C.B.; Mothersill, C. Early events in the apoptotic cascade initiated in cells treated with medium from the progeny of irradiated cells. Radiat. Prot. Dosim. 2002, 99, 169–172. [Google Scholar] [CrossRef] [PubMed]
- Nugent, S.M.; Mothersill, C.E.; Seymour, C.; McClean, B.; Lyng, F.M.; Murphy, J.E. Increased mitochondrial mass in cells with functionally compromised mitochondria after exposure to both direct gamma radiation and bystander factors. Radiat. Res. 2007, 168, 134–142. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nugent, S.; Mothersill, C.E.; Seymour, C.; McClean, B.; Lyng, F.M.; Murphy, J.E. Altered mitochondrial function and genome frequency post exposure to γ-radiation and bystander factors. Int. J. Radiat. Biol. 2010, 86, 829–841. [Google Scholar] [CrossRef] [Green Version]
- Maxwell, C.A.; Fleisch, M.C.; Costes, S.V.; Erickson, A.C.; Boissière, A.; Gupta, R.; Ravani, S.A.; Parvin, B.; Barcellos-Hoff, M.H. Targeted and nontargeted effects of ionizing radiation that impact genomic instability. Cancer Res. 2008, 68, 8304–8311. [Google Scholar] [CrossRef] [Green Version]
- Chen, S.; Zhao, Y.; Han, W.; Zhao, G.; Zhu, L.; Wang, J.; Bao, L.; Jiang, E.; Xu, A.; Hei, T.K.; et al. Mitochondria-dependent signalling pathway are involved in the early process of radiation-induced bystander effects. Br. J. Cancer 2008, 98, 1839–1844. [Google Scholar] [CrossRef] [Green Version]
- Chen, S.; Zhao, Y.; Zhao, G.; Han, W.; Bao, L.; Yu, K.N.; Wu, L. Up-regulation of ROS by mitochondria-dependent bystander signaling contributes to genotoxicity of bystander effects. Mutat. Res. 2009, 666, 68–73. [Google Scholar] [CrossRef]
- Szatmári, T.; Kis, D.; Bogdándi, E.N.; Benedek, A.; Bright, S.; Bowler, D.; Persa, E.; Kis, E.; Balogh, A.; Naszályi, L.N.; et al. Extracellular Vesicles Mediate Radiation-Induced Systemic Bystander Signals in the Bone Marrow and Spleen. Front. Immunol. 2017, 8, 347. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mukherjee, S.; Dutta, A.; Chakraborty, A. The cross-talk between Bax, Bcl2, caspases, and DNA damage in bystander HepG2 cells is regulated by γ-radiation dose and time of conditioned media transfer. Apoptosis 2022, 27, 184–205. [Google Scholar] [CrossRef] [PubMed]
- Mothersill, C.; Le, M.; Rusin, A.; Seymour, C. Biophotons in radiobiology: Inhibitors, communicators and reactors. Radiat. Prot. Dosim. 2019, 183, 136–141. [Google Scholar] [CrossRef]
- Jain, M.R.; Li, M.; Chen, W.; Liu, T.; de Toledo, S.M.; Pandey, B.N.; Li, H.; Rabin, B.M.; Azzam, E.I. In vivo space radiation-induced non-targeted responses: Late effects on molecular signaling in mitochondria. Curr. Mol. Pharmacol. 2011, 4, 106–114. [Google Scholar] [CrossRef]
- Liu, Z.; Mothersill, C.E.; McNeill, F.E.; Lyng, F.M.; Byun, S.H.; Seymour, C.B.; Prestwich, W.V. A dose threshold for a medium transfer bystander effect for a human skin cell line. Radiat. Res. 2006, 166, 19–23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lyng, F.M.; Howe, O.L.; McClean, B. Reactive oxygen species-induced release of signalling factors in irradiated cells triggers membrane signalling and calcium influx in bystander cells. Int. J. Radiat. Biol. 2011, 87, 683–695. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shao, C.; Folkard, M.; Prise, K.M. Role of TGF-beta1 and nitric oxide in the bystander response of irradiated glioma cells. Oncogene 2008, 27, 434–440. [Google Scholar] [CrossRef] [Green Version]
- Lafargue, A.; Degorre, C.; Corre, I.; Alves-Guerra, M.C.; Gaugler, M.H.; Vallette, F.; Pecqueur, C.; Paris, F. Ionizing radiation induces long-term senescence in endothelial cells through mitochondrial respiratory complex II dysfunction and superoxide generation. Free Radical. Biol. Med. 2017, 108, 750–759. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shibamoto, Y.; Nakamura, H. Overview of Biological, Epidemiological, and Clinical Evidence of Radiation Hormesis. Int. J. Mol. Sci. 2018, 19, 2387. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alexandrou, A.T.; Li, J.J. Cell cycle regulators guide mitochondrial activity in radiation-induced adaptive response. Antioxid. Redox Signal. 2014, 20, 1463–1480. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, H.S.; Seong, K.M.; Kim, J.Y.; Kim, C.S.; Yang, K.H.; Jin, Y.W.; Nam, S.Y. Chronic low-dose radiation inhibits the cells death by cytotoxic high-dose radiation increasing the level of AKT and acinus proteins via NF-κB activation. Int. J. Radiat. Biol. 2013, 89, 371–377. [Google Scholar] [CrossRef]
- Wang, X.C.; Tian, L.L.; Fan, C.X.; Duo, C.H.; Xu, K.M. The Adaptive Responses in Non-Small Cell Lung Cancer A549 Cell Lines Induced by Low-Dose Ionizing Radiation and the Variations of miRNA Expression. Dose Response 2021, 19, 1–10. [Google Scholar] [CrossRef]
- Shimura, T.; Sasatani, M.; Kamiya, K.; Kawai, H.; Inaba, Y.; Kunugita, N. Mitochondrial reactive oxygen species perturb AKT/cyclin D1 cell cycle signaling via oxidative inactivation of PP2A in low dose irradiated human fibroblasts. Oncotarget 2016, 7, 3559–3570. [Google Scholar] [CrossRef] [Green Version]
- Klokov, D.; Criswell, T.; Leskov, K.S.; Araki, S.; Mayo, L.; Boothman, D.A. IR-inducible clusterin gene expression: A protein with potential roles in ionizing radiation-induced adaptive responses, genomic instability, and bystander effects. Mutat. Res. 2004, 568, 97–110. [Google Scholar] [CrossRef]
- Ahmed, K.M.; Nantajit, D.; Fan, M.; Murley, J.S.; Grdina, D.J.; Li, J.J. Coactivation of ATM/ERK/NF-kappaB in the low-dose radiation-induced radioadaptive response in human skin keratinocytes. Free Radic. Biol. Med. 2009, 46, 1543–1550. [Google Scholar] [CrossRef] [PubMed]
- Stouten, S.; Balkenende, B.; Roobol, L.; Lunel, S.V.; Badie, C.; Dekkers, F. Hyper-radiosensitivity affects low-dose acute myeloid leukemia incidence in a mathematical model. Radiat. Environ. Biophys. 2022, 61, 361–373. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues-Moreira, S.; Moreno, S.G.; Ghinatti, G.; Lewandowski, D.; Hoffschir, F.; Ferri, F.; Gallouet, A.S.; Gay, D.; Motohashi, H.; Yamamoto, M.; et al. Low-Dose Irradiation Promotes Persistent Oxidative Stress and Decreases Self-Renewal in Hematopoietic Stem Cells. Cell Rep. 2017, 20, 3199–3211. [Google Scholar] [CrossRef] [Green Version]
- Fernando, C.; Byun, S.H.; Shi, X.; Seymour, C.B.; Mothersill, C.E. Isolation of the effects of alpha-related components from total effects of radium at low doses. Int. J. Radiat. Biol. 2022, 98, 1168–1175. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, A.; Payet, V.; Bernay, B.; Chartier-Garcia, E.; Testard, I.; Candéias, S.M.; Chevalier, F. Label-Free Direct Mass Spectrometry Analysis of the Bystander Effects Induced in Chondrocytes by Chondrosarcoma Cells Irradiated with X-rays and Carbon Ions. Front. Biosci. (Landmark Ed) 2022, 27, 1–13. [Google Scholar] [CrossRef]
- Hellweg, C.E. The Nuclear Factor κB pathway: A link to the immune system in the radiation response. Cancer Lett. 2015, 368, 275–289. [Google Scholar] [CrossRef]
- Yim, J.H.; Yun, J.M.; Kim, J.Y.; Nam, S.Y.; Kim, C.S. Estimation of low-dose radiation-responsive proteins in the absence of genomic instability in normal human fibroblast cells. Int. J. Radiat. Biol. 2017, 93, 1197–1206. [Google Scholar] [CrossRef]
- Song, K.H.; Jung, S.Y.; Park, J.I.; Ahn, J.; Park, J.K.; Hwang, S.G.; Kim, E.H.; Nam, S.Y.; Park, S.; Ha, H.; et al. Evaluation of Anti-Tumor Effects of Whole-Body Low-Dose Irradiation in Metastatic Mouse Models. Cancers 2020, 12, 1126. [Google Scholar] [CrossRef]
- Rey, N.; Ebrahimian, T.; Gloaguen, C.; Kereselidze, D.; Magneron, V.; Bontemps, C.A.; Demarquay, C.; Olsson, G.; Haghdoost, S.; Lehoux, S.; et al. Exposure to Low to Moderate Doses of Ionizing Radiation Induces a Reduction of Pro-Inflammatory Ly6chigh Monocytes and a U-Curved Response of T Cells in APOE −/− Mice. Dose Response 2021, 19, 15593258211016237. [Google Scholar] [CrossRef]
- Narasimhamurthy, R.K.; Mumbrekar, K.D.; Rao, B.S.S. Effects of low dose ionizing radiation on the brain- a functional, cellular, and molecular perspective. Toxicology 2022, 465, 153030. [Google Scholar] [CrossRef]
- Zhou, H.; Luo, J.; Mou, K.; Peng, L.; Li, X.; Lei, Y.; Wang, J.; Lin, S.; Luo, Y.; Xiang, L. Stress granules: Functions and mechanisms in cancer. Cell Biosci. 2023, 13, 86. [Google Scholar] [CrossRef] [PubMed]
- Tudor, M.; Gilbert, A.; Lepleux, C.; Temelie, M.; Hem, S.; Armengaud, J.; Brotin, E.; Haghdoost, S.; Savu, D.; Chevalier, F. A Proteomic Study Suggests Stress Granules as New Potential Actors in Radiation-Induced Bystander Effects. Int. J. Mol. Sci. 2021, 22, 7957. [Google Scholar] [CrossRef] [PubMed]
- Ladjohounlou, R.; Louati, S.; Lauret, A.; Gauthier, A.; Ardail, D.; Magne, N.; Alphonse, G.; Rodriguez-Lafrasse, C. Ceramide-Enriched Membrane Domains Contribute to Targeted and Nontargeted Effects of Radiation through Modulation of PI3K/AKT Signaling in HNSCC Cells. Int. J. Mol. Sci. 2020, 21, 7200. [Google Scholar] [CrossRef]
- Song, M.S.; Grabocka, E. Stress Granules in Cancer. Rev. Physiol. Biochem. Pharmacol. 2023, 185, 25–52. [Google Scholar] [CrossRef] [PubMed]
- Amen, T.; Kaganovich, D. Stress granules inhibit fatty acid oxidation by modulating mitochondrial permeability. Cell. Rep. 2021, 35, 109237. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Zhang, X.; Li, H.; Niu, C.; Yu, D.; Yang, G.; Liang, X.; Wen, X.; Li, M.; Cui, J. Validating the pivotal role of the immune system in low-dose radiation-induced tumor inhibition in Lewis lung cancer-bearing mice. Cancer Med. 2018, 7, 1338–1348. [Google Scholar] [CrossRef]
- Nowosielska, E.M.; Cheda, A.; Wrembel-Wargocka, J.; Janiak, M.K. Effect of low doses of low-LET radiation on the innate anti-tumor reactions in radioresistant and radiosensitive mice. Dose Response 2012, 10, 500–515. [Google Scholar] [CrossRef] [Green Version]
- Fang, F.; Xie, S.; Chen, M.; Li, Y.; Yue, J.; Ma, J.; Shu, X.; He, Y.; Xiao, W.; Tian, Z. Advances in NK cell production. Cell. Mol. Immunol. 2022, 19, 460–481. [Google Scholar] [CrossRef]
- Barsoumian, H.B.; Ramapriyan, R.; Younes, A.I.; Caetano, M.S.; Menon, H.; Comeaux, N.I.; Cushman, T.R.; Schoenhals, J.E.; Cadena, A.P.; Reilly, T.P.; et al. Low-dose radiation treatment enhances systemic antitumor immune responses by overcoming the inhibitory stroma. J. Immunother. Cancer 2020, 8, e000537. [Google Scholar] [CrossRef]
- Prakash, H.; Klug, F.; Nadella, V.; Mazumdar, V.; Schmitz-Winnenthal, H.; Umansky, L. Low doses of gamma irradiation potentially modifies immunosuppressive tumor microenvironment by retuning tumor-associated macrophages: Lesson from insulinoma. Carcinogenesis 2016, 37, 301–313. [Google Scholar] [CrossRef] [Green Version]
- Leu, J.D.; Wang, C.Y.; Lo, C.C.; Lin, M.Y.; Chang, C.Y.; Hung, W.C.; Lin, S.T.; Wang, B.S.; Lee, Y.J. Involvement of c-Myc in low dose radiation-induced senescence enhanced migration and invasion of unirradiated cancer cells. Aging 2021, 13, 22208–22231. [Google Scholar] [CrossRef] [PubMed]
- Amundson, S.A.; Lee, R.A.; Koch-Paiz, C.A.; Bittner, M.L.; Meltzer, P.; Trent, J.M.; Fornace, A.J., Jr. Differential responses of stress genes to low dose-rate gamma irradiation. Mol. Cancer Res. 2003, 1, 445–452. [Google Scholar] [PubMed]
- Collis, S.J.; Schwaninger, J.M.; Ntambi, A.J.; Keller, T.W.; Nelson, W.G.; Dillehay, L.E.; Deweese, T.L. Evasion of early cellular response mechanisms following low level radiation-induced DNA damage. J. Biol. Chem. 2004, 279, 49624–49632. [Google Scholar] [CrossRef] [Green Version]
- Khizhniak, S.V.; Stepanova, L.I.; Grubskaia, L.V.; Voĭtsitskiĭ, V.M. The functional state of the mitochondrial respiratory chain of the small intestine enterocytes of the rats under the low dose rate X-ray total external exposure. Radiatsionnaia Biol. Radioecol. 2013, 53, 592–597. [Google Scholar]
- Barjaktarovic, Z.; Merl-Pham, J.; Braga-Tanaka, I.; Tanaka, S.; Hauck, S.M.; Saran, A.; Mancuso, M.; Atkinson, M.J.; Tapio, S.; Azimzadeh, O. Hyperacetylation of Cardiac Mitochondrial Proteins Is Associated with Metabolic Impairment and Sirtuin Downregulation after Chronic Total Body Irradiation of ApoE −/− Mice. Int. J. Mol. Sci. 2019, 20, 5239. [Google Scholar] [CrossRef] [Green Version]
- Sugihara, T.; Murano, H.; Fujikawa, K.; Tanaka, I.B.; Komura, J.I. Adaptive response in mice continously irradiated with low dose-rate radiation. Radiat. Prot. Dosim. 2022, 198, 1196–1199. [Google Scholar] [CrossRef] [PubMed]
- Tang, F.R.; Loke, W.K.; Khoo, B.C. Low-dose or low-dose-rate ionizing radiation-induced bioeffects in animal models. J. Radiat. Res. 2017, 58, 165–182. [Google Scholar] [CrossRef] [Green Version]
- Suzuki, K.; Imaoka, T.; Tomita, M.; Sasatani, M.; Doi, K.; Tanaka, S.; Kai, M.; Yamada, Y.; Kakinuma, S. Molecular and cellular basis of the dose-rate-dependent adverse effects of radiation exposure in animal models. Part I: Mammary gland and digestive tract. J. Radiat. Res. 2023, 64, 210–227, Erratum in J. Radiat. Res. 2023, 64, 633. [Google Scholar] [CrossRef]
- Suzuki, K.; Imaoka, T.; Tomita, M.; Sasatani, M.; Doi, K.; Tanaka, S.; Kai, M.; Yamada, Y.; Kakinuma, S. Molecular and cellular basis of the dose-rate-dependent adverse effects of radiation exposure in animal models. Part II: Hematopoietic system, lung and liver. J. Radiat. Res. 2023, 64, 228–249. [Google Scholar] [CrossRef]
- Courtade, M.; Caratero, A.; Jozan, S.; Pipy, B.; Caratero, C. Influence of continuous, very low-dose gamma-irradiation on the mouse immune system. Int. J. Radiat. Biol. 2001, 77, 587–592. [Google Scholar] [CrossRef]
- Lacoste-Collin, L.; Jozan, S.; Cances-Lauwers, V.; Pipy, B.; Gasset, G.; Caratero, C.; Courtade-Saïdi, M. Effect of continuous irradiation with a very low dose of gamma rays on life span and the immune system in SJL mice prone to B-cell lymphoma. Radiat. Res. 2007, 168, 725–732. [Google Scholar] [CrossRef] [PubMed]
- Ogura, K.; Ayabe, Y.; Harada, C.; Tanaka, I.B., III; Tanaka, S.; Komura, J.-I. Increased Frequency of Copy Number Variations Revealed by Array Comparative Genomic Hybridization in the Offspring of Male Mice Exposed to Low Dose-Rate Ionizing Radiation. Int. J. Mol. Sci. 2021, 22, 12437. [Google Scholar] [CrossRef] [PubMed]
- Braga-Tanaka, I., 3rd; Tanaka, S.; Kohda, A.; Takai, D.; Nakamura, S.; Ono, T.; Tanaka, K.; Komura, J.I. Experimental studies on the biological effects of chronic low dose-rate radiation exposure in mice: Overview of the studies at the Institute for Environmental Sciences. Int. J. Radiat. Biol. 2018, 94, 423–433. [Google Scholar] [CrossRef] [PubMed]
- Vo, N.T.K.; Seymour, C.B.; Mothersill, C.E. Dose rate effects of low-LET ionizing radiation on fish cells. Radiat. Environ. Biophys. 2017, 56, 433–441. [Google Scholar] [CrossRef]
- Chauhan, V.; Rowan-Carroll, A.; Gagné, R.; Kuo, B.; Williams, A.; Yauk, C.L. The use of in vitro transcriptional data to identify thresholds of effects in a human lens epithelial cell-line exposed to ionizing radiation. Int. J. Radiat. Biol. 2019, 95, 156–169. [Google Scholar] [CrossRef]
- Yentrapalli, R.; Azimzadeh, O.; Barjaktarovic, Z.; Sarioglu, H.; Wojcik, A.; Harms-Ringdahl, M.; Atkinson, M.J.; Haghdoost, S.; Tapio, S. Quantitative proteomic analysis reveals induction of premature senescence in human umbilical vein endothelial cells exposed to chronic low-dose rate gamma radiation. Proteomics 2013, 3, 1096–1107. [Google Scholar] [CrossRef]
- Yentrapalli, R.; Azimzadeh, O.; Sriharshan, A.; Malinowsky, K.; Merl, J.; Wojcik, A.; Harms-Ringdahl, M.; Atkinson, M.J.; Becker, K.-F.; Haghdoost, S.; et al. The PI3 K/Akt/mTOR pathway is implicated in the premature senescence of primary human endothelial cells exposed to chronic radiation. PLoS ONE 2013, 8, e70024. [Google Scholar] [CrossRef] [Green Version]
- Ina, Y.; Tanooka, H.; Yamada, T.; Sakai, K. Suppression of thymic lymphoma induction by life-long low-dose-rate irradiation accompanied by immune activation in C57BL/6 mice. Radiat. Res. 2005, 163, 153–158. [Google Scholar] [CrossRef]
- Edin, N.J.; Altaner, Č.; Altanerova, V.; Ebbesen, P. TGF-B3 Dependent Modification of Radiosensitivity in Reporter Cells Exposed to Serum From Whole-Body Low Dose-Rate Irradiated Mice. Dose Response 2015, 13, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Edin, N.F.; Altaner, Č.; Altanerova, V.; Ebbesen, P.; Pettersen, E.O. Low-Dose-Rate Irradiation for 1 Hour Induces Protection Against Lethal Radiation Doses but Does Not Affect Life Span of DBA/2 Mice. Dose Response 2016, 14, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Loseva, O.; Shubbar, E.; Haghdoost, S.; Evers, B.; Helleday, T.; Harms-Ringdahl, M. Chronic Low Dose Rate Ionizing Radiation Exposure Induces Premature Senescence in Human Fibroblasts that Correlates with Up Regulation of Proteins Involved in Protection against Oxidative Stress. Proteomes 2014, 2, 341–362. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ansari, M.A.; Thiruvengadam, M.; Venkidasamy, B.; Alomary, M.N.; Salawi, A.; Chung, I.M.; Shariati, M.A.; Rebezov, M. Exosome-based nanomedicine for cancer treatment by targeting inflammatory pathways: Current status and future perspectives. Semin. Cancer Biol. 2022, 86, 678–696. [Google Scholar] [CrossRef] [PubMed]
- Favaudon, V.; Fouillade, C.; Vozenin, M.C. Radiothérapie « flash » à très haut débit de dose: Un moyen d’augmenter l’indice thérapeutique par minimisation des dommages aux tissus sains? [Ultrahigh dose-rate, “flash” irradiation minimizes the side-effects of radiotherapy]. Cancer Radiother. 2015, 19, 526–531. [Google Scholar] [CrossRef]
- Mazal, A.; Prezado, Y.; Ares, C.; de Marzi, L.; Patriarca, A.; Miralbell, R.; Favaudon, V. FLASH and minibeams in radiation therapy: The effect of microstructures on time and space and their potential application to protontherapy. Br. J. Radiol. 2020, 93, 20190807. [Google Scholar] [CrossRef]
- Guo, Z.; Buonanno, M.; Harken, A.; Zhou, G.; Hei, T.K. Mitochondrial Damage Response and Fate of Normal Cells Exposed to FLASH Irradiation with Protons. Radiat. Res. 2022, 197, 569–582. [Google Scholar] [CrossRef] [PubMed]
- Faas, M.M.; de Vos, P. Mitochondrial function in immune cells in health and disease. Biochim. Biophys. Acta Mol. Basis Dis. 2020, 1866, 165845. [Google Scholar] [CrossRef] [PubMed]
- Scheibye-Knudsen, M.; Fang, E.F.; Croteau, D.L.; Wilson, D.M., 3rd; Bohr, V.A. Protecting the mitochondrial powerhouse. Trends Cell Biol. 2015, 25, 158–170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tang, L.; Wei, F.; Wu, Y.; He, Y.; Shi, L.; Xiong, F.; Gong, Z.; Guo, C.; Li, X.; Deng, H.; et al. Role of metabolism in cancer cell radioresistance and radiosensitization methods. J. Exp. Clin. Cancer Res. 2018, 37, 87. [Google Scholar] [CrossRef]
- Tan, Y.Q.; Zhang, X.; Zhang, S.; Zhu, T.; Garg, M.; Lobie, P.E.; Pandey, V. Mitochondria: The metabolic switch of cellular oncogenic transformation. Biochim. Biophys. Acta Rev. Cancer 2021, 1876, 188534. [Google Scholar] [CrossRef]
- Chen, Y.; Li, Y.; Huang, L.; Du, Y.; Gan, F.; Li, Y.; Yao, Y. Antioxidative Stress: Inhibiting Reactive Oxygen Species Production as a Cause of Radioresistance and Chemoresistance. Oxid. Med. Cell. Longev. 2021, 2021, 6620306. [Google Scholar] [CrossRef]
- Vander Heiden, M.G.; Cantley, L.C.; Thompson, C.B. Understanding the Warburg effect: The metabolic requirements of cell proliferation. Science 2009, 324, 1029–1033. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, L.; Li, W. Epithelial-mesenchymal transition in human cancer: Comprehensive reprogramming of metabolism, epigenetics, and differentiation. Pharmacol. Ther. 2015, 150, 33–46. [Google Scholar] [CrossRef] [PubMed]
- Payen, V.L.; Zampieri, L.X.; Porporato, P.E.; Sonveaux, P. Pro- and antitumor effects of mitochondrial reactive oxygen species. Cancer Metastasis Rev. 2019, 38, 189–203. [Google Scholar] [CrossRef] [Green Version]
- Vaupel, P.; Multhoff, G. Revisiting the Warburg effect: Historical dogma versus current understanding. J. Physiol. 2021, 599, 1745–1757. [Google Scholar] [CrossRef] [PubMed]
- Shimura, T.; Noma, N.; Sano, Y.; Ochiai, Y.; Oikawa, T.; Fukumoto, M.; Kunugita, N. AKT-mediated enhanced aerobic glycolysis causes acquired radioresistance by human tumor cells. Radiother. Oncol. 2014, 112, 302–307. [Google Scholar] [CrossRef]
- Dwarakanath, B.S.; Singh, D.; Banerji, A.K.; Sarin, R.; Venkataramana, N.K.; Jalali, R.; Vishwanath, P.N.; Mohanti, B.K.; Tripathi, R.P.; Kalia, V.K.; et al. Clinical studies for improving radiotherapy with 2-deoxy-D-glucose: Present status and future prospects. J. Cancer Res. Ther. 2009, 5 (Suppl. S1), S21–S26. [Google Scholar] [CrossRef]
- Cook, K.M.; Shen, H.; McKelvey, K.J.; Gee, H.E.; Hau, E. Targeting Glucose Metabolism of Cancer Cells with Dichloroacetate to Radiosensitize High-Grade Gliomas. Int. J. Mol. Sci. 2021, 22, 7265. [Google Scholar] [CrossRef]
- Rashmi, R.; Huang, X.; Floberg, J.M.; Elhammali, A.E.; McCormick, M.L.; Patti, G.J.; Spitz, D.R.; Schwarz, J.K. Radioresistant Cervical Cancers Are Sensitive to Inhibition of Glycolysis and Redox Metabolism. Cancer Res. 2018, 78, 1392–1403. [Google Scholar] [CrossRef] [Green Version]
- Huang, L.; Li, B.; Tang, S.; Guo, H.; Li, W.; Huang, X.; Yan, W.; Zou, F. Mitochondrial KATP Channels Control Glioma Radioresistance by Regulating ROS-Induced ERK Activation. Mol. Neurobiol. 2015, 52, 626–637. [Google Scholar] [CrossRef]
- Alphonse, G.; Bionda, C.; Aloy, M.T.; Ardail, D.; Rousson, R.; Rodriguez-Lafrasse, C. Overcoming resistance to gamma-rays in squamous carcinoma cells by poly-drug elevation of ceramide levels. Oncogene 2004, 23, 2703–2715. [Google Scholar] [CrossRef] [Green Version]
- Assi, M.; Rébillard, A. The Janus-Faced Role of Antioxidants in Cancer Cachexia: New Insights on the Established Concepts. Oxid. Med. Cell. Longev. 2016, 2016, 9579868. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zaffaroni, M.; Vincini, M.G.; Corrao, G.; Marvaso, G.; Pepa, M.; Viglietto, G.; Amodio, N.; Jereczek-Fossa, B.A. Unraveling Mitochondrial Determinants of Tumor Response to Radiation Therapy. Int. J. Mol. Sci. 2022, 23, 11343. [Google Scholar] [CrossRef] [PubMed]
- Vakifahmetoglu-Norberg, H.; Ouchida, A.T.; Norberg, E. The role of mitochondria in metabolism and cell death. Biochem. Biophys. Res. Commun. 2017, 482, 426–431. [Google Scholar] [CrossRef] [PubMed]
- Benej, M.; Hong, X.; Vibhute, S.; Scott, S.; Wu, J.; Graves, E.; Le, Q.T.; Koong, A.C.; Giaccia, A.J.; Yu, B.; et al. Papaverine and its derivatives radiosensitize solid tumors by inhibiting mitochondrial metabolism. Proc. Natl. Acad. Sci USA 2018, 115, 10756–10761, Erratum in Proc. Natl. Acad. Sci. USA 2018, 115, E11561. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buckley, A.M.; Dunne, M.R.; Lynam-Lennon, N.; Kennedy, S.A.; Cannon, A.; Reynolds, A.L.; Maher, S.G.; Reynolds, J.V.; Kennedy, B.N.; O’Sullivan, J. Pyrazinib (P3), [(E)-2-(2-Pyrazin-2-yl-vinyl)-phenol], a small molecule pyrazine compound enhances radiosensitivity in oesophageal adenocarcinoma. Cancer Lett. 2019, 447, 115–129. [Google Scholar] [CrossRef] [PubMed]
- Aravindan, N.; Aravindan, S.; Pandian, V.; Khan, F.H.; Ramraj, S.K.; Natt, P.; Natarajan, M. Acquired tumor cell radiation resistance at the treatment site is mediated through radiation-orchestrated intercellular communication. Int. J. Radiat. Oncol. Biol. Phys. 2014, 88, 677–685. [Google Scholar] [CrossRef] [Green Version]
- Ali, M.Y.; Oliva, C.R.; Flor, S.; Goswami, P.C.; Griguer, C.E. Cytochrome c oxidase mediates labile iron level and radioresistance in glioblastoma. Free Radic. Biol. Med. 2022, 185, 25–35. [Google Scholar] [CrossRef]
- Adamczuk, G.M.; Humeniuk, E.; Adamczuk, K.; Madej-Czerwonka, B.; Dudka, J. Disruption of mitochondrial function augments the radiosensitivity of prostate cancer cell lines. Ann. Agric. Environ. Med. 2023, 30, 65–76. [Google Scholar] [CrossRef]
- Grasso, D.; Medeiros, H.C.D.; Zampieri, L.X.; Bol, V.; Danhier, P.; van Gisbergen, M.W.; Bouzin, C.; Brusa, D.; Grégoire, V.; Smeets, H.; et al. Fitter Mitochondria Are Associated with Radioresistance in Human Head and Neck SQD9 Cancer Cells. Front. Pharmacol. 2020, 11, 263. [Google Scholar] [CrossRef]
- McCann, E.; O’Sullivan, J.; Marcone, S. Targeting cancer-cell mitochondria and metabolism to improve radiotherapy response. Transl. Oncol. 2021, 14, 100905. [Google Scholar] [CrossRef]



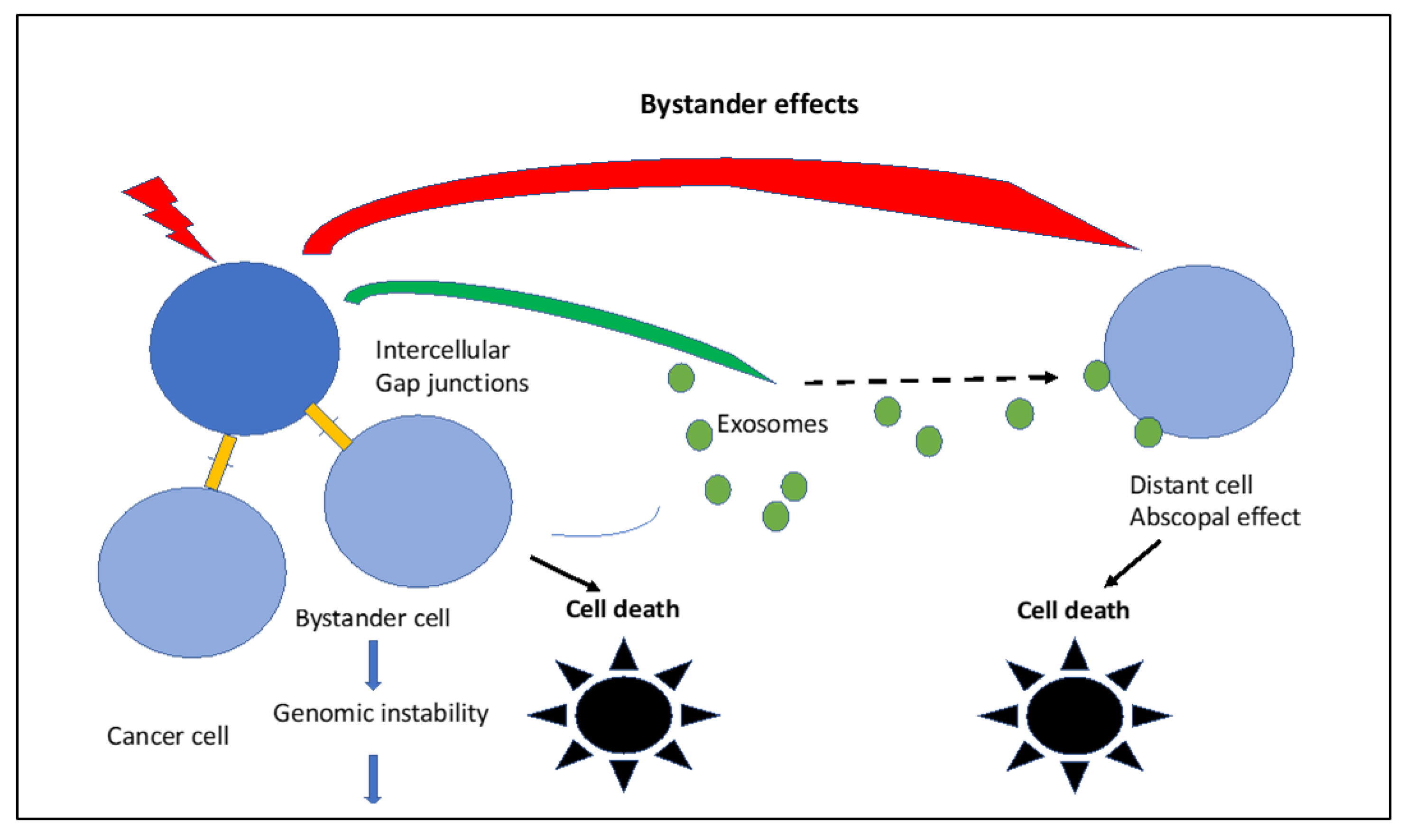

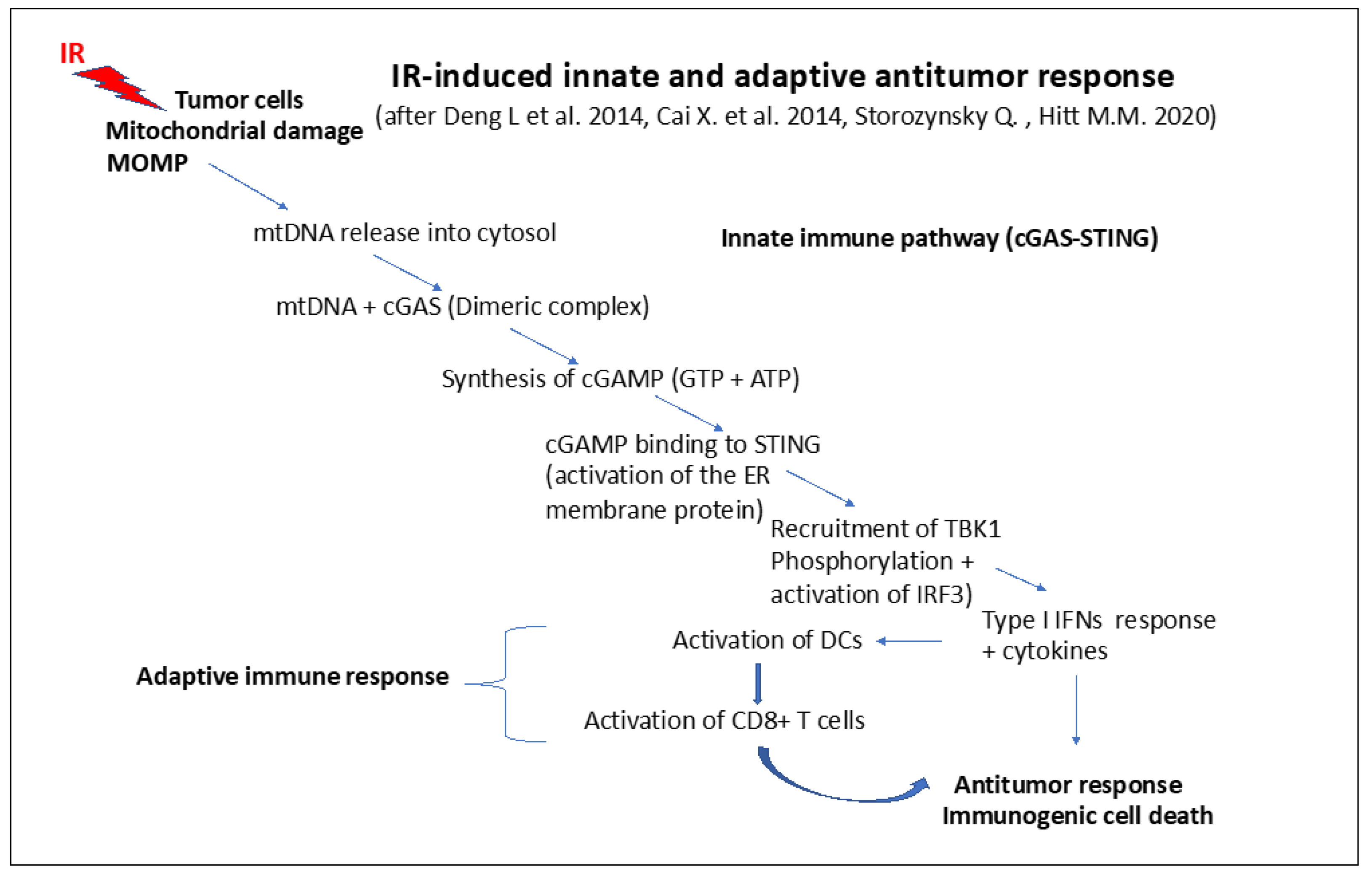
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Averbeck, D. Low-Dose Non-Targeted Effects and Mitochondrial Control. Int. J. Mol. Sci. 2023, 24, 11460. https://doi.org/10.3390/ijms241411460
Averbeck D. Low-Dose Non-Targeted Effects and Mitochondrial Control. International Journal of Molecular Sciences. 2023; 24(14):11460. https://doi.org/10.3390/ijms241411460
Chicago/Turabian StyleAverbeck, Dietrich. 2023. "Low-Dose Non-Targeted Effects and Mitochondrial Control" International Journal of Molecular Sciences 24, no. 14: 11460. https://doi.org/10.3390/ijms241411460





